Enhanced Survival of High-Risk Medulloblastoma-Bearing Mice after Multimodal Treatment with Radiotherapy, Decitabine, and Abacavir
Abstract
1. Introduction
2. Results
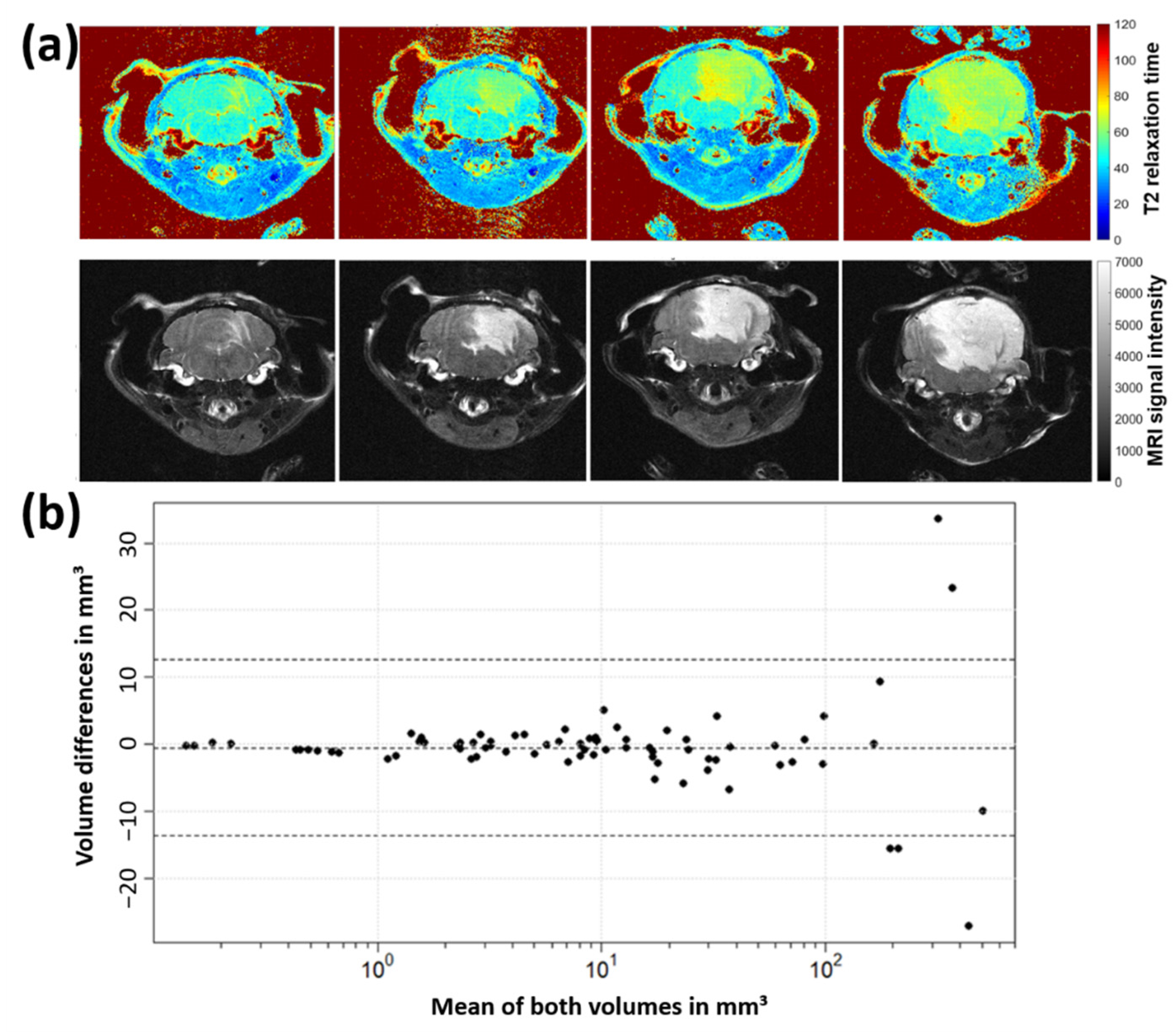
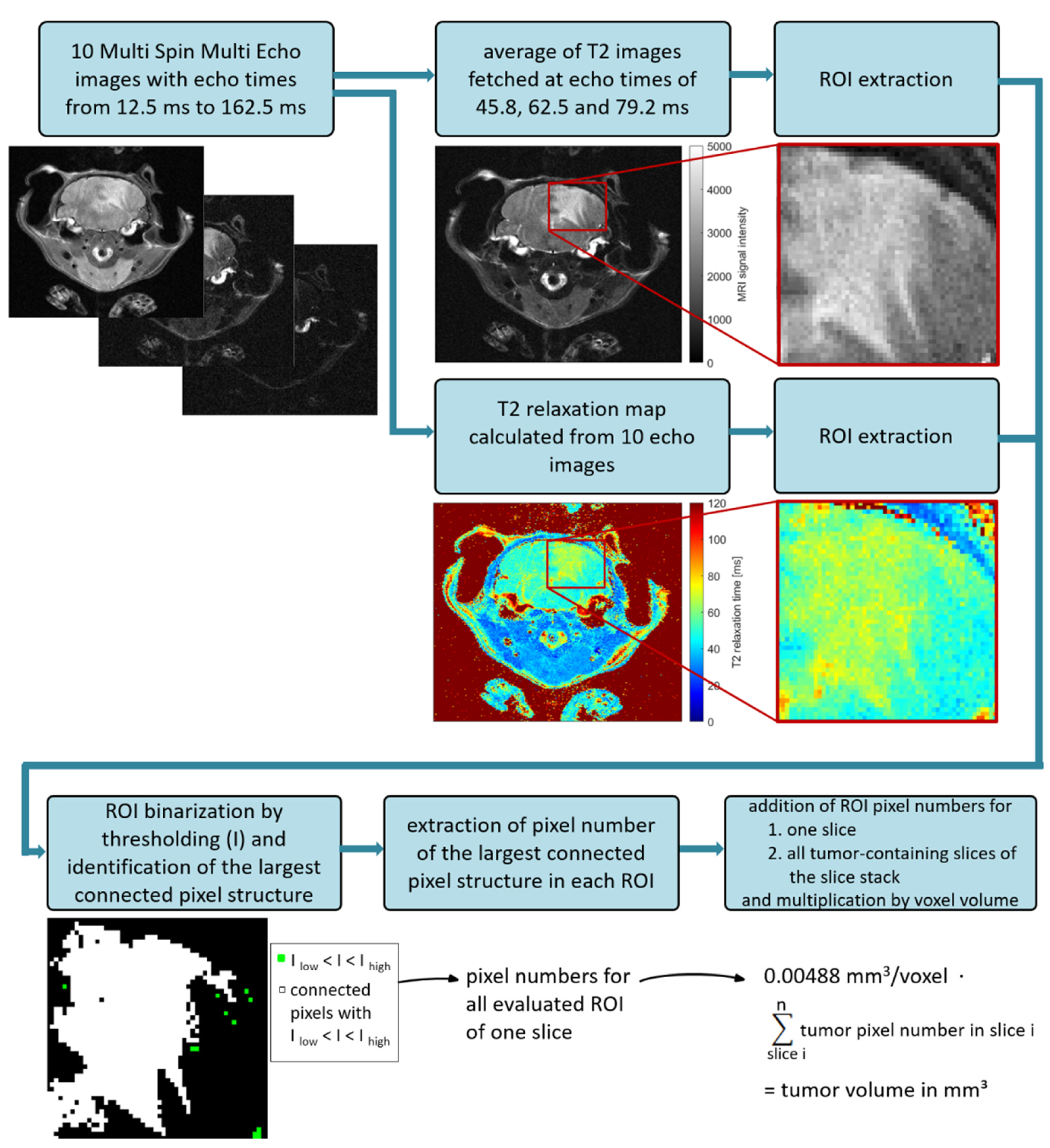
2.1. Optimization of MRI Volumetry Measurements
2.2. Dose-Finding and Toxicity Studies
2.2.1. Whole Brain Irradiation (WBI)
2.2.2. Decitabine and Abacavir
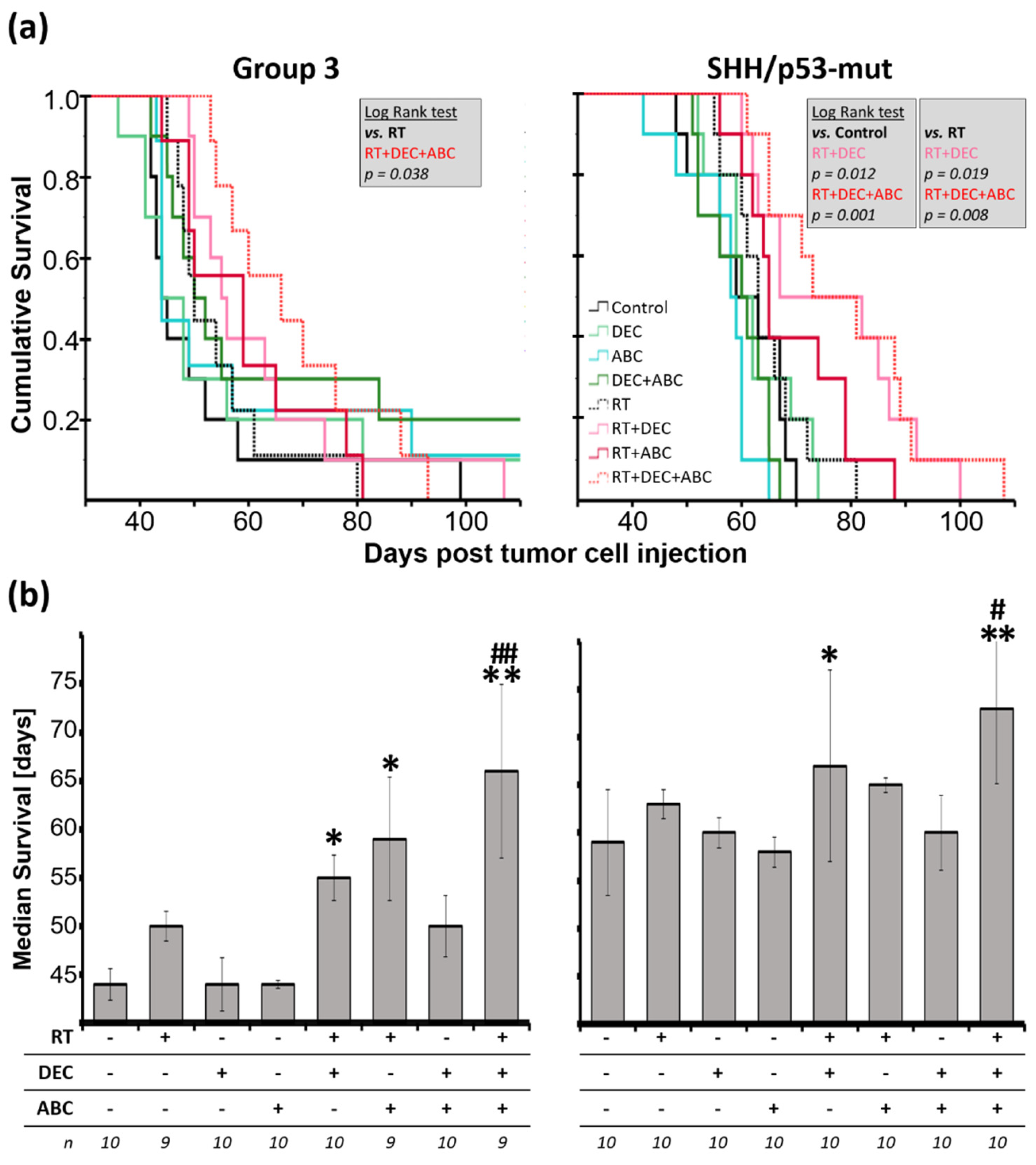
2.3. Enhanced Survival of Mice after Multimodal Therapy
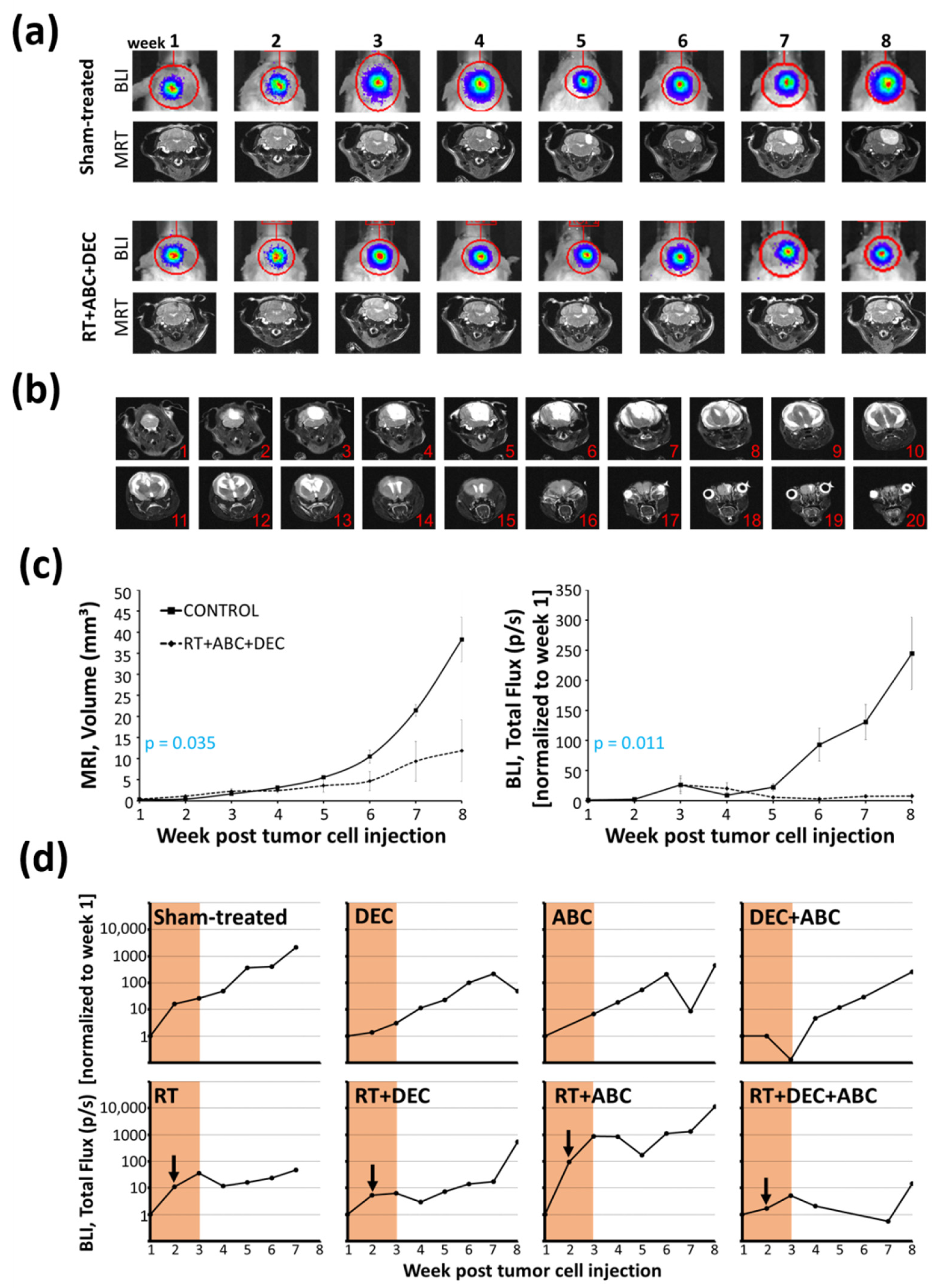
2.4. Inhibition of Tumor Growth after Multimodal Therapy in SHH/TP53-Mut MB
2.5. Strong Correlation of BLI and MRI Data
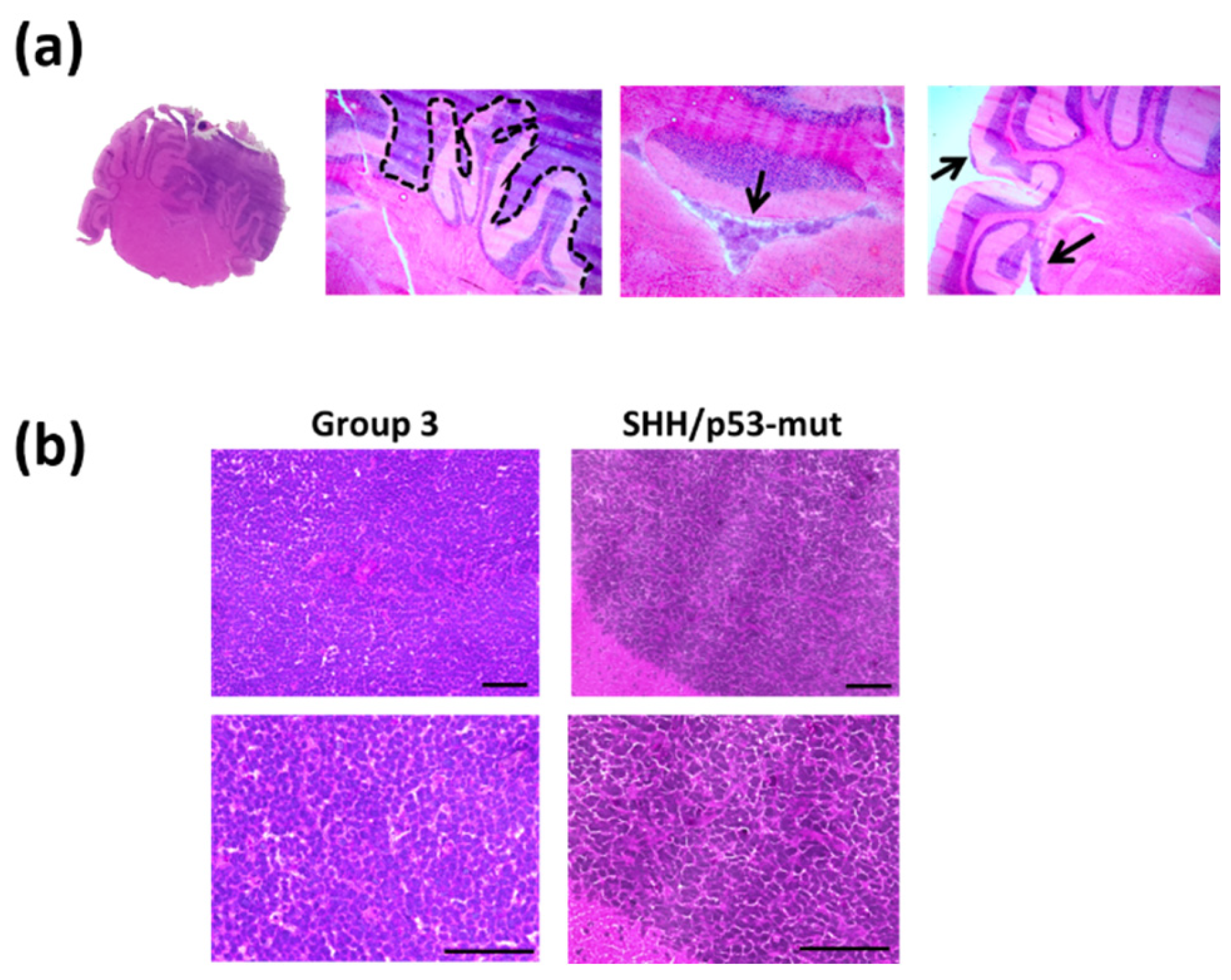
2.6. Tumor Histology
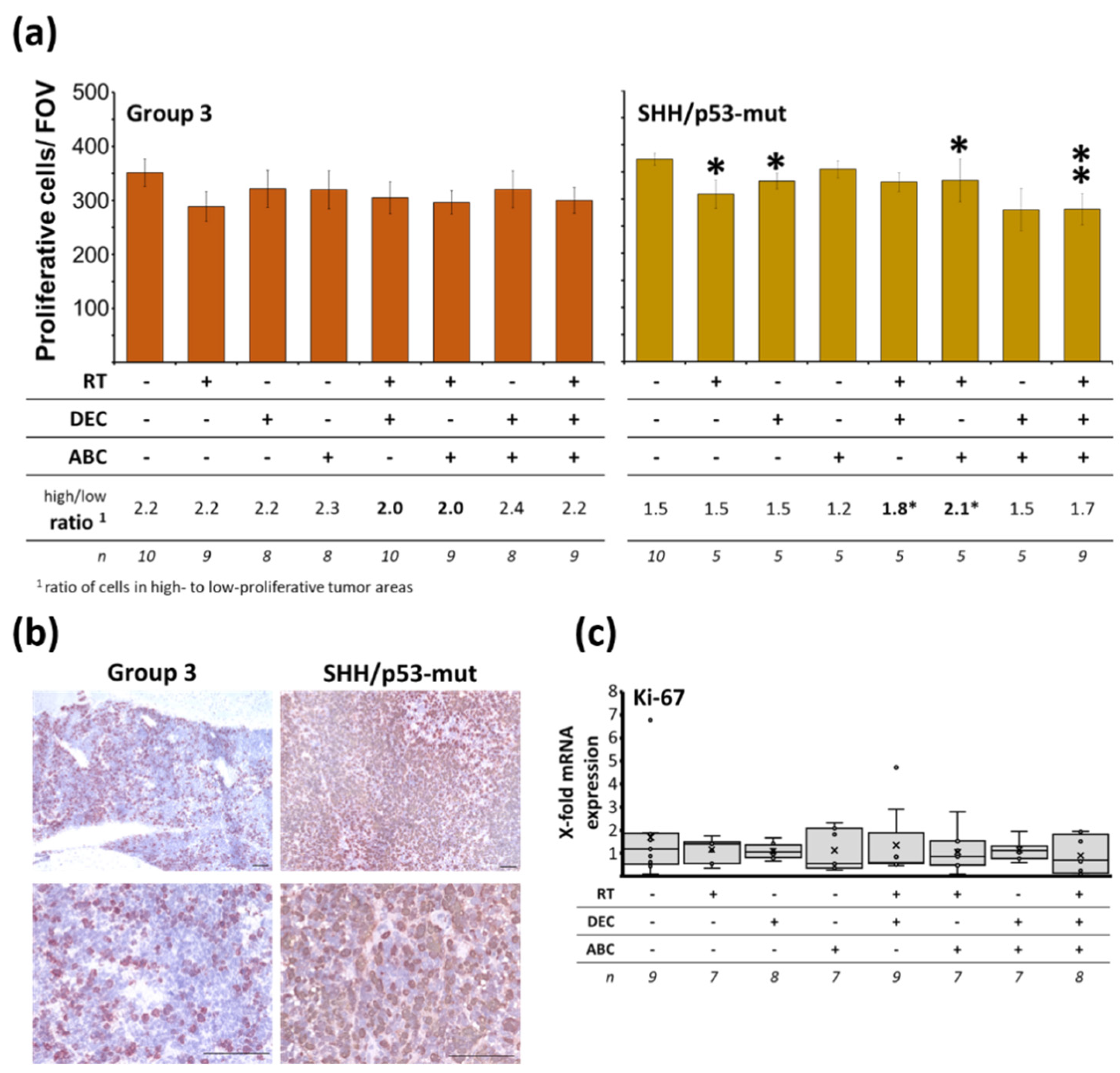
2.7. Reduced Tumor Proliferation Index after Multimodal Therapy in SHH/TP53-Mut MB
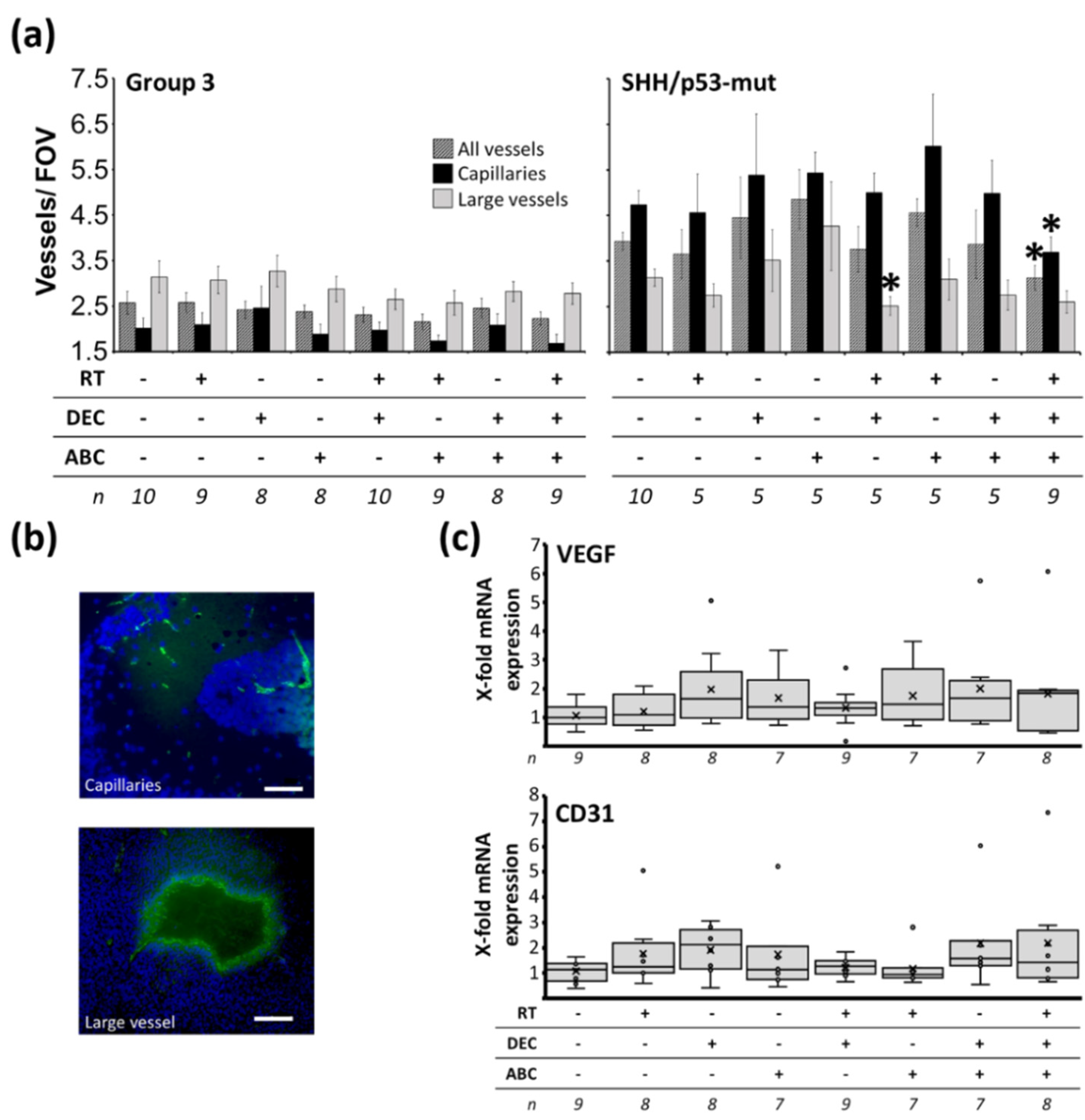
2.8. Reduced Tumor Vascularization after Multimodal Therapy in SHH/TP53-Mut MB
2.9. Tumor Stemness Remained Unchanged after Multimodal Therapy
