Purinergic Signalling in the Cochlea
Abstract
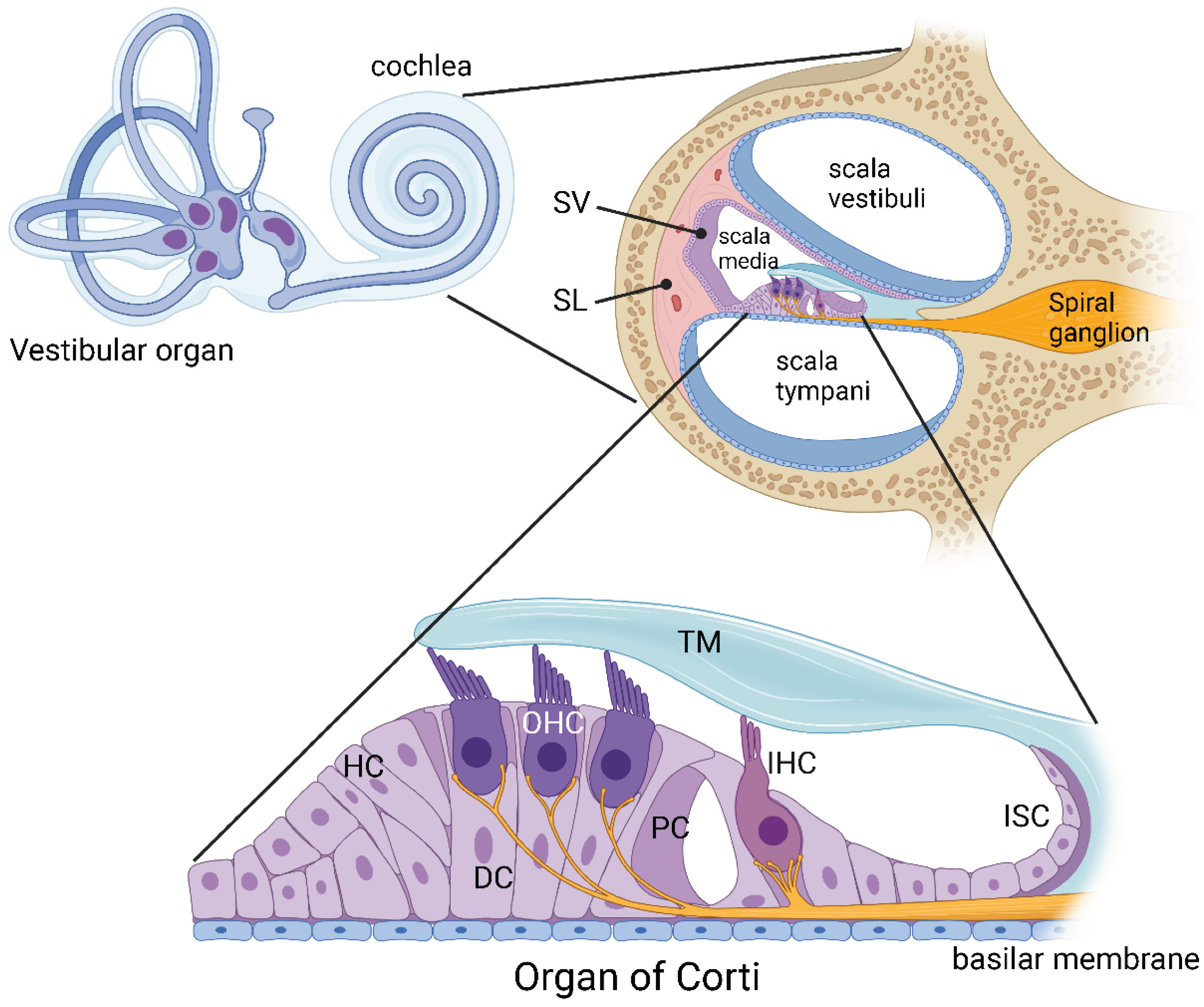
1. Introduction
2. Methodology
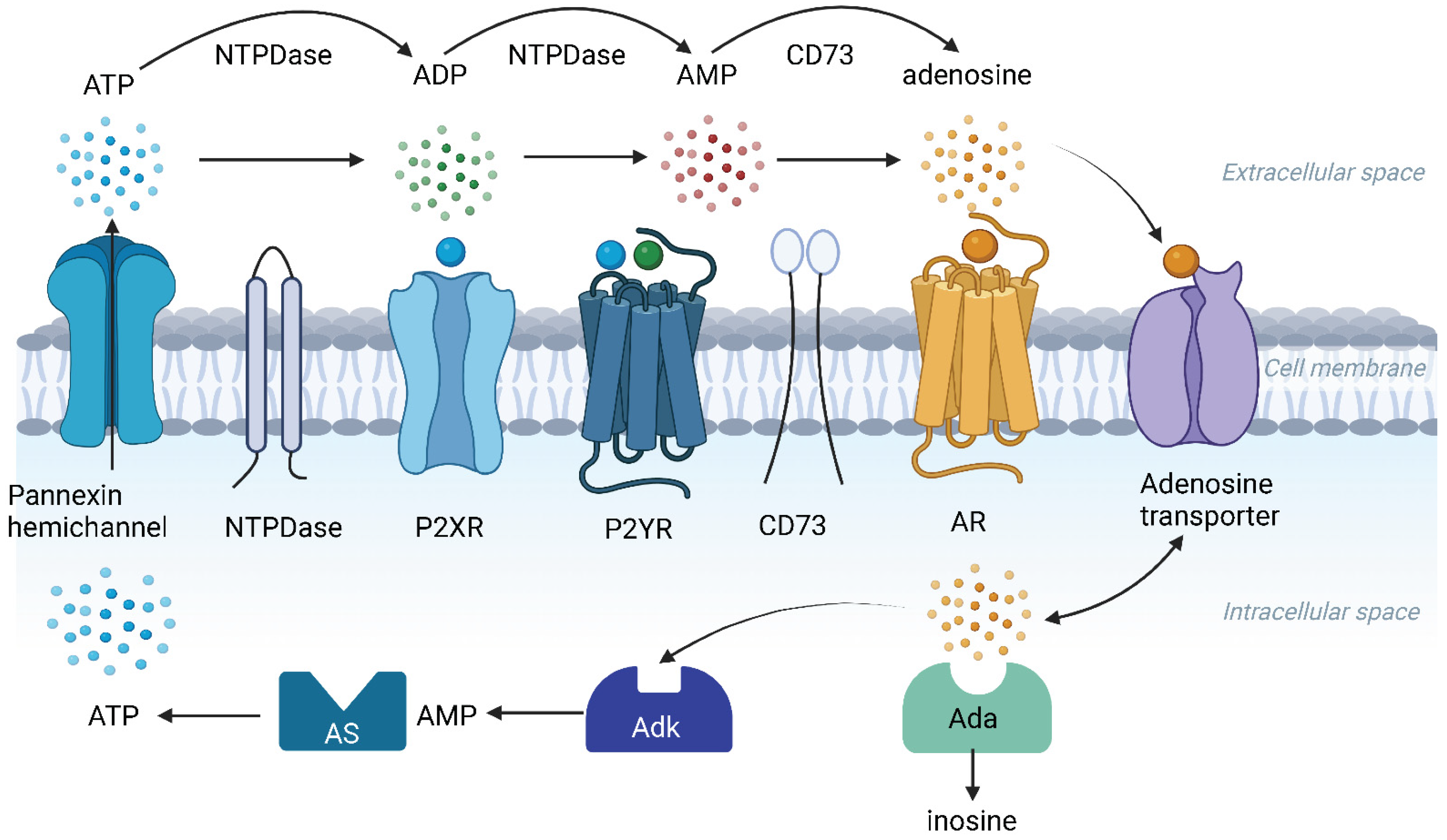
3. ATP Release in the Cochlea
4. P2 Receptors in the Cochlea
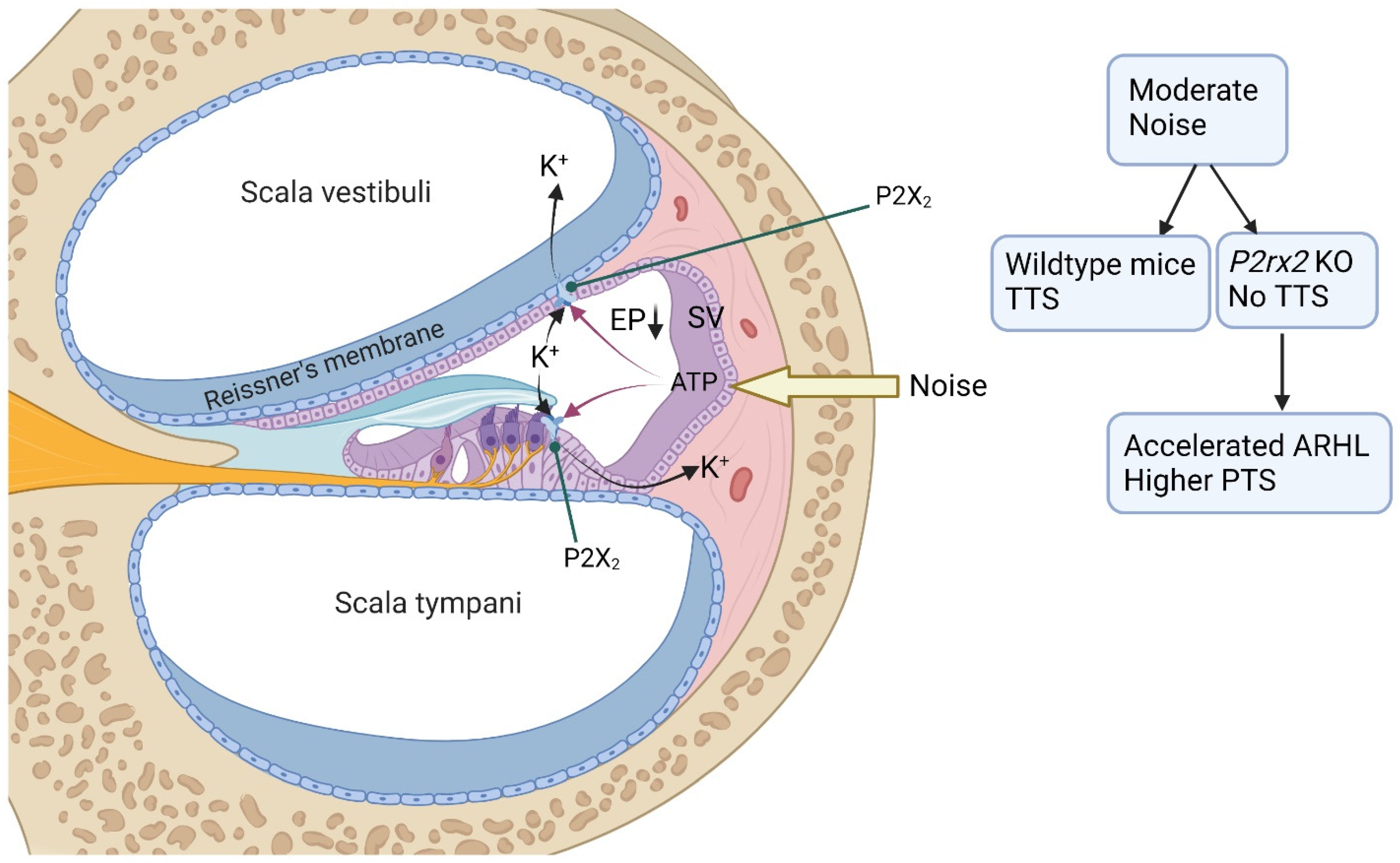
4.1. P2X Receptors in the Cochlea
4.2. P2Y Receptors in the Cochlea
5. ATP and Cochlear Development
6. Ectonucleotidases in the Cochlea
7. Adenosine Receptor Signalling in the Cochlea
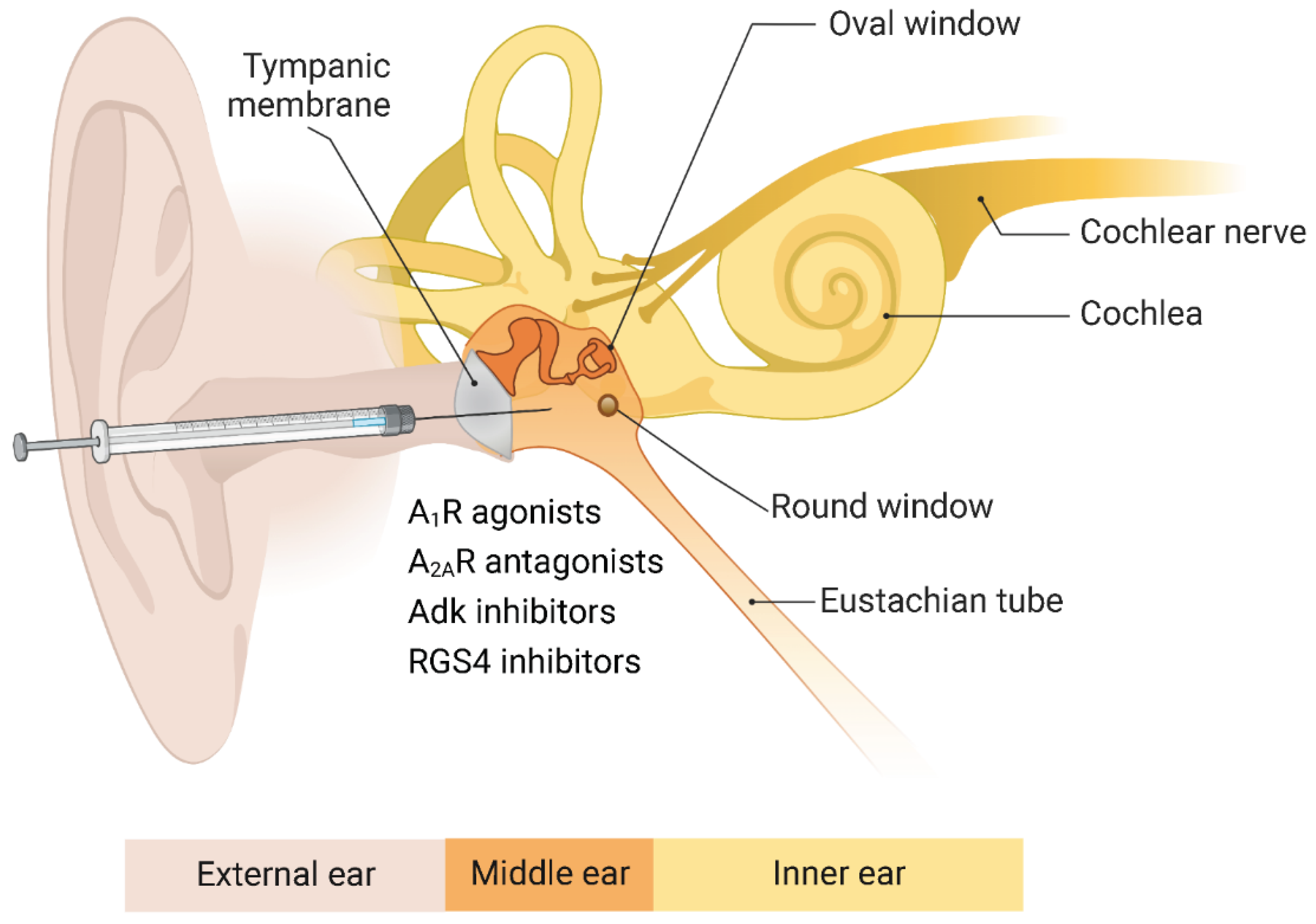
8. Adenosine A1 Receptors and Sensorineural Hearing Loss
9. Adenosine Kinase and Age-Related Hearing Loss
10. Regulators of G Protein Signalling and Hearing Loss
11. Adenosine A2A Receptors and Hearing Loss
12. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Elliott, S.J.; A Shera, C. The cochlea as a smart structure. Smart Mater. Struct. 2012, 21, 064001. [Google Scholar] [CrossRef] [PubMed]
- Raphael, Y.; A Altschuler, R. Structure and innervation of the cochlea. Brain Res. Bull. 2003, 60, 397–422. [Google Scholar] [CrossRef] [PubMed]
- Liberman, M.C.; Gao, J.; He, D.Z.Z.; Wu, X.; Jia, S.; Zuo, J. Prestin is required for electromotility of the outer hair cell and for the cochlear amplifier. Nature 2002, 419, 300–304. [Google Scholar] [CrossRef] [PubMed]
- Cohen-Salmon, M.; Ott, T.; Michel, V.; Hardelin, J.P.; Perfettini, I.; Eybalin, M.; Wu, T.; Marcus, D.C.; Wangemann, P.; Willecke, K.; et al. Targeted ablation of connexin26 in the inner ear epithelial gap junction network causes hearing impairment and cell death. Curr. Biol. 2002, 12, 1106–1111. [Google Scholar] [CrossRef] [PubMed]
- Thorne, P.R.; Munoz, D.J.; Nikolic, P.; Mander, L.; Jagger, D.J.; Greenwood, D.; Vlajkovic, S.; Housley, G.D. Potential Role of Purinergic Signalling in Cochlear Pathology. Audiol. Neurotol. 2002, 7, 180–184. [Google Scholar] [CrossRef] [PubMed]
- Housley, G.D.; Bringmann, A.; Reichenbach, A. Purinergic signaling in special senses. Trends Neurosci. 2009, 32, 128–141. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, D.J.; Kendrick, I.S.; Rassam, M.; Thorne, P.R. Vesicular storage of adenosine triphosphate in the guinea-pig cochlear lateral wall and concentrations of ATP in the endolymph during sound exposure and hypoxia. Acta Oto-Laryngol. 2001, 121, 10–15. [Google Scholar]
- Housley, G.D.; Morton-Jones, R.; Vlajkovic, S.M.; Telang, R.S.; Paramananthasivam, V.; Tadros, S.F.; Wong, A.C.Y.; Froud, K.E.; Cederholm, J.M.; Sivakumaran, Y.; et al. ATP-gated ion channels mediate adaptation to elevated sound levels. Proc. Natl. Acad. Sci. USA 2013, 110, 7494–7499. [Google Scholar] [CrossRef] [PubMed]
- Thorne, P.R.; Muñoz, D.J.B.B.; Housley, G.D. Purinergic Modulation of Cochlear Partition Resistance and Its Effect on the Endocochlear Potential in the Guinea Pig. JARO-J. Assoc. Res. Otolaryngol. 2004, 5, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Housley, G.D.; Jagger, D.J.; Greenwood, D.; Raybould, N.P.; Salih, S.G.; Järlebark, L.E.; Vlajkovic, S.M.; Kanjhan, R.; Nikolic, P.; Muñoz, D.J.M.M.; et al. Purinergic regulation of sound transduction and auditory neurotransmission. Audiol. Neuro-Otol. 2002, 7, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Berekméri, E.; Szepesy, J.; Köles, L.; Zelles, T. Purinergic signaling in the organ of Corti: Potential therapeutic targets of sensorineural hearing losses. Brain Res. Bull. 2019, 151, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Burnstock, G.; Verkhratsky, A. Special Senses. In Purinergic Signalling and the Nervous System; Springer: Berlin/Heidelberg, Germany, 2012; pp. 627–679. [Google Scholar]
