Comparative Analysis of the Cytotoxic Effect of a Complex of Selenium Nanoparticles Doped with Sorafenib, “Naked” Selenium Nanoparticles, and Sorafenib on Human Hepatocyte Carcinoma HepG2 Cells
Abstract
1. Introduction
2. Results
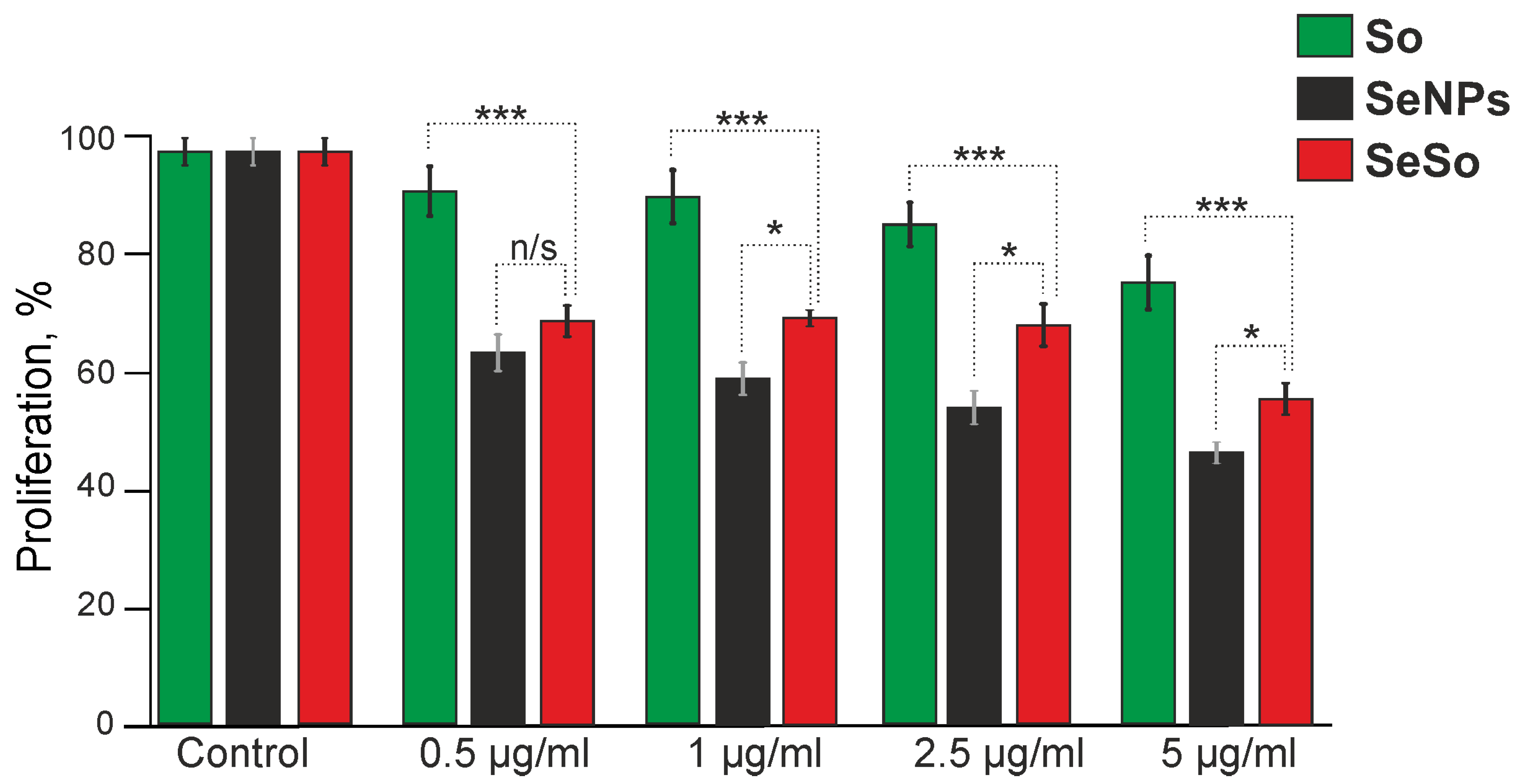
2.1. The Effect of Sorafenib, Selenium Nanoparticles, and Selenium–Sorafenib Nanocomplex on the Proliferation and Viability of HepG2 Cells
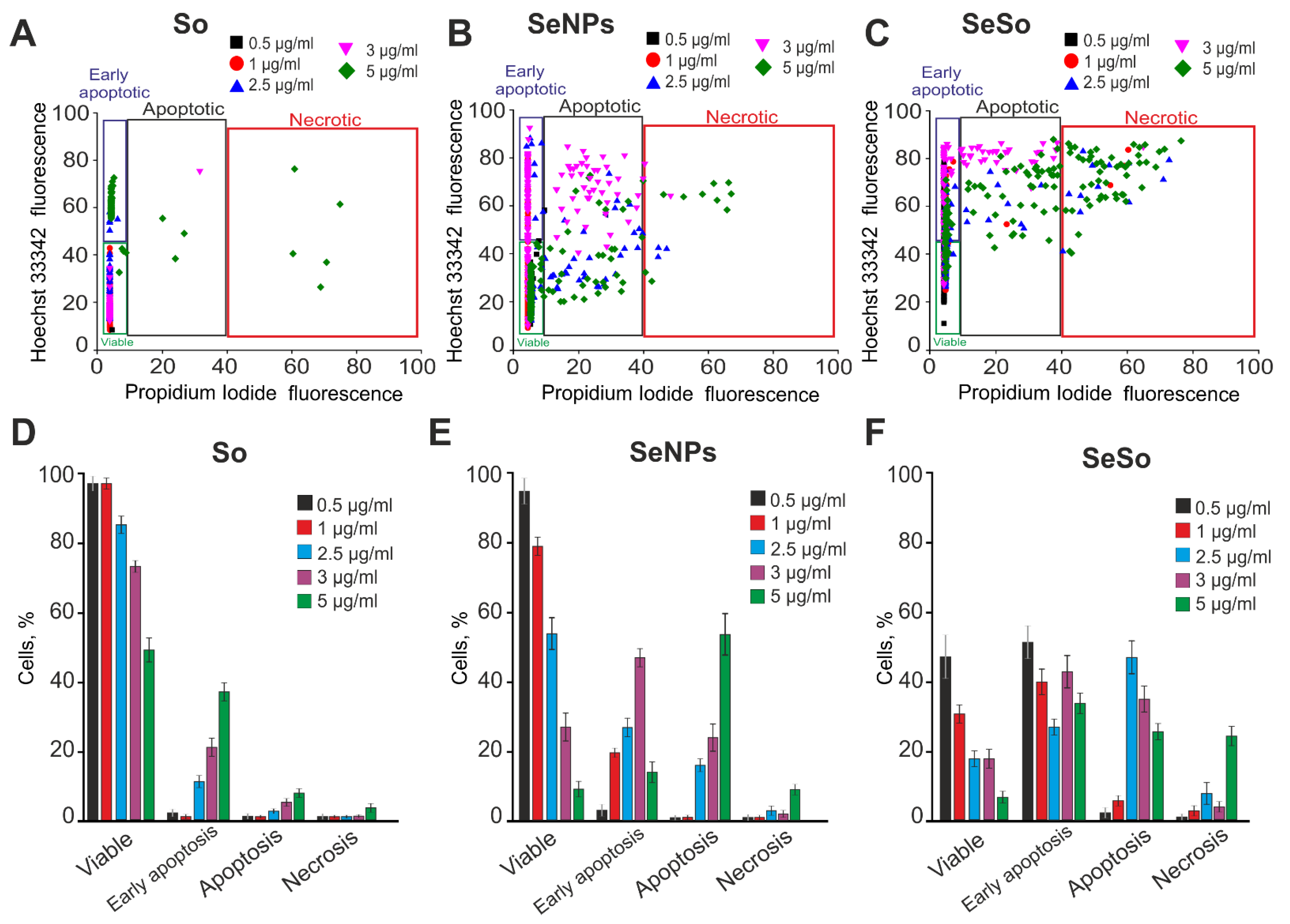
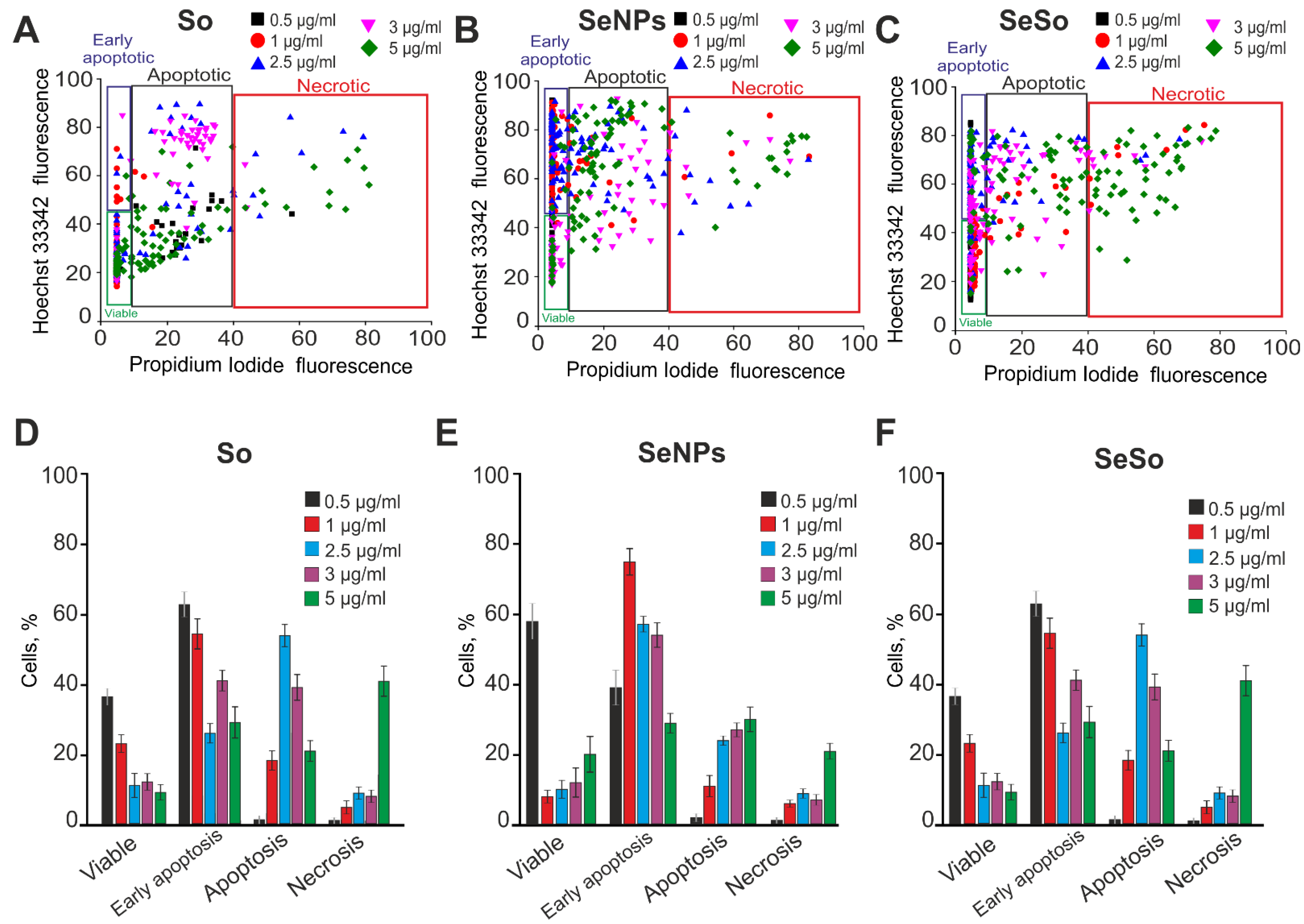
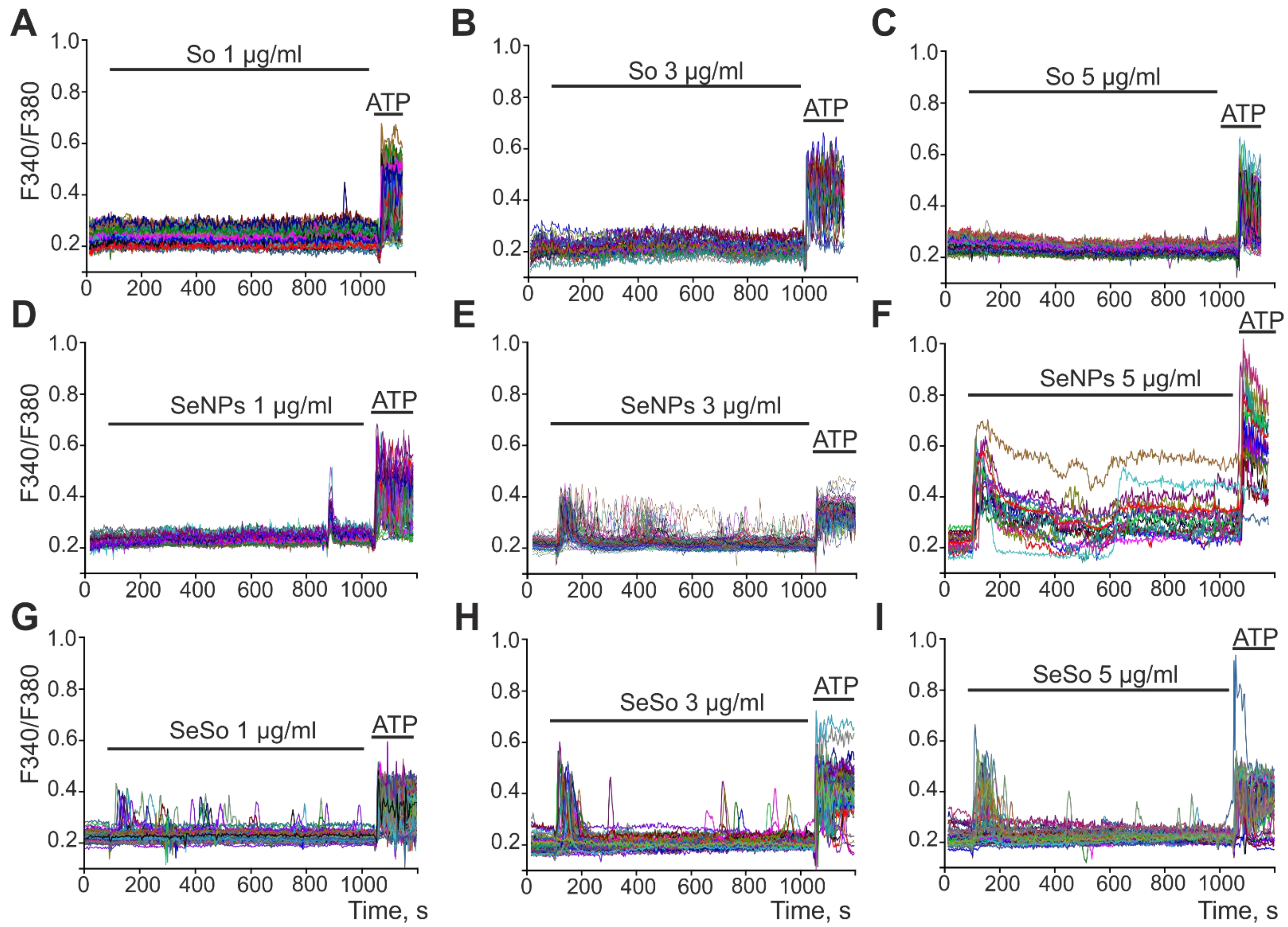
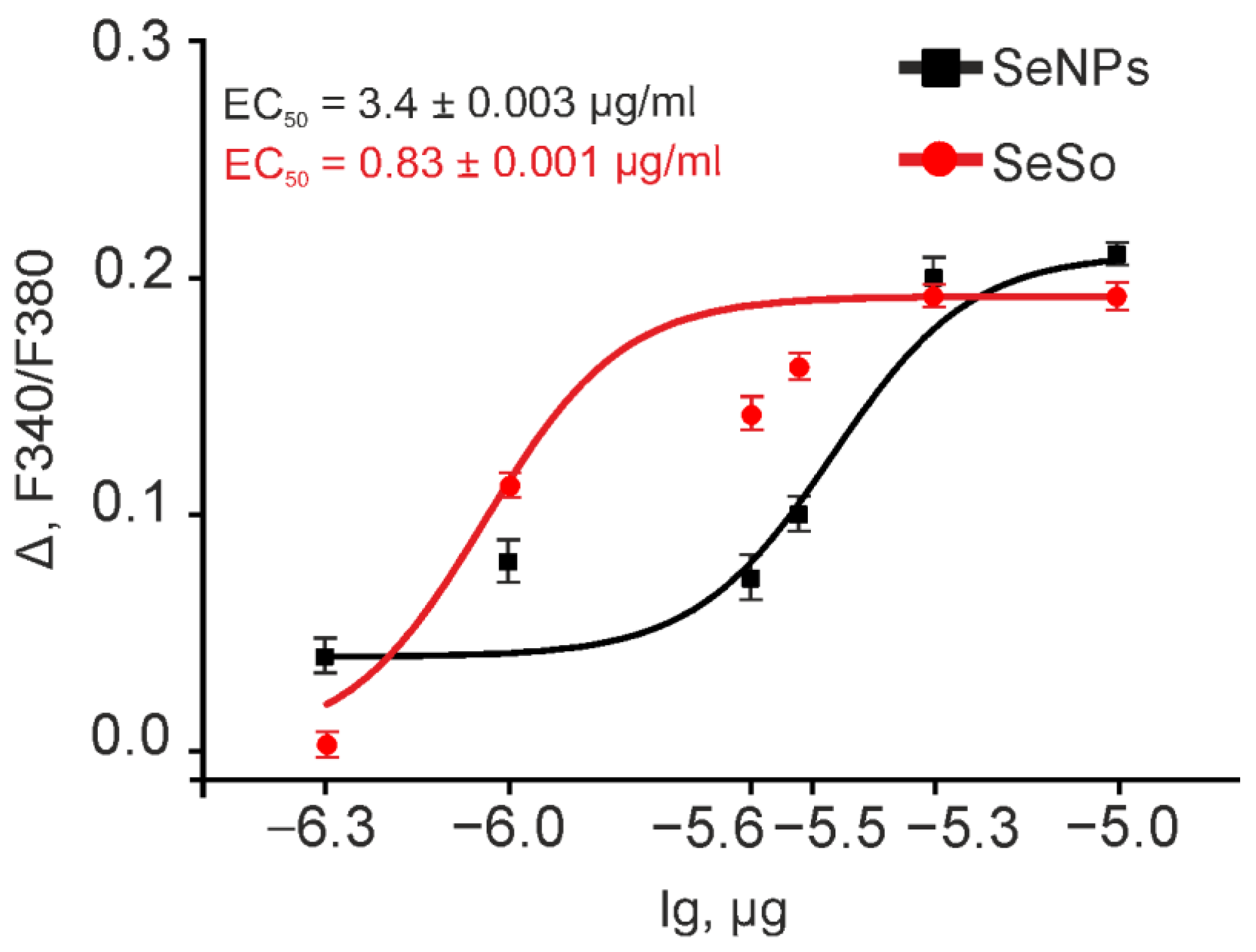
2.2. The Effect of Sorafenib, Selenium Nanoparticles, and Selenium–Sorafenib Nanocomplex on the Calcium Signaling System of HepG2 Cells
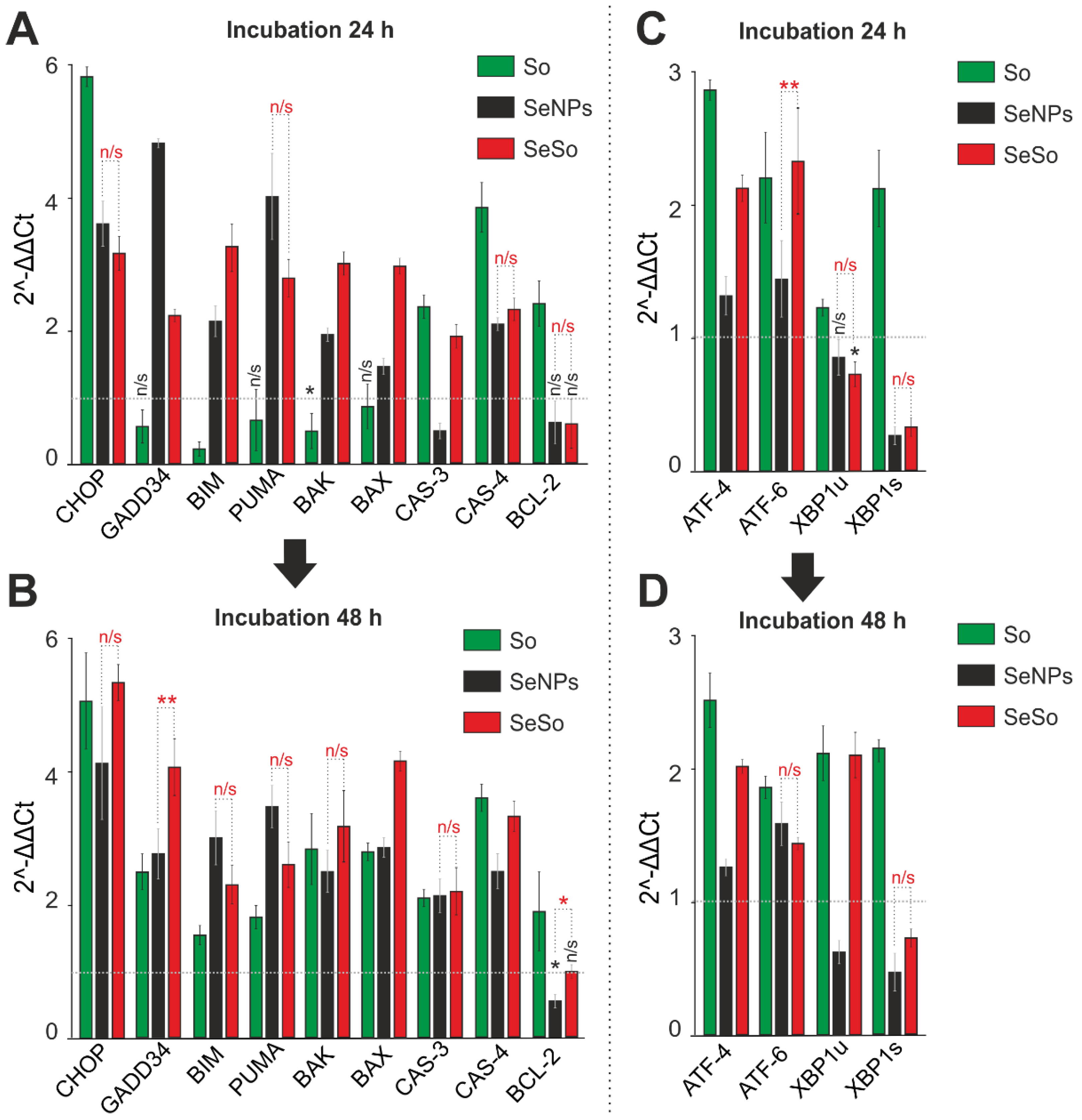
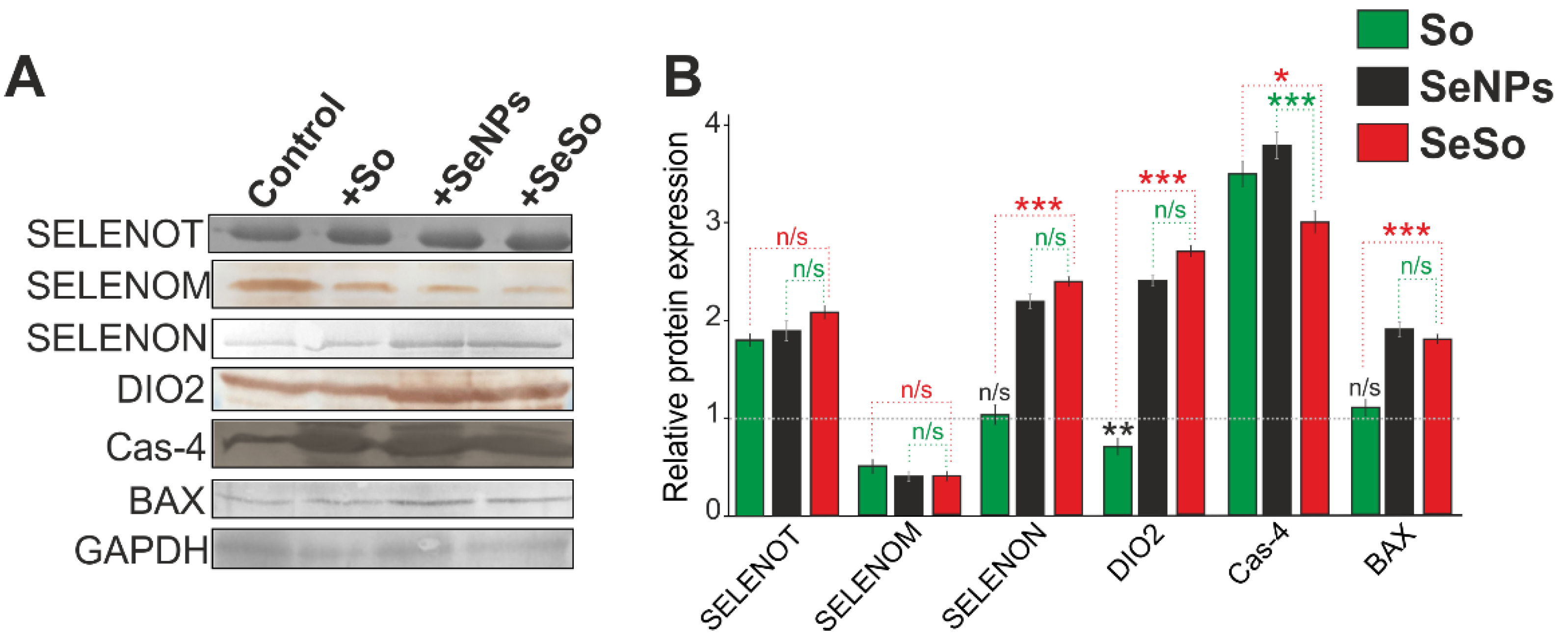
2.3. Effects of Sorafenib, Selenium Nanoparticles, and Selenium–Sorafenib Nanocomplex on the Expression of Genes Encoding Signal Kinases and Selenium-Containing Proteins
3. Discussion
4. Materials and Methods
4.1. Reagents
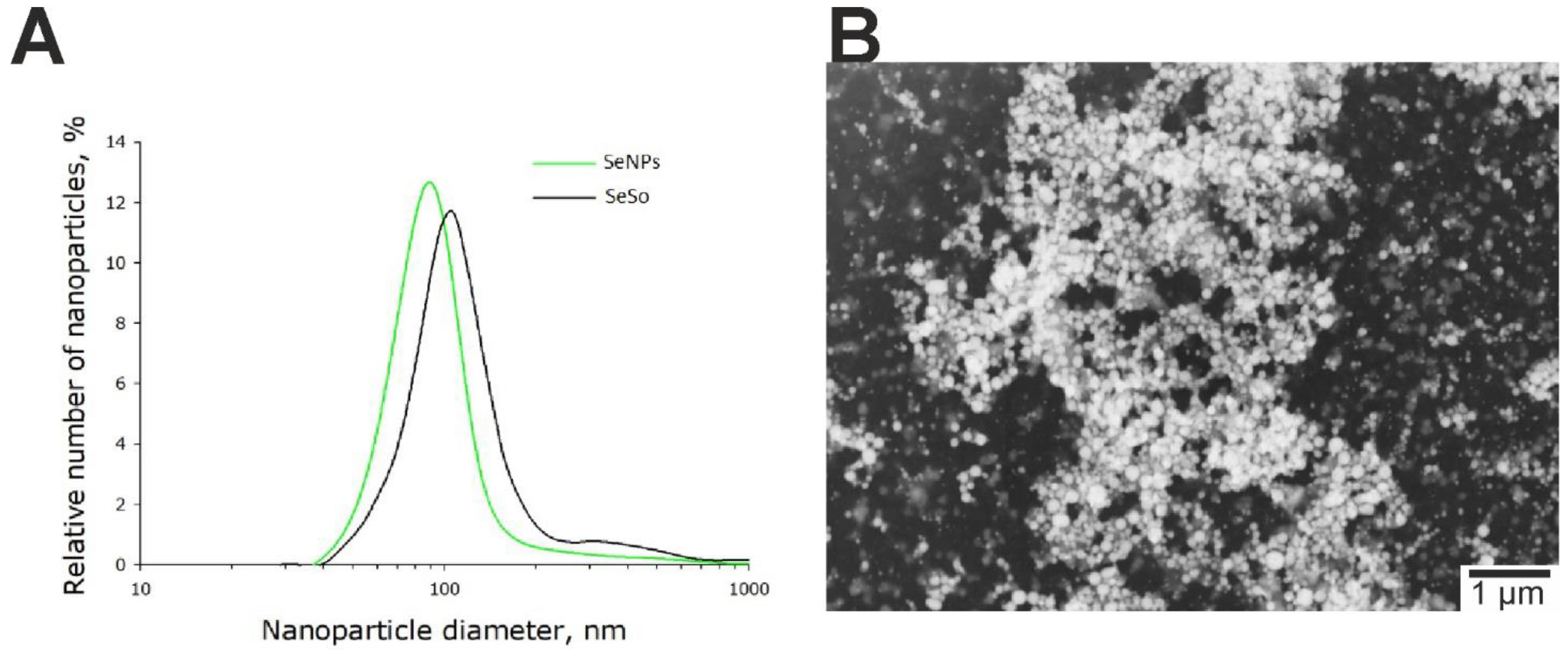
4.2. Preparation of Selenium Nanoparticles and Sorafenib-Doped Selenium Nanoparticles (SeSo)
4.3. Cell Culture
4.4. Assessment of Cell Viability and Apoptosis
4.5. MTT Assay
4.6. Fluorescent Ca2+ Measurements
4.7. Extraction of RNA and Real-Time Polymerase Chain Reaction (RT-qPCR)
4.8. Western Blotting
4.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef] [PubMed]
- Lencioni, R.; de Baere, T.; Soulen, M.C.; Rilling, W.S.; Geschwind, J.F.H. Lipiodol transarterial chemoembolization for hepatocellular carcinoma: A systematic review of efficacy and safety data. Hepatology 2016, 64, 106–116. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Fang, Y.; Zhao, R.; Le, J.; Zhang, B.; Huang, R.; Chen, Z.; Shao, J. Evolution in medicinal chemistry of sorafenib derivatives for hepatocellular carcinoma. Eur. J. Med. Chem. 2019, 179, 916–935. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, S.M.; Carter, C.; Tang, L.; Wilkie, D.; McNabola, A.; Rong, H.; Chen, C.; Zhang, X.; Vincent, P.; McHugh, M.; et al. BAY 43-9006 exhibits broad spectrum oral antitumor activity and targets the RAF/MEK/ERK pathway and receptor tyrosine kinases involved in tumor progression and angiogenesis. Cancer Res. 2004, 64, 7099–7109. [Google Scholar] [CrossRef]
- Yang, S.; Zhang, B.; Gong, X.; Wang, T.; Liu, Y.; Zhang, N. In vivo biodistribution, biocompatibility, and efficacy of Sorafenib-loaded lipid-based nanosuspensions evaluated experimentally in cancer. Int. J. Nanomed. 2016, 11, 2329–2343. [Google Scholar]
- Li, M.; Su, Y.; Zhang, F.; Chen, K.; Xu, X.; Xu, L.; Zhou, J.; Wang, W. A dual-targeting reconstituted high density lipoprotein leveraging the synergy of sorafenib and antimiRNA21 for enhanced hepatocellular carcinoma therapy. Acta Biomater. 2018, 75, 413–426. [Google Scholar] [CrossRef]