2.10. Invasion and Activation of Immune Cells
2.11. Therapy-Induced Altered Gene Set Expression
3. Discussion
3.1. MRI Volumetry: T2-Weighted Maps versus Average Images
3.2. BLI Total Flux Correlated Well with MRI-Based Tumor Volume
3.3. Survival Benefit after Multimodal Therapy with RT, DEC, and ABC
3.4. Less Proliferating Tumor Cells in the More Therapy-Resistant SHH/TP53-Mut MB Model after Multimodal Therapy with RT, DEC, and ABC
3.5. Reduced Vascularity after Multimodal Therapy with RT, DEC, and ABC in SHH/TP53-Mut MB
3.6. No Enhancement of Stem Cell Marker mRNA Expression after Multimodal Therapy with RT, DEC, and ABC
3.7. No Therapy-Induced Expression of Immune Cell Markers
3.8. Altered Expression of Genes Involved in Cell Cycle Regulation
4. Materials and Methods
4.1. Animals
4.2. Patient-Derived Xenografts (PDX)
4.3. Orthotopic PDX Mouse Model
4.4. Treatment of MB-Bearing Mice with Decitabine (DEC) and Abacavir (ABC)
4.5. Radiation Therapy of MB-Bearing Mice
4.6. Magnetic Resonance Imaging (MRI)
