Identification and Affinity Determination of Protein-Antibody and Protein-Aptamer Epitopes by Biosensor-Mass Spectrometry Combination †
Abstract
1. Introduction
2. Results and Discussion
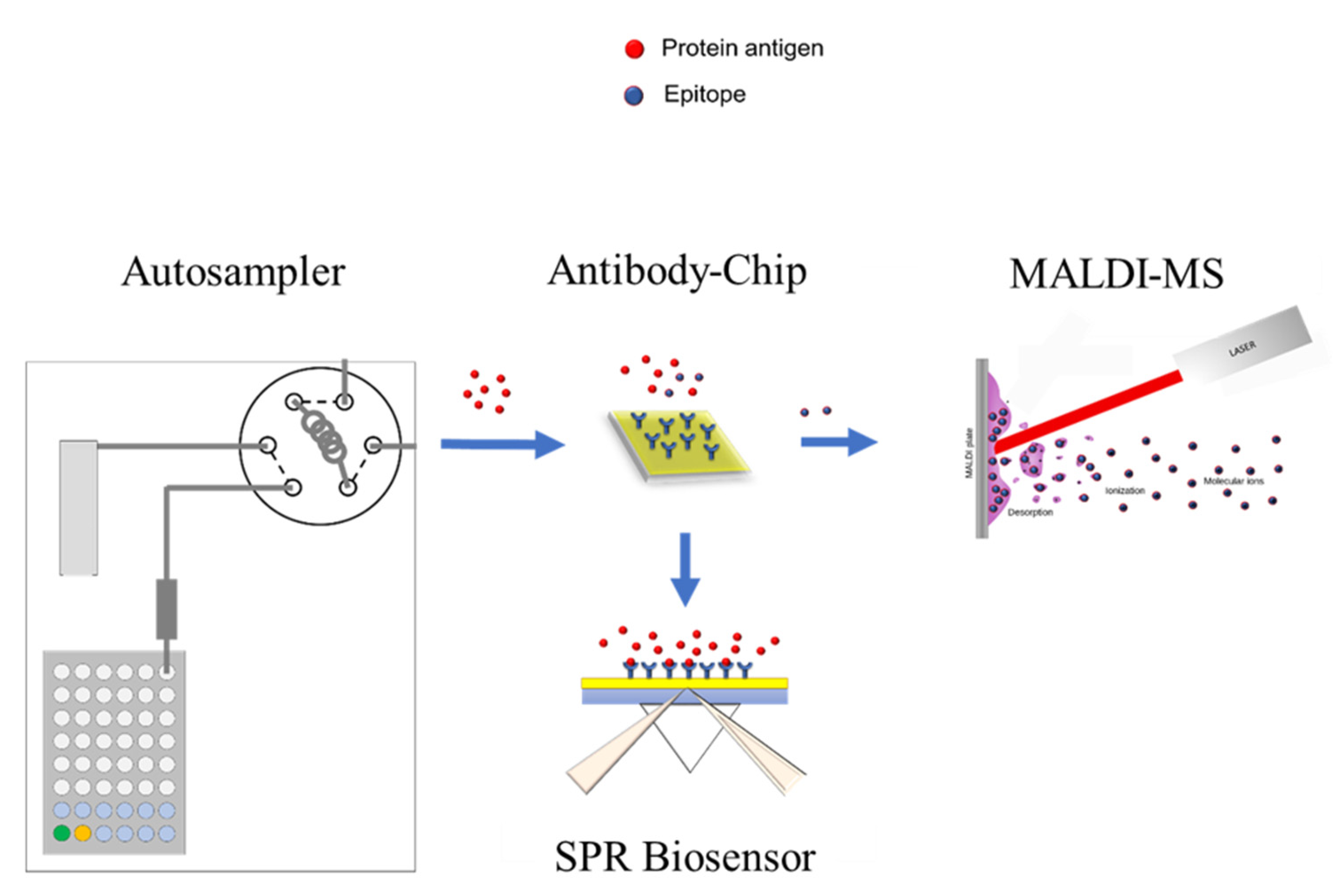
2.1. SPR-MALDI-MS Combination for Epitope and Affinity Determination
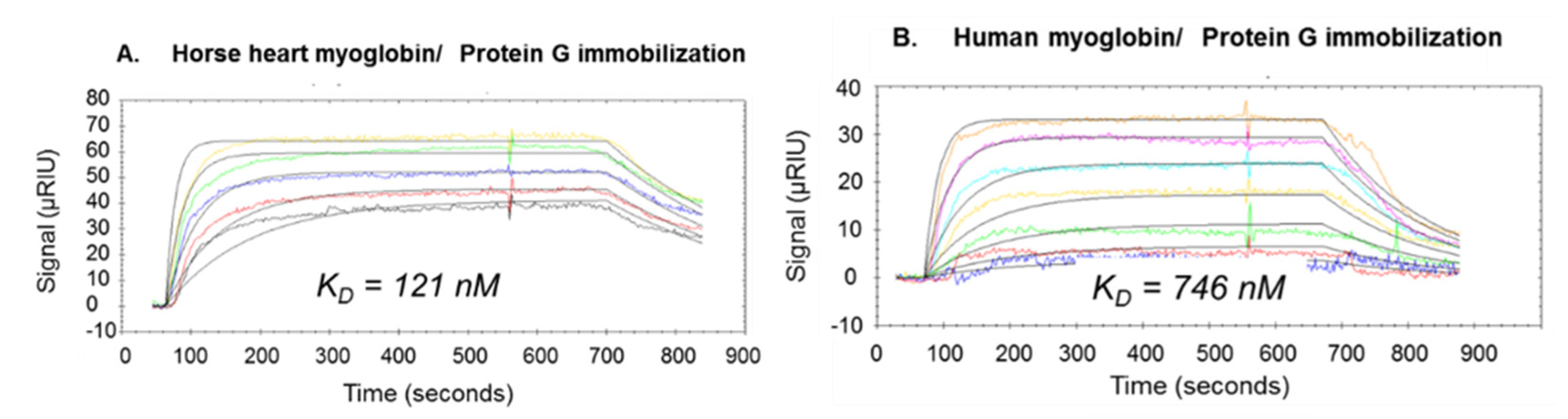
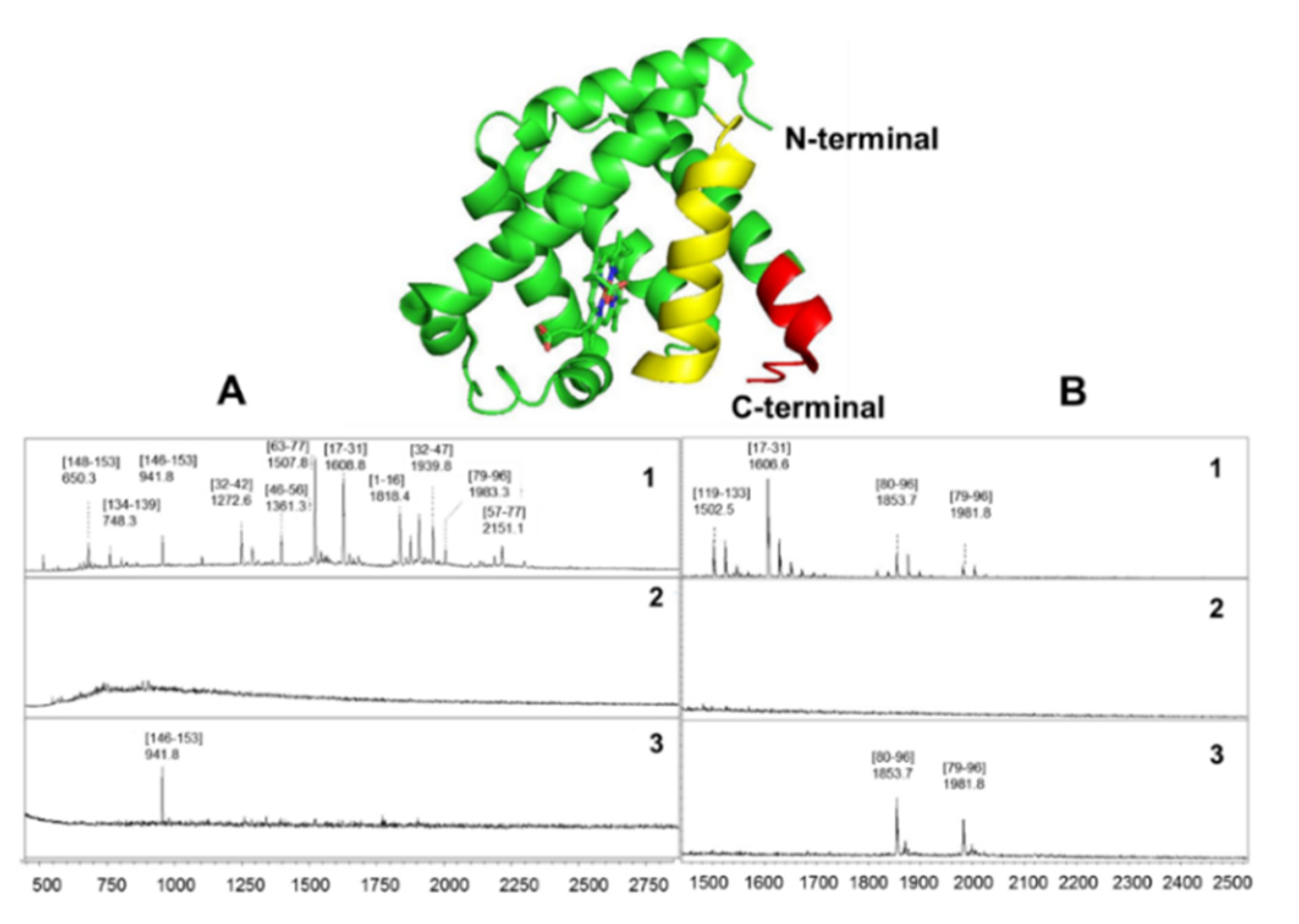
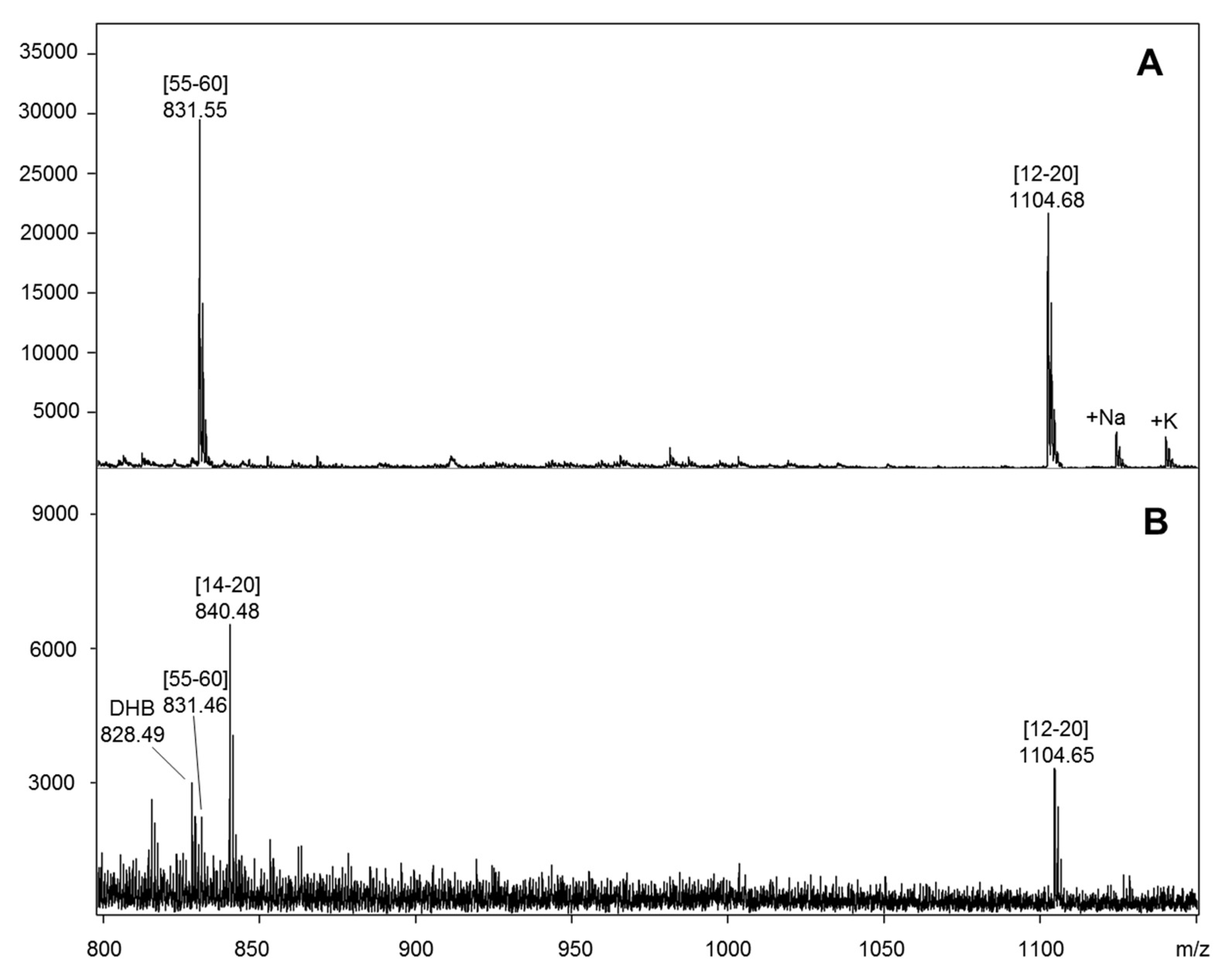
2.2. Determination of Monoclonal and Polyclonal Antibody Epitopes of Myoglobin
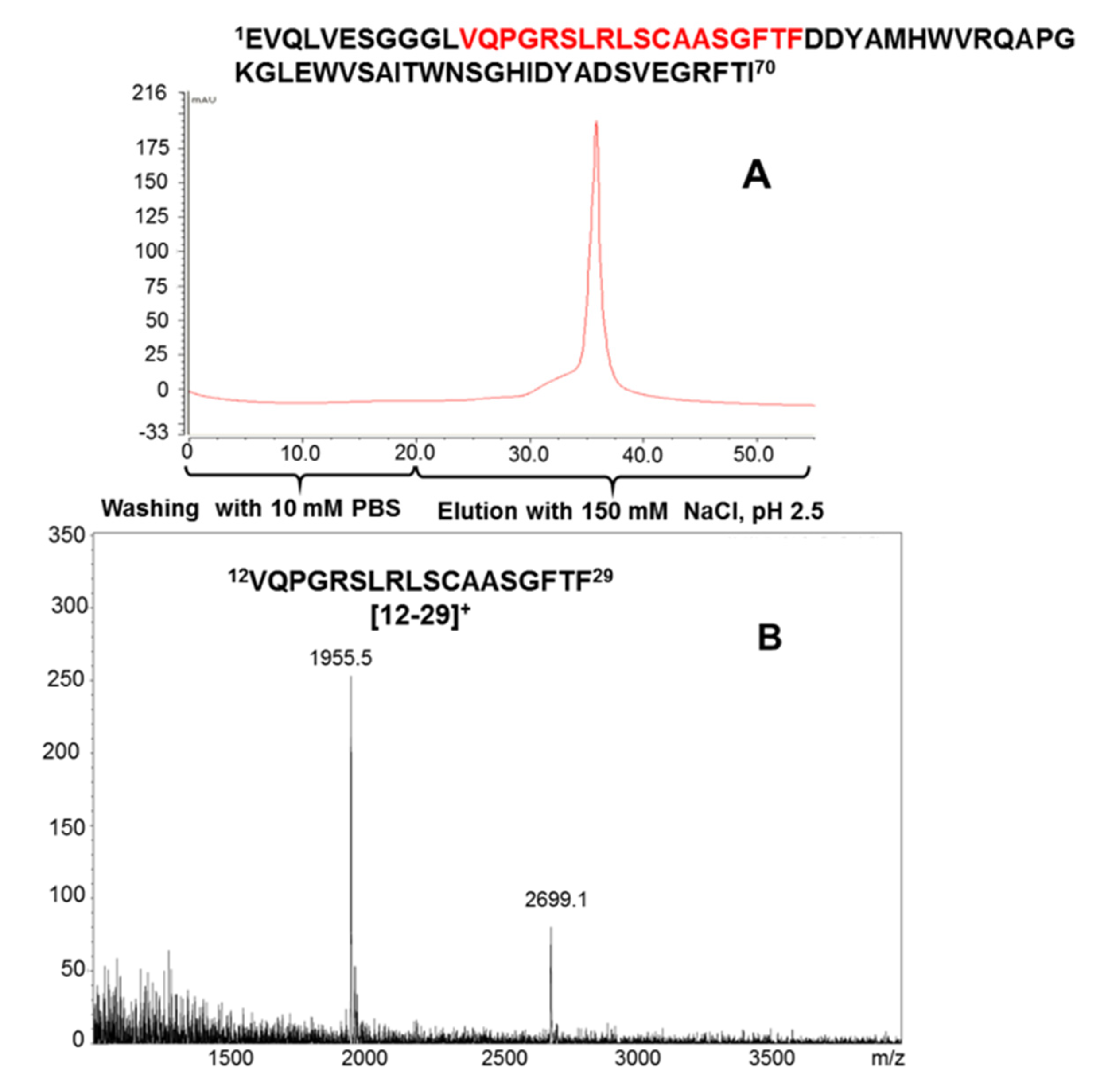
2.3. Protein G Immobilization for Antibody Epitope Determination from Biological Material
2.4. Identification of Assembled (Discontinuous) Antibody Epitopes of Interleukin-8
2.5. Epitope Determination of Pathophysiological Antibodies upon Enzyme Replacement Therapy of Lysosomal Storage Diseases
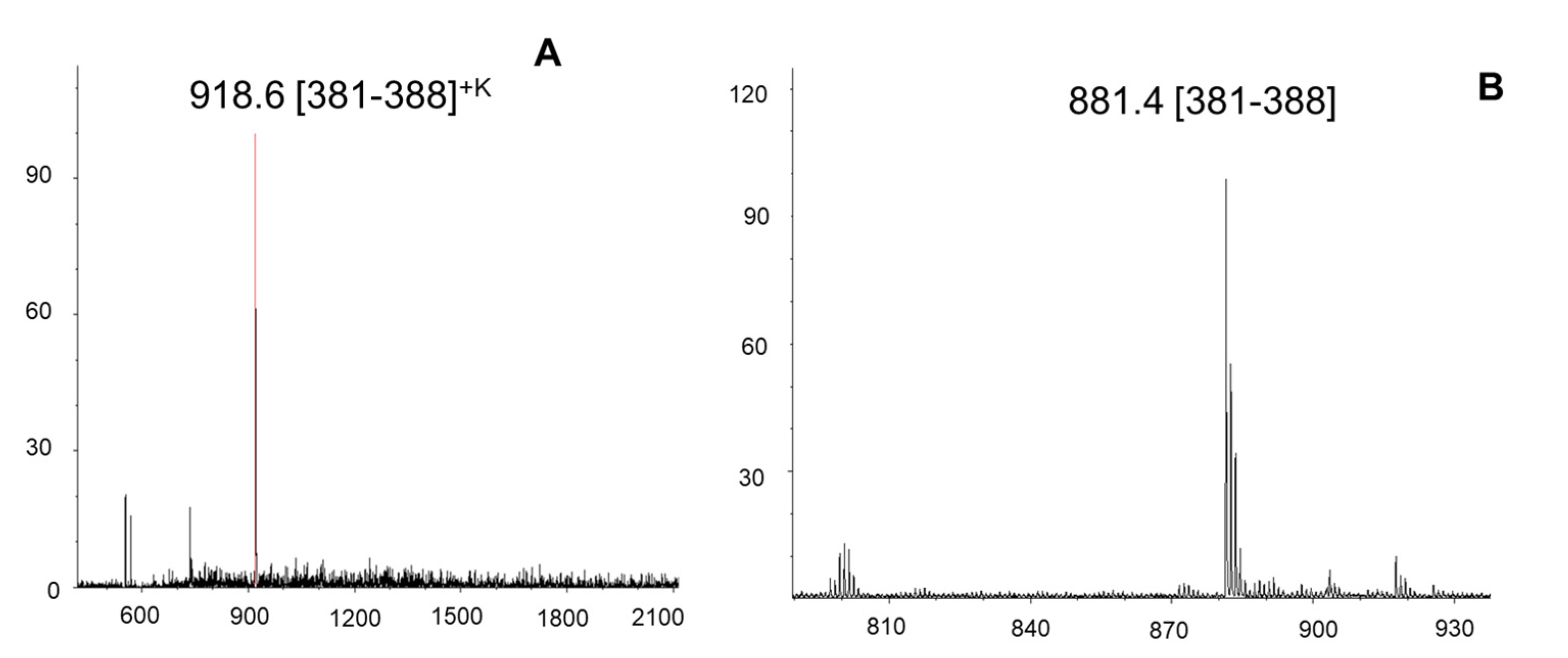
2.6. DNA-Aptamers as Alternative Antibodies: Epitope Determination of an Aptamer-C-Met Protein Complex
3. Conclusions
4. Methods and Experimental Techniques
4.1. Materials and Proteins
4.2. Proteolytic Digestion
4.3. Immobilization Procedures of Antibodies and Aptamers
4.4. SPR Biosensor Techniques
4.5. Epitope Determination by Proteolytic Extraction MS
4.6. Epitope Analyses from SPR-Chips
4.7. Mass Spectrometry
4.8. Synthesis and Affinity Characterization of Epitope Peptides
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ecker, D.M.; Jones, S.D.; Levine, H.L. The therapeutic monoclonal antibody market. mAbs 2015, 7, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Aggarval, S.R. A survey of breakthrough therapy designations. Nat. Biotechnol. 2014, 32, 323–330. [Google Scholar]
- Walsh, G. Biopharmaceutical benchmarks. Nat. Biotechnol. 2014, 32, 992–1000. [Google Scholar] [CrossRef]
- Van Regenmortel, M.H.V. Specificity, polyspecificity, and heterospecificity of antibody-antigen recognition. J. Mol. Recognit. 2014, 27, 627–639. [Google Scholar] [CrossRef]
- Barlow, D.J.; Edwards, M.S.; Thornton, J.M. Continuous and discontinuous protein antigenic determinants. Nature 1986, 322, 747–748. [Google Scholar] [CrossRef]
- Przybylski, M. Mass spectrometry. In Hand-Book of Spectroscopy; Gauglitz, G., Moore, D.S., Eds.; Wiley-VCH: Weinheim, Germany, 2014; pp. 357–405. [Google Scholar]
- Opuni, K.F.M.; Al-Majdoub, M.; Yefremova, Y.; El-Kased, R.F.; Koy, C.; Glocker, M.O. Mass Spectrometric Epitope Mapping. Mass Spectrom. Rev. 2018, 37, 229–241. [Google Scholar] [CrossRef] [PubMed]
- Petre, B.A.; Ulrich, M.; Stumbaum, M.; Bernevic, B.; Moise, A.; Döring, G.; Przybylski, M. When is mass spectrometry combined with affinity approaches essential? A case study of tyrosine nitration in proteins. J. Am. Soc. Mass Spectrom. 2012, 23, 1831–1840. [Google Scholar] [CrossRef] [PubMed]