- Köles, L.; Szepesy, J.; Berekméri, E.; Zelles, T. Purinergic signalling and cochlear injury-targeting the immune system? Int. J. Mol. Sci. 2019, 20, 2979. [Google Scholar] [CrossRef] [PubMed]
- Abbracchio, M.P.; Burnstock, G.; Verkhratsky, A.; Zimmermann, H. Purinergic signalling in the nervous system: An overview. Trends Neurosci. 2009, 32, 19–29. [Google Scholar] [CrossRef]
- Piazza, V.; Ciubotaru, C.D.; Gale, J.E.; Mammano, F. Purinergic signalling and intercellular Ca2+ wave propagation in the organ of Corti. Cell Calcium 2007, 41, 77–86. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, H. History of ectonucleotidases and their role in purinergic signaling. Biochem. Pharmacol. 2021, 187, 114322. [Google Scholar] [CrossRef]
- Vlajkovic, S.M.; Thorne, P.R.; Sévigny, J.; Robson, S.C.; Housley, G.D. NTPDase1 and NTPDase2 immunolocalization in mouse cochlea: Implications for regulation of p2 receptor signaling. J. Histochem. Cytochem. 2002, 50, 1435–1441. [Google Scholar] [CrossRef]
- Vlajkovic, S.M.; Housley, G.D.; Thorne, P.R. Adenosine and the auditory system. Curr. Neuropharmacol. 2009, 7, 246–256. [Google Scholar] [CrossRef]
- Jovanovic, S.; Milenkovic, I. Purinergic Modulation of Activity in the Developing Auditory Pathway. Neurosci. Bull. 2020, 36, 1285–1298. [Google Scholar] [CrossRef]
- Mammano, F. ATP-dependent intercellular Ca2+ signaling in the developing cochlea: Facts, fantasies and perspectives. Semin. Cell Dev. Biol. 2013, 24, 31–39. [Google Scholar] [CrossRef]
- Tritsch, N.X.; Yi, E.; Gale, J.E.; Glowatzki, E.; Bergles, D.E. The origin of spontaneous activity in the developing auditory system. Nature 2007, 450, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Housley, G.D.; von Jonquieres, G.; Pinyon, J.L.; Matheson, J.-A.T.; Pearson, L.J.; Salthouse, T.P.; Cederholm, J.M. Cochlear homeostasis: A molecular physiological perspective on maintenance of sound transduction and auditory neurotransmission with noise and ageing. Curr. Opin. Physiol. 2020, 18, 106–115. [Google Scholar] [CrossRef]
- Dahl, G. ATP release through pannexon channels. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 2015, 370, 20140191. [Google Scholar] [CrossRef]
- Chen, J.; Zhu, Y.; Liang, C.; Chen, J.; Zhao, H.B. Pannexin1 channels dominate ATP release in the cochlea ensuring endocochlear potential and auditory receptor potential generation and hearing. Sci. Rep. 2015, 5, 10762. [Google Scholar] [CrossRef]
- Belousov, A.B.; Fontes, J.D.; Freitas-Andrade, M.; Naus, C.C. Gap junctions and hemichannels: Communicating cell death in neurodevelopment and disease. BMC Cell Biol. 2017, 18 (Suppl. S1), 4. [Google Scholar] [CrossRef]
