Glutathione Depletion and MicroRNA Dysregulation in Multiple System Atrophy: A Review
Abstract
1. Introduction
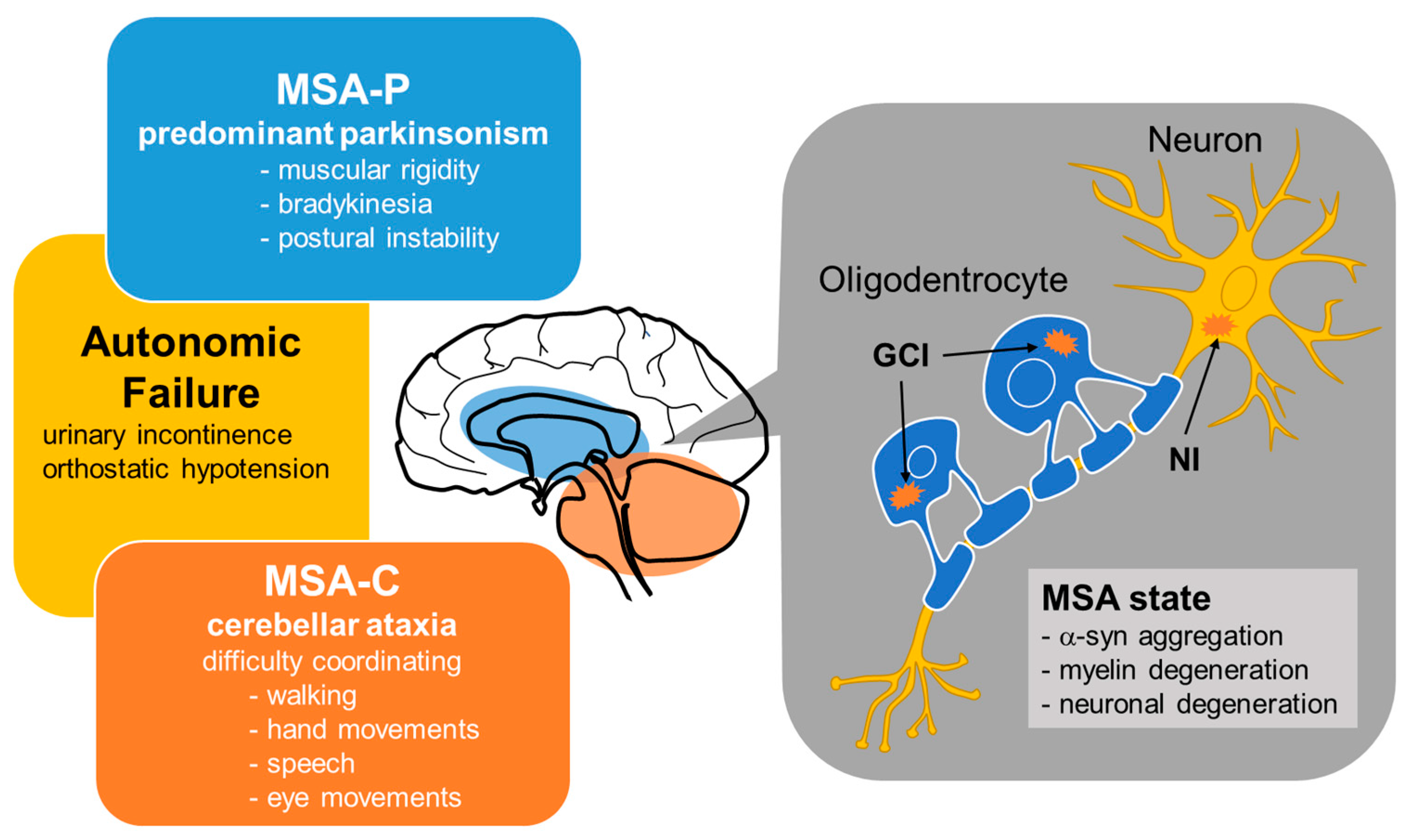
2. An Overview of MSA
2.1. Clinical Classification of MSA
2.2. Pathological Features of MSA
2.3. Distinction between MSA and Lewy Body Diseases
3. Genes Associated with MSA Features
| Gene Name | Encoding Protein | Physiological Function | Pathology | Related NDs [69] |
|---|---|---|---|---|
| SNCA | α-synuclein | regulation of synaptic vesicles and neurotransmitter release | inclusion formation | PD, LBD |
| COQ2 | polyprenyl transferase | catalyzing coenzyme Q10 biosynthesis | coenzyme Q10 deficiency, followed by mitochondrial dysfunction | PD |
| MAPT | microtubule-associated protein tau | formation and stabilization of axonal microtubules | tau accumulation | AD, FTD, PD |
| LRRK2 | leucine rich-repeat kinase 2 | involved in neuronal plasticity, autophagy, and vesicle trafficking | associated with pathologies of α-syn | PD |
| GBA1 | β-glucocerebrosidase | hydrolysis of glucosylceramide and glucosylsphingosine | α-syn accumulation | AD, FTD, PD |
| TBP | TATA-box binding protein | component of the eukaryotic transcription initiation machinery | formation of aggregates | PD, HD |
| CHCHD2 | coiled-coil-helix-coiled-coil-helix domain containing 2 | regulating electron flow in the mitochondrial electron transport chain | mitochondrial dysfunction | AD, FTD, PD, ALS, |
| c9orf72 | chromosome 9 open reading frame 72 | regulation of autophagy and vesicular trafficking | formation of aggregates | FTD, ALS |
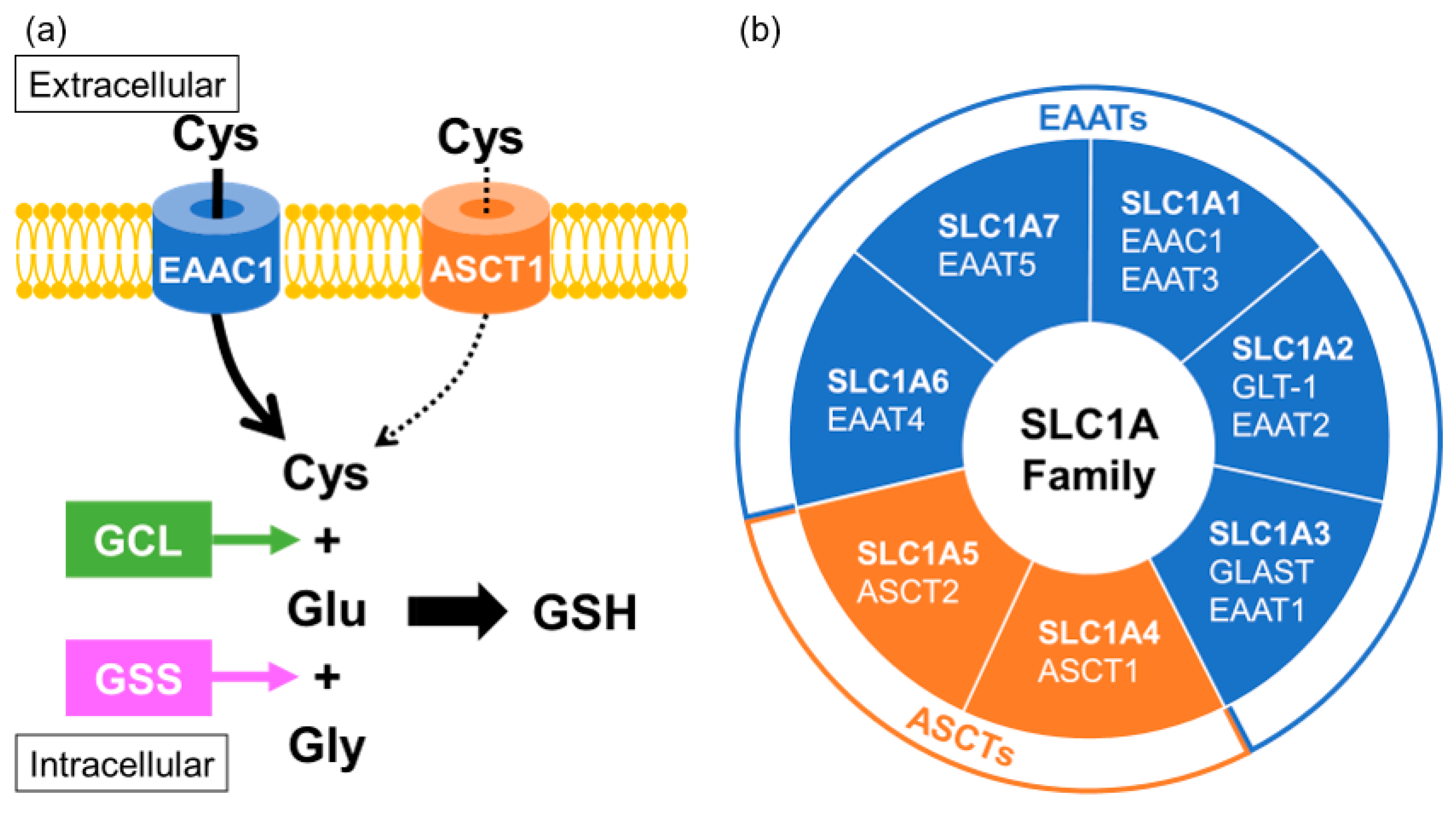
| ASCT1 | alanine/serine/cysteine/threonine transporter 1 | uptake of neutral amino acids | enhanced oxidative stress | No data |
| EAAC1 | excitatory amino acid carrier 1 | cysteine uptake | GSH depletion, followed by oxidative stress | No data |
| TauT | taurine transporter | taurine uptake | taurine depletion, followed by oxidative stress | No data |
| NOVA1 | neuro-oncological ventral antigen-1 | alternative splicing, involved in the formation and activity of the synapses | autoantigen in paraneoplastic opsoclonus myoclonus ataxia | No data |
| Oct1 | organic cation transporter1 | Translocation of organic cations across the blood–brain barrier | unknown | No data |
4. The Association between GSH Dysregulation and MSA
4.1. GSH Levels in Patients with MSA
4.2. Possible Association of the Enzymes for GSH Synthesis and Metabolism with MSA
4.3. Association between Transporters Related to GSH Biosynthesis and MSA
5. The Association of miRNA Dysregulation and MSA Pathology
5.1. Molecular Mechanisms of miRNA Biogenesis
5.2. Dysregulation of Genes and miRNAs in Patients with MSA
5.3. Candidate miRNA Biomarkers for MSA
6. Prospective for Diagnosis and Treatment of NDs including MSA
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Fanciulli, A.; Wenning, G.K. Multiple-system atrophy. N. Engl. J. Med. 2015, 372, 249–263. [Google Scholar] [CrossRef] [PubMed]
- Palma, J.A.; Norcliffe-Kaufmann, L.; Kaufmann, H. Diagnosis of multiple system atrophy. Auton. Neurosci. Basic Clin. 2018, 211, 15–25. [Google Scholar] [CrossRef]
- Schweighauser, M.; Shi, Y.; Tarutani, A.; Kametani, F.; Murzin, A.G.; Ghetti, B.; Matsubara, T.; Tomita, T.; Ando, T.; Hasegawa, K.; et al. Structures of α-synuclein filaments from multiple system atrophy. Nature 2020, 585, 464–469. [Google Scholar] [CrossRef] [PubMed]
- Jellinger, K.A. Multiple System Atrophy: An Oligodendroglioneural Synucleinopathy1. J. Alzheimer’s Dis. JAD 2018, 62, 1141–1179. [Google Scholar] [CrossRef] [PubMed]
- Kaji, S.; Maki, T.; Ishimoto, T.; Yamakado, H.; Takahashi, R. Insights into the pathogenesis of multiple system atrophy: Focus on glial cytoplasmic inclusions. Transl. Neurodegener. 2020, 9, 7. [Google Scholar] [CrossRef]
- Mészáros, L.; Hoffmann, A.; Wihan, J.; Winkler, J. Current Symptomatic and Disease-Modifying Treatments in Multiple System Atrophy. Int. J. Mol. Sci. 2020, 21, 2775. [Google Scholar] [CrossRef]
- Marmion, D.J.; Peelaerts, W.; Kordower, J.H. A historical review of multiple system atrophy with a critical appraisal of cellular and animal models. J. Neural Transm. 2021, 128, 1507–1527. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, H.; Saito, Y.; Terao, S.; Ando, T.; Kachi, T.; Mukai, E.; Aiba, I.; Abe, Y.; Tamakoshi, A.; Doyu, M.; et al. Progression and prognosis in multiple system atrophy: An analysis of 230 Japanese patients. Brain A J. Neurol. 2002, 125, 1070–1083. [Google Scholar] [CrossRef]
- Multiple-System Atrophy Research Collaboration. Mutations in COQ2 in Familial and Sporadic Multiple-System Atrophy. N. Engl. J. Med. 2013, 369, 233–244. [Google Scholar] [CrossRef]
- Ogaki, K.; Fujioka, S.; Heckman, M.G.; Rayaprolu, S.; Soto-Ortolaza, A.I.; Labbé, C.; Walton, R.L.; Lorenzo-Betancor, O.; Wang, X.; Asmann, Y.; et al. Analysis of COQ2 gene in multiple system atrophy. Mol. Neurodegener. 2014, 9, 44. [Google Scholar] [CrossRef]
- Quinzii, C.M.; Hirano, M.; DiMauro, S. Mutant COQ2 in multiple-system atrophy. N. Engl. J. Med. 2014, 371, 81–82. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.H.; Tan, E.K.; Yang, C.C.; Yi, Z.; Wu, R.M. COQ2 gene variants associate with cerebellar subtype of multiple system atrophy in Chinese. Mov. Disord. Off. J. Mov. Disord. Soc. 2015, 30, 436–437. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Yang, X.; Tian, S.; An, R.; Zheng, J.; Xu, Y. Association of the COQ2 V393A variant with risk of multiple system atrophy in East Asians: A case-control study and meta-analysis of the literature. Neurol. Sci. Off. J. Ital. Neurol. Soc. Ital. Soc. Clin. Neurophysiol. 2016, 37, 423–430. [Google Scholar] [CrossRef] [PubMed]
