Pathophysiology of Type 1 Diabetes and Gut Microbiota Role
Abstract
1. Introduction
2. Epidemiology of T1D
3. Pathophysiology of T1D
4. Role of Thymus-Dependent Immune System
5. T1D Risk Factors
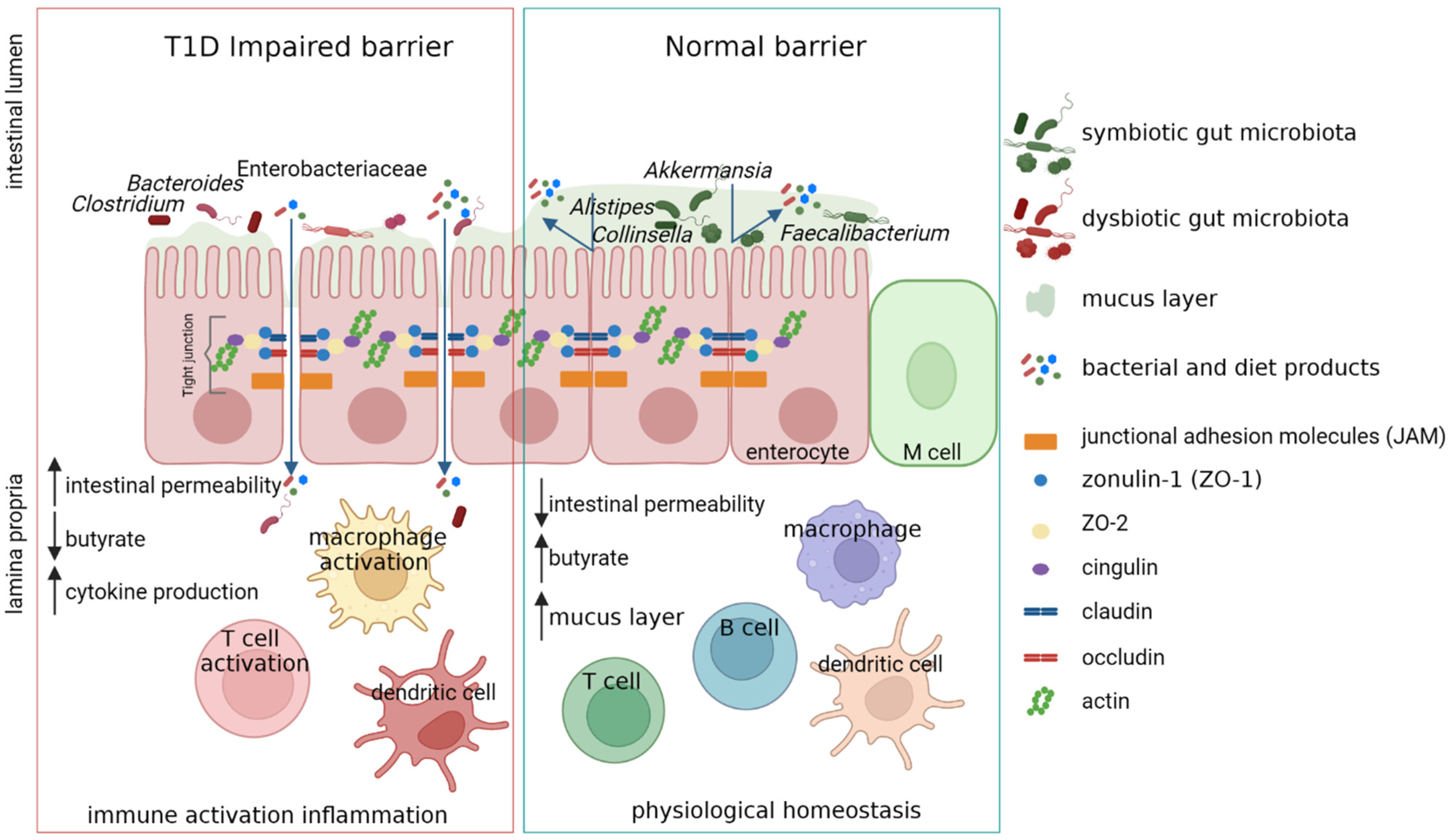
6. Role of Gut Microbiota in T1D Pathophysiology
7. Gut Microbiota Dysbiosis in T1D
8. Butyrate-Associated Barrier Dysfunction in T1D Pathophysiology
9. Probiotics-Based Treatment in T1D Patients
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Maahs, D.M.; West, N.A.; Lawrence, J.M.; Mayer-Davis, E.J. Epidemiology of Type 1 Diabetes. Endocrinol. Metab. Clin. N. Am. 2010, 39, 481–497. [Google Scholar] [CrossRef] [PubMed]
- Karuranga, S.; Malanda, B.; Saeedi, P.; Salpea, P. (Eds.) IDF Diabetes Atlas, 9th Edition Committee IDF DIABETES ATLAS Ninth Edition 2019; IDF: Brussels, Belgium, 2019; ISBN 978-2-930229-87-4. [Google Scholar]
- Lucier, J.; Weinstock, R.S. Diabetes Mellitus Type 1. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Taplin, C.E.; Barker, J.M. Autoantibodies in Type 1 Diabetes. Autoimmunity 2008, 41, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Insel, R.A.; Dunne, J.L.; Atkinson, M.A.; Chiang, J.L.; Dabelea, D.; Gottlieb, P.A.; Greenbaum, C.J.; Herold, K.C.; Krischer, J.P.; Lernmark, Å.; et al. Staging Presymptomatic Type 1 Diabetes: A Scientific Statement of JDRF, the Endocrine Society, and the American Diabetes Association. Diabetes Care 2015, 38, 1964–1974. [Google Scholar] [CrossRef] [PubMed]
- MetaHIT Consortium; Qin, J.; Li, R.; Raes, J.; Arumugam, M.; Burgdorf, K.S.; Manichanh, C.; Nielsen, T.; Pons, N.; Levenez, F.; et al. A Human Gut Microbial Gene Catalogue Established by Metagenomic Sequencing. Nature 2010, 464, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Petersen, C.; Round, J.L. Defining Dysbiosis and Its Influence on Host Immunity and Disease. Cell Microbiol. 2014, 16, 1024–1033. [Google Scholar] [CrossRef]
- Harsch, I.; Konturek, P. The Role of Gut Microbiota in Obesity and Type 2 and Type 1 Diabetes Mellitus: New Insights into “Old” Diseases. Med. Sci. 2018, 6, 32. [Google Scholar] [CrossRef] [PubMed]
- Gavin, P.G.; Hamilton-Williams, E.E. The Gut Microbiota in Type 1 Diabetes: Friend or Foe? Curr. Opin. Endocrinol. Diabetes Obes. 2019, 26, 207–212. [Google Scholar] [CrossRef]
- Zhou, H.; Sun, L.; Zhang, S.; Zhao, X.; Gang, X.; Wang, G. Evaluating the Causal Role of Gut Microbiota in Type 1 Diabetes and Its Possible Pathogenic Mechanisms. Front. Endocrinol. 2020, 11, 125. [Google Scholar] [CrossRef]
- Gale, E.A.M. Type 1 Diabetes in the Young: The Harvest of Sorrow Goes On. Diabetologia 2005, 48, 1435–1438. [Google Scholar] [CrossRef] [PubMed]
- Harjutsalo, V.; Sjöberg, L.; Tuomilehto, J. Time Trends in the Incidence of Type 1 Diabetes in Finnish Children: A Cohort Study. Lancet 2008, 371, 1777–1782. [Google Scholar] [CrossRef]
- Östman, J.; Lönnberg, G.; Arnqvist, H.J.; Blohmé, G.; Bolinder, J.; Schnell, A.E.; Eriksson, J.W.; Gudbjörnsdottir, S.; Sundkvist, G.; Nyström, L. Gender Differences and Temporal Variation in the Incidence of Type 1 Diabetes: Results of 8012 Cases in the Nationwide Diabetes Incidence Study in Sweden 1983–2002. J. Intern. Med. 2008, 263, 386–394. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Magdalena, P.; Ropero, A.B.; Carrera, M.P.; Cederroth, C.R.; Baquie, M.; Gauthier, B.R.; Nef, S.; Stefani, E.; Nadal, A. Pancreatic Insulin Content Regulation by the Estrogen Receptor ERα. PLoS ONE 2008, 3, e2069. [Google Scholar] [CrossRef] [PubMed]