4.7. MRI Image Processing and Tumor Volumetry Based on T2-Weighted Average Image
4.8. Bioluminescence Imaging (BLI)
4.9. Correlation Analysis of MRI and BLI Values
4.10. Start of the Multimodal Therapy
4.11. Survival Analysis
4.12. Preparation and Hematoxylin/Eosin Staining of Mouse Brain and Liver
4.13. Immunostaining of Ki-67 and CD31
4.14. Gene Expression RT-PCR Analyses
4.15. Gene Expression Microarray Analyses
4.16. Statistics
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ostrom, Q.T.; Gittleman, H.; Truitt, G.; Boscia, A.; Kruchko, C.; Barnholtz-Sloan, J.S. CBTRUS Statistical Report: Primary Brain and Other Central Nervous System Tumors Diagnosed in the United States in 2011–2015. Neuro-Oncology 2018, 20 (Suppl. 4), iv1–iv86. [Google Scholar] [CrossRef] [PubMed]
- Schwalbe, E.; Lindsey, J.C.; Nakjang, S.; Crosier, S.; Smith, A.J.; Hicks, D.; Rafiee, G.; Hill, R.M.; Iliasova, A.; Stone, T.; et al. Novel molecular subgroups for clinical classification and outcome prediction in childhood medulloblastoma: A cohort study. Lancet Oncol. 2017, 18, 958–971. [Google Scholar] [CrossRef]
