A Possible Role for HMG-CoA Reductase Inhibitors and Its Association with HMGCR Genetic Variation in Parkinson’s Disease
Abstract
:1. Introduction
2. Pathogenesis of Dementia in Parkinson’s Disease
3. Protective Role of Cholesterol in Neurodegenerative Diseases
4. Detrimental Role of Cholesterol in Neurodegenerative Diseases
5. Statins and HMG-CoA Reductase
6. Statins in Neuroprotection
7. Statins in PD
8. HMGCR Gene Polymorphisms and Haplotypes
9. HMGCR Genetic Variability in PD and Other Neurodegenerative Diseases
10. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Moustafa, A.A.; Chakravarthy, S.; Phillips, J.R.; Gupta, A.; Keri, S.; Polner, B.; Frank, M.J.; Jahanshahi, M. Motor symptoms in Parkinson’s disease: A unified framework. Neurosci. Biobehav. Rev. 2016, 68, 727–740. [Google Scholar] [CrossRef] [PubMed]
- Rao, G.; Fisch, L.; Srinivasan, S.; D’Amico, F.; Okada, T.; Eaton, C.; Robbins, C. Does this patient have Parkinson disease? JAMA 2003, 289, 347–353. [Google Scholar] [CrossRef] [PubMed]
- Chaudhuri, K.R.; Healy, D.G.; Schapira, A.H. National Institute for Clinical Excellence. Non-motor symptoms of Parkinson’s disease: Diagnosis and management. Lancet Neurol. 2006, 5, 235–245. [Google Scholar] [CrossRef]
- Aarsland, D. Cognitive impairment in Parkinson’s disease and dementia with Lewy bodies. Parkinsonism Relat. Disord. 2016, 22, S144–S148. [Google Scholar] [CrossRef] [PubMed]
- Larsson, V.; Torisson, G.; Londos, E. Relative survival in patients with dementia with Lewy bodies and Parkinson’s disease dementia. PLoS ONE 2018, 13, e0202044. [Google Scholar] [CrossRef]
- Ascherio, A.; Schwarzschild, M.A. The epidemiology of Parkinson’s disease: Risk factors and prevention. Lancet Neurol. 2016, 15, 1257–1272. [Google Scholar] [CrossRef]
- Jellinger, K.A. How close are we to revealing the etiology of Parkinson’s disease? Expert Rev. Neurother. 2015, 15, 1105–1107. [Google Scholar] [CrossRef]
- Fahn, S. The 200-year journey of Parkinson disease: Reflecting on the past and looking towards the future. Parkinsonism Relat. Disord. 2018, 46, S1–S5. [Google Scholar] [CrossRef]
- Niranjan, R. The role of inflammatory and oxidative stress mechanisms in the pathogenesis of Parkinson’s disease: Focus on astrocytes. Mol. Neurobiol. 2014, 49, 28–38. [Google Scholar] [CrossRef] [PubMed]
- Jellinger, K.A. Neurobiology of cognitive impairment in Parkinson’s disease. Expert Rev. Neurother. 2012, 12, 1451–1466. [Google Scholar] [CrossRef]
