Protease-Activated Receptor 1 in Human Carotid Atheroma Is Significantly Related to Iron Metabolism, Plaque Vulnerability, and the Patient’s Age
Abstract
:1. Introduction
2. Results
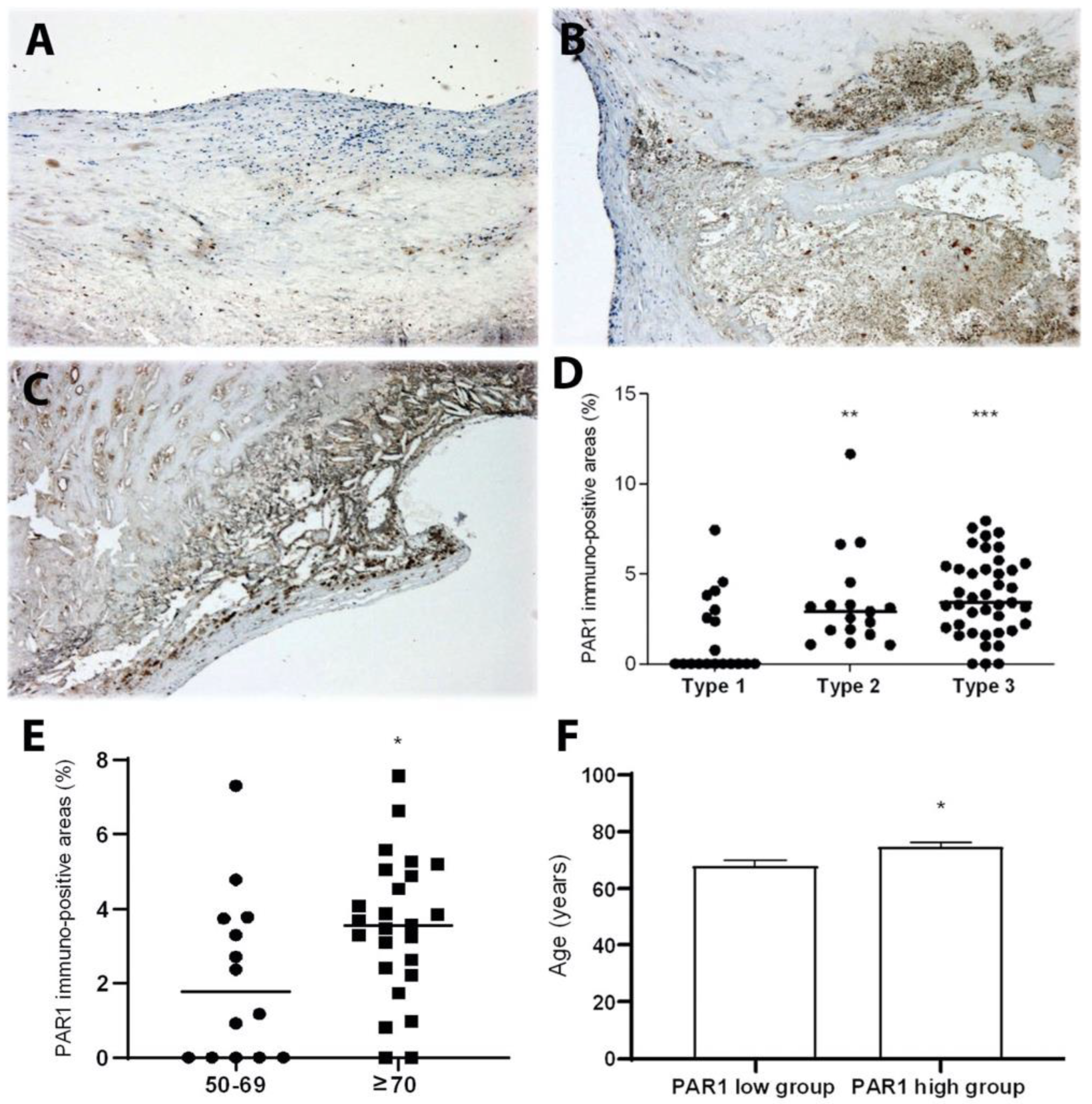
2.1. PAR1 Expression Is Significantly Increased with the Progression of Human Atherosclerotic Plaques and Patient’s Age
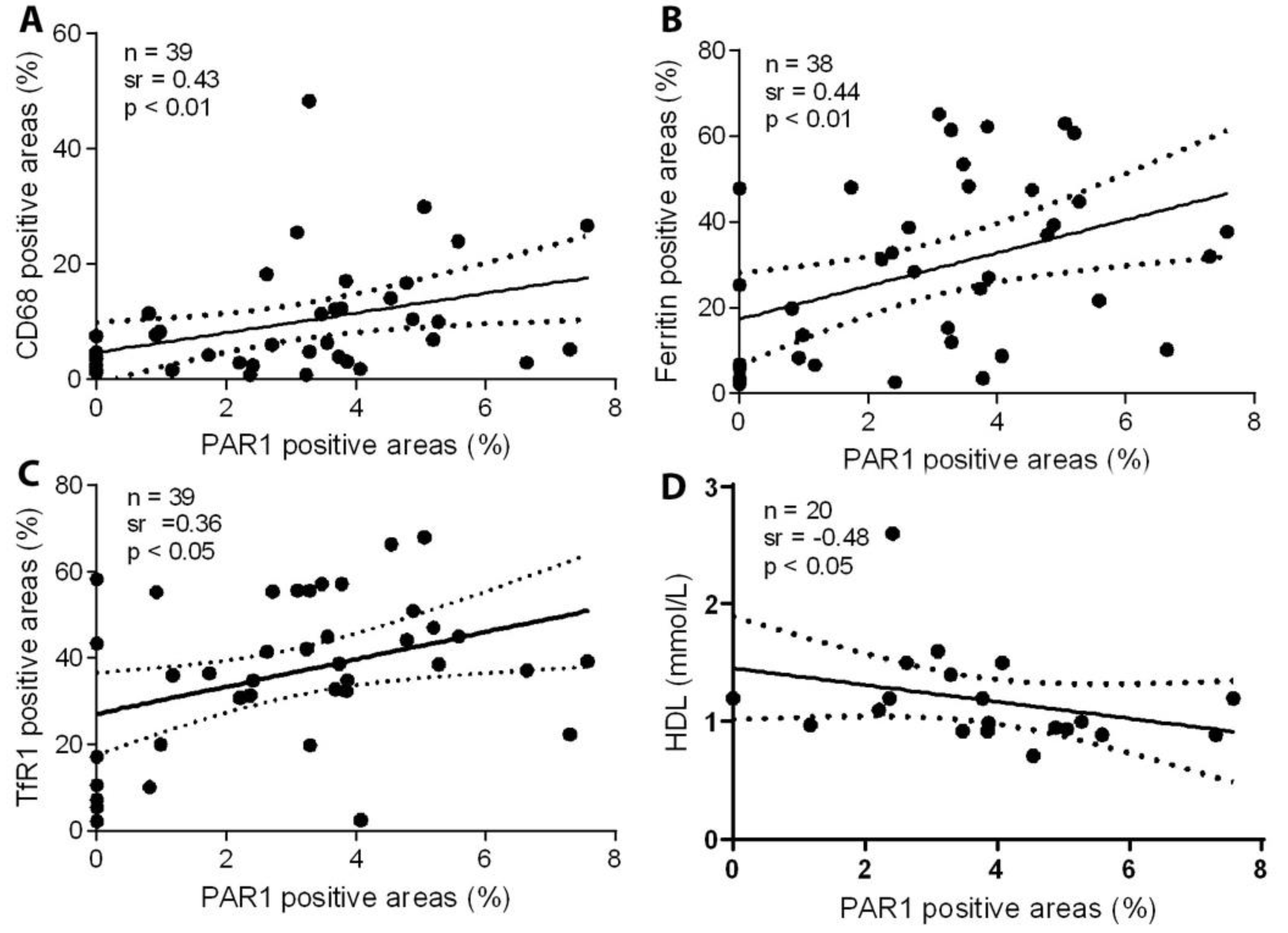
2.2. PAR1 Expression Is Significantly Correlated with Accumulation of CD68-Positive Macrophages, Ferritin, and TfR1, and Inversely Correlated with Levels of HDL
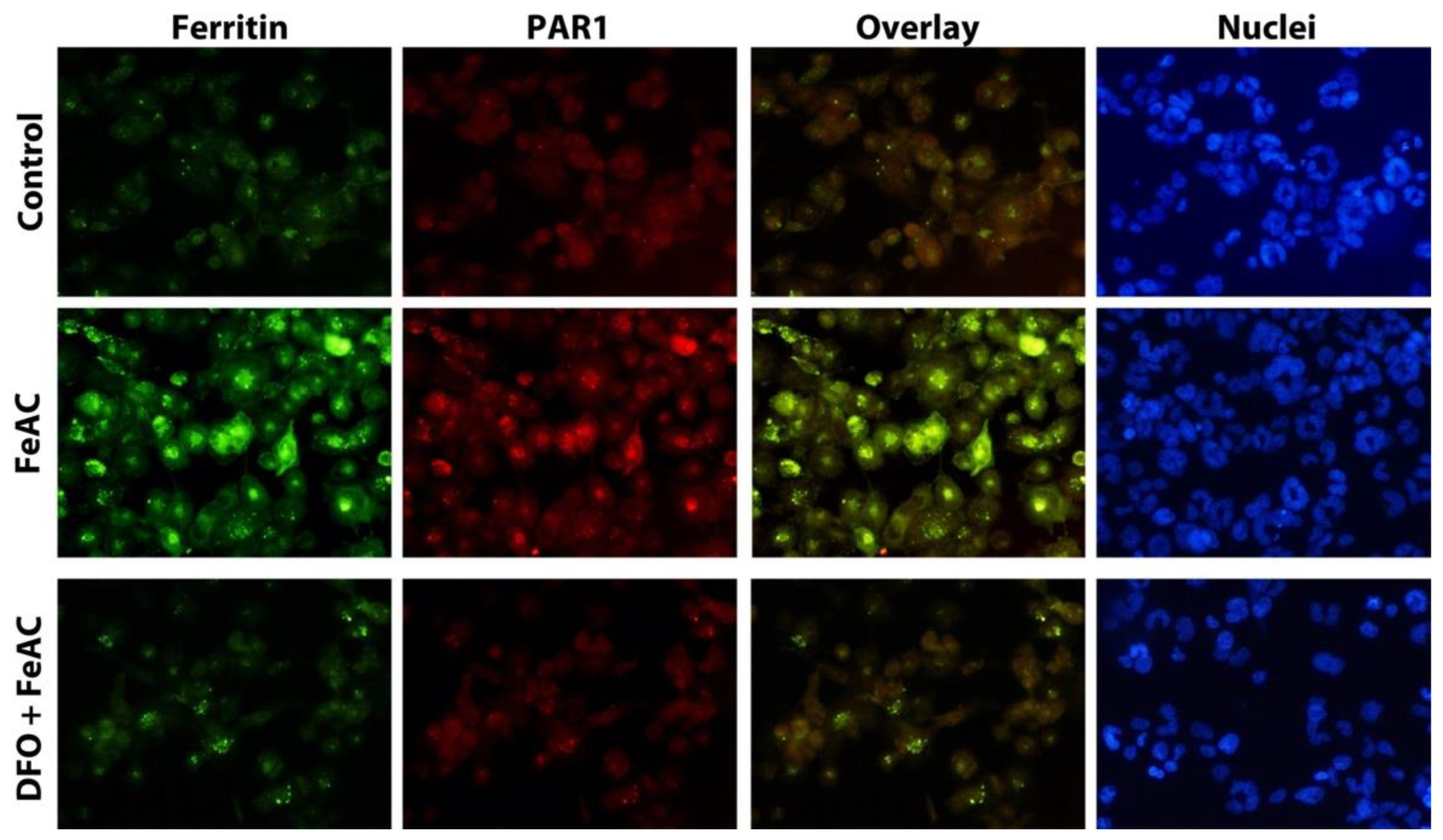
2.3. PAR1 Is Significantly Increased in Macrophages Exposed to Iron and Colocalized with Ferritin Expression
3. Discussion
Limitation
4. Materials and Methods
4.1. Collection of Carotid Artery Samples
4.2. Immunohistochemistry
4.3. Classification of the Plaques
4.4. Cell Cultures and Experimental Conditions
4.5. Double Immunocytochemistry
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Badimon, L.; Padro, T.; Vilahur, G. Atherosclerosis, platelets and thrombosis in acute ischaemic heart disease. Eur. Heart J. Acute Cardiovasc. Care 2012, 1, 60–74. [Google Scholar] [CrossRef]
- Colognato, R.; Slupsky, J.R.; Jendrach, M.; Burysek, L.; Syrovets, T.; Simmet, T. Differential expression and regulation of protease-activated receptors in human peripheral monocytes and monocyte-derived antigen-presenting cells. Blood 2003, 102, 2645–2652. [Google Scholar] [CrossRef] [Green Version]
