Inflammasomes—New Contributors to Blood Diseases
Abstract
:1. Introduction
- −
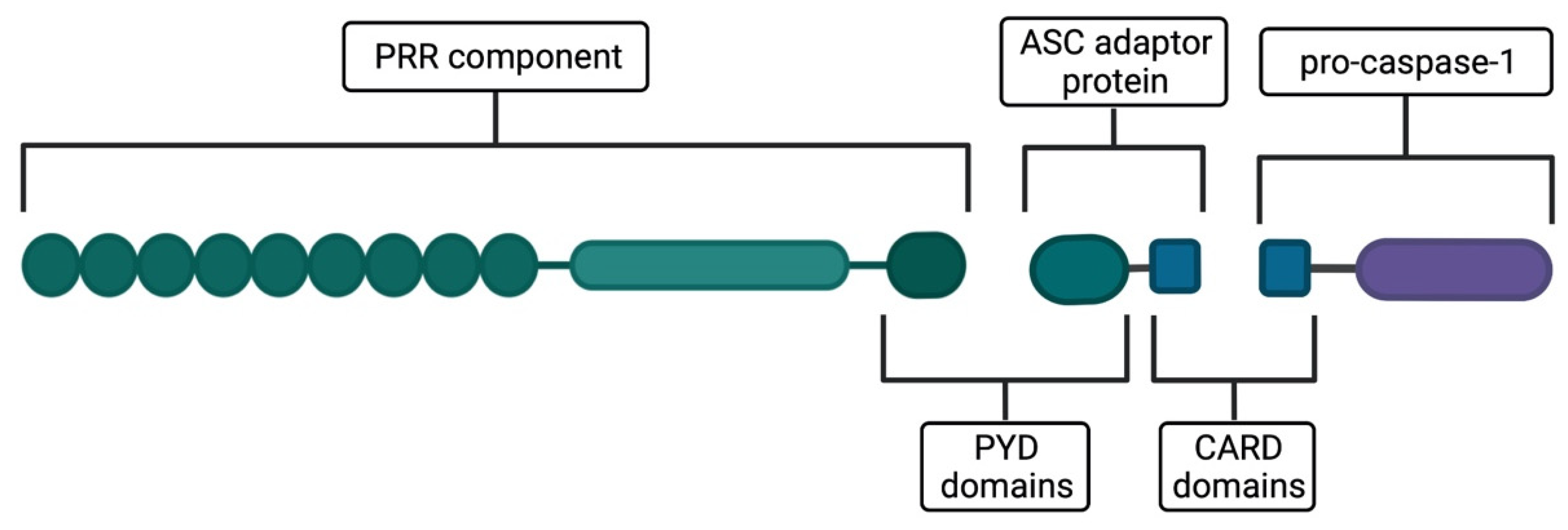
- Pattern-recognition receptors (PRRs)—molecules predominantly belonging to the NOD-like receptor (NLR) family, that detect a wide array of activators. Moreover, absent-in-melanoma-2-like (AIM2-like) receptors and pyrin are also reported as PRRs forming the inflammasomes [4], serving as exclusive exogenous DNA and toxin detectors, respectively [1,5]. The PRR component is essential for the inflammasome to initiate its function.
- −
- Apoptosis-associated speck-like protein containing a caspase-recruitment domain (ASC)—an adaptor protein composed of a pyrin domain (PYD), enabling association with the PRR component, and a caspase activation and recruitment domain (CARD), which facilitates the binding of pro-caspase-1 to the PRR–ASC complex. The adaptor protein is absent once PRR contains the CARD domain [4].
- −
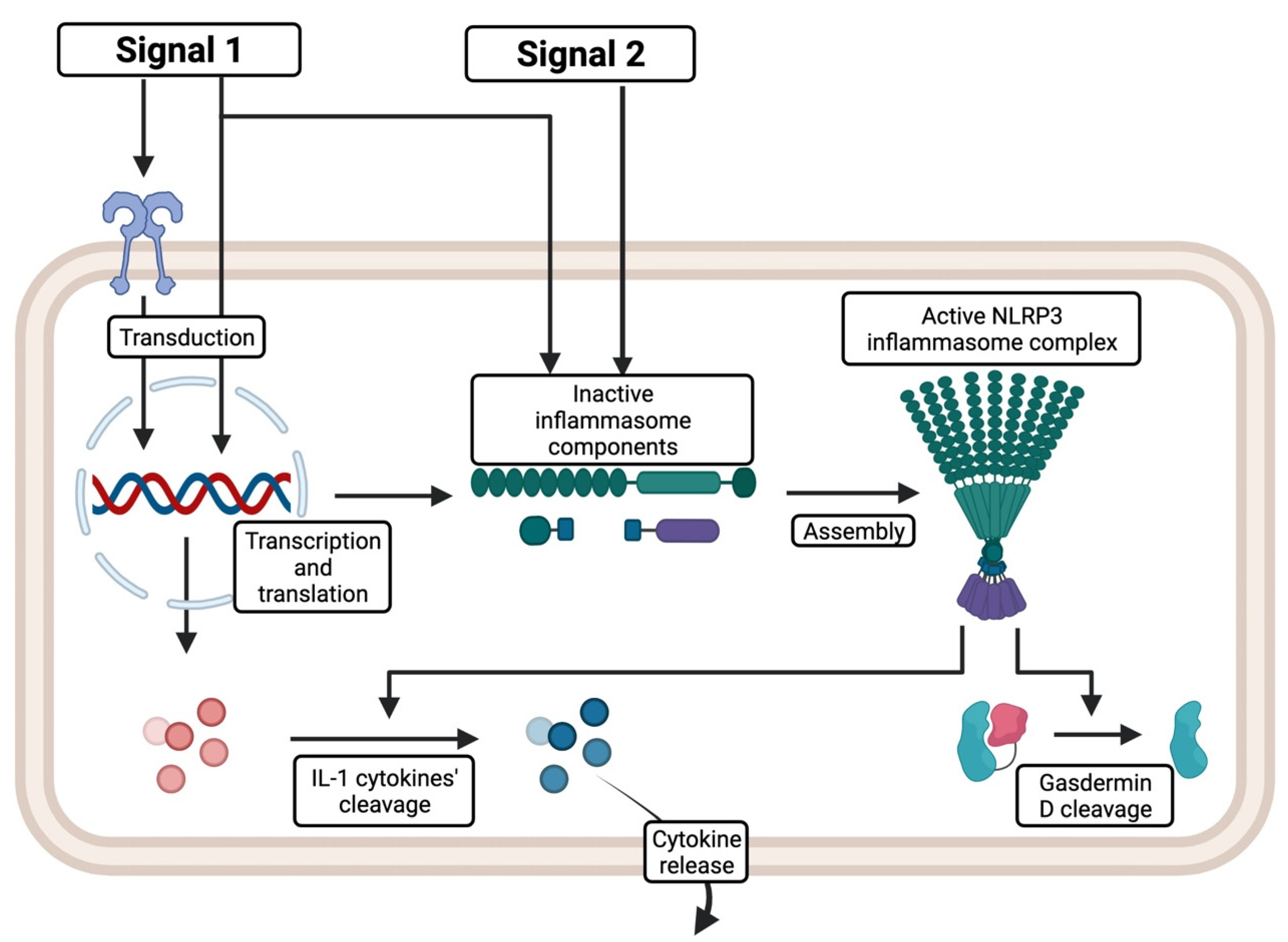
- Pro-caspase-1—an inactive form of caspase-1, a protease that cleaves the precursors of IL-1β and IL-18 cytokines and other proteins, for instance, gasdermin D [4]. It is activated upon inflammasome assembly, which is elicited by detecting a specific activating signal by PRR and subsequent association of the discussed components [4]. As a result, pro-caspase-1 undergoes autocleavage to form active caspase-1 [6].
Inflammasome Priming and Activation Mechanisms
2. Myelodysplastic Neoplasms
3. Lymphomas
4. Multiple Myeloma
5. Leukemias
5.1. Acute Myeloid Leukemia
5.2. Chronic Myeloid Leukemia
5.3. Acute Lymphoblastic Leukemia
5.4. Chronic Lymphocytic Leukemia
6. Hemophagocytic Lymphohistiocytosis
7. Graft-Versus-Host Disease
8. Sickle Cell Anemia
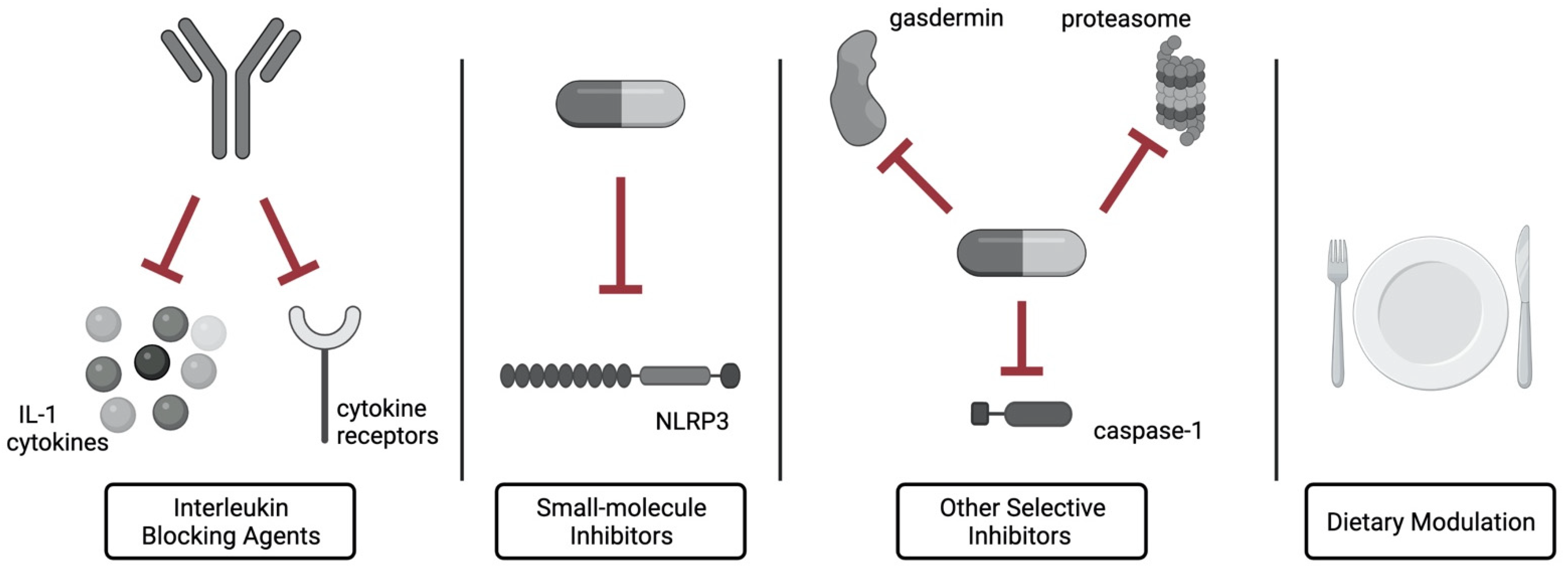
9. Inflammasomes and Immunotherapies
10. Discussion
11. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Punt, J.; Stranford, S.A.; Jones, P.P.; Owen, J.A. Innate Immunity. In Kuby Immunology; WH Freeman: New York, NY, USA, 2019; pp. 120–132. ISBN 9781319172985. [Google Scholar]
- Sharma, B.R.; Kanneganti, T.-D. NLRP3 inflammasome in cancer and metabolic diseases. Nat. Immunol. 2021, 22, 550–559. [Google Scholar] [CrossRef] [PubMed]
- Fusco, R.; Siracusa, R.; Genovese, T.; Cuzzocrea, S.; Di Paola, R. Focus on the Role of NLRP3 Inflammasome in Diseases. Int. J. Mol. Sci. 2020, 21, 4223. [Google Scholar] [CrossRef] [PubMed]
- Kelley, N.; Jeltema, D.; Duan, Y.; He, Y. The NLRP3 Inflammasome: An Overview of Mechanisms of Activation and Regulation. Int. J. Mol. Sci. 2019, 20, 3328. [Google Scholar] [CrossRef] [Green Version]
- Sharma, D.; Kanneganti, T.-D. The cell biology of inflammasomes: Mechanisms of inflammasome activation and regulation. J. Cell Biol. 2016, 213, 617–629. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, Q.; Scott, M.J. Caspase-1 as a multifunctional inflammatory mediator: Noncytokine maturation roles. J. Leukoc. Biol. 2016, 100, 961–967. [Google Scholar] [CrossRef] [PubMed]
- Urwanisch, L.; Luciano, M.; Horejs-Hoeck, J. The NLRP3 Inflammasome and Its Role in the Pathogenicity of Leukemia. Int. J. Mol. Sci. 2021, 22, 1271. [Google Scholar] [CrossRef]
- Chakraborty, S.; Shapiro, L.C.; de Oliveira, S.; Rivera-Pena, B.; Verma, A.; Shastri, A. Therapeutic targeting of the inflammasome in myeloid malignancies. Blood Cancer J. 2021, 11, 152. [Google Scholar] [CrossRef] [PubMed]