- Dragusanu, M.; Petrel, B.A.; Slamnoiu, S.; Vlad, C.; Tu, T.; Przybylski, M. Online bioaffinity-electrospray mass spectrometry for simultaneous detection, identification, and quantification of protein-ligand interactions. J. Am. Soc. Mass Spectrom. 2010, 21, 1643–1648. [Google Scholar] [CrossRef][Green Version]
- Hager-Braun, C.; Tomer, K.B. Determination of protein-derived epitopes by mass spectrometry. Expert Rev. Proteom. 2005, 2, 745–756. [Google Scholar] [CrossRef]
- Dhungana, S.; Williams, J.G.; Fessler, M.B.; Tomer, K.B. Epitope mapping by proteolysis of antigen-antibody complexes. In Epitope Mapping Protocols; Reineke, U., Schutkowskim, M., Eds.; Humana Press: New York, NY, USA, 2009; pp. 87–102. [Google Scholar]
- Defaus, S.; Avilés, M.; Andreu, D.; Gutiérrez-Gallego, R. Lectin-Binding Specificity of the Fertilization-Relevant Protein PDC-109 by Means of Surface Plasmon Resonance and Carbohydrate Recognition Domain Excision-Mass Spectrometry. Int. J. Mol. Sci. 2018, 19, 1076. [Google Scholar] [CrossRef] [PubMed]
- Paterson, Y.; Englander, S.W.; Roder, H. An antibody binding site on cytochrome c defined by hydrogen exchange and two-dimensional NMR. Science 1990, 249, 755–759. [Google Scholar] [CrossRef] [PubMed]
- Malito, E.; Carfi, A.; Bottomley, M.J. Protein Crystallography in Vaccine Research and Development. Int. J. Mol. Sci. 2015, 16, 13106–13140. [Google Scholar] [CrossRef] [PubMed]
- Carter, J.M. Epitope mapping of a protein using the Geysen (PEPSCAN) procedure. Methods Mol. Biol. 2016, 36, 207–223. [Google Scholar]
- Kristensen, C.; Kjeldsen, T.; Wiberg, F.C.; Schäffer, L.; Hach, M.; Havelund, S.; Bass, L.; Steiner, D.F.; Andersen, A.S. Alanine Scanning Mutagenesis of Insulin. J. Biol. Chem. 1997, 272, 12978–12983. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.; Liu, X.; Feng, J.; Zhang, W.; Shen, B.; Ou’yang, W.; Cao, Y.; Jin, B. Characterization of the neutralizing activity of three anti-human TNF monoclonal antibodies and prediction of their TNF epitopes by molecular modeling and mutant protein approach. Immunol. Lett. 2006, 102, 177–183. [Google Scholar] [CrossRef]
- Chen, Y.; Wiesmann, C.; Fuh, G.; Li, B.; Christinger, H.W.; McKay, P.; deVos, A.M.; Lowman, H.B. Selection and analysis of an optimized anti-VEGF antibody: Crystal structure of an affinity-matured Fab in complex with antigen. J. Mol. Biol. 1999, 293, 865–881. [Google Scholar] [CrossRef]
- Engen, J.R.; Wales, T.E. Analytical Aspects of Hydrogen Deuterium Mass Spectrometry. Ann. Rev. Anal. Chem. 2015, 8, 127–148. [Google Scholar] [CrossRef]
- Pirrhone, G.F.; Vernon, B.C.; Kent, M.S.; Engen, J.R. Hydrogen Deuterium Exchange Mass Spectrometry of Proteins at Langmuir Monolayers. Anal. Chem. 2015, 87, 7022–7029. [Google Scholar] [CrossRef]
- Chen, J.; Rempel, D.L.; Gau, B.C.; Gross, M. Fast Photochemical Oxidation of Proteins and Mass Spectromety follow submillisecond Protein Folding at the Amino Acid Level. J. Am. Chem. Soc. 2012, 134, 18724–18731. [Google Scholar] [CrossRef]
