Using Exogenous Melatonin, Glutathione, Proline, and Glycine Betaine Treatments to Combat Abiotic Stresses in Crops
Abstract
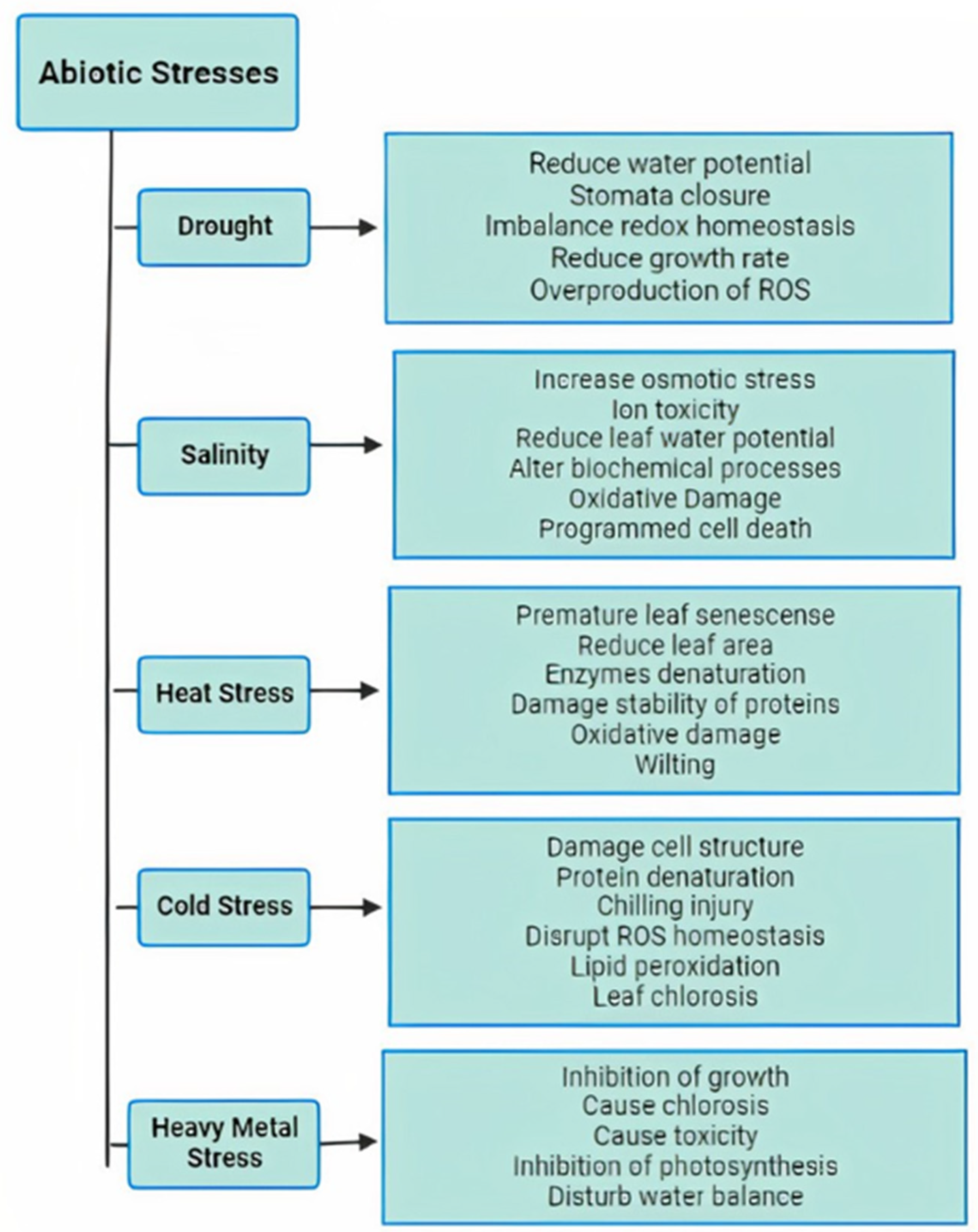
1. Introduction
2. Melatonin
2.1. Structure and Function of Melatonin
2.2. Melatonin Effect against Drought Stress
2.3. Melatonin Effect against Salinity Stress
2.4. Melatonin Effect against Heat Stress
2.5. Melatonin Effect against Cold Stress
2.6. Melatonin Effect against Heavy Metal Stress
3. Glutathione
3.1. Structure and Function of Glutathione
3.2. Glutathione Effect against Drought Stress
3.3. Glutathione Effect against Salinity Stress
3.4. Glutathione Effect against Heat Stress
3.5. Glutathione Effect against Cold Stress
3.6. Glutathione Effect against Heavy Metal Stress
4. Proline
4.1. Structure and Function of Proline
4.2. Proline Effect against Drought Stress
4.3. Proline Effect against Salinity Stress
4.4. Proline Effect against Heat Stress
4.5. Proline Effect against Cold Stress
4.6. Proline Effect against Heavy Metal Stress
5. Glycine Betaine
5.1. Structure and Function of Glycine Betaine
5.2. Glycine Betaine Effect against Drought Stress
5.3. Glycine Betaine Effect against Salinity Stress
5.4. Glycine Betaine Effect against Heat Stress
5.5. Glycine Betaine Effect against Cold Stress
5.6. Glycine Betaine Effect against Heavy Metal Stress
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zulfiqar, F.; Navarro, M.; Ashraf, M.; Akram, N.A.; Munné-Bosch, S. Nanofertilizer Use for Sustainable Agriculture: Advantages and Limitations. Plant Sci. 2019, 289, 110270. [Google Scholar] [CrossRef] [PubMed]
- Hasanuzzaman, M.; Nahar, K.; Hossain, M.S.; Anee, T.I.; Parvin, K.; Fujita, M. Nitric Oxide Pretreatment Enhances Antioxidant Defense and Glyoxalase Systems to Confer PEG-Induced Oxidative Stress in Rapeseed. J. Plant Interact. 2017, 12, 323–331. [Google Scholar] [CrossRef]
- Imran, M.; Latif Khan, A.; Shahzad, R.; Aaqil Khan, M.; Bilal, S.; Khan, A.; Kang, S.-M.; Lee, I.-J. Exogenous Melatonin Induces Drought Stress Tolerance by Promoting Plant Growth and Antioxidant Defence System of Soybean Plants. AoB Plants 2021, 13, plab026. [Google Scholar] [CrossRef]
- Moustafa-Farag, M.; Mahmoud, A.; Arnao, M.B.; Sheteiwy, M.S.; Dafea, M.; Soltan, M.; Elkelish, A.; Hasanuzzaman, M.; Ai, S. Melatonin-Induced Water Stress Tolerance in Plants: Recent Advances. Antioxidants 2020, 9, 809. [Google Scholar] [CrossRef] [PubMed]
- Koh, Y.S.; Wong, S.K.; Ismail, N.H.; Zengin, G.; Duangjai, A.; Saokaew, S.; Phisalprapa, P.; Tan, K.W.; Goh, B.H.; Tang, S.Y. Mitigation of Environmental Stress-Impacts in Plants: Role of Sole and Combinatory Exogenous Application of Glutathione. Front. Plant Sci. 2021, 12, 791205. [Google Scholar] [CrossRef] [PubMed]
- Hosseinifard, M.; Stefaniak, S.; Ghorbani Javid, M.; Soltani, E.; Wojtyla, Ł.; Garnczarska, M. Contribution of Exogenous Proline to Abiotic Stresses Tolerance in Plants: A Review. Int. J. Mol. Sci. 2022, 23, 5186. [Google Scholar] [CrossRef]
- Chowdhury, A.R.; Ghosh, M.; Lal, M.; Pal, A.; Hazra, K.; Acharya, S.; Chaurasiya, A.; Pathak, S. Foliar Spray of Synthetic Osmolytes Alleviates Terminal Heat Stress in Late-Sown Wheat. Int. J. Plant Prod. 2020, 14, 321–333. [Google Scholar] [CrossRef]
- He, X.; Richmond, M.E.; Williams, D.V.; Zheng, W.; Wu, F. Exogenous Glycinebetaine Reduces Cadmium Uptake and Mitigates Cadmium Toxicity in Two Tobacco Genotypes Differing in Cadmium Tolerance. Int. J. Mol. Sci. 2019, 20, 1612. [Google Scholar] [CrossRef]
- Huang, X.; Tanveer, M.; Min, Y.; Shabala, S. Melatonin as a Regulator of Plant Ionic Homeostasis: Implications for Abiotic Stress Tolerance. J. Exp. Bot. 2022, 73, 5886–5902. [Google Scholar] [CrossRef]
- Godoy, F.; Olivos-Hernández, K.; Stange, C.; Handford, M. Abiotic Stress in Crop Species: Improving Tolerance by Applying Plant Metabolites. Plants 2021, 10, 186. [Google Scholar] [CrossRef]
- Kong, M.; Sheng, T.; Liang, J.; Ali, Q.; Gu, Q.; Wu, H.; Chen, J.; Liu, J.; Gao, X. Melatonin and Its Homologs Induce Immune Responses via Receptors TrP47363-TrP13076 in Nicotiana Benthamiana. Front. Plant Sci. 2021, 12, 1197. [Google Scholar] [CrossRef] [PubMed]
- Liang, D.; Ni, Z.; Xia, H.; Xie, Y.; Lv, X.; Wang, J.; Lin, L.; Deng, Q.; Luo, X. Exogenous Melatonin Promotes Biomass Accumulation and Photosynthesis of Kiwifruit Seedlings under Drought Stress. Sci. Hortic. 2019, 246, 34–43. [Google Scholar] [CrossRef]
- Sharma, A.; Wang, J.; Xu, D.; Tao, S.; Chong, S.; Yan, D.; Li, Z.; Yuan, H.; Zheng, B. Melatonin Regulates the Functional Components of Photosynthesis, Antioxidant System, Gene Expression, and Metabolic Pathways to Induce Drought Resistance in Grafted Carya Cathayensis Plants. Sci. Total Environ. 2020, 713, 136675. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Jiang, C.; Ye, T.; Tan, D.-X.; Reiter, R.J.; Zhang, H.; Liu, R.; Chan, Z. Comparative Physiological, Metabolomic, and Transcriptomic Analyses Reveal Mechanisms of Improved Abiotic Stress Resistance in Bermudagrass [Cynodon dactylon (L). Pers.] by Exogenous Melatonin. J. Exp. Bot. 2015, 66, 681–694. [Google Scholar] [CrossRef]
- Lee, H.-J.; Back, K. 2-Hydroxymelatonin Promotes the Resistance of Rice Plant to Multiple Simultaneous Abiotic Stresses (Combined Cold and Drought). J. Pineal Res. 2016, 61, 303–316. [Google Scholar] [CrossRef]
- Fleta-Soriano, E.; Díaz, L.; Bonet, E.; Munné-Bosch, S. Melatonin May Exert a Protective Role against Drought Stress in Maize. J. Agron. Crop Sci. 2017, 203, 286–294. [Google Scholar] [CrossRef]
- Ye, J.; Wang, S.; Deng, X.; Yin, L.; Xiong, B.; Wang, X. Melatonin Increased Maize (Zea mays L.) Seedling Drought Tolerance by Alleviating Drought-Induced Photosynthetic Inhibition and Oxidative Damage. Acta Physiol. Plant. 2016, 38, 48. [Google Scholar] [CrossRef]
- Cui, G.; Zhao, X.; Liu, S.; Sun, F.; Zhang, C.; Xi, Y. Beneficial Effects of Melatonin in Overcoming Drought Stress in Wheat Seedlings. Plant Physiol. Biochem. 2017, 118, 138–149. [Google Scholar] [CrossRef]
- Li, D.; Zhang, D.; Wang, H.; Li, H.; Song, S.; Li, H.; Li, R. Effects of Melatonin on Germination and Amino Acid Content in Different Wheat Varieties Seeds under Polyethylene Glycol Stress. bioRxiv 2019, 710954. [Google Scholar]
- Hossain, Md.S.; Li, J.; Sikdar, A.; Hasanuzzaman, M.; Uzizerimana, F.; Muhammad, I.; Yuan, Y.; Zhang, C.; Wang, C.; Feng, B. Exogenous Melatonin Modulates the Physiological and Biochemical Mechanisms of Drought Tolerance in Tartary Buckwheat (Fagopyrum tataricum (L.) Gaertn). Molecules 2020, 25, 2828. [Google Scholar] [CrossRef]
- Li, M.; Hasan, M.K.; Li, C.; Ahammed, G.J.; Xia, X.; Shi, K.; Zhou, Y.; Reiter, R.J.; Yu, J.; Xu, M. Melatonin Mediates Selenium-induced Tolerance to Cadmium Stress in Tomato Plants. J. Pineal Res. 2016, 61, 291–302. [Google Scholar] [CrossRef]
- Wei, W.; Li, Q.-T.; Chu, Y.-N.; Reiter, R.J.; Yu, X.-M.; Zhu, D.-H.; Zhang, W.-K.; Ma, B.; Lin, Q.; Zhang, J.-S. Melatonin Enhances Plant Growth and Abiotic Stress Tolerance in Soybean Plants. J. Exp. Bot. 2015, 66, 695–707. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; He, S.; Zhan, Y.; Qin, B.; Jin, X.; Wang, M.; Zhang, Y.; Hu, G.; Teng, Z.; Wu, Y. Exogenous Melatonin Reduces the Inhibitory Effect of Osmotic Stress on Photosynthesis in Soybean. PLoS ONE 2019, 14, e0226542. [Google Scholar] [CrossRef] [PubMed]
- Ding, Z.; Tie, W.; Fu, L.; Yan, Y.; Liu, G.; Yan, W.; Li, Y.; Wu, C.; Zhang, J.; Hu, W. Strand-Specific RNA-Seq Based Identification and Functional Prediction of Drought-Responsive LncRNAs in Cassava. BMC Genomics 2019, 20, 214. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Xiao, S.; Zhang, Z.; Zhang, Y.; Sun, H.; Zhang, K.; Wang, X.; Bai, Z.; Li, C.; Liu, L. Melatonin Improves the Germination Rate of Cotton Seeds under Drought Stress by Opening Pores in the Seed Coat. PeerJ 2020, 8, e9450. [Google Scholar] [CrossRef]
- Antoniou, C.; Chatzimichail, G.; Xenofontos, R.; Pavlou, J.J.; Panagiotou, E.; Christou, A.; Fotopoulos, V. Melatonin Systemically Ameliorates Drought Stress-induced Damage in M Edicago Sativa Plants by Modulating Nitro-oxidative Homeostasis and Proline Metabolism. J. Pineal Res. 2017, 62, e12401. [Google Scholar] [CrossRef]
- Liang, B.; Ma, C.; Zhang, Z.; Wei, Z.; Gao, T.; Zhao, Q.; Ma, F.; Li, C. Long-Term Exogenous Application of Melatonin Improves Nutrient Uptake Fluxes in Apple Plants under Moderate Drought Stress. Environ. Exp. Bot. 2018, 155, 650–661. [Google Scholar] [CrossRef]
- Liu, J.; Wang, W.; Wang, L.; Sun, Y. Exogenous Melatonin Improves Seedling Health Index and Drought Tolerance in Tomato. Plant Growth Regul. 2015, 77, 317–326. [Google Scholar] [CrossRef]
- Karaca, P.; Cekic, F.Ö. Exogenous Melatonin-Stimulated Defense Responses in Tomato Plants Treated with Polyethylene Glycol. Int. J. Veg. Sci. 2019, 25, 601–609. [Google Scholar] [CrossRef]
- Kaya, A.; Doganlar, Z.B. Melatonin Improves the Multiple Stress Tolerance in Pepper (Capsicum annuum). Sci. Hortic. 2019, 256, 108509. [Google Scholar] [CrossRef]
- Lee, H.-J.; Back, K. 2-Hydroxymelatonin Confers Tolerance against Combined Cold and Drought Stress in Tobacco, Tomato, and Cucumber as a Potent Anti-Stress Compound in the Evolution of Land Plants. Melatonin Res. 2019, 2, 35–46. [Google Scholar] [CrossRef]
- Khan, M.N.; Zhang, J.; Luo, T.; Liu, J.; Rizwan, M.; Fahad, S.; Xu, Z.; Hu, L. Seed Priming with Melatonin Coping Drought Stress in Rapeseed by Regulating Reactive Oxygen Species Detoxification: Antioxidant Defense System, Osmotic Adjustment, Stomatal Traits and Chloroplast Ultrastructure Perseveration. Ind. Crops Prod. 2019, 140, 111597. [Google Scholar] [CrossRef]
