Respiratory Depression as Antibacterial Mechanism of Linalool against Pseudomonas fragi Based on Metabolomics
Abstract
:1. Introduction
2. Results
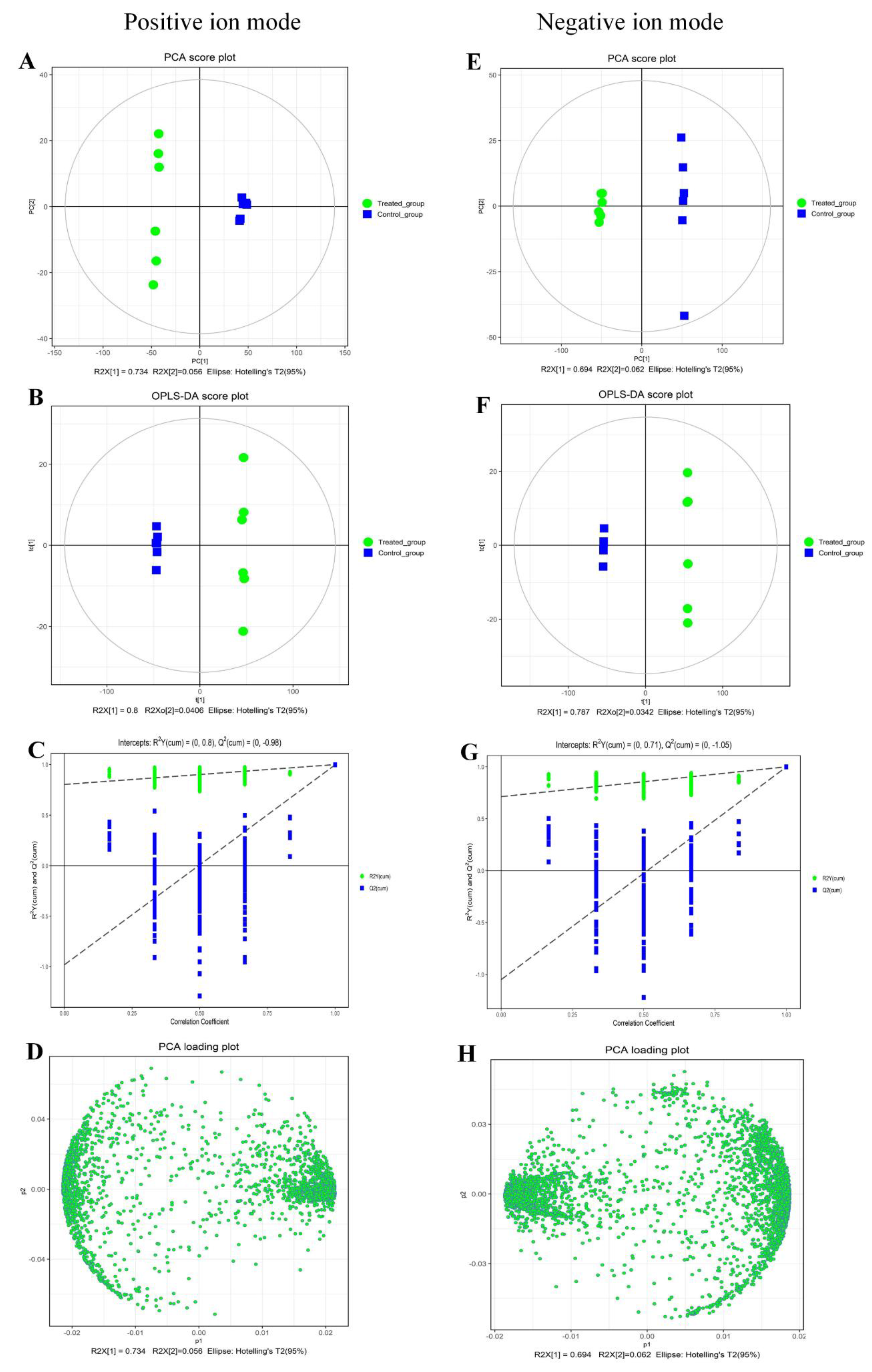
2.1. Global Analysis of Metabolomic Response
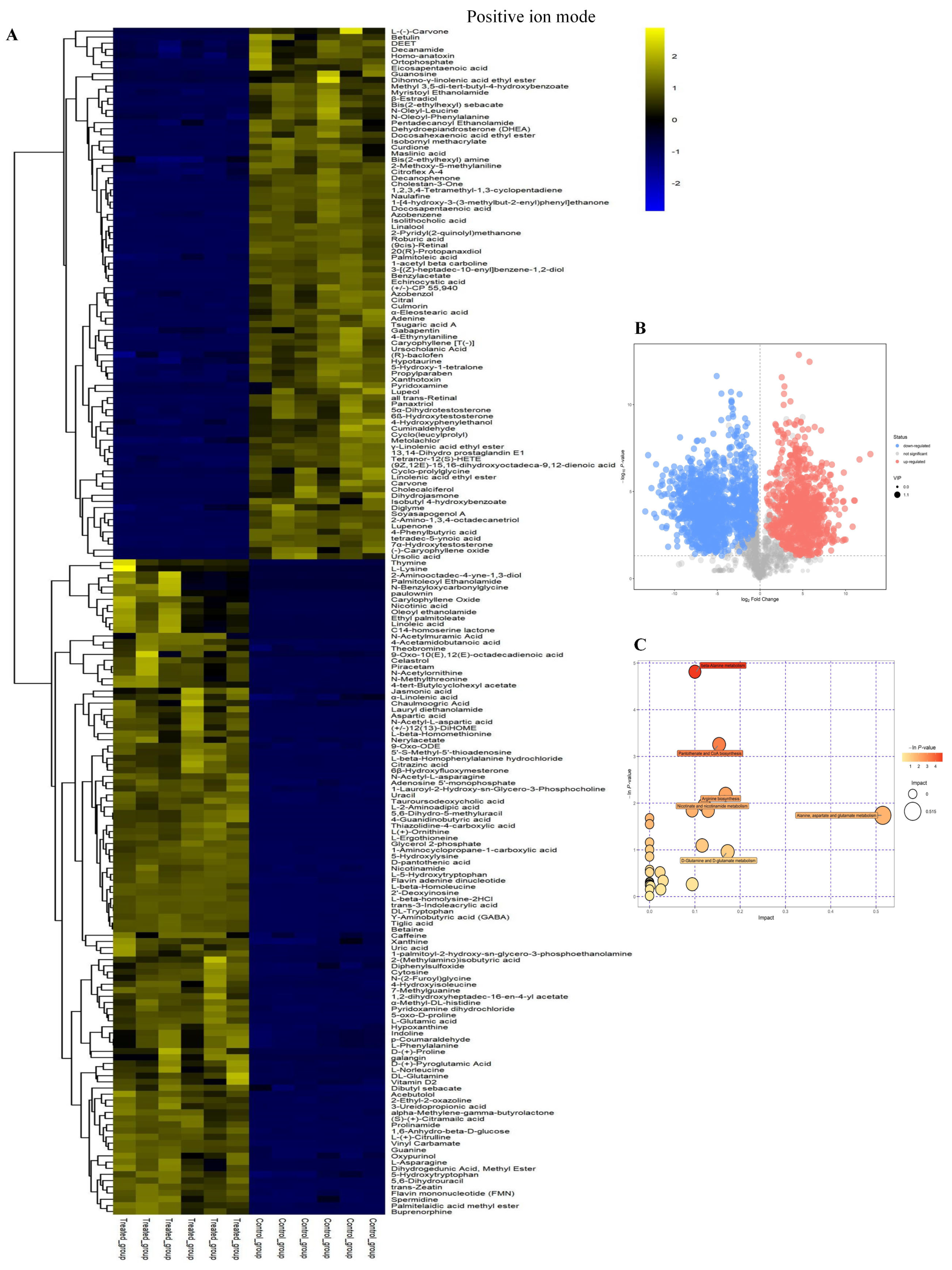
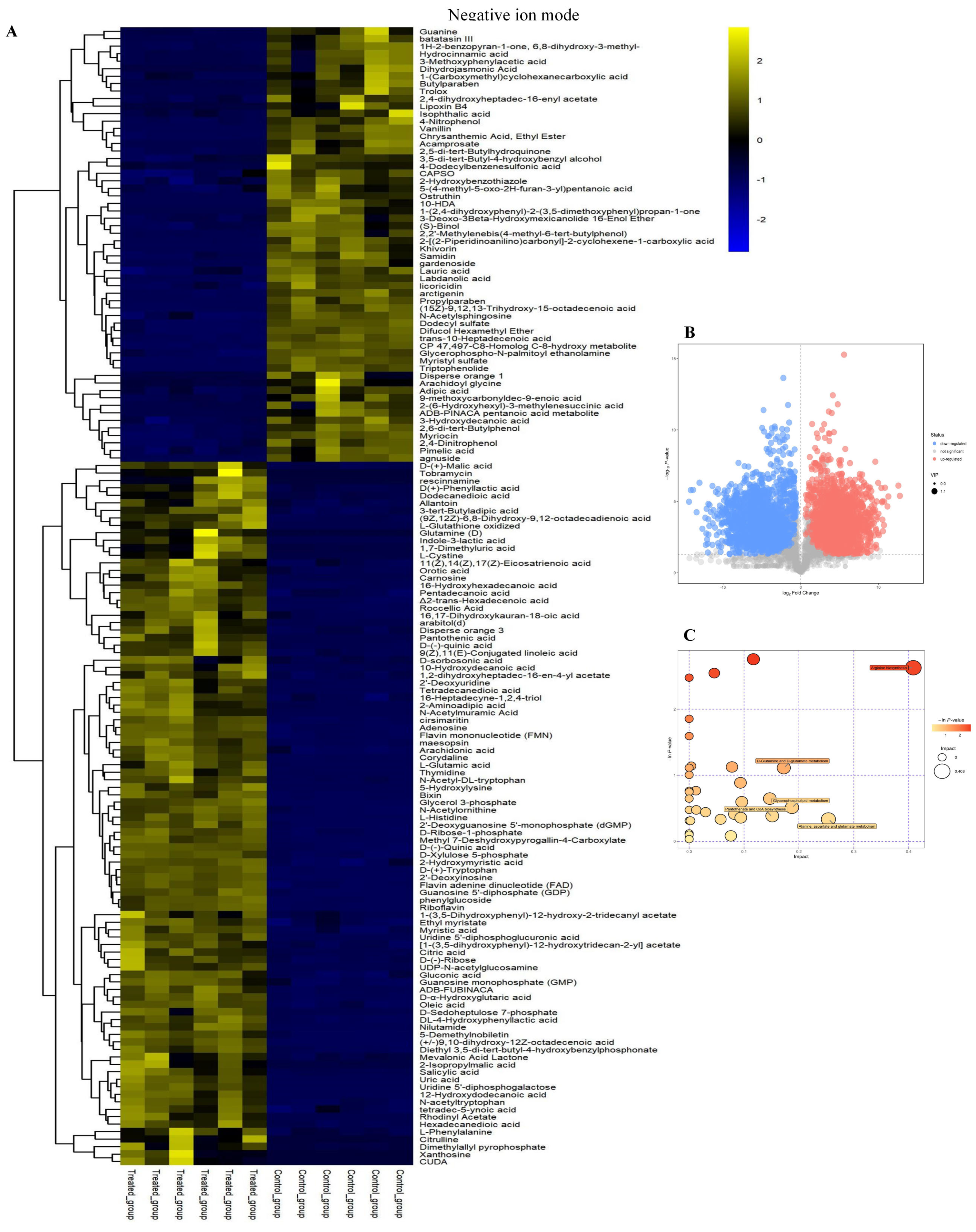
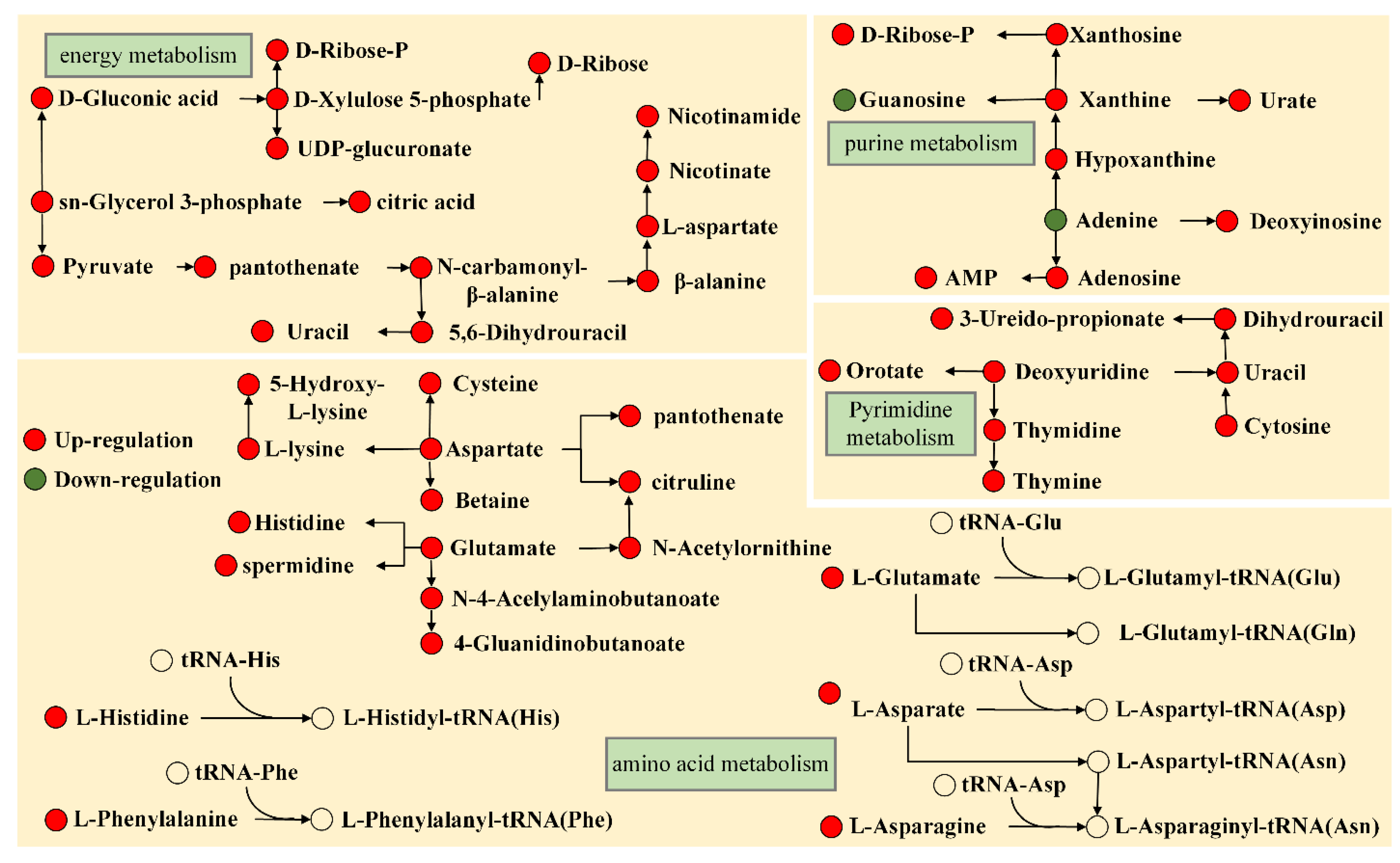
2.2. Cluster Analysis of Differential Metabolites
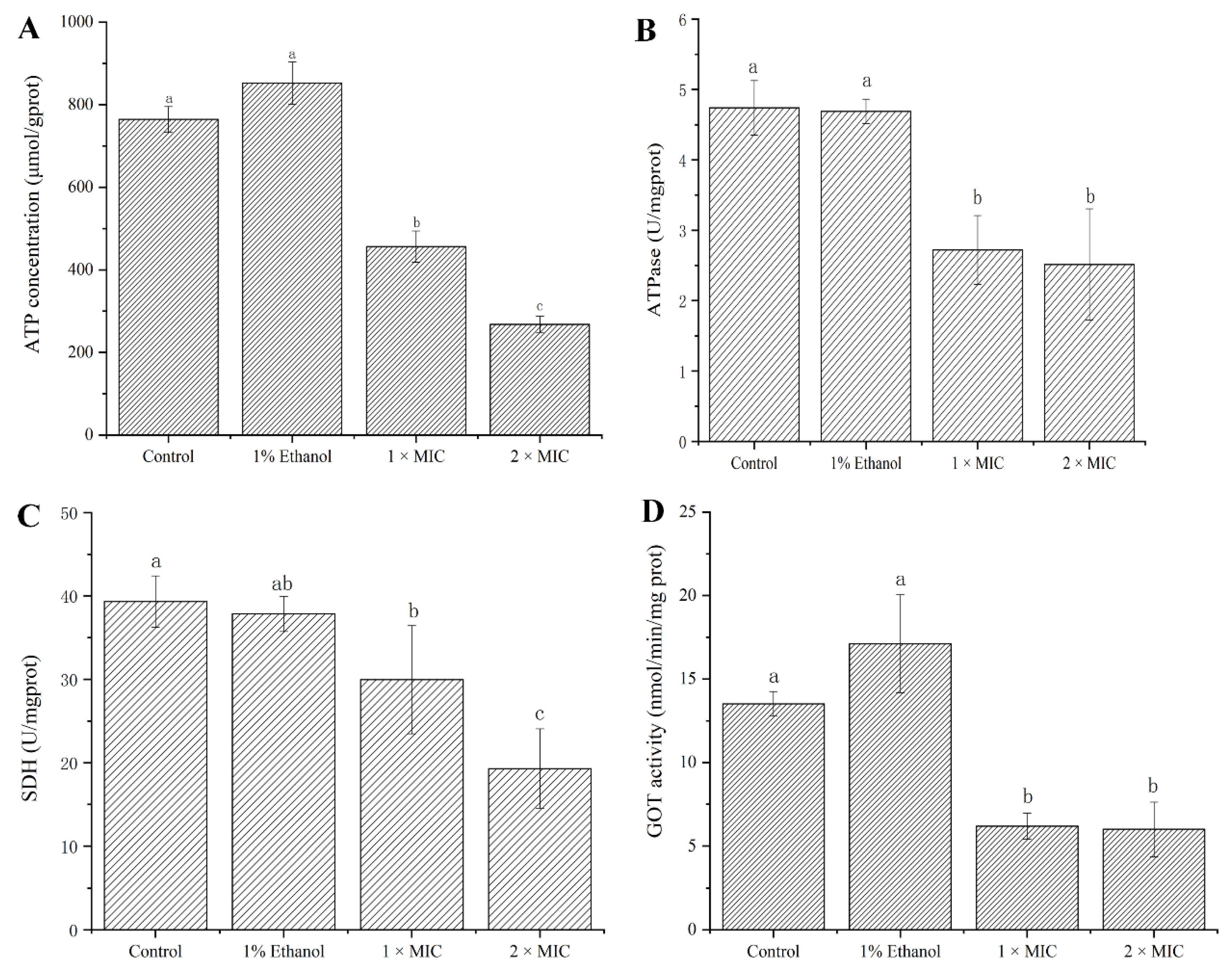
2.3. Intracellular Enzyme Activity (SDH, ATPase and GOT) and Composition (ATP) Analysis
3. Discussion
4. Materials and Methods
4.1. Materials and Bacterial Culture
4.2. Intracellular Enzyme Activity and Composition Determination
4.3. Metabolite Sample Preparation and Metabolomics Analysis
4.3.1. Sample Preparation
4.3.2. Extraction of the Intracellular Metabolites
4.3.3. UPLC-MS/MS Analysis
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Casaburi, A.; Piombino, P.; Nychas, G.J.; Villani, F.; Ercolini, D. Bacterial populations and the volatilome associated to meat spoilage. Food Microbiol. 2015, 45, 83–102. [Google Scholar] [CrossRef] [PubMed]
- Solomakos, N.; Govaris, A.; Koidis, P.; Botsoglou, N. The antimicrobial effect of thyme essential oil, nisin and their combination against Escherichia coli O157:H7 in minced beef during refrigerated storage. Meat Sci. 2008, 80, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Remenant, B.; Jaffres, E.; Dousset, X.; Pilet, M.F.; Zagorec, M. Bacterial spoilers of food: Behavior, fitness and functional properties. Food Microbiol. 2015, 45, 45–53. [Google Scholar] [CrossRef]