- Mancuso, A.; Airoldi, A.; Vigano, R.; Pinzello, G. Fatal gastric bleeding during Sorafenib treatment for hepatocellular carcinoma recurrence after liver transplantation. Dig. Liver Dis. 2011, 43, 754. [Google Scholar] [CrossRef]
- Kroon, J.; Metselaar, J.M.; Storm, G.; van der Pluijm, G. Liposomal nanomedicines in the treatment of prostate cancer. Cancer Treat. Rev. 2014, 40, 578–584. [Google Scholar] [CrossRef]
- Wang, H.; Wang, H.; Yang, W.; Yu, M.; Sun, S.; Xie, B. Improved oral bioavailability and liver targeting of Sorafenib Solid lipid nanoparticles in rats. AAPS PharmSciTech 2018, 19, 761–768. [Google Scholar] [CrossRef]
- Zheng, L.; Li, C.; Huang, X.; Lin, X.; Lin, W.; Yang, F.; Chen, T. Thermosensitive hydrogels for sustained-release of sorafenib and selenium nanoparticles for localized synergistic chemoradiotherapy. Biomaterials 2019, 216, 119220. [Google Scholar] [CrossRef]
- Su, Y.; Wang, K.; Li, Y.; Song, W.; Xin, Y.; Zhao, W.; Tian, J.; Ren, L.; Lu, L. Sorafenib-loaded polymeric micelles as passive targeting therapeutic agents for hepatocellular carcinoma therapy. Nanomedicine 2018, 13, 1009–1023. [Google Scholar] [CrossRef] [PubMed]
- Vishwakarma, S.K.; Sharmila, P.; Bardia, A.; Chandrakala, L.; Raju, N.; Sravani, G.; Sastry, B.V.S.; Habeeb, M.A.; Khan, A.A.; Dhayal, M. Use of biocompatible sorafenib-gold nanoconjugates for reversal of drug resistance in human hepatoblatoma cells. Sci. Rep. 2017, 7, 8539. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Fang, Y.; Chen, X.; Deng, R.; Zhang, Y.; Shao, J. Recent advances of Sorafenib nanoformulations for cancer therapy: Smart nanosystem and combination therapy. Asian J. Pharm. Sci. 2021, 16, 318–336. [Google Scholar] [CrossRef] [PubMed]
- Ebrahim, H.M.; El-Rouby, M.N.; Morsy, M.E.; Said, M.M.; Ezz, M.K. The synergistic cytotoxic effect of laser-irradiated gold nanoparticles and Sorafenib against the growth of a human hepatocellular carcinoma cell line. Asian Pac. J. Cancer Prev. 2019, 20, 3369–3376. [Google Scholar] [CrossRef]
- Babos, G.; Biro, E.; Meiczinger, M.; Feczko, T. Dual drug delivery of Sorafenib and doxorubicin from PLGA and PEG-PLGA polymeric nanoparticles. Polymers 2018, 10, 895. [Google Scholar] [CrossRef]
- Xiong, Q.; Cui, M.; Yu, G.; Wang, J.; Song, T. Facile fabrication of reduction-responsive supramolecular nanoassemblies for co-delivery of doxorubicin and Sorafenib toward hepatoma cells. Front. Pharmacol. 2018, 9, 61. [Google Scholar] [CrossRef]
- Liu, T.; Zeng, L.; Jiang, W.; Fu, Y.; Zheng, W.; Chen, T. Rational design of cancer-targeted selenium nanoparticles to antagonize multidrug resistance in cancer cells. Nanomed. Nanotechnol. Biol. Med. 2015, 11, 947–958. [Google Scholar] [CrossRef]
- Huang, G.; Liu, Z.; He, L.; Luk, K.-H.; Cheung, S.-T.; Wong, K.-H.; Chen, T. Autophagy is an important action mode for functionalized selenium nanoparticles to exhibit anti-colorectal cancer activity. Biomater. Sci. 2018, 6, 2508–2517. [Google Scholar] [CrossRef]
