Proteomics of Cryptococcus neoformans: From the Lab to the Clinic
Abstract
:1. Introduction
2. Defining the Fungal Pathogen—Cryptococcus spp.
2.1. Distribution and Disease
2.2. Virulence Factors
2.3. Host Immune Response
2.4. Antifungal Treatment and Resistance
3. Proteomic Profiling of Cryptococcal Infection In Vitro
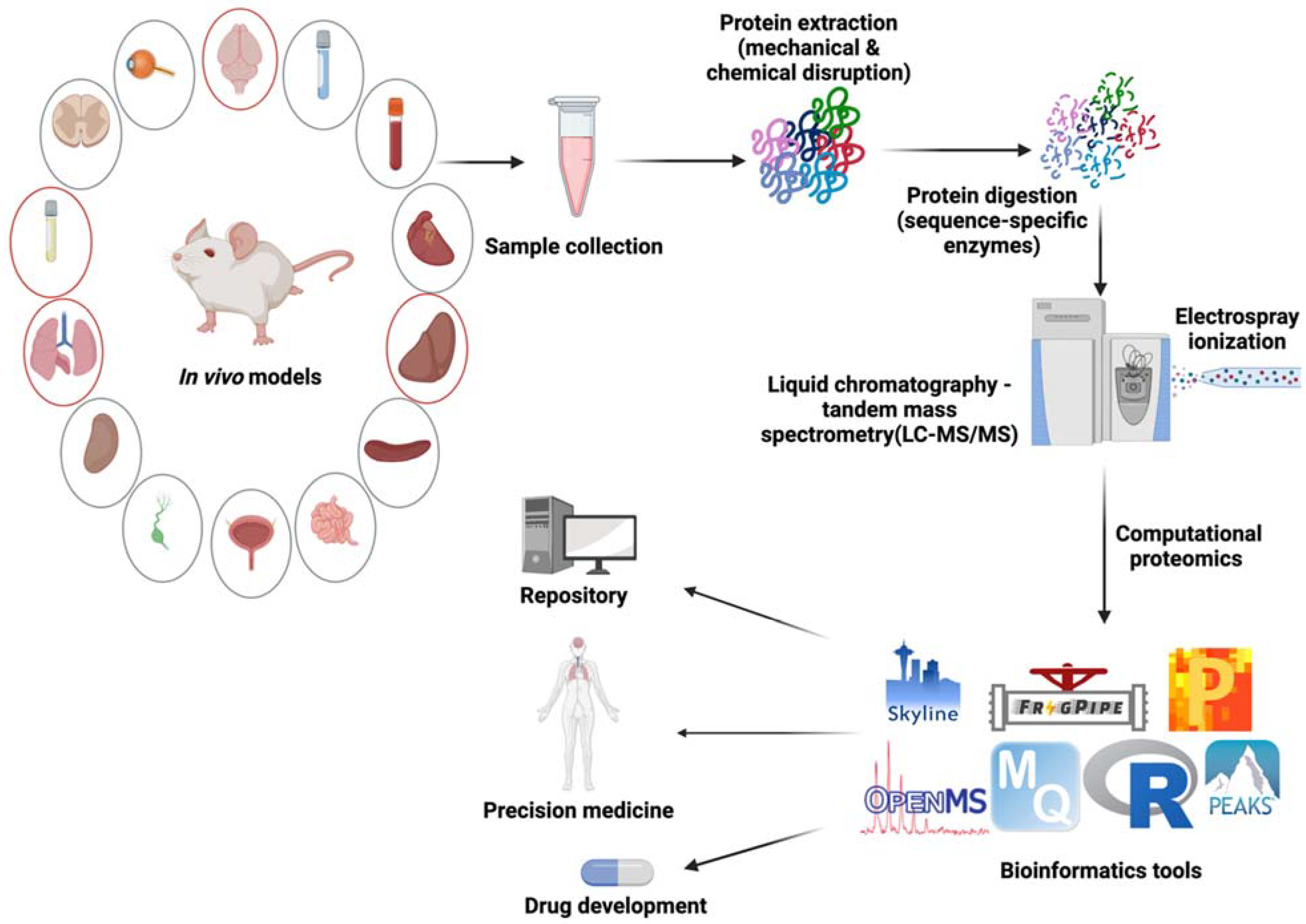
4. In Vivo Modelling of Cryptococcosis
5. Assessing Proteome Changes of Fungal Infection In Vivo
6. Future Directions and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bongomin, F.; Gago, S.; Oladele, R.; Denning, D. Global and Multi-National Prevalence of Fungal Diseases—Estimate Precision. J. Fungi 2017, 3, 57. [Google Scholar] [CrossRef]
- Dufresne, S.F.; Cole, D.C.; Denning, D.W.; Sheppard, D.C. Serious fungal infections in Canada. Eur. J. Clin. Microbiol. Infect. Dis. 2017, 36, 987–992. [Google Scholar] [CrossRef] [Green Version]
- Martin, G.S.; Mannino, D.M.; Eaton, S.; Moss, M. The Epidemiology of Sepsis in the United States from 1979 through 2000. N. Engl. J. Med. 2003, 348, 1546–1554. [Google Scholar] [CrossRef] [Green Version]
- Kronstad, J.W.; Attarian, R.; Cadieux, B.; Choi, J.; D’Souza, C.A.; Griffiths, E.J.; Geddes, J.M.H.; Hu, G.; Jung, W.H.; Kretschmer, M.; et al. Expanding fungal pathogenesis: Cryptococcus breaks out of the opportunistic box. Nat. Rev. Microbiol. 2011, 9, 193–203. [Google Scholar] [CrossRef] [Green Version]
- Perfect, J.R. The antifungal pipeline: A reality check. Nat. Rev. Drug Discov. 2017, 16, 603–616. [Google Scholar] [CrossRef] [Green Version]
- Geddes-McAlister, J.; Shapiro, R.S. New pathogens, new tricks: Emerging, drug-resistant fungal pathogens and future prospects for antifungal therapeutics. Ann. N. Y. Acad. Sci. 2019, 1435, 57–78. [Google Scholar] [CrossRef]
- Bermas, A.; Geddes-McAlister, J. Combatting the evolution of anti-fungal resistance in Cryptococcus neoformans. Mol. Microbiol. 2020, 114, 721–734. [Google Scholar] [CrossRef]
- Ball, B.; Bermas, A.; Carruthers-Lay, D.; Geddes-McAlister, J. Mass Spectrometry-Based Proteomics of Fungal Pathogenesis, Host–Fungal Interactions, and Antifungal Development. J. Fungi 2019, 5, 52. [Google Scholar] [CrossRef] [Green Version]
- Tran, J.C.; Zamdborg, L.; Ahlf, D.R.; Lee, J.E.; Catherman, A.D.; Durbin, K.R.; Tipton, J.D.; Vellaichamy, A.; Kellie, J.F.; Li, M.; et al. Mapping intact protein isoforms in discovery mode using top-down proteomics. Nature 2011, 480, 254–258. [Google Scholar] [CrossRef] [Green Version]
- Aebersold, R.; Mann, M. Mass-spectrometric exploration of proteome structure and function. Nature 2016, 537, 347–355. [Google Scholar] [CrossRef]
- Ankney, J.A.; Muneer, A.; Chen, X. Relative and Absolute Quantitation in Mass Spectrometry–Based Proteomics. Annu. Rev. Anal. Chem. 2018, 11, 49–77. [Google Scholar] [CrossRef]
