NGF and the Male Reproductive System: Potential Clinical Applications in Infertility
Abstract
1. Introduction
1.1. Male Infertility
1.1.1. Epidemiology
1.1.2. Etiology
1.1.3. Treatments
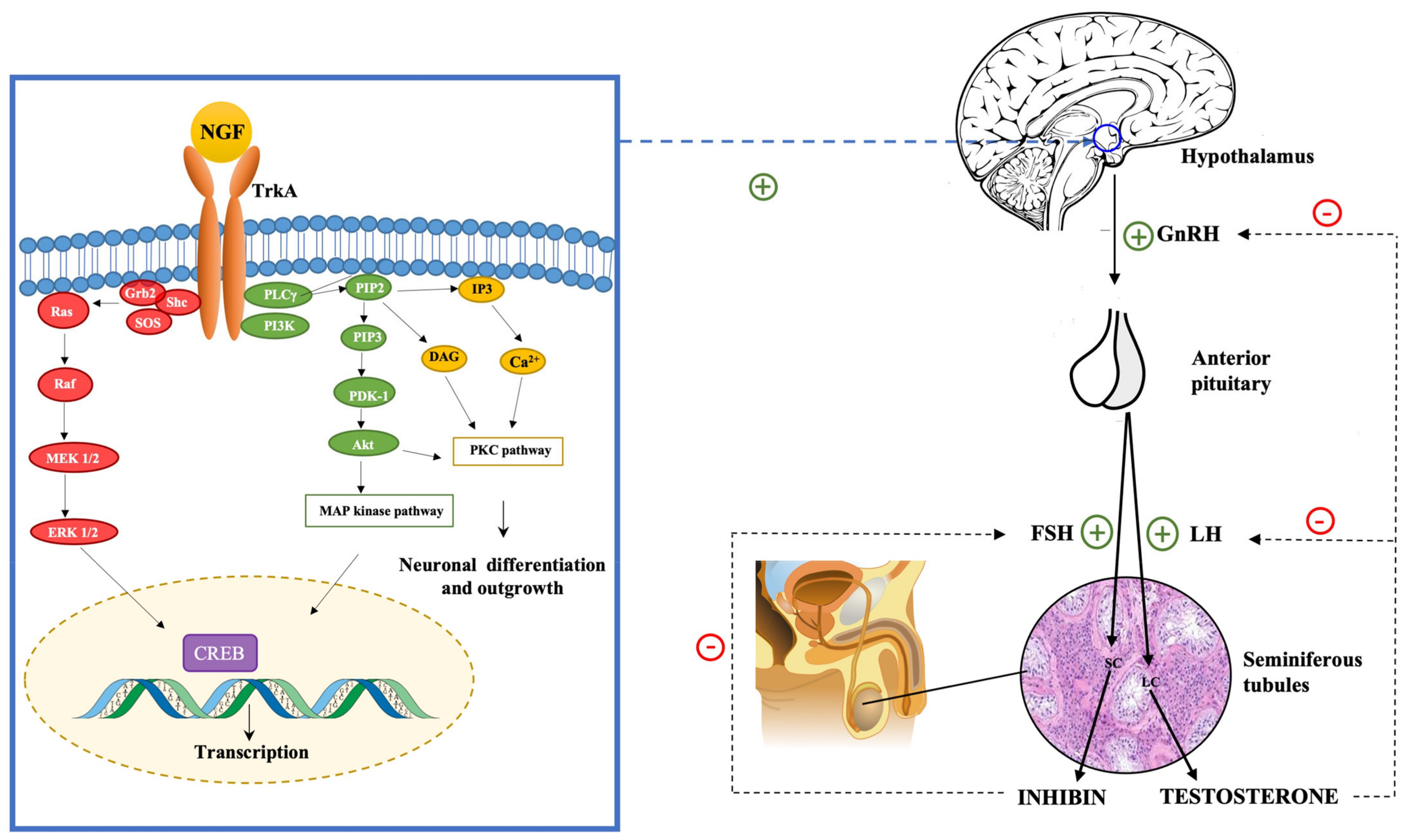
2. NGF
2.1. Neurotrophins
2.2. NGF Expression in the Reproductive System
3. NGF Specific Functions in the Male Reproductive System
3.1. NGF’s Role in Testis Morphogenesis
3.2. NGF Role in Spermatogenesis
3.3. The Impact of NGF on the Hypothalamus-Pituitary-Gonadal (HPG) Axis
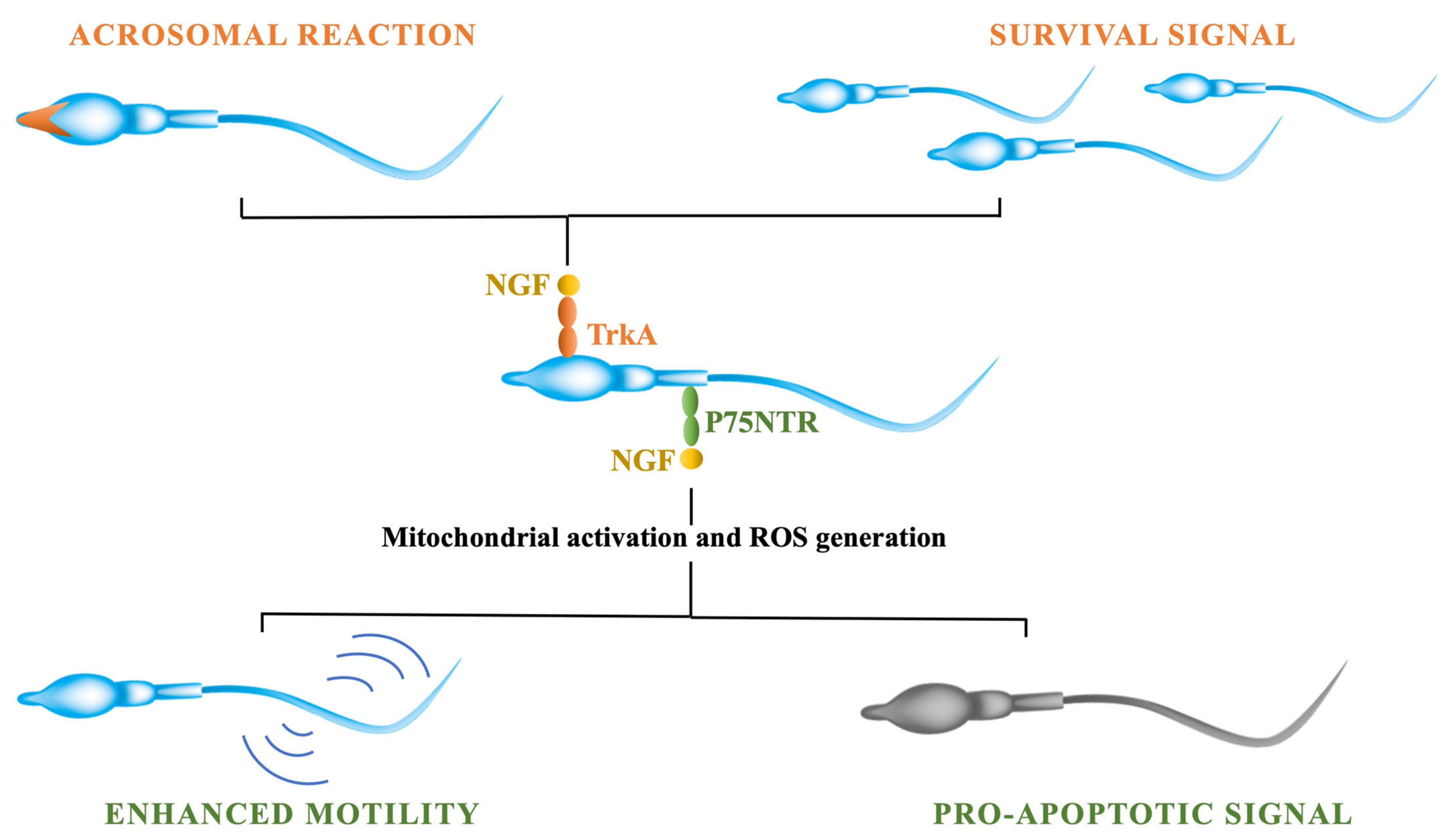
3.4. NGF Effects on Sperm Traits
4. New Therapeutical Opportunities
4.1. The Potential Role of NGF in Male Infertility Treatment
4.2. The Effect of NGF Addition to Cryopreserved Sperm
4.3. NGF Supplementation in Assisted Reproduction
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ABP | androgen-binding protein |
| AKT | protein kinase B |
| ARTs | assisted reproductive technologies |
| BDNF | brain-derived neurotrophic factor |
| CAVD | congenital absence of the vas deferens |
| CML | chronic myeloid leukemia |
| CREB | c-AMP-response element binding protein |
| DAG | diacylglycerol |
| EdU | 5-ethynyl-2′-deoxyuridine |
| ERK | extracellular signal-regulated kinase |
| FSH | follicle-stimulating hormone |
| GnRH | gonadotropin-releasing hormone |
| GRB2 | growth factor receptor-bound protein 2 |
| HH | hypogonadotropic hypogonadism |
| HPG | hypothalamus–pituitary–gonadal |
| ICSI | intracytoplasmic sperm injection |
| ILCs | immature Leydig cells |
| IP3 | inositol triphosphate |
| IUI | intrauterine insemination |
| IVF | in vitro fertilization |
| LC | Leydig cell |
| LH | luteinizing hormone |
| MAPK | mitogen-activated protein kinase |
| MEK | mitogen-activated protein kinase |
| NGF | nerve growth factor |
| NO | nitric oxide |
| NT-3 | neurotrophin-3 |
| NT-4 | neurotrophin-4 |
| OA | obstructive azoospermia |
| PADAM | partial androgen deficiency of the aging male |
| PDK | 3-phosphoinositide-dependent kinase 1 |
| PI3K | phosphoinositide 3-kinase |
| PIP2 | phosphatidylinositol 4,5-bisphosphate |
| PIP3 | phosphatidylinositol 3,4,5-trisphosphate |
| PKC | protein kinase C |
| PLC-γ | phosphoinositide-specific phospholipase C-γ |
| PLCs | progenitor Leydig cells |
| PM | progressive motility |
| RAF | rapidly accelerated fibrosarcoma |
| ROS | reactive oxygen species |
| SC | Sertoli cell |
| SERMs | selective estrogen receptor modulators |
| SHC | src honology and collagen |
| SLCs | stem Leydig cells |
| TST | testosterone supplementation therapy |
| WHO | World Health Organization |
References
- Zegers-Hochschild, F.; Adamson, G.D.; Dyer, S.; Racowsky, C.; de Mouzon, J.; Sokol, R.; Rienzi, L.; Sunde, A.; Schmidt, L.; Cooke, I.D.; et al. The International Glossary on Infertility and Fertility Care, 2017. Fertil. Steril. 2017, 108, 393–406. [Google Scholar] [CrossRef] [PubMed]
- Fainberg, J.; Kashanian, J.A. Recent advances in understanding and managing male infertility. F1000Research 2019, 8, 670. [Google Scholar] [CrossRef] [PubMed]
- Vander Borght, M.; Wyns, C. Fertility and infertility: Definition and epidemiology. Clin. Biochem. 2018, 62, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Thoma, M.E.; McLain, A.; Louis, J.F.; King, R.B.; Trumble, A.C.; Sundaram, R.; Louis, G.B. Prevalence of infertility in the United States as estimated by the current duration approach and a traditional constructed approach. Fertil. Steril. 2013, 99, 1324–1331.e1. [Google Scholar] [CrossRef]
- Bak, C.W.; Seok, H.H.; Song, S.H.; Kim, E.S.; Her, Y.S.; Yoon, T.K. Hormonal imbalances and psychological scars left behind in infertile men. J. Androl. 2012, 33, 181–189. [Google Scholar] [CrossRef]
- Wu, A.K.; Elliott, P.; Katz, P.P.; Smith, J.F. Time costs of fertility care: The hidden hardship of building a family. Fertil. Steril. 2013, 99, 2025–2030. [Google Scholar] [CrossRef]
- Dimitriadis, F.; Adonakis, G.; Kaponis, A.; Mamoulakis, C.; Takenaka, A.; Sofikitis, N. Pre-Testicular, Testicular, and Post-Testicular Causes of Male Infertility. In Endocrinology of the Testis and Male Reproduction; Simoni, M., Huhtaniemi, I., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 1–47. [Google Scholar] [CrossRef]
- Lenzi, A.; Balercia, G.; Bellastella, A.; Colao, A.; Fabbri, A.; Foresta, C.; Galdiero, M.; Gandini, L.; Krausz, C.; Lombardi, G.; et al. Epidemiology; diagnosis, and treatment of male hypogonadotropic hypogonadism. J. Endocrinol. Investig. 2009, 32, 934–938. [Google Scholar] [CrossRef]
- Huhtaniemi, I.; Alevizaki, M. Mutations along the hypothalamic-pituitary-gonadal axis affecting male reproduction. Reprod. Biomed. Online 2007, 15, 622–632. [Google Scholar] [CrossRef]
- Bianco, S.D.C.; Kaiser, U.B. The genetic and molecular basis of idiopathic hypogonadotropic hypogonadism. Nat. Rev. Endocrinol. 2009, 5, 569–576. [Google Scholar] [CrossRef]
- Whitcomb, R.W.; Crowley, W.F. Male hypogonadotropic hypogonadism. Endocrinol. Metab. Clin. N. Am. 1993, 22, 125–143. [Google Scholar] [CrossRef]
- Fraietta, R.; Zylberstejn, D.S.; Esteves, S.C. Hypogonadotropic hypogonadism revisited. Clinics 2013, 68 (Suppl. S1), 81–88. [Google Scholar] [CrossRef]
- Aust, T.R.; Lewis-Jones, D.I. Retrograde ejaculation and male infertility. Hosp. Med. 2004, 65, 361–364. [Google Scholar] [CrossRef] [PubMed]
- Kondoh, N. Ejaculatory dysfunction as a cause of infertility. Reprod. Med. Biol. 2012, 11, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Lotti, F.; Maggi, M. Sexual dysfunction and male infertility. Nat. Rev. Urol. 2018, 15, 287–307. [Google Scholar] [CrossRef] [PubMed]
- Wiser, H.J.; Sandlow, J.; Köhler, T.S. Causes of male infertility. In Male Infertility; Parekattil, S., Agarwal, A., Eds.; Springer: New York, NY, USA, 2012; pp. 3–14. [Google Scholar] [CrossRef]
- Wong, T.W.; Straus, F.H.; Jones, T.M.; Warner, N.E. Pathological aspects of the infertile testis. Urol. Clin. N. Am. 1978, 5, 503–530. [Google Scholar] [CrossRef]
- Cocuzza, M.; Alvarenga, C.; Pagani, R. The epidemiology and etiology of azoospermia. Clinics 2013, 68, 15–26. [Google Scholar] [CrossRef]
- Merino, G.; Carranza-Lira, S.; Murrieta, S.; Rodriguez, L.; Cuevas, E.; Moran, C. Bacterial infection and semen characteristics in infertile men. Syst. Biol. Reprod. Med. 1995, 35, 43–47. [Google Scholar] [CrossRef]
- Diemer, T.; Huwe, P.; Ludwig, M.; Hauck, E.W.; Weidner, W. Urogenital infection and sperm motility. Andrologia 2003, 35, 283–287. [Google Scholar] [CrossRef]
- Krausz, C.; Mills, C.; Rogers, S.; Tan, S.L.; Aitken, R.J. Stimulation of oxidant generation by human sperm suspensions using phorbol esters and formyl peptides: Relationships with motility and fertilization in vitro. Fertil. Steril. 1994, 62, 599–605. [Google Scholar] [CrossRef]
- Restrepo, B.; Cardona Maya, W. Antisperm antibodies and fertility asociation. Actas Urol. Esp. 2013, 37, 571–578. [Google Scholar] [CrossRef]
- Bieth, E.; Hamdi, S.M.; Mieusset, R. Genetics of the congenital absence of the vas deferens. Hum. Genet. 2021, 140, 59–76. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Minhas, S.; Dhillo, W.S.; Jayasena, C.N. Male infertility due to testicular disorders. J. Clin. Endocrinol. Metab. 2021, 106, E442–E459. [Google Scholar] [CrossRef] [PubMed]
- Villumsen, A.L.; Zachau-Christiansen, B. Spontaneous alterations in position of the testes. Arch. Dis. Child. 1966, 41, 198–200. [Google Scholar] [CrossRef]
- Wohlfahrt-Veje, C.; Boisen, K.A.; Boas, M.; Damgaard, I.N.; Kai, C.M.; Schmidt, I.M.; Chellakooty, M.; Suomi, A.-M.; Toppari, J.; Skakkebaek, N.E.; et al. Acquired cryptorchidism is frequent in infancy and childhood. Int. J. Androl. 2009, 32, 423–428. [Google Scholar] [CrossRef] [PubMed]
- Main, K.M.; Skakkebæk, N.E.; Virtanen, H.E.; Toppari, J. Genital anomalies in boys and the environment. Best Pract. Res. Clin. Endocrinol. Metab. 2010, 24, 279–289. [Google Scholar] [CrossRef] [PubMed]
- Kantartzi, P.D.; Goulis, C.D.; Goulis, G.D.; Papadimas, I. Male infertility and varicocele: Myths and reality. Hippokratia 2007, 11, 99–104. [Google Scholar]