- Yu, B.; Liu, T.; Du, Y.; Luo, Z.; Zheng, W.; Chen, T. X-ray-responsive selenium nanoparticles for enhanced cancer chemo-radiotherapy. Colloids Surf. B Biointerfaces 2016, 139, 180–189. [Google Scholar] [CrossRef]
- Varlamova, E.G.; Turovsky, E.A.; Blinova, E.V. Therapeutic potential and main methods of obtaining selenium nanoparticles. Int. J. Mol. Sci. 2021, 22, 10808. [Google Scholar] [CrossRef]
- Varlamova, E.G.; Goltyaev, M.V.; Mal’tseva, V.N.; Turovsky, E.A.; Sarimov, R.M.; Simakin, A.V.; Gudkov, S.V. Mechanisms of the cytotoxic effect of selenium nanoparticles in different human cancer cell lines. Int. J. Mol. Sci. 2021, 22, 7798. [Google Scholar] [CrossRef]
- Varlamova, E.G.; Turovsky, E.A.; Babenko, V.A.; Plotnikov, E.Y. The mechanisms underlying the protective action of selenium nanoparticles against ischemia/reoxygenation are mediated by the activation of the Ca2+ signaling system of astrocytes and reactive astrogliosis. Int. J. Mol. Sci. 2021, 22, 12825. [Google Scholar] [CrossRef]
- Turovsky, E.A.; Mal’tseva, V.N.; Sarimov, R.M.; Simakin, A.V.; Gudkov, S.V.; Plotnikov, E.Y. Features of the cytoprotective effect of selenium nanoparticles on primary cortical neurons and astrocytes during oxygen-glucose deprivation and reoxygenation. Sci. Rep. 2022, 12, 1710. [Google Scholar] [CrossRef]
- Fekry, M.I.; Ezzat, S.M.; Salama, M.M.; Alshehri, O.Y.; Al-Abd, A.M. Bioactive glycoalkaloides isolated from Solanum melongena fruit peels with potential anticancer properties against hepatocellular carcinoma cells. Sci. Rep. 2019, 9, 1746. [Google Scholar] [CrossRef]
- Goltyaev, M.V.; Mal’tseva, V.N.; Varlamova, E.G. Expression of ER-resident selenoproteins and activation of cancer cells apoptosis mechanisms under ER-stress conditions caused by methylseleninic acid. Gene 2020, 755, 144884. [Google Scholar] [CrossRef]
- King, A.P.; WilSon, J.J. Endoplasmic reticulum stress: An arising target for metal-based anticancer agents. Chem. Soc. Rev. 2020, 49, 8113–8136. [Google Scholar] [CrossRef]
- Varlamova, E.G.; Turovsky, E.A. The main cytotoxic effects of methylseleninic acid on various cancer cells. Int. J. Mol. Sci. 2021, 22, 6614. [Google Scholar] [CrossRef]
- Patergnani, S.; Danese, A.; Bouhamida, E.; Aguiari, G.; Previati, M.; Pinton, P.; Giorgi, C. Various aspects of calcium signaling in the regulation of apoptosis, autophagy, cell proliferation, and cancer. Int. J. Mol. Sci. 2020, 21, 8323. [Google Scholar] [CrossRef]
- Kuznetsova, Y.P.; Goltyaev, M.V.; Gorbacheva, O.S.; Novoselov, S.V.; Varlamova, E.G.; Fesenko, E.E. Influence of sodium selenite on the mRNA expression of the mammalian selenocysteine-containing protein genes in testicle and prostate cancer cells. Dokl. Biochem. Biophys. 2018, 480, 131–134. [Google Scholar] [CrossRef]
- Varlamova, E.G.; Goltyaev, M.V.; Kuznetsova, J.P. Effect of sodium selenite on gene expression of SELF, SELW, and TGR selenoproteins in adenocarcinoma cells of the human prostate. Mol. Biol. 2018, 52, 519–526. [Google Scholar] [CrossRef]