- Zhu, Y.; Zhao, H.B. ATP activates P2X receptors to mediate gap junctional coupling in the cochlea. Biochem. Biophys. Res. Commun. 2012, 426, 528–532. [Google Scholar] [CrossRef]
- Ceriani, F.; Pozzan, T.; Mammano, F. Critical role of ATP-induced ATP release for Ca2+ signaling in nonsensory cell networks of the developing cochlea. Proc. Natl. Acad. Sci. USA 2016, 113, E7194–E7201. [Google Scholar] [CrossRef]
- Locovei, S.; Wang, J.; Dahl, G. Activation of pannexin 1 channels by ATP through P2Y receptors and by cytoplasmic calcium. FEBS Lett. 2006, 580, 239–244. [Google Scholar] [CrossRef]
- Baroja-Mazo, A.; Barberà-Cremades, M.; Pelegrín, P. The participation of plasma membrane hemichannels to purinergic signaling. Biochim. Et Biophys. Acta-Biomembr. 2013, 1828, 79–93. [Google Scholar] [CrossRef]
- Sahu, G.; Sukumaran, S.; Bera, A.K. Pannexins form gap junctions with electrophysiological and pharmacological properties distinct from connexins. Sci. Rep. 2014, 4, 4955. [Google Scholar] [CrossRef]
- Zhao, HB. Expression and function of pannexins in the inner ear and hearing. BMC Cell Biol. 2016, 17 (Suppl. S1), 16. [Google Scholar] [CrossRef]
- Zhu, Y.; Zhao, H.B. ATP-mediated potassium recycling in the cochlear supporting cells. Purinergic Signal. 2010, 6, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.-B.; Zhu, Y.; Liang, C.; Chen, J. Pannexin 1 deficiency can induce hearing loss. Biochem. Biophys. Res. Commun. 2015, 463, 143–147. [Google Scholar] [CrossRef] [PubMed]
- Nikolic, P.; Housley, G.D.; Luo, L.; Ryan, A.F.; Thorne, P.R. Transient expression of P2X1 receptor subunits of ATP-gated ion channels in the developing rat cochlea. Dev. Brain Res. 2001, 126, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Järlebark, L.E.; Housley, G.D.; Raybould, N.P.; Vlajkovic, S.; Thorne, P.R. ATP-gated ion channels assembled from P2X2 receptor subunits in the mouse cochlea. Neuroreport 2002, 13, 1979–1984. [Google Scholar] [CrossRef]
- Järlebark, L.E.; Housley, G.D.; Thorne, P.R. Immunohistochemical localization of adenosine 5’-triphosphate-gated ion channel P2X2 receptor subunits in adult and developing rat cochlea. J. Comp. Neurol. 2000, 421, 289–301. [Google Scholar] [CrossRef]
- Huang, L.C.; Ryan, A.F.; Cockayne, D.A.; Housley, G.D. Developmentally regulated expression of the P2X3 receptor in the mouse cochlea. Histochem. Cell Biol. 2006, 125, 681–692. [Google Scholar] [CrossRef]
- Greenwood, D.; Jagger, D.J.; Huang, L.C.; Hoya, N.; Thorne, P.R.; Wildman, S.S.; King, B.F.; Pak, K.; Ryan, A.F.; Housley, G.D. P2X receptor signaling inhibits BDNF-mediated spiral ganglion neuron development in the neonatal rat cochlea. Development 2007, 134, 1407–1417. [Google Scholar] [CrossRef]
- Szücs, A.; Szappanos, H.; Tóth, A.; Farkas, Z.; Panyi, G.; Csernoch, L.; Sziklai, I. Differential expression of purinergic receptor subtypes in the outer hair cells of the guinea pig. Hear. Res. 2004, 196, 2–7. [Google Scholar] [CrossRef]
- Wu, T.; Dai, M.; Shi, X.R.; Jiang, Z.G.; Nuttall, A.L. Functional expression of P2X4 receptor in capillary endothelial cells of the cochlear spiral ligament and its role in regulating the capillary diameter. Am. J. Physiol. Circ. Physiol. 2011, 301, H69–H78. [Google Scholar] [CrossRef]