- Sharma, T.; Schwalbe, E.C.; Williamson, D.; Sill, M.; Hovestadt, V.; Mynarek, M.; Rutkowski, S.; Robinson, G.W.; Gajjar, A.; Cavalli, F.; et al. Second-generation molecular subgrouping of medulloblastoma: An international meta-analysis of Group 3 and Group 4 subtypes. Acta Neuropathol. 2019, 138, 309–326. [Google Scholar] [CrossRef] [PubMed]
- Orr, B.A. Pathology, diagnostics, and classification of medulloblastoma. Brain Pathol. 2020, 30, 664–678. [Google Scholar] [CrossRef]
- Cavalli, F.M.G.; Remke, M.; Rampasek, L.; Peacock, J.; Shih, D.J.H.; Luu, B.; Garzia, L.; Torchia, J.; Nor, C.; Morrissy, A.S.; et al. Intertumoral Heterogeneity within Medulloblastoma Subgroups. Cancer Cell 2017, 31, 737–754.e6. [Google Scholar] [CrossRef] [PubMed]
- Grimes, E. An International Prospective Trial on High-Risk Medulloblastoma in Patients Older than 3 Years. Available online: https://www.clinicaltrialsregister.eu/ctr-search/trial/2018-004250-17/AT#B (accessed on 25 August 2021).