- Saravi, S.S.S.; Saravi, S.S.S.; Arefidoust, A.; Dehpour, A.R. The beneficial effects of HMG-CoA reductase inhibitors in the processes of neurodegeneration. Metab. Brain Dis. 2017, 32, 949–965. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Qiao, L.; Tian, J.; Liu, A.; Wu, J.; Huang, J.; Shen, M.; Lai, X. Effect of statins on Parkinson’s disease: A systematic review and meta-analysis. Medicine 2019, 98, e14852. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Chen, H.; Miller, W.C.; Mailman, R.B.; Woodard, J.L.; Chen, P.C.; Xiang, D.; Murrow, R.W.; Wang, Y.Z.; Poole, C. Lower low-density lipoprotein cholesterol levels are associated with Parkinson’s disease. Mov. Disord. 2007, 22, 377–381. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leduc, V.; De Beaumont, L.; Théroux, L.; Dea, D.; Aisen, P.; Petersen, R.C.; Alzheimer’s Disease Neuroimaging Initiative; Dufour, R.; Poirier, J. HMGCR is a genetic modifier for risk, age of onset and MCI conversion to Alzheimer’s disease in a three cohorts study. Mol. Psychiatry 2015, 20, 867–873. [Google Scholar] [CrossRef] [Green Version]
- Schultz, B.G.; Patten, D.K.; Berlau, D.J. The role of statins in both cognitive impairment and protection against dementia: A tale of two mechanisms. Transl. Neurodegener. 2018, 7, 5. [Google Scholar] [CrossRef] [PubMed]
- Aarsland, D.; Andersen, K.; Larsen, J.P.; Lolk, A.; Kragh-Sørensen, P. Prevalence and characteristics of dementia in Par-kinson disease: An 8-year prospective study. Arch. Neurol. 2003, 60, 387–392. [Google Scholar] [CrossRef] [Green Version]
- Zhu, K.; van Hilten, J.J.; Marinus, J. Predictors of dementia in Parkinson’s disease; findings from a 5-year prospective study using the SCOPA-COG. Parkinsonism Relat. Disord. 2014, 20, 980–985. [Google Scholar] [CrossRef]
- Hanganu, A.; Bedetti, C.; Jubault, T.; Gagnon, J.F.; Mejia-Constain, B.; Degroot, C.; Lafontaine, A.L.; Chouinard, S.; Monchi, O. Mild cognitive impairment in patients with Parkinson’s disease is associated with increased cortical degeneration. Mov. Disord. 2013, 28, 1360–1369. [Google Scholar] [CrossRef]
- Horvath, J.; Herrmann, F.R.; Burkhard, P.R.; Bouras, C.; Kövari, E. Neuropathology of dementia in a large cohort of patients with Parkinson’s disease. Parkinsonism Relat. Disord. 2013, 19, 864–868. [Google Scholar] [CrossRef]
- Jellinger, K.A. Interaction between pathogenic proteins in neurodegenerative disorders. J. Cell. Mol. Med. 2012, 16, 1166–1183. [Google Scholar] [CrossRef]
- Jellinger, K.A. The neuropathologic substrate of Parkinson disease dementia. Acta Neuropathol. 2010, 119, 151–153. [Google Scholar] [CrossRef] [PubMed]
- Orth, M.; Bellosta, S. Cholesterol: Its regulation and role in central nervous system disorders. Cholesterol 2012, 2012, 292598. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vance, J.E. Dysregulation of cholesterol balance in the brain: Contribution to neurodegenerative diseases. Dis. Model. Mech. 2012, 5, 746–755. [Google Scholar] [CrossRef] [Green Version]