- Tang, K.; Luo, C.; Li, Y.; Lu, C.; Zhou, W.; Huang, H.; Chen, X. The study of a novel sorafenib derivative HLC-080 as an antitumor agent. PLoS ONE 2014, 9, e101889. [Google Scholar] [CrossRef]
- Chen, K.-F.; Tai, W.-T.; Hsu, C.-Y.; Huang, J.-W.; Liu, C.-Y.; Chen, P.-J.; Kim, I.; Shiau, C.-W. Blockade of STAT3 activation by sorafenib derivatives through enhancing SHP-1 phosphatase activity. Eur. J. Med. Chem. 2012, 55, 220–227. [Google Scholar] [CrossRef]
- Tai, W.-T.; Cheng, A.-L.; Shiau, C.-W.; Huang, H.-P.; Huang, J.-W.; Chen, P.-J.; Chen, K.-F. Signal transducer and activator of transcription 3 is a major kinase-independent target of sorafenib in hepatocellular carcinoma. J. Hepatol. 2011, 55, 1041–1048. [Google Scholar] [CrossRef]
- Poojari, R.; Sawant, A.V.; Kini, S.; Srivastava, R.; Panda, D. Antihepatoma activity of multifunctional polymeric nanoparticles via inhibition of microtubules and tyrosine kinases. Nanomedicine 2020, 15, 381–396. [Google Scholar] [CrossRef]
- Schmidt, C.M.; McKillop, I.H.; Cahill, P.A.; Sitzmann, J.V. Increased MAPK expression and activity in primary human hepatocellular carcinoma. Biochem. Biophys. Res. Commun. 1997, 236, 54–58. [Google Scholar] [CrossRef]
- Sonntag, R.; Gassler, N.; Bangen, J.M.; Trautwein, C.; Liedtke, C. Pro-apoptotic sorafenib signaling in murine hepatocytes depends on malignancy and is associated with PUMA expression in vitro and in vivo. Cell Death Dis. 2014, 5, 1030. [Google Scholar] [CrossRef]
- Han, M.-K.; Song, E.-K.; Guo, Y.; Ou, X.; Mantel, C.; Broxmeyer, H.E. SIRT1 regulates apoptosis and Nanog expression in mouse embryonic stem cells by controlling p53 subcellular localization. Cell Stem Cell 2008, 2, 241–251. [Google Scholar] [CrossRef]
- Gillissen, B.; Richter, A.; Richter, A.; Preissner, R.; Schulze-Osthoff, K.; Essmann, F.; Daniel, P.T. Bax/Bak-independent mitochondrial depolarization and reactive oxygen species induction by sorafenib overcome resistance to apoptosis in renal cell carcinoma. J. Biol. Chem. 2017, 292, 6478–6492. [Google Scholar] [CrossRef]
- Jeong, K.-Y.; Park, M.; Sim, J.-J.; Kim, H.M. Combination antitumor effect of sorafenib via calcium-dependent deactivation of focal adhesion kinase targeting colorectal cancer cells. Molecules 2020, 25, 5299. [Google Scholar] [CrossRef]
- Ito, K.; Okamoto, I.; Araki, N.; Kawano, Y.; Nakao, M.; Fujiyama, S.; Tomita, K.; Mimori, T.; Saya, H. Calcium influx triggers the sequential proteolysis of extracellular and cytoplasmic domains of E-cadherin, leading to loss of β-catenin from cell-cell contacts. Oncogene 1999, 18, 7080–7090. [Google Scholar] [CrossRef]
- Rahmani, M.; Davis, E.M.; Crabtree, T.R.; Habibi, J.R.; Nguyen, T.K.; Dent, P.; Grant, S. The kinase inhibitor Sorafenib induces cell death through a process involving induction of endoplasmic reticulum stress. Mol. Cell. Biol. 2007, 27, 5499–5513. [Google Scholar] [CrossRef] [PubMed]
