Metabolomic Profiling and Assessment of Phenolic Compounds Derived from Vitis davidii Foex Cane and Stem Extracts
Abstract
1. Introduction
2. Results and Discussion
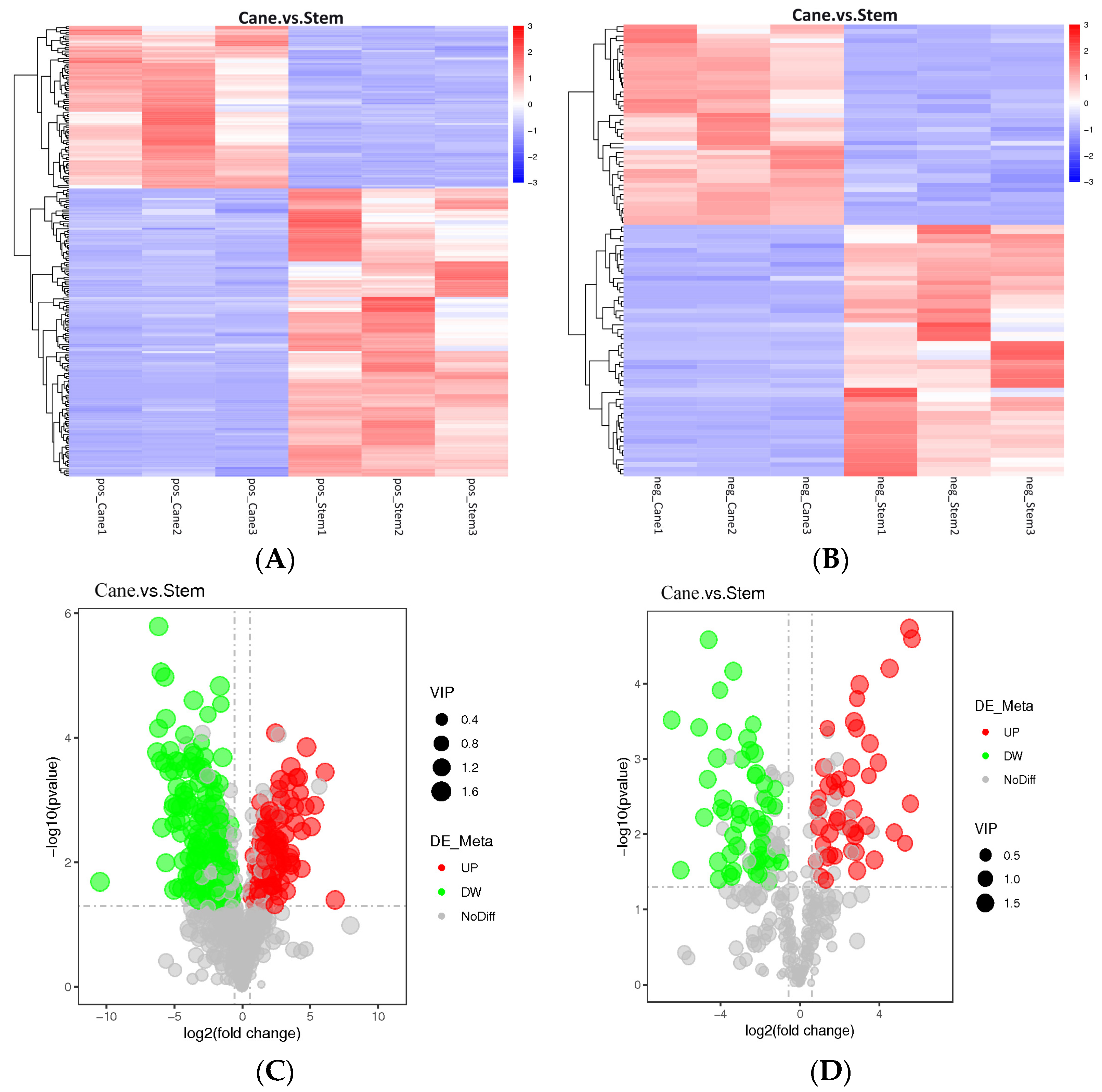
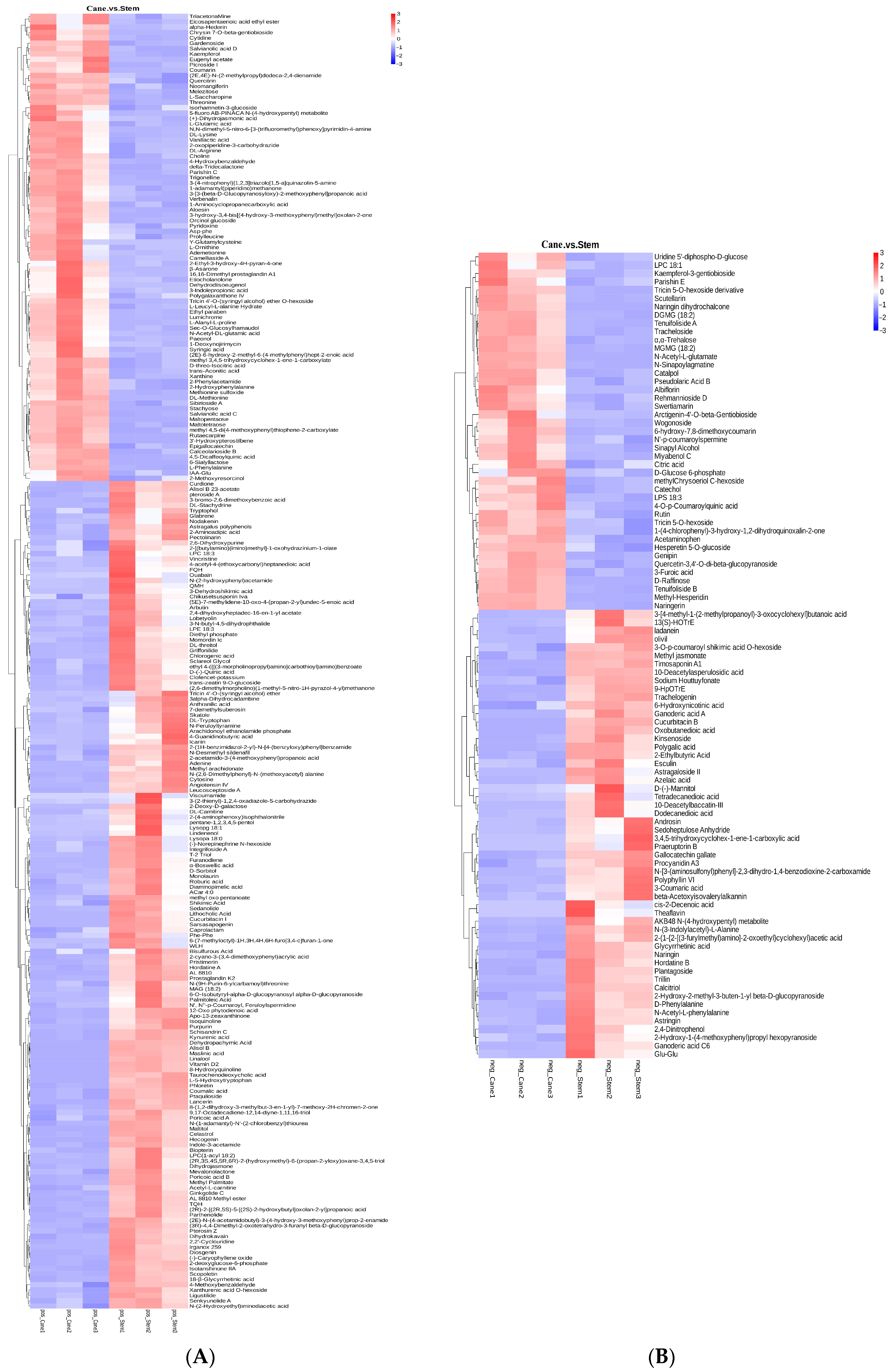
2.1. Metabolic Profiles of Vine Canes and Berry Stem Extracts
2.2. Composition of Phenolic Compounds
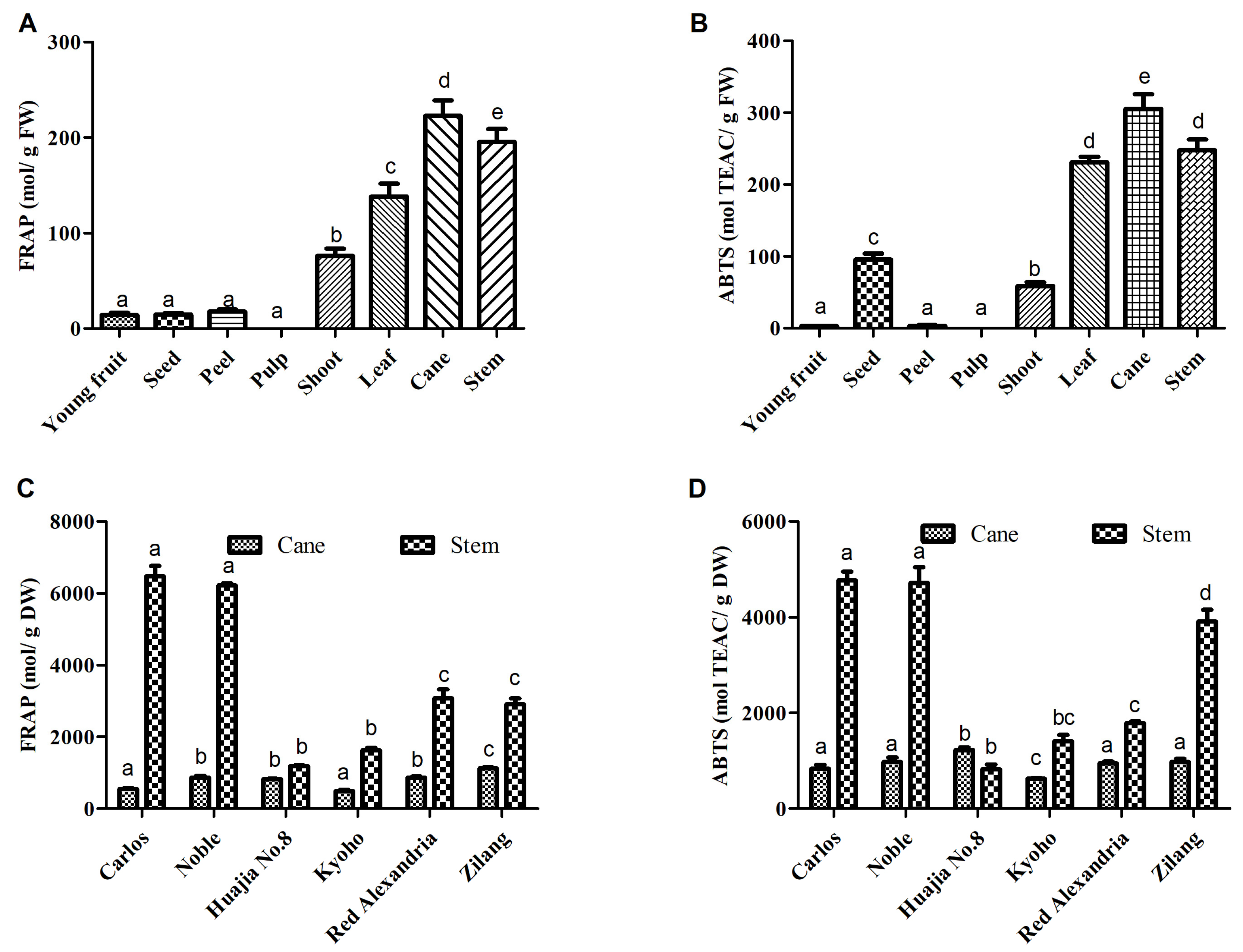
2.3. Antioxidant Activity
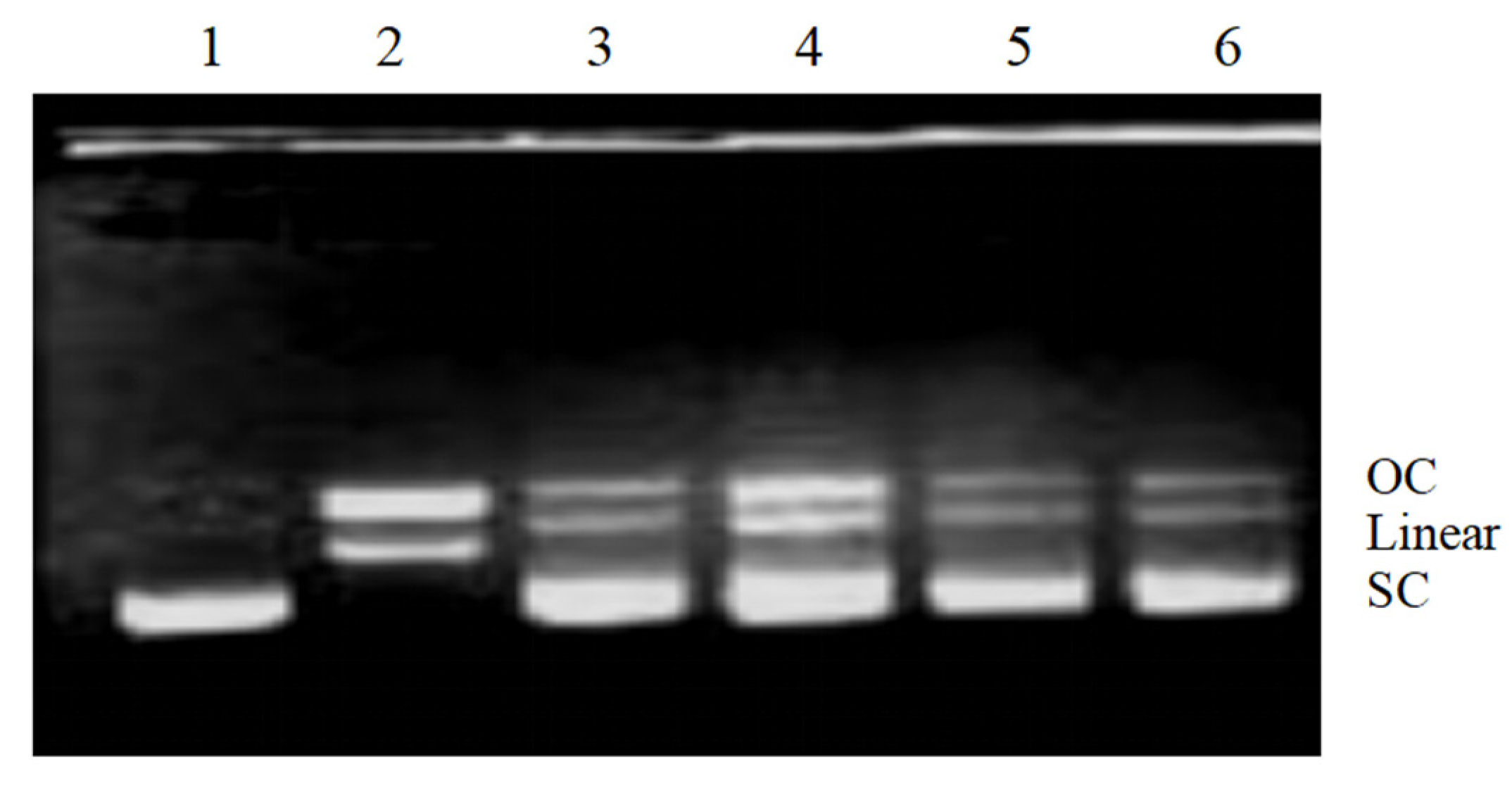
2.4. DNA Damage-Preventing Activity against Reactive Oxygen Species (ROS)
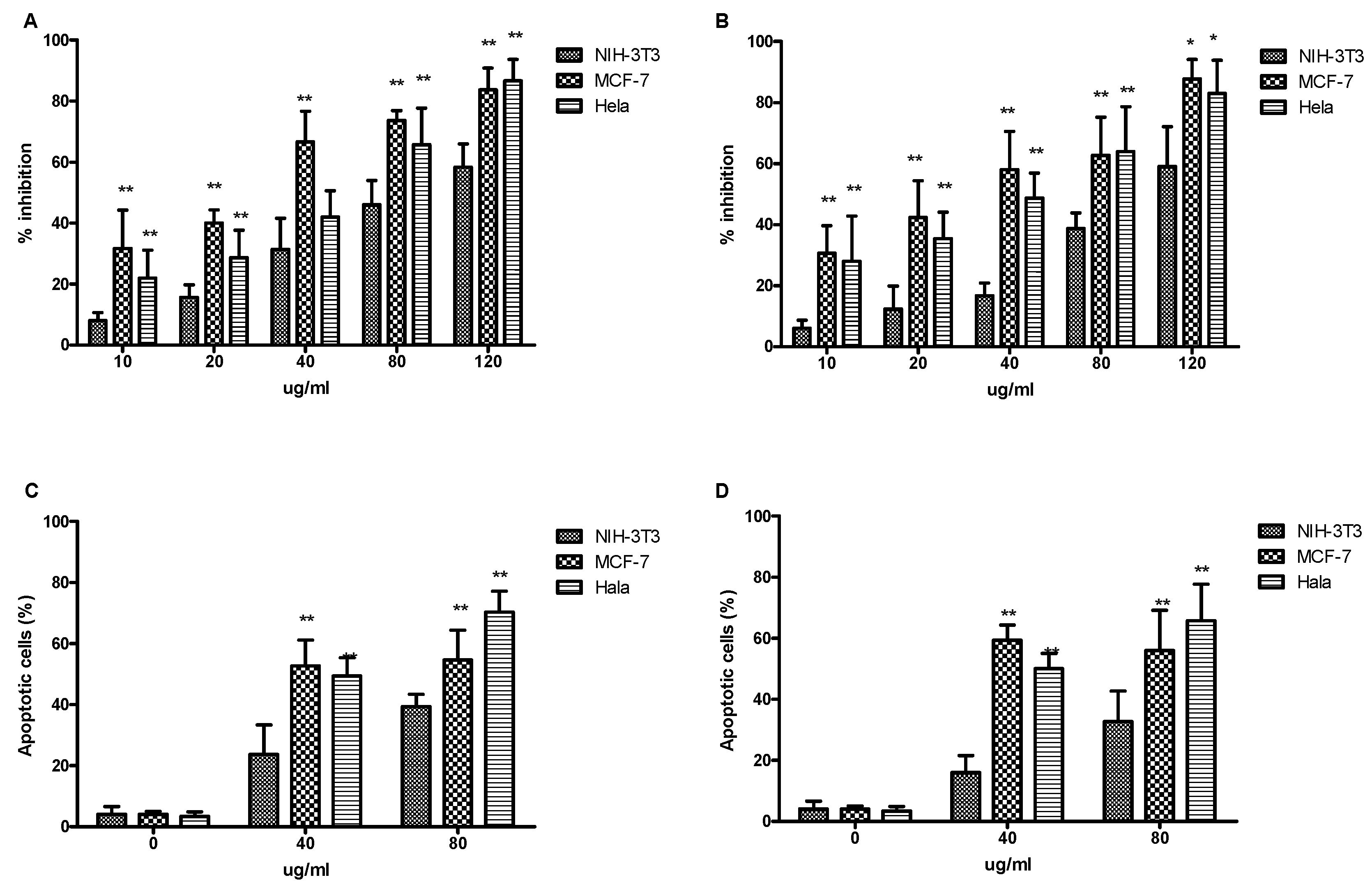
2.5. Inhibition of Cancer Cell Growth in the MCF-7 and HeLa Lines
3. Materials and Methods
3.1. Characteristics of the Experimental Site
3.2. Materials
3.3. Metabolomic Profiling and Analysis
3.4. Extraction of Phenolic Compounds
3.5. Analysis of Polyphenols by HPLC
3.6. Measurement of Antioxidant Properties by ABTS and FRAP
3.7. Hydroxyl Radical-Induced DNA Strand Cleavage Assay
3.8. Anticancer Activity
3.8.1. Cell Culture Conditions and Reagents
3.8.2. MTT Assay for Inhibition of Cell Proliferation
3.8.3. Assessment of Apoptotic Cells
3.9. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Xu, Q.; Fu, Q.; Li, Z.; Liu, H.; Wang, Y.; Lin, X.; He, R.; Zhang, X.; Ju, Z.; Campisi, J.; et al. The flavonoid procyanidin C1 has senotherapeutic activity and increases lifespan in mice. Nat. Metab. 2021, 3, 1706–1726. [Google Scholar] [CrossRef] [PubMed]
- Apostolou, A.; Stagos, D.; Galitsiou, E.; Spyrou, A.; Haroutounian, S.; Portesis, N.; Trizoglou, I.; Wallace Hayes, A.; Tsatsakis, A.M.; Kouretas, D. Assessment of polyphenolic content, antioxidant activity, protection against ROS-induced DNA damage and anticancer activity of Vitis vinifera stem extracts. Food Chem. Toxicol. 