LC-PDA-MS and GC-MS Analysis of Scorzonera hispanica Seeds and Their Effects on Human Breast Cancer Cell Lines
Abstract
1. Introduction
2. Results
2.1. GC-MS Analysis of SH1, SH9-SH12
2.2. LC-PDA-MS Characterization of SH1-SH8
2.2.1. Qualitative Analysis
2.2.2. Quantitative Analysis
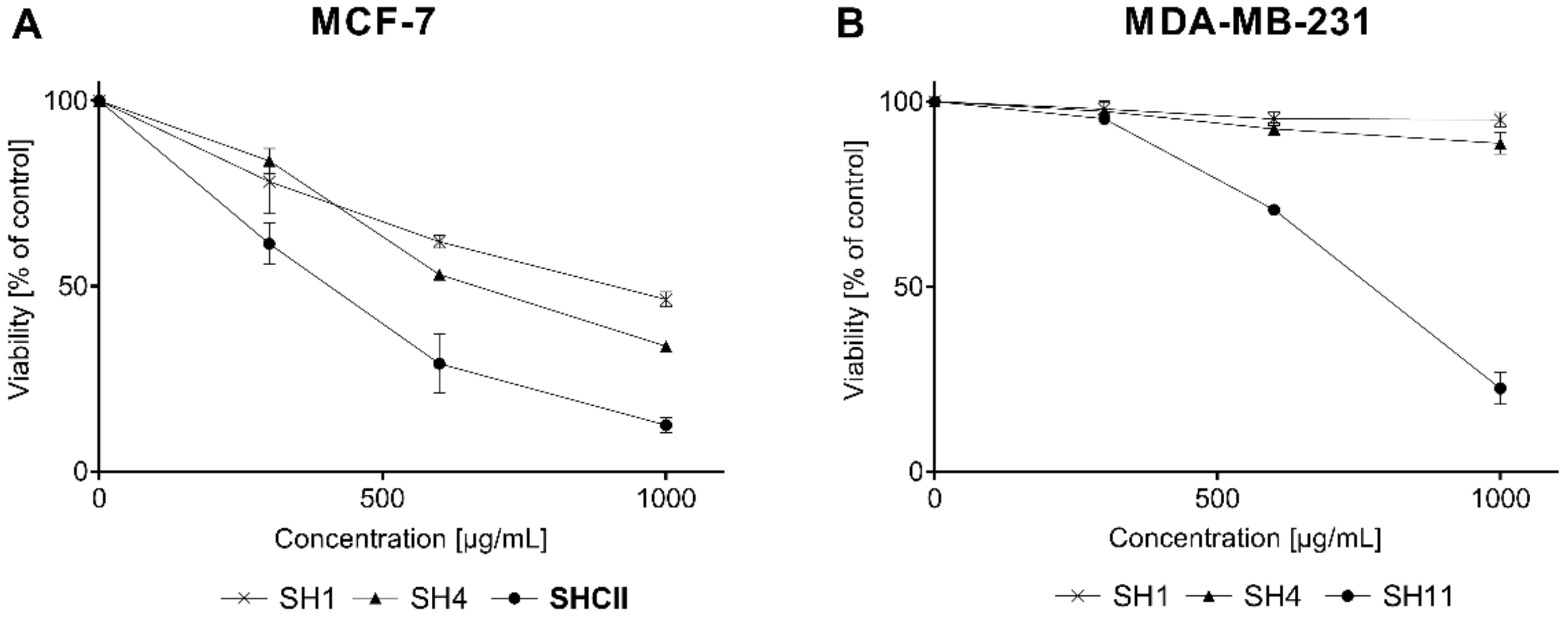
2.3. Cell Viability Assay
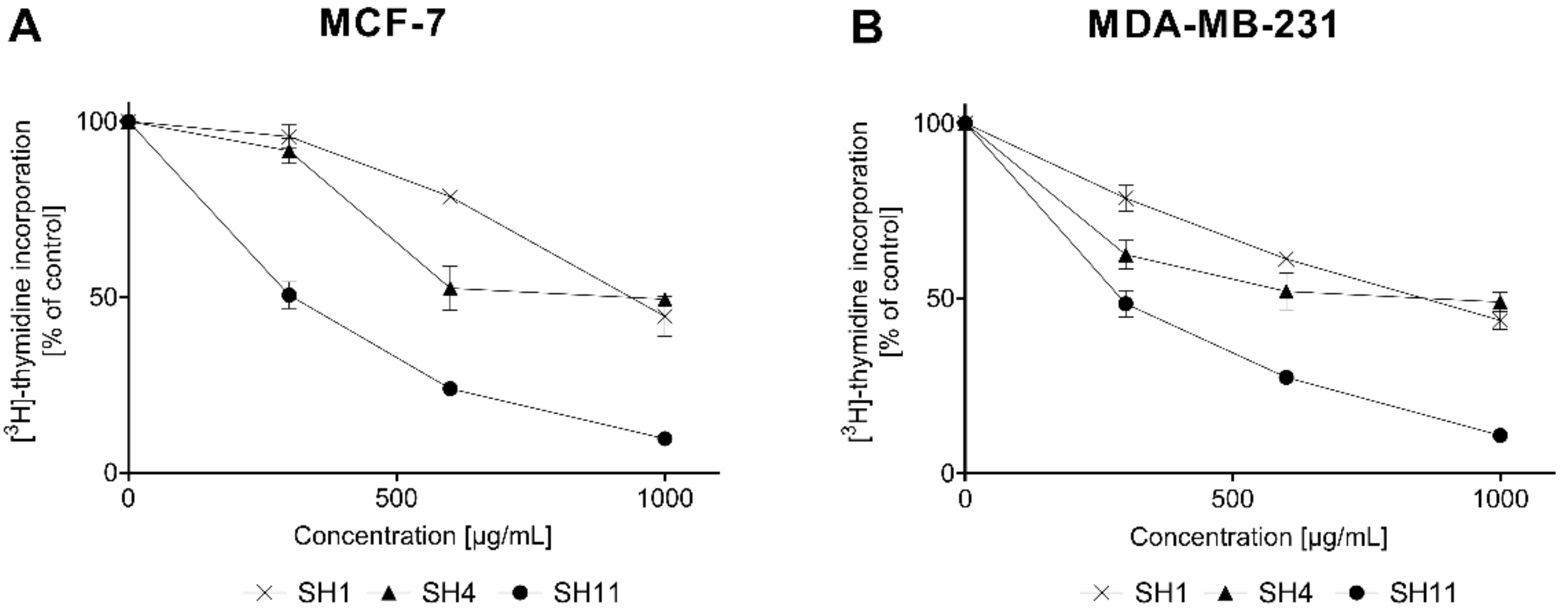
2.4. DNA Biosynthesis Assay
2.5. Annexin V/PI Binding Assay
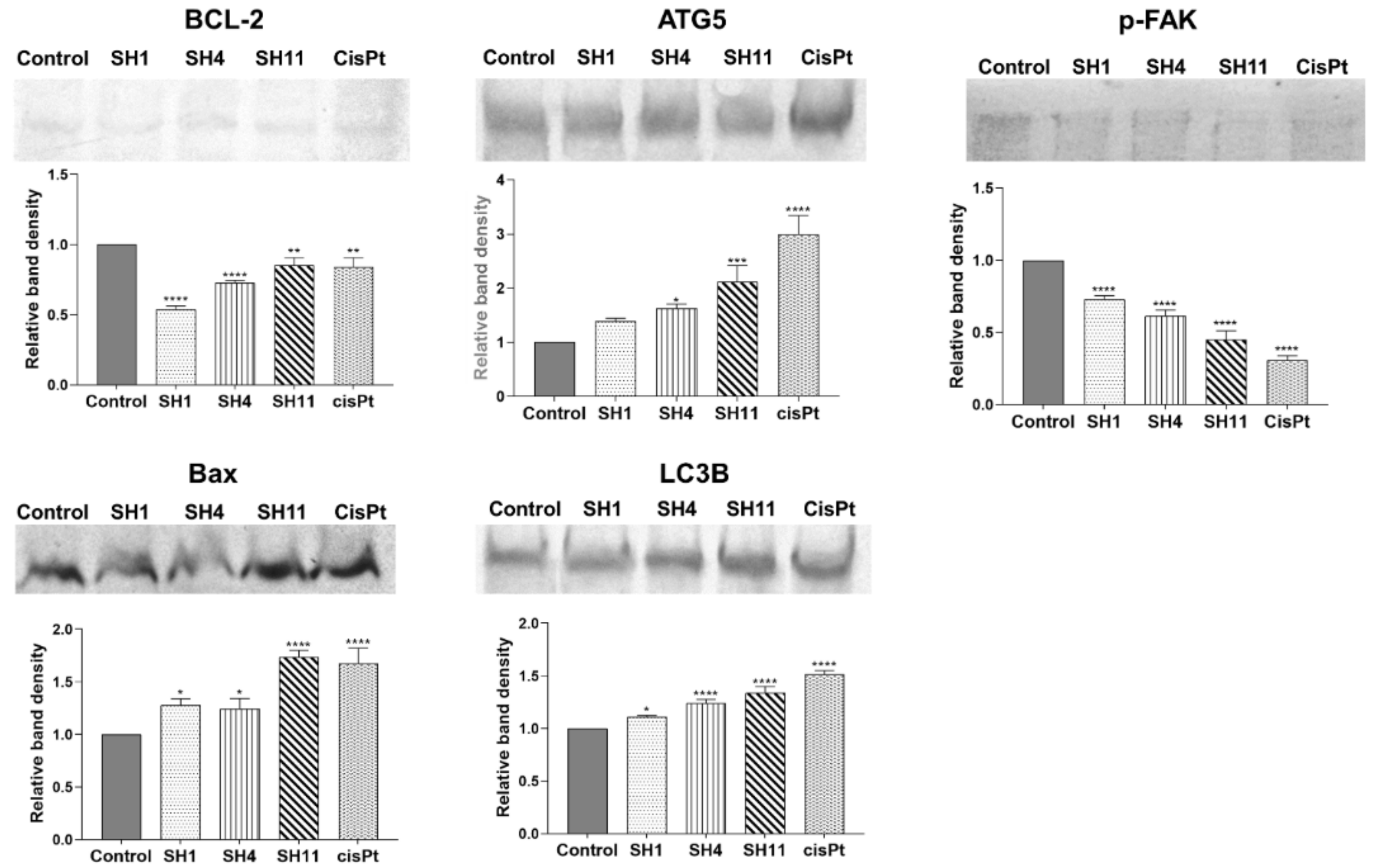
2.6. Western Blot Evaluation of the Expression of Apoptosis- and Autophagy-Related Protein
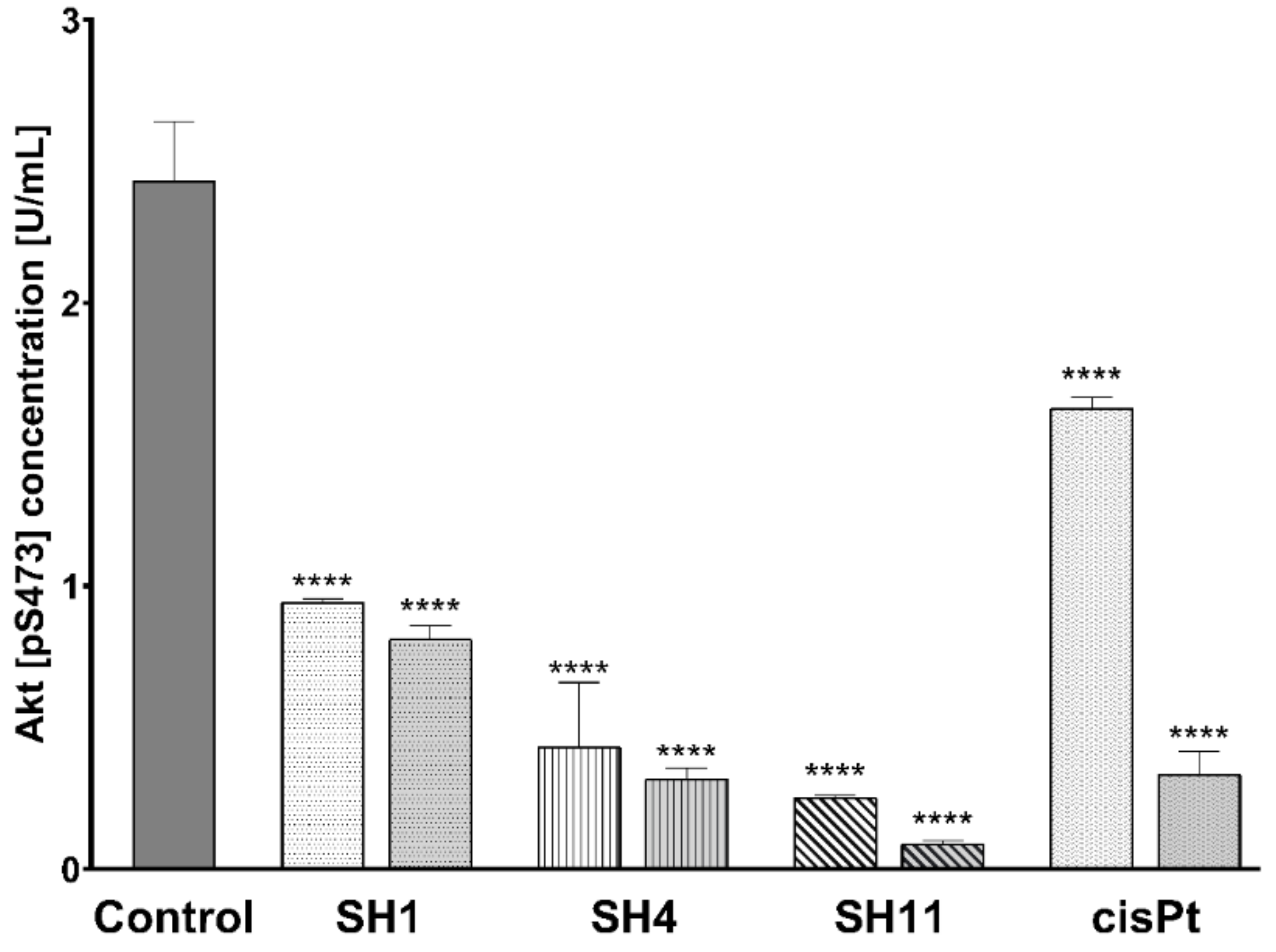
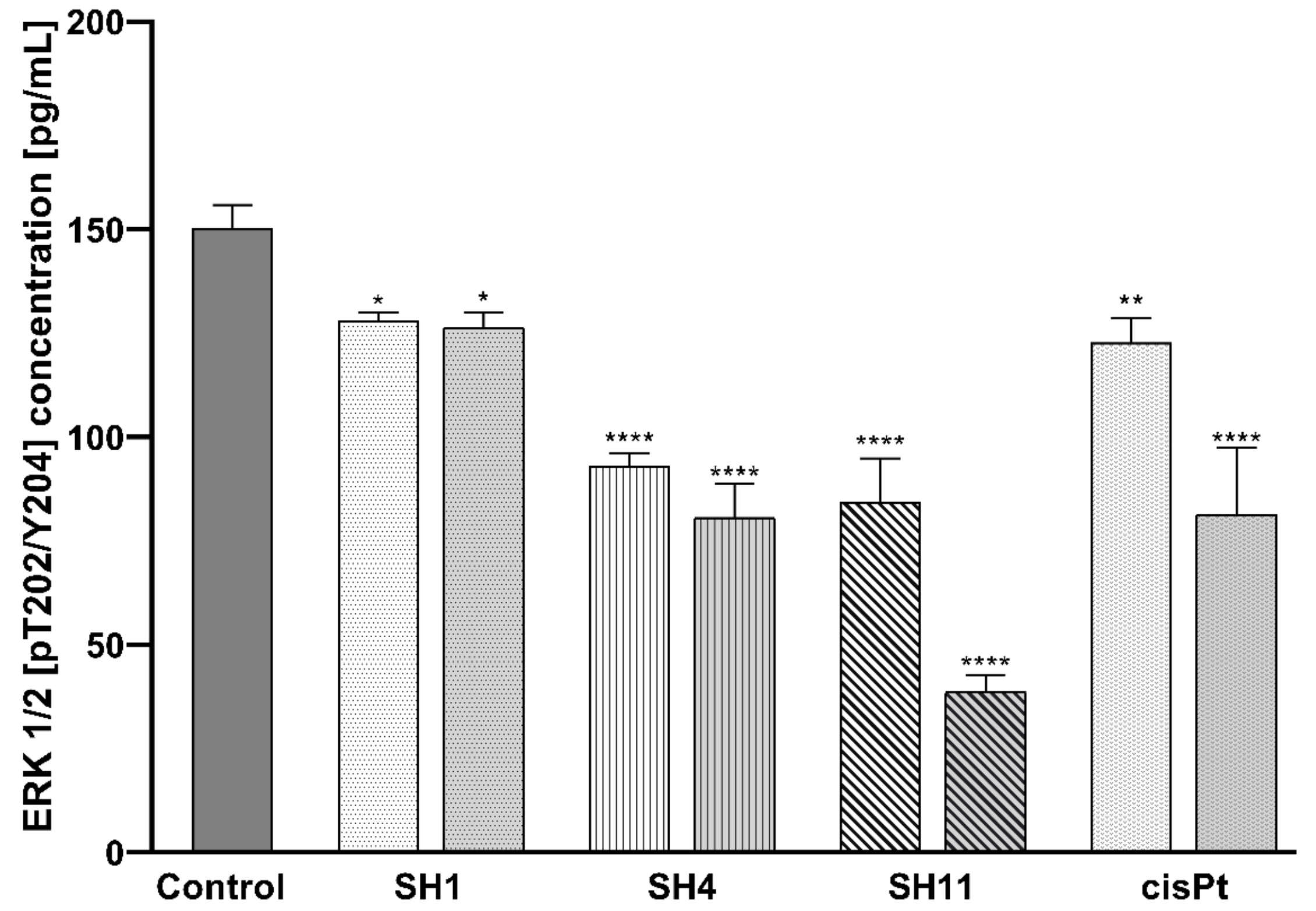
2.7. Influence of SH1, SH4, and SH11 on the Expression of Proteins Related to Cell Survival and Proliferation
2.8. Influence of SH1, SH4, and SH11 on the Concentration of TNF-α, Interleukin-8, and Interleukin-10
3. Discussion
4. Materials and Methods
4.1. Chemicals and Equipment
4.2. Plant Material, Preparation of Extracts, Fractions, and Oil
4.3. GC-MS Analysis of SH1, SH9-SH12
4.4. Identification of the Chemical Composition of SH1, SH9-SH12
4.5. LC-PDA-MS Conditions
4.6. LC-PDA-MS Optimization and Validation
4.6.1. Preparation of Standard Solutions and Samples
4.6.2. Chromatographic Optimization
4.7. Cell Culture
4.8. Cell Viability Assay
4.9. DNA Biosynthesis Assay
4.10. Flow Cytometry Evaluation of Annexin V Binding
4.11. Analysis of Protein Expression Using Western Blot Technique
4.12. Analysis of Protein Concentration Using ELISA Technique
4.13. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| 3:5-dCQa | 3:5-di-caffeoylquinic acid |
| 3-CQa | 3-caffeoylquinic acid |
| 4,5-dCQa | 4,5-di-caffeoylquinic acid |
| 4-CQa | 4-caffeoylquinic acid |
| 5-CQa | 5-caffeoylquinic acid |
| A | Apigenin |
| ACN | Acetonitrile |
| Akt | Protein kinase B |
| ATG5 | Autophagy-related protein 5 |
| BAK | BCL-2 homologous antagonist |
| Bax | BCL-2-like protein 4 |
| BCL-2 | B-cell lymphoma 2 |
| BCL-xL | B-cell lymphoma extra-large |
| CA | Caffeic acid |
| cisPt | Cisplatin |
| CLA | Conjugated linoleic acid |
| DMEM | Dulbecco’s modification of Eagle medium |
| EDTA | Ethylenediaminetetraacetic acid |
| EI | Electron ionization |
| ERK | Extracellular signal-regulated kinase |
| FA | Formic acid |
| FAK | Focal adhesion kinase |
| FBS | Fetal bovine serum |
| GC-MS | Gas chromatography-mass spectrometry |
| IC25 | Quarter-maximal inhibitory concentration |
| IC50 | Half-maximal inhibitory concentration |
| IL-8 | Interleukin 8 |
| LA | Linoleic acid |
| LC-PDA-MS | Liquid chromatography-photodiode array-mass spectrometry |
| LC3B | Light chain 3B |
| LOD | Limit of detection |
| LOQ | Limit of quantification |
| MTT | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide |
| NF-κB | Nuclear factor kappa B |
| OA | Oleic acid |
| PA | Palmitic acid |
| PAGE | Polyacrylamide gel electrophoresis |
| PBS | Phosphate-buffered saline |
| PI | Propidium iodide |
| PI3K | Phosphoinositide 3-kinase |
| QA | Quinic acid |
| RI | Retention index |
| SDS | Sodium dodecyl sulfate |
| TBS-T | TRIS-buffered saline with Tween 20 |
| TCA | Trichloroacetic acid |
| TIC | Total ion current |
| TMS | Trimethylsilyl |
| TNF-α | Tumor necrosis factor-alpha |
| TRIS | Tris(hydroxymethyl)aminomethane |
| UFA | Unsaturated fatty acids |
| UPW | Ultra-pure water |
References
- Duran, A.; Hamzaoğlu, E. A New Species of Scorzonera L. (Asteraceae) from South Anatolia, Turkey. Biologia 2004, 59, 47–50. [Google Scholar]
- Zaika, M.A.; Kilian, N.; Jones, K.; Krinitsina, A.A.; Nilova, M.V.; Speranskaya, A.S.; Sukhorukov, A.P. Scorzonera sensu lato (Asteraceae, Cichorieae)—Taxonomic reassessment in the light of new molecular phylogenetic and carpological analyses. PhytoKeys 2020, 137, 1–85. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Edrada-Ebel, R.; Tsevegsuren, N.; Sendker, J.; Braun, M.; Wray, V.; Lin, W.; Proksch, P. Dihydrostilbene derivatives from the Mongolian medicinal plant Scorzonera radiata. J. Nat. Prod. 2009, 72, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Buranov, A.U.; Elmuradov, B.J. Extraction and characterization of latex and natural rubber from rubber-bearing plants. J. Agric. Food Chem. 2010, 58, 734–743. [Google Scholar] [CrossRef]
- Bahadır Acıkara, Ö.; Citoğlu Gülçin, S.; Dall’Acqua, S.; Özbek, H.; Cvačka, J.; Žemlička, M.; Šmejkal, K. Bioassay-guided isolation of the antinociceptive compounds motiol and β-sitosterol from Scorzonera latifolia root extract. Pharmazie 2014, 69, 711–714. [Google Scholar] [CrossRef]
- Karakaya, S.; Polat, A.; Aksakal, Ö.; Sümbüllü, Y.Z.; İncekara, Ü. Ethnobotanical study of medicinal plants in Aziziye district (Erzurum, Turkey). Turk. J. Pharm. Sci. 2020, 17, 211–220. [Google Scholar] [CrossRef]
- Yaldiz, G.; Koca Çalişkan, U.; Aka, C. In vitro screening of natural drug potentials for mass production. Not. Bot. Horti Agrobot. Cluj-Napoca 2017, 45, 292–300. [Google Scholar] [CrossRef][Green Version]