- Wu, Y.; Lian, H.; Mu, X.; Wang, X.; Zhang, Y. Effects of Foliar Spraying Exogenous Melatonin on Physiological and Biochemical Characteristics of Dendranthema Morifolium’Chuju’seedlings under Drought Stress. Acta Bot. Boreali-Occident. Sin. 2016, 36, 2241–2246. [Google Scholar]
- Kabiri, R.; Hatami, A.; Oloumi, H.; Naghizadeh, M.; Nasibi, F.; Tahmasebi, Z. Foliar Application of Melatonin Induces Tolerance to Drought Stress in Moldavian Balm Plants (Dracocephalum moldavica) through Regulating the Antioxidant System. Folia Hortic. 2018, 30, 155. [Google Scholar] [CrossRef]
- Ma, X.; Zhang, J.; Burgess, P.; Rossi, S.; Huang, B. Interactive Effects of Melatonin and Cytokinin on Alleviating Drought-Induced Leaf Senescence in Creeping Bentgrass (Agrostis stolonifera). Environ. Exp. Bot. 2018, 145, 1–11. [Google Scholar] [CrossRef]
- Alam, M.N.; Wang, Y.; Chan, Z. Physiological and Biochemical Analyses Reveal Drought Tolerance in Cool-Season Tall Fescue (Festuca arundinacea) Turf Grass with the Application of Melatonin. Crop Pasture Sci. 2018, 69, 1041–1049. [Google Scholar] [CrossRef]
- Zamani, Z.; Amiri, H.; Ismaili, A. Improving Drought Stress Tolerance in Fenugreek (Trigonella Foenum-Graecum) by Exogenous Melatonin. Plant Biosyst. Int. J. Deal. Asp. Plant Biol. 2020, 154, 643–655. [Google Scholar] [CrossRef]
- Campos, C.N.; Ávila, R.G.; de Souza, K.R.D.; Azevedo, L.M.; Alves, J.D. Melatonin Reduces Oxidative Stress and Promotes Drought Tolerance in Young Coffea arabica L. Plants. Agric. Water Manag. 2019, 211, 37–47. [Google Scholar] [CrossRef]
- Li, J.; Yang, Y.; Sun, K.; Chen, Y.; Chen, X.; Li, X. Exogenous Melatonin Enhances Cold, Salt and Drought Stress Tolerance by Improving Antioxidant Defense in Tea Plant (Camellia sinensis (L.) O. Kuntze). Molecules 2019, 24, 1826. [Google Scholar] [CrossRef]
- Mosaad, I.S.; Serag, A.H.; Moustafa-Farag, M.; Seadh, A.K. Effect of Exogenous Proline Application on Maize Yield and the Optimum Rate of Mineral Nitrogen under Salinity Stress. J. Plant Nutr. 2020, 43, 354–370. [Google Scholar] [CrossRef]
- Elkelish, A.A.; Soliman, M.H.; Alhaithloul, H.A.; El-Esawi, M.A. Selenium Protects Wheat Seedlings against Salt Stress-Mediated Oxidative Damage by up-Regulating Antioxidants and Osmolytes Metabolism. Plant Physiol. Biochem. 2019, 137, 144–153. [Google Scholar] [CrossRef]
- Sheikhalipour, M.; Mohammadi, S.A.; Esmaielpour, B.; Zareei, E.; Kulak, M.; Ali, S.; Nouraein, M.; Bahrami, M.K.; Gohari, G.; Fotopoulos, V. Exogenous Melatonin Increases Salt Tolerance in Bitter Melon by Regulating Ionic Balance, Antioxidant System and Secondary Metabolism-Related Genes. BMC Plant Biol. 2022, 22, 380. [Google Scholar] [CrossRef]
- Rajora, N.; Vats, S.; Raturi, G.; Thakral, V.; Kaur, S.; Rachappanavar, V.; Kumar, M.; Kesarwani, A.K.; Sonah, H.; Sharma, T.R. Seed Priming with Melatonin: A Promising Approach to Combat Abiotic Stress in Plants. Plant Stress 2022, 4, 100071. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, L.; Wang, Z.; Feng, G.; Gao, Q.; Li, X. Induction of Low Temperature Tolerance in Wheat by Pre-Soaking and Parental Treatment with Melatonin. Molecules 2021, 26, 1192. [Google Scholar] [CrossRef] [PubMed]
- Talaat, N.B. Polyamine and Nitrogen Metabolism Regulation by Melatonin and Salicylic Acid Combined Treatment as a Repressor for Salt Toxicity in Wheat (Triticum aestivum L.) Plants. Plant Growth Regul. 2021, 95, 315–329. [Google Scholar] [CrossRef]
- Bahcesular, B.; Yildirim, E.D.; Karaçocuk, M.; Kulak, M.; Karaman, S. Seed Priming with Melatonin Effects on Growth, Essential Oil Compounds and Antioxidant Activity of Basil (Ocimum basilicum L.) under Salinity Stress. Ind. Crops Prod. 2020, 146, 112165. [Google Scholar] [CrossRef]
- Dawood, M.G.; El-Awadi, M.E. Alleviation of Salinity Stress on Vicia faba L. Plants via Seed Priming with Melatonin. Acta Biológica Colomb. 2015, 20, 223–235. [Google Scholar] [CrossRef]
- Shukla, M.R.; Bajwa, V.S.; Freixas-Coutin, J.A.; Saxena, P.K. Salt Stress in Arabidopsis Thaliana Seedlings: Role of Indoleamines in Stress Alleviation. Melatonin Res. 2021, 4, 70–83. [Google Scholar] [CrossRef]
- Zhao, G.; Zhao, Y.; Yu, X.; Kiprotich, F.; Han, H.; Guan, R.; Wang, R.; Shen, W. Nitric Oxide Is Required for Melatonin-Enhanced Tolerance against Salinity Stress in Rapeseed (Brassica napus L.) Seedlings. Int. J. Mol. Sci. 2018, 19, 1912. [Google Scholar] [CrossRef]
- Park, H.-S.; Kazerooni, E.A.; Kang, S.-M.; Al-Sadi, A.M.; Lee, I.-J. Melatonin Enhances the Tolerance and Recovery Mechanisms in Brassica juncea (L.) Czern. under Saline Conditions. Front. Plant Sci. 2021, 12, 593717. [Google Scholar] [CrossRef]
- Castañares, J.L.; Bouzo, C.A. Effect of Exogenous Melatonin on Seed Germination and Seedling Growth in Melon (Cucumis melo L.) under Salt Stress. Hortic. Plant J. 2019, 5, 79–87. [Google Scholar] [CrossRef]
- Liang, C.; Zheng, G.; Li, W.; Wang, Y.; Hu, B.; Wang, H.; Wu, H.; Qian, Y.; Zhu, X.; Tan, D. Melatonin Delays Leaf Senescence and Enhances Salt Stress Tolerance in Rice. J. Pineal Res. 2015, 59, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Moustafa-Farag, M.; Elkelish, A.; Dafea, M.; Khan, M.; Arnao, M.B.; Abdelhamid, M.T.; El-Ezz, A.A.; Almoneafy, A.; Mahmoud, A.; Awad, M. Role of Melatonin in Plant Tolerance to Soil Stressors: Salinity, PH and Heavy Metals. Molecules 2020, 25, 5359. [Google Scholar] [CrossRef]
- Martinez, V.; Nieves-Cordones, M.; Lopez-Delacalle, M.; Rodenas, R.; Mestre, T.C.; Garcia-Sanchez, F.; Rubio, F.; Nortes, P.A.; Mittler, R.; Rivero, R.M. Tolerance to Stress Combination in Tomato Plants: New Insights in the Protective Role of Melatonin. Molecules 2018, 23, 535. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Chang, J.; Chen, H.; Wang, Z.; Gu, X.; Wei, C.; Zhang, Y.; Ma, J.; Yang, J.; Zhang, X. Exogenous Melatonin Confers Salt Stress Tolerance to Watermelon by Improving Photosynthesis and Redox Homeostasis. Front. Plant Sci. 2017, 8, 295. [Google Scholar] [CrossRef]
- Iqbal, N.; Fatma, M.; Gautam, H.; Umar, S.; Sofo, A.; D’ippolito, I.; Khan, N.A. The Crosstalk of Melatonin and Hydrogen Sulfide Determines Photosynthetic Performance by Regulation of Carbohydrate Metabolism in Wheat under Heat Stress. Plants 2021, 10, 1778. [Google Scholar] [CrossRef]
- Zhang, J.; Shi, Y.; Zhang, X.; Du, H.; Xu, B.; Huang, B. Melatonin Suppression of Heat-Induced Leaf Senescence Involves Changes in Abscisic Acid and Cytokinin Biosynthesis and Signaling Pathways in Perennial Ryegrass (Lolium perenne L.). Environ. Exp. Bot. 2017, 138, 36–45. [Google Scholar] [CrossRef]
- Jahan, M.S.; Shu, S.; Wang, Y.; Chen, Z.; He, M.; Tao, M.; Sun, J.; Guo, S. Melatonin Alleviates Heat-Induced Damage of Tomato Seedlings by Balancing Redox Homeostasis and Modulating Polyamine and Nitric Oxide Biosynthesis. BMC Plant Biol. 2019, 19, 414. [Google Scholar] [CrossRef]
- ur Rehman, H.; Alharby, H.F.; Bamagoos, A.A.; Abdelhamid, M.T.; Rady, M.M. Sequenced Application of Glutathione as an Antioxidant with an Organic Biostimulant Improves Physiological and Metabolic Adaptation to Salinity in Wheat. Plant Physiol. Biochem. 2021, 158, 43–52. [Google Scholar] [CrossRef]
- Yao, M.; Ge, W.; Zhou, Q.; Zhou, X.; Luo, M.; Zhao, Y.; Wei, B.; Ji, S. Exogenous Glutathione Alleviates Chilling Injury in Postharvest Bell Pepper by Modulating the Ascorbate-Glutathione (AsA-GSH) Cycle. Food Chem. 2021, 352, 129458. [Google Scholar] [CrossRef]
- Khan, A.; Numan, M.; Khan, A.L.; Lee, I.-J.; Imran, M.; Asaf, S.; Al-Harrasi, A. Melatonin: Awakening the Defense Mechanisms during Plant Oxidative Stress. Plants 2020, 9, 407. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Guo, Y.; Lan, Z.; Xu, K.; Chang, J.; Ahammed, G.J.; Ma, J.; Wei, C.; Zhang, X. Methyl Jasmonate Mediates Melatonin-Induced Cold Tolerance of Grafted Watermelon Plants. Hortic. Res. 2021, 8, 57. [Google Scholar] [CrossRef] [PubMed]
- Ding, F.; Wang, M.; Liu, B.; Zhang, S. Exogenous Melatonin Mitigates Photoinhibition by Accelerating Non-Photochemical Quenching in Tomato Seedlings Exposed to Moderate Light during Chilling. Front. Plant Sci. 2017, 8, 244. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; Hu, Z.; Xie, Y.; Chan, Z.; Chen, K.; Amombo, E.; Chen, L.; Fu, J. Alleviation of Cold Damage to Photosystem II and Metabolisms by Melatonin in Bermudagrass. Front. Plant Sci. 2015, 6, 925. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Wei, J.-P.; Scott, E.R.; Liu, J.-W.; Guo, S.; Li, Y.; Zhang, L.; Han, W.-Y. Exogenous Melatonin Alleviates Cold Stress by Promoting Antioxidant Defense and Redox Homeostasis in Camellia sinensis L. Molecules 2018, 23, 165. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Fu, X.; Han, L.; Xu, C.; Liu, C.; Bi, H.; Ai, X. Nitric Oxide Functions as a Downstream Signal for Melatonin-Induced Cold Tolerance in Cucumber Seedlings. Front. Plant Sci. 2021, 12, 1432. [Google Scholar] [CrossRef]
- Tiwari, G.; Duraivadivel, P.; Sharma, S. 1-Aminocyclopropane-1-Carboxylic Acid Deaminase Producing Beneficial Rhizobacteria Ameliorate the Biomass Characters of Panicum Maximum Jacq. by Mitigating Drought and Salt Stress. Sci. Rep. 2018, 8, 17513. [Google Scholar] [CrossRef]