2013, 61, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Terra, X.; Valls, J.; Vitrac, X.; Mérrillon, J.M.; Arola, L.; Ardèvol, A.; Bladé, C.; Fernandez-Larrea, J.; Pujadas, G.; Salvadó, J.; et al. Grape-seed procyanidins act as antiinflammatory agents in endotoxin-stimulated RAW 264.7 macrophages by inhibiting NFkB signaling pathway. J. Agric. Food Chem. 2007, 55, 4357–4365. [Google Scholar] [CrossRef] [PubMed]
- Mollica, A.; Scioli, G.; Della Valle, A.; Cichelli, A.; Novellino, E.; Bauer, M.; Kamysz, W.; Llorent-Martinez, E.J.; Fernandez-de Cordova, M.L.; Castillo-López, R.; et al. Phenolic analysis and in vitro biological activity of red wine, pomace and grape seeds oil derived from Vitis vinifera L. cv. Montepulciano d’abruzzo. Antioxidants 2021, 10, 1704. [Google Scholar] [CrossRef] [PubMed]
- Shrotriya, S.; Deep, G.; Gu, M.; Kaur, M.; Jain, A.K.; Inturi, S.; Agarwal, R.; Agarwal, C. Generation of reactive oxygen species by grape seed extract causes irreparable DNA damage leading to G2/M arrest and apoptosis selectively in head and neck squamous cell carcinoma cells. Carcinogenesis 2012, 33, 848–858. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Huo, X.C.; Sun, F.D.; Dong, R.Q. Polyphenol-rich extract of salvia chinensis exhibits anticancer activity in different cancer cell lines, and induces cell cycle arrest at the G0/G1-phase, apoptosis and loss of mitochondrial membrane potential in pancreatic cancer cells. Mol. Med. Rep. 2015, 12, 4843–4850. [Google Scholar] [CrossRef]