- Jakacki, R.I.; Burger, P.C.; Zhou, T.; Holmes, E.J.; Kocak, M.; Onar, A.; Goldwein, J.; Mehta, M.; Packer, R.J.; Tarbell, N.; et al. Outcome of children with metastatic medulloblastoma treated with carboplatin during craniospinal radiotherapy: A Children’s Oncology Group Phase I/II study. J. Clin. Oncol. 2012, 30, 2648–2653. [Google Scholar] [CrossRef] [PubMed]
- Leary, S.E.; Packer, R.J.; Li, Y.; Billups, C.A.; Smith, K.S.; Jaju, A.; Heier, L.; Burger, P.; Walsh, K.; Han, Y.; et al. Efficacy of Carboplatin and Isotretinoin in Children With High-risk Medulloblastoma: A Randomized Clinical Trial From the Children’s Oncology Group. JAMA Oncol. 2021, 7, 1313–1321. [Google Scholar] [CrossRef]
- Castellino, S.M.; Ullrich, N.J.; Whelen, M.J.; Lange, B.J. Developing interventions for cancer-related cognitive dysfunction in childhood cancer survivors. J. Nat. Cancer Inst. 2014, 106, dju186. [Google Scholar] [CrossRef] [PubMed]
- Marusak, H.A.; Iadipaolo, A.S.; Harper, F.W.; Elrahal, F.; Taub, J.W.; Goldberg, E.; Rabinak, C.A. Neurodevelopmental consequences of pediatric cancer and its treatment: Applying an early adversity framework to understanding cognitive, behavioral, and emotional outcomes. Neuropsychol. Rev. 2018, 28, 123–175. [Google Scholar] [CrossRef] [PubMed]
- Robinson, G.W.; Rudneva, V.; Buchhalter, I.; Billups, C.A.; Waszak, S.; Smith, K.S.; Bowers, D.; Bendel, A.; Fisher, P.; Partap, S.; et al. Risk-adapted therapy for young children with medulloblastoma (SJYC07): Therapeutic and molecular outcomes from a multicentre, phase 2 trial. Lancet Oncol. 2018, 19, 768–784. [Google Scholar] [CrossRef]
- Maddrey, A.M.; Bergeron, J.A.; Lombardo, E.R.; McDonald, N.K.; Mulne, A.F.; Barenberg, P.D.; Bowers, D. Neuropsychological performance and quality of life of 10 year survivors of childhood medulloblastoma. J. Neurooncol. 2005, 72, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Merchant, T.E.; Schreiber, J.E.; Wu, S.; Lukose, R.; Xiong, X.; Gajjar, A. Critical combinations of radiation dose and volume predict intelligence quotient and academic achievement scores after craniospinal irradiation in children with medulloblastoma. Int. J. Radiat. Oncol. Biol. Phys. 2014, 90, 554–561. [Google Scholar] [CrossRef] [PubMed]
- Ms, K.E.R.; Kuttesch, J.F.; Champion, J.E.; Bs, C.F.A.; Hipp, D.W.; Bettis, A.; Barnwell, A.; Compas, B.E. A quantitative meta-analysis of neurocognitive sequelae in survivors of pediatric brain tumors. Pediatr. Blood Cancer 2010, 55, 525–531. [Google Scholar]
- Greene-Schloesser, D.; Robbins, M.E. Radiation-induced cognitive impairment—from bench to bedside. Neuro-Oncology 2012, 14 (Suppl. 4), iv37–iv44. [Google Scholar] [CrossRef] [PubMed]
- Monje, M.L.; Vogel, H.; Masek, M.; Ligon, K.L.; Fisher, P.G.; Palmer, T.D. Impaired human hippocampal neurogenesis after treatment for central nervous system malignancies. Ann. Neurol. 2007, 62, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Monje, M.L.; Mizumatsu, S.; Fike, J.R.; Palmer, T.D. Irradiation induces neural precursor-cell dysfunction. Nat. Med. 2002, 8, 955–962. [Google Scholar] [CrossRef]
- Mynarek, M.; Von Hoff, K.; Pietsch, T.; Ottensmeier, H.; Warmuth-Metz, M.; Bison, B.; Pfister, S.; Korshunov, A.; Sharma, T.; Jaeger, N.; et al. Nonmetastatic Medulloblastoma of Early Childhood: Results From the Prospective Clinical Trial HIT-2000 and An Extended Validation Cohort. J. Clin. Oncol. 2020, 38, 2028–2040. [Google Scholar] [CrossRef] [PubMed]
- Rutkowski, S.; Bode, U.; Deinlein, F.; Ottensmeier, H.; Warmuth-Metz, M.; Soerensen, N.; Graf, N.; Emser, A.; Pietsch, T.; Wolff, J.E.; et al. Treatment of early childhood medulloblastoma by postoperative chemotherapy alone. N. Engl. J. Med. 2005, 352, 978–986. [Google Scholar] [CrossRef] [PubMed]
- Saha, A.; Salley, C.; Saigal, P.; Ms, L.R.; Goldberg, J.D.; Scott, S.; Olshefski, R.; Hukin, J.; Sands, S.A.; Finlay, J.; et al. Late effects in survivors of childhood CNS tumors treated on Head Start I and II protocols. Pediatr. Blood Cancer 2014, 61, 1644–1672. [Google Scholar] [CrossRef]
- Hutchinson, A.D.; Pfeiffer, S.M.; Wilson, C. Cancer-related cognitive impairment in children. Curr. Opin. Support.Palliat. Care 2017, 11, 70–75. [Google Scholar] [CrossRef]
- Krull, K.R.; Hockenberry, M.J.; Miketova, P.; Carey, M.; Moore, I.M. Chemotherapy-related changes in central nervous system phospholipids and neurocognitive function in childhood acute lymphoblastic leukemia. Leuk. Lymphoma 2012, 54, 535–540. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Monje, M.; Dietrich, J. Cognitive side effects of cancer therapy demonstrate a functional role for adult neurogenesis. Behav. Brain Res. 2012, 227, 376–379. [Google Scholar] [CrossRef] [PubMed]
- Dietrich, J.; Han, R.; Yang, Y.; Mayer-Pröschel, M.; Noble, M. CNS progenitor cells and oligodendrocytes are targets of chemotherapeutic agents in vitro and in vivo. J. Biol. 2006, 5, 22. [Google Scholar] [CrossRef] [PubMed]
- De Bont, J.M.; Packer, R.J.; Michiels, E.M.; den Boer, M.L.; Pieters, R. Biological background of pediatric medulloblastoma and ependymoma: A review from a translational research perspective. Neuro. Oncol. 2008, 10, 1040–1060. [Google Scholar] [CrossRef] [PubMed]
- Lindsey, J.C.; Lusher, M.E.; Anderton, J.A.; Bailey, S.; Gilbertson, R.J.; Pearson, A.D.; Ellison, D.W.; Clifford, S.C. Identification of tumour-specific epigenetic events in medulloblastoma development by hypermethylation profiling. Carcinogenesis 2003, 25, 661–668. [Google Scholar] [CrossRef] [PubMed]
- Haltom, A.R.; Toll, S.A.; Cheng, D.; Maegawa, S.; Gopalakrishnan, V.; Khatua, S. Medulloblastoma epigenetics and the path to clinical innovation. J. Neurooncol. 2020, 150, 35–46. [Google Scholar] [CrossRef]
- Schwalbe, E.; Williamson, D.; Lindsey, J.; Hamilton, D.; Ryan, S.L.; Megahed, H.; Garami, M.; Hauser, P.; Dembowska-Baginska, B.; Perek, D.; et al. DNA methylation profiling of medulloblastoma allows robust subclassification and improved outcome prediction using formalin-fixed biopsies. Acta Neuropathol. 2013, 125, 359–371. [Google Scholar] [CrossRef] [PubMed]
- Patties, I.; Jahns, J.; Hildebrandt, G.; Kortmann, R.-D.; Glasow, A. Additive effects of 5-aza-2’-deoxycytidine and irradiation on clonogenic survival of human medulloblastoma cell lines. Strahlenther. Onkol. 2009, 185, 331–338. [Google Scholar] [CrossRef]
- Lindsey, J.C.; Lusher, M.E.; Anderton, J.A.; Gilbertson, R.J.; Ellison, D.W.; Clifford, S.C. Epigenetic deregulation of multiple S100 gene family members by differential hypomethylation and hypermethylation events in medulloblastoma. Br. J. Cancer 2007, 97, 267–274. [Google Scholar] [CrossRef]
- Palii, S.S.; van Emburgh, B.O.; Sankpal, U.T.; Brown, K.D. Robertson KDDNA methylation inhibitor 5-Aza-2’-deoxycytidine induces reversible genome-wide DNA damage that is distinctly influenced by DNA methyltransferases 1 and 3B. Mol. Cell. Biol. 2008, 28, 752–771. [Google Scholar] [CrossRef]