- Pasanen, P.; Myllykangas, L.; Siitonen, M.; Raunio, A.; Kaakkola, S.; Lyytinen, J.; Tienari, P.J.; Pöyhönen, M.; Paetau, A. Novel α-synuclein mutation A53E associated with atypical multiple system atrophy and Parkinson’s disease-type pathology. Neurobiol. Aging 2014, 35, e2181–e2185. [Google Scholar] [CrossRef]
- Danka Mohammed, C.P.; Park, J.S.; Nam, H.G.; Kim, K. MicroRNAs in brain aging. Mech. Ageing Dev. 2017, 168, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, C.; Aoyama, K.; Nakaki, T. microRNA as a new agent for regulating neuronal glutathione synthesis and metabolism. AIMS Mol. Sci. 2015, 2, 124–143. [Google Scholar] [CrossRef]
- Kinoshita, C.; Kubota, N.; Aoyama, K. Interplay of RNA-Binding Proteins and microRNAs in Neurodegenerative Diseases. Int. J. Mol. Sci. 2021, 22, 5292. [Google Scholar] [CrossRef]
- Ubhi, K.; Rockenstein, E.; Kragh, C.; Inglis, C.; Spencer, B.; Michael, S.; Mante, M.; Adame, A.; Galasko, D.; Masliah, E. Widespread microRNA dysregulation in multiple system atrophy—Disease-related alteration in miR-96. Eur. J. Neurosci. 2014, 39, 1026–1041. [Google Scholar] [CrossRef]
- Singh, A.; Kukreti, R.; Saso, L.; Kukreti, S. Oxidative Stress: A Key Modulator in Neurodegenerative Diseases. Molecules 2019, 24, 1583. [Google Scholar] [CrossRef]
- Shukla, D.; Mandal, P.K.; Mishra, R.; Punjabi, K.; Dwivedi, D.; Tripathi, M.; Badhautia, V. Hippocampal Glutathione Depletion and pH Increment in Alzheimer’s Disease: An in vivo MRS Study. J. Alzheimer’s Dis. JAD 2021, 84, 1139–1152. [Google Scholar] [CrossRef]
- Mandal, P.K.; Dwivedi, D.; Shukla, D.; Samkaria, A.; Roy, R.G.; Arora, Y.; Jindal, K. Interplay Between Hippocampal Glutathione Depletion and pH Increment in Alzheimer’s Disease. J. Alzheimer’s Dis. JAD 2022, 88, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Chiang, G.C.; Mao, X.; Kang, G.; Chang, E.; Pandya, S.; Vallabhajosula, S.; Isaacson, R.; Ravdin, L.D.; Shungu, D.C. Relationships among Cortical Glutathione Levels, Brain Amyloidosis, and Memory in Healthy Older Adults Investigated In Vivo with (1)H-MRS and Pittsburgh Compound-B PET. AJNR Am. J. Neuroradiol. 2017, 38, 1130–1137. [Google Scholar] [CrossRef] [PubMed]
- Jenner, P. Presymptomatic detection of Parkinson’s disease. Journal of neural transmission. Supplementum 1993, 40, 23–36. [Google Scholar]
- Pearce, R.K.B.; Owen, A.; Daniel, S.; Jenner, P.; Marsden, C.D. Alterations in the distribution of glutathione in the substantia nigra in Parkinson’s disease. J. Neural Transm. 1997, 104, 661–677. [Google Scholar] [CrossRef]
- Andronesi, O.C.; Nicholson, K.; Jafari-Khouzani, K.; Bogner, W.; Wang, J.; Chan, J.; Macklin, E.A.; Levine-Weinberg, M.; Breen, C.; Schwarzschild, M.A.; et al. Imaging Neurochemistry and Brain Structure Tracks Clinical Decline and Mechanisms of ALS in Patients. Front. Neurol. 2020, 11, 590573. [Google Scholar] [CrossRef]
- Chen, J.J.; Thiyagarajah, M.; Song, J.; Chen, C.; Herrmann, N.; Gallagher, D.; Rapoport, M.J.; Black, S.E.; Ramirez, J.; Andreazza, A.C.; et al. Altered central and blood glutathione in Alzheimer’s disease and mild cognitive impairment: A meta-analysis. Alzheimer’s Res. Ther. 2022, 14, 23. [Google Scholar] [CrossRef]
- Sofic, E.; Lange, K.W.; Jellinger, K.; Riederer, P. Reduced and oxidized glutathione in the substantia nigra of patients with Parkinson’s disease. Neurosci. Lett. 1992, 142, 128–130. [Google Scholar] [CrossRef]
- Fanciulli, A.; Stankovic, I.; Krismer, F.; Seppi, K.; Levin, J.; Wenning, G.K. Multiple system atrophy. Int. Rev. Neurobiol. 2019, 149, 137–192. [Google Scholar] [CrossRef]
- Graham, J.G.; Oppenheimer, D.R. Orthostatic hypotension and nicotine sensitivity in a case of multiple system atrophy. J. Neurol. Neurosurg. Psychiatry 1969, 32, 28–34. [Google Scholar] [CrossRef]
- Yoshida, M. Multiple system atrophy: Alpha-synuclein and neuronal degeneration. Neuropathol. Off. J. Jpn. Soc. Neuropathol. 2007, 27, 484–493. [Google Scholar] [CrossRef]
- Gilman, S.; Low, P.A.; Quinn, N.; Albanese, A.; Ben-Shlomo, Y.; Fowler, C.J.; Kaufmann, H.; Klockgether, T.; Lang, A.E.; Lantos, P.L.; et al. Consensus statement on the diagnosis of multiple system atrophy. J. Neurol. Sci. 1999, 163, 94–98. [Google Scholar] [CrossRef]
- Gilman, S.; Wenning, G.K.; Low, P.A.; Brooks, D.J.; Mathias, C.J.; Trojanowski, J.Q.; Wood, N.W.; Colosimo, C.; Dürr, A.; Fowler, C.J.; et al. Second consensus statement on the diagnosis of multiple system atrophy. Neurology 2008, 71, 670–676. [Google Scholar] [CrossRef]
- Woerman, A.L.; Watts, J.C.; Aoyagi, A.; Giles, K.; Middleton, L.T.; Prusiner, S.B. α-Synuclein: Multiple System Atrophy Prions. Cold Spring Harb. Perspect. Med. 2018, 8, a024588. [Google Scholar] [CrossRef]
- Lashuel, H.A.; Overk, C.R.; Oueslati, A.; Masliah, E. The many faces of α-synuclein: From structure and toxicity to therapeutic target. Nature reviews. Neuroscience 2013, 14, 38–48. [Google Scholar] [CrossRef]
- Pukaß, K.; Richter-Landsberg, C. Oxidative Stress Promotes Uptake, Accumulation, and Oligomerization of Extracellular α-Synuclein in Oligodendrocytes. J. Mol. Neurosci. 2014, 52, 339–352. [Google Scholar] [CrossRef]
- Jaganjac, M.; Milkovic, L.; Zarkovic, N.; Zarkovic, K. Oxidative stress and regeneration. Free. Radic. Biol. Med. 2022, 181, 154–165. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.; Mercado-Ayon, E.; Mercado-Ayon, Y.; Dong, Y.N.; Halawani, S.; Ngaba, L.; Lynch, D.R. Mitochondrial dysfunction in the development and progression of neurodegenerative diseases. Arch. Biochem. Biophys. 2021, 702, 108698. [Google Scholar] [CrossRef]
- Priesnitz, C.; Becker, T. Pathways to balance mitochondrial translation and protein import. Genes Dev. 2018, 32, 1285–1296. [Google Scholar] [CrossRef] [PubMed]
- Monzio Compagnoni, G.; Di Fonzo, A. Understanding the pathogenesis of multiple system atrophy: State of the art and future perspectives. Acta Neuropathol. Commun. 2019, 7, 113. [Google Scholar] [CrossRef] [PubMed]