- Simonetti, G.; Brasili, E.; Pasqua, G. Antifungal activity of phenolic and polyphenolic compounds from different matrices of Vitis vinifera L. against human pathogens. Molecules 2020, 25, 3748. [Google Scholar] [CrossRef] [PubMed]
- Dani, C.; Oliboni, L.S.; Agostini, F.; Funchal, C.; Serafini, L.; Henriques, J.A.; Salvador, M. Phenolic content of grapevine leaves (Vitis labrusca var. Bordo) and its neuroprotective effect against peroxide damage. Toxicol. In Vitro 2010, 24, 148–153. [Google Scholar] [CrossRef]
- Ashwin, K.; Pattanaik, A.K.; Howarth, G.S. Polyphenolic bioactives as an emerging group of nutraceuticals for promotion of gut health: A review. Food Biosci. 2021, 44, 101376. [Google Scholar] [CrossRef]
- Zhu, L.; Zhang, Y.; Lu, J. Phenolic contents and compositions in skins of red wine grape cultivars among various genetic backgrounds and originations. Int. J. Mol. Sci. 2012, 13, 3492–3510. [Google Scholar] [CrossRef] [PubMed]
- Chiavaroli, A.; Balaha, M.; Acquaviva, A.; Ferrante, C.; Cataldi, A.; Menghini, L.; Rapino, M.; Orlando, G.; Brunetti, L.; Leone, S.; et al. Phenolic characterization and neuroprotective properties of grape pomace extracts. Molecules 2021, 26, 6216. [Google Scholar] [CrossRef] [PubMed]
- Pantelić, M.M.; Dabić Zagorac, D.Č.; Davidović, S.M.; Todić, S.R.; Bešlić, Z.S.; Gašić, U.M.; Tešić, Ž.L.; Natić, M.M. Identification and quantification of phenolic compounds in berry skin, pulp, and seeds in 13 grapevine varieties grown in Serbia. Food Chem. 2016, 211, 243–252. [Google Scholar] [CrossRef] [PubMed]
- Peixoto, C.M.; Dias, M.I.; Alves, M.J.; Calhelha, R.C.; Barros, L.; Pinho, S.P.; Ferreira, I.C.F.R. Grape pomace as a source of phenolic compounds and diverse bioactive properties. Food Chem. 2018, 253, 132–138. [Google Scholar] [CrossRef]