- Ferlin, A.; Arredi, B.; Foresta, C. Genetic causes of male infertility. Reprod. Toxicol. 2006, 22, 133–141. [Google Scholar] [CrossRef]
- Linn, E.; Ghanem, L.; Bhakta, H.; Greer, C.; Avella, M. Genes Regulating Spermatogenesis and Sperm Function Associated With Rare Disorders. Front. Cell Dev. Biol. 2021, 9, 634536. [Google Scholar] [CrossRef]
- Leaver, R.B. Male infertility: An overview of causes and treatment options. Br. J. Nurs. 2016, 25, S35–S40. [Google Scholar] [CrossRef]
- Kamischke, A.; Nieschlag, E. Analysis of medical treatment of male infertility. Hum. Reprod. 1999, 14, 1–23. [Google Scholar] [CrossRef]
- Dabaja, A.A.; Schlegel, P.N. Medical treatment of male infertility. Transl. Androl. Urol. 2014, 3, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Nachtigall, L.B.; Boepple, P.A.; Pralong, F.P.; Crowley, W.F. Adult-Onset Idiopathic Hypogonadotropic Hypogonadism—A Treatable Form of Male Infertility. N. Engl. J. Med. 1997, 336, 410–415. [Google Scholar] [CrossRef] [PubMed]
- Conn, P.M.; Hansen, J.R. Gonadotropin-releasing hormone and its analogs. Iowa Med. 1986, 76, 372–377. [Google Scholar] [CrossRef] [PubMed]
- Zitzmann, M.; Nieschlag, E. Hormone substitution in male hypogonadism. Mol. Cell. Endocrinol. 2000, 161, 73–88. [Google Scholar] [CrossRef]
- Blumenfeld, Z.; Frisch, L.; Conn, P.M. Gonadotropin-releasing hormone (GnRH) antibodies formation in hypogonadotropic azoospermic men treated with pulsatile GnRH-Diagnosis and possible alternative treatment. Fertil. Steril. 1988, 50, 622–629. [Google Scholar] [CrossRef]
- Wei, C.; Long, G.; Zhang, Y.; Wang, T.; Wang, S.; Liu, J.; Ma, D.; Liu, X. Spermatogenesis of male patients with congenital hypogonadotropic hypogonadism receiving pulsatile gonadotropin-releasing hormone therapy versus gonadotropin therapy: A systematic review and meta-analysis. World J. Mens Health 2020, 38, 654–665. [Google Scholar] [CrossRef]
- Liu, L.; Banks, S.M.; Barnes, K.M.; Sherins, R.J. Two-year comparison of testicular responses to pulsatile gonadotropin-releasing hormone and exogenous gonadotropins from the inception of therapy in men with isolated hypogonadotropic hypogonadism. J. Clin. Endocrinol. Metab. 1988, 67, 1140–1145. [Google Scholar] [CrossRef]
- Madhukar, D.; Rajender, S. Hormonal treatment of male infertility: Promise and pitfalls. J. Androl. 2009, 30, 95–112. [Google Scholar] [CrossRef]
- Burgués, S.; Calderón, M.D. Subcutaneous self-administration of highly purified follicle stimulating hormone and human chorionic gonadotrophin for the treatment of male hypogonadotrophic hypogonadism. Hum. Reprod. 1997, 12, 980–986. [Google Scholar] [CrossRef]
- Dabbous, Z.; Atkin, S.L. Hyperprolactinaemia in male infertility: Clinical case scenarios. Arab J. Urol. 2018, 16, 44–52. [Google Scholar] [CrossRef]
- Cannarella, R.; Condorelli, R.A.; Mongioì, L.M.; Barbagallo, F.; Calogero, A.E.; La Vignera, S. Effects of the selective estrogen receptor modulators for the treatment of male infertility: A systematic review and meta-analysis. Expert Opin. Pharmacother. 2019, 20, 1517–1525. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, S.R.; Siddhanti, S.; Ciaccia, A.V.; Plouffe, L. A pharmacological review of selective oestrogen receptor modulators. Hum. Reprod. Update 2000, 6, 212–224. [Google Scholar] [CrossRef] [PubMed]
- Velasquez, M.; Tanrikut, C. Surgical management of male infertility: An update. Transl. Androl. Urol. 2014, 3, 64–76. [Google Scholar] [CrossRef] [PubMed]
- Schiff, J.D.; Ramírez, M.L.; Bar-Chama, N. Medical and Surgical Management Male Infertility. Endocrinol. Metab. Clin. N. Am. 2007, 36, 313–331. [Google Scholar] [CrossRef] [PubMed]
- Tournaye, H. Male factor infertility and ART. Asian J. Androl. 2012, 14, 103–108. [Google Scholar] [CrossRef]
- Duran, H.E.; Morshedi, M.; Kruger, T.; Oehninger, S. Intrauterine insemination: A systematic review on determinants of success. Hum. Reprod. Update 2002, 8, 373–384. [Google Scholar] [CrossRef]
- Huang, J.Y.J.; Rosenwaks, Z. Assisted reproductive techniques. Methods Mol. Biol. 2014, 1154, 171–231. [Google Scholar] [CrossRef]
- Aboulghar, M.; Baird, D.T.; Collins, J.; Evers, J.L.H.; Fauser, B.C.J.M.; Lambalk, C.B.; Somigliana, E.; Sunde, A.; Tarlatzis, B.; Crosignani, P.G.; et al. Intrauterine insemination. Hum. Reprod. Update 2009, 15, 265–277. [Google Scholar] [CrossRef]
- Keck, C.; Gerber-Schafer, C.; Wilhelm, C.; Vogelgesang, D.; Breckwoldt, M. Intrauterine insemination for treatment of male infertility. Int. J. Androl. Suppl. 1997, 20, 55–64. [Google Scholar]
- Hasler, J.F.; Barfield, J.P. Vitro Fertilization. In Bovine Reproduction; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2021; pp. 1124–1141. [Google Scholar] [CrossRef]
- Palermo, G.D.; Neri, Q.V.; Hariprashad, J.J.; Davis, O.K.; Veeck, L.L.; Rosenwaks, Z. ICSI and its outcome. Semin. Reprod. Med. 2000, 18, 161–169. [Google Scholar] [CrossRef]
- O’Neill, C.L.; Chow, S.; Rosenwaks, Z.; Palermo, G.D. Development of ICSI. Reproduction 2018, 156, F51–F58. [Google Scholar] [CrossRef] [PubMed]
- Justice, T.; Christensen, G. Sperm Cryopreservation Methods. Methods Mol. Biol. 2013, 927, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Skaper, S.D. Neurotrophic Factors: An Overview. Methods Mol. Biol. 2018, 1727, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Bothwell, M. Keeping track of neurotrophin receptors. Cell 1991, 65, 915–918. [Google Scholar] [CrossRef]
- Chao, M.V. Neurotrophins and their receptors: A convergence point for many signalling pathways. Nat. Rev. Neurosci. 2003, 4, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Casaccia-Bonnefil, P.; Carter, B.D.; Dobrowsky, R.T.; Chao, M.V. Death of oligodendrocytes mediated by the interaction of nerve growth factor with its receptor p75. Nature 1996, 383, 716–719. [Google Scholar] [CrossRef]
- Friedman, W.J.; Greene, L.A. Neurotrophin signaling via Trks and p75. Exp. Cell Res. 1999, 253, 131–142. [Google Scholar] [CrossRef]
- Pearson, G.; Robinson, F.; Beers Gibson, T.; Xu, B.E.; Karandikar, M.; Berman, K.; Cobb, M.H. Mitogen-Activated Protein (MAP) Kinase Pathways: Regulation and Physiological Functions. Endocr. Rev. 2001, 22, 153–183. [Google Scholar] [CrossRef]
- Yuan, X.-B.; Jin, M.; Xu, X.; Song, Y.-Q.; Wu, C.-P.; Poo, M.-M.; Duan, S. Signalling and crosstalk of Rho GTPases in mediating axon guidance. Nat. Cell Biol. 2003, 5, 38–45. [Google Scholar] [CrossRef]
- Rose, C.R.; Blum, R.; Pichler, B.; Lepier, A.; Kafitz, K.W.; Konnerth, A. Truncated TrkB-T1 mediates neurotrophin-evoked calcium signalling in glia cells. Nature 2003, 426, 74–78. [Google Scholar] [CrossRef]
- Toledo-Aral, J.J.; Brehm, P.; Halegoua, S.; Mandel, G. A single pulse of nerve growth factor triggers long-term neuronal excitability through sodium channel gene induction. Neuron 1995, 14, 607–611. [Google Scholar] [CrossRef]
- Minichiello, L.; Calella, A.M.; Medina, D.L.; Bonhoeffer, T.; Klein, R.; Korte, M. Mechanism of TrkB-mediated hippocampal long-term potentiation. Neuron 2002, 36, 121–137. [Google Scholar] [CrossRef]
- Nahum, L.H. the Nerve Growth Factor (Ngf). Conn. Med. 1964, 28, 508–512. [Google Scholar] [PubMed]
- Fiore, M.; Amendola, T.; Triaca, V.; Tirassa, P.; Alleva, E.; Aloe, L. Agonistic encounters in aged male mouse potentiate the expression of endogenous brain NGF and BDNF: Possible implication for brain progenitor cells’ activation. Eur. J. Neurosci. 2003, 17, 1455–1464. [Google Scholar] [CrossRef][Green Version]
- Ciafrè, S.; Ferraguti, G.; Tirassa, P.; Iannitelli, A.; Ralli, M.; Greco, A.; Chaldakov, G.N.; Rosso, P.; Fico, E.; Messina, M.P.; et al. Nerve growth factor in the psychiatric brain. Riv. Psichiatr. 2020, 55, 4–15. [Google Scholar] [CrossRef]
- Wood, S.J.; Pritchard, J.; Sofroniew, M.V. Re-expression of Nerve Growth Factor Receptor after Axonal Injury Recapitulates a Developmental Event in Motor Neurons: Differential Regulation when Regeneration is Allowed or Prevented. Eur. J. Neurosci. 1990, 2, 650–657. [Google Scholar] [CrossRef]