- Santos, R.S.; Batista, T.M.; Camargo, R.L.; Morato, P.N.; Borck, P.C.; Leite, N.C.; Kurauti, M.A.; Wanschel, A.C.; Nadal, Á.; Clegg, D.J. Lacking of Estradiol Reduces Insulin Exocytosis from Pancreatic β-Cells and Increases Hepatic Insulin Degradation. Steroids 2016, 114, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Kooptiwut, S.; Mahawong, P.; Hanchang, W.; Semprasert, N.; Kaewin, S.; Limjindaporn, T.; Yenchitsomanus, P. Estrogen Reduces Endoplasmic Reticulum Stress to Protect against Glucotoxicity Induced-Pancreatic β-Cell Death. J. Steroid Biochem. Mol. Biol. 2014, 139, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Moltchanova, E.V.; Schreier, N.; Lammi, N.; Karvonen, M. Seasonal Variation of Diagnosis of Type 1 Diabetes Mellitus in Children Worldwide. Diabet. Med. 2009, 26, 673–678. [Google Scholar] [CrossRef] [PubMed]
- Kahn, H.S.; Morgan, T.M.; Case, L.D.; Dabelea, D.; Mayer-Davis, E.J.; Lawrence, J.M.; Marcovina, S.M.; Imperatore, G. SEARCH for Diabetes in Youth Study Group Association of Type 1 Diabetes with Month of Birth among U.S. Youth: The SEARCH for Diabetes in Youth Study. Diabetes Care 2009, 32, 2010–2015. [Google Scholar] [CrossRef]
- Forouhi, N.G.; Wareham, N.J. Epidemiology of Diabetes. Medicine (Abingdon) 2014, 42, 698–702. [Google Scholar] [CrossRef]
- DIAMOND Project Group. Incidence and Trends of Childhood Type 1 Diabetes Worldwide 1990–1999. Diabet Med. 2006, 23, 857–866. [Google Scholar] [CrossRef]
- EURODIAB ACE Study Group. Variation and Trends in Incidence of Childhood Diabetes in Europe. Lancet 2000, 355, 873–876. [Google Scholar] [CrossRef]
- Adeloye, D.; Chan, K.Y.; Thorley, N.; Jones, C.; Johnstone, D.; L’Heveder, A.; Saftic, V.; Henderson, D.; Chopra, M.; Campbell, H.; et al. Global and Regional Estimates of the Morbidity Due to Type I Diabetes among Children Aged 0–4 Years: A Systematic Review and Analysis. J. Glob. Health 2018, 8, 021101. [Google Scholar] [CrossRef]
- Patterson, C.C.; Gyürüs, E.; Rosenbauer, J.; Cinek, O.; Neu, A.; Schober, E.; Parslow, R.C.; Joner, G.; Svensson, J.; Castell, C.; et al. Trends in Childhood Type 1 Diabetes Incidence in Europe during 1989-2008: Evidence of Non-Uniformity over Time in Rates of Increase. Diabetologia 2012, 55, 2142–2147. [Google Scholar] [CrossRef] [PubMed]
- Podar, T.; Solntsev, A.; Karvonen, M.; Padaiga, Z.; Brigis, G.; Urbonaite, B.; Viik-Kajander, M.; Reunanen, A.; Tuomilehto, J. Increasing Incidence of Childhood-Onset Type I Diabetes in 3 Baltic Countries and Finland 1983–1998. Diabetologia 2001, 44 (Suppl. S3), B17–B20. [Google Scholar] [CrossRef] [PubMed]
- Mayer-Davis, E.J.; Lawrence, J.M.; Dabelea, D.; Divers, J.; Isom, S.; Dolan, L.; Imperatore, G.; Linder, B.; Marcovina, S.; Pettitt, D.J.; et al. Incidence Trends of Type 1 and Type 2 Diabetes among Youths, 2002–2012. N. Engl. J. Med. 2017, 376, 1419–1429. [Google Scholar] [CrossRef] [PubMed]
- DiMeglio, L.A.; Evans-Molina, C.; Oram, R.A. Type 1 Diabetes. Lancet 2018, 391, 2449–2462. [Google Scholar] [CrossRef]