- Esparza, I.; Moler, J.A.; Arteta, M.; Jiménez-Moreno, N.; Ancín-Azpilicueta, C. Phenolic composition of grape stems from different Spanish varieties and vintages. Biomolecules 2021, 11, 1221. [Google Scholar] [CrossRef] [PubMed]
- Anastasiadi, M.; Chorianopoulos, N.G.; Nychas, G.J.; Haroutounian, S.A. Antilisterial activities of polyphenol-rich extracts of grapes and vinification byproducts. J. Agric. Food Chem. 2009, 57, 457–463. [Google Scholar] [CrossRef]
- Liang, N.N.; Pan, Q.H.; He, F.; Wang, J.; Reeves, M.J.; Duan, C.Q. Phenolic profiles of Vitis davidii and Vitis quinquangularis species native to China. J. Agric. Food Chem. 2013, 61, 6016–6027. [Google Scholar] [CrossRef]
- Han, F.; Ju, Y.; Ruan, X.; Zhao, X.; Yue, X.; Zhuang, X.; Qin, M.; Fang, Y. Color, anthocyanin, and antioxidant characteristics of young wines produced from spine grapes (Vitis davidii Foex) in China. Food Nutr. Res. 2017, 61, 1339552. [Google Scholar] [CrossRef]
- Kim, J.Y.; Jeong, H.Y.; Lee, H.K.; Kim, S.; Hwang, B.Y.; Bae, K.; Seong, Y.H. Neuroprotection of the leaf and stem of Vitis amurensis and their active compounds against ischemic brain damage in rats and excitotoxicity in cultured neurons. Phytomedicine 2012, 19, 150–159. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.; Fang, Y.; Wang, H.; Li, H.; Zhang, Z. Free-radical scavenging properties and reducing power of grape cane extracts from 11 selected grape cultivars widely grown in China. Molecules 2011, 16, 10104–10122. [Google Scholar] [CrossRef] [PubMed]
- Khan, Y.; Mulk, K.S.; ul Haq, I.; Farzana, F.; Abdullah, A.; Abbasi, A.M.; Alamri, S.; Hashem, M.; Sakhi, S.; Asif, M.; et al. Antioxidant potential in the leaves of grape varieties (Vitis vinifera L.) grown in different soil compositions. Arab. J. Chem. 2021, 14, 103412. [Google Scholar] [CrossRef]