- Tsevegsuren, N.; Edrada, R.A.; Lin, W.; Ebel, R.; Torre, C.; Ortlepp, S.; Wray, V.; Proksch, P. Biologically active natural products from Mongolian medicinal plants Scorzonera divaricata and Scorzonera pseudodivaricata. J. Nat. Prod. 2007, 70, 962–967. [Google Scholar] [CrossRef]
- Lendzion, K.; Gornowicz, A.; Bielawski, K.; Bielawska, A. phytochemical composition and biological activities of Scorzonera species. Int. J. Mol. Sci. 2021, 22, 5128. [Google Scholar] [CrossRef]
- Wu, Q.-X.; Su, Y.-B.; Zhu, Y. Triterpenes and steroids from the roots of Scorzonera austriaca. Fitoterapia 2011, 82, 493–496. [Google Scholar] [CrossRef]
- Yang, Y.-J.; Yao, J.; Jin, X.-J.; Shi, Z.-N.; Shen, T.-F.; Fang, J.-G.; Yao, X.-J.; Zhu, Y. Sesquiterpenoids and tirucallane triterpenoids from the roots of Scorzonera divaricata. Phytochemistry 2016, 124, 86–98. [Google Scholar] [CrossRef] [PubMed]
- Bahadır-Acıkara, Ö.; Özbilgin, S.; Saltan-İşcan, G.; Dall’Acqua, S.; Rjašková, V.; Özgökçe, F.; Suchý, V.; Šmejkal, K. Phytochemical analysis of Podospermum and Scorzonera n-hexane extracts and the HPLC quantitation of triterpenes. Molecules 2018, 23, 1813. [Google Scholar] [CrossRef] [PubMed]
- Bahadır Acıkara, Ö.; Akkol, E.K.; Süntar, I.; Ergene, B.; Saltan-Çitoğlu, G.; Çoban, T. Assessment of anti-inflammatory and free radical scavenger activities of selected Scorzonera species and determination of active components. Int. J. Pharmacogn. Phytochem. Res. 2014, 6, 492–498. [Google Scholar]
- Donia, A.E.R.M. Phytochemical and pharmacological studies on Scorzonera alexandrina Boiss. J. Saudi Chem. Soc. 2016, 20, S433–S439. [Google Scholar] [CrossRef]
- Akkol, E.K.; Acikara, O.B.; Süntar, I.; Citolu, G.S.; Kele, H.; Ergene, B. Enhancement of wound healing by topical application of Scorzonera species: Determination of the constituents by HPLC with new validated reverse phase method. J. Ethnopharmacol. 2011, 137, 1018–1027. [Google Scholar] [CrossRef]
- Akkol, E.K.; Šmejkal, K.; Kurtul, E.; Ilhan, M.; Güragac, F.T.; Çitoğlu, G.S.; Acıkara, Ö.B.; Cvačka, J.; Buděšínský, M. Inhibitory activity of Scorzonera latifolia and its components on enzymes connected with healing process. J. Ethnopharmacol. 2019, 245, 112168. [Google Scholar] [CrossRef]
- Granica, S.; Lohwasser, U.; Jöhrer, K.; Zidorn, C. Qualitative and quantitative analyses of secondary metabolites in aerial and subaerial of Scorzonera hispanica L. (black salsify). Food Chem. 2015, 173, 321–331. [Google Scholar] [CrossRef]
- Zidorn, C.; Ellmerer-Müller, E.P.; Stuppner, H. Sesquiterpenoids from Scorzonera hispanica L. Pharmazie 2000, 55, 550–551. [Google Scholar]
- Granica, S.; Zidorn, C. Phenolic compounds from aerial parts as chemosystematic markers in the Scorzonerinae (Asteraceae). Biochem. Syst. Ecol. 2015, 58, 102–113. [Google Scholar] [CrossRef]
- Petkova, N. Characterization of inulin from black salsify (Scorzonera hispanica L.) for food and pharmaceutical purposes. Asian J. Pharm. Clin. Res. 2018, 11, 221–225. [Google Scholar] [CrossRef]
- Park, B.Y.; Oh, S.R.; Ahn, K.S.; Kwon, O.K.; Lee, H.K. (-)-Syringaresinol inhibits proliferation of human promyelocytic HL-60 leukemia cells via G1 arrest and apoptosis. Int. Immunopharmacol. 2008, 8, 967–973. [Google Scholar] [CrossRef] [PubMed]
- Jeong, Y.H.; Chung, S.Y.; Han, A.R.; Sung, M.K.; Jang, D.S.; Lee, J.; Kwon, Y.; Lee, H.J.; Seo, E.K. P-glycoprotein inhibitory activity of two phenolic compounds, (-)-syringaresinol and tricin from Sasa borealis. Chem. Biodivers. 2007, 4, 12–16. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Zhang, L.; Shi, J.; Liu, Y.; Zhou, L.; Hou, K.; Qu, X.; Teng, Y. clinical significance of pAkt and pErk1/2 expression in early-stage breast cancer patients treated with anthracycline-based adjuvant chemotherapy. Oncol Lett 2015, 9, 1707–1714. [Google Scholar] [CrossRef] [PubMed]
- Han, Q.; Xiao, F.; Ma, L.; Zhou, J.; Wang, L.; Cheng, H.; Zhu, J.; Yao, F.; Lyu, J.; Du, L. DDR1 promotes migration and invasion of breast cancer by modulating the Src-FAK signaling. Neoplasma 2022, 220316N289. [Google Scholar] [CrossRef] [PubMed]
- Sulzmaier, F.J.; Jean, C.; Schlaepfer, D.D. FAK in cancer: Mechanistic findings and clinical applications. Nat. Rev. Cancer 2014, 14, 598–610. [Google Scholar] [CrossRef]
- Lindsay, J.; Esposti, M.D.; Gilmore, A.P. Bcl-2 Proteins and Mitochondria—Specificity in membrane targeting for death. Biochim. Biophys. Acta (BBA) Mol. Cell Res. 2011, 1813, 532–539. [Google Scholar] [CrossRef]
- Marquez, R.T.; Xu, L. Bcl-2:Beclin 1 Complex: Multiple, Mechanisms Regulating Autophagy/Apoptosis Toggle Switch. Am. J. Cancer Res. 2012, 2, 214. [Google Scholar]
- Buzun, K.; Gornowicz, A.; Lesyk, R.; Bielawski, K.; Bielawska, A. Autophagy Modulators in Cancer Therapy. Int. J. Mol. Sci. 2021, 22, 5804. [Google Scholar] [CrossRef]
- Waugh, D.J.J.; Wilson, C. The Interleukin-8 pathway in Cancer. Clin. Cancer Res. 2008, 14, 6735–6741. [Google Scholar] [CrossRef]
- Cruceriu, D.; Baldasici, O.; Balacescu, O.; Berindan-Neagoe, I. The Dual Role of Tumor Necrosis Factor-Alpha (TNF-α) in Breast Cancer: Molecular Insights and Therapeutic Approaches. Cell. Oncol. 2020, 43, 1–8. [Google Scholar] [CrossRef]
