Relative Abundance of Lipid Metabolites in Spermatozoa across Three Compartments
Abstract
1. Introduction
2. Results
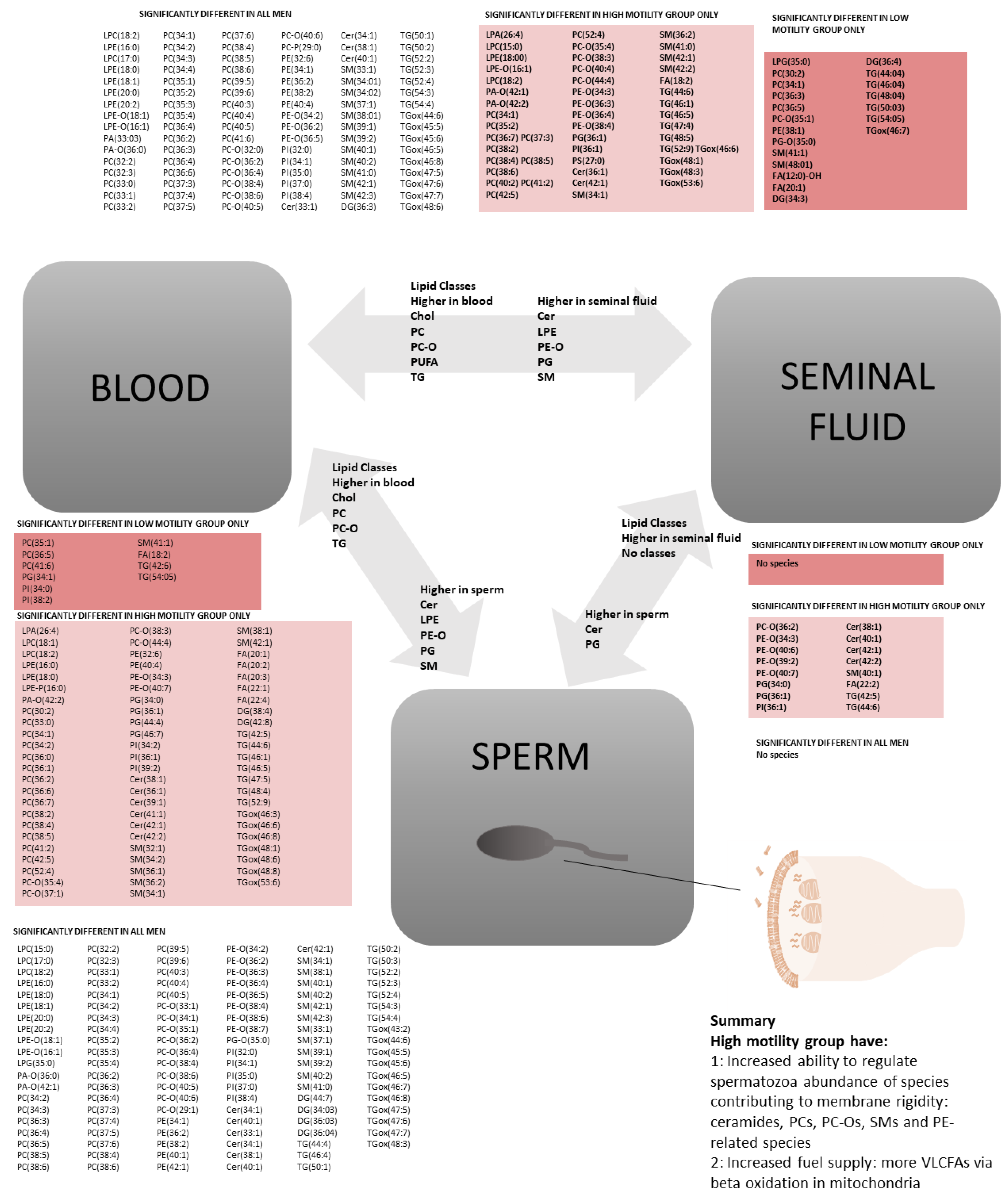
2.1. All Participants: Lipid Class Analysis between Three Compartments
2.2. All Participants: Lipid Species Analysis between Three Compartments
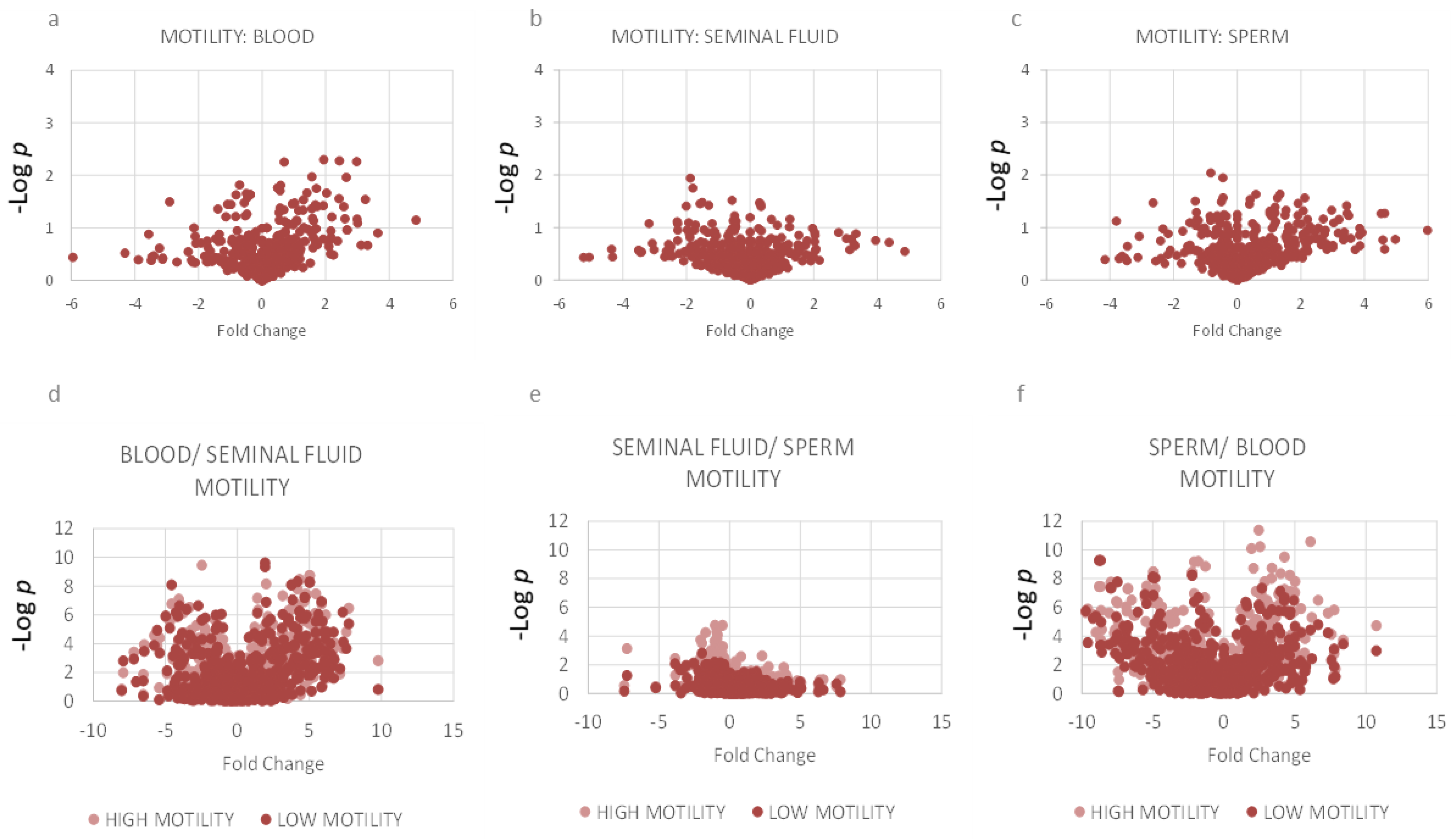
2.3. Motility: Lipidomics Analysis between Three Compartments
3. Discussion
3.1. Strengths and Weaknesses
3.2. Relevance of Lipidomic Findings to Human Health
3.3. Effects of Lipid Composition on Sperm Cell Membrane
3.4. Effects of Lipid Composition on Cellular Adhesion
3.5. Effects of Lipid Composition on Cell Signalling
3.6. Effect of the Abundance of Lipid Species on Cell Fuel Supply and Reserves
3.7. Associations between Reduced Motility and Lipid Composition
3.8. Areas for Future Research
4. Materials and Methods
4.1. Participants
4.2. Semen Collection and Analysis
4.3. Blood Sample Collection and Analysis
4.4. Specimen Processing and Mass Spectrometry
4.5. Data Processing
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Harris, I.D.; Fronczak, C.; Roth, L.; Meacham, R.B. Fertility and the aging male. Rev. Urol. 2011, 13, e184–e190. [Google Scholar] [PubMed]
- Elbashir, S.; Magdi, Y.; Rashed, A.; Ibrahim, M.A.; Edris, Y.; Abdelaziz, A.M. Relationship between sperm progressive motility and DNA integrity in fertile and infertile men. Middle East Fertil. Soc. J. 2018, 23, 195–198. [Google Scholar] [CrossRef]
- Kumar, N.; Singh, A. Trends of male factor infertility, an important cause of infertility: A review of literature. J. Hum. Reprod. Sci. 2015, 8, 191–196. [Google Scholar] [CrossRef] [PubMed]
- La Vignera, S.; Condorelli, R.; Vicari, E.; D’Agata, R.; Calogero, A.E. Diabetes Mellitus and Sperm Parameters. J. Androl. 2012, 33, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Condorelli, R.A.; La Vignera, S.; Mongioì, L.M.; Alamo, A.; Calogero, A.E. Diabetes Mellitus and Infertility: Different Pathophysiological Effects in Type 1 and Type 2 on Sperm Function. Front. Endocrinol. 2018, 9, 268. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.-F.; Ding, G.-L.; Liu, Y.; Liu, M.-E.; Pan, J.-X.; Guo, M.-X.; Sheng, J.-Z. The effects of diabetes on male fertility and epigenetic regulation during spermatogenesis. Asian J. Androl. 2015, 17, 948–953. [Google Scholar] [CrossRef]