- Atak, A.; Goksel, Z.; Yilmaz, Y. Changes in major phenolic compounds of seeds, skins, and pulps from various Vitis spp. and the effect of powdery and downy mildew diseases on their levels in grape leaves. Plants 2021, 10, 2554. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, J.A.; Matthews, M.A.; Waterhouse, A.L. Changes in grape seed polyphenols during fruit ripening. Phytochemistry 2000, 1, 77–85. [Google Scholar] [CrossRef]
- Teixeira, A.; Eiras-Dias, J.; Castellarin, S.; Gerós, H. Berry phenolics of grapevine under challenging environments. Int. J. Mol. Sci. 2013, 14, 18711–18739. [Google Scholar] [CrossRef]
- Razgonova, M.; Zakharenko, A.; Pikula, K.; Manakov, Y.; Ercisli, S.; Derbush, I.; Kislin, E.; Seryodkin, I.; Sabitov, A.; Kalenik, T.; et al. LC-MS/MS screening of phenolic compounds in wild and cultivated grapes Vitis amurensis Rupr. Molecules 2021, 26, 3650. [Google Scholar] [CrossRef]
- Kurt-Celebi, A.; Colak, N.; Hayirlioglu-Ayaz, S.; Kostadinović Veličkovska, S.; Ilieva, F.; Esatbeyoglu, T.; Ayaz, F.A. Accumulation of phenolic compounds and antioxidant capacity during berry development in black ‘isabel’ grape (Vitis vinifera L. × Vitis labrusca L.). Molecules 2020, 25, 3845. [Google Scholar] [CrossRef]
- Loupit, G.; Prigent, S.; Franc, C.; De Revel, G.; Richard, T.; Cookson, S.J.; Fonayet, J.V. Polyphenol profiles of just pruned grapevine canes from wild Vitis accessions and Vitis vinifera cultivars. J. Agric. Food Chem. 2020, 68, 13397–13407. [Google Scholar] [CrossRef]