- Gau, B.; Chen, J.; Gross, M.L. Fast Photochemical Oxidation of Proteins for Comparing Solvent-accessible Changes Accompanying Protein Folding. Biochim. Biophys. Acta 2013, 1834, 1230–1238. [Google Scholar] [CrossRef]
- Narang, D.; Lento, C.; Wilson, J.D. HDX-MS: An Analytical Tool to Capture Protein Motion in Action. Biomedicines 2020, 8, 224. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.L.; Legge, F.S.; Lebani, K.; Mahler, S.M.; Young, P.R.; Watterson, D.; Treutlein, H.R.; Zeng, J. Computational Identification of Antibody Epitopes on the Dengue Virus NS1 Protein. Molecules 2017, 22, 607. [Google Scholar] [CrossRef]
- Demolombe, V.; de Brevern, A.G.; Molina, F.; Lavigne, G.; Granier, C.; Moreau, V. Benchmarking the PEPOP methods for mimicking discontinuous epitopes. BMC Bioinform. 2019, 20, 738. [Google Scholar] [CrossRef]
- Suckau, D.; Kohl, J.; Karwath, G.; Schneider, K.; Casaretto, M.; Bitter-Suermann, D.; Przybylski, M. Molecular epitope identification by limited proteolysis of an antigen-antibody complex and mass spectrometric peptide mapping. Proc. Natl. Acad. Sci. USA 1990, 87, 9848–9852. [Google Scholar] [CrossRef] [PubMed]
- Stefanescu, R.; Iacob, R.E.; Damoc, E.N.; Marquardt, A.; Amstalden, E.; Manea, M.; Perdivara, I.; Maftei, M.; Paraschiv, G.; Przybylski, M. Mass spectrometric approaches for elucidation of antigen-antibody recognition structures in molecular immunology. Eur. J. Mass Spectrom. 2007, 13, 69–75. [Google Scholar] [CrossRef]
- Legros, V.; Jolivet-Reynaud, C.; Battail-Poirot, N.; Saint-Pierre, C.; Forest, E. Characterization of an anti-Borrelia burgdorferi OspA conformational epitope by limited proteolysis of monoclonal antibody-bound antigen and mass spectrometric peptide mapping. Protein Sci. 2000, 9, 1002–1010. [Google Scholar] [CrossRef] [PubMed]
- McLaurin, J.; Cecal, R.; Kierstead, M.E.; Tian, X.; Phinney, A.L.; Manea, M.; French, J.E.; Lambermon, M.H.; Darabie, A.A.; Brown, M.E. Therapeutically effective antibodies against amyloid-beta peptide target amyloid-beta residues 4-10 and inhibit cytotoxicity and fibrillogenesis. Nat. Med. 2002, 8, 1263–1269. [Google Scholar] [CrossRef]
- Juszczyk, P.; Paraschiv, G.; Szymanska, A.; Kolodziejczyk, A.S.; Rodziewicz-Motowidlo, S.; Grzonka, Z.; Przybylski, M. Binding epitopes and interaction structure of the neuroprotective protease inhibitor cystatin C with beta-amyloid revealed by proteolytic excision mass spectrometry and molecular docking simulation. J. Med. Chem. 2009, 52, 2420–2428. [Google Scholar] [CrossRef] [PubMed]
- Iurascu, M.I.; Marroquin Belaunzanar, O.; Cozma, C.; Petrausch, U.; Renner, C.; Przybylski, M. An HLA-B27 Homodimer Specific Antibody Recognizes a Discontinuous Mixed- Disulfide Epitope as Identified by Affinity-Mass Spectrometry. J. Am. Soc. Mass Spectrom. 2016, 27, 1105–1112. [Google Scholar] [CrossRef]
- Paraschiv, G.; Vincke, C.; Czaplewska, P.; Manea, M.; Muyldermans, S.; Przybylski, M. Epitope structure and binding affinity of single chain llama anti-β-amyloid antibodies revealed by proteolytic excision affinity-mass spectrometry. J. Mol. Recognit. 2013, 26, 1–9. [Google Scholar] [CrossRef]