- Zhu, Y.; Aebersold, R.; Mann, M.; Guo, T. SnapShot: Clinical proteomics. Cell 2021, 184, 4840. [Google Scholar] [CrossRef]
- Jannetto, P.J.; Fitzgerald, R.L. Effective Use of Mass Spectrometry in the Clinical Laboratory. Clin. Chem. 2016, 62, 92–98. [Google Scholar] [CrossRef] [Green Version]
- Fu, Q.; Chen, Z.; Zhang, S.; Parker, S.J.; Fu, Z.; Tin, A.; Liu, X.; Van Eyk, J.E. Multiple and Selective Reaction Monitoring Using Triple Quadrupole Mass Spectrometer: Preclinical Large Cohort Analysis. Methods Mol. Biol. 2016, 1410, 249–264. [Google Scholar] [CrossRef]
- Geyer, P.E.; Kulak, N.A.; Pichler, G.; Holdt, L.M.; Teupser, D.; Mann, M. Plasma Proteome Profiling to Assess Human Health and Disease. Cell Syst. 2016, 2, 185–195. [Google Scholar] [CrossRef] [Green Version]
- Zhang, S.; Raedschelders, K.; Venkatraman, V.; Huang, L.; Holewinski, R.; Fu, Q.; Van Eyk, J.E. A Dual Workflow to Improve the Proteomic Coverage in Plasma Using Data-Independent Acquisition-MS. J. Proteome Res. 2020, 19, 2828–2837. [Google Scholar] [CrossRef]
- Macklin, A.; Khan, S.; Kislinger, T. Recent advances in mass spectrometry based clinical proteomics: Applications to cancer research. Clin. Proteom. 2020, 17, 17. [Google Scholar] [CrossRef]
- Hughes, C.S.; Morin, G.B. Using Public Data for Comparative Proteome Analysis in Precision Medicine Programs. Proteom. Clin. Appl. 2018, 12, 1600179. [Google Scholar] [CrossRef]
- Retanal, C.; Ball, B.; Geddes-McAlister, J. Post-Translational Modifications Drive Success and Failure of Fungal–Host Interactions. J. Fungi 2021, 7, 124. [Google Scholar] [CrossRef]
- Meissner, F.; Geddes-McAlister, J.; Mann, M.; Bantscheff, M. The emerging role of mass spectromerty based proteomics in drug discovery. Nat. Rev. Drug Discov. 2021, in press. [Google Scholar]
- Ball, B.; Woroszchuk, E.; Sukumaran, A.; Afaq, A.; Carruthers-Lay, D.; Gee, L.; Langille, M.; Geddes-McAlister, J. Zinc availability alters the proteome and secretome of Cryptococcus neoformans, identifying Wos2 as a subtle influencer of fungal virulence. BMC Microbiol. 2021, in press. [Google Scholar]
- Blum, B.C.; Mousavi, F.; Emili, A. Single-platform “multi-omic” profiling: Unified mass spectrometry and computational workflows for integrative proteomics-metabolomics analysis. Mol. Omics 2018, 14, 307–319. [Google Scholar] [CrossRef] [Green Version]
- Coman, C.; Solari, F.A.; Hentschel, A.; Sickmann, A.; Zahedi, R.P.; Ahrends, R. Simultaneous metabolite, protein, lipid extraction (SIMPLEX): A combinatorial multimolecular omics approach for systems biology. Mol. Cell. Proteom. 2016, 15, 1453–1466. [Google Scholar] [CrossRef] [Green Version]
- Cox, J.; Mann, M. MaxQuant enables high peptide identification rates, individualized p.p.b.-range mass accuracies and proteome-wide protein quantification. Nat. Biotechnol. 2008, 26, 1367–1372. [Google Scholar] [CrossRef]