- Stankovic, I.; Fanciulli, A.; Sidoroff, V.; Wenning, G.K. A Review on the Clinical Diagnosis of Multiple System Atrophy. Cerebellum 2022, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Koga, S.; Aoki, N.; Uitti, R.J.; van Gerpen, J.A.; Cheshire, W.P.; Josephs, K.A.; Wszolek, Z.K.; Langston, J.W.; Dickson, D.W. When DLB, PD, and PSP masquerade as MSA. Neurology 2015, 85, 404. [Google Scholar] [CrossRef] [PubMed]
- Grossauer, A.; Sidoroff, V.; Heim, B.; Seppi, K. Symptomatic Care in Multiple System Atrophy: State of the Art. Cerebellum 2022, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Fahn, S.; Oakes, D.; Shoulson, I.; Kieburtz, K.; Rudolph, A.; Lang, A.; Olanow, C.W.; Tanner, C.; Marek, K.; Parkinson Study Group. Levodopa and the Progression of Parkinson’s Disease. N. Engl. J. Med. 2004, 351, 2498–2508. [Google Scholar] [CrossRef] [PubMed]
- Koga, S.; Sekiya, H.; Kondru, N.; Ross, O.A.; Dickson, D.W. Neuropathology and molecular diagnosis of Synucleinopathies. Mol. Neurodegener. 2021, 16, 83. [Google Scholar] [CrossRef] [PubMed]
- Malfertheiner, K.; Stefanova, N.; Heras-Garvin, A. The Concept of α-Synuclein Strains and How Different Conformations May Explain Distinct Neurodegenerative Disorders. Front. Neurol. 2021, 12, 737195. [Google Scholar] [CrossRef]
- Martinez-Valbuena, I.; Visanji, N.P.; Kim, A.; Lau, H.H.C.; So, R.W.L.; Alshimemeri, S.; Gao, A.; Seidman, M.A.; Luquin, M.R.; Watts, J.C.; et al. Alpha-synuclein seeding shows a wide heterogeneity in multiple system atrophy. Transl. Neurodegener. 2022, 11, 7. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki, T.R.; Holmes, B.B.; Furman, J.L.; Dhavale, D.D.; Su, B.W.; Song, E.S.; Cairns, N.J.; Kotzbauer, P.T.; Diamond, M.I. Parkinson’s disease and multiple system atrophy have distinct α-synuclein seed characteristics. J. Biol. Chem. 2019, 294, 1045–1058. [Google Scholar] [CrossRef] [PubMed]
- Jellinger, K.A. Heterogeneity of Multiple System Atrophy: An Update. Biomedicines 2022, 10, 599. [Google Scholar] [CrossRef] [PubMed]
- Strohäker, T.; Jung, B.C.; Liou, S.-H.; Fernandez, C.O.; Riedel, D.; Becker, S.; Halliday, G.M.; Bennati, M.; Kim, W.S.; Lee, S.-J.; et al. Structural heterogeneity of α-synuclein fibrils amplified from patient brain extracts. Nat. Commun. 2019, 10, 5535. [Google Scholar] [CrossRef]
- Koga, S. The Nomenclature and Classification of Sporadic Spinocerebellar Degeneration. Brain Nerve = Shinkei Kenkyu No Shinpo 2016, 68, 1453–1457. [Google Scholar] [CrossRef]
- Hirayama, K.; Takayanagi, T.; Nakamura, R.; Yanagisawa, N.; Hattori, T.; Kita, K.; Yanagimoto, S.; Fujita, M.; Nagaoka, M.; Satomura, Y.; et al. Spinocerebellar degenerations in Japan: A nationwide epidemiological and clinical study. Acta Neurol. Scand. Suppl. 1994, 153, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Ma, Q.; Zhao, X.; Wang, C.; Wu, H.; Li, J.; Yang, W. Dilemma of multiple system atrophy and spinocerebellar ataxias. J. Neurol. 2018, 265, 2764–2772. [Google Scholar] [CrossRef] [PubMed]
- Kiely, A.P.; Asi, Y.T.; Kara, E.; Limousin, P.; Ling, H.; Lewis, P.; Proukakis, C.; Quinn, N.; Lees, A.J.; Hardy, J.; et al. α-Synucleinopathy associated with G51D SNCA mutation: A link between Parkinson’s disease and multiple system atrophy? Acta Neuropathol. 2013, 125, 753–769. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Ohta, Y.; Yamashita, T.; Sato, K.; Takemoto, M.; Hishikawa, N.; Abe, K. New susceptible variant of COQ2 gene in Japanese patients with sporadic multiple system atrophy. Neurology. Genetics 2016, 2, e54. [Google Scholar] [CrossRef]
- Quinzii, C.; Naini, A.; Salviati, L.; Trevisson, E.; Navas, P.; Dimauro, S.; Hirano, M. A mutation in para-hydroxybenzoate-polyprenyl transferase (COQ2) causes primary coenzyme Q10 deficiency. Am. J. Hum. Genet. 2006, 78, 345–349. [Google Scholar] [CrossRef]
- Rauchová, H. Coenzyme Q10 effects in neurological diseases. Physiol. Res. 2021, 70, S683–S714. [Google Scholar] [CrossRef]
- Barca, E.; Kleiner, G.; Tang, G.; Ziosi, M.; Tadesse, S.; Masliah, E.; Louis, E.D.; Faust, P.; Kang, U.J.; Torres, J.; et al. Decreased Coenzyme Q10 Levels in Multiple System Atrophy Cerebellum. J. Neuropathol. Exp. Neurol. 2016, 75, 663–672. [Google Scholar] [CrossRef]
- Hsiao, J.T.; Purushothuman, S.; Jensen, P.H.; Halliday, G.M.; Kim, W.S. Reductions in COQ2 Expression Relate to Reduced ATP Levels in Multiple System Atrophy Brain. Front. Neurosci. 2019, 13, 1187. [Google Scholar] [CrossRef]
- Chen, Y.P.; Zhao, B.; Cao, B.; Song, W.; Guo, X.; Wei, Q.Q.; Yang, Y.; Yuan, L.X.; Shang, H.F. Mutation scanning of the COQ2 gene in ethnic Chinese patients with multiple-system atrophy. Neurobiol. Aging 2015, 36, 1222.e7–1222.e11. [Google Scholar] [CrossRef]
- Procopio, R.; Gagliardi, M.; Brighina, L.; Nicoletti, G.; Morelli, M.; Ferrarese, C.; Annesi, G.; Quattrone, A. Genetic mutation analysis of the COQ2 gene in Italian patients with multiple system atrophy. Gene 2019, 716, 144037. [Google Scholar] [CrossRef]
- Mikasa, M.; Kanai, K.; Li, Y.; Yoshino, H.; Mogushi, K.; Hayashida, A.; Ikeda, A.; Kawajiri, S.; Okuma, Y.; Kashihara, K.; et al. COQ2 variants in Parkinson’s disease and multiple system atrophy. J. Neural Transm. 2018, 125, 937–944. [Google Scholar] [CrossRef] [PubMed]
- Schottlaender, L.V.; Holton, J.L.; Houlden, H. Multiple system atrophy and repeat expansions in C9orf72. JAMA Neurol. 2014, 71, 1190–1191. [Google Scholar] [CrossRef] [PubMed]
- Riboldi, G.M.; Palma, J.A.; Cortes, E.; Iida, M.A.; Sikder, T.; Henderson, B.; Raj, T.; Walker, R.H.; Crary, J.F.; Kaufmann, H.; et al. Early-onset pathologically proven multiple system atrophy with LRRK2 G2019S mutation. Mov. Disord. Off. J. Mov. Disord. Soc. 2019, 34, 1080–1082. [Google Scholar] [CrossRef] [PubMed]