- Pretre, V.; Papadopoulos, D.; Regard, J.; Pelletier, M.; Woo, J. Interleukin-1 (IL-1) and the inflammasome in cancer. Cytokine 2022, 153, 155850. [Google Scholar] [CrossRef]
- Garlanda, C.; Dinarello, C.A.; Mantovani, A. The Interleukin-1 Family: Back to the Future. Immunity 2013, 39, 1003–1018. [Google Scholar] [CrossRef] [Green Version]
- Ratajczak, M.Z.; Kucia, M. The Nlrp3 inflammasome—The evolving story of its positive and negative effects on hematopoiesis. Curr. Opin. Hematol. 2021, 28, 251–261. [Google Scholar] [CrossRef]
- Ward, G.A.; McGraw, K.L.; Abbas-Aghababazadeh, F.; Meyer, B.S.; McLemore, A.F.; Vincelette, N.D.; Lam, N.B.; Aldrich, A.L.; Al Ali, N.H.; Padron, E.; et al. Oxidized mitochondrial DNA released after inflammasome activation is a disease biomarker for myelodysplastic syndromes. Blood Adv. 2021, 5, 2216–2228. [Google Scholar] [CrossRef] [PubMed]
- Kucia, M.; Ratajczak, J.; Bujko, K.; Adamiak, M.; Ciechanowicz, A.; Chumak, V.; Brzezniakiewicz-Janus, K.; Ratajczak, M.Z. An evidence that SARS-CoV-2/COVID-19 spike protein (SP) damages hematopoietic stem/progenitor cells in the mechanism of pyroptosis in Nlrp3 inflammasome-dependent manner. Leukemia 2021, 35, 3026–3029. [Google Scholar] [CrossRef] [PubMed]
- Khoury, J.D.; Solary, E.; Abla, O.; Akkari, Y.; Alaggio, R.; Apperley, J.F.; Bejar, R.; Berti, E.; Busque, L.; Chan, J.K.C.; et al. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Myeloid and Histiocytic/Dendritic Neoplasms. Leukemia 2022, 36, 1703–1719. [Google Scholar] [CrossRef] [PubMed]
- Fenaux, P.; Haase, D.; Santini, V.; Sanz, G.; Platzbecker, U.; Mey, U. Myelodysplastic syndromes: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2021, 32, 142–156. [Google Scholar] [CrossRef]
- Basiorka, A.A.; McGraw, K.L.; Eksioglu, E.A.; Chen, X.; Johnson, J.; Zhang, L.; Zhang, Q.; Irvine, B.A.; Cluzeau, T.; Sallman, D.A.; et al. The NLRP3 inflammasome functions as a driver of the myelodysplastic syndrome phenotype. Blood 2016, 128, 2960–2975. [Google Scholar] [CrossRef]
- Graf, J.R.; Forster, S.; Bruehl, F.K.; Banz, Y.; Hallal, M.; Brodard, J.; Bacher, V.U.; Allam, R.; Schürch, C.M.; Bonadies, N. Diagnostic and Prognostic Implications of Caspase-1 and PD-L1 Co-Expression Patterns in Myelodysplastic Syndromes. Cancers 2021, 13, 5712. [Google Scholar] [CrossRef]
- Shi, L.; Zhao, Y.; Fei, C.; Guo, J.; Jia, Y.; Wu, D.; Wu, L.; Chang, C. Cellular senescence induced by S100A9 in mesenchymal stromal cells through NLRP3 inflammasome activation. Aging 2019, 11, 9626–9642. [Google Scholar] [CrossRef]
- Sallman, D.A.; List, A. The central role of inflammatory signaling in the pathogenesis of myelodysplastic syndromes. Blood 2019, 133, 1039–1048. [Google Scholar] [CrossRef] [Green Version]
- Yin, C.; He, N.; Li, P.; Zhang, C.; Yu, J.; Hua, M.; Ji, C.; Ma, D. Polymorphisms of Interlukin-1β rs16944 confer susceptibility to myelodysplastic syndromes. Life Sci. 2016, 165, 109–112. [Google Scholar] [CrossRef]
- Alaggio, R.; Amador, C.; Anagnostopoulos, I.; Attygalle, A.D.; Araujo, I.B.D.O.; Berti, E.; Bhagat, G.; Borges, A.M.; Boyer, D.; Calaminici, M.; et al. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Lymphoid Neoplasms. Leukemia 2022, 36, 1720–1748. [Google Scholar] [CrossRef]
- Armitage, J.O.; Gascoyne, R.D.; Lunning, M.A.; Cavalli, F. Non-Hodgkin lymphoma. Lancet 2017, 390, 298–310. [Google Scholar] [CrossRef]