- Tyanova, S.; Temu, T.; Sinitcyn, P.; Carlson, A.; Hein, M.Y.; Geiger, T.; Mann, M.; Cox, J. The Perseus computational platform for comprehensive analysis of (prote)omics data. Nat. Methods 2016, 13, 731–740. [Google Scholar] [CrossRef]
- Röst, H.L.; Sachsenberg, T.; Aiche, S.; Bielow, C.; Weisser, H.; Aicheler, F.; Andreotti, S.; Ehrlich, H.C.; Gutenbrunner, P.; Kenar, E.; et al. OpenMS: A flexible open-source software platform for mass spectrometry data analysis. Nat. Methods 2016, 13, 741–748. [Google Scholar] [CrossRef]
- MacLean, B.; Tomazela, D.M.; Shulman, N.; Chambers, M.; Finney, G.L.; Frewen, B.; Kern, R.; Tabb, D.L.; Liebler, D.C.; MacCoss, M.J. Skyline: An open source document editor for creating and analyzing targeted proteomics experiments. Bioinformatics 2010, 26, 966–968. [Google Scholar] [CrossRef] [Green Version]
- Werness, S.A.; Anderson, D.J. R-Studio User Manual; R-Tools Technology Inc.: Richmond Hill, ON, Canada, 2011; ISBN 3032783534. [Google Scholar]
- Kong, A.T.; Leprevost, F.V.; Avtonomov, D.M.; Mellacheruvu, D.; Nesvizhskii, A.I. MSFragger: Ultrafast and comprehensive peptide identification in mass spectrometry–based proteomics. Nat. Methods 2017, 14, 513–520. [Google Scholar] [CrossRef] [Green Version]
- Rajasingham, R.; Smith, R.M.; Park, B.J.; Jarvis, J.N.; Govender, N.P.; Chiller, T.M.; Denning, D.W.; Loyse, A.; Boulware, D.R. Global burden of disease of HIV-associated cryptococcal meningitis: An updated analysis. Lancet Infect. Dis. 2017, 17, 873–881. [Google Scholar] [CrossRef] [Green Version]
- Datta, K.; Bartlett, K.H.; Baer, R.; Byrnes, E.; Galanis, E.; Heitman, J.; Hoang, L.; Leslie, M.J.; MacDougall, L.; Magill, S.S.; et al. Spread of Cryptococcus gattii into Pacific Northwest Region of the United States. Emerg. Infect. Dis. 2009, 15, 1185. [Google Scholar] [CrossRef]
- May, R.C.; Stone, N.R.H.; Wiesner, D.L.; Bicanic, T.; Nielsen, K. Cryptococcus: From environmental saprophyte to global pathogen. Nat. Rev. Microbiol. 2016, 14, 106–117. [Google Scholar] [CrossRef]
- Dromer, F.; Levitz, S.M. Invasion of Cryptococcus into the Central Nervous System. 2014. Available online: https://www.researchgate.net/publication/344112827_Invasion_of_Cryptococcus_into_the_Central_Nervous_System (accessed on 14 November 2021).
- Chang, Y.C.; Stins, M.F.; McCaffery, M.J.; Miller, G.F.; Pare, D.R.; Dam, T.; Paul-Satyasee, M.; Kim, K.S.; Kwon-Chung, K.J. Cryptococcal yeast cells invade the central nervous system via transcellular penetration of the blood-brain barrier. Infect. Immun. 2004, 72, 4985–4995. [Google Scholar] [CrossRef] [Green Version]
- Warkentien, T.; Crum-Cianflone, N.F. An Update on Cryptococcosis among HIV-Infected Persons. Int. J. STD AIDS 2011, 21, 679–684. [Google Scholar] [CrossRef] [Green Version]