- Patties, I.; Kortmann, R.-D.; Glasow, A. Inhibitory effects of epigenetic modulators and differentiation inducers on human medulloblastoma cell lines. J. Exp. Clin. Cancer Res. 2013, 32, 27–28. [Google Scholar] [CrossRef]
- Patties, I.; Kortmann, R.-D.; Menzel, F.; Glasow, A. Enhanced inhibition of clonogenic survival of human medulloblastoma cells by multimodal treatment with ionizing irradiation, epigenetic modifiers, and differentiation-inducing drugs. J. Exp. Clin. Cancer Res. 2016, 35, 94. [Google Scholar] [CrossRef] [PubMed]
- Seelan, R.S.; Mukhopadhyay, P.; Pisano, M.M.; Greene, R.M. Effects of 5-Aza-2’-deoxycytidine (decitabine) on gene expression. Drug Metab. Rev. 2017, 50, 193–207. [Google Scholar] [CrossRef] [PubMed]
- Chang, Q.; Pang, J.C.-S.; Li, J.; Hu, L.; Kong, X.; Ng, H.-K. Molecular analysis of PinX1 in medulloblastomas. Int. J. Cancer. 2004, 109, 309–314. [Google Scholar] [CrossRef]
- Didiano, D.; Shalaby, T.; Lang, D.; Grotzer, M.A. Telomere maintenance in childhood primitive neuroectodermal brain tumors. Neuro-Oncology 2004, 6, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.D.; Leão, R.; Komosa, M.; Gallo, M.; Zhang, C.H.; Lipman, T.; Remke, M.; Heidari, A.; Nunes, N.M.; Apolonio, J.D.; et al. DNA hypermethylation within TERT promoter upregulates TERT expression in cancer. J. Clin. Investig. 2019, 129, 223–229. [Google Scholar] [CrossRef]
- Minasi, S.; Baldi, C.; Pietsch, T.; Donofrio, V.; Pollo, B.; Antonelli, M.; Massimino, M.; Giangaspero, F.; Buttarelli, F.R. Telomere elongation via alternative lengthening of telomeres (ALT) and telomerase activation in primary metastatic medulloblastoma of childhood. J. Neurooncol. 2019, 142, 435–444. [Google Scholar] [CrossRef]
- Lindsey, J.; Schwalbe, E.; Potluri, S.; Bailey, S.; Williamson, D.; Clifford, S.C. TERT promoter mutation and aberrant hypermethylation are associated with elevated expression in medulloblastoma and characterise the majority of non-infant SHH subgroup tumours. Acta Neuropathol. 2014, 127, 307–309. [Google Scholar] [CrossRef] [PubMed]
- Remke, M.; Ramaswamy, V.; Peacock, J.; Shih, D.J.H.; Koelsche, C.; Northcott, P.A.; Hill, N.; Cavalli, F.M.G.; Kool, M.; Wang, X.; et al. TERT promoter mutations are highly recurrent in SHH subgroup medulloblastoma. Acta Neuropathol. 2013, 126, 917–929. [Google Scholar] [CrossRef] [PubMed]
- Varatharajan, L.; Thomas, S.A. The transport of anti-HIV drugs across blood-CNS interfaces: Summary of current knowledge and recommendations for further research. Antiviral Res. 2009, 82, A99–A109. [Google Scholar] [CrossRef] [PubMed]
- Rossi, A.; Russo, G.; Puca, A.; La Montagna, R.; Caputo, M.; Mattioli, E.; Lopez, M.; Giordano, A.; Pentimalli, F. The antiretroviral nucleoside analogue Abacavir reduces cell growth and promotes differentiation of human medulloblastoma cells. Int. J. Cancer 2009, 125, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Huybrechts, S.; Le Teuff, G.; Tauziède-Espariat, A.; Rossoni, C.; Chivet, A.; Indersie, .; Varlet, P.; Puget, S.; Abbas, R.; Ayrault, O.; et al. Prognostic Clinical and Biologic Features for Overall Survival after Relapse in Childhood Medulloblastoma. Cancers 2020, 13, 53. [Google Scholar] [CrossRef] [PubMed]
- Remke, M.; Ramaswamy, V. Infant medulloblastoma—learning new lessons from old strata. Nat. Rev. Clin. Oncol. 2018, 15, 659–660. [Google Scholar] [CrossRef] [PubMed]
- Hill, R.M.; Kuijper, S.; Lindsey, J.; Petrie, K.; Schwalbe, E.; Barker, K.; Boult, J.K.; Williamson, D.; Ahmad, Z.; Hallsworth, A.; et al. Combined MYC and P53 defects emerge at medulloblastoma relapse and define rapidly progressive, therapeutically targetable disease. Cancer Cell 2015, 27, 72–84. [Google Scholar] [CrossRef] [PubMed]
- Ramaswamy, V.; Remke, M.; Bouffet, E.; Faria, C.C.; Perreault, S.; Cho, Y.-J.; Shih, D.J.; Luu, B.; Dubuc, A.M.; Northcott, P.A.; et al. Recurrence patterns across medulloblastoma subgroups: An integrated clinical and molecular analysis. Lancet Oncol. 2013, 14, 1200–1207. [Google Scholar] [CrossRef]
- Stewart, E.; Federico, S.M.; Chen, X.; Shelat, A.A.; Bradley, C.; Gordon, B.; Karlstrom, A.; Twarog, N.R.; Clay, M.R.; Bahrami, A.; et al. Orthotopic patient-derived xenografts of paediatric solid tumours. Nature 2017, 549, 96–100. [Google Scholar] [CrossRef]
- Williams, J.A. Using PDX for Preclinical Cancer Drug Discovery: The Evolving Field. J. Clin. Med. 2018, 7, 41. [Google Scholar] [CrossRef]
- Zhao, X.; Liu, Z.; Yu, L.; Zhang, Y.; Baxter, P.; Voicu, H.; Gurusiddappa, S.; Luan, J.; Su, J.M.; Leung, H.-C.E.; et al. Global gene expression profiling confirms the molecular fidelity of primary tumor-based orthotopic xenograft mouse models of medulloblastoma. Neuro-Oncology 2012, 14, 574–583. [Google Scholar] [CrossRef]
- Brabetz, S.; Leary, S.E.S.; Gröbner, S.N.; Nakamoto, M.W.; Şeker-Cin, H.; Girard, E.; Cole, B.; Strand, A.D.; Bloom, K.L.; Hovestadt, V.; et al. A biobank of patient-derived pediatric brain tumor models. Nat Med. 2018, 24, 1752–1761. [Google Scholar] [CrossRef]
- Smith, S.M.; Bianski, B.M.; Orr, B.A.; Harknett, G.; Onar-Thomas, A.; Gilbertson, R.J.; Merchant, T.E.; Roussel, M.F.; Tinkle, C.L. Preclinical Modeling of Image-Guided Craniospinal Irradiation for Very-High-Risk Medulloblastoma. Int. J. Radiat. Oncol. 2018, 103, 728–737. [Google Scholar] [CrossRef]
- Chung, S.H.; Cerussi, A.E.; Klifa, C.; Baek, H.M.; Birgul, O.; Gulsen, G.; Merritt, S.I.; Hsiang, D.; Tromberg, B.J. In vivo water state measurements in breast cancer using broadband diffuse optical spectroscopy. Phys. Med. Biol. 2008, 53, 6713–6727. [Google Scholar] [CrossRef] [PubMed]
- Mahapatra, S.; Amsbaugh, M.J. Medulloblastoma; StatPearls: Treasure Island, FL, USA, 2021. [Google Scholar]
- Rehemtulla, A.; Stegman, L.D.; Cardozo, S.J.; Gupta, S.; Hall, D.E.; Contag, C.H.; Ross, B.D. Rapid and quantitative assessment of cancer treatment response using in vivo bioluminescence imaging. Neoplasia 2000, 2, 491–495. [Google Scholar] [CrossRef] [PubMed]
- Szentirmai, O.; Baker, C.H.; Lin, N.; Szucs, S.; Takahashi, M.; Kiryu, S.; Kung, A.L.; Mulligan, R.C.; Carter, B.S. Noninvasive bioluminescence imaging of luciferase expressing intracranial U87 xenografts: Correlation with magnetic resonance imaging determined tumor volume and longitudinal use in assessing tumor growth and antiangiogenic treatment effect. Neurosurgery 2006, 58, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Jost, S.C.; Collins, L.; Travers, S.; Piwnica-Worms, D.; Garbow, J.R. Measuring brain tumor growth: Combined bioluminescence imaging-magnetic resonance imaging strategy. Mol. Imaging 2009, 8, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Crowther, A.J.; Ocasio, J.; Fang, F.; Meidinger, J.; Wu, J.; Deal, A.M.; Chang, S.X.; Yuan, H.; Schmid, R.; Davis, I.; et al. Radiation Sensitivity in a Preclinical Mouse Model of Medulloblastoma Relies on the Function of the Intrinsic Apoptotic Pathway. Cancer Res. 2016, 76, 3211–3223. [Google Scholar] [CrossRef] [PubMed]