- Han, X.-J.; Chae, J.-K.; Lee, M.-J.; You, K.-R.; Lee, B.-H.; Kim, D.-G. Involvement of GADD153 and cardiac ankyrin repeat protein in hypoxia-induced apoptosis of H9c2 cells. J. Biol. Chem. 2005, 280, 23122–23129. [Google Scholar] [CrossRef] [PubMed]
- Schneider, C.; Wallner, M.; Kolesnik, E.; Herbst, V.; Mächler, H.; Pichler, M.; von Lewinski, D.; Sedej, S.; Rainer, P.P. The Anti-Cancer Multikinase Inhibitor Sorafenib Impairs Cardiac Contractility by Reducing Phospholamban Phosphorylation and Sarcoplasmic Calcium Transients. Sci. Rep. 2018, 8, 5295. [Google Scholar] [CrossRef] [PubMed]
- Turovsky, E.A.; Varlamova, E.G. Mechanism of Ca2+-dependent pro-apoptotic action of selenium nanoparticles, mediated by activation of Cx43 hemichannels. Biology 2021, 10, 743. [Google Scholar] [CrossRef]
- Yoshida, H.; Matsui, T.; Yamamoto, A.; Okada, T.; Mori, K. XBP1 mRNA is induced by ATF6 and spliced by IRE1 in response to ER stress to produce a highly active transcription factor. Cell 2001, 107, 881–891. [Google Scholar] [CrossRef]
- Calfon, M.; Zeng, H.; Urano, F.; Till, J.H.; Hubbard, S.R.; Harding, H.P.; Clark, S.G.; Ron, D. IRE1 couples endoplasmic reticulum load to secretory capacity by processing the XBP-1 mRNA. Nature 2002, 415, 92–96. [Google Scholar] [CrossRef]
- Nishitoh, H.; Matsuzawa, A.; Tobiume, K.; Saegusa, K.; Takeda, K.; Inoue, K.; Hori, S.; Kakizuka, A.; Ichijo, H. ASK1 is essential for endoplasmic reticulum stress-induced neuronal cell death triggered by expanded polyglutamine repeats. Genes Dev. 2002, 16, 1345–1355. [Google Scholar] [CrossRef]
- Dahmer, M.K. Caspases-2, -3, and -7 are involved in thapsigargin-induced apoptosis of SH-SY5Y neuroblastoma cells. J. Neurosci. Res. 2005, 80, 576–583. [Google Scholar] [CrossRef]
- Hitomi, J.; Katayama, T.; Eguchi, Y.; Kudo, T.; Taniguchi, M.; Koyama, Y.; Manabe, T.; Yamagishi, S.; Bando, Y.; Imaizumi, K.; et al. Involvement of caspase-4 in endoplasmic reticulum stress-induced apoptosis and Abeta-induced cell death. J. Cell Biol. 2004, 165, 347–356. [Google Scholar] [CrossRef]
- Yoshida, I.; Monji, A.; Tashiro, K.; Nakamura, K.; Inoue, R.; Kanba, S. Depletion of intracellular Ca2+ store itself may be a major factor in thapsigargin-induced ER stress and apoptosis in PC12 cells. Neurochem. Int. 2006, 48, 696–702. [Google Scholar] [CrossRef]
- Treiman, M. Regulation of the endoplasmic reticulum calcium storage during the unfolded protein response-significance in tissue ischemia? Trends Cardiovasc. Med. 2002, 12, 57–62. [Google Scholar] [CrossRef]
- Malhi, H.; Kaufman, R.J. Endoplasmic reticulum stress in liver disease. J. Hepatol. 2011, 54, 795–809. [Google Scholar] [CrossRef] [PubMed]
- Prieto-Domínguez, N.; Ordóñez, R.; Fernández, A.; García-Palomo, A.; Muntané, J.; González-Gallego, J.; Mauriz, J.L. Modulation of Autophagy by Sorafenib: Effects on Treatment Response. Front. Pharmacol. 2016, 7, 151. [Google Scholar] [CrossRef] [PubMed]