- Lu, J.-C.; Jing, J.; Yao, Q.; Fan, K.; Wang, G.-H.; Feng, R.-X.; Liang, Y.-J.; Chen, L.; Ge, Y.-F.; Yao, B. Relationship between Lipids Levels of Serum and Seminal Plasma and Semen Parameters in 631 Chinese Subfertile Men. PLoS ONE 2016, 11, e0146304. [Google Scholar] [CrossRef]
- Schisterman, E.F.; Mumford, S.L.; Chen, Z.; Browne, R.W.; Boyd Barr, D.; Kim, S.; Louis, G.M.B. Lipid concentrations and semen quality: The LIFE study. Andrology 2014, 2, 408–415. [Google Scholar] [CrossRef]
- Alessandra, F.; Mariangela, D.G.; Natalina, M.; Vincenzo, Z. Obesity and Male Infertility: Role of Fatty Acids in the Modulation of Sperm Energetic Metabolism. Eur. J. Lipid Sci. Technol. 2018, 120, 1700451. [Google Scholar]
- Amaral, A.; Castillo, J.; Ramalho-Santos, J.; Oliva, R. The combined human sperm proteome: Cellular pathways and implications for basic and clinical science. Hum. Reprod. Update 2013, 20, 40–62. [Google Scholar] [CrossRef]
- Amaral, A.; Castillo, J.; Estanyol, J.M.; Ballescà, J.L.; Ramalho-Santos, J.; Oliva, R. Human Sperm Tail Proteome Suggests New Endogenous Metabolic Pathways. Mol. Cell. Proteomics 2013, 12, 330–342. [Google Scholar] [CrossRef] [PubMed]
- Swegen, A.; Curry, B.J.; Gibb, Z.; Lambourne, S.R.; Smith, N.D.; Aitken, R.J. Investigation of the stallion sperm proteome by mass spectrometry. Reproduction 2015, 149, 235–244. [Google Scholar] [CrossRef] [PubMed]
- Fraisl, P.; Tanaka, H.; Forss-Petter, S.; Lassmann, H.; Nishimune, Y.; Berger, J. A novel mammalian bubblegum-related acyl-CoA synthetase restricted to testes and possibly involved in spermatogenesis. Arch. Biochem. Biophys. 2006, 451, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Hashemitabar, M.; Sabbagh, S.; Orazizadeh, M.; Ghadiri, A.; Bahmanzadeh, M. A proteomic analysis on human sperm tail: Comparison between normozoospermia and asthenozoospermia. J. Assist. Reprod. Genet. 2015, 32, 853–863. [Google Scholar] [CrossRef] [PubMed]
- Ferramosca, A.; Moscatelli, N.; Di Giacomo, M.; Zara, V. Dietary fatty acids influence sperm quality and function. Andrology 2017, 5, 423–430. [Google Scholar] [CrossRef]