- Rozani, V.; Gurevich, T.; Giladi, N.; El-Ad, B.; Tsamir, J.; Hemo, B.; Peretz, C. Higher serum cholesterol and decreased Parkinson’s disease risk: A statin-free cohort study. Mov. Disord. 2018, 33, 1298–1305. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Alonso, A.; Guo, X.; Umbach, D.M.; Lichtenstein, M.L.; Ballantyne, C.M.; Mailman, R.B.; Mosley, T.H.; Chen, H. Statins, plasma cholesterol, and risk of Parkinson’s disease: A prospective study. Mov. Disord. 2015, 30, 552–559. [Google Scholar] [CrossRef]
- Sterling, N.W.; Lichtenstein, M.; Lee, E.Y.; Lewis, M.M.; Evans, A.; Eslinger, P.J.; Du, G.; Gao, X.; Chen, H.; Kong, L.; et al. Higher plasma LDL-cholesterol is associated with preserved executive and fine motor functions in Parkinson’s disease. Aging Dis. 2016, 7, 237–245. [Google Scholar] [CrossRef] [Green Version]
- Musanti, R.; Parati, E.; Lamperti, E.; Ghiselli, G. Decreased Cholesterol Biosynthesis in Fibroblasts from Patients with Par-kinson Disease. Biochem. Med. Metab. Biol. 1993, 49, 133–142. [Google Scholar] [CrossRef]
- Jin, U.; Park, S.J.; Park, S.M. Cholesterol metabolism in the brain and its association with Parkinson’s disease. Exp. Neurobiol. 2019, 28, 554–567. [Google Scholar] [CrossRef] [Green Version]
- Sławek, J.; Roszmann, A.; Robowski, P.; Dubaniewicz, M.; Sitek, E.J.; Honczarenko, K.; Gorzkowska, A.; Budrewicz, S.; Mak, M.; Gołąb-Janowska, M.; et al. The impact of MRI white matter hyperintensities on dementia in Parkinson’s disease in rela-tion to the homocysteine level and other vascular risk factors. Neurodegener. Dis. 2013, 12, 1–12. [Google Scholar] [CrossRef]
- Paul, R.; Choudhury, A.; Borah, A. Cholesterol—A putative endogenous contributor towards Parkinson’s disease. Neurochem. Int. 2015, 90, 125–133. [Google Scholar] [CrossRef]
- Hu, G.; Antikainen, R.; Jousilahti, P.; Kivipelto, M.; Tuomilehto, J. Total cholesterol and the risk of Parkinson disease. Neurology 2008, 70, 1972–1979. [Google Scholar] [CrossRef] [PubMed]
- Anstey, K.J.; Ashby-Mitchell, K.; Peters, R. Updating the evidence on the association between serum cholesterol and risk of late-life dementia: Review and meta-analysis. J. Alzheimer’s Dis. 2017, 56, 215–228. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peng, Z.; Dong, S.; Tao, Y.; Huo, Y.; Zhou, Z.; Huang, W.; Qu, H.; Liu, J.; Chen, Y.; Xu, Z.; et al. Metabolic syndrome contributes to cognitive impairment in patients with Parkinson’s disease. Parkinsonism Relat. Disord. 2018, 55, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Doiron, M.; Langlois, M.; Dupré, N.; Simard, M. The influence of vascular risk factors on cognitive function in early Parkinson’s disease. Int. J. Geriatr. Psychiatry 2018, 33, 288–297. [Google Scholar] [CrossRef] [PubMed]
- Jasinska-Myga, B.; Opala, G.; Goetz, C.G.; Tustanowski, J.; Ochudlo, S.; Gorzkowska, A.; Tyrpa, J. Apolipoprotein E gene polymorphism, total plasma cholesterol level, and Parkinson disease dementia. Arch. Neurol. 2007, 64, 261–265. [Google Scholar] [CrossRef] [Green Version]