- Heuberger, D.M.; Schuepbach, R.A. Protease-activated receptors (PARs): Mechanisms of action and potential therapeutic modulators in PAR-driven inflammatory diseases. Thromb. J. 2019, 17, 1–24. [Google Scholar]
- Nelken, N.A.; Soifer, S.J.; O′Keefe, J.; Vu, T.K.; Charo, I.F.; Coughlin, S.R. Thrombin receptor expression in normal and atherosclerotic human arteries. J. Clin. Investig. 1992, 90, 1614–1621. [Google Scholar] [CrossRef]
- Ku, D.D.; Dai, J. Expression of thrombin receptors in human atherosclerotic coronary arteries leads to an exaggerated vasoconstrictory response in vitro. J. Cardiovasc. Pharmacol. 1997, 30, 649–657. [Google Scholar] [CrossRef]
- Rana, R.; Huang, T.; Koukos, G.; Fletcher, E.K.; Turner, S.E.; Shearer, A.; Gurbel, P.A.; Rade, J.J.; Kimmelstiel, C.D.; Bliden, K.P.; et al. Noncanonical matrix metalloprotease 1-protease-activated receptor 1 signaling drives progression of atherosclerosis. Arter. Thromb. Vasc. Biol. 2018, 38, 1368–1380. [Google Scholar] [CrossRef] [Green Version]
- Friebel, J.; Moritz, E.; Witkowski, M.; Jakobs, K.; Strässler, E.; Dörner, A.; Steffens, D.; Puccini, M.; Lammel, S.; Glauben, R.; et al. Pleiotropic effects of the protease-activated receptor 1 (PAR1) inhibitor, vorapaxar, on atherosclerosis and vascular inflammation. Cells 2021, 10, 3517. [Google Scholar] [CrossRef]
- Yuan, X.M.; Ward, L.J.; Forssell, C.; Siraj, N.; Li, W. Carotid atheroma from men has significantly higher levels of inflammation and iron metabolism enabled by macrophages. Stroke 2018, 49, 419–425. [Google Scholar] [CrossRef]