- Mohareb, F.; Iriondo, M.; Doulgeraki, A.I.; Van Hoek, A.; Aarts, H.; Cauchi, M.; Nychas, G.J.E. Identification of meat spoilage gene biomarkers in Pseudomonas putida using gene profiling. Food Control 2015, 57, 152–160. [Google Scholar] [CrossRef]
- Ercolini, D.; Casaburi, A.; Nasi, A.; Ferrocino, I.; Di Monaco, R.; Ferranti, P.; Mauriello, G.; Villani, F. Different molecular types of Pseudomonas fragi have the same overall behaviour as meat spoilers. Int. J. Food Microbiol. 2010, 142, 120–131. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.Y.; Ma, F.; Zeng, L.Y.; Bai, Y.; Wang, H.H.; Xu, X.L.; Zhou, G.H. Modified atmosphere packaging decreased Pseudomonas fragi cell metabolism and extracellular proteolytic activities on meat. Food Microbiol. 2018, 76, 443–449. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Fei, P.; Man, C.X.; Lou, B.B.; Niu, J.T.; Feng, J.; Sun, L.H.; Li, M.Y.; Jiang, Y.J. Tea polyphenols inactivate Cronobacter sakazakii isolated from powdered infant formula. J. Dairy Sci. 2016, 99, 1019–1028. [Google Scholar] [CrossRef]
- Bajpai, V.K.; Baek, K.H.; Kang, S.C. Control of Salmonella in foods by using essential oils: A review. Food Res. Int. 2012, 45, 722–734. [Google Scholar] [CrossRef]
- Vasilijevic, B.; Mitic-Culafic, D.; Djekic, I.; Markovic, T.; Knezevic-Vukcevic, J.; Tomasevic, I.; Velebit, B.; Nikolic, B. Antibacterial effect of Juniperus communis and Satureja montana essential oils against Listeria monocytogenes in vitro and in wine marinated beef. Food Control 2019, 100, 247–256. [Google Scholar] [CrossRef]