- McFarland, A.J.; Anoopkumar-Dukie, S.; Arora, D.S.; Grant, G.D.; McDermott, C.M.; Perkins, A.V.; Davey, A.K. Molecular mechanisms underlying the effects of statins in the central nervous system. Int. J. Mol. Sci. 2014, 15, 20607–20637. [Google Scholar] [CrossRef] [Green Version]
- Barone, E.; Di Domenico, F.; Butterfield, D.A. Statins more than cholesterol lowering agents in Alzheimer disease: Their pleiotropic functions as potential therapeutic targets. Biochem. Pharmacol. 2014, 88, 605–616. [Google Scholar] [CrossRef] [Green Version]
- Ward, N.C.; Watts, G.F.; Eckel, R.H. Statin Toxicity. Mechanistic Insights and Clinical Implications. Circ. Res. 2019, 124, 328–350. [Google Scholar] [CrossRef]
- Bai, S.; Song, Y.; Huang, X.; Peng, L.; Jia, J.; Liu, Y.; Lu, H. Statin use and the risk of Parkinson’s disease: An updated me-ta-analysis. PLoS ONE 2016, 11, e0152564. [Google Scholar] [CrossRef] [Green Version]
- Medina, M.W.; Gao, F.; Ruan, W.; Rotter, J.I.; Krauss, R.M. Alternative splicing of 3-hydroxy-3-methylglutaryl coenzyme a reductase is associated with plasma low-density lipoprotein cholesterol response to simvastatin. Circulation 2008, 118, 355–362. [Google Scholar] [CrossRef] [Green Version]
- Deana, R.; Meneghello, R.; Manzi, L.; Gregolin, C. Formation of acetoacetate from 3-hydroxy-3-methylglutarate by rat liver and isolation of a mitochondrial coenzyme A-transferase activity involved. Biochem. J. 1974, 138, 481–486. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schachter, M. Chemical, pharmacokinetic and pharmacodynamic properties of statins: An update. Fundam. Clin. Pharmacol. 2005, 19, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Michalets, E.L. Update: Clinically significant cytochrome P-450 drug interactions. Pharmacotherapy 1998, 18, 84–112. [Google Scholar]
- Sirtori, C.R. The pharmacology of statins. Pharmacol. Res. 2014, 88, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Hamelin, B.A.; Turgeon, J. Hydrophilicity/Lipophilicity: Relevance for the pharmacology and clinical effects of HMG-CoA reductase inhibitors. Trends Pharmacol. Sci. 1998, 19, 26–37. [Google Scholar] [CrossRef]
- Wood, W.G.; Mΰller, W.E.; Eckert, G.P. Statins and Neuroprotection: Basic Pharmacology Needed. Mol. Neurobiol. 2014, 50, 214–220. [Google Scholar] [CrossRef]
- Endres, M.; Laufs, U.; Liao, J.K.; Moskowitz, M.A. Targeting eNOS for stroke protection. Trends Neurosci. 2004, 27, 283–289. [Google Scholar] [CrossRef]
- Kureishi, Y.; Luo, Z.; Shiojima, I.; Bialik, A.; Fulton, D.; Lefer, D.J.; Sessa, W.C.; Walsh, K. The HMG-CoA reductase inhibitor simvastatin activates the protein kinase Akt and promotes angiogenesis in normocholesterolemic animals. Nat. Med. 2000, 6, 1004–1010. [Google Scholar] [CrossRef]