- Li, W.; Xu, L.H.; Forssell, C.; Sullivan, J.L.; Yuan, X.M. Overexpression of transferrin receptor and ferritin related to clinical symptoms and destabilization of human carotid plaques. Exp. Biol. Med. 2008, 233, 818–826. [Google Scholar] [CrossRef]
- Cheng, Y.; Xi, G.; Jin, H.; Keep, R.F.; Feng, J.; Hua, Y. Thrombin-induced cerebral hemorrhage: Role of protease-activated receptor-1. Transl. Stroke Res. 2014, 5, 472–475. [Google Scholar] [CrossRef] [Green Version]
- Yuan, X.M.; Olsson, A.G.; Brunk, U.T. Macrophage erythrophagocytosis and iron exocytosis. Redox Rep. 1996, 2, 9–17. [Google Scholar] [CrossRef]
- Lege, A.J.; Covic, L.; Kuliopulos, A. Protease-activated receptors in cardiovascular diseases. Circulation 2006, 114, 1070–1077. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, X.; Yu, J.; Song, S.; Yue, X.; Li, Q. Protease-activated receptor-1 (PAR-1): A promising molecular target for cancer. Oncotarget 2017, 8, 107334–107345. [Google Scholar] [CrossRef] [Green Version]
- Tang, X.; Zhang, Z.; Fang, M.; Han, Y.; Wang, G.; Wang, S.; Xue, M.; Li, Y.; Zhang, L.; Wu, J.; et al. Transferrin plays a central role in coagulation balance by interacting with clotting factors. Cell Res. 2020, 30, 119–132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Olejarz, W.; Łacheta, D.; Kubiak-Tomaszewska, G. Matrix metalloproteinases as biomarkers of atherosclerotic plaque instability. Int. J. Mol. Sci. 2020, 21, 3946. [Google Scholar] [CrossRef] [PubMed]
- Pavlic, V.; Peric, D.; Kalezic, I.S.; Madi, M.; Bhat, S.G.; Brkic, Z.; Staletovic, D. Identification of periopathogens in atheromatous plaques obtained from carotid and coronary arteries. Biomed. Res. Int. 2021, 2021, 9986375. [Google Scholar] [CrossRef]