- Atkinson, M.A. The Pathogenesis and Natural History of Type 1 Diabetes. Cold Spring Harb. Perspect. Med. 2012, 2, a007641. [Google Scholar] [CrossRef]
- Mallone, R.; Eizirik, D.L. Presumption of Innocence for Beta Cells: Why Are They Vulnerable Autoimmune Targets in Type 1 Diabetes? Diabetologia 2020, 63, 1999–2006. [Google Scholar] [CrossRef]
- Damond, N.; Engler, S.; Zanotelli, V.R.T.; Schapiro, D.; Wasserfall, C.H.; Kusmartseva, I.; Nick, H.S.; Thorel, F.; Herrera, P.L.; Atkinson, M.A.; et al. A Map of Human Type 1 Diabetes Progression by Imaging Mass Cytometry. Cell Metab. 2019, 29, 755–768.e5. [Google Scholar] [CrossRef]
- von Herrath, M.; Sanda, S.; Herold, K. Type 1 Diabetes as a Relapsing–Remitting Disease? Nat. Rev. Immunol. 2007, 7, 988–994. [Google Scholar] [CrossRef]
- Burkart, V.; Strassburger, K.; Zivehe, F.; Markgraf, D.; Herder, C.; Müssig, K.; Szendroedi, J.; Schloot, N.; Roden, M.; GDS Group. Inverse Association of Insulin Antibody Levels with Insulin Sensitivity in Adults with Type 1 Diabetes. Diabet Med. 2018, 35, 595–601. [Google Scholar] [CrossRef]
- Jelenik, T.; Séquaris, G.; Kaul, K.; Ouwens, D.M.; Phielix, E.; Kotzka, J.; Knebel, B.; Weiß, J.; Reinbeck, A.L.; Janke, L.; et al. Tissue-Specific Differences in the Development of Insulin Resistance in a Mouse Model for Type 1 Diabetes. Diabetes 2014, 63, 3856–3867. [Google Scholar] [CrossRef]
- Chen, C.; Cohrs, C.M.; Stertmann, J.; Bozsak, R.; Speier, S. Human Beta Cell Mass and Function in Diabetes: Recent Advances in Knowledge and Technologies to Understand Disease Pathogenesis. Mol. Metab. 2017, 6, 943–957. [Google Scholar] [CrossRef] [PubMed]
- Oram, R.A.; Sims, E.K.; Evans-Molina, C. Beta Cells in Type 1 Diabetes: Mass and Function; Sleeping or Dead? Diabetologia 2019, 62, 567–577. [Google Scholar] [CrossRef] [PubMed]
- Clark, M.; Kroger, C.J.; Tisch, R.M. Type 1 Diabetes: A Chronic Anti-Self-Inflammatory Response. Front. Immunol. 2017, 8, 1898. [Google Scholar] [CrossRef] [PubMed]
- Mendes-da-Cruz, D.A.; Lemos, J.P.; Passos, G.A.; Savino, W. Abnormal T-Cell Development in the Thymus of Non-Obese Diabetic Mice: Possible Relationship With the Pathogenesis of Type 1 Autoimmune Diabetes. Front. Endocrinol. 2018, 9, 381. [Google Scholar] [CrossRef]
- Holohan, D.R.; Van Gool, F.; Bluestone, J.A. Thymically-Derived Foxp3+ Regulatory T Cells Are the Primary Regulators of Type 1 Diabetes in the Non-Obese Diabetic Mouse Model. PLoS ONE 2019, 14, e0217728. [Google Scholar] [CrossRef]
- Spence, A.; Purtha, W.; Tam, J.; Dong, S.; Kim, Y.; Ju, C.-H.; Sterling, T.; Nakayama, M.; Robinson, W.H.; Bluestone, J.A.; et al. Revealing the Specificity of Regulatory T Cells in Murine Autoimmune Diabetes. Proc. Natl. Acad. Sci. USA 2018, 115, 5265–5270. [Google Scholar] [CrossRef]
- Pugliese, A. Autoreactive T Cells in Type 1 Diabetes. J. Clin. Investig. 2017, 127, 2881–2891. [Google Scholar] [CrossRef]
- Ehlers, M.R.; Rigby, M.R. Targeting Memory T Cells in Type 1 Diabetes. Curr. Diab. Rep. 2015, 15, 84. [Google Scholar] [CrossRef]
- Narsale, A.; Davies, J.D. Memory T Cells in Type 1 Diabetes: The Devil Is in the Detail. Curr. Diab. Rep. 2017, 17, 61. [Google Scholar] [CrossRef]
- Lee, H.S.; Hwang, J.S. Genetic Aspects of Type 1 Diabetes. Ann. Pediatr. Endocrinol. Metab. 2019, 24, 143–148. [Google Scholar] [CrossRef]