- Ni, J.; Wang, Q.; Shah, F.A.; Liu, W.; Wang, D.; Huang, S.; Fu, S.; Wu, L. Exogenous Melatonin Confers Cadmium Tolerance by Counterbalancing the Hydrogen Peroxide Homeostasis in Wheat Seedlings. Molecules 2018, 23, 799. [Google Scholar] [CrossRef]
- Gu, Q.; Chen, Z.; Yu, X.; Cui, W.; Pan, J.; Zhao, G.; Xu, S.; Wang, R.; Shen, W. Melatonin Confers Plant Tolerance against Cadmium Stress via the Decrease of Cadmium Accumulation and Reestablishment of MicroRNA-Mediated Redox Homeostasis. Plant Sci. 2017, 261, 28–37. [Google Scholar] [CrossRef]
- Lin, L.; Li, J.; Chen, F.; Liao, M.; Tang, Y.; Liang, D.; Xia, H.; Lai, Y.; Wang, X.; Chen, C. Effects of Melatonin on the Growth and Cadmium Characteristics of Cyphomandra Betacea Seedlings. Environ. Monit. Assess. 2018, 190, 119. [Google Scholar] [CrossRef]
- Nawaz, M.A.; Jiao, Y.; Chen, C.; Shireen, F.; Zheng, Z.; Imtiaz, M.; Bie, Z.; Huang, Y. Melatonin Pretreatment Improves Vanadium Stress Tolerance of Watermelon Seedlings by Reducing Vanadium Concentration in the Leaves and Regulating Melatonin Biosynthesis and Antioxidant-Related Gene Expression. J. Plant Physiol. 2018, 220, 115–127. [Google Scholar] [CrossRef] [PubMed]
- Kołodziejczyk, I.; Dzitko, K.; Szewczyk, R.; Posmyk, M.M. Exogenous Melatonin Expediently Modifies Proteome of Maize (Zea mays L.) Embryo during Seed Germination. Acta Physiol. Plant. 2016, 38, 146. [Google Scholar] [CrossRef]
- David, O.; Jolayemi, O.; Akomolafe, G.; Adegoke, M. Lead Sequestration, Immobilization and Xylem Cavitation in Melatonin-Primed Amaranthus Cruentus. Plant Physiol. Rep. 2021, 26, 162–171. [Google Scholar] [CrossRef]
- El-Beltagi, H.S.; Mohamed, H.I.; Sofy, M.R. Role of Ascorbic Acid, Glutathione and Proline Applied as Singly or in Sequence Combination in Improving Chickpea Plant through Physiological Change and Antioxidant Defense under Different Levels of Irrigation Intervals. Molecules 2020, 25, 1702. [Google Scholar] [CrossRef] [PubMed]
- Hasanuzzaman, M.; Fujita, M. Selenium Pretreatment Upregulates the Antioxidant Defense and Methylglyoxal Detoxification System and Confers Enhanced Tolerance to Drought Stress in Rapeseed Seedlings. Biol. Trace Elem. Res. 2011, 143, 1758–1776. [Google Scholar] [CrossRef] [PubMed]
- Garg, B.; Jaiswal, J.P.; Misra, S.; Tripathi, B.N.; Prasad, M. A Comprehensive Study on Dehydration-Induced Antioxidative Responses during Germination of Indian Bread Wheat (Triticum aestivum L. Em Thell) Cultivars Collected from Different Agroclimatic Zones. Physiol. Mol. Biol. Plants 2012, 18, 217–228. [Google Scholar] [CrossRef]
- Nahar, K.; Hasanuzzaman, M.; Alam, M.; Fujita, M. Glutathione-Induced Drought Stress Tolerance in Mung Bean: Coordinated Roles of the Antioxidant Defence and Methylglyoxal Detoxification Systems. AoB Plants 2015, 7, plv069. [Google Scholar] [CrossRef]
- Chen, J.-H.; Jiang, H.-W.; Hsieh, E.-J.; Chen, H.-Y.; Chien, C.-T.; Hsieh, H.-L.; Lin, T.-P. Drought and Salt Stress Tolerance of an Arabidopsis Glutathione S-Transferase U17 Knockout Mutant Are Attributed to the Combined Effect of Glutathione and Abscisic Acid. Plant Physiol. 2012, 158, 340–351. [Google Scholar] [CrossRef]
- Al-Elwany, O.A.; Mohamed, G.F.; Abdurrahman, H.A.; LATEF, A.A.A. Exogenous Glutathione-Mediated Tolerance to Deficit Irrigation in Salt-Affected Capsicum frutescence (L.) Plants Is Connected with Higher Antioxidant Content and Ionic Homeostasis. Not. Bot. Hortic. Agrobot. Cluj Napoca 2020, 48, 1957–1979. [Google Scholar] [CrossRef]
- Seleiman, M.F.; Semida, W.M.; Rady, M.M.; Mohamed, G.F.; Hemida, K.A.; Alhammad, B.A.; Hassan, M.M.; Shami, A. Sequential Application of Antioxidants Rectifies Ion Imbalance and Strengthens Antioxidant Systems in Salt-Stressed Cucumber. Plants 2020, 9, 1783. [Google Scholar] [CrossRef]
- Semida, W.M.; El-Mageed, A.; Taia, A.; Abdalla, R.M.; Hemida, K.A.; Howladar, S.; Leilah, A.A.; Rady, M.O. Sequential Antioxidants Foliar Application Can Alleviate Negative Consequences of Salinity Stress in Vicia faba L. Plants 2021, 10, 914. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Wen, Z.; Zhang, J.; Chen, X.; Cui, J.; Xu, W.; Liu, H. Exogenous Glutathione Alleviates Salt-Induced Oxidative Stress in Tomato Seedlings by Regulating Glutathione Metabolism, Redox Status, and the Antioxidant System. Sci. Hortic. 2017, 220, 90–101. [Google Scholar] [CrossRef]
- El-Shabrawi, H.; Kumar, B.; Kaul, T.; Reddy, M.K.; Singla-Pareek, S.L.; Sopory, S.K. Redox Homeostasis, Antioxidant Defense, and Methylglyoxal Detoxification as Markers for Salt Tolerance in Pokkali Rice. Protoplasma 2010, 245, 85–96. [Google Scholar] [CrossRef]
- Ding, X.; Jiang, Y.; He, L.; Zhou, Q.; Yu, J.; Hui, D.; Huang, D. Exogenous Glutathione Improves High Root-Zone Temperature Tolerance by Modulating Photosynthesis, Antioxidant and Osmolytes Systems in Cucumber Seedlings. Sci. Rep. 2016, 6, 35424. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Cai, J.; Liu, F.; Dai, T.; Cao, W.; Wollenweber, B.; Jiang, D. Multiple Heat Priming Enhances Thermo-Tolerance to a Later High Temperature Stress via Improving Subcellular Antioxidant Activities in Wheat Seedlings. Plant Physiol. Biochem. 2014, 74, 185–192. [Google Scholar] [CrossRef]
- Zou, M.; Yuan, L.; Zhu, S.; Liu, S.; Ge, J.; Wang, C. Response of Osmotic Adjustment and Ascorbate-Glutathione Cycle to Heat Stress in a Heat-Sensitive and a Heat-Tolerant Genotype of Wucai (Brassica campestris L.). Sci. Hortic. 2016, 211, 87–94. [Google Scholar] [CrossRef]
- Almeselmani, M.; Deshmukh, P.; Sairam, R. High Temperature Stress Tolerance in Wheat Genotypes: Role of Antioxidant Defence Enzymes. Acta Agron. Hung. 2009, 57, 1–14. [Google Scholar] [CrossRef]
- Park, S.-I.; Kim, J.-J.; Kim, H.-S.; Kim, Y.-S.; Yoon, H.-S. Enhanced Glutathione Content Improves Lateral Root Development and Grain Yield in Rice Plants. Plant Mol. Biol. 2021, 105, 365–383. [Google Scholar] [CrossRef]
- Lukatkin, A.S.; Anjum, N.A. Control of Cucumber (Cucumis sativus L.) Tolerance to Chilling Stress—Evaluating the Role of Ascorbic Acid and Glutathione. Front. Environ. Sci. 2014, 2, 62. [Google Scholar] [CrossRef]
- Li, Z.-G.; Yuan, L.-X.; Wang, Q.-L.; Ding, Z.-L.; Dong, C.-Y. Combined Action of Antioxidant Defense System and Osmolytes in Chilling Shock-Induced Chilling Tolerance in Jatropha Curcas Seedlings. Acta Physiol. Plant. 2013, 35, 2127–2136. [Google Scholar] [CrossRef]
- Alamri, S.; Kushwaha, B.K.; Singh, V.P.; Siddiqui, M.H.; Al-Amri, A.A.; Alsubaie, Q.D.; Ali, H.M. Ascorbate and Glutathione Independently Alleviate Arsenate Toxicity in Brinjal but Both Require Endogenous Nitric Oxide. Physiol. Plant. 2021, 173, 276–286. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Lin, K.; Chen, H.; Qi, Z.; Liu, B.; Cao, F.; Chen, H.; Wu, F. Metabolome Analysis Revealed the Mechanism of Exogenous Glutathione to Alleviate Cadmium Stress in Maize (Zea mays L.) Seedlings. Plants 2021, 10, 105. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Nahar, K.; Rahman, A.; Mahmud, J.A.; Alharby, H.F.; Fujita, M. Exogenous Glutathione Attenuates Lead-Induced Oxidative Stress in Wheat by Improving Antioxidant Defense and Physiological Mechanisms. J. Plant Interact. 2018, 13, 203–212. [Google Scholar] [CrossRef]
- Wang, F.; Chen, F.; Cai, Y.; Zhang, G.; Wu, F. Modulation of Exogenous Glutathione in Ultrastructure and Photosynthetic Performance against Cd Stress in the Two Barley Genotypes Differing in Cd Tolerance. Biol. Trace Elem. Res. 2011, 144, 1275–1288. [Google Scholar] [CrossRef] [PubMed]
- Hasan, M.K.; Liu, C.; Wang, F.; Ahammed, G.J.; Zhou, J.; Xu, M.-X.; Yu, J.-Q.; Xia, X.-J. Glutathione-Mediated Regulation of Nitric Oxide, S-Nitrosothiol and Redox Homeostasis Confers Cadmium Tolerance by Inducing Transcription Factors and Stress Response Genes in Tomato. Chemosphere 2016, 161, 536–545. [Google Scholar] [CrossRef]
- Cao, F.; Cai, Y.; Liu, L.; Zhang, M.; He, X.; Zhang, G.; Wu, F. Differences in Photosynthesis, Yield and Grain Cadmium Accumulation as Affected by Exogenous Cadmium and Glutathione in the Two Rice Genotypes. Plant Growth Regul. 2015, 75, 715–723. [Google Scholar] [CrossRef]
- Anjum, N.A.; Umar, S.; Ahmad, A.; Iqbal, M.; Khan, N.A. Sulphur Protects Mustard (Brassica campestris L.) from Cadmium Toxicity by Improving Leaf Ascorbate and Glutathione. Plant Growth Regul. 2008, 54, 271–279. [Google Scholar] [CrossRef]
- Chen, F.; Wang, F.; Wu, F.; Mao, W.; Zhang, G.; Zhou, M. Modulation of Exogenous Glutathione in Antioxidant Defense System against Cd Stress in the Two Barley Genotypes Differing in Cd Tolerance. Plant Physiol. Biochem. 2010, 48, 663–672. [Google Scholar] [CrossRef]
- Cai, Y.; Cao, F.; Cheng, W.; Zhang, G.; Wu, F. Modulation of Exogenous Glutathione in Phytochelatins and Photosynthetic Performance against Cd Stress in the Two Rice Genotypes Differing in Cd Tolerance. Biol. Trace Elem. Res. 2011, 143, 1159–1173. [Google Scholar] [CrossRef]
- Ilyas, N.; Amjid, M.W.; Saleem, M.A.; Khan, W.; Wattoo, F.M.; Rana, R.M.; Maqsood, R.H.; Zahid, A.; Shah, G.A.; Anwar, A. Quantitative Trait Loci (QTL) Mapping for Physiological and Biochemical Attributes in a Pasban90/Frontana Recombinant Inbred Lines (RILs) Population of Wheat (Triticum Aestivum) under Salt Stress Condition. Saudi J. Biol. Sci. 2020, 27, 341–351. [Google Scholar] [CrossRef]
- Farooq, M.; Nawaz, A.; Chaudhry, M.; Indrasti, R.; Rehman, A. Improving Resistance against Terminal Drought in Bread Wheat by Exogenous Application of Proline and Gamma-aminobutyric Acid. J. Agron. Crop Sci. 2017, 203, 464–472. [Google Scholar] [CrossRef]
