Overexpression of Sweet Potato Carotenoid Cleavage Dioxygenase 4 (IbCCD4) Decreased Salt Tolerance in Arabidopsis thaliana
Abstract
:1. Introduction
2. Results
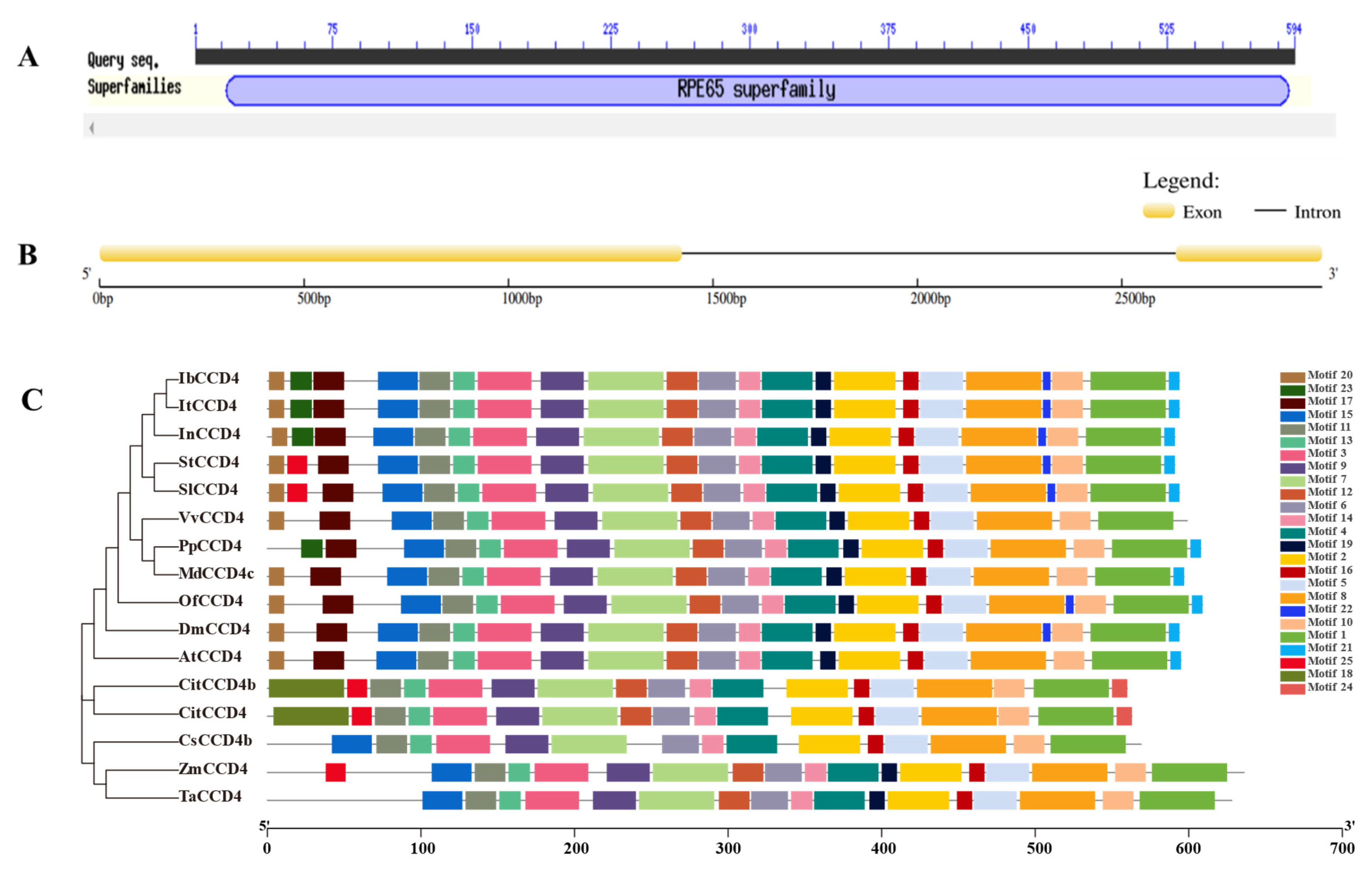
2.1. Bioinformatics Analysis of IbCCD4
2.2. IbCCD4 Response to Salt and Dehydration Stress
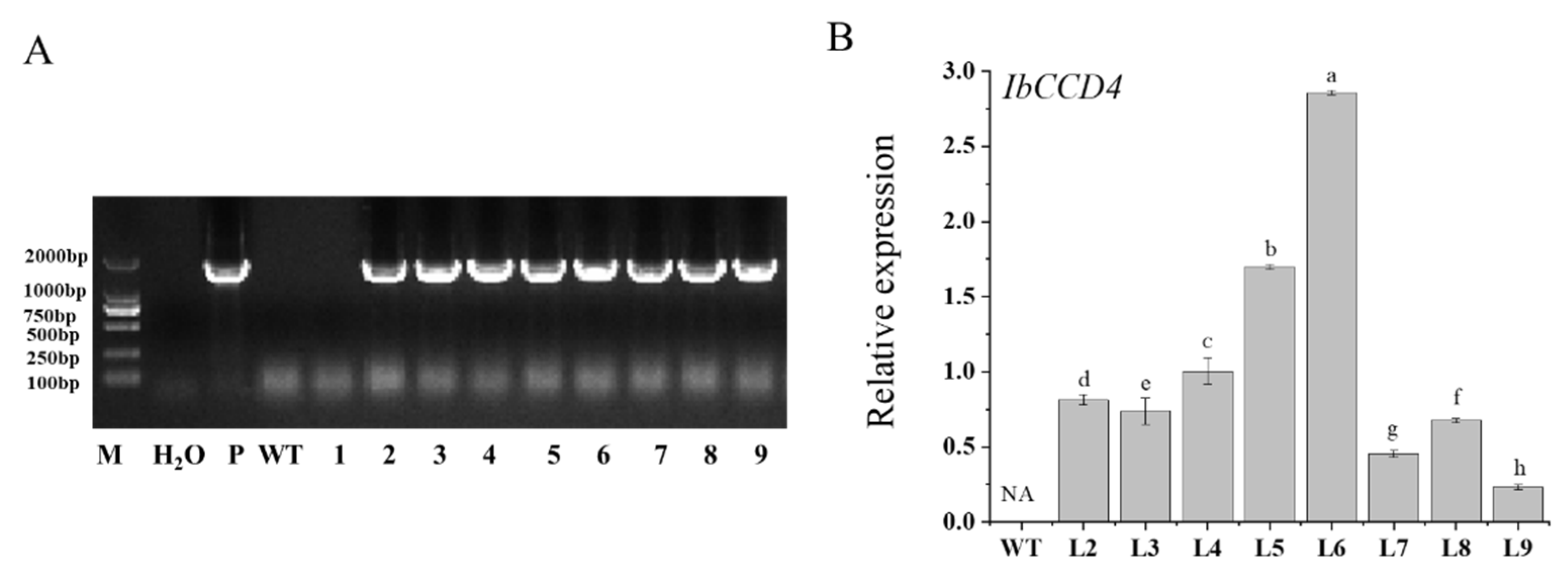
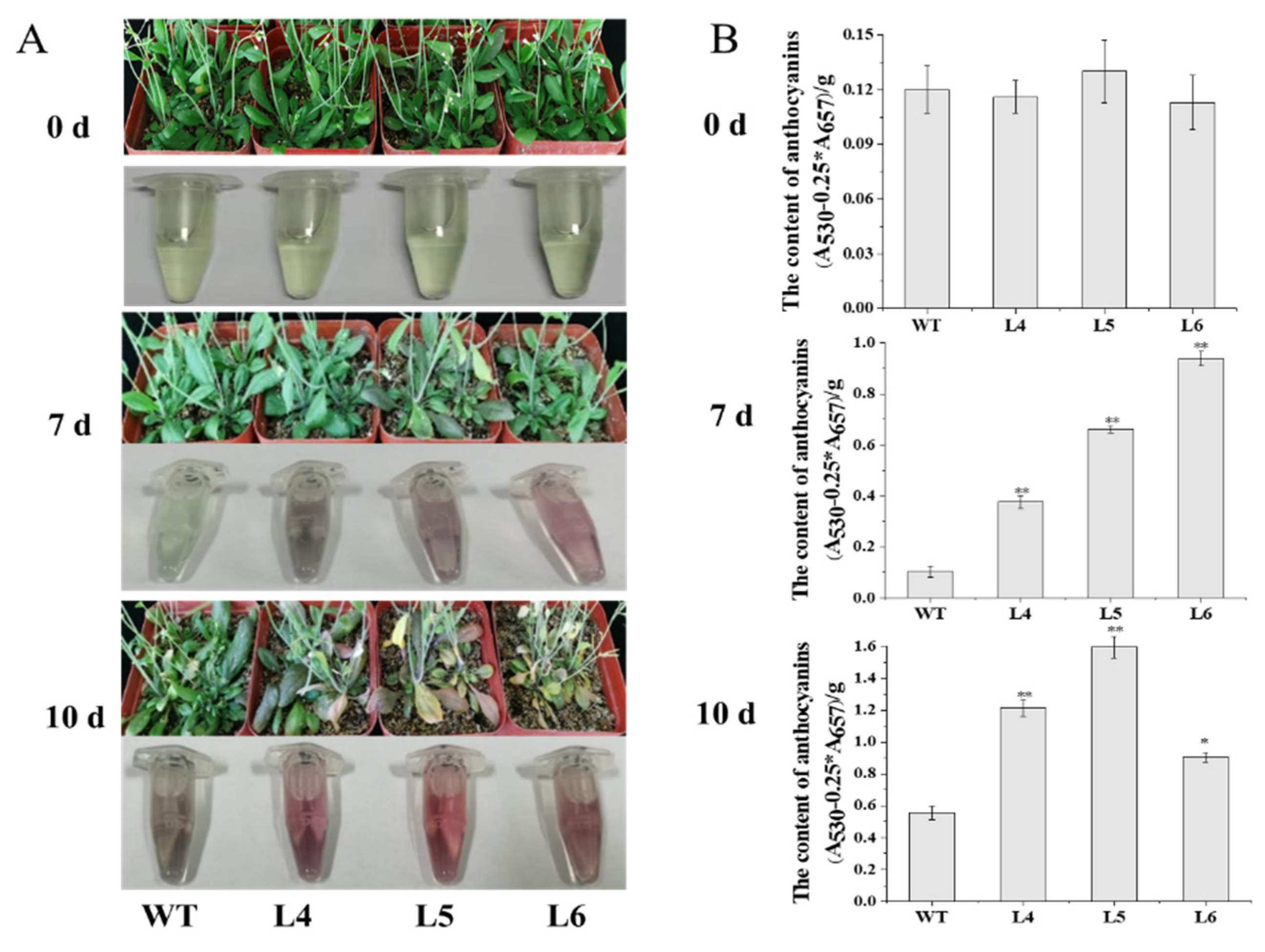
2.3. The Ectopic Expression of IbCCD4 Reduced Salt Tolerance in Transgenic Arabidopsis
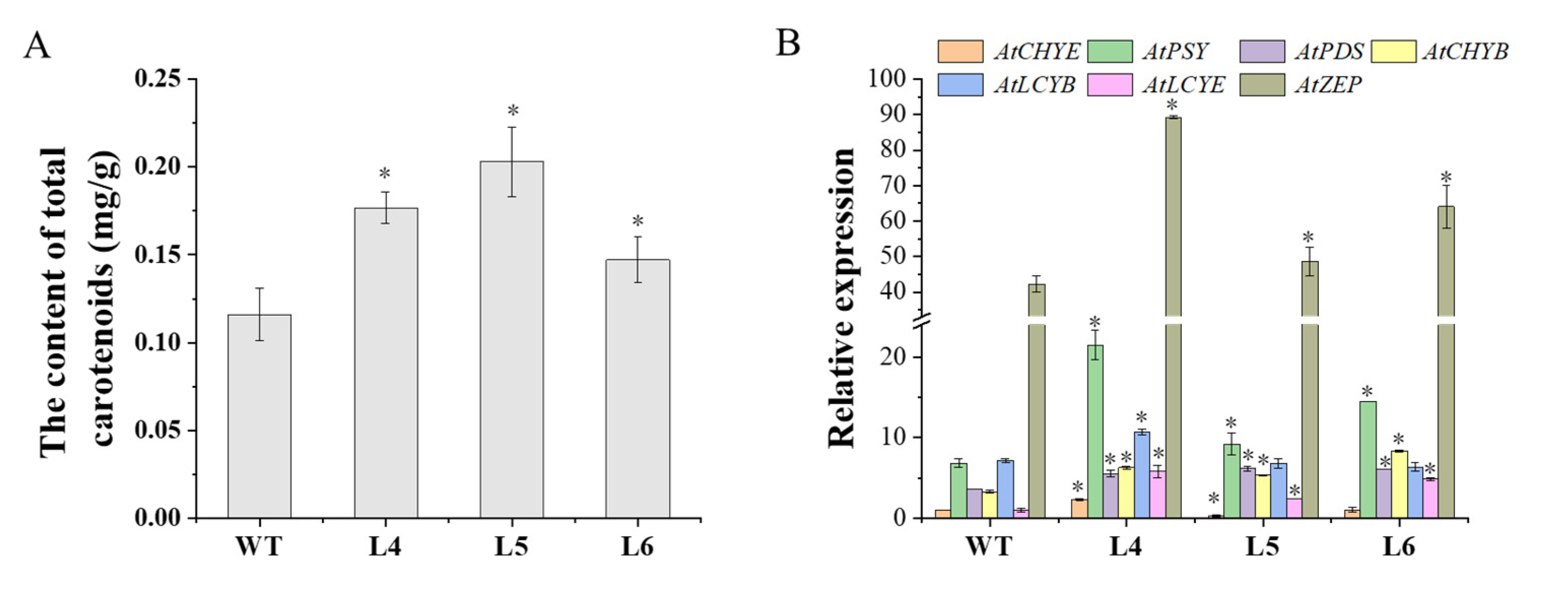
2.4. The Ectopic Expression of IbCCD4 Increased the Total Carotenoid Content and the Expression Levels of Most Carotenoid Biosynthesis Genes in Transgenic Arabidopsis
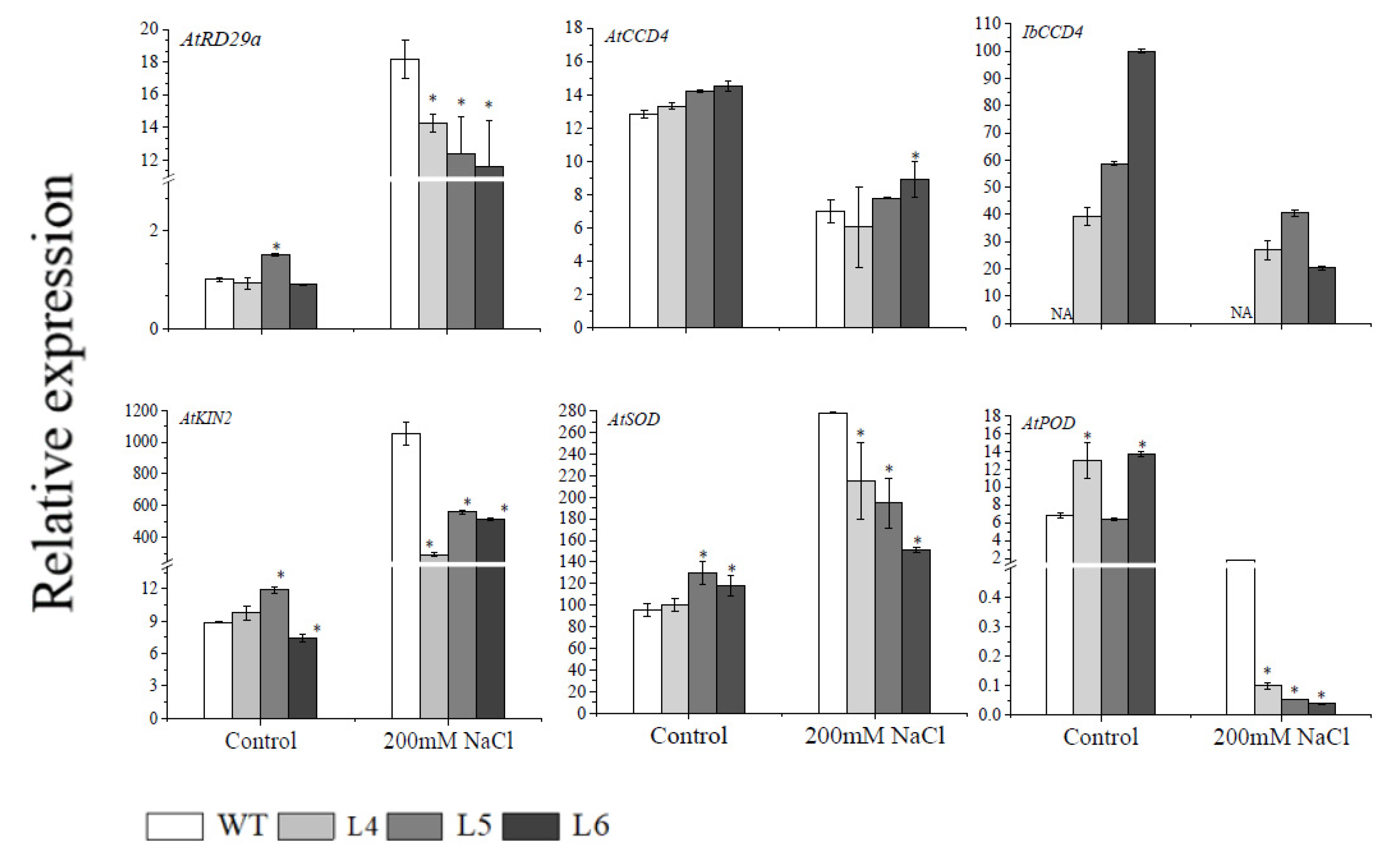
2.5. The Ectopic Expression of IbCCD4 Decreased the Expression of Resistance-Related Genes in Transgenic Arabidopsis under Salt Stress
2.6. The Ectopic Expression of IbCCD4 Decreased Antioxidant Enzyme Activity in Transgenic Arabidopsis under Salt Stress
3. Discussion
4. Materials and Methods
4.1. Plant Materials
4.2. IbCCD4 Multiple Sequence Alignment, Phylogenetic Tree Construction, and Motif Analysis
4.3. Salt and Dehydration Stress Treatment
4.4. Total RNA Extraction, cDNA Synthesis, and Expression Analysis by qRT-PCR
4.5. Cloning of the IbCCD4 Genomic Sequence and Gene Structure Analysis
4.6. Generation of Transgenic Arabidopsis
4.7. Salt Stress Assay of IbCCD4-Overexpressing Arabidopsis
4.8. Statistical Analysis of Data
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Munns, R.; Tester, M. Mechanisms of salinity tolerance. Annu. Rev. Plant Biol. 2008, 59, 651–681. [Google Scholar] [CrossRef] [PubMed]
- Shahid, S.A.; Zaman, M.; Heng, L. Soil salinity: Historical perspectives and a world overview of the problem. In Guideline for Salinity Assessment, Mitigation and Adaptation Using Nuclear And Related Techniques; Springer: Cham, Switzerland, 2018; pp. 43–53. [Google Scholar]
- Ivuskin, K.; Bartholomeus, H.; Bregt, A.K.; Pulatov, A.; Kempen, B.; de Sousa, L. Global mapping of soil salinity change. Remote Sens. Envi. 2019, 231, 111260. [Google Scholar] [CrossRef]