- Alves, S.; Duarte, A.; Sousa, S.; Domingues, F.C. Study of the major essential oil compounds of Coriandrum sativum against Acinetobacter baumannii and the effect of linalool on adhesion, biofilms and quorum sensing. Biofouling 2016, 32, 155–165. [Google Scholar] [CrossRef]
- Pereira, I.; Severino, P.; Santos, A.C.; Silva, A.M.; Souto, E.B. Linalool bioactive properties and potential applicability in drug delivery systems. Colloids Surf. B Biointerfaces 2018, 171, 566–578. [Google Scholar] [CrossRef] [PubMed]
- Aprotosoaie, A.C.; Hancianu, M.; Costache, I.I.; Miron, A. Linalool: A review on a key odorant molecule with valuable biological properties. Flavour Frag. J. 2014, 29, 193–219. [Google Scholar] [CrossRef]
- Herman, A.; Tambor, K.; Herman, A. Linalool Affects the Antimicrobial Efficacy of Essential Oils. Curr. Microbiol. 2016, 72, 165–172. [Google Scholar] [CrossRef]
- Batista, P.A.; Werner, M.F.; Oliveira, E.C.; Burgos, L.; Pereira, P.; Brum, L.F.; Santos, A.R. Evidence for the involvement of ionotropic glutamatergic receptors on the antinociceptive effect of (-)-linalool in mice. Neurosci. Lett. 2008, 440, 299–303. [Google Scholar] [CrossRef]
- Linck, V.M.; da Silva, A.L.; Figueiro, M.; Caramao, E.B.; Moreno, P.R.H.; Elisabetsky, E. Effects of inhaled Linalool in anxiety, social interaction and aggressive behavior in mice. Phytomedicine 2010, 17, 679–683. [Google Scholar] [CrossRef] [PubMed]