- Rikitake, Y.; Liao, J.K. Rho GTPases, statins, and nitric oxide. Circ. Res. 2005, 97, 1232–1235. [Google Scholar] [CrossRef] [Green Version]
- Laufs, U.; Liao, J.K. Post-transcriptional regulation of endothelial nitric oxide synthase mRNA stability by Rho GTPase. J. Biol. Chem. 1998, 273, 24266–24271. [Google Scholar] [CrossRef] [Green Version]
- Endres, M.; Laufs, U.; Huang, Z.; Nakamura, T.; Huang, P.; Moskowitz, M.A.; Liao, J.K. Stroke protection by 3-hydroxy-3-methylglutaryl (HMG)-CoA reductase inhibitors mediated by endothelial nitric oxide synthase. Proc. Natl. Acad. Sci. USA 1998, 95, 8880–8885. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stone, D.K.; Kiyota, T.; Mosley, R.L.; Gendelman, H.E. A model of nitric oxide induced α-synuclein misfolding in Parkinson’s disease. Neurosci. Lett. 2012, 523, 167–173. [Google Scholar] [CrossRef] [Green Version]
- van der Most, P.J.; Dolga, A.M.; Nijholt, I.M.; Luiten, P.G.; Eisel, U.L. Statins: Mechanisms of neuroprotection. Prog. Neurobiol. 2009, 88, 64–75. [Google Scholar] [CrossRef] [PubMed]
- Barnham, K.J.; Masters, C.L.; Bush, A.I. Neurodegenerative diseases and oxidative stress. Nat. Rev. Drug Discov. 2004, 3, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Wu, L.; Jiang, T.; Wang, Y.; Zhao, H.; Gao, Q.; Pan, Y.; Tian, Y.; Zhang, Y. Angiotensin AT2 receptor stimulation inhibits activation of NADPH oxidase and ameliorates oxidative stress in rotenone model of Parkinson’s disease in CATH.a cells. Neurotoxicol. Teratol. 2015, 47, 16–24. [Google Scholar] [CrossRef]
- Li, Q.; Zhuang, Q.K.; Yang, J.N.; Zhang, Y.Y. Statins excert neuroprotection on cerebral ischemia independent of their lipid-lowering action: The potential molecular mechanisms. Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 1113–1126. [Google Scholar]
- Poli, G.; Biasi, F.; Leonarduzzi, G. Oxysterols in the pathogenesis of major chronic diseases. Redox Biol. 2013, 1, 125–130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seet, R.C.; Lee, C.Y.; Lim, E.C.; Tan, J.J.; Quek, A.M.; Chong, W.L.; Looi, W.F.; Huang, S.H.; Wang, H.; Chan, Y.H.; et al. Oxidative damage in Parkinson disease: Measurement using accurate biomarkers. Free Radic. Biol. Med. 2010, 48, 560–566. [Google Scholar] [CrossRef]
- Kumagai, R.; Oki, C.; Muramatsu, Y.; Kurosaki, R.; Kato, H.; Araki, T. Pitavastatin, a 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase inhibitor, reduces hippocampal damage after transient cerebral ischemia in gerbils. J. Neural Transm. 2004, 111, 1103–1120. [Google Scholar] [CrossRef]
- Rogers, J.; Mastroeni, D.; Leonard, B.; Joyce, J.; Grover, A. Neuroinflammation in Alzheimer’s Disease and Parkinson’s Disease: Are Microglia Pathogenic in Either Disorder? Int. Rev. Neurobiol. 2007, 82, 235–246. [Google Scholar] [CrossRef]