- Moise, A.; André, S.; Eggers, F.; Krzeminski, M.; Przybylski, M.; Gabius, H.J. Toward bioinspired galectin mimetics: Identification of ligand-contacting peptides by proteolytic-excision mass spectrometry. J. Am. Chem. Soc. 2011, 133, 14844–14847. [Google Scholar] [CrossRef] [PubMed]
- Stefanescu, R.; Born, R.; Moise, A.; Ernst, B.; Przybylski, M. Epitope structure of the carbohydrate recognition domain of asialoglycoprotein receptor to a monoclonal antibody revealed by high-resolution proteolytic excision mass spectrometry. J. Am. Soc. Mass Spectrom. 2011, 22, 148–157. [Google Scholar] [CrossRef]
- Lakayan, D.; Haselberg, R.; Gahoual, R.; Somsen, G.W.; Kool, J. Affinity profiling of monoclonal antibody and antibody-drug-conjugate preparations by coupled liquid chromatography-surface plasmon resonance biosensing. Anal. Bioanal. Chem. 2018, 410, 7837–7848. [Google Scholar] [CrossRef]
- Florinskaya, A.; Ershov, P.; Mezentsev, Y. SPR biosensors in direct molecular Fishing: Implications for Protein Interactomics. Sensors 2018, 18, 1616. [Google Scholar] [CrossRef]
- Bouffartigues, E.; Leh, H.; Anger-Leroy, M.; Rimsky, S.; Buckle, M. Rapid coupling of Surface Plasmon Resonance (SPR and SPRi) and ProteinChip based mass spectrometry for the identification of proteins in nucleoprotein interactions. Nucleic Acids Res. 2007, 35, e39. [Google Scholar] [CrossRef] [PubMed]
- Mihoc, D.; Lupu, L.M.; Wiegand, P.; Kleinekofort, W.; Müller, O.; Völklein, F.; Glocker, M.O.; Barka, F.; Barka, G.; Przybylski, M. Antibody Epitope and Affinity Determination of the Myocardial Infarction Marker Myoglobin by SPR-Biosensor Mass Spectrometry. J. Am. Soc. Mass Spectrom. 2021, 6, 106–113. [Google Scholar] [CrossRef]
- Atassi, M.Z.; Tarlowski, D.P.; Paull, J.H. Immunochemistry of sperm whale myoglobin, vll. Correlation of immunochemical cross-reaction of eight myoglobins with structural similarity and its dependence on conformation. Biochim. Biophys. Acta Protein Struct. 1970, 221, 623–640. [Google Scholar] [CrossRef]
- Berkower, I.; Buckenmeyer, G.K.; Gurd, F.R.; Berzofsky, J.A. A possible immunodominant epitope recognized by murine T lymphocytes immune to different myoglobins. Proc. Natl. Acad. Sci. USA 1982, 79, 4723–4727. [Google Scholar] [CrossRef]
- Macht, M.; Fiedler, W.; Kuerzinger, K.; Przybylski, M. Mass Spectrometric Mapping of Protein Epitope Structures of Myocardial Infarct Markers Myoglobin and Troponin, T. Biochemistry 1996, 35, 15633–15639. [Google Scholar] [CrossRef]
- Bjorck, L.; Kronvall, G. Purification and some properties of streptococcal protein G, a novel IgG-binding reagent. J. Immunol. 1984, 133, 969–974. [Google Scholar] [PubMed]