- Day, S.M.; Duquaine, D.; Mundada, L.V.; Menon, R.G.; Khan, B.V.; Rajagopalan, S.; Fay, W.P. Chronic iron administration increases vascular oxidative stress and accelerates arterial thrombosis. Circulation 2003, 107, 2601–2606. [Google Scholar] [CrossRef]
- Yuan, X.M.; Anders, W.L.; Olsson, A.G.; Brunk, U.T. Iron in human atheroma and LDL oxidation by macrophages following erythrophagocytosis. Atherosclerosis 1996, 124, 61–73. [Google Scholar] [CrossRef]
- Li, W.; Ostblom, M.; Xu, L.H.; Hellsten, A.; Leanderson, P.; Liedberg, B.; Brunk, U.T.; Eaton, J.W.; Yuan, X.M. Cytocidal effects of atheromatous plaque components: The death zone revisited. FASEB J. 2006, 20, 2281–2290. [Google Scholar] [CrossRef] [Green Version]
- Seehafer, S.S.; Pearce, D.A. You say lipofuscin, we say ceroid: Defining autofluorescent storage material. Neurobiol. Aging. 2006, 27, 576–588. [Google Scholar] [CrossRef] [PubMed]
- Ward, L.J.; Olausson, P.; Li, W.; Yuan, X.M. Proteomics and multivariate modelling reveal sex-specific alterations in distinct regions of human carotid atheroma. Biol. Sex. Differ. 2018, 9, 54. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kopriva, D.; Kisheev, A.; Meena, D.; Pelle, S.; Karnitsky, M.; Lavoie, A.; Buttigieg, J. The nature of iron deposits differs between symptomatic and asymptomatic carotid atherosclerotic plaques. PLoS ONE 2015, 10, e0143138. [Google Scholar] [CrossRef] [PubMed]