- Wernick, A.I.; Walton, R.L.; Koga, S.; Soto-Beasley, A.I.; Heckman, M.G.; Gan-Or, Z.; Ren, Y.; Rademakers, R.; Uitti, R.J.; Wszolek, Z.K.; et al. GBA variation and susceptibility to multiple system atrophy. Park. Relat. Disord. 2020, 77, 64–69. [Google Scholar] [CrossRef]
- Nicoletti, G.; Gagliardi, M.; Procopio, R.; Iannello, G.; Morelli, M.; Annesi, G.; Quattrone, A. A new CHCHD2 mutation identified in a southern italy patient with multiple system atrophy. Park. Relat. Disord. 2018, 47, 91–93. [Google Scholar] [CrossRef]
- Wernick, A.I.; Walton, R.L.; Soto-Beasley, A.I.; Koga, S.; Heckman, M.G.; Valentino, R.R.; Milanowski, L.M.; Hoffman-Zacharska, D.; Koziorowski, D.; Hassan, A.; et al. Frequency of spinocerebellar ataxia mutations in patients with multiple system atrophy. Clin. Auton. Res. Off. J. Clin. Auton. Res. Soc. 2021, 31, 117–125. [Google Scholar] [CrossRef]
- Vilariño-Güell, C.; Soto-Ortolaza, A.I.; Rajput, A.; Mash, D.C.; Papapetropoulos, S.; Pahwa, R.; Lyons, K.E.; Uitti, R.J.; Wszolek, Z.K.; Dickson, D.W.; et al. MAPT H1 haplotype is a risk factor for essential tremor and multiple system atrophy. Neurology 2011, 76, 670–672. [Google Scholar] [CrossRef]
- Piñero, J.; Saüch, J.; Sanz, F.; Furlong, L.I. The DisGeNET cytoscape app: Exploring and visualizing disease genomics data. Comput. Struct. Biotechnol. J. 2021, 19, 2960–2967. [Google Scholar] [CrossRef]
- Porto, K.J.; Hirano, M.; Mitsui, J.; Chikada, A.; Matsukawa, T.; Ishiura, H.; Toda, T.; Kusunoki, S.; Tsuji, S. COQ2 V393A confers high risk susceptibility for multiple system atrophy in East Asian population. J. Neurol. Sci. 2021, 429, 117623. [Google Scholar] [CrossRef]
- Rink, C.; Khanna, S. Significance of brain tissue oxygenation and the arachidonic acid cascade in stroke. Antioxid. Redox Signal. 2011, 14, 1889–1903. [Google Scholar] [CrossRef]
- Bazinet, R.P.; Layé, S. Polyunsaturated fatty acids and their metabolites in brain function and disease. Nat. Rev. Neurosci. 2014, 15, 771–785. [Google Scholar] [CrossRef]
- Aoyama, K. Glutathione in the Brain. Int. J. Mol. Sci. 2021, 22, 5010. [Google Scholar] [CrossRef]
- Shukla, G.S.; Hussain, T.; Srivastava, R.S.; Chandra, S.V. Glutathione peroxidase and catalase in liver, kidney, testis and brain regions of rats following cadmium exposure and subsequent withdrawal. Ind. Health 1989, 27, 59–69. [Google Scholar] [CrossRef]
- Szymonik-Lesiuk, S.; Czechowska, G.; Stryjecka-Zimmer, M.; SŁomka, M.; MĄldro, A.; CeliŃski, K.; Wielosz, M. Catalase, superoxide dismutase, and glutathione peroxidase activities in various rat tissues after carbon tetrachloride intoxication. J. Hepato-Biliary-Pancreat. Surg. 2003, 10, 309–315. [Google Scholar] [CrossRef]
- Dringen, R.; Hirrlinger, J. Glutathione Pathways in the Brain. Biol. Chem. 2003, 384, 505–516. [Google Scholar] [CrossRef] [PubMed]
- Couto, N.; Wood, J.; Barber, J. The role of glutathione reductase and related enzymes on cellular redox homoeostasis network. Free. Radic. Biol. Med. 2016, 95, 27–42. [Google Scholar] [CrossRef] [PubMed]
- Fitzmaurice, P.S.; Ang, L.; Guttman, M.; Rajput, A.H.; Furukawa, Y.; Kish, S.J. Nigral glutathione deficiency is not specific for idiopathic Parkinson’s disease. Mov. Disord. 2003, 18, 969–976. [Google Scholar] [CrossRef] [PubMed]
- Jenner, P.; Dexter, D.T.; Sian, J.; Schapira, A.H.V.; Marsden, C.D. Oxidative stress as a cause of nigral cell death in Parkinson’s disease and incidental lewy body disease. Ann. Neurol. 1992, 32, S82–S87. [Google Scholar] [CrossRef]
- Sian, J.; Dexter, D.T.; Lees, A.J.; Daniel, S.; Agid, Y.; Javoy-Agid, F.; Jenner, P.; Marsden, C.D. Alterations in glutathione levels in Parkinson’s disease and other neurodegenerative disorders affecting basal ganglia. Ann. Neurol. 1994, 36, 348–355. [Google Scholar] [CrossRef]
- Paik, S.R.; Lee, D.; Cho, H.-J.; Lee, E.-N.; Chang, C.-S. Oxidized glutathione stimulated the amyloid formation of α-synuclein. FEBS Lett. 2003, 537, 63–67. [Google Scholar] [CrossRef]
- Xu, B.; Wu, S.-W.; Lu, C.-W.; Deng, Y.; Liu, W.; Wei, Y.-G.; Yang, T.-Y.; Xu, Z.-F. Oxidative stress involvement in manganese-induced alpha-synuclein oligomerization in organotypic brain slice cultures. Toxicology 2013, 305, 71–78. [Google Scholar] [CrossRef]
- Tanaka, K.-I.; Sonoda, K.; Asanuma, M. Effect of Alteration of Glutathione Content on Cell Viability in α-Synuclein-Transfected SH-SY5Y Cells. Adv. Park. Dis. 2017, 6, 93. [Google Scholar] [CrossRef][Green Version]
- Clark, J.; Clore, E.L.; Zheng, K.; Adame, A.; Masliah, E.; Simon, D.K. Oral N-acetyl-cysteine attenuates loss of dopaminergic terminals in alpha-synuclein overexpressing mice. PLoS ONE 2010, 5, e12333. [Google Scholar] [CrossRef]
- Flohé, L. Glutathione peroxidase. Basic Life Sci. 1988, 49, 663–668. [Google Scholar] [CrossRef]
- Kish, S.J.; Morito, C.L.; Hornykiewicz, O. Brain glutathione peroxidase in neurodegenerative disorders. Neurochem. Pathol. 1986, 4, 23–28. [Google Scholar] [CrossRef]
- Koo, H.J.; Yang, J.E.; Park, J.H.; Lee, D.; Paik, S.R. α-Synuclein-mediated defense against oxidative stress via modulation of glutathione peroxidase. Biochim. Et Biophys. Acta 2013, 1834, 972–976. [Google Scholar] [CrossRef]
- Hayes, J.D.; Flanagan, J.U.; Jowsey, I.R. Glutathione transferases. Annu. Rev. Pharmacol. Toxicol. 2005, 45, 51–88. [Google Scholar] [CrossRef]
- Probst-Cousin, S.; Bergmann, M.; Kuchelmeister, K.; Schröder, R.; Schmid, K.W. Ubiquitin-positive inclusions in different types of multiple system atrophy: Distribution and specificity. Pathol. Res. Pract. 1996, 192, 453–461. [Google Scholar] [CrossRef] [PubMed]
- Aoyama, K.; Nakaki, T. Impaired glutathione synthesis in neurodegeneration. Int. J. Mol. Sci. 2013, 14, 21021–21044. [Google Scholar] [CrossRef] [PubMed]