- Liang, Z.; Yang, Y.; Cheng, L.; Zhong, G.Y. Characterization of polyphenolic metabolites in the seeds of Vitis germplasm. J. Agric. Food Chem. 2012, 60, 1291–1299. [Google Scholar] [CrossRef]
- Liang, Z.; Owens, C.L.; Zhong, G.Y.; Cheng, L. Polyphenolic profiles detected in the ripe berries of Vitis vinifera germplasm. Food Chem. 2011, 129, 940–950. [Google Scholar] [CrossRef]
- You, Q.; Chen, F.; Sharp, J.L.; Wang, X.; You, Y.; Zhang, C. High-performance liquid chromatography-mass spectrometry and evaporative light-scattering detector to compare phenolic profiles of muscadine grapes. J. Chromatogr. A 2012, 1240, 96–103. [Google Scholar] [CrossRef]
- Liang, Z.; Yang, Y.; Cheng, L.; Zhong, G.-Y. Polyphenolic composition and content in the ripe berries of wild Vitis species. Food Chem. 2012, 132, 730–738. [Google Scholar] [CrossRef]
- Lindahl, T.; Barnes, D.E. Repair of endogenous DNA damage. Cold Spring Harb. Symp. Quant. Biol. 2000, 65, 127–133. [Google Scholar] [CrossRef]
- Hertog, M.G.; Kromhout, D.; Aravanis, C.; Blackburn, H.; Buzina, R.; Fidanza, F.; Giampaoli, S.; Jansen, A.; Menotti, A.; Nedeljkovic, S. Flavonoid intake and long-term risk of coronary heart disease and cancer in the seven countries study. Arch. Intern. Med. 1995, 155, 381–386. [Google Scholar] [CrossRef]
- Sahpazidou, D.; Geromichalos, G.D.; Stagos, D.; Apostolou, A.; Haroutounian, S.A.; Tsatsakis, A.M.; Tzanakakis, G.N.; Hayes, A.W.; Kouretas, D. Anticarcinogenic activity of polyphenolic extracts from grape stems against breast, colon, renal and thyroid cancer cells. Toxicol. Lett. 2014, 230, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Yi, W.; Akoh, C.C.; Fischer, J.; Krewer, G. Effects of phenolic compounds in blueberries and muscadine grapes on HepG2 cell viability and apoptosis. Food Res. Int. 2006, 39, 628–638. [Google Scholar] [CrossRef]