- Li, P.; Wu, J.; Qian, H. Regulation of secondary soil salinization in semi-arid regions: A simulation research in the Nanshantaizi area along the silk road, Northwest China. Environ. Earth Sci. 2016, 75, 698. [Google Scholar] [CrossRef]
- Abbas, A.; Khan, S.; Hussain, N.; Hanjra, M.A.; Akbar, S. Characterizing soil salinity in irrigated agriculture using a remote sensing approach. Phys. Chem. Earth 2011, 55, 43–52. [Google Scholar] [CrossRef]
- Ahrazem, O.; Diretto, G.; Argandona, J.; Rubio-Moraga, A.; Julve, J.M.; Orzaez, D.; Granell, A.; Gomez-Gomez, L. Evolutionarily distinct carotenoid cleavage dioxygenases are responsible for crocetin production in Buddleja davidii. J. Exp. Bot. 2017, 68, 4663–4677. [Google Scholar] [CrossRef]
- De, L.; Alvarez-Parrilla, E.; Gonzlez-Aguilar, G.A. chemistry, stability, and biological actions of carotenoids. In Fruit and Vegetable Phytochemicals; Wiley-Blackwell: Ames, IA, USA, 2009; pp. 177–222. [Google Scholar]
- Qi, Z.; Fan, X.; Zhu, C.; Chang, D.; Pei, J.; Zhao, L. Overexpression and characterization of a novel plant carotenoid cleavage dioxygenase 1 from Morus notabilis. Chem. Biodivers. 2022, 19, e202100735. [Google Scholar] [CrossRef]
- Liang, Y.S.; Jeon, Y.-A.; Lim, S.-H.; Kim, J.K.; Lee, J.-Y.; Kim, Y.-M.; Lee, Y.-H.; Ha, S.-H. Vascular-specific activity of the Arabidopsis carotenoid cleavage dioxygenase 7 gene promoter. Plant Cell Rep. 2011, 30, 973–980. [Google Scholar] [CrossRef]
- Ahrazem, O.; Gomez-Gomez, L.; Rodrigo, M.J.; Avalos, J.; Limon, M.C. Carotenoid Cleavage Oxygenases from Microbes and Photosynthetic Organisms: Features and Functions. Int. J. Mol. Sci. 2016, 17, 1781. [Google Scholar] [CrossRef]
- Ohmiya, A. Carotenoid cleavage dioxygenases and their apocarotenoid products in plants. Plant Biotechnol. 2009, 26, 351–358. [Google Scholar] [CrossRef]
- Langi, P.; Kiokias, S.; Varzakas, T.; Proestos, C. Carotenoids: From plants to food and feed industries. Methods Mol. Biol. 2018, 1852, 57–71. [Google Scholar] [CrossRef]
- Kim, S.H.; Ahn, Y.O.; Ahn, M.J.; Lee, H.S.; Kwak, S.S. Down-regulation of beta-carotene hydroxylase increases beta-carotene and total carotenoids enhancing salt stress tolerance in transgenic cultured cells of sweetpotato. Phytochemistry 2012, 74, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Jeong, J.C.; Park, S.; Bae, J.Y.; Ahn, M.J.; Lee, H.S.; Kwak, S.S. Down-regulation of sweetpotato lycopene beta-cyclase gene enhances tolerance to abiotic stress in transgenic calli. Mol. Biol. Rep. 2014, 41, 8137–8148. [Google Scholar] [CrossRef]