- Verge, V.M.K.; Richardson, P.M.; Benoit, R.; Riopelle, R.J. Histochemical characterization of sensory neurons with high-affinity receptors for nerve growth factor. J. Neurocytol. 1989, 18, 583–591. [Google Scholar] [CrossRef]
- Ruit, K.G.; Osborne, P.A.; Schmidt, R.E.; Johnson, E.M.; Snider, W.D. Nerve growth factor regulates sympathetic ganglion cell morphology and survival in the adult mouse. J. Neurosci. 1990, 10, 2412–2419. [Google Scholar] [CrossRef]
- Heumann, R.; Lindholm, D.; Bandtlow, C.; Meyer, M.; Radeke, M.J.; Misko, T.P.; Shooter, E.; Thoenen, H. Differential regulation of mRNA encoding nerve growth factor and its receptor in rat sciatic nerve during development, degeneration, and regeneration: Role of macrophages. Proc. Natl. Acad. Sci. USA 1987, 84, 8735–8739. [Google Scholar] [CrossRef]
- Chaldakov, G.N.; Stankulov, I.S.; Fiore, M.; Ghenev, P.I.; Aloe, L. Nerve growth factor levels and mast cell distribution in human coronary atherosclerosis. Atherosclerosis 2001, 159, 57–66. [Google Scholar] [CrossRef]
- Aloe, L.; Alleva, E.; Fiore, M. Stress and nerve growth factor: Findings in animal models and humans. Pharmacol. Biochem. Behav. 2002, 73, 159–166. [Google Scholar] [CrossRef]
- Schulte-Herbruggen, O.; Braun, A.; Rochlitzer, S.; Jockers-Scherubl, M.C.; Hellweg, R. Neurotrophic factors—A tool for therapeutic strategies in neurological, neuropsychiatric and neuroimmunological diseases? Curr. Med. Chem. 2007, 14, 2318–2329. [Google Scholar] [CrossRef]
- Bruscolini, A.; Sacchetti, M.; La Cava, M.; Nebbioso, M.; Iannitelli, A.; Quartini, A.; Lambiase, A.; Ralli, M.; de Virgilio, A.; Greco, A. Quality of life and neuropsychiatric disorders in patients with Graves’ Orbitopathy: Current concepts. Autoimmun. Rev. 2018, 17, 639–643. [Google Scholar] [CrossRef] [PubMed]
- Chaldakov, G.N.; Fiore, M.; Tonchev, A.B.; Aloe, L. Neuroadipology: A novel component of neuroendocrinology. Cell Biol. Int. 2010, 34, 1051–1053. [Google Scholar] [CrossRef] [PubMed]
- Tore, F.; Tonchev, A.; Fiore, M.; Tuncel, N.; Atanassova, P.; Aloe, L.; Chaldakov, G. From Adipose Tissue Protein Secretion to Adipopharmacology of Disease. Immunol. Endocr. Metab. Agents Med. Chem. 2007, 7, 149–155. [Google Scholar] [CrossRef]
- Carito, V.; Ceccanti, M.; Tarani, L.; Ferraguti, G.; Chaldakov, G.N.; Fiore, M. Neurotrophins’ Modulation by Olive Polyphenols. Curr. Med. Chem. 2016, 23, 3189–3197. [Google Scholar] [CrossRef] [PubMed]
- Petrella, C.; Di Certo, M.G.; Gabanella, F.; Barbato, C.; Ceci, F.M.; Greco, A.; Ralli, M.; Polimeni, A.; Angeloni, A.; Severini, C.; et al. Mediterranean Diet, Brain and Muscle: Olive Polyphenols and Resveratrol Protection in Neurodegenerative and Neuromuscular Disorders. Curr. Med. Chem. 2021, 28, 7595–7613. [Google Scholar] [CrossRef]
- Ceci, F.M.; Ferraguti, G.; Petrella, C.; Greco, A.; Tirassa, P.; Iannitelli, A.; Ralli, M.; Vitali, M.; Ceccanti, M.; Chaldakov, G.N.; et al. Nerve Growth Factor, Stress and Diseases. Curr. Med. Chem. 2020, 28, 2943–2959. [Google Scholar] [CrossRef]
- Rosso, P.; Iannitelli, A.; Pacitti, F.; Quartini, A.; Fico, E.; Fiore, M.; Greco, A.; Ralli, M.; Tirassa, P. Vagus nerve stimulation and Neurotrophins: A biological psychiatric perspective. Neurosci. Biobehav. Rev. 2020, 113, 338–353. [Google Scholar] [CrossRef]
- Carito, V.; Ceccanti, M.; Ferraguti, G.; Coccurello, R.; Ciafrè, S.; Tirassa, P.; Fiore, M. NGF and BDNF Alterations by Prenatal Alcohol Exposure. Curr. Neuropharmacol. 2019, 17, 308–317. [Google Scholar] [CrossRef]
- Ciafrè, S.; Ferraguti, G.; Greco, A.; Polimeni, A.; Ralli, M.; Ceci, F.M.; Ceccanti, M.; Fiore, M. Alcohol as an early life stressor: Epigenetics, metabolic, neuroendocrine and neurobehavioral implications. Neurosci. Biobehav. Rev. 2020, 118, 654–668. [Google Scholar] [CrossRef] [PubMed]
- Ceci, F.M.; Ferraguti, G.; Petrella, C.; Greco, A.; Ralli, M.; Iannitelli, A.; Carito, V.; Tirassa, P.; Chaldakov, G.N.; Messina, M.P.; et al. Nerve Growth Factor in Alcohol Use Disorders. Curr. Neuropharmacol. 2020, 19, 45–60. [Google Scholar] [CrossRef] [PubMed]
- Fiore, M.; Triaca, V.; Amendola, T.; Tirassa, P.; Aloe, L. Brain NGF and EGF administration improves passive avoidance response and stimulates brain precursor cells in aged male mice. Physiol. Behav. 2002, 77, 437–443. [Google Scholar] [CrossRef]
- Chaldakov, G.; Fiore, M.; Tonchev, A.; Dimitrov, D.; Pancheva, R.; Rancic, G.; Aloe, L. Homo obesus: A Metabotrophin-Deficient Species. Pharmacol. Nutr. Insight Curr. Pharm. Des. 2007, 13, 2176–2179. [Google Scholar] [CrossRef]
- Miranda, M.; Morici, J.F.; Zanoni, M.B.; Bekinschtein, P. Brain-Derived Neurotrophic Factor: A Key Molecule for Memory in the Healthy and the Pathological Brain. Front. Cell. Neurosci. 2019, 13, 363. [Google Scholar] [CrossRef] [PubMed]
- Otten, U.; Ehrhard, P.; Peck, R. Nerve growth factor induces growth and differentiation of human B lymphocytes. Proc. Natl. Acad. Sci. USA 1989, 86, 10059–10063. [Google Scholar] [CrossRef] [PubMed]
- Torcia, M.; Bracci-Laudiero, L.; Lucibello, M.; Nencioni, L.; Labardi, D.; Rubartelli, A.; Cozzolino, F.; Aloe, L.; Garaci, E. Nerve growth factor is an autocrine survival factor for memory B lymphocytes. Cell 1996, 85, 345–356. [Google Scholar] [CrossRef]
- Kannan, Y.; Usami, K.; Okada, M.; Shimizu, S.; Matsuda, H. Nerve growth factor suppresses apoptosis of murine neutrophils. Biochem. Biophys. Res. Commun. 1992, 186, 1050–1056. [Google Scholar] [CrossRef]
- Horigome, K.; Bullock, E.D.; Johnson, E.M. Effects of nerve growth factor on rat peritoneal mast cells. Survival promotion and immediate-early gene induction. J. Biol. Chem. 1994, 269, 2695–2702. [Google Scholar] [CrossRef]
- Shaoxia, P.U.; Changwei, Q.U.; Zhi, L.I.; Yansen, L.I.; Chunmei, L.I. Expression of nerve growth factor (NGF) and its receptors TrkA and p75 in the reproductive organs of laying hens. Rev. Bras. Cienc. Avic. 2016, 18, 187–192. [Google Scholar] [CrossRef]
- Perrard, M.-H.; Vigier, M.; Damestoy, A.; Chapat, C.; Silandre, D.; Rudkin, B.B.; Durand, P. Β-Nerve Growth Factor Participates in an Auto/Paracrine Pathway of Regulation of the Meiotic Differentiation of Rat Spermatocytes. J. Cell. Physiol. 2007, 210, 51–62. [Google Scholar] [CrossRef] [PubMed]
- Artico, M.; Bronzetti, E.; Saso, L.; Felici, L.M.; D’Ambrosio, A.; Forte, F.; Grande, C.; Ortolani, F. Immunohistochemical profile of some neurotransmitters and neurotrophins in the seminiferous tubules of rats treated by lonidamine. Eur. J. Histochem. 2007, 51, 19–24. [Google Scholar] [PubMed]
- Adams, G.P.; Ratto, M.H. Ovulation-inducing factor in seminal plasma: A review. Anim. Reprod. Sci. 2013, 136, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Jia, L.; Zhang, Y.; Ji, W.; Li, H. Activation of the NGF/TrkA signaling pathway attenuates diabetic erectile dysfunction. Oncotarget 2017, 8, 105692–105702. [Google Scholar] [CrossRef] [PubMed]
- Spinnler, K.; Fröhlich, T.; Arnold, G.J.; Kunz, L.; Mayerhofer, A. Human tryptase cleaves pro-nerve growth factor (Pro-NGF): Hints of local, mast cell-dependent regulation of NGF/PRO-NGF action. J. Biol. Chem. 2011, 286, 31707–31713. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Zhu, P.; Wu, C.; Yu, L.; Zhao, S.; Gu, X. In silico analysis indicates a similar gene expression pattern between human brain and testis. Cytogenet. Genome Res. 2003, 103, 58–62. [Google Scholar] [CrossRef]
- Graves, J.A.M. Review: Sex Chromosome Evolution and the Expression of Sex-Specific Genes in the Placenta. Placenta 2010, 31, S27–S32. [Google Scholar] [CrossRef]
- Ramírez-Reveco, A.; Villarroel-Espíndola, F.; Rodríguez-Gil, J.E.; Concha, I.I. Neuronal signaling repertoire in the mammalian sperm functionality. Biol. Reprod. 2017, 96, 505–524. [Google Scholar] [CrossRef]
- Ayer-LeLievre, C.; Olson, L.; Ebendal, T.; Hallbook, F.; Persson, H. Nerve growth factor mRNA and protein in the testis and epididymis of mouse and rat. Proc. Natl. Acad. Sci. USA 1988, 85, 2628–2632. [Google Scholar] [CrossRef]
- Harper, G.P.; Barde, Y.A.; Burnstock, G.; Carstairs, J.R.; Dennison, M.E.; Suda, K.; Vernon, C.A. Guinea pig prostate is a rich source of nerve growth factor. Nature 1979, 279, 160–162. [Google Scholar] [CrossRef]