- Voelz, K.; May, R.C. Cryptococcal interactions with the host immune system. Eukaryot. Cell 2010, 9, 835–846. [Google Scholar] [CrossRef] [Green Version]
- Setianingrum, F.; Rautemaa-Richardson, R.; Denning, D.W. Pulmonary cryptococcosis: A review of pathobiology and clinical aspects. Med. Mycol. 2019, 57, 133–150. [Google Scholar] [CrossRef]
- Brown, G.D. Innate antifungal immunity. Annu. Rev. Immunol. 2012, 29, 1–21. [Google Scholar] [CrossRef] [Green Version]
- Mansour, M.K.; Reedy, J.L.; Tam, J.M.; Vyas, J.M. Macrophage-Cryptococcus interactions: An update. Curr. Fungal Infect. Rep. 2014, 8, 109–115. [Google Scholar] [CrossRef]
- Chen, G.H.; Olszewski, M.A.; McDonald, R.A.; Wells, J.C.; Paine, R.; Huffnagle, G.B.; Toews, G.B. Role of granulocyte macrophage colony-stimulating factor in host defense against pulmonary Cryptococcus neoformans infection during murine allergic bronchopulmonary mycosis. Am. J. Pathol. 2007, 170, 1028–1040. [Google Scholar] [CrossRef] [Green Version]
- Capilla, J.; Maffei, C.M.L.; Clemons, K.V.; Sobel, R.A.; Stevens, D.A. Experimental systemic infection with Cryptococcus neoformans var. grubii and Cryptococcus gattii in normal and immunodeficient mice. Med. Mycol. 2006, 44, 601–610. [Google Scholar] [CrossRef] [Green Version]
- Normile, T.G.; Bryan, A.M.; Del Poeta, M. Animal Models of Cryptococcus neoformans in Identifying Immune Parameters Associated With Primary Infection and Reactivation of Latent Infection. Front. Immunol. 2020, 11, 2197. [Google Scholar] [CrossRef]
- Geddes, J.M.H.; Caza, M.; Croll, D.; Stoynov, N.; Foster, L.J.; Kronstad, J.W. Analysis of the protein kinase a-regulated proteome of Cryptococcus neoformans identifies a role for the ubiquitin-proteasome pathway in capsule formation. MBio 2016, 7, e01862-15. [Google Scholar] [CrossRef] [Green Version]
- Boersema, P.J.; Aye, T.T.; Van Veen, T.A.B.; Heck, A.J.R.; Mohammed, S. Triplex protein quantification based on stable isotope labeling by peptide dimethylation applied to cell and tissue lysates. Proteomics 2008, 8, 4624–4632. [Google Scholar] [CrossRef]
- Cox, J.; Neuhauser, N.; Michalski, A.; Scheltema, R.A.; Olsen, J.V.; Mann, M. Andromeda: A peptide search engine integrated into the MaxQuant environment. J. Proteome Res. 2011, 10, 1794–1805. [Google Scholar] [CrossRef]
- Szklarczyk, D.; Gable, A.L.; Lyon, D.; Junge, A.; Wyder, S.; Huerta-Cepas, J.; Simonovic, M.; Doncheva, N.T.; Morris, J.H.; Bork, P.; et al. STRING v11: Protein-protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res. 2019, 47, D607–D613. [Google Scholar] [CrossRef] [Green Version]
- Geddes, J.M.H.; Croll, D.; Caza, M.; Stoynov, N.; Foster, L.J.; Kronstad, J.W. Secretome profiling of Cryptococcus neoformans reveals regulation of a subset of virulence-associated proteins and potential biomarkers by protein kinase A. BMC Microbiol. 2015, 15, 1–26. [Google Scholar] [CrossRef] [Green Version]