- Harvald, E.B.; Olsen, A.S.; Faergeman, N.J. Autophagy in the light of sphingolipid metabolism. Apoptosis 2015, 20, 658–670. [Google Scholar] [CrossRef] [PubMed]
- Ordoñez, R.; Fernández, A.; Prieto-Domínguez, N.; Martínez, L.; García-Ruiz, C.; Fernández-Checa, J.C.; Mauriz, J.L.; González-Gallego, J. Ceramide metabolism regulates autophagy and apoptotic cell death induced by melatonin in liver cancer cells. J. Pineal Res. 2015, 59, 178–189. [Google Scholar] [CrossRef] [PubMed]
- Petersen, M.C.; Vatner, D.F.; Shulman, G.I. Regulation of hepatic glucose metabolism in health and disease. Nat. Rev. Endocrinol. 2017, 13, 572–587. [Google Scholar] [CrossRef] [PubMed]
- Luo, H.; Yang, Y.; Duan, J.; Wu, P.; Jiang, Q.; Xu, C. PTEN-regulated AKT/FoxO3a/Bim signaling contributes to reactive oxygen species-mediated apoptosis in selenite-treated colorectal cancer cells. Cell Death Dis. 2013, 4, 481. [Google Scholar] [CrossRef]
- Zhao, Z.; Barcus, M.; Kim, J.; Lum, K.L.; Mills, C.; Lei, X.G. High dietary selenium intake alters lipid metabolism and protein synthesis in liver and muscle of pigs. J. Nutr. 2016, 146, 1625–1633. [Google Scholar] [CrossRef]
- Zheng, X.; Ren, B.; Li, X.; Yan, H.; Xie, Q.; Liu, H.; Zhou, J.; Tian, J.; Huang, K. Selenoprotein F knockout leads to glucose and lipid metabolism disorders in mice. J. Biol. Inorg. Chem. 2020, 25, 1009–1022. [Google Scholar] [CrossRef]
- Varlamova, E.G.; Goltyaev, M.V.; Turovsky, E.A. The role of selenoproteins SELENOM and SELENOT in the regulation of apoptosis, ER-stress, and calcium homeostasis in human glioblastoma cells. Biology 2022, in press. [Google Scholar] [CrossRef]
- Lee, D.; Xu, I.M.-J.; Chiu, D.K.-C.; Leibold, J.; Tse, A.P.-W.; Bao, M.H.-R.; Yuen, V.W.-H.; Chan, C.Y.-K.; Lai, R.K.-H.; Chin, D.W.-C.; et al. Induction of oxidative stress through inhibition of thioredoxin reductase 1 is an effective therapeutic approach for hepatocellular carcinoma. Hepatology 2019, 69, 1768–1786. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.; Zhang, G.; Zheng, Y.; Yang, Y.; Chen, C.; Xia, J.; Liang, L.; Lei, C.; Hu, Y.; Cai, X.; et al. SLC27A5 deficiency activates NRF2/TXNRD1 pathway by increased lipid peroxidation in HCC. Cell Death Differ. 2020, 27, 1086–1104. [Google Scholar] [CrossRef] [PubMed]
- Wei, R.; Qiu, H.; Xu, J.; Mo, J.; Liu, Y.; Gui, Y.; Huang, G.; Zhang, S.; Yao, H.; Huang, X.; et al. Expression and prognostic potential of GPX1 in human cancers based on data mining. Ann. Transl. Med. 2020, 8, 124. [Google Scholar] [CrossRef]
- Cheng, Y.; Xu, T.; Li, S.; Ruan, H. GPX1, a biomarker for the diagnosis and prognosis of kidney cancer, promotes the progression of kidney cancer. Aging 2019, 11, 12165–12176. [Google Scholar] [CrossRef] [PubMed]
- Mousa, A.B. Sorafenib in the treatment of advanced hepatocellular carcinoma. Saudi J. Gastroenterol. 2008, 14, 40–42. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, H.; Alves, A.M.; Marques, M.; Vale, A.M.; Pereira, P.; Macedo, G. Hepatocellular carcinoma treatment with sorafenib: Real-life evaluation of prognostic factors and a practical clue for patient management. GE Port. J. Gastroenterol. 2016, 23, 243–248. [Google Scholar] [CrossRef]