- Dennis, K.L.; Blatner, N.R.; Gounari, F.; Khazaie, K. Current Status of IL-10 and Regulatory T-Cells in Cancer. Curr. Opin. Oncol. 2013, 25, 637. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.M.; Lam, H.Y.P.; Hsu, H.J.; Jiang, S.J. Interleukin-10: A double-edged sword in breast cancer. Tzu Chi Med. J. 2021, 33, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Gornowicz, A.; Kałuża, Z.; Bielawska, A.; Gabryel-Porowska, H.; Czarnomysy, R.; Bielawski, K. Cytotoxic efficacy of a novel dinuclear platinum(II) complex used with anti-MUC1 in human breast cancer cells. Mol. Cell. Biochem. 2014, 392, 161–174. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lopes, C.M.; Dourado, A.; Oliveira, R. Phytotherapy and nutritional supplements on breast cancer. Biomed. Res. Int. 2017, 2017, 7207983. [Google Scholar] [CrossRef] [PubMed]
- Puskulluoglu, M.; Uchańska, B.; Tomaszewski, K.A.; Zygulska, A.L.; Zielińska, P.; Grela-Wojewoda, A. Use of complementary and alternative medicine among Polish cancer patients. Nowotwory. J. Oncol. 2021, 71, 274–281. [Google Scholar] [CrossRef]
- Drozdoff, L.; Klein, E.; Kiechle, M.; Paepke, D. Use of biologically-based complementary medicine in breast and gynecological cancer patients during systemic therapy. BMC Complement. Altern. Med. 2018, 18, 259. [Google Scholar] [CrossRef]
- Leggett, S.; Koczwara, B.; Miller, M. The impact of complementary and alternative medicines on cancer symptoms, treatment side effects, quality of life, and survival in women with breast cancer—A systematic review. Nutr. Cancer 2015, 67, 373–391. [Google Scholar] [CrossRef]
- McGrowder, D.A.; Miller, F.G.; Nwokocha, C.R.; Anderson, M.S.; Wilson-Clarke, C.; Vaz, K.; Anderson-Jackson, L.; Brown, J. medicinal herbs used in traditional management of breast cancer: Mechanisms of action. Medicines 2020, 7, 47. [Google Scholar] [CrossRef]
- Gharby, S.; Harhar, H.; Bouzoubaa, Z.; Asdadi, A.; El Yadini, A.; Charrouf, Z. Chemical characterization and oxidative stability of seeds and oil of sesame grown in Morocco. J. Saudi Soc. Agric. Sci. 2017, 16, 105–111. [Google Scholar] [CrossRef]
- Xu, H.; Li, Y.; Han, B.; Li, Z.; Wang, B.; Jiang, P.; Zhang, J.; Ma, W.; Zhou, D.; Li, X.; et al. Anti-breast-cancer activity exerted by β-sitosterol- d -glucoside from sweet potato via upregulation of microRNA-10a and via the PI3K-Akt signaling pathway. J. Agric. Food Chem. 2018, 66, 9704–9718. [Google Scholar] [CrossRef]
- Zhao, M.; Zhong, Q.; Tian, M.; Han, R.; Ren, Y. Comparative transcriptome analysis reveals differentially expressed genes associated with the development of Jerusalem artichoke tuber (Helianthus tuberosus L.). Ind. Crops Prod. 2020, 151, 112455. [Google Scholar] [CrossRef]
- Yilmaz, E.; Güneşer, B.A. Cold pressed versus solvent extracted lemon (Citrus limon L.) seed oils: Yield and properties. J. Food Sci. Technol. 2017, 54, 1891–1900. [Google Scholar] [CrossRef]
- Konopka, I.; Roszkowska, B.; Czaplicki, S.; Tańska, M. Optimization of pumpkin oil recovery by using aqueous enzymatic extraction and comparison of the quality of the obtained oil with the quality of cold-pressed oil. Food Technol. Biotechnol. 2016, 54, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Tantivejkul, K.; Vucenik, I.; Eiseman, J.; Shamsuddin, A.K.M. Inositol hexaphosphate (IP6) enhances the anti-proliferative effects of adriamycin and tamoxifen in breast cancer. Breast Cancer Res. Treat. 2003, 79, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Teng, Y.N.; Wang, C.C.N.; Liao, W.C.; Lan, Y.H.; Hung, C.C. caffeic acid attenuates multi-drug resistance in cancer cells by inhibiting efflux function of human P-glycoprotein. Molecules 2020, 25, 247. [Google Scholar] [CrossRef]
- Rosendahl, A.H.; Perks, C.M.; Zeng, L.; Markkula, A.; Simonsson, M.; Rose, C.; Ingvar, C.; Holly, J.M.P.; Jernström, H. Caffeine and caffeic acid inhibit growth and modify estrogen receptor and insulin-like growth factor I receptor levels in human breast cancer. Clin. Cancer Res. 2015, 21, 1877–1887. [Google Scholar] [CrossRef] [PubMed]
- Ghasemzadeh, A.; Talei, D.; Jaafar, H.Z.E.; Juraimi, A.S.; Mohamed, M.T.M.; Puteh, A.; Halim, M.R.A. Plant-growth regulators alter phytochemical constituents and pharmaceutical quality in Sweet potato (Ipomoea batatas L.). BMC Complement. Altern. Med. 2016, 16, 152. [Google Scholar] [CrossRef]
- Elansary, H.O.; Szopa, A.; Kubica, P.; Ekiert, H.; Al-Mana, F.A.; El-Shafei, A.A. Polyphenols of Frangula alnus and Peganum harmala leaves and associated biological activities. Plants 2020, 9, 1086. [Google Scholar] [CrossRef]
- Shendge, A.K.; Chaudhuri, D.; Basu, T.; Mandal, N. A natural flavonoid, apigenin isolated from Clerodendrum viscosum leaves, induces G2/M phase cell cycle arrest and apoptosis in MCF-7 cells through the regulation of p53 and caspase-cascade pathway. Clin. Transl. Oncol. 2021, 23, 718–730. [Google Scholar] [CrossRef]