- Rodrigues, M.L.; Nakayasu, E.S.; Almeida, I.C.; Nimrichter, L. The impact of proteomics on the understanding of functions and biogenesis of fungal extracellular vesicles. J. Proteom. 2014, 97, 177–186. [Google Scholar] [CrossRef] [Green Version]
- Rodrigues, M.L.; Nakayasu, E.S.; Oliveira, D.L.; Nimrichter, L.; Nosanchuk, J.D.; Almeida, I.C.; Casadevall, A. Extracellular vesicles produced by Cryptococcus neoformans contain protein components associated with virulence. Eukaryot. Cell 2008, 7, 58–67. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.; Zhang, K.; Li, H.; Coelho, C.; Gonçalves, D.S.; Fu, S.; Li, X.; Nakayasu, E.S.; Kim, Y.-M.; Liao, W.; et al. Cryptococcus neoformans- Infected Macrophages Release Proin fl ammatory Extracellular Vesicles: Insight into Their Components by Mulit-OMICS. MBio 2021, 12, e00279-21. [Google Scholar] [CrossRef]
- Nakayasu, E.S.; Nicora, C.D.; Sims, A.C.; Burnum-Johnson, K.E.; Kim, Y.-M.; Kyle, J.E.; Matzke, M.M.; Shukla, A.K.; Chu, R.K.; Schepmoes, A.A.; et al. MPLEx: A Robust and Universal Protocol for Single-Sample Integrative Proteomic, Metabolomic, and Lipidomic Analyses. MSystems 2016, 1, e00043-16. [Google Scholar] [CrossRef] [Green Version]
- Santi, L.; Beys-Da-Silva, W.O.; Berger, M.; Calzolari, D.; Guimarães, J.A.; Moresco, J.J.; Yates, J.R. Proteomic profile of Cryptococcus neoformans biofilm reveals changes in metabolic processes. J. Proteome Res. 2014, 13, 1545–1559. [Google Scholar] [CrossRef]
- Eng, J.K.; McCormack, A.L.; Yates, J.R. An Approach to correlate Tandem Mass Spectral Data of Peptides with Amino Acid Sequences in a Protien Database. J. Am. Soc. Mass Spectrom. 1994, 5, 976–989. [Google Scholar] [CrossRef] [Green Version]
- Schirmer, E.C.; Yates, J.R., 3rd; Gerace, L. MudPIT: A powerful proteomics tool for discovery. Discov. Med. 2003, 3, 38–39. [Google Scholar]
- Selvan, L.D.N.; Renuse, S.; Kaviyil, J.E.; Sharma, J.; Pinto, S.M.; Yelamanchi, S.D.; Puttamallesh, V.N.; Ravikumar, R.; Pandey, A.; Prasad, T.S.K.; et al. Phosphoproteome of Cryptococcus neoformans. J. Proteom. 2014, 97, 287–295. [Google Scholar] [CrossRef]
- Thingholm, T.E.; Jensen, O.N. Enrichment and characterization of phosphopeptides by immobilized metal affinity chromatography (IMAC) and mass spectrometry. Methods Mol. Biol. 2009, 527, 47–56. [Google Scholar]
- Pinkse, M.W.H.; Uitto, P.M.; Hilhorst, M.J.; Ooms, B.; Heck, A.J.R. Selective isolation at the femtomole level of phosphopeptides from proteolytic digests using 2D-NanoLC-ESI-MS/MS and titanium oxide precolumns. Anal. Chem. 2004, 76, 3935–3943. [Google Scholar] [CrossRef]