- Horvat, R.; Palade, G.E. The functional thrombin receptor is associated with the plasmalemma and a large endosomal network in cultured human umbilical vein endothelial cells. J. Cell Sci. 1995, 108, 1155–1164. [Google Scholar] [CrossRef] [PubMed]
- Xepapadaki, E.; Zvintzou, E.; Kalogeropoulou, C.; Filou, S.; Kypreos, K.E. Τhe antioxidant function of HDL in atherosclerosis. Angiology 2020, 71, 112–121. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, W.; Osman, E.; Forssell, C.; Yuan, X.-M. Protease-Activated Receptor 1 in Human Carotid Atheroma Is Significantly Related to Iron Metabolism, Plaque Vulnerability, and the Patient’s Age. Int. J. Mol. Sci. 2022, 23, 6363. https://doi.org/10.3390/ijms23126363
Li W, Osman E, Forssell C, Yuan X-M. Protease-Activated Receptor 1 in Human Carotid Atheroma Is Significantly Related to Iron Metabolism, Plaque Vulnerability, and the Patient’s Age. International Journal of Molecular Sciences. 2022; 23(12):6363. https://doi.org/10.3390/ijms23126363
Chicago/Turabian StyleLi, Wei, Ehab Osman, Claes Forssell, and Xi-Ming Yuan. 2022. "Protease-Activated Receptor 1 in Human Carotid Atheroma Is Significantly Related to Iron Metabolism, Plaque Vulnerability, and the Patient’s Age" International Journal of Molecular Sciences 23, no. 12: 6363. https://doi.org/10.3390/ijms23126363
APA StyleLi, W., Osman, E., Forssell, C., & Yuan, X.-M. (2022). Protease-Activated Receptor 1 in Human Carotid Atheroma Is Significantly Related to Iron Metabolism, Plaque Vulnerability, and the Patient’s Age. International Journal of Molecular Sciences, 23(12), 6363. https://doi.org/10.3390/ijms23126363