- Elewa, T.A.; Sadak, M.S.; Saad, A.M. Proline Treatment Improves Physiological Responses in Quinoa Plants under Drought Stress. Biosci. Res. 2017, 14, 21–33. [Google Scholar]
- Zouari, M.; Hassena, A.B.; Trabelsi, L.; Rouina, B.B.; Decou, R.; Labrousse, P. Exogenous Proline-Mediated Abiotic Stress Tolerance in Plants: Possible Mechanisms. In Osmoprotectant-Mediated Abiotic Stress Tolerance in Plants; Springer: Berlin/Heidelberg, Germany, 2019; pp. 99–121. [Google Scholar]
- Kaya, C.; Tuna, A.L.; Ashraf, M.; Altunlu, H. Improved Salt Tolerance of Melon (Cucumis melo L.) by the Addition of Proline and Potassium Nitrate. Environ. Exp. Bot. 2007, 60, 397–403. [Google Scholar] [CrossRef]
- Huang, Y.; Bie, Z.; Liu, Z.; Zhen, A.; Wang, W. Protective Role of Proline against Salt Stress Is Partially Related to the Improvement of Water Status and Peroxidase Enzyme Activity in Cucumber. Soil Sci. Plant Nutr. 2009, 55, 698–704. [Google Scholar] [CrossRef]
- Sabagh, A.E.; Sorour, S.; Ragab, A.; Saneoka, H.; Islam, M. The Effect of Exogenous Application of Proline and Glycine Betaineon the Nodule Activity of Soybean under Saline Condition. J. Agric. Biotechnol. 2017, 2, 01–05. [Google Scholar]
- Iqbal, A.; Iftikhar, I.; Nawaz, H.; Nawaz, M. Role of Proline to Induce Salinity Tolerance in Sunflower (Helianthus annusl). Sci. Technol. Dev. 2014, 33, 88–93. [Google Scholar]
- de Freitas, P.A.F.; de Souza Miranda, R.; Marques, E.C.; Prisco, J.T.; Gomes-Filho, E. Salt Tolerance Induced by Exogenous Proline in Maize Is Related to Low Oxidative Damage and Favorable Ionic Homeostasis. J. Plant Growth Regul. 2018, 37, 911–924. [Google Scholar]
- Rady, M.M.; Kuşvuran, A.; Alharby, H.F.; Alzahrani, Y.; Kuşvuran, S. Pretreatment with Proline or an Organic Bio-Stimulant Induces Salt Tolerance in Wheat Plants by Improving Antioxidant Redox State and Enzymatic Activities and Reducing the Oxidative Stress. J. Plant Growth Regul. 2019, 38, 449–462. [Google Scholar]
- de Freitas, P.A.F.; de Carvalho, H.H.; Costa, J.H.; Miranda, R.d.S.; Saraiva, K.D.d.C.; de Oliveira, F.D.B.; Coelho, D.G.; Prisco, J.T.; Gomes-Filho, E. Salt Acclimation in Sorghum Plants by Exogenous Proline: Physiological and Biochemical Changes and Regulation of Proline Metabolism. Plant Cell Rep. 2019, 38, 403–416. [Google Scholar] [CrossRef]
- El Moukhtari, A.; Cabassa-Hourton, C.; Farissi, M.; Savouré, A. How Does Proline Treatment Promote Salt Stress Tolerance during Crop Plant Development? Front. Plant Sci. 2020, 11, 1127. [Google Scholar] [CrossRef]
- Wani, A.; Ahmad, A.; Hayat, S.; Tahir, I. Is Foliar Spray of Proline Sufficient for Mitigation of Salt Stress in Brassica Juncea Cultivars? Environ. Sci. Pollut. Res. 2016, 23, 13413–13423. [Google Scholar] [CrossRef] [PubMed]
- Jamil, M.; Kharal, M.A.; Ahmad, M.; Abbasi, G.H.; Nazli, F.; Hussain, A.; Akhtar, M.F.-Z. Inducing Salinity Tolerance in Red Pepper (Capsicum annuum L.) through Exogenous Application of Proline and L-Tryptophan. Soil Environ. 2018, 37, 160–168. [Google Scholar]
- Qirat, M.; Shahbaz, M.; Perveen, S. Beneficial Role of Foliar-Applied Proline on Carrot (Daucus carota L.) under Saline Conditions. Pak J Bot 2018, 50, 1735–1744. [Google Scholar]
- Abdelhamid, M.T.; Rady, M.M.; Osman, A.S.; Abdalla, M.A. Exogenous Application of Proline Alleviates Salt-Induced Oxidative Stress in Phaseolus vulgaris L. Plants. J. Hortic. Sci. Biotechnol. 2013, 88, 439–446. [Google Scholar] [CrossRef]
- Shaddad, M. The Effect of Proline Application on the Physiology Ofraphanus Sativus Plants Grown under Salinity Stress. Biol. Plant. 1990, 32, 104–112. [Google Scholar] [CrossRef]
- Orsini, F.; Pennisi, G.; Mancarella, S.; Al Nayef, M.; Sanoubar, R.; Nicola, S.; Gianquinto, G. Hydroponic Lettuce Yields Are Improved under Salt Stress by Utilizing White Plastic Film and Exogenous Applications of Proline. Sci. Hortic. 2018, 233, 283–293. [Google Scholar] [CrossRef]
- Hussain, R.; Ayyub, C.M.; Shaheen, M.R.; Rashid, S.; Nafees, M.; Ali, S.; Butt, M.; Ali, M.; Maqsood, A.; Fiaz, S. Regulation of Osmotic Balance and Increased Antioxidant Activities under Heat Stress in Abelmoschus esculentus L. Triggered by Exogenous Proline Application. Agronomy 2021, 11, 685. [Google Scholar] [CrossRef]
- Priya, M.; Sharma, L.; Singh, I.; Bains, T.; Siddique, K.H.; Bindumadhava, H.; Nair, R.M.; Nayyar, H. Securing Reproductive Function in Mungbean Grown under High Temperature Environment with Exogenous Application of Proline. Plant Physiol. Biochem. 2019, 140, 136–150. [Google Scholar] [CrossRef]
- Mohammadrezakhani, S.; Hajilou, J.; Rezanejad, F.; Zaare-Nahandi, F. Assessment of Exogenous Application of Proline on Antioxidant Compounds in Three Citrus Species under Low Temperature Stress. J. Plant Interact. 2019, 14, 347–358. [Google Scholar] [CrossRef]
- Alyemeni, M.N.; Hayat, Q.; Hayat, S.; Faizan, M.; Faraz, A. Exogenous Proline Application Enhances the Efficiency of Nitrogen Fixation and Assimilation in Chickpea Plants Exposed to Cadmium. Legume Res. Int. J. 2016, 39, 221–227. [Google Scholar] [CrossRef]
- Zouari, M.; Ahmed, C.B.; Elloumi, N.; Bellassoued, K.; Delmail, D.; Labrousse, P.; Abdallah, F.B.; Rouina, B.B. Impact of Proline Application on Cadmium Accumulation, Mineral Nutrition and Enzymatic Antioxidant Defense System of Olea europaea L. Cv Chemlali Exposed to Cadmium Stress. Ecotoxicol. Environ. Saf. 2016, 128, 195–205. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, M.; Sharma, S.; Kaur, N.; Pathania, D.; Bhandhari, K.; Kaushal, N.; Kaur, R.; Singh, K.; Srivastava, A.; Nayyar, H. Exogenous Proline Application Reduces Phytotoxic Effects of Selenium by Minimising Oxidative Stress and Improves Growth in Bean (Phaseolus vulgaris L.) Seedlings. Biol. Trace Elem. Res. 2011, 140, 354–367. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Riaz, M.; Jiang, C. Exogenous Application of Proline Alleviates B-Deficiency-Induced Injury While Aggravates Aluminum Toxicity in Trifoliate Orange Seedlings. Sci. Hortic. 2020, 268, 109372. [Google Scholar] [CrossRef]
- Singh, M.; Singh, V.P.; Dubey, G.; Prasad, S.M. Exogenous Proline Application Ameliorates Toxic Effects of Arsenate in Solanum Melongena L. Seedlings. Ecotoxicol. Environ. Saf. 2015, 117, 164–173. [Google Scholar] [CrossRef]
- Karalija, E.; Selović, A. The Effect of Hydro and Proline Seed Priming on Growth, Proline and Sugar Content, and Antioxidant Activity of Maize under Cadmium Stress. Environ. Sci. Pollut. Res. 2018, 25, 33370–33380. [Google Scholar] [CrossRef] [PubMed]
- Zulfiqar, F.; Ashraf, M.; Siddique, K.H. Role of Glycine Betaine in the Thermotolerance of Plants. Agronomy 2022, 12, 276. [Google Scholar] [CrossRef]
- Annunziata, M.G.; Ciarmiello, L.F.; Woodrow, P.; Dell’Aversana, E.; Carillo, P. Spatial and Temporal Profile of Glycine Betaine Accumulation in Plants under Abiotic Stresses. Front. Plant Sci. 2019, 10, 230. [Google Scholar] [CrossRef]
- Gupta, N.; Thind, S.K.; Bains, N.S. Glycine Betaine Application Modifies Biochemical Attributes of Osmotic Adjustment in Drought Stressed Wheat. Plant Growth Regul. 2014, 72, 221–228. [Google Scholar] [CrossRef]
- Raza, M.; Saleem, M.; Shah, G.; Khan, I.; Raza, A. Exogenous Application of Glycinebetaine and Potassium for Improving Water Relations and Grain Yield of Wheat under Drought. J. Soil Sci. Plant Nutr. 2014, 14, 348–364. [Google Scholar] [CrossRef]
- Anjum, S.; Farooq, M.; Wang, L.C.; Xue, L.; Wang, S.; Wang, L.; Zhang, S.; Chen, M. Gas Exchange and Chlorophyll Synthesis of Maize Cultivars Are Enhanced by Exogenously-Applied Glycinebetaine under Drought Conditions. Plant Soil Environ. 2011, 57, 326–331. [Google Scholar] [CrossRef]
- Jokinen, K.; Somersalo, S.; Mäkelä, P.; Urbano, P.; Rojo, C.; González, J.; Soler, J.; Usano, M.; Moure, J.; Moya, M. Glycinebetaine from Sugar Beet Enhances the Yield of ‘Field-Grown’Tomatoes. In VI International Symposium on Processing Tomato & Workshop on Irrigation & Fertigation of Processing Tomato; ISHS: Leuven, Belgium, 1998; pp. 233–236. [Google Scholar]
- Osman, H.S. Enhancing Antioxidant–Yield Relationship of Pea Plant under Drought at Different Growth Stages by Exogenously Applied Glycine Betaine and Proline. Ann. Agric. Sci. 2015, 60, 389–402. [Google Scholar] [CrossRef]
- Agboma, P.; Sinclair, T.; Jokinen, K.; Peltonen-Sainio, P.; Pehu, E. An Evaluation of the Effect of Exogenous Glycinebetaine on the Growth and Yield of Soybean: Timing of Application, Watering Regimes and Cultivars. Field Crops Res. 1997, 54, 51–64. [Google Scholar] [CrossRef]
- Manaf, H.H. Beneficial Effects of Exogenous Selenium, Glycine Betaine and Seaweed Extract on Salt Stressed Cowpea Plant. Ann. Agric. Sci. 2016, 61, 41–48. [Google Scholar] [CrossRef]
- Osman, H.S.; Salim, B.B. Influence of Exogenous Application of Some Phytoprotectants on Growth, Yield and Pod Quality of Snap Bean under NaCl Salinity. Ann. Agric. Sci. 2016, 61, 1–13. [Google Scholar] [CrossRef][Green Version]
- Malekzadeh, P. Influence of Exogenous Application of Glycinebetaine on Antioxidative System and Growth of Salt-Stressed Soybean Seedlings (Glycine max L.). Physiol. Mol. Biol. Plants 2015, 21, 225–232. [Google Scholar] [CrossRef]
- Hu, L.; Hu, T.; Zhang, X.; Pang, H.; Fu, J. Exogenous Glycine Betaine Ameliorates the Adverse Effect of Salt Stress on Perennial Ryegrass. J. Am. Soc. Hortic. Sci. 2012, 137, 38–46. [Google Scholar] [CrossRef]