- Chen, P.K.; Lan, J.L.; Chen, Y.M.; Chen, H.H.; Chang, S.H.; Chung, C.M.; Rutt, N.H.; Tan, T.M.; Mamat, R.N.R.; Anuar, N.D.; et al. Anti-TROVE2 Antibody Determined by Immune-Related Array May Serve as a Predictive Marker for Adalimumab Immunogenicity and Effectiveness in RA. J. Immunol. Res. 2021, 2021, 6656121. [Google Scholar] [CrossRef]
- Homann, A.; Röckendorf, N.; Kromminga, A.; Frey, A.; Platts-Mills, T.A.; Jappe, U. Glycan and Peptide IgE Epitopes of the TNF-alpha Blockers Infliximab and Adalimumab—Precision Diagnostics by Cross-Reactivity Immune Profiling of Patient Sera. Theranostics 2017, 7, 4699–4709. [Google Scholar] [CrossRef]
- Van Schouwenburg, P.A.; Kruithof, S.; Votsmeier, C.; van Schie, K.; Hart, M.H.; de Jong, R.N.; van Buren, E.E.; van Ham, M.; Aarden, L.; Wolbink, G.; et al. Functional analysis of the anti-adalimumab response using patient-derived monoclonal antibodies. J. Biol. Chem. 2014, 289, 34482–34488. [Google Scholar] [CrossRef]
- Krayukhina, E.; Noda, M.; Ishii, K.; Maruno, T.; Wakabayashi, H.; Tada, M.; Suzuki, T.; Ishii-Watabe, A.; Kato, M.; Uchiyama, S. Analytical ultracentrifugation with fluorescence detection system reveals differences in complex formation between recombinant human TNF and different biological TNF antagonists in various environments. mAbs 2017, 9, 664–679. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, M.; Ozawa, F.; Sugimoto, W.; Aso, S. Miniature surface-plasmon resonance immunosensors-rapid and repetitive procedure. Anal. Bioanal. Chem. 2002, 372, 301–304. [Google Scholar] [CrossRef] [PubMed]
- Bergström, G.; Mandenius, C.F. Orientation and capturing of antibody affinity ligands: Applications to surface plasmon resonance biochips. Sens. Actuators B 2011, 158, 265–270. [Google Scholar] [CrossRef]
- Rusche, H. Identification of Adalimumab Epitopes Recognized by Anti-drug Antibodies. Master’s Thesis, University of Konstanz, Konstanz, Germany, 2017. [Google Scholar]
- Lowman, H.B.; Fairbrother, W.J.; Slagle, P.H.; Kabakoff, R.; Liu, J.; Shire, S.; Hebert, C.A. Monomeric variants of IL-8: Effects of side chain substitutions and solution conditions upon dimer formation. Protein Sci. 1997, 6, 598–608. [Google Scholar] [CrossRef] [PubMed]
- Helmer, D.; Rink, I.; Dalton, J.A.R.; Brahm, K.; Jöst, M.; Nargang, T.M.; Blum, W.; Wadhwani, P.; Brenner-Weiss, G.; Rapp, B.E.; et al. Rational design of a peptide capture agent for CXCL8 based on a model of the CXCL8:CXCR1 complex. RSC Adv. 2015, 5, 25657–25668. [Google Scholar] [CrossRef]
- Clubb, R.T.; Omichinski, J.G.; Clore, G.M.; Gronenborn, A.M. Mapping the binding surface of interleukin-8 complexed with an N-terminal fragment of the Type 1 human interleukin-8 receptor. FEBS Lett. 1994, 338, 93–97. [Google Scholar] [CrossRef]
- Wiegand, P.; Lupu, L.; Huettmann, N.; Wack, J.; Rawer, S.; Przybylski, M.; Schmitz, K. Epitope Identification and Affinity Determination of an Inhibiting Human Antibody to Interleukin IL8 (CXCL8) by SPR- Biosensor—Mass Spectrometry Combination. J. Am. Soc. Mass Spectrom. 2020, 31, 109. [Google Scholar] [CrossRef]