- Hostanska, K.; Nisslein, T.; Freudenstein, J.; Reichling, J.; Saller, R. evaluation of cell death caused by triterpene glycosides and phenolic substances from Cimicifuga racemosa extract in human MCF-7 breast cancer cells. Biol. Pharm. Bull. 2004, 27, 1970–1975. [Google Scholar] [CrossRef]
- Whyte, J.; Bergin, O.; Bianchi, A.; McNally, S.; Martin, F. Key signalling nodes in mammary gland development and cancer. Mitogen-activated protein kinase signalling in experimental models of breast cancer progression and in mammary gland development. Breast Cancer Res. 2009, 11, 209. [Google Scholar] [CrossRef] [PubMed]
- Seo, J.H.; Moon, H.S.; Kim, I.Y.; Guo, D.D.; Lee, H.G.; Choi, Y.J.; Cho, C.S. PEGylated conjugated linoleic acid stimulation of apoptosis via a p53-mediated signaling pathway in MCF-7 breast cancer cells. Eur. J. Pharm. Biopharm. 2008, 70, 621–626. [Google Scholar] [CrossRef] [PubMed]
- Dan, H.C.; Cooper, M.J.; Cogswell, P.C.; Duncan, J.A.; Ting, J.P.Y.; Baldwin, A.S. Akt-dependent regulation of NF-κB is controlled by mTOR and Raptor in association with IKK. Genes Dev. 2008, 22, 1490–1500. [Google Scholar] [CrossRef] [PubMed]
- Kello, M.; Takac, P.; Kubatka, P.; Kuruc, T.; Petrova, K.; Mojzis, J. Oxidative stress-induced DNA damage and apoptosis in clove buds-treated MCF-7 cells. Biomolecules 2020, 10, 139. [Google Scholar] [CrossRef] [PubMed]
- Chun, J.; Han, L.; Xu, M.Y.; Wang, B.; Cheng, M.S.; Kim, Y.S. The induction of apoptosis by a newly synthesized diosgenyl saponin through the suppression of estrogen receptor-α in MCF-7 human breast cancer cells. Arch. Pharm. Res. 2014, 37, 1477–1486. [Google Scholar] [CrossRef] [PubMed]
- Luo, M.; Guan, J.L. Focal Adhesion Kinase: A prominent determinant in breast cancer initiation, progression and metastasis. Cancer Lett 2010, 289, 127–139. [Google Scholar] [CrossRef]
- Kello, M.; Kulikova, L.; Vaskova, J.; Nagyova, A.; Mojzis, J. Fruit Peel Polyphenolic Extract-Induced Apoptosis in Human Breast Cancer Cells Is Associated with ROS Production and Modulation of p38MAPK/Erk1/2 and the Akt Signaling Pathway. Nutr. Cancer 2017, 69, 920–931. [Google Scholar] [CrossRef]
- Yu, C.H.; Chu, S.C.; Yang, S.F.; Hsieh, Y.S.; Lee, C.Y.; Chen, P.N. Induction of apoptotic but not autophagic cell death by Cinnamomum cassia extracts on human oral cancer cells. J. Cell. Physiol. 2019, 234, 5289–5303. [Google Scholar] [CrossRef]
- Marie Hardwick, J.; Soane, L. Multiple functions of BCL-2 family proteins. Cold Spring Harb. Perspect. Biol. 2013, 5, a008722. [Google Scholar] [CrossRef]
- Edlich, F. BCL-2 proteins and apoptosis: Recent insights and unknowns. Biochem. Biophys. Res. Commun. 2018, 500, 26–34. [Google Scholar] [CrossRef]
- Irshad, M.; Jafar Mehdi, S.; Al-Fatlawi, A.A.; Zafaryab, M.; Ali, A.; Ahmad, I.; Singh, M.; Moshahid Rizvi, M.A. Phytochemical composition of Cassia fistula fruit extracts and its anticancer activity against human cancer cell lines. J. Biol. Act. Prod. Nat. 2014, 4, 158–170. [Google Scholar] [CrossRef]
- Ruwizhi, N.; Aderibigbe, B.A. Cinnamic acid derivatives and their biological efficacy. Int. J. Mol. Sci. 2020, 21, 5712. [Google Scholar] [CrossRef] [PubMed]
- Yousefi, S.; Perozzo, R.; Schmid, I.; Ziemiecki, A.; Schaffner, T.; Scapozza, L.; Brunner, T.; Simon, H.U. Calpain-mediated cleavage of Atg5 switches autophagy to apoptosis. Nat. Cell Biol. 2006, 8, 1124–1132. [Google Scholar] [CrossRef] [PubMed]
- Koukourakis, M.I.; Kalamida, D.; Giatromanolaki, A.; Zois, C.E.; Sivridis, E.; Pouliliou, S.; Mitrakas, A.; Gatter, K.C.; Harris, A.L. Autophagosome proteins LC3A, LC3B and LC3C have distinct subcellular distribution kinetics and expression in cancer cell lines. PLoS ONE 2015, 10, e0137675. [Google Scholar] [CrossRef] [PubMed]
- Weng, J.R.; Yen, M.H.; Lin, W.Y. Cytotoxic constituents from Celastrus paniculatus induce apoptosis and autophagy in breast cancer cells. Phytochemistry 2013, 94, 211–219. [Google Scholar] [CrossRef]
- Tokgun, O.; Tokgun, P.E.; Turel, S.; Inal, B.; Inci, K.; Tan, S.; Can Alvur, O. Bryonia multiflora extract induces autophagy via regulating long non-coding RNAs in breast cancer cells. Nutr. Cancer 2021, 73, 1792–1803. [Google Scholar] [CrossRef]
- Esquivel-Velázquez, M.; Ostoa-Saloma, P.; Palacios-Arreola, M.I.; Nava-Castro, K.E.; Castro, J.I.; Morales-Montor, J. The role of cytokines in breast cancer development and progression. J. Interf. Cytokine Res. 2015, 35, 1–16. [Google Scholar] [CrossRef]
- Santos, A.F.; Santos Mota, N.S.R.; Schiefer, E.M.; da Cunha, R.S.; Junkert, A.M.; Stinghen, A.E.M.; Pontarolo, R.; Crisma, A.R.; Weffort-Santos, A.M.; Pedrosa, R.C.; et al. The toxicity of Aspidosperma subincanum to MCF7 cells is related to modulation of oxidative status and proinflammatory pathways. J. Ethnopharmacol. 2021, 281, 114512. [Google Scholar] [CrossRef]
- Kim, S.J.; Pham, T.H.; Bak, Y.; Ryu, H.W.; Oh, S.R.; Yoon, D.Y. 7-Methoxy-luteolin-8-C-β-6-deoxy-xylo-pyranos-3-uloside exactly (mLU8C-PU) isolated from Arthraxon hispidus inhibits migratory and invasive responses mediated via downregulation of MMP-9 and IL-8 expression in MCF-7 breast cancer cells. Environ. Toxicol. 2018, 33, 1143–1152. [Google Scholar] [CrossRef]