- Wen, B.; Mei, Z.; Zeng, C.; Liu, S. metaX: A flexible and comprehensive software for processing metabolomics data. BMC Bioinform. 2017, 18, 183. [Google Scholar] [CrossRef]
- Sandhu, A.K.; Gray, D.J.; Lu, J.; Gu, L. Effects of exogenous abscisic acid on antioxidant capacities, anthocyanins, and flavonol contents of muscadine grape (Vitis rotundifolia) skins. Food Chem. 2011, 126, 982–988. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Sun, B.; Ricardo-da-Silva, J.M.; Spranger, I. Critical factors of vanillin assay for catechins and proanthocyanidins. J. Agric. Food Chem. 1998, 46, 4267–4274. [Google Scholar] [CrossRef]
- Miller, N.J.; Rice-Evans, C.; Davies, M.J.; Gopinathan, V.; Milner, A. A novel method for measuring antioxidant capacity and its application to monitoring the antioxidant status in premature neonates. Clin. Sci. 1993, 84, 407–412. [Google Scholar] [CrossRef] [PubMed]
- Benzie, I.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of antioxidant power: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Tian, B.; Hua, Y. Concentration dependence of prooxidant and antioxidant effects of aloin and aloe-emodin on DNA. Food Chem. 2005, 91, 413–418. [Google Scholar] [CrossRef]

| Peak Number | RT (Min) | λmax (nm) | Identity | Content (mg/g DW) | |
|---|---|---|---|---|---|
| Canes | Stems | ||||
| 1 | 2.89 | 280 | Proanthocyanins | 3.25 ± 0.61 d | 11.29 ± 1.15 f |
| 2 | 4.06 | 280 | Gallic acid | 0.28 ± 0.02 a | 0.6 ± 0.04 ab |
| 3 | 7.01 | 320 | Caffeic acid | 0.13 ± 0.00 a | 0.6 ± 0.01 ab |