- Ramaswamy, V.; Nör, C.; Taylor, M.D. p53 and Meduloblastoma. Cold Spring Harb Perspect Med. 2015, 6, a026278. [Google Scholar] [CrossRef]
- Hollenbach, P.W.; Nguyen, A.N.; Brady, H.; Williams, M.; Ning, Y.; Richard, N.; Krushel, L.; Aukerman, S.L.; Heise, C.; Macbeth, K.J. A comparison of azacitidine and decitabine activities in acute myeloid leukemia cell lines. PLoS ONE 2010, 5, e9001. [Google Scholar] [CrossRef]
- Wright, W.E.; Brasiskyte, D.; Piatyszek, M.A.; Shay, J.W. Experimental elongation of telomeres extends the lifespan of immortal x normal cell hybrids. EMBO J. 1996, 15, 1734–1741. [Google Scholar] [CrossRef]
- Vaziri, H.; West, M.D.; Allsopp, R.C.; Davison, T.S.; Wu, Y.-S.; Arrowsmith, C.; Poirier, G.G.; Benchimol, S. ATM-dependent telomere loss in aging human diploid fibroblasts and DNA damage lead to the post-translational activation of p53 protein involving poly(ADP-ribose) polymerase. EMBO J. 1997, 16, 6018–6033. [Google Scholar] [CrossRef]
- Grotzer, M.A.; Geoerger, B.; Janss, A.J.; Zhao, H.; Rorke, L.B.; Phillips, P.C. Prognostic significance of Ki-67 (MIB-1) proliferation index in childhood primitive neuroectodermal tumors of the central nervous system. Med. Pediatr. Oncol. 2001, 36, 268–273. [Google Scholar] [CrossRef]
- Das, P.; Puri, T.; Suri, V.; Sharma, M.C.; Sharma, B.S.; Sarkar, C. Medulloblastomas: A correlative study of MIB-1 proliferation index along with expression of c-Myc, ERBB2, and anti-apoptotic proteins along with histological typing and clinical outcome. Childs Nerv. Syst. 2009, 25, 825–835. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, A.F.; Araújo, M.B.M.; Aguiar, P.H.; Plese, J.P.P. Medulloblastoma: Evaluation of proliferative index by monoclonal antibody Mib-1, its prognostic correlation and therapeutic implications. Arq. Neuro-Psiquiatr. 2003, 61, 547–551. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cheng, L.; Sebo, T.J.; Cheville, J.C.; Pisansky, T.M.; Slezak, J.; Bergstralh, E.J.; Pacelli, A.; Neumann, R.M.; Zincke, H.; Bostwick, D.G. p53 Protein overexpression is associated with increased cell proliferation in patients with locally recurrent prostate carcinoma after radiation therapy. Cancer 1999, 85, 1293–1299. [Google Scholar] [CrossRef]
- Phoenix, T.N.; Patmore, D.M.; Boop, S.; Boulos, N.; Jacus, M.O.; Patel, Y.T.; Roussel, M.F.; Finkelstein, D.; Goumnerova, L.; Perreault, S.; et al. Medulloblastoma Genotype Dictates Blood Brain Barrier Phenotype. Cancer Cell 2016, 29, 508–522. [Google Scholar] [CrossRef]
- Cosse, J.-P.; Michiels, C. Tumour hypoxia affects the responsiveness of cancer cells to chemotherapy and promotes cancer progression. ACAMC 2008, 8, 790–797. [Google Scholar] [CrossRef]
- Singh, S.K.; Hawkins, C.; Clarke, I.D.; Squire, J.A.; Bayani, J.; Hide, T.; Henkelman, R.M.; Cusimano, M.D.; Dirks, P.B. Identification of human brain tumour initiating cells. Nature 2004, 432, 396–401. [Google Scholar] [CrossRef]
- Read, T.-A.; Fogarty, M.P.; Markant, S.L.; McLendon, R.E.; Wei, Z.; Ellison, D.W.; Febbo, P.G.; Wechsler-Reya, R.J. Identification of CD15 as a marker for tumor-propagating cells in a mouse model of medulloblastoma. Cancer Cell 2009, 15, 135–147. [Google Scholar] [CrossRef]
- Sutter, R.; Shakhova, O.; Bhagat, H.; Behesti, H.; Sutter, C.; Penkar, S.; Santuccione, A.; Bernays, R.; Heppner, F.L.; Schüller, U.; et al. Cerebellar stem cells act as medulloblastoma-initiating cells in a mouse model and a neural stem cell signature characterizes a subset of human medulloblastomas. Oncogene 2010, 29, 1845–1856. [Google Scholar] [CrossRef]
- Yu, C.-C.; Chiou, G.-Y.; Lee, Y.-Y.; Chang, Y.-L.; Huang, P.-I.; Cheng, Y.-W.; Tai, L.-K.; Ku, H.-H.; Chiou, S.-H.; Wong, T.-T. Medulloblastoma-derived tumor stem-like cells acquired resistance to TRAIL-induced apoptosis and radiosensitivity. Child Nerv. Syst. 2010, 26, 897–904. [Google Scholar] [CrossRef]
- Hambardzumyan, D.; Becher, O.J.; Rosenblum, M.K.; Pandolfi, P.P.; Manova-Todorova, K.; Holland, E.C. PI3K pathway regulates survival of cancer stem cells residing in the perivascular niche following radiation in medulloblastoma in vivo. Genes Dev. 2008, 22, 436–448. [Google Scholar] [CrossRef]
- Huang, G.-H.; Xu, Q.-F.; Cui, Y.-H.; Li, N.; Bian, X.-W.; Lv, S.-Q. Medulloblastoma stem cells: Promising targets in medulloblastoma therapy. Cancer Sci. 2016, 107, 583–589. [Google Scholar] [CrossRef] [PubMed]
- Baumann, M.; Krause, M.; Hill, R. Exploring the role of cancer stem cells in radioresistance. Nat. Rev. Cancer. 2008, 8, 545–554. [Google Scholar] [CrossRef] [PubMed]
- Rycaj, K.; Tang, D.G. Cancer stem cells and radioresistance. Int. J. Radiat. Biol. 2014, 90, 615–621. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Jeong, E.K.; Ju, M.K.; Jeon, H.M.; Kim, M.Y.; Kim, C.H.; Park, H.G.; Han, S.I.; Kang, H.S. Induction of metastasis, cancer stem cell phenotype, and oncogenic metabolism in cancer cells by ionizing radiation. Mol. Cancer 2017, 16, 10. [Google Scholar] [CrossRef]
- Lagadec, C.; Vlashi, E.; Della Donna, L.; Meng, Y.; Dekmezian, C.; Kim, K.; Pajonk, F. Survival and self-renewing capacity of breast cancer initiating cells during fractionated radiation treatment. Breast Cancer Res. 2010, 12, R13. [Google Scholar] [CrossRef]
- Liu, L.; Chen, L.; Wu, X.; Li, X.; Song, Y.; Mei, Q.; Nie, J.; Han, W. Low-dose DNA-demethylating agent enhances the chemosensitivity of cancer cells by targeting cancer stem cells via the upregulation of microRNA-497. J. Cancer Res. Clin. Oncol. 2016, 142, 1431–1439. [Google Scholar] [CrossRef]
- Bockmayr, M.; Mohme, M.; Klauschen, F.; Winkler, B.; Budczies, J.; Rutkowski, S.; Schüller, U. Subgroup-specific immune and stromal microenvironment in medulloblastoma. OncoImmunology 2018, 7, e1462430. [Google Scholar] [CrossRef]
- Vermeulen, J.F.; Van Hecke, W.; Adriaansen, E.J.M.; Jansen, M.K.; Bouma, R.G.; Hidalgo, J.V.; Fisch, P.; Broekhuizen, R.; Spliet, W.G.M.; Kool, M.; et al. Prognostic relevance of tumor-infiltrating lymphocytes and immune checkpoints in pediatric medulloblastoma. OncoImmunology 2018, 7, e1398877. [Google Scholar] [CrossRef]
- Pham, C.D.; Flores, C.; Yang, C.; Pinheiro, E.M.; Yearley, J.H.; Sayour, E.J.; Pei, Y.; E Moore, C.; McLendon, R.E.; Huang, J.; et al. Differential Immune Microenvironments and Response to Immune Checkpoint Blockade among Molecular Subtypes of Murine Medulloblastoma. Clin Cancer Res. 2016, 22, 582–595. [Google Scholar] [CrossRef]
- Diao, S.; Gu, C.; Zhang, H.; Yu, C. Immune cell infiltration and cytokine secretion analysis reveal a non-inflammatory microenvironment of medulloblastoma. Oncol. Lett. 2020, 20, 397. [Google Scholar] [CrossRef]
- The Jackson Laboratory. NSG Mice. Available online: https://www.jax.org/strain/005557 (accessed on 12 February 2021).