- Bahadır Acikara, Ö.; Hošek, J.; Babula, P.; Cvačka, J.; Budešínský, M.; Dračinský, M.; Saltan İşcan, G.; Kadlecová, D.; Ballová, L.; Šmejkal, K. Turkish Scorzonera species extracts attenuate cytokine secretion via inhibition of NF-κB activation, showing anti-inflammatory effect in vitro. Molecules 2016, 21, 43. [Google Scholar] [CrossRef]
- Costantini, S.; Rusolo, F.; De Vito, V.; Moccia, S.; Picariello, G.; Capone, F.; Guerriero, E.; Castello, G.; Volpe, M.G. Potential anti-inflammatory effects of the hydrophilic fraction of pomegranate (Punica granatum L.) seed oil on breast cancer cell lines. Molecules 2014, 19, 8644–8660. [Google Scholar] [CrossRef] [PubMed]
- Nazaruk, J.; Jakoniuk, P. Flavonoid composition and antimicrobial activity of Cirsium rivulare (Jacq.) All. flowers. J. Ethnopharmacol. 2005, 102, 208–212. [Google Scholar] [CrossRef] [PubMed]
- Juszczak, A.M.; Czarnomysy, R.; Strawa, J.W.; Končić, M.Z.; Bielawski, K.; Tomczyk, M. In vitro anticancer potential of Jasione montana and its main components against human amelanotic melanoma cells. Int. J. Mol. Sci. 2021, 22, 3345. [Google Scholar] [CrossRef] [PubMed]
- Isidorov, V.A.; Smolewska, M.; Purzyåska-Pugacewicz, A.; Tyszkiewicz, Z. Chemical composition of volatile and extractive compounds of pine and spruce leaf litter in the initial stages of decomposition. Biogeosciences 2010, 7, 2785–2794. [Google Scholar] [CrossRef]
- National Institute of Standard and Technology. NIST Chemistry WebBook; National Institute of Standard and Technology: Gaithersburg, MD, USA, 2013. [Google Scholar]
- Isidorov, V.A.; Kotowska, U.; Vinogorova, V.T. GC identification of organic compounds based on partition coefficients of their TMS derivatives in a hexane-acetonitrile system and retention indices. Anal. Sci. 2005, 21, 1483–1489. [Google Scholar] [CrossRef]
- Isidorov, V.A.; Szczepaniak, L. Gas chromatographic retention indices of biologically and environmentally important organic compounds on capillary columns with low-polar stationary phases. J. Chromatogr. A 2009, 1216, 8998–9007. [Google Scholar] [CrossRef]
- Isidorov, V.A.; Stocki, M.; Vetchinikova, L. Inheritance of specific secondary volatile metabolites in buds of white birch Betula pendula and Betula pubescens hybrids. Trees Struct. Funct. 2019, 33, 1329–1344. [Google Scholar] [CrossRef]
- Babushok, V.I.; Linstrom, P.J.; Zenkevich, I.G. Retention indices for frequently reported compounds of plant essential oils. J. Phys. Chem. Ref. Data 2011, 40, 043101. [Google Scholar] [CrossRef]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectrometry, 4th ed.; Allured Publishing Corporation: Carol Streams, IL, USA, 2007; ISBN 1932633219. [Google Scholar]
- ICH. In Proceedings of the 6th International Conference on Harmonization. Geneva, Switzerland, 1 November 2005. [Google Scholar]
- Carmichael, J.; DeGraff, W.G.; Gazdgar, A.F.; Minna, J.D.; Mitchel, J.B. evaluation of a tetrazolium-based semiautomated colorimetric assay: Assessment of chemosensitivity testing. Cancer Res. 1987, 47, 936–942. [Google Scholar]
- Gornowicz, A.; Szymanowski, W.; Bielawski, K.; Kałuża, Z.; Michalak, O.; Bielawska, A. Mucin 1 as a molecular target of a novel diisoquinoline derivative combined with anti-MUC1 antibody in AGS gastric cancer cells. Molecules 2021, 26, 6504. [Google Scholar] [CrossRef]
- Gornowicz, A.; Szymanowski, W.; Czarnomysy, R.; Bielawski, K.; Bielawska, A. Anti-HER2 monoclonal antibodies intensify the susceptibility of human gastric cancer cells to etoposide by promoting apoptosis, but not autophagy. PLoS ONE 2021, 16, e0255585. [Google Scholar] [CrossRef] [PubMed]
- Buzun, K.; Gornowicz, A.; Lesyk, R.; Kryshchyshyn-Dylevych, A.; Gzella, A.; Czarnomysy, R.; Latacz, G.; Olejarz-Maciej, A.; Handzlik, J.; Bielawski, K.; et al. 2-{5-[(Z,2Z)-2-Chloro-3-(4-nitrophenyl)-2-propenylidene]-4-oxo-2-thioxothiazolidin-3-yl}-3-methylbutanoic acid as a potential anti-breast cancer molecule. Int. J. Mol. Sci. 2022, 23, 4091. [Google Scholar] [CrossRef] [PubMed]
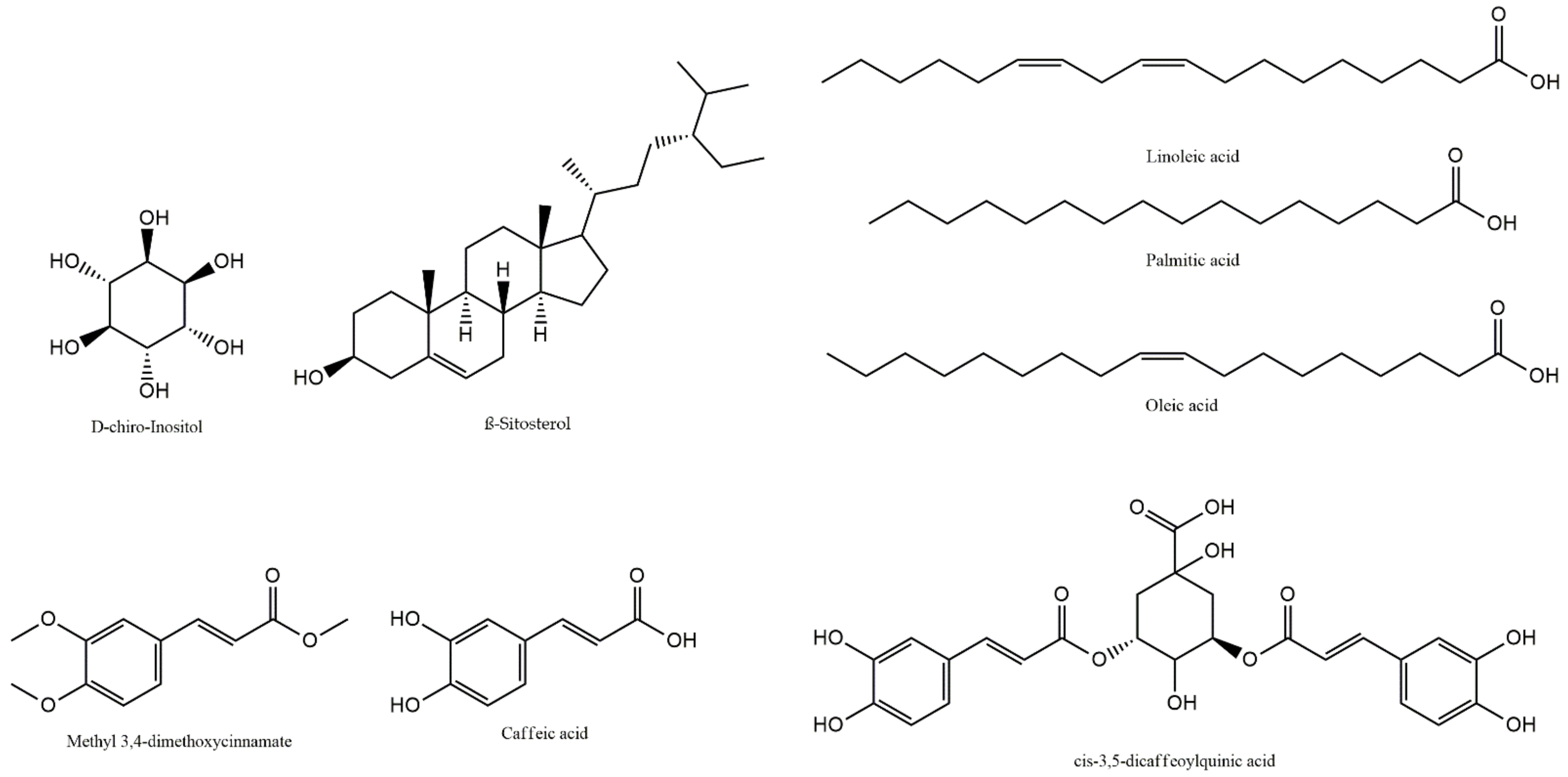


| Compounds | Analytical Parameters | Relative Composition, % | |||||||
|---|---|---|---|---|---|---|---|---|---|
| RIExp | RILit | Target Ions, m/z | M+ | SH1 | SH9 | SH10 | SH11 | SH12 | |
| Organic Acids | 11.2 | 41.7 | 53.0 | 41.0 | 62.2 | ||||
| Fatty acids | 7.5 | 41.7 | 44.2 | 33.0 | 61.8 | ||||
| Linoleic acid (LA, 18:2), mono-TMS | 2220 | 2215 | 73 (100), 75 (99), 337 (86), 67 (62), 81 (59) | 352 | 3.7 | 38.7 | 18.8 | 9.9 | 27.2 |
| Oleic acid (OA, 18:1), mono-TMS | 2225 | 2220 | 339 (100), 73 (93), 117 (92), 75 (86), 129 (76) | 354 | 1.4 | 9.0 | 6.5 | 16.0 | |
| Palmitic acid (PA, 16:0), mono-TMS | 2054 | 2052 | 313 (100), 117 (91), 73 (71), 75 (51), 132 (42) | 328 | 1.2 | 6.9 | 5.9 | 15.7 | |
| Conjugated linoleic acid (CLA, 18:2), mono-TMS | 73 (100), 75 (83), 117 (62), 129 (50), 105 (48) | 352 | 0.3 | 2.1 | 0.4 | 2.4 | |||