- Janáky, R.; Varga, V.; Hermann, A.; Saransaari, P.; Oja, S.S. Mechanisms of L-cysteine neurotoxicity. Neurochem. Res. 2000, 25, 1397–1405. [Google Scholar] [CrossRef] [PubMed]
- Paul, B.D.; Sbodio, J.I.; Snyder, S.H. Cysteine Metabolism in Neuronal Redox Homeostasis. Trends Pharmacol. Sci. 2018, 39, 513–524. [Google Scholar] [CrossRef]
- Hughes, C.E.; Coody, T.K.; Jeong, M.Y.; Berg, J.A.; Winge, D.R.; Hughes, A.L. Cysteine Toxicity Drives Age-Related Mitochondrial Decline by Altering Iron Homeostasis. Cell 2020, 180, 296–310.e218. [Google Scholar] [CrossRef]
- Richman, P.G.; Meister, A. Regulation of gamma-glutamyl-cysteine synthetase by nonallosteric feedback inhibition by glutathione. J. Biol. Chem. 1975, 250, 1422–1426. [Google Scholar] [CrossRef]
- Lu, S.C. Regulation of glutathione synthesis. Mol. Asp. Med. 2009, 30, 42–59. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, G.; Bridge, W. Glutamate cysteine ligase and the age-related decline in cellular glutathione: The therapeutic potential of γ-glutamylcysteine. Arch. Biochem. Biophys. 2016, 593, 12–23. [Google Scholar] [CrossRef]
- Njålsson, R. Glutathione synthetase deficiency. Cell. Mol. Life Sci. CMLS 2005, 62, 1938–1945. [Google Scholar] [CrossRef] [PubMed]
- Aoyama, K.; Nakaki, T. Glutathione in Cellular Redox Homeostasis: Association with the Excitatory Amino Acid Carrier 1 (EAAC1). Molecules 2015, 20, 8742–8758. [Google Scholar] [CrossRef] [PubMed]
- Divito, C.B.; Underhill, S.M. Excitatory amino acid transporters: Roles in glutamatergic neurotransmission. Neurochem. Int. 2014, 73, 172–180. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bianchi, M.G.; Bardelli, D.; Chiu, M.; Bussolati, O. Changes in the expression of the glutamate transporter EAAT3/EAAC1 in health and disease. Cell. Mol. Life Sci. 2014, 71, 2001–2015. [Google Scholar] [CrossRef] [PubMed]
- Aoyama, K.; Suh, S.W.; Hamby, A.M.; Liu, J.; Chan, W.Y.; Chen, Y.; Swanson, R.A. Neuronal glutathione deficiency and age-dependent neurodegeneration in the EAAC1 deficient mouse. Nat. Neurosci. 2006, 9, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Kanai, Y.; Hediger, M.A. The glutamate and neutral amino acid transporter family: Physiological and pharmacological implications. Eur. J. Pharmacol. 2003, 479, 237–247. [Google Scholar] [CrossRef]
- Watts, S.D.; Torres-Salazar, D.; Divito, C.B.; Amara, S.G. Cysteine Transport through Excitatory Amino Acid Transporter 3 (EAAT3). PLoS ONE 2014, 9, e109245. [Google Scholar] [CrossRef] [PubMed]
- Sedláčková, L.; Laššuthová, P.; Štěrbová, K.; Vlčková, M.; Kudr, M.; Buksakowska, I.; Staněk, D.; Seeman, P. Severe neurodevelopmental disorder with intractable seizures due to a novel SLC1A4 homozygous variant. Eur. J. Med. Genet. 2021, 64, 104263. [Google Scholar] [CrossRef]
- Abdelrahman, H.A.; Al-Shamsi, A.; John, A.; Ali, B.R.; Al-Gazali, L. A Novel SLC1A4 Mutation (p.Y191*) Causes Spastic Tetraplegia, Thin Corpus Callosum, and Progressive Microcephaly (SPATCCM) With Seizure Disorder. Child Neurol. Open 2019, 6, 2329048x19880647. [Google Scholar] [CrossRef] [PubMed]
- Pironti, E.; Salpietro, V.; Cucinotta, F.; Granata, F.; Mormina, E.; Efthymiou, S.; Scuderi, C.; Gagliano, A.; Houlden, H.; Di Rosa, G. A novel SLC1A4 homozygous mutation causing congenital microcephaly, epileptic encephalopathy and spastic tetraparesis: A video-EEG and tractography—Case study. J. Neurogenet. 2018, 32, 316–321. [Google Scholar] [CrossRef]
- Heimer, G.; Marek-Yagel, D.; Eyal, E.; Barel, O.; Oz Levi, D.; Hoffmann, C.; Ruzzo, E.K.; Ganelin-Cohen, E.; Lancet, D.; Pras, E.; et al. SLC1A4 mutations cause a novel disorder of intellectual disability, progressive microcephaly, spasticity and thin corpus callosum. Clin. Genet. 2015, 88, 327–335. [Google Scholar] [CrossRef]
- Deng, X.; Sagata, N.; Takeuchi, N.; Tanaka, M.; Ninomiya, H.; Iwata, N.; Ozaki, N.; Shibata, H.; Fukumaki, Y. Association study of polymorphisms in the neutral amino acid transporter genes SLC1A4, SLC1A5 and the glycine transporter genes SLC6A5, SLC6A9 with schizophrenia. BMC Psychiatry 2008, 8, 58. [Google Scholar] [CrossRef] [PubMed]
- Damseh, N.; Simonin, A.; Jalas, C.; Picoraro, J.A.; Shaag, A.; Cho, M.T.; Yaacov, B.; Neidich, J.; Al-Ashhab, M.; Juusola, J.; et al. Mutations in SLC1A4, encoding the brain serine transporter, are associated with developmental delay, microcephaly and hypomyelination. J. Med. Genet. 2015, 52, 541–547. [Google Scholar] [CrossRef] [PubMed]
- Soma, H.; Yabe, I.; Takei, A.; Fujiki, N.; Yanagihara, T.; Sasaki, H. Associations between multiple system atrophy and polymorphisms of SLC1A4, SQSTM1, and EIF4EBP1 genes. Mov. Disord. Off. J. Mov. Disord. Soc. 2008, 23, 1161–1167. [Google Scholar] [CrossRef]
- Kinoshita, C.; Aoyama, K. The Role of Non-Coding RNAs in the Neuroprotective Effects of Glutathione. Int. J. Mol. Sci. 2021, 22, 4245. [Google Scholar] [CrossRef]
- Pérez-Soriano, A.; Bravo, P.; Soto, M.; Infante, J.; Fernández, M.; Valldeoriola, F.; Muñoz, E.; Compta, Y.; Tolosa, E.; Garrido, A.; et al. MicroRNA Deregulation in Blood Serum Identifies Multiple System Atrophy Altered Pathways. Mov. Disord. Off. J. Mov. Disord. Soc. 2020, 35, 1873–1879. [Google Scholar] [CrossRef] [PubMed]
- Kume, K.; Iwama, H.; Deguchi, K.; Ikeda, K.; Takata, T.; Kokudo, Y.; Kamada, M.; Fujikawa, K.; Hirose, K.; Masugata, H.; et al. Serum microRNA expression profiling in patients with multiple system atrophy. Mol. Med. Rep. 2018, 17, 852–860. [Google Scholar] [CrossRef]
- Uwatoko, H.; Hama, Y.; Iwata, I.T.; Shirai, S.; Matsushima, M.; Yabe, I.; Utsumi, J.; Sasaki, H. Identification of plasma microRNA expression changes in multiple system atrophy and Parkinson’s disease. Mol. Brain 2019, 12, 49. [Google Scholar] [CrossRef]
- Kim, T.; Valera, E.; Desplats, P. Alterations in Striatal microRNA-mRNA Networks Contribute to Neuroinflammation in Multiple System Atrophy. Mol. Neurobiol. 2019, 56, 7003–7021. [Google Scholar] [CrossRef]