- Pandey, A.; Ding, S.L.; Qin, Q.M.; Gupta, R.; Gomez, G.; Lin, F.; Feng, X.; Fachini da Costa, L.; Chaki, S.P.; Katepalli, M.; et al. Global Reprogramming of Host Kinase Signaling in Response to Fungal Infection. Cell Host Microbe 2017, 21, 637–649. [Google Scholar] [CrossRef]
- Jung, C.H.; Jun, C.B.; Ro, S.H.; Kim, Y.M.; Otto, N.M.; Cao, J.; Kundu, M.; Kim, D.H. ULK-Atg13-FIP200 complexes mediate mTOR signaling to the autophagy machinery. Mol. Biol. Cell 2009, 20, 1992–2003. [Google Scholar] [CrossRef] [Green Version]
- Hurley, J.H.; Young, L.N. Mechanisms of Autophagy Initiation. Annu. Rev. Biochem. 2017, 86, 225–244. [Google Scholar] [CrossRef]
- Sun, X.; Chiu, J.-F.; He, Q.-Y. Application of immobilized metal affinity chromatography in proteomics. Expert Rev. Proteom. 2005, 2, 649–657. [Google Scholar] [CrossRef]
- Feng, S.; Ye, M.; Zhou, H.; Jiang, X.; Jiang, X.; Zou, H.; Gong, B. Immobilized Zirconium Ion Affinity Chromatography for Specific Enrichment of Phosphopeptides in Phosphoproteome Analysis. Mol. Cell. Proteom. 2007, 6, 1656–1665. [Google Scholar] [CrossRef] [Green Version]
- Mukaremera, L.; McDonald, T.R.; Nielsen, J.N.; Molenaar, C.J.; Akampurira, A.; Schutz, C.; Taseera, K.; Muzoora, C.; Meintjes, G.; Meya, D.B.; et al. The mouse inhalation model of Cryptococcus neoformans infection recapitulates strain virulence in humans and shows that closely related strains can possess differential virulence. Infect. Immun. 2019, 87, e00046-19. [Google Scholar] [CrossRef] [Green Version]
- Desnos-Ollivier, M.; Patel, S.; Spaulding, A.R.; Charlier, C.; Garcia-Hermoso, D.; Nielsen, K.; Dromer, F. Mixed infections and in vivo evolution in the human fungal pathogen Cryptococcus neoformans. MBio 2010, 1, e00091-10. [Google Scholar] [CrossRef] [Green Version]
- Lortholary, O.; Improvisi, L.; Fitting, C.; Cavaillon, J.M.; Dromer, F. Influence of gender and age on course of infection and cytokine responses in mice with disseminated Cryptococcus neoformans infection. Clin. Microbiol. Infect. 2002, 8, 31–37. [Google Scholar] [CrossRef] [Green Version]
- Coelho, C.; Camacho, E.; Salas, A.; Alanio, A.; Casadevall, A. Intranasal Inoculation of Cryptococcus neoformans in Mice Produces Nasal Infection with Rapid Brain Dissemination. mSphere 2019, 4, e00483-19. [Google Scholar] [CrossRef] [Green Version]
- Wozniak, K.L.; Vyas, J.M.; Levitz, S.M. In vivo role of dendritic cells in a murine model of pulmonary cryptococcosis. Infect. Immun. 2006, 74, 3817–3824. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Li, Y.; Sun, T.; Du, W.; Zhang, Z.; Li, D.; Ding, C. Integrative Proteome and Acetylome Analyses of Murine Responses to Cryptococcus neoformans Infection. Front. Microbiol. 2020, 11, 575. [Google Scholar] [CrossRef] [Green Version]
- Selbach, M.; Schwanhäusser, B.; Thierfelder, N.; Fang, Z.; Khanin, R.; Rajewsky, N. Widespread changes in protein synthesis induced by microRNAs. Nature 2008, 455, 58–63. [Google Scholar] [CrossRef]