- Bigger, B.W.; Saif, M.; Linthorst, G.E. The role of antibodies in enzyme treatments and therapeutic strategies. Best Pract. Res. Clin. Endocrinol. Metab. 2015, 29, 183–194. [Google Scholar] [CrossRef]
- Ortolano, S.; Vieitez, I.; Navarro, C.; Spuch, C. Treatment of Lysosomal Storage Diseases: Recent Patents and Strategies. Rec. Pat. Endocr. Metab. Immune Drug Discov. 2014, 8, 1872–2148. [Google Scholar] [CrossRef]
- Desnik, R.J. Enzyme replacement therapy for Fabry disease: Lessons from two galactosidase A orphan products and one FDA approval. Expert Opin. Biol. Ther. 2004, 4, 1167–1176. [Google Scholar] [CrossRef] [PubMed]
- Banugaria, S.N.; Prater, S.N.; Ng, Y.K.; Kobori, J.A.; Finkel, R.S.; Ladda, R.L.; Chen, Y.T.; Rosenberg, A.S.; Kishani, P.S. The impact of antibodies on clinical outcomes in diseases treated with therapeutic protein: Lessons learned from infantile Pompe disease. Genet. Med. 2011, 13, 729–736. [Google Scholar] [CrossRef] [PubMed]
- Banugaria, S.G.; Patel, T.T.; Mackey, J.; Das, S.; Amalfitano, A.; Rosenberg, A.S.; Charrow, J.; Chen, Y.T.; Kishani, P.S. Persistence of high sustained antibodies to enzyme replacement therapy despite extensive immunomodulatory therapy in an infant with Pompe disease: Need for agents to target antibody secreting plasma cells. Mol. Genet. Metab. 2012, 105, 677–680. [Google Scholar] [CrossRef]
- Valaianopoulos, V.; Wijburg, F.A. Therapy for the Mucopolysaccharidoses. Rheumatology 2011, 50, v49–v59. [Google Scholar] [CrossRef] [PubMed]
- Hennermann, J.B.; Gökce, S.; Solyom, A.; Mengel, E.; Schuchman, E.H.; Simonaro, C.M. Treatment with pentosan polysulphate in patients with MPS I: Results from an open label, randomized, monocentric phase II study. J. Inherit. Metab. Dis. 2016, 39, 831–837. [Google Scholar] [CrossRef]
- Langereis, E.J.; van Vlies, N.; Church, H.J.; Geskus, R.B.; Hollak, C.E.; Jones, S.A.; Kulik, W.; van Lenthe, H.; Mercer, J.; Schreider, L.; et al. Biomarker responses correlate with antibody status in mucopolysaccharidosis type I patients on long-term enzyme replacement therapy. Mol. Genet. Metab. 2015, 114, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Kukacka, Z.; Iurascu, M.; Lupu, L.; Rusche, H.; Murphy, M.; Altamore, L.; Borri, F.; Maeser, S.; Papini, A.M.; Hennermann, J.; et al. Antibody Epitope of Human alpha-Galactosidase A Revealed by Affinity Mass Spectrometry: A Basis for Reversing Immunoreactivity in Enzyme Replacement Therapy of Fabry Disease. ChemMedChem 2018, 13, 909–915. [Google Scholar] [CrossRef]
- Moise, A.; Maeser, S.; Rawer, S.; Eggers, F.; Murphy, M.; Bornheim, J.; Przybylski, M. Substrate and Substrate-Mimetic Chaperone Binding Sites in Human alpha-Galactosidase A Revealed by Affinity-Mass Spectrometry. J. Am. Soc. Mass. Spectrom. 2016, 27, 1071–1078. [Google Scholar] [CrossRef] [PubMed]