- Wan, J.; Liu, T.; Mei, L.; Li, J.; Gong, K.; Yu, C.; Li, W. Synergistic antitumour activity of sorafenib in combination with tetrandrine is mediated by reactive oxygen species (ROS)/Akt signaling. Br. J. Cancer 2013, 109, 342–350. [Google Scholar] [CrossRef]
- Tang, W.; Chen, Z.; Zhang, W.; Cheng, Y.; Zhang, B.; Wu, F.; Wang, Q.; Wang, S.; Rong, D.; Reiter, F.P.; et al. The mechanisms of sorafenib resistance in hepatocellular carcinoma: Theoretical basis and therapeutic aspects. Signal Transduct. Target. Ther. 2020, 5, 87. [Google Scholar] [CrossRef]
- Baimler, I.V.; Simakin, A.V.; Chevokin, V.K.; Podvyaznikov, V.A.; Gudkov, S.V. Features of optical breakdown of aqueous colloidal Solutions of ferric oxide (Fe2O3) nanoparticles occurring on individual or on two closely located nanoparticles. Chem. Phys. Lett. 2021, 776, 138697. [Google Scholar] [CrossRef]
- Baymler, I.V.; Simakin, A.V.; Gudkov, S.V. Investigation of the laser-induced breakdown plasma, acoustic vibrations and dissociation processes of water molecules caused by laser breakdown of colloidal Solutions containing Ni nanoparticles. Plasma Sources Sci. Technol. 2021, 30, 125015. [Google Scholar] [CrossRef]
- Drexler, H.G.; Uphoff, C.C. Mycoplasma contamination of cell cultures: Incidence, sources, effects, detection, elimination, prevention. Cytotechnology 2002, 39, 75–90. [Google Scholar] [CrossRef] [PubMed]
- Gaidin, S.G.; Turovskaya, M.V.; Gavrish, M.S.; Babaev, A.A.; Mal’tseva, V.N.; Blinova, E.V.; Turovsky, E.A. The selective BDNF overexpression in neurons protects neuroglial networks against OGD and glutamate-induced excitotoxicity. Int. J. Neurosci. 2020, 130, 363–383. [Google Scholar] [CrossRef] [PubMed]
- Schmid, I.; Uittenbogaart, C.; Jamieson, B.D. Live-cell assay for detection of apoptosis by dual-laser flow cytometry using Hoechst 33342 and 7-amino-actino-mycin D. Nat. Protoc. 2007, 2, 187–190. [Google Scholar] [CrossRef] [PubMed]


| Gene Name | Forward Primer 5′→3′ | Reverse Primer 5′→3′ |
|---|---|---|
| GAPDH | ACATCGCTCAGACACCATG | GCCAGTGAGCTTCCCGTT |
| SELENOT | TCTCCTAGTGGCGGCGTC | GTCTATATATTGGTTGAGGGAGG |
| SELENOM | AGCCTCCTGTTGCCTCCGC | AGGTCAGCGTGGTCCGAAG |
| SELENOF | TACGGTTGTTGTTGGCGAC | CAAATTGTGCTTCCTCCTGAC |
| SELENOK | TTTACATCTCGAACGGACAAG | CAGCCTTCCACTTCTTGATG |
| SELENOS | TGGGACAGCATGCAAGAAG | GCGTCCAGGTCTCCAGG |
| SELENON | TGATCTGCCTGCCCAATG | TCAGGAACTGCATGTAGGTGG |
| DIO2 | AGCTTCCTCCTCGATGCC | AAAGGAGGTCAAGTGGCTG |
| GPX1 | CTACTTATCGAGAATGTGGCG | CGAAGAGCATGAAGTTGGG |
| GPX4 | CCATGCACGAGTTTTCCG | AATTTGACGTTGTAGCCCG |
| TXNRD1 | GGTCTGGCAGCTGCTAAGG | TAGCCCCAATTCAAAGAGC |
| TXNRD3 | CCTTTGCTTTGTTGTTTCTGTG | TAGTGAGTGTGAGGGTGAAGC |
| CHOP | GCTCTGATTGACCGAATGG | TCTGGGAAAGGTGGGTAGTG |