- Redondo, M.J.; Steck, A.K.; Pugliese, A. Genetics of Type 1 Diabetes. Pediatr. Diabetes 2018, 19, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Pociot, F. Type 1 Diabetes Genome-Wide Association Studies: Not to Be Lost in Translation. Clin. Trans. Immunol. 2017, 6, e162. [Google Scholar] [CrossRef]
- Klak, M.; Gomółka, M.; Kowalska, P.; Cichoń, J.; Ambrożkiewicz, F.; Serwańska-Świętek, M.; Berman, A.; Wszoła, M. Type 1 Diabetes: Genes Associated with Disease Development. Cejoi 2020, 45, 439–453. [Google Scholar] [CrossRef] [PubMed]
- Nyaga, D.M.; Vickers, M.H.; Jefferies, C.; Perry, J.K.; O’Sullivan, J.M. Type 1 Diabetes Mellitus-Associated Genetic Variants Contribute to Overlapping Immune Regulatory Networks. Front. Genet. 2018, 9, 535. [Google Scholar] [CrossRef]
- Noble, J.A. Immunogenetics of Type 1 Diabetes: A Comprehensive Review. J. Autoimmun. 2015, 64, 101–112. [Google Scholar] [CrossRef] [PubMed]
- Buschard, K. What Causes Type 1 Diabetes? Lessons from Animal Models. APMIS 2011, 119, 1–19. [Google Scholar] [CrossRef]
- Oilinki, T.; Otonkoski, T.; Ilonen, J.; Knip, M.; Miettinen, P. Prevalence and Characteristics of Diabetes among Somali Children and Adolescents Living in Helsinki, Finland. Pediatr. Diabetes 2012, 13, 176–180. [Google Scholar] [CrossRef] [PubMed]
- Söderström, U.; Åman, J.; Hjern, A. Being Born in Sweden Increases the Risk for Type 1 Diabetes–a Study of Migration of Children to Sweden as a Natural Experiment. Acta Paediatr. 2012, 101, 73–77. [Google Scholar] [CrossRef]
- Rewers, M.; Ludvigsson, J. Environmental Risk Factors for Type 1 Diabetes. Lancet 2016, 387, 2340–2348. [Google Scholar] [CrossRef] [PubMed]
- Esposito, S.; Toni, G.; Tascini, G.; Santi, E.; Berioli, M.G.; Principi, N. Environmental Factors Associated with Type 1 Diabetes. Front. Endocrinol. 2019, 10, 592. [Google Scholar] [CrossRef] [PubMed]
- Infante, M.; Ricordi, C.; Sanchez, J.; Clare-Salzler, M.J.; Padilla, N.; Fuenmayor, V.; Chavez, C.; Alvarez, A.; Baidal, D.; Alejandro, R.; et al. Influence of Vitamin D on Islet Autoimmunity and Beta-Cell Function in Type 1 Diabetes. Nutrients 2019, 11, 2185. [Google Scholar] [CrossRef] [PubMed]
- Vaarala, O.; Atkinson, M.A.; Neu, J. The “Perfect Storm” for Type 1 Diabetes: The Complex Interplay between Intestinal Microbiota, Gut Permeability, and Mucosal Immunity. Diabetes 2008, 57, 2555–2562. [Google Scholar] [CrossRef]
- Bibbò, S.; Dore, M.P.; Pes, G.M.; Delitala, G.; Delitala, A.P. Is There a Role for Gut Microbiota in Type 1 Diabetes Pathogenesis? Ann. Med. 2017, 49, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Uzzau, S.; Goldblum, S.E.; Fasano, A. Human Zonulin, a Potential Modulator of Intestinal Tight Junctions. J. Cell Sci. 2000, 113 Pt 24, 4435–4440. [Google Scholar] [CrossRef] [PubMed]
- Asmar, R.; Gosse, P.; Topouchian, J.; N’tela, G.; Dudley, A.; Shepherd, G.L. Effects of Telmisartan on Arterial Stiffness in Type 2 Diabetes Patients with Essential Hypertension. J. Renin. Angiotensin. Aldosterone Syst. 2002, 3, 176–180. [Google Scholar] [CrossRef] [PubMed]
- Watts, T.; Berti, I.; Sapone, A.; Gerarduzzi, T.; Not, T.; Zielke, R.; Fasano, A. Role of the Intestinal Tight Junction Modulator Zonulin in the Pathogenesis of Type I Diabetes in BB Diabetic-Prone Rats. Proc. Natl. Acad. Sci. USA 2005, 102, 2916–2921. [Google Scholar] [CrossRef] [PubMed]