- Xiang, Z.; Bo, X.; Burnstock, G. P2X receptor immunoreactivity in the rat cochlea, vestibular ganglion and cochlear nucleus. Hear. Res. 1999, 128, 190–196. [Google Scholar] [CrossRef]
- Jagger, D.J.; Housley, G.D. Membrane properties of type II spiral ganglion neurones identified in a neonatal rat cochlear slice. J. Physiol. 2003, 552, 525–533. [Google Scholar] [CrossRef]
- Nikolic, P.; Housley, G.D.; Thorne, P.R. Expression of the P2X7 Receptor Subunit of the Adenosine 5′-Triphosphate-Gated Ion Channel in the Developing and Adult Rat Cochlea. Audiol. Neurotol. 2003, 8, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Prades, S.; Heard, G.; Gale, J.E.; Engel, T.; Kopp, R.; Nicke, A.; Smith, K.E.; Jagger, D.J. Functional P2X7 Receptors in the Auditory Nerve of Hearing Rodents Localize Exclusively to Peripheral Glia. J. Neurosci. 2021, 41, 2615–2629. [Google Scholar] [CrossRef] [PubMed]
- Morton-Jones, R.T.; Vlajkovic, S.M.; Thorne, P.R.; Cockayne, D.A.; Ryan, A.F.; Housley, G.D. Properties of ATP-gated ion channels assembled from P2X2 subunits in mouse cochlear Reissner’s membrane epithelial cells. Purinergic Signal. 2015, 11, 551–560. [Google Scholar] [CrossRef] [PubMed]
- Cederholm, J.M.E.; Ryan, A.F.; Housley, G.D. Onset kinetics of noise-induced purinergic adaptation of the ‘cochlear amplifier’. Purinergic Signal. 2019, 15, 343–355. [Google Scholar] [CrossRef] [PubMed]
- Yan, D.; Zhu, Y.; Walsh, T.; Xie, D.; Yuan, H.; Sirmaci, A.; Fujikawa, T.; Wong, A.C.Y.; Loh, T.L.; Du, L.; et al. Mutation of the ATP-gated P2X 2 receptor leads to progressive hearing loss and increased susceptibility to noise. Proc. Natl. Acad. Sci. USA 2013, 110, 2228–2233. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Abad, C.; Chen, Z.-Y.; I Young, J.; Gurumurthy, C.B.; Walz, K.; Liu, X.Z. Generation and characterization of a P2rx2 V60L mouse model for DFNA41. Hum. Mol. Genet. 2021, 30, 985–995. [Google Scholar] [CrossRef]
- George, B.; Swartz, K.J.; Li, M. Hearing loss mutations alter the functional properties of human P2X2 receptor channels through distinct mechanisms. Proc. Natl. Acad. Sci. USA 2019, 116, 22862–22871. [Google Scholar] [CrossRef]
- Horváth, T.; Polony, G.; Fekete, Á.; Aller, M.; Halmos, G.; Lendvai, B.; Heinrich, A.; Sperlágh, B.; Vizi, E.S.; Zelles, T. ATP-Evoked Intracellular Ca2+ Signaling of Different Supporting Cells in the Hearing Mouse Hemicochlea. Neurochem. Res. 2016, 41, 364–375. [Google Scholar] [CrossRef][Green Version]
- Lahne, M.; Gale, J.E. Damage-induced cell–cell communication in different cochlear cell types via two distinct ATP-dependent Ca2+ waves. Purinergic Signal. 2010, 6, 189–200. [Google Scholar] [CrossRef]
- Sirko, P.; E Gale, J.; Ashmore, J.F. Intercellular Ca2+signalling in the adult mouse cochlea. J. Physiol. 2019, 597, 303–317. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Glowatzki, E.; Fuchs, P.A. Unmyelinated type II afferent neurons report cochlear damage. Proc. Natl. Acad. Sci. USA 2015, 112, 14723–14727. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.C.; Thorne, P.R.; Vlajkovic, S.M.; Housley, G.D. Differential expression of P2Y receptors in the rat cochlea during development. Purinergic Signal. 2010, 6, 231–248. [Google Scholar] [CrossRef] [PubMed]