- Zamay, T.N.; Zamay, G.S.; Kolovskaya, O.S.; Zukov, R.A.; Petrova, M.M.; Gargaun, A.; Berezovski, M.V.; Kichkailo, A.S. Current and Prospective Protein Biomarkers of Lung Cancer. Cancers 2017, 9, 155. [Google Scholar] [CrossRef] [PubMed]
- Zamay, G.S.; Ivanchenko, T.I.; Zamay, T.N.; Grigorieva, V.L.; Glazyrin, Y.E.; Kolovskaya, O.S.; Garanzha, I.V.; Barinov, A.A.; Krat, A.V.; Mironov, G.G.; et al. DNA Aptamers for the Characterization of Histological Structure of Lung Adenocarcinoma. Mol. Ther. Nucleic Acids 2017, 6, 150–162. [Google Scholar] [CrossRef] [PubMed]
- Xie, S.; Walton, S.P. Development of a dual-aptamer-based multiplex protein biosensor. Biosens Bioelectron. 2010, 25, 2663–2668. [Google Scholar] [CrossRef] [PubMed]
- Sabri, M.Z.; Abdul Hamid, A.A.; Sayed Hitam, S.M.; Abdul Rahim, M.Z. In Silico Screening of Aptamers Configuration against Hepatitis B Surface Antigen. Adv. Bioinform. 2019, 2019, 6912914. [Google Scholar] [CrossRef] [PubMed]
- Labib, M.; Zamay, A.S.; Muharemagic, D.; Chechik, A.V.; Bell, J.C.; Berezovski, M.V. Electrochemical differentiation of epitope-specific aptamers. Anal. Chem. 2012, 84, 2548–2556. [Google Scholar] [CrossRef]
- Lupu, L.; Wiegand, P.; Hüttmann, N.; Rawer, S.; Kleinekofort, W.; Shugureva, I.; Kichkailo, A.S.; Tomilin, F.N.; Lazarev, A.; Berezovski, M.V.; et al. Molecular Epitope Determination of Aptamer Complexes of the Multidomain Protein C-Met by Proteolytic Affinity-Mass Spectrometry. ChemMedChem 2020, 15, 363–369. [Google Scholar] [CrossRef]
- Famulok, M.; Mayer, G. Aptamers and SELEX in Chemistry & Biology. Chem. Biol. 2014, 18, 1055–1058. [Google Scholar]
- Bouattour, M.; Raymond, E.; Qin, S.; Cheng, A.L.; Stammberger, U.; Locatelli, G.; Faivre, S. Recent developments of C-Met as a therapeutic target in hepatocellular carcinoma. Hepatology 2018, 67, 1132–1149. [Google Scholar] [CrossRef]
- Al-Majdoub, M.; Koy, C.; Lorenz, P.; Thiesen, H.J.; Glocker, M.O. Mass spectrometric and peptide chip characterization of an assembled epitope: Analysis of a polyclonal antibody model serum directed against the Sjogren/systemic lupus erythematosus autoantigen TRIM21. J. Mass Spectrom. 2013, 48, 651–659. [Google Scholar] [CrossRef]
- Ansong, C.; Miles, S.M.; Fay, P.J. Epitope mapping factor VIII A2 domain by affinity-directed mass spectrometry: Residues 497-510 and 584-593 comprise a discontinuous epitope for the monoclonal antibody R8B12. J. Thromb Haemost. 2006, 4, 842–847. [Google Scholar] [CrossRef]
- El-Kased, R.F.; Koy, C.; Deierling, T.; Lorenz, P.; Qian, Z.; Li, Y.; Thiesen, H.J.; Glocker, M.O. Mass spectrometric and peptide chip epitope mapping of rheumatoid arthritis autoantigen RA33. Eur. J. Mass Spectrom. 2009, 15, 747–759. [Google Scholar] [CrossRef]
- Glocker, M.O.; Nock, S.; Sprinzl, M.; Przybylski, M. Characterization of surface topology and binding area in complexes of the elongation factor proteins EF-Ts and EF-Tu.GDP from thermus thermophilus: A study by protein chemical modification and mass spectrometry. Chem. Eur. J. 1998, 4, 707–715. [Google Scholar] [CrossRef]