- Messing, S.A.; Gabelli, S.B.; Echeverria, I.; Vogel, J.T.; Guan, J.C.; Tan, B.C.; Klee, H.J.; McCarty, D.R.; Amzel, L.M. Structural insights into maize viviparous14, a key enzyme in the biosynthesis of the phytohormone abscisic acid. Plant Cell 2010, 22, 2970–2980. [Google Scholar] [CrossRef] [PubMed]
- Tan, B.C.; Joseph, L.M.; Deng, W.T.; Liu, L.; Li, Q.B.; Cline, K.; McCarty, D.R. Molecular characterization of the Arabidopsis 9-cis epoxycarotenoid dioxygenase gene family. Plant J. 2003, 35, 44–56. [Google Scholar] [CrossRef]
- Simkin, A.J.; Schwartz, S.H.; Auldridge, M.; Taylor, M.G.; Klee, H.J. The tomato carotenoid cleavage dioxygenase 1 genes contribute to the formation of the flavor volatiles beta-ionone, pseudoionone, and geranylacetone. Plant J. 2004, 40, 882–892. [Google Scholar] [CrossRef]
- Simkin, A.J.; Underwood, B.A.; Auldridge, M.; Loucas, H.M.; Shibuya, K.; Schmelz, E.; Clark, D.G.; Klee, H.J. Circadian regulation of the PhCCD1 carotenoid cleavage dioxygenase controls emission of beta-ionone, a fragrance volatile of petunia flowers. Plant Physiol. 2004, 136, 3504–3514. [Google Scholar] [CrossRef]
- Sun, Z.; Hans, J.; Walter, M.H.; Matusova, R.; Beekwilder, J.; Verstappen, F.W.; Ming, Z.; van Echtelt, E.; Strack, D.; Bisseling, T.; et al. Cloning and characterisation of a maize carotenoid cleavage dioxygenase (ZmCCD1) and its involvement in the biosynthesis of apocarotenoids with various roles in mutualistic and parasitic interactions. Planta 2008, 228, 789–801. [Google Scholar] [CrossRef]
- Zheng, X.; Yang, Y.; Al-Babili, S. Exploring the diversity and regulation of apocarotenoid metabolic pathways in plants. Front. Plant Sci. 2021, 12, 787049. [Google Scholar] [CrossRef]
- Nguyen, T.L.H.; Masuda, J.-I.; Miyajima, I.; Nguyen, Q.T.; Narges, M.; Hiramatsu, M.; Kim, J.H.; Okubo, H. Involvement of carotenoid cleavage dioxygenase 4 gene in 17 tepal color change in Lilium brownii var. colchesteri. J. Jpn. Soc. Hort. Sci. 2012, 81, 366–373. [Google Scholar] [CrossRef]