- Fasano, A. Intestinal Permeability and Its Regulation by Zonulin: Diagnostic and Therapeutic Implications. Clin. Gastroenterol. Hepatol. 2012, 10, 1096–1100. [Google Scholar] [CrossRef] [PubMed]
- Kelly, C.P.; Green, P.H.R.; Murray, J.A.; Dimarino, A.; Colatrella, A.; Leffler, D.A.; Alexander, T.; Arsenescu, R.; Leon, F.; Jiang, J.G.; et al. Larazotide Acetate in Patients with Coeliac Disease Undergoing a Gluten Challenge: A Randomised Placebo-Controlled Study. Aliment. Pharm. 2013, 37, 252–262. [Google Scholar] [CrossRef] [PubMed]
- Sapone, A.; De Magistris, L.; Pietzak, M.; Clemente, M.G.; Tripathi, A.; Cucca, F.; Lampis, R.; Kryszak, D.; Cartenì, M.; Generoso, M. Zonulin Upregulation Is Associated with Increased Gut Permeability in Subjects with Type 1 Diabetes and Their Relatives. Diabetes 2006, 55, 1443–1449. [Google Scholar] [CrossRef] [PubMed]
- Mønsted, M.Ø.; Falck, N.D.; Pedersen, K.; Buschard, K.; Holm, L.J.; Haupt-Jorgensen, M. Intestinal Permeability in Type 1 Diabetes: An Updated Comprehensive Overview. J. Autoimmun. 2021, 122, 102674. [Google Scholar] [CrossRef] [PubMed]
- Bosi, E.; Molteni, L.; Radaelli, M.; Folini, L.; Fermo, I.; Bazzigaluppi, E.; Piemonti, L.; Pastore, M.; Paroni, R. Increased Intestinal Permeability Precedes Clinical Onset of Type 1 Diabetes. Diabetologia 2006, 49, 2824–2827. [Google Scholar] [CrossRef] [PubMed]
- Secondulfo, M.; Iafusco, D.; Carratu, R.; Demagistris, L.; Sapone, A.; Generoso, M.; Mezzogiorno, A.; Sasso, F.; Cartenì, M.; De Rosa, R. Ultrastructural Mucosal Alterations and Increased Intestinal Permeability in Non-Celiac, Type I Diabetic Patients. Dig. Liver Dis. 2004, 36, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Paroni, R.; Fermo, I.; Molteni, L.; Folini, L.; Pastore, M.R.; Mosca, A.; Bosi, E. Lactulose and Mannitol Intestinal Permeability Detected by Capillary Electrophoresis. J. Chromatogr. B 2006, 834, 183–187. [Google Scholar] [CrossRef]
- Maffeis, C.; Martina, A.; Corradi, M.; Quarella, S.; Nori, N.; Torriani, S.; Plebani, M.; Contreas, G.; Felis, G.E. Association between Intestinal Permeability and Faecal Microbiota Composition in Italian Children with Beta Cell Autoimmunity at Risk for Type 1 Diabetes. Diabetes/Metab. Res. Rev. 2016, 32, 700–709. [Google Scholar] [CrossRef]
- Harbison, J.E.; Roth-Schulze, A.J.; Giles, L.C.; Tran, C.D.; Ngui, K.M.; Penno, M.A.; Thomson, R.L.; Wentworth, J.M.; Colman, P.G.; Craig, M.E.; et al. Gut Microbiome Dysbiosis and Increased Intestinal Permeability in Children with Islet Autoimmunity and Type 1 Diabetes: A Prospective Cohort Study. Pediatr. Diabetes 2019, 20, 574–583. [Google Scholar] [CrossRef] [PubMed]
- Ulluwishewa, D.; Anderson, R.C.; McNabb, W.C.; Moughan, P.J.; Wells, J.M.; Roy, N.C. Regulation of Tight Junction Permeability by Intestinal Bacteria and Dietary Components. J. Nutr. 2011, 141, 769–776. [Google Scholar] [CrossRef] [PubMed]
- Bedi, S.; Richardson, T.M.; Jia, B.; Saab, H.; Brinkman, F.S.L.; Westley, M. Similarities between Bacterial GAD and Human GAD65: Implications in Gut Mediated Autoimmune Type 1 Diabetes. PLoS ONE 2022, 17, e0261103. [Google Scholar] [CrossRef] [PubMed]
