Tension Stimulation of Tenocytes in Aligned Hyaluronic Acid/Platelet-Rich Plasma-Polycaprolactone Core-Sheath Nanofiber Membrane Scaffold for Tendon Tissue Engineering
Abstract
1. Introduction
2. Results and Discussion
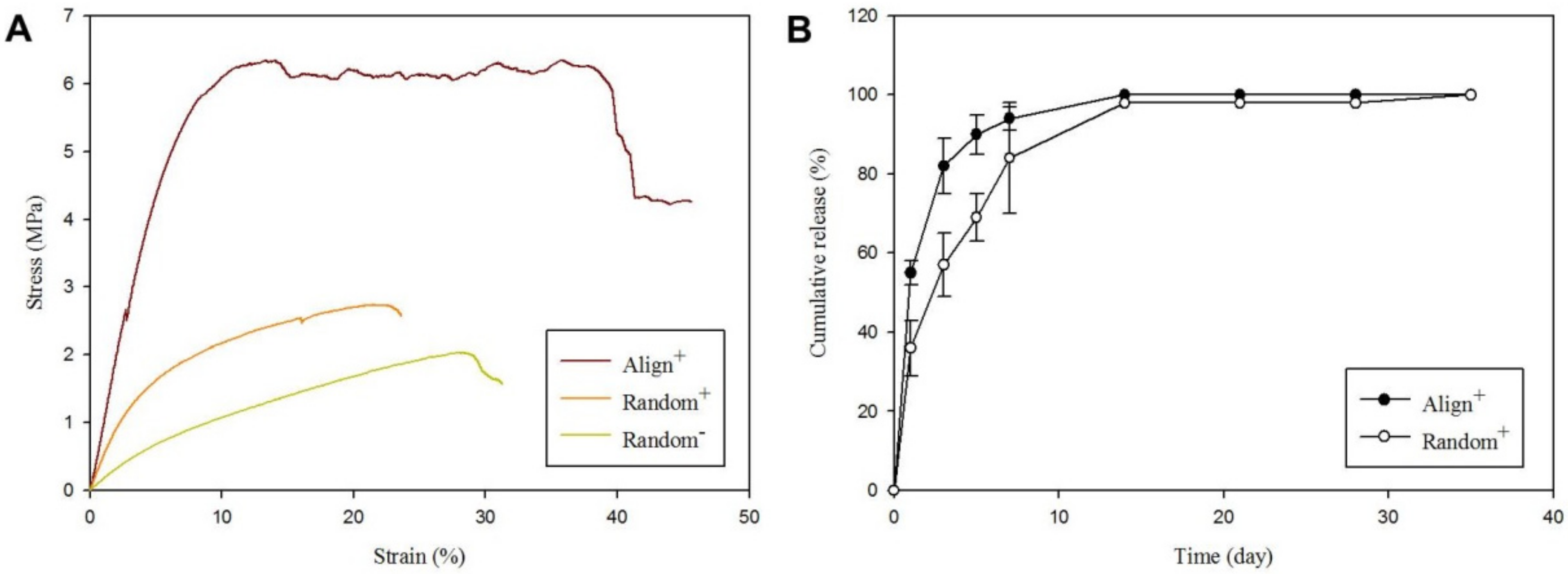
2.1. Physicochemical Properties of Core-Sheath Nanofiber Membrane Scaffolds (CSNMS)