- Wakabayashi, K.; Mori, F.; Kakita, A.; Takahashi, H.; Tanaka, S.; Utsumi, J.; Sasaki, H. MicroRNA expression profiles of multiple system atrophy from formalin-fixed paraffin-embedded samples. Neurosci. Lett. 2016, 635, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-T.; Chu, K.; Jung, K.-H.; Ban, J.-J.; Im, W.-S.; Jo, H.-Y.; Park, J.-H.; Lim, J.-Y.; Shin, J.-W.; Moon, J.; et al. Altered Expression of miR-202 in Cerebellum of Multiple-System Atrophy. Mol. Neurobiol. 2015, 51, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Vallelunga, A.; Ragusa, M.; Di Mauro, S.; Iannitti, T.; Pilleri, M.; Biundo, R.; Weis, L.; Di Pietro, C.; De Iuliis, A.; Nicoletti, A.; et al. Identification of circulating microRNAs for the differential diagnosis of Parkinson’s disease and Multiple System Atrophy. Front. Cell. Neurosci. 2014, 8, 156. [Google Scholar] [CrossRef]
- Starhof, C.; Hejl, A.M.; Heegaard, N.H.H.; Carlsen, A.L.; Burton, M.; Lilje, B.; Winge, K. The biomarker potential of cell-free microRNA from cerebrospinal fluid in Parkinsonian Syndromes. Mov. Disord. Off. J. Mov. Disord. Soc. 2019, 34, 246–254. [Google Scholar] [CrossRef]
- Seok, H.; Ham, J.; Jang, E.S.; Chi, S.W. MicroRNA Target Recognition: Insights from Transcriptome-Wide Non-Canonical Interactions. Mol. Cells 2016, 39, 375–381. [Google Scholar] [CrossRef]
- Krol, J.; Loedige, I.; Filipowicz, W. The widespread regulation of microRNA biogenesis, function and decay. Nat. Rev. Genet. 2010, 11, 597–610. [Google Scholar] [CrossRef]
- Alarcón, C.R.; Lee, H.; Goodarzi, H.; Halberg, N.; Tavazoie, S.F. N6-methyladenosine marks primary microRNAs for processing. Nature 2015, 519, 482–485. [Google Scholar] [CrossRef] [PubMed]
- Michlewski, G.; Cáceres, J.F. Post-transcriptional control of miRNA biogenesis. RNA 2019, 25, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Iwakawa, H.O.; Tomari, Y. Life of RISC: Formation, action, and degradation of RNA-induced silencing complex. Mol. Cell 2022, 82, 30–43. [Google Scholar] [CrossRef]
- Stavast, C.J.; Erkeland, S.J. The Non-Canonical Aspects of MicroRNAs: Many Roads to Gene Regulation. Cells 2019, 8, 1465. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, C.; Aoyama, K.; Matsumura, N.; Kikuchi-Utsumi, K.; Watabe, M.; Nakaki, T. Rhythmic oscillations of the microRNA miR-96-5p play a neuroprotective role by indirectly regulating glutathione levels. Nat. Commun. 2014, 5, 3823. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, C.; Kikuchi-Utsumi, K.; Aoyama, K.; Suzuki, R.; Okamoto, Y.; Matsumura, N.; Omata, D.; Maruyama, K.; Nakaki, T. Inhibition of miR-96-5p in the mouse brain increases glutathione levels by altering NOVA1 expression. Commun. Biol. 2021, 4, 182. [Google Scholar] [CrossRef] [PubMed]
- Vallelunga, A.; Iannitti, T.; Capece, S.; Somma, G.; Russillo, M.C.; Foubert-Samier, A.; Laurens, B.; Sibon, I.; Meissner, W.G.; Barone, P.; et al. Serum miR-96-5P and miR-339-5P Are Potential Biomarkers for Multiple System Atrophy and Parkinson’s Disease. Front. Aging Neurosci. 2021, 13, 632891. [Google Scholar] [CrossRef] [PubMed]
- Valera, E.; Spencer, B.; Mott, J.; Trejo, M.; Adame, A.; Mante, M.; Rockenstein, E.; Troncoso, J.C.; Beach, T.G.; Masliah, E.; et al. MicroRNA-101 Modulates Autophagy and Oligodendroglial Alpha-Synuclein Accumulation in Multiple System Atrophy. Front. Mol. Neurosci. 2017, 10, 329. [Google Scholar] [CrossRef] [PubMed]
- Vidal-Martinez, G.; Segura-Ulate, I.; Yang, B.; Diaz-Pacheco, V.; Barragan, J.A.; De-Leon Esquivel, J.; Chaparro, S.A.; Vargas-Medrano, J.; Perez, R.G. FTY720-Mitoxy reduces synucleinopathy and neuroinflammation, restores behavior and mitochondria function, and increases GDNF expression in Multiple System Atrophy mouse models. Exp. Neurol. 2020, 325, 113120. [Google Scholar] [CrossRef]
- Koepsell, H. General Overview of Organic Cation Transporters in Brain. In Organic Cation Transporters in the Central Nervous System; Daws, L.C., Ed.; Springer International Publishing: Cham, Switzerland, 2021; pp. 1–39. [Google Scholar]
- Baumgart, B.R.; Gray, K.L.; Woicke, J.; Bunch, R.T.; Sanderson, T.P.; Van Vleet, T.R. MicroRNA as biomarkers of mitochondrial toxicity. Toxicol. Appl. Pharmacol. 2016, 312, 26–33. [Google Scholar] [CrossRef]
- Chang, L.; Xia, J. MicroRNA Regulatory Network Analysis Using miRNet 2.0. In Transcription Factor Regulatory Networks; Song, Q., Tao, Z., Eds.; Springer US: New York, NY, USA, 2022; pp. 185–204. [Google Scholar]
- Huang, Z.; Shi, J.; Gao, Y.; Cui, C.; Zhang, S.; Li, J.; Zhou, Y.; Cui, Q. HMDD v3.0: A database for experimentally supported human microRNA-disease associations. Nucleic Acids Res. 2019, 47, D1013–D1017. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Liu, L.; Lu, Y.; Zhang, T.; Zhao, W. Serum aberrant expression of miR-24-3p and its diagnostic value in Alzheimer’s disease. Biomark. Med. 2021, 15, 1499–1507. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wang, X. miRDB: An online database for prediction of functional microRNA targets. Nucleic acids research 2019, 48, D127–D131. [Google Scholar] [CrossRef] [PubMed]
- Yousefi, M.; Peymani, M.; Ghaedi, K.; Irani, S.; Etemadifar, M. Significant modulations of linc001128 and linc0938 with miR-24-3p and miR-30c-5p in Parkinson disease. Sci. Rep. 2022, 12, 2569. [Google Scholar] [CrossRef]
- Di, G.; Yang, X.; Cheng, F.; Liu, H.; Xu, M. CEBPA-AS1 Knockdown Alleviates Oxygen-Glucose Deprivation/Reperfusion-Induced Neuron Cell Damage by the MicroRNA 24-3p/BOK Axis. Mol. Cell. Biol. 2021, 41, e0006521. [Google Scholar] [CrossRef]
- Matoušková, P.; Hanousková, B.; Skálová, L. MicroRNAs as Potential Regulators of Glutathione Peroxidases Expression and Their Role in Obesity and Related Pathologies. Int. J. Mol. Sci. 2018, 19, 1199. [Google Scholar] [CrossRef]