- Yamauchi, T.; Takenaka, K.; Urata, S.; Shima, T.; Kikushige, Y.; Tokuyama, T.; Iwamoto, C.; Nishihara, M.; Iwasaki, H.; Miyamoto, T.; et al. Polymorphic Sirpa is the genetic determinant for NOD-based mouse lines to achieve efficient human cell engraftment. Blood 2013, 121, 1316–1325. [Google Scholar] [CrossRef] [PubMed]
- Hutchinson, S.A.; Tooke-Locke, E.; Wang, J.; Tsai, S.; Katz, T.; Trede, N.S. Tbl3 regulates cell cycle length during zebrafish development. Dev. Biol. 2012, 368, 261–272. [Google Scholar] [CrossRef] [PubMed]
- Uhlén, M.; Zhang, C.; Lee, S.; Sjöstedt, E.; Fagerberg, L.; Bidkhori, G.; Benfeitas, R.; Arif, M.; Liu, Z.; Edfors, F.; et al. A pathology atlas of the human cancer transcriptome. Science 2017, 357, eaan2507. [Google Scholar] [CrossRef] [PubMed]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001, 29, e45. [Google Scholar] [CrossRef]
- Bijwaard, K.E.; Aguilera, N.S.I.; Monczak, Y.; Trudel, M.; Taubenberger, J.K.; Lichy, J.H. Quantitative Real-Time Reverse Transcription-PCR Assay for Cyclin D1 Expression: Utility in the Diagnosis of Mantle Cell Lymphoma. Clin. Chem. 2001, 47, 195–201. [Google Scholar] [CrossRef] [PubMed]




| Therapy | Group 3 (n = 77) | SHH/TP53-Mutated (n = 80) |
|---|---|---|
| Sham-treated | 10 | 10 |
| RT | 9 | 10 |
| DEC | 10 | 10 |
| ABC | 10 | 10 |
| RT/DEC | 10 | 10 |
| RT/ABC | 9 | 10 |
| DEC/ABC | 10 | 10 |
| RT/DEC/ABC | 9 | 10 |
| Species*/Gene *M…Mouse; H…Human NCBI Reference Sequence | Forward Primer (5′-3′) | Probe (5′-3′) | Reverse Primer (5′-3′) |
|---|---|---|---|
| mVEGF NM_009505.4 | AACGATGAAGCCCTGGAGTG | YakimaYellow™-CGTGCCCACGTCAGAGAGCAACATCA-BHQ1 | ATGTGCTGGCTTTGGTGAGG |
| mCD31 NM_001032378.2 | CCAGGTGTGCGAAATGCTCT | YakimaYellow™-AAGGACATGCCATAGGCATCAGCT-BHQ1 | GGTGGTAAGTGATGGGTGCAG |
| mIBA NM_019467.2 | GCAGGGATTTGCAGGGAGGA | HEX-CCAGCCTCTCTTCCTGCTGGGCC-BHQ1 | TGGACGGCAGATCCTCATCA |
| mCD68 NM_001291058.1 | GCTGTGGAAATGCAAGCATAG | HEX-AGGCTACAGGCTGCTCAGCTGCC-BHQ1 | GAGAAACATGGCCCGAAGT |
| hNestin NM_006617.2 | AGGAGAAACAGGGCCTACA | YakimaYellow™-CACCTCAAGATGTCCCTCAGCCTG-BHQ1 | AGGAGGGTCCTGTACGTG |
| hCD133 NM_001145852.2 | ATGAAACTCCAGAGCAAATC | YakimaYellow™-TACAACACTACCAAGGACAAGGCG-BHQ1 | GTCTCAGTCGGTCAAGAA |
| hCD15 NM_002033.4 | GGGTTTGGATGAACTTCG | YakimaYellow™-AGAGCGTCCAGTTGAAGAGGTTAC-BHQ1 | GGGTAGAGGTAGCCATAAG |
| hKi-67 NM_002417.5 | CAGAATGGAAGGAAGTCAAC | YakimaYellow™-AATACGTGAACAGGAGCCAGCA-BHQ1 | TTCTCATCAGGGTCAGAAG |
| mß2-microglobulin NM_009735.3 | TGAGACTGATACATACGCCTGCA | HEX-ATGGCCGAGCCCAAGACCGTC-BHQ1 | GATGCTTGATCACATGTCTCGATC |
| hß2-microglobulin [88] | TGACTTTGTCACAGCCCAAGATA | BHQ1-TGATGCTGCTTACATGTCTCGATCCCA-HEX | AATCCAAATGCGGCATCTTC |
| hHPRT1 NM_004048.4 | GACTTTGCTTTCCTTGGTCAG | YakimaYellow™-CCAAAGATGGTCAAGGTCGCAAGC-BHQ1 | TGGCTTATATCCAACACTTCGT |
| mhprt1 NM_009735.3 | AAACTTTGCTTTCCCTGGTTAA | YakimaYellow™-ACCAGCAAGCTTGCAACCTTAACC-BHQ1 | CCTGTATCCAACACTTCGAGA |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gringmuth, M.; Walther, J.; Greiser, S.; Toussaint, M.; Schwalm, B.; Kool, M.; Kortmann, R.-D.; Glasow, A.; Patties, I. Enhanced Survival of High-Risk Medulloblastoma-Bearing Mice after Multimodal Treatment with Radiotherapy, Decitabine, and Abacavir. Int. J. Mol. Sci. 2022, 23, 3815. https://doi.org/10.3390/ijms23073815
Gringmuth M, Walther J, Greiser S, Toussaint M, Schwalm B, Kool M, Kortmann R-D, Glasow A, Patties I. Enhanced Survival of High-Risk Medulloblastoma-Bearing Mice after Multimodal Treatment with Radiotherapy, Decitabine, and Abacavir. International Journal of Molecular Sciences. 2022; 23(7):3815. https://doi.org/10.3390/ijms23073815
Chicago/Turabian StyleGringmuth, Marieke, Jenny Walther, Sebastian Greiser, Magali Toussaint, Benjamin Schwalm, Marcel Kool, Rolf-Dieter Kortmann, Annegret Glasow, and Ina Patties. 2022. "Enhanced Survival of High-Risk Medulloblastoma-Bearing Mice after Multimodal Treatment with Radiotherapy, Decitabine, and Abacavir" International Journal of Molecular Sciences 23, no. 7: 3815. https://doi.org/10.3390/ijms23073815
APA StyleGringmuth, M., Walther, J., Greiser, S., Toussaint, M., Schwalm, B., Kool, M., Kortmann, R.-D., Glasow, A., & Patties, I. (2022). Enhanced Survival of High-Risk Medulloblastoma-Bearing Mice after Multimodal Treatment with Radiotherapy, Decitabine, and Abacavir. International Journal of Molecular Sciences, 23(7), 3815. https://doi.org/10.3390/ijms23073815