- Marques, N.F.; Castro, A.A.; Mancini, G.; Rocha, F.L.; Santos, A.R.S.; Prediger, R.D.; De Bem, A.F.; Tasca, C.I. Atorvastatin Prevents Early Oxidative Events and Modulates Inflammatory Mediators in the Striatum Following Intranasal 1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) Administration in Rats. Neurotox. Res. 2018, 33, 549–559. [Google Scholar] [CrossRef]
- McFarland, A.J.; Davey, A.K.; McDermott, C.M.; Grant, G.D.; Lewohl, J.; Anoopkumar-Dukie, S. Differences in statin asso-ciated neuroprotection corresponds with either decreased production of IL-1β or TNF-α in an in vitro model of neuroin-flammation-induced neurodegeneration. Toxicol. Appl. Pharmacol. 2018, 344, 56–73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghosh, A.; Roy, A.; Matras, J.; Brahmachari, S.; Gendelman, H.E.; Pahan, K. Simvastatin inhibits the activation of p21ras and prevents the loss of dopaminergic neurons in a mouse model of Parkinson’s disease. J. Neurosci. 2009, 29, 13543–13556. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bar-On, P.; Crews, L.; Koob, A.O.; Mizuno, H.; Adame, A.; Spencer, B.; Masliah, E. Statins reduce neuronal alpha-synuclein aggregation in in vitro models of Parkinson’s disease. J. Neurochem. 2008, 105, 1656–1667. [Google Scholar] [CrossRef] [PubMed]
- Fortin, D.L.; Troyer, M.D.; Nakamura, K.; Kubo, S.; Anthony, M.D.; Edwards, R.H. Lipid rafts mediate the synaptic locali-zation of alpha-synuclein. J. Neurosci. 2004, 24, 6715–6723. [Google Scholar] [CrossRef] [Green Version]
- Koob, A.O.; Ubhi, K.; Paulsson, J.F.; Kelly, J.; Rockenstein, E.; Mante, M.; Adame, A.; Masliah, E. Lovastatin ameliorates alpha-synuclein accumulation and oxidation in transgenic mouse models of alpha-synucleinopathies. Exp. Neurol. 2010, 221, 267–274. [Google Scholar] [CrossRef] [Green Version]
- Deck, B.L.; Rick, J.; Xie, S.X.; Chen-Plotkin, A.; Duda, J.E.; Morley, J.F.; Chahine, L.M.; Dahodwala, N.; Trojanowski, J.Q.; Weintraub, D. Statins and Cognition in Parkinson’s Disease. J. Parkinsons Dis. 2017, 7, 661–667. [Google Scholar] [CrossRef] [Green Version]
- Palermo, G.; Giannoni, S.; Giuntini, M.; Belli, E.; Frosini, D.; Siciliano, G.; Ceravolo, R. Statins in Parkinson’s Disease: Influence on Motor Progression. J. Parkinsons Dis. 2021, 11, 1651–1662. [Google Scholar] [CrossRef]
- Bykov, K.; Yoshida, K.; Weisskopf, M.G.; Gagne, J.J. Confounding of the association between statins and Parkinson disease: Systematic review and meta-analysis. Pharmacoepidemiol. Drug Saf. 2017, 26, 294–300. [Google Scholar] [CrossRef]
- Lee, Y.C.; Lin, C.H.; Wu, R.M.; Lin, M.S.; Lin, J.W.; Chang, C.H.; Lai, M.S. Discontinuation of statin therapy associates with Parkinson disease: A population-based study. Neurology 2013, 81, 410–416. [Google Scholar] [CrossRef]