- Zhao, X.; Zhang, C.; Hua, M.; Wang, R.; Zhong, C.; Yu, J.; Han, F.; He, N.; Zhao, Y.; Liu, G.; et al. NLRP3 inflammasome activation plays a carcinogenic role through effector cytokine IL-18 in lymphoma. Oncotarget 2017, 8, 108571–108583. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Z.-H.; Zhang, L.; Jing, F.-J.; Xiao, S.-X.; Gao, Y.; Bian, H.-Y.; Zhao, X. Genetic Polymorphisms in NLRP3 Inflammasome-Associated Genes in Patients with B-Cell Non-Hodgkin’s Lymphoma. J. Inflamm. Res. 2021, 14, 5687–5697. [Google Scholar] [CrossRef]
- Lu, F.; Zhao, Y.; Pang, Y.; Ji, M.; Sun, Y.; Wang, H.; Zou, J.; Wang, Y.; Li, G.; Sun, T.; et al. NLRP3 inflammasome upregulates PD-L1 expression and contributes to immune suppression in lymphoma. Cancer Lett. 2021, 497, 178–189. [Google Scholar] [CrossRef]
- Reinhart, N.M.; Akinyemi, I.A.; Frey, T.R.; Xu, H.; Agudelo, C.; Brathwaite, J.; Burton, E.M.; Burgula, S.; McIntosh, M.T.; Bhaduri-McIntosh, S. The danger molecule HMGB1 cooperates with the NLRP3 inflammasome to sustain expression of the EBV lytic switch protein in Burkitt lymphoma cells. Virology 2022, 566, 136–142. [Google Scholar] [CrossRef]
- Ozel, I.; Akkaya, I.; Oylumlu, E.; Uzel, G.; Ciraci, C. Adenosine-Induced NLRP11 in B Lymphoblasts Suppresses Human CD4+ T Helper Cell Responses. J. Immunol. Res. 2020, 2020, 1421795. [Google Scholar] [CrossRef] [PubMed]
- Ellwanger, K.; Becker, E.; Kienes, I.; Sowa, A.; Postma, Y.; Gloria, Y.C.; Weber, A.N.; Kufer, T.A. The NLR family pyrin domain–containing 11 protein contributes to the regulation of inflammatory signaling. J. Biol. Chem. 2018, 293, 2701–2710. [Google Scholar] [CrossRef] [Green Version]
- Brito-Zerón, P.; Baldini, C.; Bootsma, H.; Bowman, S.J.; Jonsson, R.; Mariette, X.; Sivils, K.; Theander, E.; Tzioufas, A.; Ramos-Casals, M. Sjögren syndrome. Nat. Rev. Dis. Prim. 2016, 2, 16047. [Google Scholar] [CrossRef]
- Vakrakou, A.G.; Boiu, S.; Ziakas, P.; Xingi, E.; Boleti, H.; Manoussakis, M.N. Systemic activation of NLRP3 inflammasome in patients with severe primary Sjögren’s syndrome fueled by inflammagenic DNA accumulations. J. Autoimmun. 2018, 91, 23–33. [Google Scholar] [CrossRef]
- Baldini, C.; Santini, E.; Rossi, C.; Donati, V.; Solini, A. The P2X7 receptor-NLRP3 inflammasome complex predicts the development of non-Hodgkin’s lymphoma in Sjogren’s syndrome: A prospective, observational, single-centre study. J. Intern. Med. 2017, 282, 175–186. [Google Scholar] [CrossRef]
- Huanosta-Murillo, E.; Alcántara-Hernández, M.; Hernández-Rico, B.; Victoria-Acosta, G.; Miranda-Cruz, P.; Domínguez-Gómez, M.A.; Jurado-Santacruz, F.; Patiño-López, G.; Pérez-Koldenkova, V.; Palma-Guzmán, A.; et al. NLRP3 Regulates IL-4 Expression in TOX+ CD4+ T Cells of Cutaneous T Cell Lymphoma to Potentially Promote Disease Progression. Front. Immunol. 2021, 12, 668369. [Google Scholar] [CrossRef]