- Ohmiya, A.; Kishimoto, S.; Aida, R.; Yoshioka, S.; Sumitomo, K. Carotenoid cleavage dioxygenase (CmCCD4a) contributes to white color formation in chrysanthemum petals. Plant Physiol. 2006, 142, 1193–1201. [Google Scholar] [CrossRef] [Green Version]
- Li, T.; Deng, Y.J.; Liu, J.X.; Duan, A.Q.; Liu, H.; Xiong, A.S. DcCCD4 catalyzes the degradation of alpha-carotene and beta-carotene to affect carotenoid accumulation and taproot color in carrot. Plant J. 2021, 108, 1116–1130. [Google Scholar] [CrossRef] [PubMed]
- Adami, M.; De Franceschi, P.; Brandi, F.; Liverani, A.; Giovannini, D.; Rosati, C.; Dondini, L.; Tartarini, S. Identifying a carotenoid cleavage dioxygenase (ccd4) gene controlling yellow/white fruit flesh color of peach. Plant Mol. Biol. Rep. 2013, 31, 1166–1175. [Google Scholar] [CrossRef]
- Bruno, M.; Beyer, P.; Al-Babili, S. The potato carotenoid cleavage dioxygenase 4 catalyzes a single cleavage of beta-ionone ring-containing carotenes and non-epoxidated xanthophylls. Arch. Biochem. Biophys. 2015, 572, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Rameau, C. Strigolactones, a novel class of plant hormone controlling shoot branching. Comptes Rendus Biol. 2010, 333, 344–349. [Google Scholar] [CrossRef] [PubMed]
- Decker, E.L.; Alder, A.; Hunn, S.; Ferguson, J.; Lehtonen, M.T.; Scheler, B.; Kerres, K.L.; Wiedemann, G.; Safavi-Rizi, V.; Nordzieke, S.; et al. Strigolactone biosynthesis is evolutionarily conserved, regulated by phosphate starvation and contributes to resistance against phytopathogenic fungi in a moss, Physcomitrella patens. New Phytol. 2017, 216, 455–468. [Google Scholar] [CrossRef]
- Wei, H.; Movahedi, A.; Liu, G.; Li, Y.; Liu, S.; Yu, C.; Chen, Y.; Zhong, F.; Zhang, J. Comprehensive analysis of carotenoid cleavage dioxygenases gene family and its expression in response to abiotic stress in poplar. Int. J. Mol. Sci. 2022, 23, 1418. [Google Scholar] [CrossRef]
- Chen, H.; Zuo, X.; Shao, H.; Fan, S.; Ma, J.; Zhang, D.; Zhao, C.; Yan, X.; Liu, X.; Han, M. Genome-wide analysis of carotenoid cleavage oxygenase genes and their responses to various phytohormones and abiotic stresses in apple (Malus domestica) Plant Physiol. Biochem. 2018, 123, 81–93. [Google Scholar] [CrossRef]