- Harper, G.P.; Thoenen, H. The Distribution of Nerve Growth Factor in the Male Sex Organs of Mammals. J. Neurochem. 1980, 34, 893–903. [Google Scholar] [CrossRef] [PubMed]
- Jin, W.Z.; Tanaka, A.; Watanabe, G.; Matsuda, H.; Taya, K. Effect of NGF on the motility and acrosome reaction of golden hamster spermatozoa in vitro. J. Reprod. Dev. 2010, 56, 437–443. [Google Scholar] [CrossRef] [PubMed]
- Jin, W.; Arai, K.Y.; Shimizu, K.; Kojima, C.; Itoh, M.; Watanabe, G.; Taya, K. Cellular localization of NGF and its receptors trkA and p75LNGFR in male reproductive organs of the Japanese monkey, Macaca fuscata fuscata. Endocrine 2006, 29, 155–160. [Google Scholar] [CrossRef]
- Levanti, M.B.; Germanà, A.; de Carlos, F.; Ciriaco, E.; Vega, J.A.; Germanà, G. Effects of increased nerve growth factor plasma levels on the expression of TrkA and p75NTR in rat testicles. J. Anat. 2006, 208, 373–379. [Google Scholar] [CrossRef]
- Kumar, S.; Sharma, V.K.; Singh, S.; Hariprasad, G.R.; Mal, G.; Srinivasan, A.; Yadav, S. Proteomic identification of camel seminal plasma: Purification of β-nerve growth factor. Anim. Reprod. Sci. 2013, 136, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Druart, X.; Rickard, J.; Mactier, S.; Kohnke, P.; Kershaw-Young, C.; Bathgate, R.; Gibb, Z.; Crossett, B.; Tsikis, G.; Labas, V.; et al. Proteomic characterization and cross species comparison of mammalian seminal plasma. J. Proteomics 2013, 91, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Harper, G.P.; Glanville, R.W.; Thoenen, H. The purification of nerve growth factor from bovine seminal plasma. Biochemical characterization and partial amino acid sequence. J. Biol. Chem. 1982, 257, 8541–8548. [Google Scholar] [CrossRef]
- Li, C.; Zheng, L.; Wang, C.; Zhou, X. Absence of nerve growth factor and comparison of tyrosine kinase receptor A levels in mature spermatozoa from oligoasthenozoospermic, asthenozoospermic and fertile men. Clin. Chim. Acta 2010, 411, 1482–1486. [Google Scholar] [CrossRef]
- Adams, G.P.; Ratto, M.H.; Silva, M.E.; Carrasco, R.A. Ovulation-inducing factor (OIF/NGF) in seminal plasma: A review and update. Reprod. Domest. Anim. 2016, 51, 4–17. [Google Scholar] [CrossRef]
- Seidl, K.; Buchberger, A.; Erck, C. Expression of nerve growth factor and neurotrophin receptors in testicular cells suggest novel roles for neurotrophins outside the nervous system. Reprod. Fertil. Dev. 1996, 8, 1075–1087. [Google Scholar] [CrossRef]
- Brill, G.; Kahane, N.; Carmeli, C.; Von Schack, D.; Barde, Y.A.; Kalcheim, C. Epithelial-mesenchymal conversion of dermatome progenitors requires neural tube-derived signals: Characterization of the role of Neurotrophin-3. Development 1995, 121, 2583–2594. [Google Scholar] [CrossRef] [PubMed]
- Dissen, G.A.; Newman Hirshfield, A.; Malamed, S.; Ojeda, S.R. Expression of neurotrophins and their receptors in the mammalian ovary is developmentally regulated: Changes at the time of folliculogenesis. Endocrinology 1995, 136, 4681–4692. [Google Scholar] [CrossRef] [PubMed]
- Mitsiadis, T.A.; Luukko, K. Neurotrophins in odontogenesis. Int. J. Dev. Biol. 1995, 39, 195–202. [Google Scholar] [PubMed]
- Ojeda, S.R.; Dissen, G.A.; Junier, M.P. Neurotrophic factors and female sexual development. Front. Neuroendocrinol. 1992, 13, 120–162. [Google Scholar]
- Onoda, M.; Pflug, B.; Djakiew, D. Germ cell mitogenic activity is associated with nerve growth factor-like protein(s). J. Cell. Physiol. 1991, 149, 536–543. [Google Scholar] [CrossRef]
- Persson, H.; Lievre, C.A.-L.; Söder, O.; Villar, M.J.; Metsis, M.; Olson, L.; Ritzen, M.; Hökfelt, T. Expression of β-nerve growth factor receptor mRNA in Sertoli cells downregulated by testosterone. Science 1990, 247, 704–707. [Google Scholar] [CrossRef]
- Lonnerberg, P.; Soder, O.; Parvinen, M.; Ritzen, E.M.; Persson, H. β-Nerve growth factor influences the expression of androgen-binding protein messenger ribonucleic acid in the rat testis. Biol. Reprod. 1992, 47, 381–388. [Google Scholar] [CrossRef]
- Wheeler, E.F.; Bothwell, M. Spatiotemporal patterns of expression of NGF and the low-affinity NGF receptor in rat embryos suggest functional roles in tissue morphogenesis and myogenesis. J. Neurosci. 1992, 12, 930–945. [Google Scholar] [CrossRef]
- Russo, M.A.; Odorisio, T.; Fradeani, A.; Rienzi, L.; De Felici, M.; Cattaneo, A.; Siracusa, G. Low-affinity nerve growth factor receptor is expressed during testicular morphogenesis and in germ cells at specific stages of spermatogenesis. Mol. Reprod. Dev. 1994, 37, 157–166. [Google Scholar] [CrossRef]
- Levine, E.; Cupp, A.S.; Skinner, M.K. Role of neurotropins in rat embryonic testis morphogenesis (Cord formation). Biol. Reprod. 2000, 62, 132–142. [Google Scholar] [CrossRef][Green Version]
- Cupp, A.S.; Kim, G.H.; Skinner, M.K. Expression and action of neurotropin-3 and nerve growth factor in embryonic and early postnatal rat testis development. Biol. Reprod. 2000, 63, 1617–1628. [Google Scholar] [CrossRef] [PubMed]
- Cupp, A.S.; Tessarollo, L.; Skinner, M.K. Testis developmental phenotypes in neurotropin receptor trkA and trkC null mutations: Role in formation of seminiferous cords and germ cell survival. Biol. Reprod. 2002, 66, 1838–1845. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Neto, F.T.L.; Bach, P.V.; Najari, B.B.; Li, P.S.; Goldstein, M. Spermatogenesis in humans and its affecting factors. Semin. Cell Dev. Biol. 2016, 59, 10–26. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, H.; L’Hernault, S.W. Spermatogenesis. Curr Biol. 2017, 27, R988–R994. [Google Scholar] [CrossRef]
- De Kretser, D.M.; Loveland, K.L.; Meinhardt, A.; Simorangkir, D.; Wreford, N. Spermatogenesis. Hum. Reprod. 1998, 13, 1–8. [Google Scholar] [CrossRef]
- Larose, H.; Kent, T.; Ma, Q.; Shami, A.N.; Harerimana, N.; Li, J.Z.; Hammoud, S.S.; Handel, M.A. Regulation of meiotic progression by Sertoli-cell androgen signaling. Mol. Biol. Cell 2020, 31, 2841–2862. [Google Scholar] [CrossRef]
- Hess, R.A.; De Franca, L.R. Spermatogenesis and cycle of the seminiferous epithelium. Adv. Exp. Med. Biol. 2008, 636, 1–15. [Google Scholar] [CrossRef]
- Holdcraft, R.W.; Braun, R.E. Hormonal regulation of spermatogenesis. Int. J. Androl. 2004, 27, 335–342. [Google Scholar] [CrossRef]
- Rossi, P.; Dolci, S. Paracrine mechanisms involved in the control of early stages of mammalian spermatogenesis. Front. Endocrinol. 2013, 4, 181. [Google Scholar] [CrossRef]
- Sofikitis, N.; Giotitsas, N.; Tsounapi, P.; Baltogiannis, D.; Giannakis, D.; Pardalidis, N. Hormonal regulation of spermatogenesis and spermiogenesis. J. Steroid Biochem. Mol. Biol. 2008, 109, 323–330. [Google Scholar] [CrossRef]
- Griswold, M.D. 50 years of spermatogenesis: Sertoli cells and their interactions with germ cells. Biol. Reprod. 2018, 99, 87–100. [Google Scholar] [CrossRef] [PubMed]
- Schlatt, S.; Meinhardt, A.; Nieschlag, E. Paracrine regulation of cellular interactions in the testis: Factors in search of a function. Eur. J. Endocrinol. 1997, 137, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Iliadou, P.K.; Tsametis, C.; Kaprara, A.; Papadimas, I.; Goulis, D.G. The sertoli cell: Novel clinical potentiality. Hormones 2015, 14, 504–514. [Google Scholar] [CrossRef]
- Galdieri, M.; Monaco, L.; Stefanini, M. Secretion of Androgen Binding Protein by Sertoli Cells Is Influenced by Contact with Germ Cells. J. Androl. 1984, 5, 409–415. [Google Scholar] [CrossRef]
- Le Magueresse, B.; Jégou, B. In vitro effects of germ cells on the secretory activity of sertoli cells recovered from rats of different ages. Endocrinology 1988, 122, 1672–1680. [Google Scholar] [CrossRef]
- Onoda, M.; Djakiew, D. A 29,000 Mr protein derived from round spermatids regulates Sertoli cell secretion. Mol. Cell. Endocrinol. 1993, 93, 53–61. [Google Scholar] [CrossRef]
- Haugen, T.B.; Landmark, B.F.; Josefsen, G.M.; Hansson, V.; Högset, A. The mature form of interleukin-1α is constitutively expressed in immature male germ cells from rat. Mol. Cell. Endocrinol. 1994, 105, R19–R23. [Google Scholar] [CrossRef]