- Krauss, R.M.; Mangravite, L.M.; Smith, J.D.; Medina, M.W.; Wang, D.; Guo, X.; Rieder, M.J.; Simon, J.A.; Hulley, S.B.; Wa-ters, D.; et al. Variation in the 3-hydroxyl-3-methylglutaryl coenzyme A reductase gene is associated with racial differences in low-density lipoprotein cholesterol response to simvastatin treatment. Circulation 2008, 117, 1537–1544. [Google Scholar] [CrossRef] [PubMed]
- Chasman, D.I.; Posada, D.; Subrahmanyan, L.; Cook, N.R.; Stanton, V.P., Jr.; Ridker, P.M. Pharmacogenetic study of statin therapy and cholesterol reduction. JAMA 2004, 291, 2821–2827. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Medina, M.W.; Sangkuhl, K.; Klein, T.E.; Altman, R.B. PharmGKB: Very important pharmacogene—HMGCR. Pharmacogenet. Genom. 2011, 21, 98–101. [Google Scholar] [CrossRef] [PubMed]
- Medina, M.W.; Krauss, R.M. The Role of HMGCR Alternative Splicing in Statin Efficacy. Trends Cardiovasc. Med. 2009, 19, 173–177. [Google Scholar] [CrossRef] [Green Version]
- Leduc, V.; Bourque, L.; Poirier, J.; Dufour, R. Role of rs3846662 and HMGCR alternative splicing in statin efficacy and baseline lipid levels in familial hypercholesterolemia. Pharmacogenet Genom. 2016, 26, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cano-Corres, R.; Candás-Estébanez, B.; Padró-Miquel, A.; Fanlo-Maresma, M.; Pintó, X.; Alía-Ramos, P. Influence of 6 genetic variants on the efficacy of statins in patients with dyslipidemia. J. Clin. Lab. Anal. 2018, 32, e22566. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chung, J.Y.; Cho, S.K.; Oh, E.S.; Lee, D.H.; Lim, L.A.; Jang, S.B.; Lee, Y.J.; Park, K.; Park, M.S. Effect of HMGCR variant alleles on low-density lipoprotein cholesterol-lowering response to atorvastatin in healthy Korean subjects. J. Clin. Pharmacol. 2012, 52, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, L.A.; Doney, A.S.; Dannfald, J.; Whitley, A.L.; Lang, C.C.; Morris, A.D.; Donnan, P.T.; Palmer, C.N. A paucimorphic variant in the HMG-CoA reductase gene is associated with lipid-lowering response to statin treatment in diabetes: A GoDARTS study. Pharmacogenet. Genom. 2008, 18, 1021–1026. [Google Scholar] [CrossRef]
- Chang, X.L.; Tan, L.; Tan, M.S.; Wang, H.F.; Tan, C.C.; Zhang, W.; Zheng, Z.J.; Kong, L.L.; Wang, Z.X.; Jiang, T.; et al. Association of HMGCR polymorphism with late-onset Alzheimer’s disease in Han Chinese. Oncotarget 2016, 7, 22746–22751. [Google Scholar] [CrossRef] [Green Version]
- Wright, S.M.; Jensen, S.L.; Cockriel, K.L.; Davis, B.; Tschanz, J.T.; Munger, R.G.; Corcoran, C.D.; Kauwe, J.S.K. Association study of rs3846662 with Alzheimer’s disease in a population-based cohort: The Cache County Study. Neurobiol. Aging 2019, 84, e1–e242. [Google Scholar] [CrossRef]