- Babola, T.A.; Li, S.; Wang, Z.; Kersbergen, C.J.; Elgoyhen, A.B.; Coate, T.M.; Bergles, D.E. Purinergic Signaling Controls Spontaneous Activity in the Auditory System throughout Early Development. J. Neurosci. 2021, 41, 594–612. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Hou, S.; Yang, J. ATP is stored in lysosomes of greater epithelial ridge supporting cells in newborn rat cochleae. J. Cell. Biochem. 2019, 120, 19469–19481. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liu, W.; Yang, J. ATP-containing vesicles in stria vascular marginal cell cytoplasms in neonatal rat cochlea are lysosomes. Sci. Rep. 2016, 6, 20903. [Google Scholar] [CrossRef]
- Liu, B.; Cao, W.; Li, J.; Liu, J. Lysosomal exocytosis of ATP is coupled to P2Y2 receptor in marginal cells in the stria vascular in neonatal rats. Cell Calcium 2018, 76, 62–71. [Google Scholar] [CrossRef]
- Dayaratne, M.W.; Vlajkovic, S.M.; Lipski, J.; Thorne, P.R. Kölliker’s organ and the development of spontaneous activity in the auditory system: Implications for hearing dysfunction. BioMed Res. Int. 2014, 2014, 367939. [Google Scholar] [CrossRef]
- Dayaratne, M.W.; Vlajkovic, S.M.; Lipski, J.; Thorne, P.R. Putative role of border cells in generating spontaneous morphological activity within Kölliker’s organ. Hear. Res. 2015, 330, 90–97. [Google Scholar] [CrossRef]
- Rabbitt, R.D.; Holman, H.A. ATP and ACh Evoked Calcium Transients in the Neonatal Mouse Cochlear and Vestibular Sensory Epithelia. Front. Neurosci. 2021, 15, 710076. [Google Scholar] [CrossRef]
- Ceriani, F.; Hendry, A.; Jeng, J.; Johnson, S.L.; Stephani, F.; Olt, J.; Holley, M.C.; Mammano, F.; Engel, J.; Kros, C.J.; et al. Coordinated calcium signalling in cochlear sensory and non-sensory cells refines afferent innervation of outer hair cells. EMBO J. 2019, 38, e99839. [Google Scholar] [CrossRef]
- Babola, T.A.; Kersbergen, C.J.; Wang, H.C.; Bergles, D.E. Purinergic signaling in cochlear supporting cells reduces hair cell excitability by increasing the extracellular space. eLife 2020, 9, 52160. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Heo, J.H.; Kim, C.H.; Chang, S.O.; Kim, C.S.; Oh, S.H. Changes in P2Y4 receptor expression in rat cochlear outer sulcus cells during development. Hear. Res. 2007, 228, 201–211. [Google Scholar] [CrossRef]
- Wang, Z.; Jung, J.S.; Inbar, T.C.; Rangoussis, K.M.; Faaborg-Andersen, C.; Coate, T.M. The purinergic receptor P2rx3 is required for spiral ganglion neuron branch refinement during development. eNeuro 2020, 7, ENEURO.0179-20.2020. [Google Scholar] [CrossRef] [PubMed]
- Jovanovic, S.; Radulovic, T.; Coddou, C.; Dietz, B.; Nerlich, J.; Stojilkovic, S.S.; Rübsamen, R.; Milenkovic, I. Tonotopic action potential tuning of maturing auditory neurons through endogenous ATP. J. Physiol. 2017, 595, 1315–1337. [Google Scholar] [CrossRef]
- Robson, S.C.; Sévigny, J.; Zimmermann, H. The E-NTPDase family of ectonucleotidases: Structure function relationships and pathophysiological significance. Purinergic Signal. 2006, 2, 409–430. [Google Scholar] [CrossRef]