| GADD34 | CTCCGAGAAGGTCACTGTCC | GACGAGCGGGAAGGTGTGG |
| PUMA | CAGATATGCGCCCAGAGAT | CCATTCGTGGGTGGTCTTC |
| BIM | GGACGACCTCAACGCACAGTACGAG | GTAAGGGCAGGAGTCCCA |
| ATF–4 | GTGTTCTCTGTGGGTCTGCC | GACCCTTTTCTTCCCCCTTG |
| ATF–6 | AACCCTAGTGTGAGCCCTGC | GTTCAGAGCACCCTGAAGA |
| XBPu | ACTCAGACTACGTGCACCTC | GTCAATACCGCCAGAATCC |
| XBPs | CTGAGTCCGCAGCGGTGCAGG | GGTCCAAGTTGTCCAGAATG |
| CAS–3 | GCATTGAGACAGACAGTGGTG | AATAGAGTTCTTTTGTGAGCATG |
| CAS–4 | CACGCCTGGCTCTCATCATA | TAGCAAATGCCCTCAGCG |
| BAX | GGGCTGGACATTGGACTTC | AACACAGTCCAAGGCAGCTG |
| BAK | GAGAGTGGCATCAATTGGGG | CAGCCACCCCTCTGTGCAATCCA |
| BCL-2 | GGTGAACTGGGGGAGGATTG | AGCCAGGAGAAATCAAACAGAG |
| RIPK1 | GCCATTCAGCTCCTTGCCAC | CAGTTTACGGGCACAGTTTTTC |
| RIPK3 | AGCCCTACCTCAACTGGAAC | CCAGGCTTCAGGATCTTTAGG |
| MLKL | TGAGAAGATCCGCAAGCTGG | TTTGTGCCTCTCCCAGCTTC |
| RAS | AACAAGTGTGACCTGGCTGC | TCCGGCACCTCCATGTCCTG |
| RAF | CCCCAAAGCAATGAAGAGGC | TCAACTAGAAGACAGGCAGCC |
| MAPK1 | AGCAGTATTACGACCCGAGTG | CTGGGAAGAAGAACACCGATG |
| MAPK3 | GCTGGCTCACCCCTACCTG | ATTTTCTAACAGTCTGGCGGG |
| PI3K | CAAATACATTAGGAGCGAAGGC | CAAATACATTAGGAGCGAAGGC |
| mTOR | TTACGACGGCATGGGAATCTC | CAAATACATTAGGAGCGAAGGC |
| KIT | AACGCTCGACTACCTGTGAAG | GCTTGAATGTTGGTCTTTTTAGG |
| FLT3 | AGAAGCGATGTATCAGAATGTG | GTGAAGCAGCAGTTGATAATAG |
| VEGFR | CTGACTCGGAAGGACACGG | CCTGGCGGCACGAAATATCC |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Varlamova, E.G.; Goltyaev, M.V.; Simakin, A.V.; Gudkov, S.V.; Turovsky, E.A. Comparative Analysis of the Cytotoxic Effect of a Complex of Selenium Nanoparticles Doped with Sorafenib, “Naked” Selenium Nanoparticles, and Sorafenib on Human Hepatocyte Carcinoma HepG2 Cells. Int. J. Mol. Sci. 2022, 23, 6641. https://doi.org/10.3390/ijms23126641
Varlamova EG, Goltyaev MV, Simakin AV, Gudkov SV, Turovsky EA. Comparative Analysis of the Cytotoxic Effect of a Complex of Selenium Nanoparticles Doped with Sorafenib, “Naked” Selenium Nanoparticles, and Sorafenib on Human Hepatocyte Carcinoma HepG2 Cells. International Journal of Molecular Sciences. 2022; 23(12):6641. https://doi.org/10.3390/ijms23126641
Chicago/Turabian StyleVarlamova, Elena G., Mikhail V. Goltyaev, Aleksander V. Simakin, Sergey V. Gudkov, and Egor A. Turovsky. 2022. "Comparative Analysis of the Cytotoxic Effect of a Complex of Selenium Nanoparticles Doped with Sorafenib, “Naked” Selenium Nanoparticles, and Sorafenib on Human Hepatocyte Carcinoma HepG2 Cells" International Journal of Molecular Sciences 23, no. 12: 6641. https://doi.org/10.3390/ijms23126641
APA StyleVarlamova, E. G., Goltyaev, M. V., Simakin, A. V., Gudkov, S. V., & Turovsky, E. A. (2022). Comparative Analysis of the Cytotoxic Effect of a Complex of Selenium Nanoparticles Doped with Sorafenib, “Naked” Selenium Nanoparticles, and Sorafenib on Human Hepatocyte Carcinoma HepG2 Cells. International Journal of Molecular Sciences, 23(12), 6641. https://doi.org/10.3390/ijms23126641