- Jamshidi, P.; Hasanzadeh, S.; Tahvildari, A.; Farsi, Y.; Arbabi, M.; Mota, J.F.; Sechi, L.A.; Nasiri, M.J. Is There Any Association between Gut Microbiota and Type 1 Diabetes? A Systematic Review. Gut Pathog. 2019, 11, 49. [Google Scholar] [CrossRef]
- Altindis, E.; Vomund, A.N.; Chow, I.-T.; Damasio, M.; Kwok, W.; Unanue, E.R.; Kahn, C.R. Identification of Cross Reactive Insulin Immunogenic Epitopes from Commensal Gut Microbes. Diabetes 2018, 67, 95-OR. [Google Scholar] [CrossRef]
- Cole, D.K.; Bulek, A.M.; Dolton, G.; Schauenberg, A.J.; Szomolay, B.; Rittase, W.; Trimby, A.; Jothikumar, P.; Fuller, A.; Skowera, A.; et al. Hotspot Autoimmune T Cell Receptor Binding Underlies Pathogen and Insulin Peptide Cross-Reactivity. J. Clin. Investig. 2016, 126, 2191–2204. [Google Scholar] [CrossRef] [PubMed]
- Girdhar, K.; Huang, Q.; Chow, I.-T.; Vatanen, T.; Brady, C.; Raisingani, A.; Autissier, P.; Atkinson, M.A.; Kwok, W.W.; Kahn, C.R.; et al. A Gut Microbial Peptide and Molecular Mimicry in the Pathogenesis of Type 1 Diabetes. Proc. Natl. Acad. Sci. USA 2022, 119, e2120028119. [Google Scholar] [CrossRef]
- Hill, J.H.; Franzosa, E.A.; Huttenhower, C.; Guillemin, K. A Conserved Bacterial Protein Induces Pancreatic Beta Cell Expansion during Zebrafish Development. Elife 2016, 5, e20145. [Google Scholar] [CrossRef] [PubMed]
- Giongo, A.; Gano, K.A.; Crabb, D.B.; Mukherjee, N.; Novelo, L.L.; Casella, G.; Drew, J.C.; Ilonen, J.; Knip, M.; Hyöty, H.; et al. Toward Defining the Autoimmune Microbiome for Type 1 Diabetes. ISME J. 2011, 5, 82–91. [Google Scholar] [CrossRef] [PubMed]
- de Goffau, M.C.; Luopajärvi, K.; Knip, M.; Ilonen, J.; Ruohtula, T.; Härkönen, T.; Orivuori, L.; Hakala, S.; Welling, G.W.; Harmsen, H.J.; et al. Fecal Microbiota Composition Differs between Children with β-Cell Autoimmunity and Those without. Diabetes 2013, 62, 1238–1244. [Google Scholar] [CrossRef] [PubMed]
- Vatanen, T.; Franzosa, E.A.; Schwager, R.; Tripathi, S.; Arthur, T.D.; Vehik, K.; Lernmark, Å.; Hagopian, W.A.; Rewers, M.J.; She, J.-X.; et al. The Human Gut Microbiome in Early-Onset Type 1 Diabetes from the TEDDY Study. Nature 2018, 562, 589–594. [Google Scholar] [CrossRef] [PubMed]
- Murri, M.; Leiva, I.; Gomez-Zumaquero, J.M.; Tinahones, F.J.; Cardona, F.; Soriguer, F.; Queipo-Ortuño, M.I. Gut Microbiota in Children with Type 1 Diabetes Differs from That in Healthy Children: A Case-Control Study. BMC Med. 2013, 11, 46. [Google Scholar] [CrossRef]
- Alkanani, A.K.; Hara, N.; Gottlieb, P.A.; Ir, D.; Robertson, C.E.; Wagner, B.D.; Frank, D.N.; Zipris, D. Alterations in Intestinal Microbiota Correlate with Susceptibility to Type 1 Diabetes. Diabetes 2015, 64, 3510–3520. [Google Scholar] [CrossRef] [PubMed]
- Kostic, A.D.; Gevers, D.; Siljander, H.; Vatanen, T.; Hyötyläinen, T.; Hämäläinen, A.-M.; Peet, A.; Tillmann, V.; Pöhö, P.; Mattila, I.; et al. The Dynamics of the Human Infant Gut Microbiome in Development and in Progression toward Type 1 Diabetes. Cell Host Microbe 2015, 17, 260–273. [Google Scholar] [CrossRef]
- Zhang, L.; Jonscher, K.R.; Zhang, Z.; Xiong, Y.; Mueller, R.S.; Friedman, J.E.; Pan, C. Islet Autoantibody Seroconversion in Type-1 Diabetes Is Associated with Metagenome-Assembled Genomes in Infant Gut Microbiomes. Nat. Commun. 2022, 13, 3551. [Google Scholar] [CrossRef] [PubMed]