- Rajasekaran, L.; Kriedemann, P.; Aspinall, D.; Paleg, L.G. Physiological Significance of Proline and Glycinebetaine: Maintaining Photosynthesis during NaCl Stress in Wheat. Photosynthetica 1998, 34, 357–366. [Google Scholar] [CrossRef]
- Li, S.; Li, F.; Wang, J.; Zhang, W.; Meng, Q.; Chen, T.H.; Murata, N.; Yang, X. Glycinebetaine Enhances the Tolerance of Tomato Plants to High Temperature during Germination of Seeds and Growth of Seedlings. Plant Cell Environ. 2011, 34, 1931–1943. [Google Scholar] [CrossRef]
- Oukarroum, A.; El Madidi, S.; Strasser, R. Exogenous Glycine Betaine and Proline Play a Protective Role in Heat-Stressed Barley Leaves (Hordeum vulgare L.): A Chlorophyll a Fluorescence Study. Plant Biosyst. Int. J. Deal. Asp. Plant Biol. 2012, 146, 1037–1043. [Google Scholar]
- Wahid, A.; Shabbir, A. Induction of Heat Stress Tolerance in Barley Seedlings by Pre-Sowing Seed Treatment with Glycinebetaine. Plant Growth Regul. 2005, 46, 133–141. [Google Scholar] [CrossRef]
- Rasheed, R.; Wahid, A.; Farooq, M.; Hussain, I.; Basra, S. Role of Proline and Glycinebetaine Pretreatments in Improving Heat Tolerance of Sprouting Sugarcane (Saccharum sp.) Buds. Plant Growth Regul. 2011, 65, 35–45. [Google Scholar] [CrossRef]
- Sorwong, A.; Sakhonwasee, S. Foliar Application of Glycine Betaine Mitigates the Effect of Heat Stress in Three Marigold (Tagetes erecta) Cultivars. Hortic. J. 2015, 84, 161–171. [Google Scholar] [CrossRef]
- Chen, W.; Li, P.; Chen, T. Glycinebetaine Increases Chilling Tolerance and Reduces Chilling-induced Lipid Peroxidation in Zea mays L. Plant Cell Environ. 2000, 23, 609–618. [Google Scholar] [CrossRef]
- Allard, F.; Houde, M.; Kröl, M.; Ivanov, A.; Huner, N.P.; Sarhan, F. Betaine Improves Freezing Tolerance in Wheat. Plant Cell Physiol. 1998, 39, 1194–1202. [Google Scholar] [CrossRef]
- Shan, T.; Jin, P.; Zhang, Y.; Huang, Y.; Wang, X.; Zheng, Y. Exogenous Glycine Betaine Treatment Enhances Chilling Tolerance of Peach Fruit during Cold Storage. Postharvest Biol. Technol. 2016, 114, 104–110. [Google Scholar] [CrossRef]
- Zhao, Y.; Aspinall, D.; Paleg, L. Protection of Membrane Integrity in Medicago sativa L. by Glycinebetaine against the Effects of Freezing. J. Plant Physiol. 1992, 140, 541–543. [Google Scholar] [CrossRef]
- Park, E.-J.; Jeknic, Z.; Chen, T.H. Exogenous Application of Glycinebetaine Increases Chilling Tolerance in Tomato Plants. Plant Cell Physiol. 2006, 47, 706–714. [Google Scholar] [CrossRef]
- Ali, S.; Chaudhary, A.; Rizwan, M.; Anwar, H.T.; Adrees, M.; Farid, M.; Irshad, M.K.; Hayat, T.; Anjum, S.A. Alleviation of Chromium Toxicity by Glycinebetaine Is Related to Elevated Antioxidant Enzymes and Suppressed Chromium Uptake and Oxidative Stress in Wheat (Triticum aestivum L.). Environ. Sci. Pollut. Res. 2015, 22, 10669–10678. [Google Scholar] [CrossRef]
- Farooq, M.; Ali, S.; Hameed, A.; Bharwana, S.; Rizwan, M.; Ishaque, W.; Farid, M.; Mahmood, K.; Iqbal, Z. Cadmium Stress in Cotton Seedlings: Physiological, Photosynthesis and Oxidative Damages Alleviated by Glycinebetaine. South Afr. J. Bot. 2016, 104, 61–68. [Google Scholar] [CrossRef]
- Ali, S.; Abbas, Z.; Seleiman, M.F.; Rizwan, M.; YavaŞ, İ.; Alhammad, B.A.; Shami, A.; Hasanuzzaman, M.; Kalderis, D. Glycine Betaine Accumulation, Significance and Interests for Heavy Metal Tolerance in Plants. Plants 2020, 9, 896. [Google Scholar] [CrossRef]
- Jabeen, N.; Abbas, Z.; Iqbal, M.; Rizwan, M.; Jabbar, A.; Farid, M.; Ali, S.; Ibrahim, M.; Abbas, F. Glycinebetaine Mediates Chromium Tolerance in Mung Bean through Lowering of Cr Uptake and Improved Antioxidant System. Arch. Agron. Soil Sci. 2016, 62, 648–662. [Google Scholar] [CrossRef]
- Fan, W.; Zhang, M.; Zhang, H.; Zhang, P. Improved Tolerance to Various Abiotic Stresses in Transgenic Sweet Potato (Ipomoea Batatas) Expressing Spinach Betaine Aldehyde Dehydrogenase. PLoS ONE 2012, 7, e37344. [Google Scholar] [CrossRef] [PubMed]
- Ali, Q.; Anwar, F.; Ashraf, M.; Saari, N.; Perveen, R. Ameliorating Effects of Exogenously Applied Proline on Seed Composition, Seed Oil Quality and Oil Antioxidant Activity of Maize (Zea mays L.) under Drought Stress. Int. J. Mol. Sci. 2013, 14, 818–835. [Google Scholar] [CrossRef] [PubMed]
- Stepien, P. Effects of the Exogenous Glycinebetaine on Photosynthetic Apparatus in Cucumber Leaves Challenging Al Stress. In Proceedings of the 18 th International Conference on Heavy Metals in the Environment, Ghent, Belgium, 12–15 September 2016. [Google Scholar]
- Kumar, P.; Tokas, J.; Singal, H. Amelioration of Chromium VI Toxicity in Sorghum (Sorghum bicolor L.) Using Glycine Betaine. Sci. Rep. 2019, 9, 16020. [Google Scholar] [CrossRef]
- Dubey, A.K.; Kumar, N.; Ranjan, R.; Gautam, A.; Pande, V.; Sanyal, I.; Mallick, S. Application of Glycine Reduces Arsenic Accumulation and Toxicity in Oryza sativa L. by Reducing the Expression of Silicon Transporter Genes. Ecotoxicol. Environ. Saf. 2018, 148, 410–417. [Google Scholar] [CrossRef] [PubMed]

| Drought Stress | |||||
|---|---|---|---|---|---|
| Crop | Common Name | Melatonin Dose | Treatment Method | Response to Treatment | Reference(s) |
| Arabidopsis thaliana | Arabidopsis | 50 µM | Supplemented with nutrient solution | Upregulated stress-responsive genes and soluble sugars | [14] |
| Oryza sativa | Rice | 100 µM | Pretreatment in distilled water for growing | Improved plant growth, osmoprotectants, stress-responsive genes, and ROS scavenging; reduced electrolyte leakage | [15] |
| Zea mays | Maize | 1 mM | Supplemented with irrigation | Upregulated photoprotection (photosystem II efficiency) | [16] |
| Zea mays | Maize | 100 µM | Foliar application | Increased stomatal conductance, photosynthesis, transpiration rates, cell turgor, and water holding capacity; increased enzymatic and non-enzymatic antioxidants, regulated osmotic potential, and ROS scavenging | [17] |
| Triticum aestivum | Wheat | 500 µM | Soil application | Regulated photosynthesis, cell turgor; increased water holding capacity and ROS scavenging; reduced membrane damage | [18] |
| Triticum aestivum | Wheat | 10 and 100 µM (dependent on variety) | Seed treatment | Increased germination percentage, radicle and plumule length, and lysine (germination-related amino acid) | [19] |
| Fagopyrum tataricum | Tartary Buckwheat | 100 µM | Foliar application | Increased osmoprotectants, water status, secondary metabolites, antioxidant enzymes, photosynthetic rate, and ROS scavenging | [20] |
| Hordeum vulgare | Barley | 1 mM | Foliar or soil application | Increased endogenous melatonin, antioxidants, ABA, water status, rate of photosynthesis, and photosystem II efficiency | [21] |
| Glycine max | Soybean | 50 µM | Seed coating | Increased seedling biomass and seedling growth; reduced electrolyte leakage | [22] |
| Glycine max | Soybean | 100 µM | Foliar and root application | Increased plant growth, flowering, seed yield, gaseous exchange, photosystem II efficiency and antioxidant enzymes | [23] |
| Minhot esculenta | Cassava | 100 µM | Soil application | Increased peroxidase activity and ROS scavenging | [24] |
| Gossypium hirsutum | Cotton | 100 µM | Seed pre-soaking | Increased number and opening of stomata, antioxidant enzyme activities, osmoprotection, and ROS scavenging | [25] |
| Medicago sativa | Alfalfa | 10 µM | Soil application | Increased chlorophyll content, stomatal conductance, and osmoprotection; upregulated nitro-oxidative homeostasis; reduced cellular redox disruption; scavenged ROS | [26] |
| Malus domestica | Apple | 100 µM | Soil application | Increased water holding capacity, rate of photosynthesis, stomatal opening regulation, and antioxidants; decreased electrolyte leakage, ROS, oxidative damage, and leaf senescence | [27] |
| Vitis vinifer | Grape | 100 µM | Root pretreatment supplemented with irrigation | Increased photoprotection, leaf thickness, stomata size, and enzymatic and non-enzymatic antioxidants; reduced oxidative damage | [4] |
| Actinidia chinensis | Kiwifruit | 100 µM | Supplemented with irrigation | Increased osmoprotectants, protein biosynthesis and photosynthesis; reduced cell membrane damage | [12] |
| Carya cathayensis | Chinese hickory | 100 µM | Foliar application pretreatment | Increased photosynthesis, antioxidants, and osmoprotectants; scavenged ROS | [13] |
| Solanum lycopersicum | Tomato | 0.1 mM | Supplemented with irrigation | Increased photosynthesis, photosystem II efficiency, and antioxidants; reduced toxic substances | [28] |
| Solanum lycopersicum | Tomato | 200 µM | Foliar application | Increased chlorophyll and antioxidant enzymes | [29] |
| Capsicum annuum | Pepper | 50 µM | Seed pretreatment | Increased water holding capacity, endogenous melatonin, carotenoids, and chlorophyll | [30] |
| Citrullus lanatus | Watermelon | 150 µM | Root pretreatment | Increased wax accumulation; reduced abscisic acid | [19] |
| Cucumis sativus | Cucumber | 100 µM | Seed priming and nutrient solution | Increased seed germination, root growth, chlorophyll, photosynthesis, antioxidant enzymes, and ROS scavenging | [4] |
| Cucumis sativus | Cucumber | 10 µM | Foliar application | Scavenged ROS; improved drought tolerance | [31] |
| Brassica napus | Rapeseed | 500 µM | Seed priming | Increased chlorophyll, stomatal regulation, cell wall expansion, antioxidant enzymes, and osmoprotectants; reduced oxidative injury | [32] |
| Dendranthema morifolium | Jinyu Chuju | 100 µM | Foliar application | Increased photosynthesis, chlorophyll, and osmoprotectants; reduced cell membrane damage and relative conductivity | [33] |