- Hasegawa, H.; Bissonnette, R.P.; Gillings, M.; Sasaki, D.; Taniguchi, H.; Kitanosono, H.; Tsuruda, K.; Kosai, K.; Uno, N.; Morinaga, Y.; et al. Induction of apoptosis by HBI-8000 in adult T-cell leukemia/lymphoma is associated with activation of Bim and NLRP3. Cancer Sci. 2016, 107, 1124–1133. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gerecke, C.; Fuhrmann, S.; Strifler, S.; Schmidt-Hieber, M.; Einsele, H.; Knop, S. The Diagnosis and Treatment of Multiple Myeloma. Dtsch. Arztebl. Int. 2016, 113, 470–476. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nakamura, K.; Kassem, S.; Cleynen, A.; Chrétien, M.-L.; Guillerey, C.; Putz, E.M.; Bald, T.; Förster, I.; Vuckovic, S.; Hill, G.R.; et al. Dysregulated IL-18 Is a Key Driver of Immunosuppression and a Possible Therapeutic Target in the Multiple Myeloma Microenvironment. Cancer Cell 2018, 33, 634–648. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hofbauer, D.; Mougiakakos, D.; Broggini, L.; Zaiss, M.; Büttner-Herold, M.; Bach, C.; Spriewald, B.; Neumann, F.; Bisht, S.; Nolting, J.; et al. β2-microglobulin triggers NLRP3 inflammasome activation in tumor-associated macrophages to promote multiple myeloma progression. Immunity 2021, 54, 1772–1787. [Google Scholar] [CrossRef]
- Li, Y.; Li, N.; Yan, Z.; Li, H.; Chen, L.; Zhang, Z.; Fan, G.; Xu, K.; Li, Z. Dysregulation of the NLRP3 inflammasome complex and related cytokines in patients with multiple myeloma. Hematology 2016, 21, 144–151. [Google Scholar] [CrossRef]
- Zhao, X.; Hua, M.; Yan, S.; Yu, J.; Han, F.; Zhong, C.; Wang, R.; Zhang, C.; Hou, M.; Ma, D. The Genetic Polymorphisms of NLRP3 Inflammasome Associated with T Helper Cells in Patients with Multiple Myeloma. J. Immunol. Res. 2018, 2018, 7569809. [Google Scholar] [CrossRef] [Green Version]
- Lust, J.A.; Lacy, M.Q.; Zeldenrust, S.R.; Dispenzieri, A.; Gertz, M.A.; Witzig, T.E.; Kumar, S.; Hayman, S.R.; Russell, S.J.; Buadi, F.K.; et al. Induction of a Chronic Disease State in Patients With Smoldering or Indolent Multiple Myeloma by Targeting Interleukin 1β-Induced Interleukin 6 Production and the Myeloma Proliferative Component. Mayo Clin. Proc. 2009, 84, 114–122. [Google Scholar] [CrossRef]
- Zhong, C.; Wang, R.; Hua, M.; Zhang, C.; Han, F.; Xu, M.; Yang, X.; Li, G.; Hu, X.; Sun, T.; et al. NLRP3 Inflammasome Promotes the Progression of Acute Myeloid Leukemia via IL-1β Pathway. Front. Immunol. 2021, 12, 661939. [Google Scholar] [CrossRef]
- Jia, Y.; Zhang, C.; Hua, M.; Wang, M.; Chen, P.; Ma, D. Aberrant NLRP3 inflammasome associated with aryl hydrocarbon receptor potentially contributes to the imbalance of T-helper cells in patients with acute myeloid leukemia. Oncol. Lett. 2017, 14, 7031–7044. [Google Scholar] [CrossRef] [Green Version]
- Liu, Q.; Hua, M.; Zhang, C.; Wang, R.; Liu, J.; Yang, X.; Han, F.; Hou, M.; Ma, D. NLRP3-activated bone marrow dendritic cells play antileukemic roles via IL-1β/Th1/IFN-γ in acute myeloid leukemia. Cancer Lett. 2021, 520, 109–120. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Wu, Y.; Wen, X.; Li, P.; Lu, F.; Shang, H. Chronic stress promotes acute myeloid leukemia progression through HMGB1/NLRP3/IL-1β signaling pathway. J. Mol. Med. 2021, 99, 403–414. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Wang, H.; Wei, S.; Wang, Z.; Ji, G. Inhibition of ER stress-related IRE1α/CREB/NLRP1 pathway promotes the apoptosis of human chronic myelogenous leukemia cell. Mol. Immunol. 2018, 101, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Zhang, A.; Yu, J.; Yan, S.; Zhao, X.; Chen, C.; Zhou, Y.; Zhao, X.; Hua, M.; Wang, R.; Zhang, C.; et al. The genetic polymorphism and expression profiles of NLRP3 inflammasome in patients with chronic myeloid leukemia. Hum. Immunol. 2018, 79, 57–62. [Google Scholar] [CrossRef]