- Torres-Vera, R.; Garcia, J.M.; Pozo, M.J.; Lopez-Raez, J.A. Do strigolactones contribute to plant defence? Mol. Plant Pathol. 2014, 15, 211–216. [Google Scholar] [CrossRef]
- Kim, Y.; Hwang, I.; Jung, H.-J.; Park, J.-I.; Kang, J.-G.; Nou, I.-S. Genome-wide classification and abiotic stress-responsive expression profiling of carotenoid oxygenase genes in Brassica rapa and Brassica oleracea. J. Plant Growth Regul. 2015, 35, 202–214. [Google Scholar] [CrossRef]
- Baba, S.A.; Jain, D.; Abbas, N.; Ashraf, N. Overexpression of Crocus carotenoid cleavage dioxygenase, CsCCD4b, in Arabidopsis imparts tolerance to dehydration, salt and oxidative stresses by modulating ROS machinery. J. Plant Physiol. 2015, 189, 114–125. [Google Scholar] [CrossRef]
- Tanaka, M.; Ishiguro, K.; Oki, T.; Okuno, S. Functional components in sweetpotato and their genetic improvement. Breed. Sci. 2017, 67, 52–61. [Google Scholar] [CrossRef] [PubMed]
- Fraser, P.D.; Bramley, P.M. The biosynthesis and nutritional uses of carotenoids. Prog. Lipid Res. 2004, 43, 228–265. [Google Scholar] [CrossRef] [PubMed]
- Eggersdorfer, M.; Wyss, A. Carotenoids in human nutrition and health. Arch. Biochem. Biophys. 2018, 652, 18–26. [Google Scholar] [CrossRef]
- Bovell-Benjamin, C.A. Sweet Potato: A review of its past, present, and future role in human nutrition. Adv. Food Nutr. Res. 2007, 52, 1–59. [Google Scholar] [CrossRef]
- Low, J.W.; Mwanga, R.O.M.; Andrade, M.; Carey, E.; Ball, A.M. Tackling vitamin A deficiency with biofortified sweetpotato in sub-Saharan Africa. Glob. Food Sec. 2017, 14, 23–30. [Google Scholar] [CrossRef]
- Cervantes-Paz, B.; Victoria-Campos, C.I.; Ornelas-Paz, J.d.J. Absorption of carotenoids and mechanisms involved in their health-related properties. Subcell Biochem. 2016, 79, 415–454. [Google Scholar] [CrossRef]
- Majeed, A.; Muhammad, Z. Salinity: A major agricultural problem—Causes, impacts on crop productivity and management strategies. In Plant Abiotic Stress Tolerance; Springer: Cham, Switzerland, 2019; pp. 83–99. [Google Scholar]