- Onoda, M.; Djakiew, D. Pachytene spermatocyte protein(s) stimulate sertoli cells grown in bicameral chambers: Dose-dependent secretion of ceruloplasmin, sulfated glycoprotein-1, sulfated glycoprotein-2, and transferrin. Vitr. Cell. Dev. Biol.-Anim. 1991, 27, 215–222. [Google Scholar] [CrossRef]
- Pineau, C.; Sharpe, R.M.; Saunders, P.T.K.; Gérard, N.; Jégou, B. Regulation of Sertoli cell inhibin production and of inhibin α-subunit mRNA levels by specific germ cell types. Mol. Cell. Endocrinol. 1990, 72, 13–22. [Google Scholar] [CrossRef]
- Onoda, M.; Djakiew, D. Modulation of Sertoli cell secretory function by rat round spermatid protein(s). Mol. Cell. Endocrinol. 1990, 73, 35–44. [Google Scholar] [CrossRef]
- Le Magueresse, B.; Jegou, B. Possible involvement of germ cells in the regulation of oestradiol-17,β and ABP secretion by immature rat sertoli cells (in vitro studies). Biochem. Biophys. Res. Commun. 1986, 141, 861–869. [Google Scholar] [CrossRef]
- Vigier, M.; Weiss, M.; Perrard, M.H.; Godet, M.; Durand, P. The effects of FSH and of testosterone on the completion of meiosis and the very early steps of spermiogenesis of the rat: An in vitro study. J. Mol. Endocrinol. 2004, 33, 729–742. [Google Scholar] [CrossRef] [PubMed]
- Hakovirta, H.; Kaipia, A.; Söder, O.; Parvinen, M. Effects of activin-A, inhibin-A, and transforming growth factor-β1 on stage-specific deoxyribonucleic acid synthesis during rat seminiferous epithelial cycle. Endocrinology 1993, 133, 1664–1668. [Google Scholar] [CrossRef] [PubMed]
- Olson, L.; Ayer-LeLievre, C.; Ebendal, T.; Seiger, Å. Nerve growth factor-like immunoreactivities in rodent salivary glands and testis. Cell Tissue Res. 1987, 248, 275–286. [Google Scholar] [CrossRef]
- MacGrogan, D.; Desprès, G.; Romand, R.; Dicou, E. Expression of the β-nerve growth factor gene in male sex organs of the mouse, rat, and guinea pig. J. Neurosci. Res. 1991, 28, 567–573. [Google Scholar] [CrossRef] [PubMed]
- Seidl, K.; Holstein, A.F. Organ culture of human seminiferous tubules: A useful tool to study the role of nerve growth factor in the testis. Cell Tissue Res. 1990, 261, 539–547. [Google Scholar] [CrossRef] [PubMed]
- Parvinen, M.; Pelto-Huikko, M.; Soder, O.; Schultz, R.; Kaipia, A.; Mali, P.; Toppari, J.; Hakovirta, H.; Lönnerberg, P.; Ritzén, E.M. Expression of β-nerve growth factor and its receptor in rat seminiferous epithelium: Specific function at the onset of meiosis. J. Cell Biol. 1992, 117, 629–641. [Google Scholar] [CrossRef]
- Djakiew, D.; Pflug, B.; Dionne, C.; Onoda, M. Postnatal expression of nerve growth factor receptors in the rat testis. Biol. Reprod. 1994, 51, 214–221. [Google Scholar] [CrossRef]
- MacGrogan, D.; Saint-André, J.-P.; Dicou, E. Expression of Nerve Growth Factor and Nerve Growth Factor Receptor Genes in Human Tissues and in Prostatic Adenocarcinoma Cell Lines. J. Neurochem. 1992, 59, 1381–1391. [Google Scholar] [CrossRef]
- Robinson, L.L.L.; Townsend, J.; Anderson, R.A. The human fetal testis is a site of expression of neurotrophins and their receptors: Regulation of the germ cell and peritubular cell population. J. Clin. Endocrinol. Metab. 2003, 88, 3943–3951. [Google Scholar] [CrossRef]
- Plant, T.M. The hypothalamo-pituitary-gonadal axis. J. Endocrinol. 2015, 226, T41–T54. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.M.; Yang, W.X. Molecular regulation of hypothalamus-pituitary-gonads axis in males. Gene 2014, 551, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Stamatiades, G.A.; Kaiser, U.B. Gonadotropin regulation by pulsatile GnRH: Signaling and gene expression. Mol. Cell. Endocrinol. 2018, 463, 131–141. [Google Scholar] [CrossRef]
- Zirkin, B.R.; Papadopoulos, V. Leydig cells: Formation, function, and regulation. Biol. Reprod. 2018, 99, 101–111. [Google Scholar] [CrossRef] [PubMed]
- Oduwole, O.O.; Peltoketo, H.; Huhtaniemi, I.T. Role of follicle-stimulating hormone in spermatogenesis. Front. Endocrinol. 2018, 9, 763. [Google Scholar] [CrossRef] [PubMed]
- Ramaswamy, S.; Weinbauer, G.F. Endocrine control of spermatogenesis: Role of FSH and LH/testosterone. Spermatogenesis 2014, 4, e996025. [Google Scholar] [CrossRef] [PubMed]
- Tilbrook, A.J.; Clarke, I.J. Negative feedback regulation of the secretion and actions of gonadotropin-releasing hormone in males. Biol. Reprod. 2001, 64, 735–742. [Google Scholar] [CrossRef]
- Corradi, P.F.; Corradi, R.B.; Greene, L.W. Physiology of the Hypothalamic Pituitary Gonadal Axis in the Male. Urol. Clin. N. Am. 2016, 43, 151–162. [Google Scholar] [CrossRef]
- Luisi, S.; Florio, P.; Reis, F.M.; Petraglia, F. Inhibins in female and male reproductive physiology: Role in gametogenesis, conception, implantation and early pregnancy. Hum. Reprod. Update 2005, 11, 123–135. [Google Scholar] [CrossRef]
- Scaccianoce, S.; Cigliana, G.; Nicolai, R.; Muscolo, L.A.; Porcu, A.; Navarra, D.; Perez-Polo, R.; Angelucci, L. Hypothalamic involvement in the activation of the pituitary-adrenocortical axis by nerve growth factor. Neuroendocrinology 1993, 58, 202–209. [Google Scholar] [CrossRef]
- Kumar, A.; Kumar, P.; Pareek, V.; Faiq, M.A.; Narayan, R.K.; Raza, K.; Prasoon, P.; Sharma, V.K. Neurotrophin mediated HPA axis dysregulation in stress induced genesis of psychiatric disorders: Orchestration by epigenetic modifications. J. Chem. Neuroanat. 2019, 102, 101688. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Yang, Y.; Zhang, T.; Su, Z.; Yu, D.; Lin, Q.; Chen, H.; Zhang, Q.; Xiang, Q.; Xue, W.; et al. Nasal delivery of nerve growth factor rescue hypogonadism by up-regulating GnRH and testosterone in aging male mice. EBioMedicine 2018, 35, 295–306. [Google Scholar] [CrossRef] [PubMed]
- Marlin, M.C.; Li, G. Biogenesis and Function of the NGF/TrkA Signaling Endosome. Int. Rev. Cell Mol. Biol. 2015, 314, 239–257. [Google Scholar] [CrossRef] [PubMed]
- Higa-Nakamine, S.; Maeda, N.; Toku, S.; Yamamoto, H. Involvement of protein kinase D1 in signal transduction from the protein kinase C pathway to the tyrosine kinase pathway in response to gonadotropin-releasing hormone. J. Biol. Chem. 2015, 290, 25974–25985. [Google Scholar] [CrossRef]
- Sasson, R.; Dearth, R.K.; White, R.S.; Chappell, P.E.; Mellon, P.L. Orexin A induces GnRH gene expression and secretion from GT1-7 hypothalamic GnRH neurons. Neuroendocrinology 2007, 84, 353–363. [Google Scholar] [CrossRef]
- Riccio, A.; Ahn, S.; Davenport, C.M.; Blendy, J.A.; Ginty, D.D. Mediation by a CREB family transcription factor of NGF-dependent survival of sympathetic neurons. Science 1999, 286, 2358–2361. [Google Scholar] [CrossRef]
- Liu, Y.Z.; Chrivia, J.C.; Latchman, D.S. Nerve growth factor up-regulates the transcriptional activity of CBP through activation of the p42/p44(MAPK) cascade. J. Biol. Chem. 1998, 273, 32400–32407. [Google Scholar] [CrossRef][Green Version]
- Wortzel, I.; Seger, R. The ERK cascade: Distinct functions within various subcellular organelles. Genes Cancer 2011, 2, 195–209. [Google Scholar] [CrossRef]
- Sanchez-Rodriguez, A.; Abad, P.; Arias-Alvarez, M.; Rebollar, P.G.; Bautista, J.M.; Lorenzo, P.L.; García-García, R.M. Recombinant rabbit beta nerve growth factor production and its biological effects on sperm and ovulation in rabbits. PLoS ONE 2019, 14, e0219780. [Google Scholar] [CrossRef]
- Li, C.; Zhou, X. The potential roles of neurotrophins in male reproduction. Reproduction 2013, 145, R89–R95. [Google Scholar] [CrossRef]
- Li, C.; Sun, Y.; Yi, K.; Ma, Y.; Zhang, W.; Zhou, X. Detection of nerve growth factor (NGF) and its specific receptor (TrkA) in ejaculated bovine sperm, and the effects of NGF on sperm function. Theriogenology 2010, 74, 1615–1622. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.-G.; Lin, K.; Xu, X.-B.; Zhang, S.-C.; Wang, N.; Fan, M. Evidence for the involvement of NGF in human sperm motility. J. Biomed. Sci. Eng. 2012, 5, 534–541. [Google Scholar] [CrossRef][Green Version]
- Lin, K.; Ding, X.-F.; Shi, C.-G.; Zeng, D.; QuZong, S.; Liu, S.-H.; Wu, Y.; LuoBu, G.; Fan, M.; Zhao, Y.-Q. Nerve growth factor promotes human sperm motility in vitro by increasing the movement distance and the number of A grade spermatozoa. Andrologia 2015, 47, 1041–1046. [Google Scholar] [CrossRef] [PubMed]