- Keller, L.; Murphy, C.; Wang, H.X.; Fratiglioni, L.; Olin, M.; Gafvels, M.; Björkhem, I.; Graff, C.; Meaney, S. A functional polymorphism in the HMGCR promoter affects transcriptional activity but not the risk for Alzheimer disease in Swedish populations. Brain Res. 2010, 1344, 185–191. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Licastro, F.; Porcellini, E.; Caruso, C.; Lio, D.; Corder, E.H. Genetic risk profiles for Alzheimer’s disease: Integration of APOE genotype and variants that up-regulate inflammation. Neurobiol. Aging 2007, 28, 1637–1643. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Rodríguez, E.; Mateo, I.; Infante, J.; Llorca, J.; García-Gorostiaga, I.; Vázquez-Higuera, J.L.; Sánchez-Juan, P.; Berciano, J.; Combarros, O. Interaction between HMGCR and ABCA1 cholesterol-related genes modulates Alzheimer’s disease risk. Brain Res. 2009, 1280, 166–171. [Google Scholar] [CrossRef]
- Benn, M.; Nordestgaard, B.G.; Frikke-Schmidt, R.; Tybjærg-Hansen, A. Low LDL cholesterol, PCSK9 and HMGCR genetic variation, and risk of Alzheimer’s disease and Parkinson’s disease: Mendelian randomisation study. BMJ 2017, 357, j1648. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, G.; Shi, M.; Mosley, J.D.; Weng, C.; Zhang, Y.; Lee, M.T.M.; Jarvik, G.P.; Hakonarson, H.; Namjou-Khales, B.; Sleiman, P.; et al. A Mendelian Randomization Approach Using 3-HMG-Coenzyme-A Reductase Gene Variation to Evaluate the Association of Statin-Induced Low-Density Lipoprotein Cholesterol Lowering With Noncardiovascular Disease Phenotypes. JAMA Netw. Open 2021, 4, e2112820. [Google Scholar] [CrossRef] [PubMed]

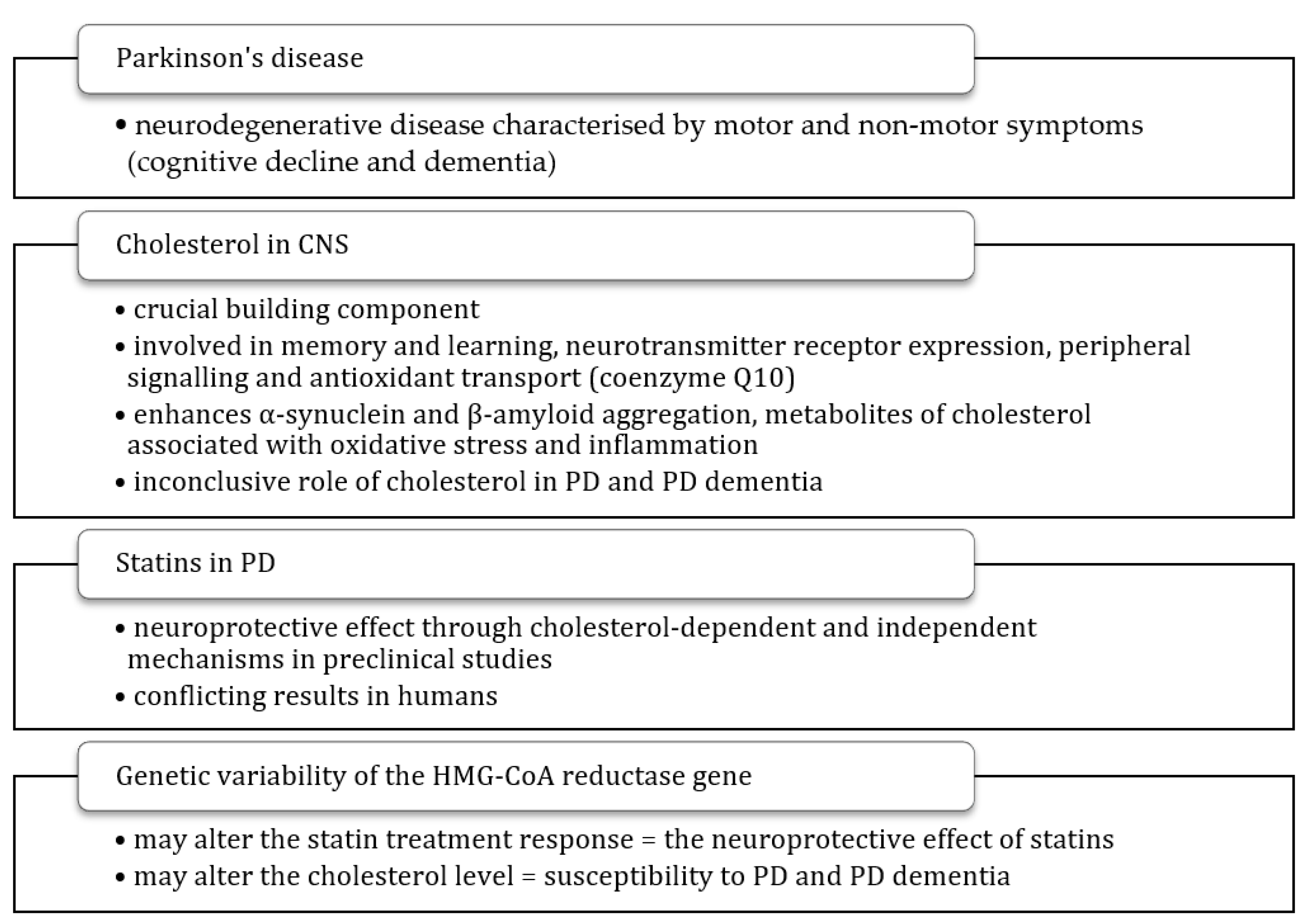
| Statins in PD | Influence of HMGCR Variants on Statin Treatment | Questions Raised |
|---|---|---|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pierzchlińska, A.; Droździk, M.; Białecka, M. A Possible Role for HMG-CoA Reductase Inhibitors and Its Association with HMGCR Genetic Variation in Parkinson’s Disease. Int. J. Mol. Sci. 2021, 22, 12198. https://doi.org/10.3390/ijms222212198
Pierzchlińska A, Droździk M, Białecka M. A Possible Role for HMG-CoA Reductase Inhibitors and Its Association with HMGCR Genetic Variation in Parkinson’s Disease. International Journal of Molecular Sciences. 2021; 22(22):12198. https://doi.org/10.3390/ijms222212198
Chicago/Turabian StylePierzchlińska, Anna, Marek Droździk, and Monika Białecka. 2021. "A Possible Role for HMG-CoA Reductase Inhibitors and Its Association with HMGCR Genetic Variation in Parkinson’s Disease" International Journal of Molecular Sciences 22, no. 22: 12198. https://doi.org/10.3390/ijms222212198
APA StylePierzchlińska, A., Droździk, M., & Białecka, M. (2021). A Possible Role for HMG-CoA Reductase Inhibitors and Its Association with HMGCR Genetic Variation in Parkinson’s Disease. International Journal of Molecular Sciences, 22(22), 12198. https://doi.org/10.3390/ijms222212198