- Yang, Z.; Dong, T.; Dai, X.; Wei, Y.; Fang, Y.; Zhang, L.; Zhu, M.; Nawaz, G.; Cao, Q.; Xu, T. Comparative analysis of salt responsive microRNAs in two sweetpotato [Ipomoea batatas (L.) Lam.] cultivars with different salt stress resistance. Front. Plant Sci. 2022, 13, 879819. [Google Scholar] [CrossRef]
- Liu, G.L.; Zheng, J.L.; Fan, W.J.; Duan, X.G.; Liu, X.M.; Peng, Z. Salinity tolerance of different sweetpotato varieties on saline-alkali soils of the yellow river delta. Plant Physiol. J. 2011, 47, 777–784. [Google Scholar]
- Wang, L.J.; He, S.Z.; Zhai, H.; Liu, D.G.; Wang, Y.N.; Liu, Q.C. Molecular cloning and functional characterization of a salt tolerance-associated gene ibnfu1 from sweetpotato. J. Integr. Agric. 2013, 12, 27–35. [Google Scholar] [CrossRef]
- Yan, M.; Nie, H.; Wang, Y.; Wang, X.; Jarret, R.; Zhao, J.; Wang, H.; Yang, J. Exploring and exploiting genetics and genomics for sweetpotato improvement: Status and perspectives. Plant Commun. 2022, 5, 100332. [Google Scholar] [CrossRef]
- Liu, Y.; Dong, B.; Zhang, C.; Yang, L.; Wang, Y.; Zhao, H. Effects of exogenous abscisic acid (ABA) on carotenoids and petal color in Osmanthus fragrans ‘Yanhonggui’. Plants 2020, 9, 454. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Norman, J.M.; Frederick, R.L.; Sieburth, L.E. BYPASS1 negatively regulates a root-derived signal that controls plant architecture. Curr. Biol. 2004, 14, 1739–1746. [Google Scholar] [CrossRef] [PubMed]
- Van Norman, J.M.; Zhang, J.; Cazzonelli, C.I.; Pogson, B.J.; Harrison, P.J.; Bugg, T.D.; Chan, K.X.; Thompson, A.J.; Benfey, P.N. Periodic root branching in Arabidopsis requires synthesis of an uncharacterized carotenoid derivative. Proc. Natl. Acad. Sci. USA 2014, 111, E1300–E1309. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Tan, H.; Ma, Z.; Huang, J. DELLA proteins promote anthocyanin biosynthesis via sequestering MYBL2 and JAZ suppressors of the MYB/bHLH/WD40 complex in Arabidopsis thaliana. Mol. Plant 2016, 9, 711–721. [Google Scholar] [CrossRef]
- Jiang, C.; Gao, X.; Liao, L.; Harberd, N.P.; Fu, X. Phosphate starvation root architecture and anthocyanin accumulation responses are modulated by the gibberellin-DELLA signaling pathway in Arabidopsis. Plant Physiol. 2007, 145, 1460–1470. [Google Scholar] [CrossRef]