| 4 | 8.66 | 280 | Catechin | 1.05 ± 0.19 b | 2.16 ± 0.26 c |
| 5 | 10.38 | 331 | 3-coumaric acid | 0.04 ± 0.00 a | 0.26 ± 0.01 a |
| 6 | 11.75 | 320 | Chlorogenic acid | 0.16 ± 0.00 a | 0.12 ± 0.00 a |
| 7 | 14.21 | 280 | Epicatechin | 3.15 ± 0.11 d | 10.43 ± 1.15 e |
| 8 | 27.51 | 365 | Rutin | 0.22 ± 0.01 a | 0.16 ± 0.00 a |
| 9 | 34.21 | 365 | Myricetin 3-O-glucoside | 0.33 ± 0.01 ab | 0.18 ± 0.00 a |
| 10 | 37.62 | 318 | Trans-resveratrol | 0.49 ± 0.02 ab | nt |
| 11 | 55.90 | 360 | Kaempferol 3-O-glucoside | 0.32 ± 0.02 ab | nt |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheng, J.; Xiang, J.; Wei, L.; Zheng, T.; Wu, J. Metabolomic Profiling and Assessment of Phenolic Compounds Derived from Vitis davidii Foex Cane and Stem Extracts. Int. J. Mol. Sci. 2022, 23, 14873. https://doi.org/10.3390/ijms232314873
Cheng J, Xiang J, Wei L, Zheng T, Wu J. Metabolomic Profiling and Assessment of Phenolic Compounds Derived from Vitis davidii Foex Cane and Stem Extracts. International Journal of Molecular Sciences. 2022; 23(23):14873. https://doi.org/10.3390/ijms232314873
Chicago/Turabian StyleCheng, Jianhui, Jiang Xiang, Lingzhu Wei, Ting Zheng, and Jiang Wu. 2022. "Metabolomic Profiling and Assessment of Phenolic Compounds Derived from Vitis davidii Foex Cane and Stem Extracts" International Journal of Molecular Sciences 23, no. 23: 14873. https://doi.org/10.3390/ijms232314873
APA StyleCheng, J., Xiang, J., Wei, L., Zheng, T., & Wu, J. (2022). Metabolomic Profiling and Assessment of Phenolic Compounds Derived from Vitis davidii Foex Cane and Stem Extracts. International Journal of Molecular Sciences, 23(23), 14873. https://doi.org/10.3390/ijms232314873