- Paugh, S.W.; Bonten, E.J.; Savic, D.; Ramsey, L.B.; E Thierfelder, W.; Gurung, P.; Malireddi, R.K.S.; Actis, M.; Mayasundari, A.; Min, J.; et al. NALP3 inflammasome upregulation and CASP1 cleavage of the glucocorticoid receptor cause glucocorticoid resistance in leukemia cells. Nat. Genet. 2015, 47, 607–614. [Google Scholar] [CrossRef] [Green Version]
- Zhang, C.; Han, F.; Yu, J.; Hu, X.; Hua, M.; Zhong, C.; Wang, R.; Zhao, X.; Shi, Y.; Ji, C.; et al. Investigation of NF-κB-94ins/del ATTG and CARD8 (rs2043211) Gene Polymorphism in Acute Lymphoblastic Leukemia. Front. Endocrinol. 2019, 10, 501. [Google Scholar] [CrossRef] [Green Version]
- Alves, F.S.; Xabregas, L.A.; Kerr, M.W.A.; Souza, G.L.; Pereira, D.S.; Magalhães-Gama, F.; Santiago, M.R.R.; Garcia, N.P.; Tarragô, A.M.; Ogusku, M.M.; et al. Genetic polymorphisms of inflammasome genes associated with pediatric acute lymphoblastic leukemia and clinical prognosis in the Brazilian Amazon. Sci. Rep. 2021, 11, 9869. [Google Scholar] [CrossRef]
- Salaro, E.; Rambaldi, A.; Falzoni, S.; Amoroso, F.S.; Franceschini, A.; Sarti, A.C.; Bonora, M.; Cavazzini, F.; Rigolin, G.M.; Ciccone, M.; et al. Involvement of the P2X7-NLRP3 axis in leukemic cell proliferation and death. Sci. Rep. 2016, 6, 26280. [Google Scholar] [CrossRef] [Green Version]
- Hayden, A.; Park, S.; Giustini, D.; Lee, A.Y.; Chen, L.Y. Hemophagocytic syndromes (HPSs) including hemophagocytic lymphohistiocytosis (HLH) in adults: A systematic scoping review. Blood Rev. 2016, 30, 411–420. [Google Scholar] [CrossRef] [Green Version]
- Andersson, U. Hyperinflammation: On the pathogenesis and treatment of macrophage activation syndrome. Acta Paediatr. 2021, 110, 2717–2722. [Google Scholar] [CrossRef]
- Schulert, G.S.; Cron, R.Q. The genetics of macrophage activation syndrome. Genes Immun. 2020, 21, 169–181. [Google Scholar] [CrossRef] [PubMed]
- Canna, S.W.; De Jesus, A.A.; Gouni, S.; Brooks, S.R.; Marrero, B.; Liu, Y.; DiMattia, M.A.; Zaal, K.J.; Sanchez, G.A.M.; Kim, H.; et al. An activating NLRC4 inflammasome mutation causes autoinflammation with recurrent macrophage activation syndrome. Nat. Genet. 2014, 46, 1140–1146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bracaglia, C.; Prencipe, G.; De Benedetti, F. Macrophage Activation Syndrome: Different mechanisms leading to a one clinical syndrome. Pediatr. Rheumatol. Online J. 2017, 15, 5. [Google Scholar] [CrossRef] [Green Version]
- Weiss, E.S.; Girard-Guyonvarc’H, C.; Holzinger, D.; De Jesus, A.A.; Tariq, Z.; Picarsic, J.; Schiffrin, E.J.; Foell, D.; Grom, A.A.; Ammann, S.; et al. Interleukin-18 diagnostically distinguishes and pathogenically promotes human and murine macrophage activation syndrome. Blood 2018, 131, 1442–1455. [Google Scholar] [CrossRef] [PubMed]
- Canna, S.W.; Girard, C.; Malle, L.; de Jesus, A.; Romberg, N.; Kelsen, J.; Surrey, L.F.; Russo, P.; Sleight, A.; Schiffrin, E.; et al. Life-threatening NLRC4-associated hyperinflammation successfully treated with IL-18 inhibition. J. Allergy Clin. Immunol. 2017, 139, 1698–1701. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hill, G.R. Inflammation and Bone Marrow Transplantation. Biol. Blood Marrow Transplant. 2009, 15, 139–141. [Google Scholar] [CrossRef] [Green Version]
- Koehn, B.H.; Zeiser, R.; Blazar, B.R. Inflammasome effects in GvHD. Oncotarget 2015, 6, 38444–38445. [Google Scholar] [CrossRef]