- Richard, A.D.; Nancy, L.P. Stress-induced phenylpropanoid metabolism. Plant Cell 1995, 7, 1085–1997. [Google Scholar] [CrossRef]
- Clotault, J.; Peltier, D.; Berruyer, R.; Thomas, M.; Briard, M.; Geoffriau, E. Expression of carotenoid biosynthesis genes during carrot root development. J. Exp. Bot. 2008, 59, 3563–3573. [Google Scholar] [CrossRef]
- Nisar, N.; Li, L.; Lu, S.; Khin, N.C.; Pogson, B. Carotenoid metabolism in plants. Mol. Plant 2015, 8, 68–82. [Google Scholar] [CrossRef]
- Xi, W.; Zhang, L.; Liu, S.; Zhao, G. The genes of CYP, ZEP, and CCD1/4 play an important role in controlling carotenoid and aroma volatile apocarotenoid accumulation of Apricot fruit. Front. Plant Sci. 2020, 11, 607715. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, N.; Zhao, M.; Jing, T.; Jin, J.; Wu, B.; Wan, X.; Schwab, W.; Song, C. Carotenoid cleavage dioxygenase 4 catalyzes the formation of carotenoid-derived volatile beta-ionone during tea (Camellia sinensis) Withering. J. Agric. Food Chem. 2020, 68, 1684–1690. [Google Scholar] [CrossRef]
- Ma, G.; Zhang, L.; Matsuta, A.; Matsutani, K.; Yamawaki, K.; Yahata, M.; Wahyudi, A.; Motohashi, R.; Kato, M. Enzymatic formation of beta-citraurin from beta-cryptoxanthin and Zeaxanthin by carotenoid cleavage dioxygenase4 in the flavedo of citrus fruit. Plant Physiol. 2013, 163, 682–695. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodrigo, M.J.; Alquezar, B.; Also, E.; Medina, V.; Carmona, L.; Bruno, M.; Al-Babili, S.; Zacarias, L. A novel carotenoid cleavage activity involved in the biosynthesis of Citrus fruit-specific apocarotenoid pigments. J. Exp. Bot. 2013, 64, 4461–4478. [Google Scholar] [CrossRef] [PubMed]
- Park, S.-C.; Kang, L.; Park, W.S.; Ahn, M.-J.; Kwak, S.-S.; Kim, H.S. Carotenoid cleavage dioxygenase 4 (CCD4) cleaves β-carotene and interacts with IbOr in sweetpotato. Plant Biot. Rep. 2020, 14, 737–742. [Google Scholar] [CrossRef]
- Ramel, F.; Birtic, S.; Ginies, C.; Soubigou-Taconnat, L.; Triantaphylides, C.; Havaux, M. Carotenoid oxidation products are stress signals that mediate gene responses to singlet oxygen in plants. Proc. Natl. Acad. Sci. USA 2012, 109, 5535–5540. [Google Scholar] [CrossRef]