- Bezerra, M.; Arruda-Alencar, J.; Martins, J.; Viana, A.; Neto, A.V.; Rêgo, J.; de Oliveira, R.V.; Lobo, M.; Moreira, A.; Moreira, R.; et al. Major seminal plasma proteome of rabbits and associations with sperm quality. Theriogenology 2019, 128, 156–166. [Google Scholar] [CrossRef] [PubMed]
- Saeednia, S.; Bahadoran, H.; Amidi, F.; Asadi, M.H.; Naji, M.; Fallahi, P.; Nejad, N.A. Nerve growth factor in human semen: Effect of nerve growth factor on the normozoospermic men during cryopreservation process. Iran. J. Basic Med. Sci. 2015, 18, 292–299. [Google Scholar] [PubMed]
- Parthipan, S.; Selvaraju, S.; Somashekar, L.; Arangasamy, A.; Sivaram, M.; Ravindra, J.P. Spermatozoal transcripts expression levels are predictive of semen quality and conception rate in bulls (Bos taurus). Theriogenology 2017, 98, 41–49. [Google Scholar] [CrossRef]
- Sari, L.M.; Zampini, R.; Argañaraz, M.E.; Carretero, M.I.; Fumuso, F.G.; Barraza, D.E.; Ratto, M.; Apichela, S.A. Expression of β-NGF and high-affinity NGF receptor (TrKA) in llama (Lama glama) male reproductive tract and spermatozoa. Mol. Reprod. Dev. 2018, 85, 934–944. [Google Scholar] [CrossRef]
- Cacialli, P. Expression of Nerve Growth Factor and Its Receptor TrkA in the Reproductive System of Adult Zebrafish. Vet. Sci. 2022, 9, 225. [Google Scholar] [CrossRef]
- Hong, W.F.; Su, Z.; Xiao, X. The Roles of Nerve Growth Factor and Its Receptors in Leydig Cells Development and Function. Int. J. Sci. 2018, 5, 176–183. [Google Scholar]
- Castellini, C.; Mattioli, S.; Bosco, A.D.; Cotozzolo, E.; Mancinelli, A.C.; Rende, M.; Stabile, A.M.; Pistilli, A. Nerve growth factor receptor role on rabbit sperm storage. Theriogenology 2020, 153, 54–61. [Google Scholar] [CrossRef]
- Suarez, S.S. Control of hyperactivation in sperm. Hum. Reprod. Update 2008, 14, 647–657. [Google Scholar] [CrossRef] [PubMed]
- Aitken, R.J.; Curry, B.J. Redox regulation of human sperm function: From the physiological control of sperm capacitation to the etiology of infertility and DNA damage in the germ line. Antioxidants Redox. Signal. 2011, 14, 367–381. [Google Scholar] [CrossRef]
- Ecroyd, H.W.; Jones, R.C.; Aitken, R.J. Endogenous redox activity in mouse spermatozoa and its role in regulating the tyrosine phosphorylation events associated with sperm capacitation. Biol. Reprod. 2003, 69, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Chandra, J.; Samali, A.; Orrenius, S. Triggering and modulation of apoptosis by oxidative stress. Free Radic. Biol. Med. 2000, 29, 323–333. [Google Scholar] [CrossRef]
- Weese, D.L.; Peaster, M.L.; Himsl, K.K.; Leach, G.E.; Lad, P.M.; Zimmern, P.E. Stimulated reactive oxygen species generation in the spermatozoa of infertile men. J. Urol. 1993, 149, 64–67. [Google Scholar] [CrossRef]
- Castellini, C.; Mattioli, S.; Bosco, A.D.; Collodel, G.; Pistilli, A.; Stabile, A.M.; Macchioni, L.; Mancuso, F.; Luca, G.; Rende, M. In vitro effect of nerve growth factor on the main traits of rabbit sperm. Reprod. Biol. Endocrinol. 2019, 17, 93. [Google Scholar] [CrossRef] [PubMed]
- Nykjaer, A.; Willnow, T.E.; Petersen, C.M. p75NTR--live or let die. Curr. Opin. Neurobiol. 2005, 15, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Castellini, C.; Mattioli, S.; Cotozzolo, E.; Pistilli, A.; Rende, M.; Bartolini, D.; Di Sante, G.; Menchetti, L.; Bosco, A.D.; Stabile, A.M. The Effect of Interaction NGF/p75(NTR) in Sperm Cells: A Rabbit Model. Cells 2022, 11, 1035. [Google Scholar] [CrossRef]
- Sanchez-Rodriguez, A.; Arias-Alvarez, M.; Timón, P.; Bautista, J.M.; Rebollar, P.G.; Lorenzo, P.L.; Garcia-Garcia, R.M. Characterization of β-Nerve Growth Factor-TrkA system in male reproductive tract of rabbit and the relationship between β-NGF and testosterone levels with seminal quality during sexual maturation. Theriogenology 2019, 126, 206–213. [Google Scholar] [CrossRef]
- Maranesi, M.; Zerani, M.; Leonardi, L.; Pistilli, A.; Arruda-Alencar, J.; Stabile, A.; Rende, M.; Castellini, C.; Petrucci, L.; Parillo, F.; et al. Gene Expression and Localization of NGF and Its Cognate Receptors NTRK1 and NGFR in the Sex Organs of Male Rabbits. Reprod. Domest. Anim. 2015, 50, 918–925. [Google Scholar] [CrossRef]
- Chaves, R.N.; Alves, A.M.C.V.; Lima, L.F.; Matos, H.M.T.; Rodrigues, A.P.R.; Figueiredo, J.R. Role of nerve growth factor (NGF) and its receptors in folliculogenesis. Zygote 2013, 21, 187–197. [Google Scholar] [CrossRef]
- D’Angelo, A.; Ceccanti, M.; Petrella, C.; Greco, A.; Tirassa, P.; Rosso, P.; Ralli, M.; Ferraguti, G.; Fiore, M.; Messina, M.P. Role of neurotrophins in pregnancy, delivery and postpartum. Eur. J. Obstet. Gynecol. Reprod. Biol. 2020, 247, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Streiter, S.; Fisch, B.; Sabbah, B.; Ao, A.; Abir, R. The importance of neuronal growth factors in the ovary. Mol. Hum. Reprod. 2015, 22, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Wang, H.; Yang, Y.; Liu, H.; Zhang, Q.; Xiang, Q.; Ge, R.; Su, Z.; Huang, Y. NGF induces adult stem Leydig cells to proliferate and differentiate during Leydig cell regeneration. Biochem. Biophys. Res. Commun. 2013, 436, 300–305. [Google Scholar] [CrossRef] [PubMed]
- Halpern, J.A.; Brannigan, R.E. Testosterone deficiency. JAMA-J. Am. Med. Assoc. 2019, 322, 1116. [Google Scholar] [CrossRef]
- Barbonetti, A.; D’Andrea, S.; Francavilla, S. Testosterone replacement therapy. Andrology 2020, 8, 1551–1566. [Google Scholar] [CrossRef]
- Grech, A.; Breck, J.; Heidelbaugh, J. Adverse effects of testosterone replacement therapy: An update on the evidence and controversy. Ther. Adv. Drug Saf. 2014, 5, 190–200. [Google Scholar] [CrossRef]
- Majzoub, A.; Sabanegh, E. Testosterone replacement in the infertile man. Transl. Androl. Urol. 2016, 5, 859–865. [Google Scholar] [CrossRef]
- Kim, E.D.; Crosnoe, L.; Bar-Chama, N.; Khera, M.; Lipshultz, L.I. The treatment of hypogonadism in men of reproductive age. Fertil. Steril. 2013, 99, 718–724. [Google Scholar] [CrossRef]
- Meng, T.; Cao, Q.; Lei, P.; Bush, A.I.; Xiang, Q.; Su, Z.; He, X.; Rogers, J.T.; Chiu, I.-M.; Zhang, Q.; et al. Tat-haFGF14–154 Upregulates ADAM10 to Attenuate the Alzheimer Phenotype of APP/PS1 Mice through the PI3K-CREB-IRE1α/XBP1 Pathway. Mol. Ther.-Nucleic Acids 2017, 7, 439–452. [Google Scholar] [CrossRef]
- Lou, G.; Zhang, Q.; Xiao, F.; Xiang, Q.; Su, Z.; Zhang, L.; Yang, P.; Yang, Y.; Zheng, Q.; Huang, Y. Intranasal administration of TAT-haFGF14-154 attenuates disease progression in a mouse model of Alzheimer’s disease. Neuroscience 2012, 223, 225–237. [Google Scholar] [CrossRef] [PubMed]
- De Rosa, R.; Garcia, A.A.; Braschi, C.; Capsoni, S.; Maffei, L.; Berardi, N.; Cattaneo, A. Intranasal administration of nerve growth factor (NGF) rescues recognition memory deficits in AD11 anti-NGF transgenic mice. Proc. Natl. Acad. Sci. USA 2005, 102, 3811–3816. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Yang, Y.; Ji, X.; He, W.; Fan, J.; Huang, Y.; Wang, Y. NGF Rescues Spermatogenesis in Azoospermic Mice. Reprod. Sci. 2021, 28, 2780–2788. [Google Scholar] [CrossRef] [PubMed]
- Begna, K.; Abdelatif, A.; Schwager, S.; Hanson, C.; Pardanani, A.; Tefferi, A. Busulfan for the treatment of myeloproliferative neoplasms: The Mayo Clinic experience. Blood Cancer J. 2016, 6, e427. [Google Scholar] [CrossRef]
- Qu, N.; Itoh, M.; Sakabe, K. Effects of chemotherapy and radiotherapy on spermatogenesis: The role of testicular immunology. Int. J. Mol. Sci. 2019, 20, 957. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Xue, X.; Fan, C.; Li, Y.; Pu, Y.; Cao, H.; Zhang, X. Establishment of recipient model for spermatogonial stem cells transplantation in Kunming mice. Tissue Cell 2014, 46, 249–254. [Google Scholar] [CrossRef]
- Skurikhin, E.G.; Pakhomova, A.V.; Pershina, O.V.; Ermolaeva, L.A.; Ermakova, N.N.; Krupin, V.A.; Pan, E.S.; Kudryashova, A.I.; Rybalkina, O.Y.; Zhdanov, V.V.; et al. Regenerative Potential of Stem and Progenitor Cells from Ischemic Testes of C57Bl/6 Mice in Culture and in the Model of Spermatogenesis Suppression Caused by Busulfan. Bull. Exp. Biol. Med. 2017, 162, 400–405. [Google Scholar] [CrossRef]