- He, R.; Zhang, Z.; Xu, L.; Chen, W.; Zhang, M.; Zhong, Q.; Chen, H.; Chen, W. Antibacterial mechanism of linalool emulsion against Pseudomonas aeruginosa and its application to cold fresh beef. World J. Microbiol. Biotechnol. 2022, 38, 56. [Google Scholar] [CrossRef] [PubMed]
- Guo, F.Y.; Chen, Q.P.; Liang, Q.; Zhang, M.; Chen, W.X.; Chen, H.M.; Yun, Y.H.; Zhong, Q.P.; Chen, W.J. Antimicrobial Activity and Proposed Action Mechanism of Linalool Against Pseudomonas fluorescens. Front. Microbiol. 2021, 12, 562094. [Google Scholar] [CrossRef]
- He, R.R.; Chen, W.J.; Chen, H.M.; Zhong, Q.P.; Zhang, H.L.; Zhang, M.; Chen, W.X. Antibacterial mechanism of linalool against L. monocytogenes, a metabolomic study. Food Control 2022, 132, 108533. [Google Scholar] [CrossRef]
- Ngome, M.T.; Alves, J.G.L.F.; de Oliveira, A.C.F.; Machado, P.D.; Mondragon-Bernal, O.L.; Piccoli, R.H. Linalool, citral, eugenol and thymol: Control of planktonic and sessile cells of Shigella flexneri. Amb. Express 2018, 8, 105. [Google Scholar] [CrossRef]
- Liu, X.; Cai, J.X.; Chen, H.M.; Zhong, Q.P.; Hou, Y.Q.; Chen, W.J.; Chen, W.X. Antibacterial activity and mechanism of linalool against Pseudomonas aeruginosa. Microb. Pathogenesis 2020, 141, 13980. [Google Scholar] [CrossRef]
- He, R.; Zhong, Q.; Chen, W.; Zhang, M.; Pei, J.; Chen, H.; Chen, W. Antimicrobial mechanism of linalool against Brochothrix thermosphacta and its application on chilled beef. Food Res. Int. 2022, 157, 111407. [Google Scholar] [CrossRef] [PubMed]