| Aliphatic monocarboxylic acids | 0.1 | 1.4 | 1.6 | 0.4 | |||||
| Hexanoic acid (6:0), mono-TMS | 1078 | 1071 | 75 (100), 173 (87), 73 (85), 117 (38), 131 (15) | 188 | 0.02 | 0.8 | 0.8 | 0.4 | |
| Nonanoic acid (9:0), mono-TMS | 1366 | 1358 | 73 (100), 75 (76), 215 (74), 117 (61), 129 (25) | 230 | 0.2 | 0.4 | |||
| Octanoic acid (8:0), mono-TMS | 1269 | 1262 | 73 (100), 75 (86), 201 (84), 117 (60), 129 (23) | 216 | 0.03 | 0.2 | 0.2 | ||
| Aliphatic dicarboxylic acid | 1.2 | 6.1 | 2.2 | ||||||
| Glutaric acid, di-TMS | 1388 | 1394 | 73 (100), 147 (85), 131 (41), 103 (23), 59 (16) | 276 | 0.3 | 4.1 | 0.4 | ||
| Azelaic acid, di-TMS | 1810 | 1812 | 73 (100), 75 (78), 317 (53), 201 (41), 129 (33) | 332 | 1.0 | 0.7 | |||
| Malic acid, tri-TMS | 1513 | 1510 | 73 (100), 147 (70), 233 (26), 245 (16), 133 (12) | 350 | 0.6 | ||||
| Sebacic acid, di-TMS | 1907 | 1905 | 73 (100), 75 (76), 331 (65), 215 (36), 129 (34) | 0.3 | 0.4 | ||||
| Aromatic acids | 0.03 | 0.3 | 0.7 | ||||||
| Benzoic acid, mono-TMS | 1247 | 1248 | 179 (100), 147 (81), 105 (69), 135 (48), 77 (46) | 194 | 0.03 | 0.1 | 0.2 | ||
| Phenylacetic acid, mono-TMS | 1300 | 1302 | 73 (100), 75 (32), 164 (19), 91 (17), 193 (16) | 208 | 0.2 | ||||
| Salicylic acid, di-TMS | 1518 | 1513 | 73 (100), 267 (46), 147 (29), 103 (24), 75 (19) | 0.2 | |||||
| Aliphatic hydroxy acids | 0.6 | 0.6 | 0.6 | ||||||
| L (+)-Lactic acid, di-TMS | 1073 | 1073 | 73 (100), 147 (77), 117 (62), 198 (18), 191 (15) | 0.1 | 0.2 | ||||
| β-Lactic acid, di-TMS | 1155 | 1145 | 147 (100), 73 (31), 177 (18), 119 (17), 148 (15) | 0.03 | 0.2 | ||||
| Glycolic acid, di-TMS | 1086 | 1085 | 147 (100), 73 (76), 148 (17), 66 (16), 133 (11) | 0.05 | 0.2 | ||||
| 2-Isopropyl-3-ketobutyrate, di-O-TMS | 1461 | 1463 | 73 (100), 273 (80), 147 (42), 155 (31), 183 (22) | 288 | 0.25 | ||||
| Glyceric acid, tri-TMS | 1351 | 1350 | 73 (100), 147 (69), 189 (39), 292 (31), 199 (27) | 0.18 | 0.17 | ||||
| Aromatic hydroxy acids | 1.8 | 0.2 | 2.0 | ||||||
| Quinic acid, penta-TMS | 1899 | 1902 | 73 (100), 345 (60), 147 (32), 75 (27), 255 (25) | 1.5 | 0.1 | ||||
| Shikimic acid, tetra-TMS | 1844 | 1845 | 252 (100), 73 (88), 204 (83), 131 (56), 103 (36) | 1.2 | |||||
| Hydroxybenzoic acid, di-TMS | 1623 | 1623 | 73 (100), 267 (42), 193 (33), 282 (32), 103 (13) | 282 | 0.02 | 0.5 | |||
| α,β-Unsaturated carboxylic acids | 0.1 | 0.1 | 0.9 | ||||||
| Cinnamic acid, mono-TMS | 1546 | 1549 | 205 (100), 131 (81), 103 (54), 161 (50), 73 (47) | 220 | 0.4 | ||||
| E-p-Coumaric acid, di-TMS | 1947 | 1947 | 73 (100), 293 (81), 219 (87), 308 (69), 249 (45) | 308 | 0.1 | 0.1 | 0.4 | ||
| Organic esters, carbonyl compounds | 0.5 | 20.1 | 3.8 | 27.9 | |||||
| Fatty acid esters | 0.3 | 18.8 | 2.7 | 4.3 | |||||
| Methyl linolelaidate | 2095 | 2095 | 73 (100), 67 (56), 81 (48), 95 (33), 55 (33) | 280 | 0.1 | 0.5 | 0.9 | ||
| 9-Octadecenoic acid, 18-TMS, methyl ester | 2434 | 2435 | 73 (100), 225 (55), 75 (42), 130 (27), 369 (19) | 384 | 0.2 | 0.7 | 0.3 | ||
| Butyl 9,12-octadecadienoate | 2470 | 2478 | 67 (100), 81 (84), 55 (62), 95 (58), 79 (56) | 336 | 8.8 | ||||
| Octadecadienoic acid (CLA), ester | 2474 | - | 55 (100), 67 (76), 81 (71), 69 (60), 95 (56) | 356 | 5.7 | ||||
| α,β-Unsaturated carboxylic esters | 0.04 | 22.8 | |||||||
| Cinnamic acid, 3,4-di-TMS, methyl ester | 2020 | 2018 | 219 (100), 238 (58),73 (41), 220 (17), 339 (16) | 338 | 0.04 | 21.16 | |||
| Cinnamic acid, methyl ester | 1856 | 1858 | 252 (100), 73 (76), 179 (71), 166 (64), 209 (64) | 1.19 | |||||
| Carbonyl compounds | 0.2 | 1.3 | 1.1 | 0.8 | |||||
| 2,4-Decadienal, (E, E)- | 1316 | 1315 | 81 (100), 41 (16), 67 (13), 83 (11), 55 (10) | 152 | 0.2 | 0.5 | 0.9 | ||
| Benzaldehyde, 3,5-dimethoxy-4-[(TMS)oxy]- | 1708 | 1711 | 224 (100), 239 (43), 223 (29), 254 (27), 73 (25) | 254 | 0.2 | ||||
| 2′,4′-Dihydroxyacetophenone, di-TMS, ether | 1703 | 1709 | 73 (100), 194 (94),70 (61), 281 (53), 44 (52) | 296 | 0.2 | ||||
| 2,4-Decadienal, (E, Z)- | 1292 | 1291 | 81 (100), 41 (23), 83 (18), 67 (18), 55 (15) | 152 | 0.3 | ||||
| 2-Decennial, E- | 1260 | 1262 | 70 (100), 55 (88), 41 (82), 43 (74), 83 (68) | 0.2 | |||||
| Organic alcohols, diols, polyols | 16.3 | 0.8 | 19.5 | 4.1 | 0.2 | ||||
| Organic alcohols | 0.3 | 0.8 | 0.1 | 4.1 | 0.2 | ||||
| 5-Allyl-1-methoxy-2,3-dihydroxybenzene, di-TMS, ether | 1953 | 1950 | 73 (100), 324 (91), 293 (56), 394 (39), 204 (38) | 324 | 0.1 | 3.7 | |||
| 3-Heptene, 4-ol, mono-TMS | 981 | 986 | 171 (100), 172 (17), 73 (10), 173 (8), 78 (7) | 186 | 0.2 | ||||
| 2-Phenylethanol, mono-TMS | 1229 | 1227 | 73 (100), 103 (42), 179 (40), 75 (35), 77 (21) | 194 | 0.1 | ||||
| Z,E-2,13-Octadecadien-1-ol | 2071 | 2076 | 99 (100), 67 (68), 55 (66), 79 (59), 81 (58) | 0.5 | |||||
| 3-Hexanol, mono-TMS | 998 | 994 | 75 (100), 159 (95), 73 (46), 103 (21), 77 (17) | 0.2 | |||||
| Diols | 0.1 | 1.6 | 0.3 | ||||||
| Ethylene glycol, di-TMS | 998 | 992 | 147 (100), 73 (41), 191 (15), 148 (15), 103 (13) | 0.05 | 0.9 | ||||