- Safarinejad, M.R. Effect of omega-3 polyunsaturated fatty acid supplementation on semen profile and enzymatic anti-oxidant capacity of seminal plasma in infertile men with idiopathic oligoasthenoteratospermia: A double-blind, placebo-controlled, randomised study. Andrologia 2011, 43, 38–47. [Google Scholar] [CrossRef]
- Cosson, J. Book Chapter. ATP: The sperm movement energizer. In Adenosine Triphosphate: Chemical Propoerties, Biosynthesis and Functions in Cells; Kuester, E., Traugott, G., Eds.; Nova Science Pub Inc.: New York, NY, USA, 2013; p. 46. [Google Scholar]
- Sugkraroek, P.; Kates, M.; Leader, A.; Tanphaichitr, N. Levels of cholesterol and phospholipids in freshly ejaculated sperm and Percoll-gradient-pelletted sperm from fertile and unexplained infertile men. Fertil. Steril. 1991, 55, 820–827. [Google Scholar] [CrossRef]
- Koynova, R.; Caffrey, M. Phases and phase transitions of the hydrated phosphatidylethanolamines. Chem. Phys. Lipids 1994, 69, 1–34. [Google Scholar] [CrossRef]
- Koynova, R.; Caffrey, M. Phases and phase transitions of the phosphatidylcholines. Biochim. Biophys. Acta 1998, 1376, 91–145. [Google Scholar] [CrossRef]
- Furse, S. The physical influence of inositides-a disproportionate effect? J. Chem. Biol. 2015, 8, 1–3. [Google Scholar] [CrossRef]
- Furse, S.; de Kroon, A.I.P.M. Phosphatidylcholine’s functions beyond that of a membrane brick. Mol. Membr. Biol. 2015, 32, 117–119. [Google Scholar] [CrossRef] [PubMed]
- Furse, S. Is phosphatidylglycerol essential for terrestrial life? J. Chem. Biol. 2017, 10, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Furse, S.; Shearman, G.C. Do lipids shape the eukaryotic cell cycle? Biochim. Biophys. Acta 2018, 1863, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Dawaliby, R.; Trubbia, C.; Delporte, C.; Noyon, C.; Ruysschaert, J.-M.; Van Antwerpen, P.; Govaerts, C. Phosphatidylethanolamine Is a Key Regulator of Membrane Fluidity in Eukaryotic Cells. J. Biol. Chem. 2016, 291, 3658–3667. [Google Scholar] [CrossRef]
- Kawano, N.; Yoshida, K.; Miyado, K.; Yoshida, M. Lipid Rafts: Keys to Sperm Maturation, Fertilization, and Early Embryogenesis. J. Lipids 2011, 2011, 10. [Google Scholar] [CrossRef]