- Jankovic, D.; Ganesan, J.; Bscheider, M.; Stickel, N.; Weber, F.C.; Guarda, G.; Follo, M.; Pfeifer, D.; Tardivel, A.; Ludigs, K.; et al. The Nlrp3 inflammasome regulates acute graft-versus-host disease. J. Exp. Med. 2013, 210, 1899–1910. [Google Scholar] [CrossRef] [Green Version]
- Koehn, B.H.; Apostolova, P.; Haverkamp, J.M.; Miller, J.S.; McCullar, V.; Tolar, J.; Munn, D.H.; Murphy, W.J.; Brickey, W.J.; Serody, J.S.; et al. GVHD-associated, inflammasome-mediated loss of function in adoptively transferred myeloid-derived suppressor cells. Blood 2015, 126, 1621–1628. [Google Scholar] [CrossRef] [Green Version]
- Koehn, B.H.; Saha, A.; McDonald-Hyman, C.; Loschi, M.; Thangavelu, G.; Ma, L.; Zaiken, M.C.; Dysthe, J.; Krepps, W.; Panthera, J.; et al. Danger-associated extracellular ATP counters MDSC therapeutic efficacy in acute GVHD. Blood 2019, 134, 1670–1682. [Google Scholar] [CrossRef]
- Chen, S.; Smith, B.; Iype, J.; Prestipino, A.; Pfeifer, D.; Grundmann, S.; Schmitt-Graeff, A.; Idzko, M.; Beck, Y.; Prinz, G.; et al. MicroRNA-155–deficient dendritic cells cause less severe GVHD through reduced migration and defective inflammasome activation. Blood 2015, 126, 103–112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, S.; Shi, P.; Lv, C.; Li, H.; Pan, B.; Chen, W.; Zhao, K.; Yan, Z.; Chen, C.; Loake, G.J.; et al. Loss of NLRP3 Function Alleviates Murine Hepatic Graft-versus-Host Disease. Biol. Blood Marrow Transplant. 2018, 24, 2409–2417. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qiao, J.; Huang, Y.; Xia, Y.; Chu, P.; Yao, H.; Xu, L.; Qi, K.; Liu, Y.; Xu, K.; Zeng, L. Busulfan and cyclosphamide induce liver inflammation through NLRP3 activation in mice after hematopoietic stem cell transplantation. Sci. Rep. 2015, 5, 17828. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cerqueira, B.A.; Boas, W.V.; Zanette, A.D.; Reis, M.G.; Goncalves, M.S. Increased concentrations of IL-18 and uric acid in sickle cell anemia: Contribution of hemolysis, endothelial activation and the inflammasome. Cytokine 2011, 56, 471–476. [Google Scholar] [CrossRef] [Green Version]
- Vogel, S.; Arora, T.; Wang, X.; Mendelsohn, L.; Nichols, J.; Allen, D.; Shet, A.S.; Combs, C.A.; Quezado, Z.M.N.; Thein, S.L. The platelet NLRP3 inflammasome is upregulated in sickle cell disease via HMGB1/TLR4 and Bruton tyrosine kinase. Blood Adv. 2018, 2, 2672–2680. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vogel, S.; Kamimura, S.; Arora, T.; Smith, M.L.; Almeida, L.E.; Combs, C.A.; Thein, S.L.; Quezado, Z.M. NLRP3 inflammasome and bruton tyrosine kinase inhibition interferes with upregulated platelet aggregation and in vitro thrombus formation in sickle cell mice. Biochem. Biophys. Res. Commun. 2021, 555, 196–201. [Google Scholar] [CrossRef]
- Pitanga, T.N.; Santana, S.S.; Zanette, D.L.; Guarda, C.C.; Santiago, R.P.; Maffili, V.V.; Lima, J.B.; Carvalho, G.Q.; Filho, J.R.; Ferreira, J.R.D.; et al. Effect of lysed and non-lysed sickle red cells on the activation of NLRP3 inflammasome and LTB4 production by mononuclear cells. Inflamm. Res. 2021, 70, 823–834. [Google Scholar] [CrossRef]
- Vats, R.; Brzoska, T.; Bennewitz, M.F.; Jimenez, M.A.; Pradhan-Sundd, T.; Tutuncuoglu, E.; Jonassaint, J.; Gutierrez, E.; Watkins, S.C.; Shiva, S.; et al. Platelet Extracellular Vesicles Drive Inflammasome–IL-1β–Dependent Lung Injury in Sickle Cell Disease. Am. J. Respir. Crit. Care Med. 2020, 201, 33–46. [Google Scholar] [CrossRef]