| Propylene glycol, di-TMS | 1014 | 1013 | 117 (100), 147 (66), 73 (64), 66 (11), 148 (10) | 0.02 | 0.5 | ||||
| 2,3-Butanediol, di-TMS, rac | 1049 | 1049 | 147 (100), 73 (97), 174 (24), 262 (18), 77 (17) | 0.3 | |||||
| Polyols | 15.8 | 17.8 | |||||||
| Glycerol, tri-TMS | 1296 | 1295 | 147 (100), 73 (95), 205 (83), 117 (40), 103 (32) | 2.1 | 15.7 | ||||
| Inositol, Hexa-OTMS, D-chiro- | 1999 | 1996 | 318 (100), 305 (85), 73 (75), 217 (71), 147 (53) | 612 | 7.2 | ||||
| D-(+)-Arabitol, Penta-TMS | 1759 | 1760 | 73 (100), 217 (92), 147 (56), 103 (48), 205 (43) | 2.0 | 0.3 | ||||
| Carbohydrates | 54.6 | 5.4 | 0.1 | ||||||
| Sucrose, octa-OTMS | 2712 | 2712 | 361 (100), 73 (56), 117 (40), 362 (33), 147 (21) | 18.0 | 2.9 | ||||
| Maltose, octa-TMS, methyloxime (isomer 2) | 2731 | 2733 | 73 (100), 361 (88), 217 (75), 289 (64), 147 (36) | 5.4 | |||||
| α-D-Fructofuranose, penta-TMS | 1845 | 1843 | 73 (100), 217 (89), 147 (34), 437 (26), 218 (17) | 3.8 | 0.3 | ||||
| β-D-Fructofuranose, penta-TMS | 1856 | 1854 | 217 (100), 73 (71), 437 (31), 147 (30), 218 (21) | 3.0 | |||||
| Phosphorous/Organophosphorous compounds | 1.6 | 10.8 | 3.8 | ||||||
| Phosphoric acid, tri-TMS | 1292 | 1285 | 299 (100), 300 (25), 73 (21), 314 (17), 301 (14) | 314 | 0.9 | 8.2 | 3.1 | ||
| Phosphoric acid, di-TMS monomethyl ester | 1192 | - | 241 (100), 242 (17), 133 (13), 73 (12), 211 (11) | 256 | 0.01 | 1.3 | 0.3 | ||
| Phosphoric acid, di-TMS, 2,3-di[(TMS)oxy]propyl ester | 1799 | 1793 | 73 (100), 357 (56), 299 (48), 147 (36), 129 (21) | 0.8 | 0.3 | ||||
| Phenols | 3.1 | 0.8 | 6.5 | ||||||
| E-Ferulic acid, di-TMS | 2102 | 2104 | 73 (100), 338 (89), 323 (48), 322 (42), 309 (39) | 338 | 0.1 | 0.2 | 4.2 | ||
| Caffeic acid, tris-TMS | 2158 | 2159 | 396 (100), 219 (94), 73 (65), 397 (36), 381 (24) | 396 | 2.7 | ||||
| Vanillin, mono-TMS | 1538 | 1545 | 194 (100), 193 (51), 209 (47), 224 (27), 73 (24) | 224 | 0.1 | 0.5 | |||
| Vanillic acid, di-TMS | 1777 | 1776 | 297 (100), 267 (71), 73 (68), 312 (56), 223 (54) | 312 | 0.04 | 0.1 | 0.3 | ||
| Sterols | 1.4 | 13.5 | 2.3 | 3.3 | 31.4 | ||||
| β-Sitosterol, mono-TMS | 3348 | 3342 | 129 (100), 357 (56),73 (58), 396 (54), 81 (47) | 486 | 0.7 | 8.0 | 1.9 | 1.3 | 21.9 |
| Stigmasterol, mono-TMS | 3290 | 3286 | 55 (100), 83 (91), 81 (78), 73 (75), 67 (67) | 484 | 0.2 | 1.1 | 0.5 | 0.7 | 2.6 |
| 5α-Stigmast-7-en-3β-ol | 3359 | 3355 | 414 (100), 255 (85), 55 (53), 81 (50), 43 (46) | 414 | 1.6 | ||||
| 5α-Stigmast-7-en-3β-ol, mono-TMS | 3401 | 3401 | 73 (100), 255 (92), 487 (79), 147 (45), 229 (20) | 486 | 4.8 | ||||
| Campesterol, mono-TMS | 3253 | 3251 | 73 (100), 129 (62), 343 (36), 147 (30), 382 (23) | 472 | 2.1 | ||||
| Amino acids | 3.0 | 0.6 | 0.3 | ||||||
| L-Proline, di- TMS | 1303 | 1302 | 142 (100), 73 (28), 143 (14), 147 (7), 216 (5) | 259 | 0.9 | 0.2 | |||
| Pyroglutamic acid, di-TMS | 1532 | 1524 | 156 (100), 73 (60), 147 (28), 157 (13), 217 (13) | 273 | 0.5 | 0.3 | |||
| Threonine, tri-TMS | 1406 | 1408 | 73 (100), 218 (60), 219 (54), 117 (41), 147 (30) | 0.3 | |||||
| Glycerolipids | 0.9 | 3.3 | 1.8 | 4.5 | |||||
| 2-Monolinolenin, di-TMS | 2776 | 2780 | 129 (100), 73 (97), 147 (60), 103 (48), 67 (41) | 498 | 0.5 | 1.1 | 2.1 | ||
| 2-Monoolein, di-TMS | 2742 | 2744 | 103 (100), 73 (84), 129 (79), 67 (43), 55 (35) | 0.2 | 0.5 | 1.5 | |||
| Glycerol 1-monolinolate | 2688 | 2697 | 67 (100), 81 (88), 55 (74), 95 (62), 79 (56) | 354 | 2.8 | ||||
| Tocopherols | 0.2 | 2.6 | 0.2 | 2.4 | |||||
| (+)-α-Tocopherol, OTMS- | 3152 | 3156 | 502 (100), 73 (80), 237 (68), 55 (51), 67 (41) | 502 | 0.2 | 0.2 | 2.4 | ||
| α-Tocopherol | 3130 | 3130 | 165 (100), 430 (86), 164 (31), 431 (28), 166 (12) | 430 | 2.3 | ||||
| α-Tocopheryl acetate | 3141 | 3132 | 165 (100), 55 (67), 430 (61), 67 (59), 81 (56) | 0.3 | |||||
| Nucleosides | 1.0 | 0.2 | |||||||
| Cytidine, 2′,3′,5′-tri-TMS ether | 2822 | 2811 | 73 (100), 217 (69), 147 (34), 147 (28), 151 (24) | 459 | 0.5 | ||||
| 5-Methyluridine, tri-TMS derivative | 2429 | 2428 | 73 (100), 217 (81), 75 (34), 55 (30), 67 (25) | 474 | 0.2 | 0.2 | |||
| Uridine, 2′,3′,5′-tri-OTMS | 2461 | 2469 | 73 (100), 217 (62), 103 (23), 259 (21), 147 (21) | 460 | 0.2 | ||||
| Terpenoids | 0.2 | 6.2 | 0.2 | ||||||
| Monoterpenes, Monoterpenoids | 0.6 | ||||||||
| p-Menthane, trans- | 984 | 978 | 97 (100), 55 (67), 41 (20), 96 (20), 57 (19) | 140 | 0.2 | ||||
| Carvone | 1241 | 1242 | 82 (100), 54 (38), 108 (36), 93 (36), 107 (25) | 150 | 0.2 | ||||
| Camphor | 1143 | 1143 | 95 (100), 81 (86), 67 (64), 152 (59), 55 (54) | 152 | 0.1 | ||||
| Triterpenes | 0.2 | 5.4 | 2.0 | ||||||
| α-Amyrin, mono-TMS | 3384 | 3382 | 218 (100), 73 (35), 189 (25), 190 (20), 219 (19) | 498 | 0.2 | 2.0 | |||
| α-Amyrin | 3376 | 3376 | 218 (100), 207 (22), 95 (22), 135 (22), 203 (22) | 426 | 4.2 | ||||
| β-Amyrin | 3330 | 3337 | 218 (100), 203 (48), 207 (26), 55 (24), 81 (23) | 426 | 1.2 | ||||
| Sesquiterpenes, Sesquiterpenoids | 0.2 | ||||||||
| α-Longipinene | 1363 | 1360 | 41 (100), 55 (96), 43 (87), 91 (78), 44 (71) | 204 | 0.1 | ||||