| Dracocephalum moldavica | Moldavian balm (Dragon head) | 100 µM | Foliar application | Increased plant growth and flowering, antioxidant activity, chlorophyll, water holding capacity, and ROS scavenging | [34] |
| Agrostis stolonifera | Creeping bentgrass | 20 µM | Foliar application | Increased photosynthetic content, water holding capacity, and photosystem II efficiency; reduced leaf senescence; scavenged ROS | [35] |
| Festuca arundinacea | Tall fescue | 20 µM | Irrigation pretreatment | Increased antioxidant enzyme activity, chlorophyll, and plant growth; scavenged ROS | [36] |
| Cynodon dactylon | Bermuda grass | 20 and 100 µM | Irrigation pretreatment | Increased plant growth, chlorophyll, antioxidant activity, stress-responsive genes, and hormonal regulation; scavenged ROS | [14] |
| Trigonella foenum-graecum | Fenugreek | 100 and 300 µM | Foliar application pretreatment | Increased endogenous melatonin and secondary metabolites, chlorophyll, and antioxidant enzymes; scavenged ROS | [37] |
| Coffea arabica | Coffee | 300 µM | Soil application | Increased photoprotection, gaseous exchange, carboxylation activity, chlorophyll, and antioxidant enzyme activities | [38] |
| Camellia sinensis | Tea | 100 µM | Foliar application pretreatment | Increased photosynthesis, antioxidant enzymes, and GSH and AsA contents; scavenged ROS | [39] |
| Nicotiana benthamiana | Tobacco | 10 µM | Foliar application | Improved drought tolerance; scavenged ROS; reduced oxidative damage | [31] |
| Salinity Stress | |||||
|---|---|---|---|---|---|
| Crop | Common Name | Melatonin Dose | Treatment Method | Response to Treatment | Reference(s) |
| Momordica charantia | Bitter melon | 150 µM | Seed priming | Increased relative water content, antioxidant enzyme activities, and gene expression levels; decreased hydrogen peroxide and malondialdehyde levels | [42] |
| Zea mays | Maize | 0.4, 0.8, and 1.6 mM | Pretreatment of seeds | Improved shoot and root lengths, germination energy, fresh and dry weights of seedling, K+ content, antioxidant enzyme activities, and relative water content. | [43] |
| Gossypium hirsutum | Cotton | 25 µM | Seed priming | Enhanced ability to scavenge ROS and improved photosynthetic efficiency | [44] |
| Triticum aestivum | Wheat | 70 µM | Seed priming | Enhanced photosynthetic pigments; indole-3-acetic acid content and growth parameters | [45] |
| Ocimum basilicum | Basil | 10 µM | Seed priming | Increased contents of flavonoid and phenolic acid | [46] |
| Vicia faba | Faba bean | 100 and 500 mM | Seed priming | Improved novel protein expressions | [47] |
| Cucumis sativum | Cucumber | 1 µM | Seed priming | Enhanced seed germination | [43] |
| Arabidopsis thaliana | Arabidopsis | 10 µM | Foliar application | Induced antioxidant defense system; scavenged ROS; upregulated abscisic acid-responsive genes | [48] |
| Brassica napus | Rapeseed | 1 µM | Foliar application | Reduced lipid peroxidation and hydrogen peroxide content; maintained redox and ion homeostasis | [49] |
| Brassica juncea | Mustard greens | 1 µM | Foliar application | Increased leaf length and width, plant height, and stem diameter; improved gaseous exchange, relative water content; increased salicylic acid and reduced abscisic acid | [50] |
| Cucumis melo | Melon | 0, 10, and 50 µM | Seed pretreatment | Increased seed germination | [51] |
| Oryza sativa | Rice | 0, 10, and 20 µM | Root irrigation | Upregulated antioxidants and leaf senescence; inhibited cell death and chlorophyll degradation | [52] |
| Glycine max | Soybean | 0–100 µM | Foliar application | Increased photosynthesis, cell division, carbohydrates, fatty acids, and ascorbate contents; reduced inhibitory effect on gene expressions | [22] |
| Malus hupehensis | Pingyitiancha | 0.1 µM | Seed pretreatment | Increased photosynthesis and ion homeostasis; reduced oxidative damage | [53] |
| Solanum lycopersicum | Tomato | 100 µM | Root irrigation | Increased protein and membrane protection, antioxidant activities, and photosynthesis | [54] |
| Citrullus lanatus | Watermelon | 50–150 µM | Seed pretreatment | Increased antioxidant enzymes, photosynthesis, and photosystem II efficiency; reduced stomatal closure and oxidative damage | [55] |
| Heat Stress | |||||
|---|---|---|---|---|---|
| Crop | Common Name | Melatonin Dose | Treatment Method | Response to Treatment | Reference(s) |
| Triticum aestivum | Wheat | 20 µM | Soil application | Increased rate of photosynthesis; reduced oxidative damage | [56] |
| Lolium perenne | Perennial Ryegrass | 100 µM | Foliar application | Regulated cytokinin biosynthesis genes; downregulated abscisic acid biosynthesis genes; enhanced endogenous melatonin level | [57] |
| Solanum lycopersicum | Tomato | 100 µM | Seed pretreatment | Enhanced phenolic acid level; regulated transcript abundances; increased endogenous melatonin levels; reduced oxidative stress | [58] |
| Arabidopsis thaliana | Arabidopsis | 20 µM | Foliar application | Upregulated heat shock factors | [59] |
| Zea mays | Maize | 100 µM | Soil application | Increased photosynthesis; reduced oxidative damage | [17] |
| Cold Stress | |||||
|---|---|---|---|---|---|
| Crop | Common Name | Melatonin Dose | Treatment Method | Response to Treatment | Reference(s) |
| Triticum aestivum | Wheat | 100 µM | Spray | Improved antioxidant enzyme activities; reduced oxidative stress | [61] |
| Citrullus lanatus | Watermelon | 150 µM | Soil treatment | Increased accumulation of hydrogen peroxide; increased tolerance | [62] |
| Solanum lycopersicum | Tomato | 100 µM | Seedling spray | Improved photosynthesis; reduced oxidative damage | [63] |
| Cynodon dactylon | Bermuda grass | 100 µM | Foliar application | Increased arabinose, mannose, and propanoic acid levels | [64] |
| Hordeum vulgare | Barley | 1 mM | Soil irrigation | Improved water status, antioxidant system, and abscisic acid level | [21] |
| Camellia sinensis | Tea plant | 100 µM | Spray | Improved production of antioxidant enzymes; reduced oxidative stress | [65] |
| Oryza sativa | Rice | N/A | Spray | Increased antioxidant enzyme activities; reduced oxidative stress | [55] |
| Cucumis sativus | Cucumber | 100 µM | Foliar spray | Improved antioxidant enzyme productions and activities; reduced oxidative stress | [66] |
| Heavy Metal Stress | |||||
|---|---|---|---|---|---|
| Crop | Common Name | Melatonin Dose | Treatment Method | Response to Treatment | Reference(s) |
| Triticum aestivum | Wheat | 50 µM | Soil treatment | Increased antioxidant enzymes against cadmium metal stress | [68] |
| Medicago sativa | Alfalfa | 50 µM | Foliar application | Increased ABC transporters; decreased cadmium accumulation | [69] |
| Solanum lycopersicum | Tomato | 100 µM | Seed priming | Increased antioxidants and plant growth; reduced electrolyte leakage and photoinhibition under cadmium metal stress | [21] |
| Nicotiana benthamiana | Tobacco | 15 µM | Foliar application | Increased cell growth and viability; decreased DNA damage against lead heavy metal | [53] |
| Cyphomandra betacea | Tree tomato | 50 µM | Soil treatment | Increased antioxidants and plant biomass under cadmium stress | [70] |
| Glycine max | Soybean | 100 mM | Seed priming | Increased photosynthesis and antioxidant enzymes under aluminum stress | [53] |
| Brassica oleracea | Red cabbage | 10 µM | Foliar application | Increased germination and fresh weight against copper metal stress | [53] |
| Citrullus lanatus | Watermelon | 50 mg/L | Seed priming | Increased plant growth, photosynthesis, chlorophyll, antioxidant enzymes, and scavenging of ROS against vanadium metal stress | [71] |
| Zea mays | Maize | 500 µM | Soil treatment | Induced additional proteins related to stress reduction during germination | [72] |
| Cucumis sativus | Cucumber | 100 and 150 µM | Soil irrigation | Reduced stress-promoted expression of genes e.g., CsHA2 under cadmium metal stress | [43] |
| Amaranthus viridis | Amaranthus | 400 µM | Foliar application | Decreased accumulation of metals (e.g., lead) in roots | [73] |
| Drought Stress | |||||
|---|---|---|---|---|---|
| Crop | Common Name | Glutathione Dose | Treatment Method | Response to Treatment | Reference(s) |
| Cicer arietinum | Chickpea | 0.75 mM | Seed soaking | Increased growth parameters, chlorophyll, photosynthesis, endogenous proline, and antioxidant enzyme activities | [74] |
| Oryza sativa | Rice | 0.2 mM | Spraying | Increased root and shoot lengths, dry and fresh weights, chlorophyll pigment, relative water content, and antioxidant enzyme activities | [5] |
| Brassica napus | Rapeseed | N/A | Foliar application | Scavenged ROS; reduced oxidative damage | [75] |
| Triticum aestivum | Wheat | N/A | Sprayed | Improved tolerance compared with non-treated cultivar | [76] |
| Vigna radiata | Mung bean | N/A | Exogenous application | Lessened drought-induced oxidative damage through the enhancement of the capacity of the antioxidant system and glyoxalase activity | [77] |
| Arabidopsis thaliana | Arabidopsis | N/A | Spraying | Increased abscisic acid level and tolerance against drought stress; improved plant health under stressful conditions | [78] |
| Salinity Stress | |||||
|---|---|---|---|---|---|
| Crop | Common Name | Glutathione Dose | Treatment Method | Response to Treatment | Reference(s) |