- Chis, A.R.; Moatar, A.I.; Dijmarescu, C.; Rosca, C.; Vorovenci, R.J.; Krabbendam, I.; Dolga, A.; Bejinar, C.; Marian, C.; Sirbu, I.O.; et al. Plasma hsa-mir-19b is a potential LevoDopa therapy marker. J. Cell. Mol. Med. 2021, 25, 8715–8724. [Google Scholar] [CrossRef]
- Cao, X.Y.; Lu, J.M.; Zhao, Z.Q.; Li, M.C.; Lu, T.; An, X.S.; Xue, L.J. MicroRNA biomarkers of Parkinson’s disease in serum exosome-like microvesicles. Neurosci. Lett. 2017, 644, 94–99. [Google Scholar] [CrossRef]
- Soto, M.; Iranzo, A.; Lahoz, S.; Fernández, M.; Serradell, M.; Gaig, C.; Melón, P.; Martí, M.J.; Santamaría, J.; Camps, J.; et al. Serum MicroRNAs Predict Isolated Rapid Eye Movement Sleep Behavior Disorder and Lewy Body Diseases. Mov. Disord. Off. J. Mov. Disord. Soc. 2022, 37, 2086–2098. [Google Scholar] [CrossRef]
- Joilin, G.; Gray, E.; Thompson, A.G.; Bobeva, Y.; Talbot, K.; Weishaupt, J.; Ludolph, A.; Malaspina, A.; Leigh, P.N.; Newbury, S.F.; et al. Identification of a potential non-coding RNA biomarker signature for amyotrophic lateral sclerosis. Brain Commun. 2020, 2, fcaa053. [Google Scholar] [CrossRef]
- Arakawa, Y.; Itoh, S.; Fukazawa, Y.; Ishiguchi, H.; Kohmoto, J.; Hironishi, M.; Ito, H.; Kihira, T. Association between oxidative stress and microRNA expression pattern of ALS patients in the high-incidence area of the Kii Peninsula. Brain Res. 2020, 1746, 147035. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Wei, W.; Shen, J.; Lu, L.; Lu, T.; Wang, H.; Xue, X. Alisol A 24-acetate protects oxygen-glucose deprivation-induced brain microvascular endothelial cells against apoptosis through miR-92a-3p inhibition by targeting the B-cell lymphoma-2 gene. Pharm. Biol. 2021, 59, 513–524. [Google Scholar] [CrossRef] [PubMed]
- Eisele, Y.S.; Monteiro, C.; Fearns, C.; Encalada, S.E.; Wiseman, R.L.; Powers, E.T.; Kelly, J.W. Targeting protein aggregation for the treatment of degenerative diseases. Nat. Rev. Drug Discov. 2015, 14, 759–780. [Google Scholar] [CrossRef] [PubMed]
- Kametani, F.; Hasegawa, M. Reconsideration of Amyloid Hypothesis and Tau Hypothesis in Alzheimer’s Disease. Front. Neurosci. 2018, 12, 25. [Google Scholar] [CrossRef] [PubMed]
- Grosso Jasutkar, H.; Oh, S.E.; Mouradian, M.M. Therapeutics in the Pipeline Targeting α-Synuclein for Parkinson’s Disease. Pharmacol. Rev. 2022, 74, 207–237. [Google Scholar] [CrossRef]
- Ballatori, N.; Krance, S.M.; Notenboom, S.; Shi, S.; Tieu, K.; Hammond, C.L. Glutathione dysregulation and the etiology and progression of human diseases. Biol. Chem. 2009, 390, 191–214. [Google Scholar] [CrossRef]
- Bottino, F.; Lucignani, M.; Napolitano, A.; Dellepiane, F.; Visconti, E.; Rossi Espagnet, M.C.; Pasquini, L. In Vivo Brain GSH: MRS Methods and Clinical Applications. Antioxidants 2021, 10, 1407. [Google Scholar] [CrossRef]
- Mandal, P.K.; Saharan, S.; Tripathi, M.; Murari, G. Brain Glutathione Levels—A Novel Biomarker for Mild Cognitive Impairment and Alzheimer’s Disease. Biol. Psychiatry 2015, 78, 702–710. [Google Scholar] [CrossRef]
- Duffy, S.L.; Lagopoulos, J.; Hickie, I.B.; Diamond, K.; Graeber, M.B.; Lewis, S.J.; Naismith, S.L. Glutathione relates to neuropsychological functioning in mild cognitive impairment. Alzheimer’s Dement. J. Alzheimer’s Assoc. 2014, 10, 67–75. [Google Scholar] [CrossRef]
- Saharan, S.; Mandal, P.K. The emerging role of glutathione in Alzheimer’s disease. J. Alzheimer’s Dis. JAD 2014, 40, 519–529. [Google Scholar] [CrossRef]
- Weiduschat, N.; Mao, X.; Hupf, J.; Armstrong, N.; Kang, G.; Lange, D.J.; Mitsumoto, H.; Shungu, D.C. Motor cortex glutathione deficit in ALS measured in vivo with the J-editing technique. Neurosci. Lett. 2014, 570, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Cheong, I.; Marjańska, M.; Deelchand, D.K.; Eberly, L.E.; Walk, D.; Öz, G. Ultra-High Field Proton MR Spectroscopy in Early-Stage Amyotrophic Lateral Sclerosis. Neurochem. Res. 2017, 42, 1833–1844. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, C.; Aoyama, K.; Nakaki, T. Neuroprotection afforded by circadian regulation of intracellular glutathione levels: A key role for miRNAs. Free. Radic. Biol. Med. 2018, 119, 17–33. [Google Scholar] [CrossRef] [PubMed]
- Recasens, A.; Perier, C.; Sue, C.M. Role of microRNAs in the Regulation of α-Synuclein Expression: A Systematic Review. Front. Mol. Neurosci. 2016, 9, 128. [Google Scholar] [CrossRef] [PubMed]



| miRNA | Number of Reports | Detection Area | Features | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Serum [112,113,118] | Plasma [114,119] | CSF [119] | Cerebellum [116,117] | Pons [116] | Striatum [115] | Frontal Cortex [18] | Related NDs [133,134] | Regulatory Function [135] | ||
| hsa-miR-24-3p | 6 | ↑↑↑ | ↓ | n.d. | n.d. | n.d. | ↑ | ↑ | AD, PD, HD | regulation of neuronal differentiation |
| hsa-miR-19b-3p | 4 | ↑ | ↓ | ↑ | n.d. | n.d. | n.d. | ↑ | AD, PD | regulation of neural proliferation |
| hsa-miR-25-3p | 4 | ↑↑ | n.d. | n.d. | n.d. | n.d. | ↑ | ↑ | AD, PD | aggravating Aβ -induced neuron injury |
| hsa-miR-92a-3p | 4 | ↑ | n.d. | ↓ | n.d. | n.d. | ↑ | ↑ | AD, ALS | aggravating oxidative stress |
| hsa-let-7b-5p | 3 | ↑↑ | n.d. | n.d. | n.d. | n.d. | ↑ | n.d. | AD, PD, ALS | regulation of neuronal apoptosis |
| hsa-miR-15b-5p | 3 | ↑ | ↓ | n.d. | n.d. | n.d. | n.d. | ↑ | AD, ALS | protection against neuronal damage |
| hsa-miR-16-5p | 3 | ↑ | n.d. | n.d. | n.d. | n.d. | n.d. | ↑ | AD, PD, ALS | associated with Aβ deposition |
| hsa-miR-17-5p | 3 | ↑↑ | n.d. | n.d. | n.d. | n.d. | n.d. | ↑ | AD, PD | protection against neurotoxicity |
| hsa-miR-23a-3p | 3 | ↑ | n.d. | n.d. | n.d. | ↑ | n.d. | ↑ | ALS, HD | associated with neuronal apoptosis |
| hsa-miR-93-5p | 3 | ↑ | n.d. | n.d. | n.d. | n.d. | ↑ | ↑ | AD, ALS | inhibition of microglial activation and inflammatory reaction |