- Vogel, S.; Thein, S.L. Platelets at the crossroads of thrombosis, inflammation and haemolysis. Br. J. Haematol. 2018, 180, 761–767. [Google Scholar] [CrossRef]
- Liu, D.; Xu, X.; Dai, Y.; Zhao, X.; Bao, S.; Ma, W.; Zha, L.; Liu, S.; Liu, Y.; Zheng, J.; et al. Blockade of AIM2 inflammasome or α1-AR ameliorates IL-1β release and macrophage-mediated immunosuppression induced by CAR-T treatment. J. Immunother. Cancer 2021, 9, e001466. [Google Scholar] [CrossRef]
- Zahid, A.; Li, B.; Kombe, A.J.K.; Jin, T.; Tao, J. Pharmacological Inhibitors of the NLRP3 Inflammasome. Front. Immunol. 2019, 10, 2538. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chauhan, D.; vande Walle, L.; Lamkanfi, M. Therapeutic modulation of inflammasome pathways. Immunol. Rev. 2020, 297, 123–138. [Google Scholar] [CrossRef] [PubMed]
- Zmora, N.; Levy, M.; Pevsner-Fishcer, M.; Elinav, E. Inflammasomes and intestinal inflammation. Mucosal Immunol. 2017, 10, 865–883. [Google Scholar] [CrossRef] [PubMed]
- Fauteux-Daniel, S.; Viel, S.; Besson, L.; Zhang, J.; Marotel, M.; Mathieu, A.-L.; Pescarmona, R.; Charrier, E.; Henry, T.; Belot, A.; et al. Deletion of Inflammasome Components Is Not Sufficient To Prevent Fatal Inflammation in Models of Familial Hemophagocytic Lymphohistiocytosis. J. Immunol. 2018, 200, 3769–3776. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.; Gerstein, M.; Snyder, M. RNA-Seq: A revolutionary tool for transcriptomics. Nat. Rev. Genet. 2009, 10, 57–63. [Google Scholar] [CrossRef]
- Khan, S.; Kaihara, K.A. Single-Cell RNA-Sequencing of Peripheral Blood Mononuclear Cells with ddSEQ. Methods Mol. Biol. 2019, 1979, 155–176. [Google Scholar] [CrossRef]
- Budden, C.F.; Gearing, L.J.; Kaiser, R.; Standke, L.; Hertzog, P.J.; Latz, E. Inflammasome-induced extracellular vesicles harbour distinct RNA signatures and alter bystander macrophage responses. J. Extracell. Vesicles 2021, 10, e12127. [Google Scholar] [CrossRef]
- Sheth, V.S.; Gauthier, J. Taming the beast: CRS and ICANS after CAR T-cell therapy for ALL. Bone Marrow Transplant. 2020, 56, 552–566. [Google Scholar] [CrossRef]
| Disease | Inflammasome Type | Expression Status | Key Cytokines | Role in Pathogenesis | Related Signaling Pathways | Ref. |
|---|---|---|---|---|---|---|
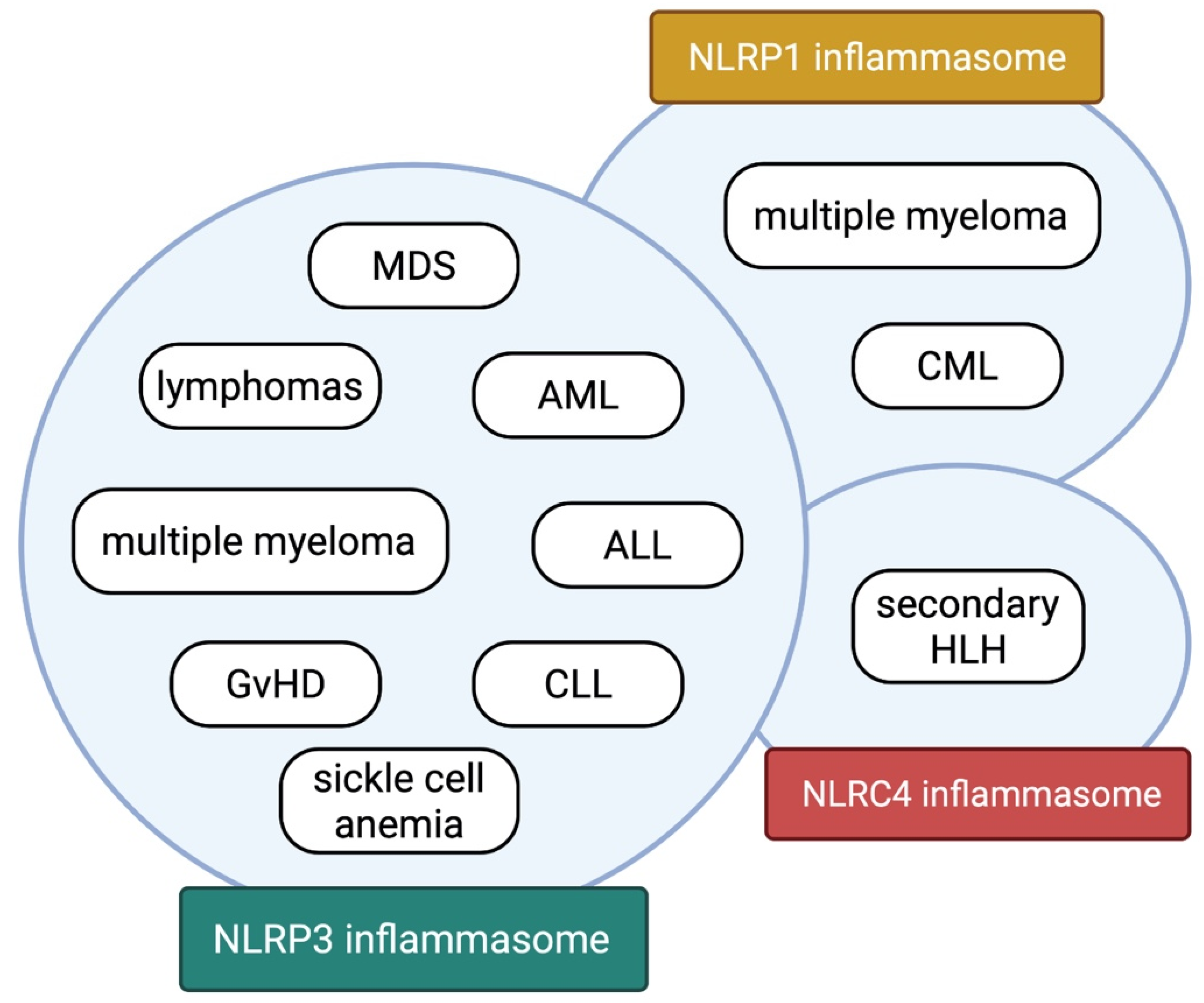
| Myelodysplastic Neoplasms (MDS) | NLRP3 | Upregulated | IL-1β and IL-18 | pyroptosis of MDS cells triggers an inflammatory response and promotes the proliferation of neoplastic cells | TLR4/MyD88/IRAK1, IRAK4/TRAF6/NF-κB WNT/β -catenin | [8,12,16,17,18,19] |
| Diffuse Large B-Cell Lymphoma (DLBCL) | NLRP3 | Upregulated | IL-18 | NLRP3 inflammasome promotes cell proliferation and apoptosis inhibition and reduces the therapeutic effect of dexamethasone | NLRP3/IL-18/IFN-Y/JAK-STAT/IRF/PD-L1 | [23,25] |
| EBV-positive Burkitt Lymphoma | NLRP3 | Downregulated | IL-1β | downregulated NLRP3 inflammasome cannot prevent latent EBV infection NLRP11 molecule represses NLRP3 expression by inhibiting the NF-κB pathway | TLR4/MyD88/TRAF6/NF-κB pathway | [26,27,28] |
| Marginal Zone Lymphoma | NLRP3 | Upregulated | IL-1β and IL-18 | lymphomagenesis, NLRP3 inflammasome promotes formation of GC-like structures | P2 × 7R-NLRP3 axis | [30,31] |