- Frenette, G.; Girouard, J.; Sullivan, R. Comparison Between Epididymosomes Collected in the Intraluminal Compartment of the Bovine Caput and Cauda Epididymidis1. Biol. Reprod. 2006, 75, 885–890. [Google Scholar] [CrossRef] [PubMed]
- Sanders, F.W.B.; Acharjee, A.; Walker, C.; Marney, L.; Roberts, L.D.; Imamura, F.; Jenkins, B.; Case, J.; Ray, S.; Virtue, S.; et al. Hepatic steatosis risk is partly driven by increased de novo lipogenesis following carbohydrate consumption. Genome Biol. 2018, 19, 79. [Google Scholar] [CrossRef]
- Chen, S.; Wang, M.; Li, L.; Wang, J.; Ma, X.; Zhang, H.; Cai, Y.; Kang, B.; Huang, J.; Li, B. High-coverage targeted lipidomics revealed dramatic lipid compositional changes in asthenozoospermic spermatozoa and inverse correlation of ganglioside GM3 with sperm motility. Reprod. Biol. Endocrinol. 2021, 19, 105. [Google Scholar] [CrossRef]
- Parks, J.E.; Graham, J.K. Effects of cryopreservation procedures on sperm membranes. Theriogenology 1992, 38, 209–222. [Google Scholar] [CrossRef]
- Evans, H.C.; Dinh, T.T.N.; Hardcastle, M.L.; Gilmore, A.A.; Ugur, M.R.; Hitit, M.; Jousan, F.D.; Nicodemus, M.C.; Memili, E. Advancing Semen Evaluation Using Lipidomics. Front. Vet. Sci. 2021, 8, 601794. [Google Scholar] [CrossRef]
- Martínez-Soto, J.C.; Landeras, J.; Gadea, J. Spermatozoa and seminal plasma fatty acids as predictors of cryopreservation success. Andrology 2013, 1, 365–375. [Google Scholar] [CrossRef] [PubMed]
- Alonso, A.; Goñi, F.M. The Physical Properties of Ceramides in Membranes. Ann. Rev. Biophys. 2018, 47, 633–654. [Google Scholar] [CrossRef]
- Fanani, M.L.; Maggio, B. The many faces (and phases) of ceramide and sphingomyelin I—Single lipids. Biophys. Rev. 2017, 9, 589–600. [Google Scholar] [CrossRef]
- Rog, T.; Koivuniemi, A. The biophysical properties of ethanolamine plasmalogens revealed by atomistic molecular dynamics simulations. Biochim. Biophys. Acta 2016, 1858, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Messias, M.C.F.; Mecatti, G.C.; Priolli, D.G.; de Oliveira Carvalho, P. Plasmalogen lipids: Functional mechanism and their involvement in gastrointestinal cancer. Lipids Health Dis. 2018, 17, 41. [Google Scholar] [CrossRef] [PubMed]