- Biassoni, R.; Di Marco, E.; Squillario, M.; Barla, A.; Piccolo, G.; Ugolotti, E.; Gatti, C.; Minuto, N.; Patti, G.; Maghnie, M.; et al. Gut Microbiota in T1DM-Onset Pediatric Patients: Machine-Learning Algorithms to Classify Microorganisms as Disease Linked. J. Clin. Endocrinol. Metab. 2020, 105, dgaa407. [Google Scholar] [CrossRef] [PubMed]
- Davis-Richardson, A.G.; Triplett, E.W. A Model for the Role of Gut Bacteria in the Development of Autoimmunity for Type 1 Diabetes. Diabetologia 2015, 58, 1386–1393. [Google Scholar] [CrossRef] [PubMed]
- Del Chierico, F.; Conta, G.; Matteoli, M.C.; Fierabracci, A.; Reddel, S.; Macari, G.; Gardini, S.; Guarrasi, V.; Levi Mortera, S.; Marzano, V.; et al. Gut Microbiota Functional Traits, Blood PH, and Anti-GAD Antibodies Concur in the Clinical Characterization of T1D at Onset. Int. J. Mol. Sci. 2022, 23, 10256. [Google Scholar] [CrossRef] [PubMed]
- van Heck, J.I.P.; Gacesa, R.; Stienstra, R.; Fu, J.; Zhernakova, A.; Harmsen, H.J.M.; Weersma, R.K.; Joosten, L.A.B.; Tack, C.J. The Gut Microbiome Composition Is Altered in Long-Standing Type 1 Diabetes and Associates with Glycemic Control and Disease-Related Complications. Diabetes Care 2022, 45, 2084–2094. [Google Scholar] [CrossRef]
- Brown, C.T.; Davis-Richardson, A.G.; Giongo, A.; Gano, K.A.; Crabb, D.B.; Mukherjee, N.; Casella, G.; Drew, J.C.; Ilonen, J.; Knip, M.; et al. Gut Microbiome Metagenomics Analysis Suggests a Functional Model for the Development of Autoimmunity for Type 1 Diabetes. PLoS ONE 2011, 6, e25792. [Google Scholar] [CrossRef]
- de Goffau, M.C.; Fuentes, S.; van den Bogert, B.; Honkanen, H.; de Vos, W.M.; Welling, G.W.; Hyöty, H.; Harmsen, H.J.M. Aberrant Gut Microbiota Composition at the Onset of Type 1 Diabetes in Young Children. Diabetologia 2014, 57, 1569–1577. [Google Scholar] [CrossRef] [PubMed]
- de Groot, P.F.; Belzer, C.; Aydin, Ö.; Levin, E.; Levels, J.H.; Aalvink, S.; Boot, F.; Holleman, F.; van Raalte, D.H.; Scheithauer, T.P.; et al. Distinct Fecal and Oral Microbiota Composition in Human Type 1 Diabetes, an Observational Study. PLoS ONE 2017, 12, e0188475. [Google Scholar] [CrossRef]
- Chambers, E.S.; Preston, T.; Frost, G.; Morrison, D.J. Role of Gut Microbiota-Generated Short-Chain Fatty Acids in Metabolic and Cardiovascular Health. Curr. Nutr. Rep. 2018, 7, 198–206. [Google Scholar] [CrossRef]
- Parada Venegas, D.; De la Fuente, M.K.; Landskron, G.; González, M.J.; Quera, R.; Dijkstra, G.; Harmsen, H.J.M.; Faber, K.N.; Hermoso, M.A. Short Chain Fatty Acids (SCFAs)-Mediated Gut Epithelial and Immune Regulation and Its Relevance for Inflammatory Bowel Diseases. Front. Immunol. 2019, 10, 277. [Google Scholar] [CrossRef]
- Silva, Y.P.; Bernardi, A.; Frozza, R.L. The Role of Short-Chain Fatty Acids From Gut Microbiota in Gut-Brain Communication. Front. Endocrinol. (Lausanne) 2020, 11, 25. [Google Scholar] [CrossRef]
- Liu, H.; Wang, J.; He, T.; Becker, S.; Zhang, G.; Li, D.; Ma, X. Butyrate: A Double-Edged Sword for Health? Adv. Nutr. 2018, 9, 21–29. [Google Scholar] [CrossRef]
- Lopetuso, L.R.; Scaldaferri, F.; Petito, V.; Gasbarrini, A. Commensal Clostridia: Leading Players in the Maintenance of Gut Homeostasis. Gut Pathog. 2013, 5, 23. [Google Scholar] [CrossRef] [PubMed]