- Hezavehei, M.; Sharafi, M.; Kouchesfahani, H.M.; Henkel, R.; Agarwal, A.; Esmaeili, V.; Shahverdi, A. Sperm cryopreservation: A review on current molecular cryobiology and advanced approaches. Reprod. Biomed. Online 2018, 37, 327–339. [Google Scholar] [CrossRef]
- Jang, T.H. Cryopreservation and its clinical applications. Integr. Med. Res. 2017, 6, 12–18. [Google Scholar] [CrossRef]
- Agarwal, A.; Cho, C.L.; Esteves, S.C.; Majzoub, A. Development of treatment strategies in men with vulnerable sperm. Transl. Androl. Urol. 2017, 6, S476–S478. [Google Scholar] [CrossRef]
- Dohle, G.R. Male infertility in cancer patients: Review of the literature. Int. J. Urol. 2010, 17, 327–331. [Google Scholar] [CrossRef] [PubMed]
- Anger, J.T.; Gilbert, B.R.; Goldstein, M. Cryopreservation of sperm: Indications, methods and results. J. Urol. 2003, 170, 1079–1084. [Google Scholar] [CrossRef] [PubMed]
- Asimakopoulos, B.; Tiptiri-Kourpeti, A.; Metallinou, C. IGF-I and NGFβ enhance in vitro progressive motility and vitality of human spermatozoa. Reprod. Med. Biol. 2021, 20, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Holoch, P.; Wald, M. Current options for preservation of fertility in the male. Fertil. Steril. 2011, 96, 286–290. [Google Scholar] [CrossRef]
- Di Santo, M.; Tarozzi, N.; Nadalini, M.; Borini, A. Human sperm cryopreservation: Update on techniques, effect on DNA integrity, and implications for ART. Adv. Urol. 2012, 2012, 854837. [Google Scholar] [CrossRef]
- Agca, Y.; Critser, J.K. Cryopreservation of spermatozoa in assisted reproduction. Semin. Reprod. Med. 2002, 20, 15–23. [Google Scholar] [CrossRef]
- Muller, I.; Oude Ophuis, R.J.A.; Broekmans, F.J.M.; Lock, T.M.T.W. Semen cryopreservation and usage rate for assisted reproductive technology in 898 men with cancer. Reprod. Biomed. Online 2016, 32, 147–153. [Google Scholar] [CrossRef]
- Hourvitz, A.; Goldschlag, D.E.; Davis, O.K.; Gosden, L.V.; Palermo, G.D.; Rosenwaks, Z. Intracytoplasmic sperm injection (ICSI) using cryopreserved sperm from men with malignant neoplasm yields high pregnancy rates. Fertil. Steril. 2008, 90, 557–563. [Google Scholar] [CrossRef]
- Talaei, T.; Esmaeelpour, T.; Aekiyash, F.; Bahmanpour, S. Effects of cryopreservation on plasma membrane glycoconjugates of human spermatozoa. Iran. J. Reprod. Med. 2010, 8, 119–124. [Google Scholar]
- Yeste, M. Sperm cryopreservation update: Cryodamage, markers, and factors affecting the sperm freezability in pigs. Theriogenology 2016, 85, 47–64. [Google Scholar] [CrossRef]
- Oehninger, S.; Duru, N.K.; Srisombut, C.; Morshedi, M. Assessment of sperm cryodamage and strategies to improve outcome. Mol. Cell Endocrinol. 2000, 169, 3–10. [Google Scholar] [CrossRef]
- Peris-Frau, P.; Soler, A.J.; Iniesta-Cuerda, M.; Martín-Maestro, A.; Sánchez-Ajofrín, I.; Medina-Chávez, D.A.; Fernández-Santos, M.R.; García-Álvarez, O.; Maroto-Morales, A.; Montoro, V.; et al. Sperm cryodamage in ruminants: Understanding the molecular changes induced by the cryopreservation process to optimize sperm quality. Int. J. Mol. Sci. 2020, 21, 2781. [Google Scholar] [CrossRef] [PubMed]
- Nijs, M.; Creemers, E.; Cox, A.; Janssen, M.; Vanheusden, E.; Castro-Sanchez, Y.; Thijs, H.; Ombelet, W. Influence of freeze-thawing on hyaluronic acid binding of human spermatozoa. Reprod. Biomed. Online 2009, 19, 202–206. [Google Scholar] [CrossRef]
- Ozkavukcu, S.; Erdemli, E.; Isik, A.; Oztuna, D.; Karahuseyinoglu, S. Effects of cryopreservation on sperm parameters and ultrastructural morphology of human spermatozoa. J. Assist. Reprod. Genet. 2008, 25, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Mazzilli, F.; Rossi, T.; Sabatini, L.; Pulcinelli, F.M.; Rapone, S.; Dondero, F.; Gazzaniga, P.P. Human sperm cryopreservation and reactive oxygen species (ROS) production. Acta Eur. Fertil. 1995, 26, 145–148. [Google Scholar] [PubMed]
- Taylor, K.; Roberts, P.; Sanders, K.; Burton, P. Effect of antioxidant supplementation of cryopreservation medium on post-thaw integrity of human spermatozoa. Reprod. Biomed. Online 2009, 18, 184–189. [Google Scholar] [CrossRef]
- McLaughlin, E.A.; Ford, W.C.L.; Hull, M.G.R. Motility characteristics and membrane integrity of cryopreserved human spermatozoa. J. Reprod. Fertil. 1992, 95, 527–534. [Google Scholar] [CrossRef]
- Critser, J.K.; Arneson, B.W.; Aaker, D.V.; Huse-Benda, A.R.; Ball, G.D. Cryopreservation of human spermatozoa. II. Postthaw chronology of motility and of zona-free hamster ova penetration. Fertil. Steril. 1987, 47, 980–984. [Google Scholar] [CrossRef]
- Bansal, A.K.; Bilaspuri, G.S. Impacts of oxidative stress and antioxidants on semen functions. Vet. Med. Int. 2011, 2011, 686137. [Google Scholar] [CrossRef]
- Thomson, L.K.; Fleming, S.D.; Aitken, R.J.; De Iuliis, G.N.; Zieschang, J.A.; Clark, A.M. Cryopreservation-induced human sperm DNA damage is predominantly mediated by oxidative stress rather than apoptosis. Hum. Reprod. 2009, 24, 2061–2070. [Google Scholar] [CrossRef]
- Cross, N.L.; Hanks, S.E. Effects of cryopreservation on human sperm acrosomes. Hum. Reprod. 1991, 6, 1279–1283. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Jiang, S.; Su, J.; Zhang, J.; Bao, X.; Ding, R.; Shi, P.; Li, S.; Wu, C.; Zhao, G.; et al. The effects of cryopreservation on the acrosome structure, enzyme activity, motility, and fertility of bovine, ovine, and goat sperm. Anim. Reprod. 2020, 17, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Gualtieri, R.; Kalthur, G.; Barbato, V.; Di Nardo, M.; Adiga, S.K.; Talevi, R. Mitochondrial dysfunction and oxidative stress caused by cryopreservation in reproductive cells. Antioxidants 2021, 10, 337. [Google Scholar] [CrossRef] [PubMed]
- Said, T.M.; Gaglani, A.; Agarwal, A. Implication of apoptosis in sperm cryoinjury. Reprod. Biomed. Online 2010, 21, 456–462. [Google Scholar] [CrossRef]
- Baumber, J.; Ball, B.A.; Linfor, J.J.; Meyers, S.A. Reactive Oxygen Species and Cryopreservation Promote DNA Fragmentation in Equine Spermatozoa. J. Androl. 2003, 24, 621–628. [Google Scholar] [CrossRef] [PubMed]
- Amidi, F.; Pazhohan, A.; Shabani Nashtaei, M.; Khodarahmian, M.; Nekoonam, S. The role of antioxidants in sperm freezing: A review. Cell Tissue Bank 2016, 17, 745–756. [Google Scholar] [CrossRef] [PubMed]
- Sieme, H.; Oldenhof, H.; Wolkers, W.F. Mode of action of cryoprotectants for sperm preservation. Anim. Reprod. Sci. 2016, 169, 2–5. [Google Scholar] [CrossRef]
- Bahmyari, R.; Zare, M.; Sharma, R.; Agarwal, A.; Halvaei, I. The efficacy of antioxidants in sperm parameters and production of reactive oxygen species levels during the freeze-thaw process: A systematic review and meta-analysis. Andrologia 2020, 52, e13514. [Google Scholar] [CrossRef]
- Najafi, A.; Asadi, E.; Moawad, A.R.; Mikaeili, S.; Amidi, F.; Adutwum, E.; Safa, M.; Sobhani, A.G. Supplementation of freezing and thawing media with brain-derived neurotrophic factor protects human sperm from freeze-thaw-induced damage. Fertil. Steril. 2016, 106, 1658–1665.e4. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Dicou, E.; Djakiew, D. Characterization of nerve growth factor precursor protein expression in rat round spermatids and the trophic effects of nerve growth factor in the maintenance of Sertoli cell viability. Mol. Cell. Endocrinol. 1997, 127, 129–136. [Google Scholar] [CrossRef]
- Oberoi, B.; Kumar, S.; Talwar, P. Study of human sperm motility post cryopreservation. Med. J. Armed. Forces India 2014, 70, 349–353. [Google Scholar] [CrossRef] [PubMed][Green Version]
- O’Connell, M.; McClure, N.; Lewis, S.E.M. The effects of cryopreservation on sperm morphology, motility and mitochondrial function. Hum. Reprod. 2002, 17, 704–709. [Google Scholar] [CrossRef] [PubMed]
- Bell, M.; Wang, R.U.N.; Hellstrom, W.J.; Sikka, S.C. Effect of Cryoprotective Additives and Cryopreservation Protocol on Sperm Membrane Lipid Peroxidation and Recovery of Motile Human Sperm. J. Androl. 1993, 14, 472–478. [Google Scholar] [CrossRef] [PubMed]