| Capsicum frutescence | Pepper | 0.4 and 0.8 mM | Foliar spray | Increased water use efficiency, growth, fresh and dry weights of roots and shoots, yield, osmoprotectants, and antioxidants | [79] |
| Cucumis sativus | Cucumber | 0.5 mM | Seed soaking | Increased growth, fresh and dry weights, relative water content, photosynthetic activity, and stomatal conductance | [80] |
| Vicia faba | Faba bean | 0.5 mM | Foliar spray | Increased growth, fresh and dry weights, relative water content, photosynthetic activity, stomatal conductance, and antioxidant enzyme activities | [81] |
| Triticum aestivum | Wheat | 1 mM | Foliar spray | Increased plant growth, membrane stability, and accumulation of osmoprotectants | [59] |
| Glycine max | Soybean | 1 mM | Foliar spray | Increased growth, photosynthesis, membrane stability, soluble sugars, and antioxidant enzyme activities | [5] |
| Phaseolus vulgaris | Common bean | 0.75 mM | Foliar spray | Increased plant length, number and surface area of leaves, fresh and dry weights of plant, relative water content, photosynthesis, and soluble sugars | [5] |
| Arabidopsis thaliana | Arabidopsis | N/A | Foliar application | Increased abscisic acid and tolerance against drought stress; improved plant health under stressful conditions | [78] |
| Solanum lycopersicum | Tomato | N/A | Exogenous application | Improved tolerance against salinity and oxidative stresses; decreased lipid peroxidation | [82] |
| Oryza sativa | Rice | N/A | sprayed | Improved activities of antioxidant enzymes; decreased ROS accumulation and ROS-induced DNA damage | [83] |
| Heat Stress | |||||
|---|---|---|---|---|---|
| Crop | Common Name | Glutathione Dose | Treatment Method | Response to Treatment | Reference(s) |
| Triticum aestivum | Wheat | N/A | External application | Increased antioxidant enzyme activities and resistance to heat stress | [85,87] |
| Vigna radiata | Mung bean | N/A | Seed pretreatment | Increased antioxidant enzyme activities; enhanced stress resistance; decreased ROS level | [77] |
| Cucumis sativus | Cucumber | N/A | External application | Enhanced heat resistance, plant growth, chlorophyll content, and photosynthetic rate | [84] |
| Brassica campestris | Mustard | N/A | External application | Maintained relative water content; increased ROS scavenging and antioxidants | [86] |
| Cold Stress | |||||
|---|---|---|---|---|---|
| Crop | Common Name | Glutathione Dose | Treatment Method | Response to Treatment | Reference(s) |
| Oryza sativa | Rice | 0.5 Mm | Spraying | Increased lengths of root and shoot, fresh and dry weights, and endogenous glutathione level | [88] |
| Capsicum annum | Pepper | 0.5 Mm | Spraying | Increased lengths of root and shoot, fresh and dry weights, and endogenous glutathione level | [60] |
| Cucumis sativus | Cucumber | N/A | Foliar application | Decreased electrolyte leakage and lipid peroxidation | [89] |
| Jatropha curcas | Purging nut | N/A | External application | Enhanced resistance and antioxidant enzyme activities | [90] |
| Heavy Metal Stress | |||||
|---|---|---|---|---|---|
| Crop | Common Name | Glutathione Dose | Treatment Method | Response to Treatment | Reference(s) |
| Triticum aestivum | Wheat | 20 µM | Foliar spray | Increased photosynthetic pigments and endogenous glutathione level against cadmium metal | [62] |
| Solanum melongena | Brinjal(Aubergine) | 1 mM | Seed pretreatment | Mitigated adverse effects of stress and protein damage against arsenate metal stress | [91] |
| Zea mays | Maize | 30 µM | Foliar application | Increased secondary metabolites and flavonoids; alleviated oxidative damage under cadmium metal stress | [92] |
| Lolium multiflorum | Italian ryegrass | 200 µM | External application | Increased stress tolerance and biomass of roots and shoots against lead stress | [93] |
| Hordeum vulgare | Barley | N/A | External application | Improved antioxidant defense system and photosynthesis; decreased ROS accumulation against cadmium metal stress | [94,98] |
| Solanum lycopersicum | Tomato | N/A | External application | Synchronized transcript levels of several stress-responsive transcription factors; improved nitric oxide contents against cadmium metal stress | [95] |
| Oryza sativa | Rice | N/A | External application | Elevated endogenous glutathione level, mineral elements and pigment contents; upregulated phytochelatins; synchronized antioxidant enzyme activities under cadmium metal stress | [96,99] |
| Brassica campestris | Mustard | N/A | Exogenous application | Reduced cadmium levels in roots and leaves and the accumulation of ROS; protected against stress | [97] |
| Drought Stress | |||||
|---|---|---|---|---|---|
| Crop | Common Name | Proline Dose | Treatment Method | Response to Treatment | Reference(s) |
| Zea mays | Maize | 1 mM | Seed priming | Increased photosynthetic and transpiration rates and stomatal conductance | [6] |
| Zea mays | Maize | N/A | Foliar application | Promoted uptake and accumulation of nitrogen, phosphorus, and potassium, as well as tolerance against drought stress | [6] |
| Triticum aestivum | Wheat | 150 ppm | Foliar application | Reduced malondialdehyde level and lipid peroxidation | [101] |
| Chenopodium quinoa | Quinoa | N/A | Foliar application | Improved photosynthetic pigments, phenols, free amino acids, plant height, and dry and fresh weights of roots and shoots | [102] |
| Arabidopsis thaliana | Arabidopsis | N/A | Spraying | Scavenged ROS; protected the integrity of plasma lemma | [100] |
| Pisum sativum | Pea | 4 mM | Foliar spray | Increased yield, the non-enzymatic antioxidant defense system, and soluble protein concentration | [103] |
| Salinity Stress | |||||
|---|---|---|---|---|---|
| Crop | Common Name | Proline Dose | Treatment Method | Response to Treatment | Reference(s) |
| Cucumis melo | Muskmelon | 10 mM | Foliar spray | Increased growth, chlorophyll and proline contents, and relative water content | [104] |
| Cucumis sativus | Cucumber | 10 mM | Nutrient solution | Increased growth, proline content, and antioxidant enzyme activities | [105] |
| Glycine max | Soybean | 25 mM | External application | Increased growth, proline content, antioxidant enzyme activities, and nitrogen fixation | [106] |
| Helianthus annus | Sunflower | 30 and 60 mM | Foliar spray | Increased growth, proline and amino acids contents, and antioxidant enzyme activities | [107] |
| Zea mays | Maize | 30 mM | Foliar spray | Increased growth and proline content | [108] |
| Triticum durum | Durum wheat | 12 mM | Seed pretreatment | Increased growth, photosynthetic activity, proline content, and antioxidant enzyme activities | [109] |
| Sorghum bicolor | Great millet | 30 mM | Foliar spray | Increased growth, relative water content, gaseous exchange, and amino acids and proline contents | [110] |
| Oryza sativa | Rice | 1, 5, and 10 mM | Seed pretreatment | Increased growth, seed germination, and chlorophyll and proline content | [111] |
| Brassica juncea | Mustard greens | 20 mM | Foliar application | Improved yield and stress tolerance | [112] |
| Capsicum annum | Red pepper | 0.8 mM | Foliar application | Improved antioxidant enzyme activities, photosynthetic and transpiration rates, plant dry and fresh weights, and root and shoot lengths | [113] |
| Daucus carota | Wild carrot | N/A | Foliar application | Improved antioxidant enzyme activities, and potassium and calcium contents in roots and shoots | [114] |
| Phaseolus vulgaris | Bean | N/A | Foliar application | Improved antioxidant enzyme activities and endogenous proline level | [115] |
| Raphanus sativus | Radish | N/A | Foliar application | Improved transpiration rate, stomatal conductance, pigment contents, and levels of proteins and some nutrients | [116] |
| Vicia faba | Faba bean | N/A | Foliar application | Improved levels of photosynthetic pigments, soluble carbohydrates, endogenous proline, and free amino acids | [6] |
| Lactuca sativa | Lettuce | 5 µM | Foliar application | Improved plant growth, photosynthetic rate, chlorophyll content and yield | [117] |
| Heat Stress | |||||
|---|---|---|---|---|---|
| Crop | Common Name | Proline Dose | Treatment Method | Response to Treatment | Reference(s) |
| Abelmoschus esculentus | Okra | N/A | Foliar application | Improved shoot length, number of leaves per plant, and free amino acids content | [118] |
| Lactuca sativa | Lettuce | 5µM | Foliar application | Improved plant growth, photosynthetic rate, chlorophyll content and yield | [117] |
| Vigna radiata | Mung bean | N/A | Foliar application | Improved carbon dioxide (CO2) assimilation capacity and heat tolerance | [119] |
| Cold Stress | |||||
|---|---|---|---|---|---|
| Crop | Common Name | Proline Dose | Treatment Method | Response to Treatment | Reference(s) |
| Citrus reticulata | Mandarin orange | N/A | Foliar application | Increased contents of phenolic acids, flavonoids and endogenous proline; increased antioxidant enzyme activities | [120] |
| Citrus sinensis | Sweet orange | N/A | Foliar application | Increased contents of phenolic acids, flavonoids, and endogenous proline; increased antioxidant enzyme activity | [120] |
| Citrus paradisi | Grapefruit | N/A | Foliar application | Increased contents of phenolic acids, flavonoids and endogenous proline; increased antioxidant enzyme activity | [120] |
| Capsicum annum | Red pepper | 24 mM | Foliar application | Increased endogenous proline level and antioxidant enzyme activities | [6] |
| Heavy Metal Stress | |||||