| hsa-miR-99a-5p | 3 | ↑ | n.d. | ↓ | n.d. | n.d. | n.d. | ↑ | AD, PD | localized at post-synaptic densities in forebrain |
| hsa-miR-106a-5p | 3 | ↑↑ | n.d. | ↓ | n.d. | n.d. | n.d. | n.d. | PD | regulation of neurogenesis |
| hsa-mIR-124-3p | 3 | n.d. | n.d. | n.d. | n.d. | ↓ | ↓ | ↑ | AD, ALS | regulation of neuronal development |
| hsa-miR-129-5p | 3 | n.d. | n.d. | n.d. | ↓↓ | ↓ | n.d. | n.d. | AD, PD, ALS | regulation of apoptosis and neuroinflammation |
| hsa-miR-186-5p | 3 | ↑ | n.d. | n.d. | n.d. | n.d. | ↑ | ↑ | AD, ALS | suppression of BACE1, enzyme for Aβ generation |
| hsa-miR-484 | 3 | ↑ | n.d. | n.d. | ↓↓ | n.d. | n.d. | n.d. | no data | regulation of neuronal apoptosis |
| hsa-let-7a-5p | 2 | ↑ | n.d. | n.d. | n.d. | n.d. | n.d. | ↑ | AD, PD, ALS | differentiation of neural stem cells |
| hsa-let-7c-5p | 2 | ↑ | n.d. | n.d. | n.d. | n.d. | n.d. | ↑ | PD | regulation of neural stem cell differentiation |
| hsa-let-7d-5p | 2 | ↑ | n.d. | n.d. | n.d. | n.d. | n.d. | ↑ | AD, PD, ALS | regulation of neural cell fate and neurogenesis |
| hsa-let-7i-5p | 2 | ↑ | n.d. | n.d. | n.d. | n.d. | n.d. | ↑ | AD, PD, ALS | protection against brain damage |
| hsa-miR-20a-5p | 2 | ↑ | n.d. | n.d. | n.d. | n.d. | n.d. | ↑ | AD | protection against neurotoxicity |
| hsa-miR-21-5p | 2 | n.d. | n.d. | n.d. | n.d. | ↑ | n.d. | ↑ | AD, PD | protection against neuronal damage |
| hsa-miR-27a-3p | 2 | ↑ | n.d. | n.d. | n.d. | n.d. | n.d. | ↑ | AD | regulator of tight junction at brain endothelium |
| hsa-miR-30b-5p | 2 | n.d. | ↑ | n.d. | n.d. | ↑ | n.d. | n.d. | AD, PD, ALS | protection against neurotoxicity |
| hsa-miR-30d-5p | 2 | ↑ | n.d. | n.d. | n.d. | n.d. | n.d. | ↑ | AD, PD | regulation of neuronal autophagy and apoptosis |
| hsa-miR-96-5p | 2 | ↑ | n.d. | n.d. | n.d. | n.d. | n.d. | ↑ | PD, ALS | regulation of neuronal glutathione level |
| hsa-miR-100-5p | 2 | n.d. | n.d. | ↓ | n.d. | n.d. | ↑ | n.d. | AD | leading microglial accumulation |
| hsa-miR-103a-3p | 2 | ↑↑ | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | AD, PD, ALS | promotion neural outgrowth |
| hsa-miR-107 | 2 | ↑↑ | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | AD | prevention of Aβ -induced neurotoxicity |
| hsa-miR-127-3p | 2 | n.d. | n.d. | n.d. | ↓ | ↓ | n.d. | n.d. | AD | regulation of neuronal autophagy |
| hsa-miR-129-2-3p | 2 | n.d. | n.d. | n.d. | ↓ | ↓ | n.d. | n.d. | no data | targeting GABAA receptor to protect epilepsy |
| hsa-miR-130a-3p | 2 | ↑ | n.d. | n.d. | n.d. | n.d. | n.d. | ↑ | PD, ALS | promotion of the neuronal differentiation |
| hsa-miR-132-3p | 2 | n.d. | n.d. | n.d. | ↓↓ | n.d. | n.d. | n.d. | AD, PD, ALS, HD | regulation of neuronal differentiation, maturation and functioning |
| hsa-miR-138-5p | 2 | n.d. | n.d. | n.d. | ↓ | ↓ | n.d. | n.d. | AD | control of hippocampal interneuron function |
| hsa-miR-142-5p | 2 | n.d. | n.d. | n.d. | ↑ | n.d. | n.d. | ↑ | AD, PD | improvement of neural differentiation |
| hsa-miR-155-5p | 2 | n.d. | n.d. | n.d. | n.d. | n.d. | ↑ | ↑ | AD, ALS | pro-inflammatory mediator of the central nervous system |
| hsa-miR-181a-5p | 2 | ↑ | n.d. | n.d. | n.d. | n.d. | ↑ | n.d. | AD, PD | promotion of neuronal degeneration |
| hsa-mIR-185-5p | 2 | ↑ | n.d. | n.d. | n.d. | n.d. | ↑ | n.d. | AD | inhibition of neuronal autophagy and apoptosis |
| hsa-miR-191-5p | 2 | ↑ | n.d. | n.d. | n.d. | n.d. | n.d. | ↑ | AD | alleviation of microglial cell injury |
| hsa-miR-219a-2-3p | 2 | n.d. | n.d. | n.d. | ↓ | n.d. | ↑ | n.d. | no data | unknown |
| hsa-miR-339-5p | 2 | ↓↓ | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | AD | negative regulation of BACE1 activity |
| hsa-miR-371b-3p | 2 | n.d. | ↑ | n.d. | n.d. | ↓ | n.d. | n.d. | no data | unknown |
| hsa-miR-380-3p | 2 | ↓ | n.d. | n.d. | ↓ | n.d. | n.d. | n.d. | AD | enhancement of neurotoxicity |
| hsa-miR-425-5p | 2 | ↑ | n.d. | n.d. | n.d. | n.d. | n.d. | ↑ | AD, ALS | promotion of neuronal necroptosis |
| hsa-miR-486-5p | 2 | ↑ | n.d. | n.d. | ↓ | n.d. | n.d. | n.d. | ALS | targeting NeuroD6, scavenger gene of ROS |
| hsa-mIR-539-5p | 2 | n.d. | n.d. | n.d. | ↓ | n.d. | ↓ | n.d. | no data | inhibition of inflammatory response of neuron |
| hsa-miR-1233-3p | 2 | n.d. | n.d. | n.d. | ↓ | ↓ | n.d. | n.d. | no data | unknown |
| hsa-miR-1290 | 2 | n.d. | n.d. | n.d. | ↑ | ↑ | n.d. | n.d. | no data | regulation of neuronal differentiation |
| hsa-miR-3663-5p | 2 | n.d. | n.d. | n.d. | ↓ | ↓ | n.d. | n.d. | no data | unknown |
| hsa-miR-4428 | 2 | n.d. | n.d. | n.d. | ↑ | ↑ | n.d. | n.d. | no data | unknown |
| hsa-miR-4440 | 2 | n.d. | n.d. | n.d. | ↓ | ↓ | n.d. | n.d. | no data | unknown |
| hsa-miR-4726-3p | 2 | n.d. | ↓ | n.d. | n.d. | ↓ | n.d. | n.d. | no data | unknown |
| hsa-miR-4739 | 2 | n.d. | n.d. | n.d. | ↓ | ↓ | n.d. | n.d. | no data | unknown |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kinoshita, C.; Kubota, N.; Aoyama, K. Glutathione Depletion and MicroRNA Dysregulation in Multiple System Atrophy: A Review. Int. J. Mol. Sci. 2022, 23, 15076. https://doi.org/10.3390/ijms232315076
Kinoshita C, Kubota N, Aoyama K. Glutathione Depletion and MicroRNA Dysregulation in Multiple System Atrophy: A Review. International Journal of Molecular Sciences. 2022; 23(23):15076. https://doi.org/10.3390/ijms232315076
Chicago/Turabian StyleKinoshita, Chisato, Noriko Kubota, and Koji Aoyama. 2022. "Glutathione Depletion and MicroRNA Dysregulation in Multiple System Atrophy: A Review" International Journal of Molecular Sciences 23, no. 23: 15076. https://doi.org/10.3390/ijms232315076
APA StyleKinoshita, C., Kubota, N., & Aoyama, K. (2022). Glutathione Depletion and MicroRNA Dysregulation in Multiple System Atrophy: A Review. International Journal of Molecular Sciences, 23(23), 15076. https://doi.org/10.3390/ijms232315076