- Wallner, S.; Orsó, E.; Grandl, M.; Konovalova, T.; Liebisch, G.; Schmitz, G. Phosphatidylcholine and phosphatidylethanolamine plasmalogens in lipid loaded human macrophages. PLoS ONE 2018, 13, e0205706. [Google Scholar] [CrossRef]
- Sheriff, D.S.; Ali, E.F. Perspective on plasma membrane cholesterol efflux and spermatozoal function. J. Hum. Reprod. Sci. 2010, 3, 68–75. [Google Scholar] [CrossRef]
- Cross, N.L. Role of cholesterol in sperm capacitation. Biol. Reprod. 1998, 59, 7–11. [Google Scholar] [CrossRef]
- Brites, P.; Waterham, H.R.; Wanders, R.J. Functions and biosynthesis of plasmalogens in health and disease. Biochim. Biophys. Acta 2004, 1636, 219–231. [Google Scholar] [CrossRef]
- Li, L.H.; Hui, S.W. The effect of lipid molecular packing stress on cationic liposome-induced rabbit erythrocyte fusion. Biochim. Biophys. Acta (BBA) Biomembr. 1997, 1323, 105–116. [Google Scholar] [CrossRef]
- Poole, A.R.; Howell, J.I.; Lucy, J.A. Lysolecithin and Cell Fusion. Nature 1970, 227, 810–814. [Google Scholar] [CrossRef] [PubMed]
- Rodemer, C.; Thai, T.P.; Brugger, B.; Kaercher, T.; Werner, H.; Nave, K.A.; Wieland, F.; Gorgas, K.; Just, W.W. Inactivation of ether lipid biosynthesis causes male infertility, defects in eye development and optic nerve hypoplasia in mice. Hum. Mol. Genet. 2003, 12, 1881–1895. [Google Scholar] [CrossRef] [PubMed]
- Pinto, S.N.; Silva, L.C.; Futerman, A.H.; Prieto, M. Effect of ceramide structure on membrane biophysical properties: The role of acyl chain length and unsaturation. Biochim. Biophys. Acta (BBA) Biomembr. 2011, 1808, 2753–2760. [Google Scholar] [CrossRef] [PubMed]
- Mangold, H.K.; Weber, N. Biosynthesis and biotransformation of ether lipids. Lipids 1987, 22, 789–799. [Google Scholar] [CrossRef]
- Tavilani, H.; Doosti, M.; Abdi, K.; Vaisiraygani, A.; Joshaghani, H.R. Decreased polyunsaturated and increased saturated fatty acid concentration in spermatozoa from asthenozoospermic males as compared with normozoospermic males. Andrologia 2006, 38, 173–178. [Google Scholar] [CrossRef] [PubMed]
- WHO Laboratory Manual for the Examination and Processing of Human Semen. Available online: https://www.who.int/publications/i/item/9789240030787 (accessed on 14 April 2022).