- Calo, J.R.; Crandall, P.G.; O’Bryan, C.A.; Ricke, S.C. Essential oils as antimicrobials in food systems—A review. Food Control 2015, 54, 111–119. [Google Scholar] [CrossRef]
- Gaupp, R.; Lei, S.L.; Reed, J.M.; Peisker, H.; Boyle-Vavra, S.; Bayer, A.S.; Bischoff, M.; Herrmann, M.; Daum, R.S.; Powers, R.; et al. Staphylococcus aureus Metabolic Adaptations during the Transition from a Daptomycin Susceptibility Phenotype to a Daptomycin Nonsusceptibility Phenotype. Antimicrob. Agents Chemother. 2015, 59, 4226–4238. [Google Scholar] [CrossRef] [PubMed]
- Tolstikov, V.; Nikolayev, A.; Dong, S.; Zhao, G.; Kuo, M.S. Metabolomics Analysis of Metabolic Effects of Nicotinamide Phosphoribosyltransferase (NAMPT) Inhibition on Human Cancer Cells. PLoS ONE 2014, 9, e114019. [Google Scholar]
- Guo, F.; Liang, Q.; Zhang, M.; Chen, W.; Chen, H.; Yun, Y.; Zhong, Q.; Chen, W. Antibacterial Activity and Mechanism of Linalool against Shewanella putrefaciens. Molecules 2021, 26, 245. [Google Scholar] [CrossRef]
- Want, E.J.; Nordstrom, A.; Morita, H.; Siuzdak, G. From exogenous to endogenous: The inevitable imprint of mass spectrometry in metabolomics. J. Proteome Res. 2007, 6, 459–468. [Google Scholar] [CrossRef]
- Zhang, R.R.; Shi, Y.G.; Gu, Q.; Fang, M.; Chen, Y.W.; Fang, S.; Dang, Y.L.; Chen, J.S. Antimicrobial effect and mechanism of non-antibiotic alkyl gallates against Pseudomonas fluorescens on the surface of Russian sturgeon (Acipenser gueldenstaedti). Int. J. Food Microbiol. 2021, 342, 109093. [Google Scholar] [CrossRef]