- Vlajkovic, S.M.; Housley, G.D.; Muñoz, D.J.; Robson, S.C.; Sevigny, J.; Wang, C.J.; Thorne, P.R. Noise exposure induces up-regulation of ecto-nucleoside triphosphate diphosphohydrolases 1 and 2 in rat cochlea. Neuroscience 2004, 126, 763–773. [Google Scholar] [CrossRef]
- Vlajkovic, S.M.; Thorne, P.R.; Sévigny, J.; Robson, S.C.; Housley, G.D. Distribution of ectonucleoside triphosphate diphosphohydrolases 1 and 2 in rat cochlea. Hear. Res. 2002, 170, 127–138. [Google Scholar] [CrossRef]
- Vlajkovic, S.M.; Vinayagamoorthy, A.; Thorne, P.R.; Robson, S.C.; Wang, C.J.; Housley, G.D. Noise-induced up-regulation of NTPDase3 expression in the rat cochlea: Implications for auditory transmission and cochlear protection. Brain Res. 2006, 1104, 55–63. [Google Scholar] [CrossRef]
- O’Keeffe, M.G.; Thorne, P.R.; Housley, G.D.; Robson, S.C.; Vlajkovic, S.M. Distribution of NTPDase5 and NTPDase6 and the regulation of P2Y receptor signalling in the rat cochlea. Purinergic Signal. 2010, 6, 249–261. [Google Scholar] [CrossRef]
- O’Keeffe, M.G.; Thorne, P.R.; Housley, G.D.; Robson, S.C.; Vlajkovic, S.M. Developmentally regulated expression of ectonucleotidases NTPDase5 and NTPDase6 and UDP-responsive P2Y receptors in the rat cochlea. Histochem. Cell Biol. 2010, 133, 425–436. [Google Scholar] [CrossRef] [PubMed]
- O’Keeffe, M.G.; Thorne, P.R.; Housley, G.D.; Robson, S.C.; Vlajkovic, S.M. Hair cell specific NTPDase6 immunolocalisation in vestibular end organs: Potential role of purinergic signaling in vestibular sensory transduction. J. Vestib. Res. 2012, 22, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, K.; Gao, Z.-G. Adenosine receptors as therapeutic targets. Nat. Rev. Drug Discov. 2006, 5, 247–264. [Google Scholar] [CrossRef] [PubMed]
- Ijzerman, A.P.; Jacobson, K.A.; Müller, C.E.; Cronstein, B.N.; Cunha, R.A. International Union of Basic and Clinical Pharmacology. CXII: Adenosine Receptors: A Further Update. Pharmacol. Rev. 2022, 74, 340–372. [Google Scholar] [CrossRef]
- Sheth, S.; Mukherjea, D.; Rybak, L.P.; Ramkumar, V. The contribution of anti-oxidant and anti-inflammatory functions of adenosine A1 Receptor in mediating otoprotection. In Inflammatory Mechanisms in Mediating Hearing Loss; Springer: Berlin/Heidelberg, Germany, 2018; pp. 149–164. [Google Scholar]
- Vlajkovic, S.M.; Ambepitiya, K.; Barclay, M.; Boison, D.; Housley, G.D.; Thorne, P.R. Adenosine receptors regulate susceptibility to noise-induced neural injury in the mouse cochlea and hearing loss. Hear. Res. 2017, 345, 43–51. [Google Scholar] [CrossRef]
- Vlajkovic, S.M.; Abi, S.; Wang, C.J.H.; Housley, G.D.; Thorne, P.R. Differential distribution of adenosine receptors in rat cochlea. Cell Tissue Res. 2007, 328, 461–471. [Google Scholar] [CrossRef]
- Kaur, T.; Borse, V.; Sheth, S.; Sheehan, K.; Ghosh, S.; Tupal, S.; Jajoo, S.; Mukherjea, D.; Rybak, L.P.; Ramkumar, V. Adenosine A1 receptor protects against cisplatin ototoxicity by suppressing the NOX3/STAT1 inflammatory pathway in the cochlea. J. Neurosci. 2016, 36, 3962–3977. [Google Scholar] [CrossRef]