- Louis, P.; Flint, H.J. Formation of Propionate and Butyrate by the Human Colonic Microbiota. Environ. Microbiol. 2017, 19, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Jacob, N.; Jaiswal, S.; Maheshwari, D.; Nallabelli, N.; Khatri, N.; Bhatia, A.; Bal, A.; Malik, V.; Verma, S.; Kumar, R.; et al. Butyrate Induced Tregs Are Capable of Migration from the GALT to the Pancreas to Restore Immunological Tolerance during Type-1 Diabetes. Sci. Rep. 2020, 10, 19120. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, A.; Krieg, R.; Massey, H.D.; Carl, D.; Ghosh, S.; Gehr, T.W.B.; Ghosh, S.S. Sodium Butyrate Ameliorates Insulin Resistance and Renal Failure in CKD Rats by Modulating Intestinal Permeability and Mucin Expression. Nephrol. Dial Transpl. 2019, 34, 783–794. [Google Scholar] [CrossRef]
- Ziegler, A.-G.; Arnolds, S.; Kölln, A.; Achenbach, P.; Berner, R.; Bonifacio, E.; Casteels, K.; Elding Larsson, H.; Gündert, M.; Hasford, J.; et al. Supplementation with Bifidobacterium Longum Subspecies Infantis EVC001 for Mitigation of Type 1 Diabetes Autoimmunity: The GPPAD-SINT1A Randomised Controlled Trial Protocol. BMJ Open 2021, 11, e052449. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Kumar, R.; Rohilla, L.; Jacob, N.; Yadav, J.; Sachdeva, N. A High Potency Multi-strain Probiotic Improves Glycemic Control in Children with New-onset Type 1 Diabetes Mellitus: A Randomized, Double-blind, and Placebo-controlled Pilot Study. Pediatr. Diabetes 2021, 22, 1014–1022. [Google Scholar] [CrossRef] [PubMed]
- de Groot, P.; Nikolic, T.; Pellegrini, S.; Sordi, V.; Imangaliyev, S.; Rampanelli, E.; Hanssen, N.; Attaye, I.; Bakker, G.; Duinkerken, G.; et al. Faecal Microbiota Transplantation Halts Progression of Human New-Onset Type 1 Diabetes in a Randomised Controlled Trial. Gut 2021, 70, 92–105. [Google Scholar] [CrossRef] [PubMed]
- Groele, L.; Szajewska, H.; Szalecki, M.; Świderska, J.; Wysocka-Mincewicz, M.; Ochocińska, A.; Stelmaszczyk-Emmel, A.; Demkow, U.; Szypowska, A. Lack of Effect of Lactobacillus Rhamnosus GG and Bifidobacterium Lactis Bb12 on Beta-Cell Function in Children with Newly Diagnosed Type 1 Diabetes: A Randomised Controlled Trial. BMJ Open Diab. Res. Care 2021, 9, e001523. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Del Chierico, F.; Rapini, N.; Deodati, A.; Matteoli, M.C.; Cianfarani, S.; Putignani, L. Pathophysiology of Type 1 Diabetes and Gut Microbiota Role. Int. J. Mol. Sci. 2022, 23, 14650. https://doi.org/10.3390/ijms232314650
Del Chierico F, Rapini N, Deodati A, Matteoli MC, Cianfarani S, Putignani L. Pathophysiology of Type 1 Diabetes and Gut Microbiota Role. International Journal of Molecular Sciences. 2022; 23(23):14650. https://doi.org/10.3390/ijms232314650
Chicago/Turabian StyleDel Chierico, Federica, Novella Rapini, Annalisa Deodati, Maria Cristina Matteoli, Stefano Cianfarani, and Lorenza Putignani. 2022. "Pathophysiology of Type 1 Diabetes and Gut Microbiota Role" International Journal of Molecular Sciences 23, no. 23: 14650. https://doi.org/10.3390/ijms232314650
APA StyleDel Chierico, F., Rapini, N., Deodati, A., Matteoli, M. C., Cianfarani, S., & Putignani, L. (2022). Pathophysiology of Type 1 Diabetes and Gut Microbiota Role. International Journal of Molecular Sciences, 23(23), 14650. https://doi.org/10.3390/ijms232314650