- Macht, M.; Marquardt, A.; Deininger, S.O.; Damoc, E.; Kohlmann, M.; Przybylski, M. Affinity-proteomics: Direct protein identification from biological material using mass spectrometric epitope mapping. Anal. Bioanal. Chem. 2004, 378, 1102–1111. [Google Scholar] [CrossRef] [PubMed]
- Obungu, V.H.; Gelfanova, V.; Huang, L. Epitope mapping of antibodies by mass spectroscopy: A case study. Methods Mol. Biol. 2013, 988, 291–302. [Google Scholar] [PubMed]
- Sinz, A. Chemical cross-linking and mass spectrometry to map threedimensional protein structures and protein-protein interactions. Mass Spectrom. Rev. 2006, 25, 663–682. [Google Scholar] [CrossRef]

| Method | Random KD (µM) (a) | Protein G KD (µM) (b) |
|---|---|---|
| Kinetics evaluation | 0.028 | 0.121 |
| Affinity plot | 0.280 | 0.145 |
| Mean Value with error | 0.154 ± 0.126 | 0.133 ± 0.12 |
| Protein/Peptide | KD (µM) | |
|---|---|---|
| Intact protein | α-Gal (1–429) | 0.015 |
| Epitope | α-Gal (309–332) | 0.038 |
| Peptide I | α-Gal (309–320) | 151 |
| Peptide II | α-Gal (315–326) | 43.4 |
| Peptide III | α-Gal (321–332) | 7.05 |
| Peptide IV | α-Gal (309–316) | 816 |
| Peptide V | α-Gal (317–324) | 1600 |
| Peptide VI | α-Gal (325–332) | 493 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lupu, L.-M.; Wiegand, P.; Holdschick, D.; Mihoc, D.; Maeser, S.; Rawer, S.; Völklein, F.; Malek, E.; Barka, F.; Knauer, S.; et al. Identification and Affinity Determination of Protein-Antibody and Protein-Aptamer Epitopes by Biosensor-Mass Spectrometry Combination. Int. J. Mol. Sci. 2021, 22, 12832. https://doi.org/10.3390/ijms222312832
Lupu L-M, Wiegand P, Holdschick D, Mihoc D, Maeser S, Rawer S, Völklein F, Malek E, Barka F, Knauer S, et al. Identification and Affinity Determination of Protein-Antibody and Protein-Aptamer Epitopes by Biosensor-Mass Spectrometry Combination. International Journal of Molecular Sciences. 2021; 22(23):12832. https://doi.org/10.3390/ijms222312832
Chicago/Turabian StyleLupu, Loredana-Mirela, Pascal Wiegand, Daria Holdschick, Delia Mihoc, Stefan Maeser, Stephan Rawer, Friedemann Völklein, Ebrahim Malek, Frederik Barka, Sascha Knauer, and et al. 2021. "Identification and Affinity Determination of Protein-Antibody and Protein-Aptamer Epitopes by Biosensor-Mass Spectrometry Combination" International Journal of Molecular Sciences 22, no. 23: 12832. https://doi.org/10.3390/ijms222312832
APA StyleLupu, L.-M., Wiegand, P., Holdschick, D., Mihoc, D., Maeser, S., Rawer, S., Völklein, F., Malek, E., Barka, F., Knauer, S., Uth, C., Hennermann, J., Kleinekofort, W., Hahn, A., Barka, G., & Przybylski, M. (2021). Identification and Affinity Determination of Protein-Antibody and Protein-Aptamer Epitopes by Biosensor-Mass Spectrometry Combination. International Journal of Molecular Sciences, 22(23), 12832. https://doi.org/10.3390/ijms222312832