- Fisch, B.; Kraicer, R.K.; Amit, S.; Zukerman, Z.; Ovadia, J.; Tadir, Y. The relationship between sperm parameters and fertilizing capacity in vitro: A predictive role for swim-up migration. J. Vitr. Fertil. Embryo Transf. 1990, 7, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Dcunha, R.; Hussein, R.S.; Ananda, H.; Kumari, S.; Adiga, S.K.; Kannan, N.; Zhao, Y.; Kalthur, G. Current Insights and Latest Updates in Sperm Motility and Associated Applications in Assisted Reproduction. Reprod. Sci. 2022, 29, 7–25. [Google Scholar] [CrossRef]
- Bongso, A.; Ng, S.-C.; Mok, H.; Lim, M.N.; Teo, H.L.; Wong, P.; Ratnam, S. Improved sperm concentration, motility, and fertilization rates following Ficoll treatment of sperm in a human in vitro fertilization program. Fertil. Steril. 1989, 51, 850–854. [Google Scholar] [CrossRef]
- Hosseini, A.; Khalili, M.A. Improvement of motility after culture of testicular spermatozoa: The effects of incubation timing and temperature. Transl. Androl. Urol. 2017, 6, 271–276. [Google Scholar] [CrossRef]
- Rajfer, J. Enhancement of sperm motility in assisted reproduction. Rev. Urol. 2006, 8, 88. [Google Scholar]
- Mbizvo, M.T.; Johnston, R.C.; Baker, G.H.W. The effect of the motility stimulants, caffeine, pentoxifylline, and 2- deoxyadenosine on hyperactivation of cryopreserved human sperm. Fertil. Steril. 1993, 59, 1112–1117. [Google Scholar] [CrossRef]
- Scott, L.; Smith, S. Human sperm motility-enhancing agents have detrimental effects on mouse oocytes and embryos. Fertil. Steril. 1995, 63, 166–175. [Google Scholar] [CrossRef]
- Weng, Q.; Shi, Z.Q.; Tukada, J.; Watanabe, G.; Taya, K. Immunodetection of NGF, trkA, p75 and inhibin α-Subunit in interstitial cells of golden hamsters treated with hCG. J. Reprod. Dev. 2009, 55, 622–628. [Google Scholar] [CrossRef] [PubMed]
- Amiss, E.; Stewart, J.W.; Negrón-Pérez, V.M.; Jones, K.; Haines, H.; Rhoads, M.L.; Lima, F.S. 117 Supplementation of IVF medium with nerve growth factor improved bovine embryonic cleavage rates during summer months. Reprod. Fertil Dev. 2020, 32, 185. [Google Scholar] [CrossRef]
| Ref. | Methods | Results | Potential Clinical Applications |
|---|---|---|---|
| [197] | The effects of NGF on Leydig-cell (LC) regeneration were investigated by measuring mRNA levels in the adult rat testis after ethane dimethanesulfonate (EDS) treatment. The established organ culture model of rat seminiferous tubules was used to examine the regulation of NGF during SLC proliferation and differentiation using EdU staining, real-time PCR and Western blotting. Progenitor Leydig cells (PLCs) and immature Leydig cells (ILCs) were also used to investigate the effects of NGF on LCs at different developmental stages. | NGF significantly promoted SLCs proliferation with an obvious dose–response relationship. There is strong evidence that NGF can induce SLCs to differentiate into LCs. | NGF pathway might be a potential target for the development of new therapies for diseases related to the dysfunction of Leydig cells, such as Partial Androgen Deficiency of the Aging Male (PADAM). |
| [165] | The ten-month-old aging male senescence accelerate mouse P8 (SAMP8) mice with age-related hypogonadotropic hypogonadism were used to study the role of NGF in hypogonadism. The age-matched accelerated senescence-resistant mouse R1 (SAMR1) served as a control. The ten-month-old SAMP8 mice were treated with NGF twice per week for 12 weeks. Sexual hormones, sexual behaviors, and fertility were analyzed after NGF treatment. The mechanisms of NGF in sex hormones’ sexual function were also studied. | NGF could enhance the sexual function, improve the quality of the sperm, and restore the fertility of aging male SAMP8 mice with age-related hypogonadism by activating gonadotropin-releasing hormone (GnRH) neurons and regulating the secretion of GnRH. NGF regulated the GnRH release through the PKC/p-ERK1/2/p-CREB signal pathway. | NGF administration might represent an alternative therapeutic option for hypogonadism treatment. |
| [206] | A model with azoospermia induced by a single intraperitoneal injection of busulfan was established. NGF pre-encapsulated with liposomes (25, 50, and 100 μg/kg) was delivered via internasal administration. Three weeks after busulfan injection, NGF treatments were performed twice a week for 8 weeks; the change in sperm quality, testis and epididymis histopathology, and androgenic hormone were analyzed to evaluate sperm regeneration. | NGF with nasal administration could significantly upregulate the markers expressing meiotic spermatogonia (Stra8) and spermatocytes (SYCP3), restore spermatogenesis, and improve sperm quality. | NGF therapy might be used as a protective strategy against gonadal dysfunction related to busulfan treatment or other anticancer agents. |
| Ref. | Methods | Results | Potential Clinical Applications |
|---|---|---|---|
| [178] | Semen samples were collected from 25 normozoospermic men and were divided into fresh semen samples as the control group, frozen–thawed semen samples without the addition of exogenous NGF, and three groups of semen samples cryopreserved with addition of exogenous NGF (0.5, 1, and 5 ng/mL) in freezing medium. Viability, motility, and NO concentration were evaluated. | Results showed that exogenous NGF at 0.5 ng/mL could significantly (p-value < 0.05) influence viability, motility, nitric oxide, and DNA fragmentation content. | NGF supplementation to cryopreserved semen might be useful to improve spermatozoa viability and motility and reduce cells’ cryodamage. |
| [216] | Forty-three volunteers gave semen samples after 2–3 days of sexual abstinence. Each sample was processed with density gradient centrifugation and sperm washing. The pellet was divided into 3 aliquots. An aliquot containing one million progressively motile spermatozoa was incubated for an hour (37 °C) in a standard culture medium (control group), and two aliquots with the same number of progressively motile spermatozoa were incubated in a medium supplemented with IGF-I or NGFβ. Two concentrations of IGF-I (100 ng/mL and 1000 ng/mL) and NGFβ (0.5 ng/mL and 5 ng/mL) were tested. | Both growth factors significantly increased PM and vitality in comparison with control either at the low or the high concentration. IGF-I seemed to be more effective than NGFβ. The effects did not seem to be dose-dependent, with the exception of the effect of IGF-I on vitality. | Incubating spermatozoa in a medium supplemented with IGF-I or NGF can enhance in vitro vitality and progressive motility of spermatozoa, suggesting their potential role in improving assisted reproduction outcomes. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferraguti, G.; Fanfarillo, F.; Tarani, L.; Blaconà, G.; Tarani, F.; Barbato, C.; Minni, A.; Ralli, M.; Francati, S.; Greco, A.; et al. NGF and the Male Reproductive System: Potential Clinical Applications in Infertility. Int. J. Mol. Sci. 2022, 23, 13127. https://doi.org/10.3390/ijms232113127
Ferraguti G, Fanfarillo F, Tarani L, Blaconà G, Tarani F, Barbato C, Minni A, Ralli M, Francati S, Greco A, et al. NGF and the Male Reproductive System: Potential Clinical Applications in Infertility. International Journal of Molecular Sciences. 2022; 23(21):13127. https://doi.org/10.3390/ijms232113127
Chicago/Turabian StyleFerraguti, Giampiero, Francesca Fanfarillo, Luigi Tarani, Giovanna Blaconà, Francesca Tarani, Christian Barbato, Antonio Minni, Massimo Ralli, Silvia Francati, Antonio Greco, and et al. 2022. "NGF and the Male Reproductive System: Potential Clinical Applications in Infertility" International Journal of Molecular Sciences 23, no. 21: 13127. https://doi.org/10.3390/ijms232113127
APA StyleFerraguti, G., Fanfarillo, F., Tarani, L., Blaconà, G., Tarani, F., Barbato, C., Minni, A., Ralli, M., Francati, S., Greco, A., Petrella, C., & Fiore, M. (2022). NGF and the Male Reproductive System: Potential Clinical Applications in Infertility. International Journal of Molecular Sciences, 23(21), 13127. https://doi.org/10.3390/ijms232113127