- Furse, S.; Fernandez-Twinn, D.; Jenkins, B.; Meek, C.L.; Williams, H.E.; Smith, G.C.S.; Charnock-Jones, S.; Ozanne, S.; Koulman, A. A high throughput platform for detailed lipidomic analysis of a range of mouse and human tissues. Anal. Bioanal. Chem. 2020, 412, 2851–2862. [Google Scholar] [CrossRef]
- Furse, S.; Koulman, A. The Lipid and Glyceride Profiles of Infant Formula Differ by Manufacturer, Region and Date Sold. Nutrients 2019, 11, 1122. [Google Scholar] [CrossRef]
- Harshfield, E.L.; Koulman, A.; Ziemek, D.; Marney, L.; Fauman, E.B.; Paul, D.S.; Stacey, D.; Rasheed, A.; Lee, J.-J.; Shah, N.; et al. An unbiased lipid phenotyping approach to study the genetic determinants of lipids and their association with coronary heart disease risk factors. J. Proteome Res. 2019, 18, 2397–2410. [Google Scholar] [CrossRef]
- Cassim, A.M.; Gouguet, P.; Gronnier, J.; Laurent, N.; Germain, V.; Grison, M.; Boutté, Y.; Gerbeau-Pissot, P.; Simon-Plas, F.; Mongrand, S. Plant lipids: Key players of plasma membrane organization and function. Prog. Lipid Res. 2018, 73, 1–27. [Google Scholar] [CrossRef]

| All Participants | Motility Analysis | ||
|---|---|---|---|
| LOW MOTILITY | HIGH MOTILITY | ||
| n = 26 | n = 12 | n = 14 | |
| Age, years | 35 (20–45) | 36.5 (24–45) | 32.5 (20–40) |
| BMI, kg/m2 | 30.4 (24–36.5) | 30.2 (24–36.5) | 30.4 (26–33) |
| Abstinence, days | 4 (2–9) | 4 (3–8) | 3 (2–9) |
| Semen volume, mL | 3.4 (1.8–8.3) | 4.35 (2.8–8.3) | 3.2 (1.8–5.7) |
| Semen pH | 8.5 (8–8.5) | 8.5 (8–8.5) | 8.5 (8–8.5) |
| Sperm concentration million/mL | 26.75 (2.6–209) | 21.2 (2.6–209) | 27.25 (7.5–196) |
| Total motility % | 56 (28–73) | 45 (28–56) | 62.5 (54–73) |
| Progressive motility % | 50 (24–68) | 40 (24–49) | 58 (50–68) |
| Non-progressive motility % | 5 (1–13) | 6 (2–9) | 5 (1–13) |
| Immotile % | 44 (27–72) | 55 (44–72) | 38 (27–46) |
| Total sperm number millions/ejaculate | 91.825 (8.68–921.2) | 89.9 (8.68–606.1) | 93.325 (24–921.2) |
| HbA1c mmol/mol | 37 (28–49) | 37.5 (28–49) | 37 (32–42) |
| Random plasma glucose mmol/L | 5.2 (4.4–8.2) | 5.3 (4.4–8.2) | 5.2 (4.7–6.5) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Furse, S.; Kusinski, L.C.; Ray, A.; Glenn-Sansum, C.; Williams, H.E.L.; Koulman, A.; Meek, C.L. Relative Abundance of Lipid Metabolites in Spermatozoa across Three Compartments. Int. J. Mol. Sci. 2022, 23, 11655. https://doi.org/10.3390/ijms231911655
Furse S, Kusinski LC, Ray A, Glenn-Sansum C, Williams HEL, Koulman A, Meek CL. Relative Abundance of Lipid Metabolites in Spermatozoa across Three Compartments. International Journal of Molecular Sciences. 2022; 23(19):11655. https://doi.org/10.3390/ijms231911655
Chicago/Turabian StyleFurse, Samuel, Laura C. Kusinski, Alison Ray, Coralie Glenn-Sansum, Huw E. L. Williams, Albert Koulman, and Claire L. Meek. 2022. "Relative Abundance of Lipid Metabolites in Spermatozoa across Three Compartments" International Journal of Molecular Sciences 23, no. 19: 11655. https://doi.org/10.3390/ijms231911655
APA StyleFurse, S., Kusinski, L. C., Ray, A., Glenn-Sansum, C., Williams, H. E. L., Koulman, A., & Meek, C. L. (2022). Relative Abundance of Lipid Metabolites in Spermatozoa across Three Compartments. International Journal of Molecular Sciences, 23(19), 11655. https://doi.org/10.3390/ijms231911655