- Xia, H.; Tang, H.; Wang, F.; Yang, X.; Wang, Z.; Liu, H.; Pan, D.; Yang, C.; Wang, S.; Sun, G. An untargeted metabolomics approach reveals further insights of Lycium barbarum polysaccharides in high fat diet and streptozotocin-induced diabetic rats. Food Res. Int. 2019, 116, 20–29. [Google Scholar] [CrossRef]
- Jia, L.N.; Kosgey, J.C.; Wang, J.L.; Yang, J.X.; Nyamao, R.M.; Zhao, Y.; Teng, X.; Gao, L.; Wabo, M.C.; Vasilyeva, N.V.; et al. Antimicrobial and mechanism of antagonistic activity of Bacillus sp. A2 against pathogenic fungus and bacteria: The implication on honey’s regulatory mechanism on host’s microbiota. Food Sci. Nutr. 2020, 8, 4857–4867. [Google Scholar] [CrossRef]
- Yu, M.; Zhu, Y.; Cong, Q.; Wu, C. Metabonomics Research Progress on Liver Diseases. Can. J. Gastroenterol. Hepatol. 2017, 2017, 8467192. [Google Scholar] [CrossRef]
- Liu, M.M.; Feng, M.X.; Yang, K.; Cao, Y.F.; Zhang, J.; Xu, J.N.; Hernandez, S.H.; Wei, X.Y.; Fan, M.T. Transcriptomic and metabolomic analyses reveal antibacterial mechanism of astringent persimmon tannin against Methicillin-resistant Staphylococcus aureus isolated from pork. Food Chem. 2020, 309, 125692. [Google Scholar] [CrossRef] [PubMed]
- Leonardi, R.; Jackowski, S. Biosynthesis of Pantothenic Acid and Coenzyme A. EcoSal Plus 2007, 2. [Google Scholar] [CrossRef] [PubMed]
- Stincone, A.; Prigione, A.; Cramer, T.; Wamelink, M.M.; Campbell, K.; Cheung, E.; Olin-Sandoval, V.; Gruning, N.M.; Kruger, A.; Tauqeer Alam, M.; et al. The return of metabolism: Biochemistry and physiology of the pentose phosphate pathway. Biol. Rev. Camb. Philos. Soc. 2015, 90, 927–963. [Google Scholar] [CrossRef] [PubMed]