|---|---|---|---|---|---|
| Crop | Common Name | Proline Dose | Treatment Method | Response to Treatment | Reference(s) |
| Cicer arietinum | Chickpea | N/A | Foliar application | Improved nitrogen fixation, nitrogen content in leaves, and antioxidant enzyme activities against cadmium stress | [121] |
| Olea europaea | Olive | 20 mM | Irrigation | Enhanced proline and oil contents, antioxidant enzyme activities; reduced hydrogen peroxide against cadmium stress | [122] |
| Phaseolus vulgaris | Bean | N/A | Culture medium | Improved relative water content, chlorophyll and endogenous proline contents, and antioxidant enzyme activities under selenium heavy metal stress | [6,123] |
| Pisum sativum | Pea | N/A | Foliar application | Enhanced growth, photosynthetic activity, relative water content and organic osmolyte contents | [6] |
| Poncirus trifoliata | Trifoliate orange | N/A | Nutrient solution | Enhanced protein and cellulose contents against aluminum stress | [124] |
| Solanum melongena | Aubergine | N/A | Seedling treatment | Increased endogenous proline level and antioxidant enzyme activities under arsenate stress | [6,125] |
| Triticum aestivum | Wheat | 80 mM | Foliar spray | Reduced ROS; increased plant height, weight, and photosynthetic capacity | [103] |
| Zea mays | Maize | N/A | Exogenous application | Improved defensive mechanism and sugar biosynthesis against cadmium stress | [126] |
| Drought Stress | |||||
|---|---|---|---|---|---|
| Crop | Common Name | Glycine Betaine Dose | Treatment Method | Response to Treatment | Reference(s) |
| Triticum aestivum | Wheat | N/A | Exogenous application | Improved stress tolerance index; enhanced osmolyte and relative water contents | [129] |
| Triticum aestivum | Wheat | N/A | Exogenous application | Increased spike length, number of spikelets per spike, number of grains, yield, and leaf turgor potential | [130] |
| Zea mays | Maize | 100 mM | Foliar application | Enhanced growth, yield, and antioxidant enzyme activities | [131] |
| Solanum lycopersicum | Tomato | N/A | Exogenous application | Improved yield | [132] |
| Pisum sativum | Pea | N/A | Exogenous application | Enhanced growth, number of pods and leaves per plant; increased level of soluble sugars and soluble protein in leaves; increased antioxidant enzyme activities | [133] |
| Nicotiana tabacum | Tobacco | 80 mM | Foliar application | Improved plant growth, osmotic adjustment, photosynthesis, and antioxidant enzyme activities | [128] |
| Glycine max | Soybean | 3 kg/ha | Exogenous application | Increased seed number | [134] |
| Salinity Stress | |||||
|---|---|---|---|---|---|
| Crop | Common Name | Glycine Betaine Dose | Treatment Method | Response to Treatment | Reference(s) |
| Vigna unguiculata | Cowpeas | 5–10 mM | Foliar application | Increased soluble sugar contents and antioxidant enzymes | [135] |
| Phaseolus vulgaris | Common bean | N/A | Exogenous application | Increased plant fresh weight, leaf area ratio, relative water content, and soluble sugar and free amino acid contents | [136] |
| Oryza sativa | Rice | N/A | Foliar application | Increased plant height, fresh and dry weights, and chlorophyll content; reduced malondialdehyde content | [128] |
| Solanum lycopersicum | Tomato | N/A | Exogenous application | Increased photosynthesis and stomatal conductance; decreased photorespiration | [128] |
| Glycine max | Soybean | N/A | Exogenous application | Reduced ROS and lipid peroxidation; increased antioxidant enzyme activities | [137] |
| Lolium perenne | Perennial ryegrass | 0, 20, 50 mM | Exogenous application | Increased fresh weight and relative water content; reduced electrolyte leakage and malondialdehyde content | [138] |
| Triticum aestivum | Wheat | N/A | Exogenous application | Increased rate of photosynthesis | [139] |
| Heat Stress | |||||
|---|---|---|---|---|---|
| Crop | Common Name | Glycine Betaine Dose | Treatment Method | Response to Treatment | Reference(s) |
| Solanum lycopersicum | Tomato | N/A | External application | Increased fruit yield and rate of photosynthesis | [128] |
| Solanum lycopersicum | Tomato | 1, 5 mM | External application | Improved seed germination, expression of heat shock genes, and accumulation of heat shock proteins | [140] |
| Hordeum vulgare | Barley | N/A | External application | Increased tolerance of photosystem Ⅱ; protective effect on oxygen-evolving complex | [141] |
| Hordeum vulgare | Barley | 10 mM | External application | protective effect on oxygen-evolving complex; greater photosystem Ⅱ stability | [141] |
| Hordeum vulgare | Barley | 10, 20, 30, 40 and 50 mM | External application | Improved growth, photosynthesis, and water relations; decreased ion leakage | [142] |
| Triticum aestivum | Wheat | 100 mM | External application | Maintained higher chlorophyll content, photosystem Ⅱ photochemical activity, net photosynthetic rate, and accumulation of endogenous glycine betaine | [85] |
| Triticum aestivum | Wheat | 50 and 100 mM | External application | Improved yield and relative membrane permeability | [7] |
| Saccharum officinarum | Sugarcane | 20 mM | External application | Improved bud sprouting, soluble sugar accumulation, and endogenous level of osmolytes; decreased hydrogen peroxide | [143] |
| Tagetes erecta | Marigold | 0.5 and 1 mM | External application | Improved gaseous exchange; reduced ROS accumulation | [144] |
| Cold Stress | |||||
|---|---|---|---|---|---|
| Crop | Common Name | Glycine Betaine Dose | Treatment Method | Response to Treatment | Reference(s) |
| Zea mays | Maize | 100 mM | Foliar application | Prevented chlorosis; reduced lipid peroxidation of membrane | [145] |
| Triticum aestivum | Wheat | 100 mM | Foliar spray | Increased osmolality and photosynthesis | [146] |
| Prunus persica | Peach | 10 mM | External application | Lowered malondialdehyde content; increased endogenous glycine betaine level | [147] |
| Medicago sativa | Alfalfa | 0.2 M | Seedling sprayed | Decreased ion leakage from shoot tissues | [148] |
| Solanum lycopersicum | Tomato | 0.1 mM | Foliar spray | Increased catalase activity; reduced hydrogen peroxide | [149] |
| Hordeum vulgare | Barley | N/A | External application | Increased osmolality and endogenous level of glycine betaine | [146] |
| Heavy Metal Stress | |||||
|---|---|---|---|---|---|
| Crop | Common Name | Glycine Betaine Dose | Treatment Method | Response to Treatment | Reference(s) |
| Triticum aestivum | Wheat | 0–100 mM | Spraying on leaves | Improved growth, chlorophyll contents, and biomass and protein contents against chromium stress | [150] |
| Gossypium hirsutum | Cotton | 1 mM | Exogenous application | Improved plant growth, antioxidant enzyme activities and photosynthetic rate and gaseous exchange; alleviated cadmium stress | [151] |
| Gossypium hirsutum | Cotton | N/A | Foliar application | Improved plant growth and gas attributes; alleviated lead stress | [152] |
| Vigna radiata | Mung bean | 0, 50, 100 mM | Foliar application | Improved plant growth; alleviated chromium stress | [153] |
| Amaranthus tricolor | Amaranth | N/A | Exogenous application | Improved photosynthesis and chlorophyll content of leaves; alleviated cadmium stress | [154] |
| Lolium perenne | Perennial ryegrass | N/A | Exogenous application | Improved membrane stability; reduced lipid peroxidation; alleviated cadmium stress | [155] |
| Nicotiana tabacum | Tobacco | N/A | Exogenous application | Reduced stomatal closure, accumulation of malondialdehyde, and leaf damage; alleviated cadmium stress | [8] |
| Cucumis sativus | Cucumber | N/A | Foliar application | Significant protective effect on chlorophyll content; alleviated aluminum stress | [156] |
| Sorghum bicolor | Millet | 50–100 mM | Exogenous application | Improved quality and yield; alleviated chromium stress | [157] |
| Oryza sativa | Asian rice | N/A | Exogenous application | Increased GST and GRX gene expressions; alleviated arsenic stress | [158] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khalid, M.; Rehman, H.M.; Ahmed, N.; Nawaz, S.; Saleem, F.; Ahmad, S.; Uzair, M.; Rana, I.A.; Atif, R.M.; Zaman, Q.U.; et al. Using Exogenous Melatonin, Glutathione, Proline, and Glycine Betaine Treatments to Combat Abiotic Stresses in Crops. Int. J. Mol. Sci. 2022, 23, 12913. https://doi.org/10.3390/ijms232112913
Khalid M, Rehman HM, Ahmed N, Nawaz S, Saleem F, Ahmad S, Uzair M, Rana IA, Atif RM, Zaman QU, et al. Using Exogenous Melatonin, Glutathione, Proline, and Glycine Betaine Treatments to Combat Abiotic Stresses in Crops. International Journal of Molecular Sciences. 2022; 23(21):12913. https://doi.org/10.3390/ijms232112913
Chicago/Turabian StyleKhalid, Memoona, Hafiz Mamoon Rehman, Nisar Ahmed, Sehar Nawaz, Fozia Saleem, Shakeel Ahmad, Muhammad Uzair, Iqrar Ahmad Rana, Rana Muhammad Atif, Qamar U. Zaman, and et al. 2022. "Using Exogenous Melatonin, Glutathione, Proline, and Glycine Betaine Treatments to Combat Abiotic Stresses in Crops" International Journal of Molecular Sciences 23, no. 21: 12913. https://doi.org/10.3390/ijms232112913
APA StyleKhalid, M., Rehman, H. M., Ahmed, N., Nawaz, S., Saleem, F., Ahmad, S., Uzair, M., Rana, I. A., Atif, R. M., Zaman, Q. U., & Lam, H.-M. (2022). Using Exogenous Melatonin, Glutathione, Proline, and Glycine Betaine Treatments to Combat Abiotic Stresses in Crops. International Journal of Molecular Sciences, 23(21), 12913. https://doi.org/10.3390/ijms232112913