| Adult T-cell Lymphoma (ATL) | NLRP3 | Upregulated artificially by HBI-8000 drug | IL-1β | NLRP3 inflammasome prevents latent HTLV-1 infection | not mentioned in the reference | [33] |
| Multiple Myeloma (MM) | NLRP1, NLRP3 | Upregulated | IL-18 | NLRP1-dependent activation of MDSCs enables MM cells to evade immune control β2 microglobulin accumulation in myeloma-associated macrophages causes NLRP3-dependent inflammatory response promoting myeloma cell proliferation | unknown | [35,36] |
| Acute Myeloid Leukemia (AML) | NLRP3 | Upregulated | IL-1β and IL-18 | NLRP3 inflammasome overexpression in AML cells promotes proliferation and survival NLRP3 inflammasome activation in bone marrow dendritic cells induces Th1 response promoting apoptosis and inhibiting proliferation of AML cells | HMGB1/TLR4, TLR2, RAGE/MyD88/NF-κB IFN-γ/STAT1 pathway in Th1 lymphocytes | [40,42,43] |
| Chronic Myeloid Leukemia (CML) | NLRP1 | Upregulated | IL-1β | NLRP1 inflammasome suppresses apoptosis and promotes proliferation of CML cells and imatinib resistance | IRE1α/CREB/NLRP1 pathway associated with Endoplasmic Reticulum Stress | [44] |
| Acute Lymphoblastic Leukemia (ALL) | NLRP3 | Upregulated | - | caspase-1 cleaves glucocorticoid receptors in their transactivation domain, contributing to glucocorticoid resistance | hypomethylation of NLRP3 and CASP1 (caspase-1 gene) promoters | [46] |
| Chronic Lymphocytic Leukemia (CLL) | NLRP3 | Downregulated | IL-1β | NLRP3 inflammasome downregulation promotes proliferation of CLL cells | P2 × 7R/NLRP3 axis | [49] |
| Hemophagocytic Lymphohistiocytosis (HLH) | NLRC4 | Upregulated | IL-18 | T cell exhaustion death causes a release of alarmins activating macrophages, which subsequently undergo pyroptosis contributing to inflammation in the positive feedback loop | unspecified TLR and IFN-γ signaling pathways | [51,52,54,75] |
| Graft-versus-Host Disease (GvHD) | NLRP3 | Upregulated | IL-1β and IL-18 | NLRP3 inflammasome activation induces overexpression of costimulatory molecules on APCs, promoting alloreactive T cells response NLRP3 inflammasome activation in MDSCs impedes their anti-inflammatory function by reducing arginase production conditioning-induced mucosal damage promotes the inflammasome-dependent response | TLR4/MyD88/TRIF/NF-κB | [58,59,60,61] |
| Sickle Cell Anemia (SCA) | NLRP3 | Upregulated | IL-1β and IL-18 | hemolysis-related alarmins activate NLRP3 inflammasomes in platelets leading to aggregation of platelets | HMGB1/TLR4/MyD88/IRAK4/NF-κB BTK kinase signaling | [66,67,68,69,70] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tomasik, J.; Basak, G.W. Inflammasomes—New Contributors to Blood Diseases. Int. J. Mol. Sci. 2022, 23, 8129. https://doi.org/10.3390/ijms23158129
Tomasik J, Basak GW. Inflammasomes—New Contributors to Blood Diseases. International Journal of Molecular Sciences. 2022; 23(15):8129. https://doi.org/10.3390/ijms23158129
Chicago/Turabian StyleTomasik, Jaromir, and Grzegorz Władysław Basak. 2022. "Inflammasomes—New Contributors to Blood Diseases" International Journal of Molecular Sciences 23, no. 15: 8129. https://doi.org/10.3390/ijms23158129
APA StyleTomasik, J., & Basak, G. W. (2022). Inflammasomes—New Contributors to Blood Diseases. International Journal of Molecular Sciences, 23(15), 8129. https://doi.org/10.3390/ijms23158129