- Tokuda, K.; Baron, B.; Yamashiro, C.; Kuramitsu, Y.; Kitagawa, T.; Kobayashi, M.; Sonoda, K.H.; Kimura, K. Up-regulation of the pentose phosphate pathway and HIF-1alpha expression during neural progenitor cell induction following glutamate treatment in rat ex vivo retina. Cell Biol. Int. 2019, 44, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Park, J.Y.; Jo, S.K.; Park, K.M.; Yu, H.; Bai, J.; Ryu, S.; Chang, P.S. Transcriptomic analysis of Staphylococcus aureus under the stress condition of antibacterial erythorbyl laurate by RNA sequencing. Food Control 2019, 96, 1–8. [Google Scholar] [CrossRef]
- Xie, M.D.; Chen, W.Q.; Lai, X.C.; Dai, H.B.; Sun, H.; Zhou, X.Y.; Chen, T.B. Metabolic responses and their correlations with phytochelatins in Amaranthus hypochondriacus under cadmium stress. Environ. Pollut. 2019, 252, 1791–1800. [Google Scholar] [CrossRef]
- Shayanfar, S.; Broumand, A.; Pillai, S.D. Acid stress induces differential accumulation of metabolites in Escherichia coli O26:H11. J. Appl. Microbiol. 2018, 125, 1911–1919. [Google Scholar] [CrossRef]
- Nazzaro, F.; Fratianni, F.; De Martino, L.; Coppola, R.; De Feo, V. Effect of essential oils on pathogenic bacteria. Pharmaceuticals 2013, 6, 1451–1474. [Google Scholar] [CrossRef]
- Hu, W.; Li, C.Z.; Dai, J.M.; Cui, H.Y.; Lin, L. Antibacterial activity and mechanism of Litsea cubeba essential oil against methicillin-resistant Staphylococcus aureus (MRSA). Ind. Crop Prod. 2019, 130, 34–41. [Google Scholar] [CrossRef]