- Buccitelli, C.; Selbach, M. mRNAs, proteins and the emerging principles of gene expression control. Nat. Rev. Genet. 2020, 21, 630–644. [Google Scholar] [CrossRef]
- Rosa, R.L.; Berger, M.; Santi, L.; Driemeier, D.; Barros Terraciano, P.; Campos, A.R.; Guimarães, J.A.; Vainstein, M.H.; Yates, J.R.; Beys-Da-Silva, W.O. Proteomics of Rat Lungs Infected by Cryptococcus gattii Reveals a Potential Warburg-like Effect. J. Proteome Res. 2019, 18, 3885–3895. [Google Scholar] [CrossRef]
- Liberti, M.V.; Locasale, J.W. The Warburg Effect: How Does it Benefit Cancer Cells? Trends Biochem. Sci. 2016, 41, 211–218. [Google Scholar] [CrossRef] [Green Version]
- Selvan, L.D.N.; Sreenivasamurthy, S.K.; Kumar, S.; Yelamanchi, S.D.; Madugundu, A.K.; Anil, A.K.; Renuse, S.; Nair, B.G.; Gowda, H.; Mathur, P.P.; et al. Characterization of host response to Cryptococcus neoformans through quantitative proteomic analysis of cryptococcal meningitis co-infected with HIV. Mol. Biosyst. 2015, 11, 2529–2540. [Google Scholar] [CrossRef]
- Mann, S.P.; Treit, P.V.; Geyer, P.E.; Omenn, G.S.; Mann, M. Ethical Principles, Constraints, and Opportunities in Clinical Proteomics. Mol. Cell. Proteom. 2021, 20, 100046. [Google Scholar] [CrossRef]
- Bandeira, N.; Deutsch, E.W.; Kohlbacher, O.; Martens, L.; Vizcaíno, J.A. Data Management of Sensitive Human Proteomics Data: Current Practices, Recommendations, and Perspectives for the Future. Mol. Cell. Proteom. 2021, 20, 100071. [Google Scholar] [CrossRef]
- Kabanda, T.; Siedner, M.J.; Klausner, J.D.; Muzoora, C.; Boulware, D.R. Point-of-care diagnosis and prognostication of cryptococcal meningitis with the cryptococcal antigen lateral flow assay on cerebrospinal fluid. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2014, 58, 113–116. [Google Scholar] [CrossRef] [Green Version]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muselius, B.; Durand, S.-L.; Geddes-McAlister, J. Proteomics of Cryptococcus neoformans: From the Lab to the Clinic. Int. J. Mol. Sci. 2021, 22, 12390. https://doi.org/10.3390/ijms222212390
Muselius B, Durand S-L, Geddes-McAlister J. Proteomics of Cryptococcus neoformans: From the Lab to the Clinic. International Journal of Molecular Sciences. 2021; 22(22):12390. https://doi.org/10.3390/ijms222212390
Chicago/Turabian StyleMuselius, Ben, Shay-Lynn Durand, and Jennifer Geddes-McAlister. 2021. "Proteomics of Cryptococcus neoformans: From the Lab to the Clinic" International Journal of Molecular Sciences 22, no. 22: 12390. https://doi.org/10.3390/ijms222212390
APA StyleMuselius, B., Durand, S.-L., & Geddes-McAlister, J. (2021). Proteomics of Cryptococcus neoformans: From the Lab to the Clinic. International Journal of Molecular Sciences, 22(22), 12390. https://doi.org/10.3390/ijms222212390