- Khan, A.F.; Thorne, P.R.; Muñoz, D.J.; Wang, C.J.; Housley, G.D.; Vlajkovic, S.M. Nucleoside transporter expression and adenosine uptake in the rat cochlea. NeuroReport 2007, 18, 235–239. [Google Scholar] [CrossRef]
- Manalo, J.M.; Liu, H.; Ding, D.; Hicks, J.; Sun, H.; Salvi, R.; Kellems, R.E.; Pereira, F.A.; Xia, Y. Adenosine A2B receptor: A pathogenic factor and a therapeutic target for sensorineural hearing loss. FASEB J. 2020, 34, 15771–15787. [Google Scholar] [CrossRef]
- Xu, X.; Negandhi, J.; Min, W.; Tsui, M.; Post, M.; Harrison, R.V.; Grunebaum, E. Early enzyme replacement therapy improves hearing and immune defects in adenosine deaminase deficient-mice. Front. Immunol. 2019, 10, 416. [Google Scholar] [CrossRef]
- World Health Organization. World Report on Hearing—Executive Summary; World Health Organization: Geneva, Switzerland, 2021. [Google Scholar]
- Ramkumar, V.; Whitworth, C.A.; Pingle, S.C.; Hughes, L.F.; Rybak, L.P. Noise induces A1 adenosine receptor expression in the chinchilla cochlea. Hear. Res. 2004, 188, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Ramkumar, V.; AJhaveri, K.; Xie, X.; Jajoo, S.; A Toth, L. Nuclear factor κB and adenosine receptors: Biochemical and behavioral profiling. Curr. Neuropharmacol. 2011, 9, 342–349. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.H.; Zheng, X.Y.; McFadden, S.L.; Kopke, R.D.; Henderson, D. R-phenylisopropyladenosine attenuates noise-induced hearing loss in the chinchilla. Hear. Res. 1997, 113, 198–206. [Google Scholar] [CrossRef]
- Wong, A.C.Y.; Guo, C.X.; Gupta, R.; Housley, G.D.; Thorne, P.R.; Vlajkovic, S.M. Post exposure administration of A1 adenosine receptor agonists attenuates noise-induced hearing loss. Hear. Res. 2010, 260, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Vlajkovic, S.M.; Chang, H.; Paek, S.Y.; Chi, H.H.-T.; Sreebhavan, S.; Telang, R.S.; Tingle, M.; Housley, G.D.; Thorne, P.R. Adenosine Amine Congener as a Cochlear Rescue Agent. BioMed Res. Int. 2014, 2014, 841489. [Google Scholar] [CrossRef] [PubMed]
- Vlajkovic, S.M.; Lee, K.-H.; Wong, A.C.Y.; Guo, C.X.; Gupta, R.; Housley, G.D.; Thorne, P.R. Adenosine amine congener mitigates noise-induced cochlear injury. Purinergic Signal. 2010, 6, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.; Telang, R.S.; Sreebhavan, S.; Tingle, M.; Thorne, P.R.; Vlajkovic, S.M. Pharmacokinetic Properties of Adenosine Amine Congener in Cochlear Perilymph after Systemic Administration. BioMed Res. Int. 2017, 2017, 8091462. [Google Scholar] [CrossRef]
- Ford, M.S.; Nie, Z.; Whitworth, C.; Rybak, L.P.; Ramkumar, V. Up-regulation of adenosine receptors in the cochlea by cisplatin. Hear. Res. 1997, 111, 143–152. [Google Scholar] [CrossRef]
- Whitworth, C.A.; Ramkumar, V.; Jones, B.; Tsukasaki, N.; Rybak, L.P. Protection against cisplatin ototoxicity by adenosine agonists. Biochem. Pharmacol. 2004, 67, 1801–1807. [Google Scholar] [CrossRef]
- Gunewardene, N.; Guo, C.X.; Wong, A.C.; Thorne, P.R.; Vlajkovic, S.M. Adenosine amine congener ameliorates cisplatin-induced hearing loss. World J. Otorhinolaryngol. 2013, 3, 100–107. [Google Scholar] [CrossRef]