- Li, Y.S.; He, R.R.; Cai, J.X.; Chen, D.; Chen, W.X. Antibacterial activity and mechanism of linalool against Pseudomonas fragi. J. Food Sci. Technol. 2022, 40, 55–63. [Google Scholar]
| Strain | Control | The Concentration of Inhibitor (mL/L) | |||||
|---|---|---|---|---|---|---|---|
| P. fragi | Water | 1% Ethanol | 0.375 | 0.75 | 1.5 | 3 | 6 |
| +++ | +++ | +++ | ++ | - | - | - | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Y.; He, R.; Chen, H.; Chen, D.; Chen, W. Respiratory Depression as Antibacterial Mechanism of Linalool against Pseudomonas fragi Based on Metabolomics. Int. J. Mol. Sci. 2022, 23, 11586. https://doi.org/10.3390/ijms231911586
Li Y, He R, Chen H, Chen D, Chen W. Respiratory Depression as Antibacterial Mechanism of Linalool against Pseudomonas fragi Based on Metabolomics. International Journal of Molecular Sciences. 2022; 23(19):11586. https://doi.org/10.3390/ijms231911586
Chicago/Turabian StyleLi, Yuansong, Rongrong He, Haiming Chen, Da Chen, and Wenxue Chen. 2022. "Respiratory Depression as Antibacterial Mechanism of Linalool against Pseudomonas fragi Based on Metabolomics" International Journal of Molecular Sciences 23, no. 19: 11586. https://doi.org/10.3390/ijms231911586
APA StyleLi, Y., He, R., Chen, H., Chen, D., & Chen, W. (2022). Respiratory Depression as Antibacterial Mechanism of Linalool against Pseudomonas fragi Based on Metabolomics. International Journal of Molecular Sciences, 23(19), 11586. https://doi.org/10.3390/ijms231911586