| β-E-Caryophyllene | 1415 | 1416 | 41 (100), 91 (99), 105 (93), 55 (91), 79 (89) | 204 | 0.1 | ||||
| Hydrocarbons | 4.8 | ||||||||
| Aliphatic hydrocarbons | 3.9 | ||||||||
| Alicyclic hydrocarbons | 0.6 | ||||||||
| Aromatic hydrocarbons | 0.3 | ||||||||
| Flavonoids | 0.4 | ||||||||
| Other compounds | 1.2 | 1.1 | 0.8 | 1.9 | 0.4 | ||||
| Non-identified compounds | 3.5 | 6.0 | 2.3 | 6.5 | 0.6 | ||||
| No | Retention Time [min] | UV λ max [nm] | [M-H]-[m/z] | Compound Name |
|---|---|---|---|---|
| 1 | 13.46 | 290 sh, 325 | 250, 300, 310 | unknown |
| 2 | 18.23 | 290 sh, 326 | 353 | 5-CQa S |
| 3 | 19.16 | 290 sh, 340 | 339 | 3-p-CoumQa |
| 4 | 21.61 | 250, 290 sh, 325 | 191, 353, 705 | 3-CQa S |
| 5 | 22.45 | 290 sh, 325 | 353 | 4-CQa S |
| 6 | 23.06 | 295 sh, 325 | 292 | unknown |
| 7 | 23.27 | 295 sh, 326 | 179 | CA S |
| 8 | 23.91 | 325 | 306 | unknown |
| 9 | 24.89 | 325 | 530 | unknown |
| 10 | 28.65 | 310 | 367 | methylated 4-CQa |
| 11 | 31.83 | 295 sh, 325 | 435 | unknown |
| 12 | 45.38 | 300 sh, 325 | 133, 161, 387, 549 | p-CoumQA derivatives |
| 13 | 48.73 | 295 sh, 325 | 147, 353, 515 | 3,5-dCQa S |
| 14 | 50.26 | 295 sh, 325 | 353, 515 | cis-3,5-dCQa |
| 15 | 52.39 | 265, 338 | 269, 445 | apigenin 7-O-glucuronide S |
| 16 | 53.47 | 295 sh, 325 | 353, 515 | 4,5-dCQa S |
| 17 | 57.16 | 295 sh, 325 | 507 | unknown |
| 18 | 57.47 | 295 sh, 320 | 529 | methylated-diCQa |
| 19 | 58.42 | 265 sh, 338 | 268, 459 | luteolin 7-O-glucuronide |
| 20 | 58.76 | 295 sh, 320 | 339, 529 | methylated-diCQa |
| 21 | 59.26 | 295 sh, 328 | 437 | unknown |
| 22 | 59.58 | 265 sh, 338 | 285 | luteolin S |
| 23 | 60.26 | 295 sh, 325 | 353, 515, 677 | triCQa |
| 24 | 63.17 | 265, 340 | 151, 269 | apigenin (A) S |
| No. | Content mg Per g of Extract/Fraction a | |||||||
|---|---|---|---|---|---|---|---|---|
| SH1 | SH2 | SH3 | SH4 | SH5 | SH6 | SH7 | SH8 | |
| 2 | nd | blq | nd | nd | nd | blq | blq | blq |
| 4 | 3.80 ± 0.03 | 12.37 ± 0.18 | blq | blq | nd | 54.34 ± 0.13 | 6.23 ± 0.06 | 13.39 ± 0.18 |
| 5 | nd | nd | nd | nd | 2.90 ± 0.10 | 2.25 ± 0.03 | blq | nd |
| 7 | nd | nd | 1.16 ± 0.03 | 20.97 ± 0.07 | nd | nd | nd | nd |
| 10 | nd | nd | nd | 4.87 ± 0.07 | nd | nd | nd | nd |
| 14 | 7.75 ± 0.04 | 13.57 ± 0.19 | blq | 36.54 ± 0.2 | 242.00 ± 0.20 | 13.54 ± 0.13 | nd | 20.54 ± 0.33 |
| 15 | nd | nd | nd | 5.55 ± 0.7 | nd | nd | nd | nd |
| 16 | 3.96 ± 0.17 | 8.48 ± 0.09 | nd | nd | nd | 48.18 ± 0.41 | nd | 10.36 ± 0.12 |
| 17 | blq | blq | nd | 16.82 ± 0.49 | 55.6 ± 0.20 | 10.31 ± 0.13 | nd | blq |
| 19 | nd | nd | nd | 1.60 ± 0.04 | nd | nd | nd | nd |
| 21 | nd | nd | nd | blq | nd | nd | nd | nd |
| 24 | blq | blq | nd | 13.98 ± 0.37 | 6.00 ± 0.10 | nd | nd | 0.80 ± 0.29 |
| 25 | 1.98 ± 0.09 | 1.10 ± 0.15 | blq | 5.90 ± 0.11 | nd | nd | nd | blq |
| Total CQa | 11.55 | 25.95 | 1.16 | 100.33 | 306.5 | 80.44 | 6.23 | 34.73 |
| Sample Name | IC50 for MCF-7 [μg/mL] | IC50 for MDA-MB-231 [μg/mL] |
|---|---|---|
| SH1 | 943.23 ± 55.5 | 863.21 ± 35.81 |
| SH4 | 630.52 ± 64.96 | 648.61 ± 182.62 |
| SH11 | 293.64 ± 16.61 | 265.05 ± 25.44 |
| Parameter | 5CQa | A |
|---|---|---|
| Linear Range [µg/mL] | 0.5–100 | 2.5–100 |
| r2 (n = 6) | 0.9998 | 0.9995 |
| Regression Equation a | y = 19.692x + 10.238 | y = 11.494x + 7.493 |
| LOD [µg/mL] | 0.64 | 0.91 |
| LOQ [µg/mL] | 1.92 | 2.76 |
| Accuracy [%] | 101.45 ± 4.47 | 101.77 ± 6.59 |
| Intraday precision (%CV) (N = 6) | 1.28 | 0.97 |
| Interday precision (%CV) (N = 9) | 1.82 | 0.87 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lendzion, K.; Gornowicz, A.; Strawa, J.W.; Bielawska, K.; Czarnomysy, R.; Popławska, B.; Bielawski, K.; Tomczyk, M.; Miltyk, W.; Bielawska, A. LC-PDA-MS and GC-MS Analysis of Scorzonera hispanica Seeds and Their Effects on Human Breast Cancer Cell Lines. Int. J. Mol. Sci. 2022, 23, 11584. https://doi.org/10.3390/ijms231911584
Lendzion K, Gornowicz A, Strawa JW, Bielawska K, Czarnomysy R, Popławska B, Bielawski K, Tomczyk M, Miltyk W, Bielawska A. LC-PDA-MS and GC-MS Analysis of Scorzonera hispanica Seeds and Their Effects on Human Breast Cancer Cell Lines. International Journal of Molecular Sciences. 2022; 23(19):11584. https://doi.org/10.3390/ijms231911584
Chicago/Turabian StyleLendzion, Karolina, Agnieszka Gornowicz, Jakub W. Strawa, Katarzyna Bielawska, Robert Czarnomysy, Bożena Popławska, Krzysztof Bielawski, Michał Tomczyk, Wojciech Miltyk, and Anna Bielawska. 2022. "LC-PDA-MS and GC-MS Analysis of Scorzonera hispanica Seeds and Their Effects on Human Breast Cancer Cell Lines" International Journal of Molecular Sciences 23, no. 19: 11584. https://doi.org/10.3390/ijms231911584
APA StyleLendzion, K., Gornowicz, A., Strawa, J. W., Bielawska, K., Czarnomysy, R., Popławska, B., Bielawski, K., Tomczyk, M., Miltyk, W., & Bielawska, A. (2022). LC-PDA-MS and GC-MS Analysis of Scorzonera hispanica Seeds and Their Effects on Human Breast Cancer Cell Lines. International Journal of Molecular Sciences, 23(19), 11584. https://doi.org/10.3390/ijms231911584