- Sheth, S.; Sheehan, K.; Dhukhwa, A.; Al Aameri, R.F.; Mamillapalli, C.; Mukherjea, D.; Rybak, L.P.; Ramkumar, V. Oral administration of caffeine exacerbates cisplatin-induced hearing loss. Sci. Rep. 2019, 9, 9571. [Google Scholar] [CrossRef] [PubMed]
- Schacht, J.; Talaska, A.E.; Rybak, L.P. Cisplatin and Aminoglycoside Antibiotics: Hearing Loss and Its Prevention. Anat. Rec. 2012, 295, 1837–1850. [Google Scholar] [CrossRef]
- Lin, S.C.Y.; Thorne, P.R.; Housley, G.D.; Vlajkovic, S.M. Purinergic Signaling and Aminoglycoside Ototoxicity: The Opposing Roles of P1 (Adenosine) and P2 (ATP) Receptors on Cochlear Hair Cell Survival. Front. Cell. Neurosci. 2019, 13, 207. [Google Scholar] [CrossRef] [PubMed]
- Gates, G.A.; Mills, J.H. Presbycusis. Lancet 2005, 366, 1111–1120. [Google Scholar] [CrossRef]
- Vlajkovic, S.M.; Guo, C.X.; Telang, R.; Wong, A.C.Y.; Paramananthasivam, V.; Boison, D.; Housley, G.D.; Thorne, P.R. Adenosine kinase inhibition in the cochlea delays the onset of age-related hearing loss. Exp. Gerontol. 2011, 46, 905–914. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.R.; Erway, L.C.; Cook, S.A.; Willott, J.F.; Zheng, Q.Y. A major gene affecting age-related hearing loss in C57BL/6J mice. Hear. Res. 1997, 114, 83–92. [Google Scholar] [CrossRef]
- Fok, C.; Bogosanovic, M.; Pandya, M.; Telang, R.; Thorne, P.R.; Vlajkovic, S.M. Regulator of G Protein Signalling 4 (RGS4) as a Novel Target for the Treatment of Sensorineural Hearing Loss. Int. J. Mol. Sci. 2020, 22, 3. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Liu, Y.; Cottingham, C.; McMahon, L.; Jiao, K.; Greengard, P.; Wang, Q. Neurabin Scaffolding of Adenosine Receptor and RGS4 Regulates Anti-Seizure Effect of Endogenous Adenosine. J. Neurosci. 2012, 32, 2683–2695. [Google Scholar] [CrossRef] [PubMed]
- Dhukhwa, A.; Al Aameri, R.F.H.; Sheth, S.; Mukherjea, D.; Rybak, L.; Ramkumar, V. Regulator of G protein signaling 17 represents a novel target for treating cisplatin induced hearing loss. Sci. Rep. 2021, 11, 8116. [Google Scholar] [CrossRef] [PubMed]
- Han, B.R.; Lin, S.C.; Espinosa, K.; Thorne, P.R.; Vlajkovic, S.M. Inhibition of the Adenosine A2A Receptor Mitigates Excitotoxic Injury in Organotypic Tissue Cultures of the Rat Cochlea. Cells 2019, 8, 877. [Google Scholar] [CrossRef]
- Shin, M.; Pandya, M.; Espinosa, K.; Telang, R.; Boix, J.; Thorne, P.; Vlajkovic, S. Istradefylline Mitigates Age-Related Hearing Loss in C57BL/6J Mice. Int. J. Mol. Sci. 2021, 22, 8000. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vlajkovic, S.M.; Thorne, P.R. Purinergic Signalling in the Cochlea. Int. J. Mol. Sci. 2022, 23, 14874. https://doi.org/10.3390/ijms232314874
Vlajkovic SM, Thorne PR. Purinergic Signalling in the Cochlea. International Journal of Molecular Sciences. 2022; 23(23):14874. https://doi.org/10.3390/ijms232314874
Chicago/Turabian StyleVlajkovic, Srdjan M., and Peter R. Thorne. 2022. "Purinergic Signalling in the Cochlea" International Journal of Molecular Sciences 23, no. 23: 14874. https://doi.org/10.3390/ijms232314874
APA StyleVlajkovic, S. M., & Thorne, P. R. (2022). Purinergic Signalling in the Cochlea. International Journal of Molecular Sciences, 23(23), 14874. https://doi.org/10.3390/ijms232314874