- Ramel, F.; Mialoundama, A.S.; Havaux, M. Nonenzymic carotenoid oxidation and photooxidative stress signalling in plants. J. Exp. Bot. 2013, 64, 799–805. [Google Scholar] [CrossRef]
- Havaux, M. Carotenoid oxidation products as stress signals in plants. Plant J. 2014, 79, 597–606. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Clough, S.J.; Bent, A.F. Floral dip: A simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 1998, 16, 735–743. [Google Scholar] [CrossRef]
- Mehrtens, F.; Kranz, H.; Bednarek, P.; Weisshaar, B. The Arabidopsis transcription factor MYB12 is a flavonol-specific regulator of phenylpropanoid biosynthesis. Plant Physiol. 2005, 138, 1083–1096. [Google Scholar] [CrossRef]
- Chen, T.C.; Chou, S.Y.; Chen, M.C.; Lin, J.S. IbTLD modulates reactive oxygen species scavenging and DNA protection to confer salinity stress tolerance in tobacco. Plant Sci. 2022, 323, 111415. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, J.; He, L.; Dong, J.; Zhao, C.; Tang, R.; Jia, X. Overexpression of Sweet Potato Carotenoid Cleavage Dioxygenase 4 (IbCCD4) Decreased Salt Tolerance in Arabidopsis thaliana. Int. J. Mol. Sci. 2022, 23, 9963. https://doi.org/10.3390/ijms23179963
Zhang J, He L, Dong J, Zhao C, Tang R, Jia X. Overexpression of Sweet Potato Carotenoid Cleavage Dioxygenase 4 (IbCCD4) Decreased Salt Tolerance in Arabidopsis thaliana. International Journal of Molecular Sciences. 2022; 23(17):9963. https://doi.org/10.3390/ijms23179963
Chicago/Turabian StyleZhang, Jie, Liheng He, Jingjing Dong, Cailiang Zhao, Ruimin Tang, and Xiaoyun Jia. 2022. "Overexpression of Sweet Potato Carotenoid Cleavage Dioxygenase 4 (IbCCD4) Decreased Salt Tolerance in Arabidopsis thaliana" International Journal of Molecular Sciences 23, no. 17: 9963. https://doi.org/10.3390/ijms23179963
APA StyleZhang, J., He, L., Dong, J., Zhao, C., Tang, R., & Jia, X. (2022). Overexpression of Sweet Potato Carotenoid Cleavage Dioxygenase 4 (IbCCD4) Decreased Salt Tolerance in Arabidopsis thaliana. International Journal of Molecular Sciences, 23(17), 9963. https://doi.org/10.3390/ijms23179963