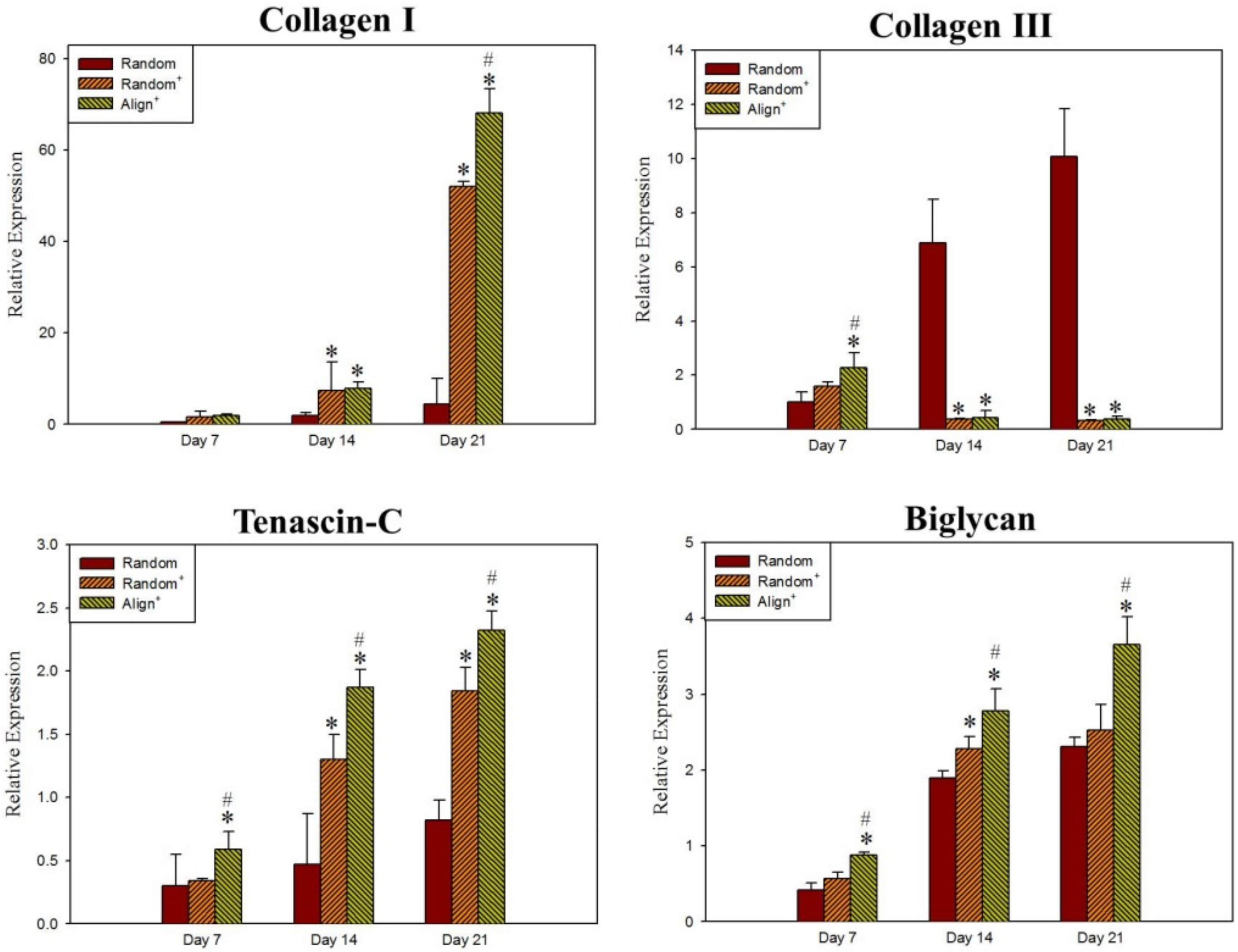
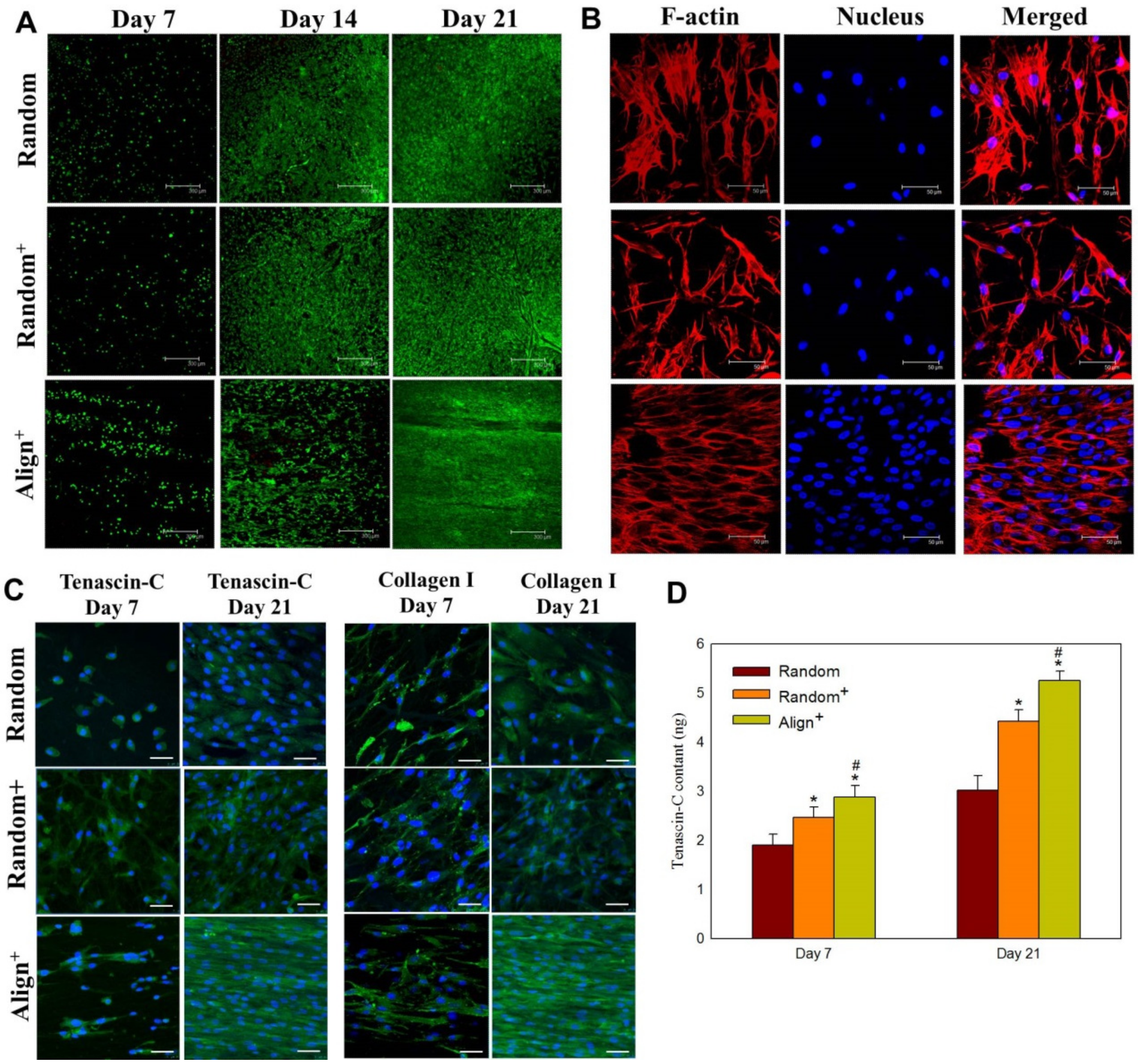
2.2. In Vitro Cell Culture
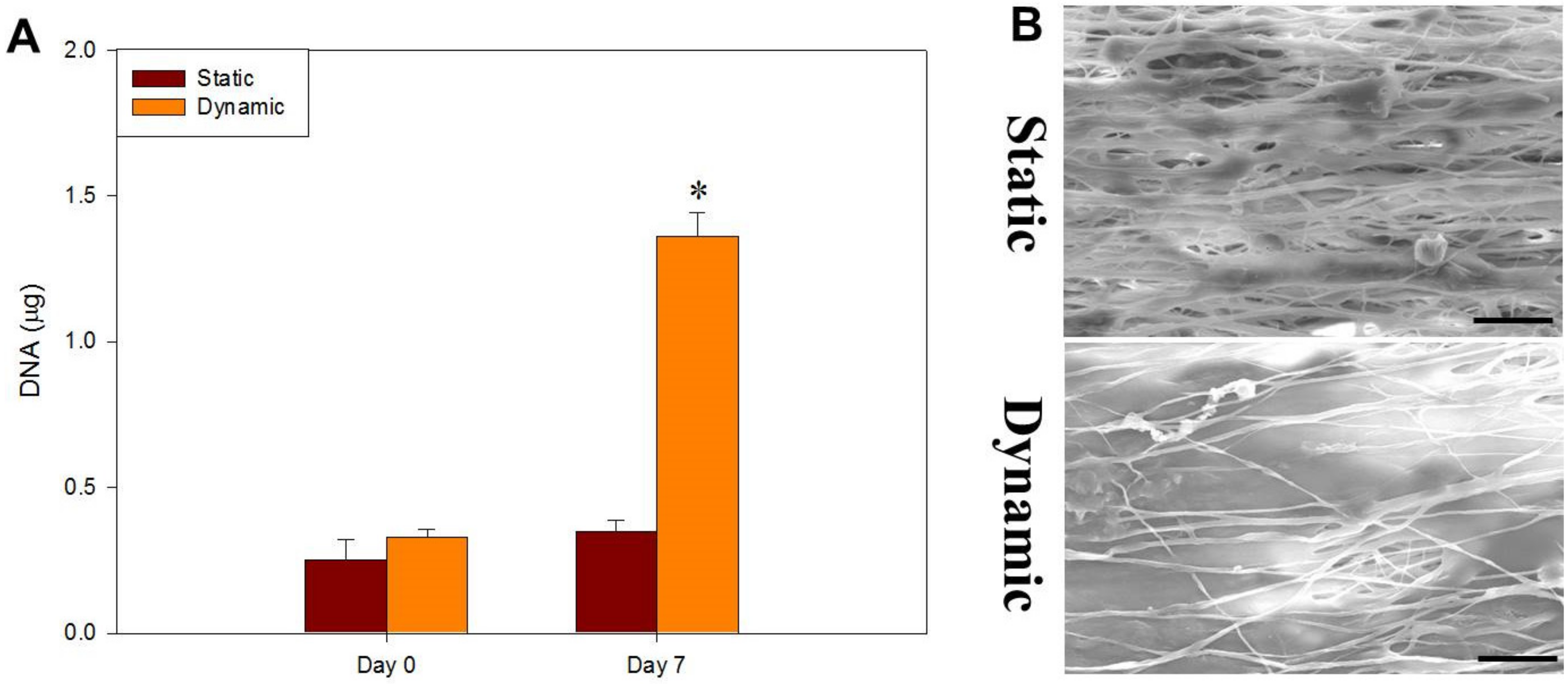
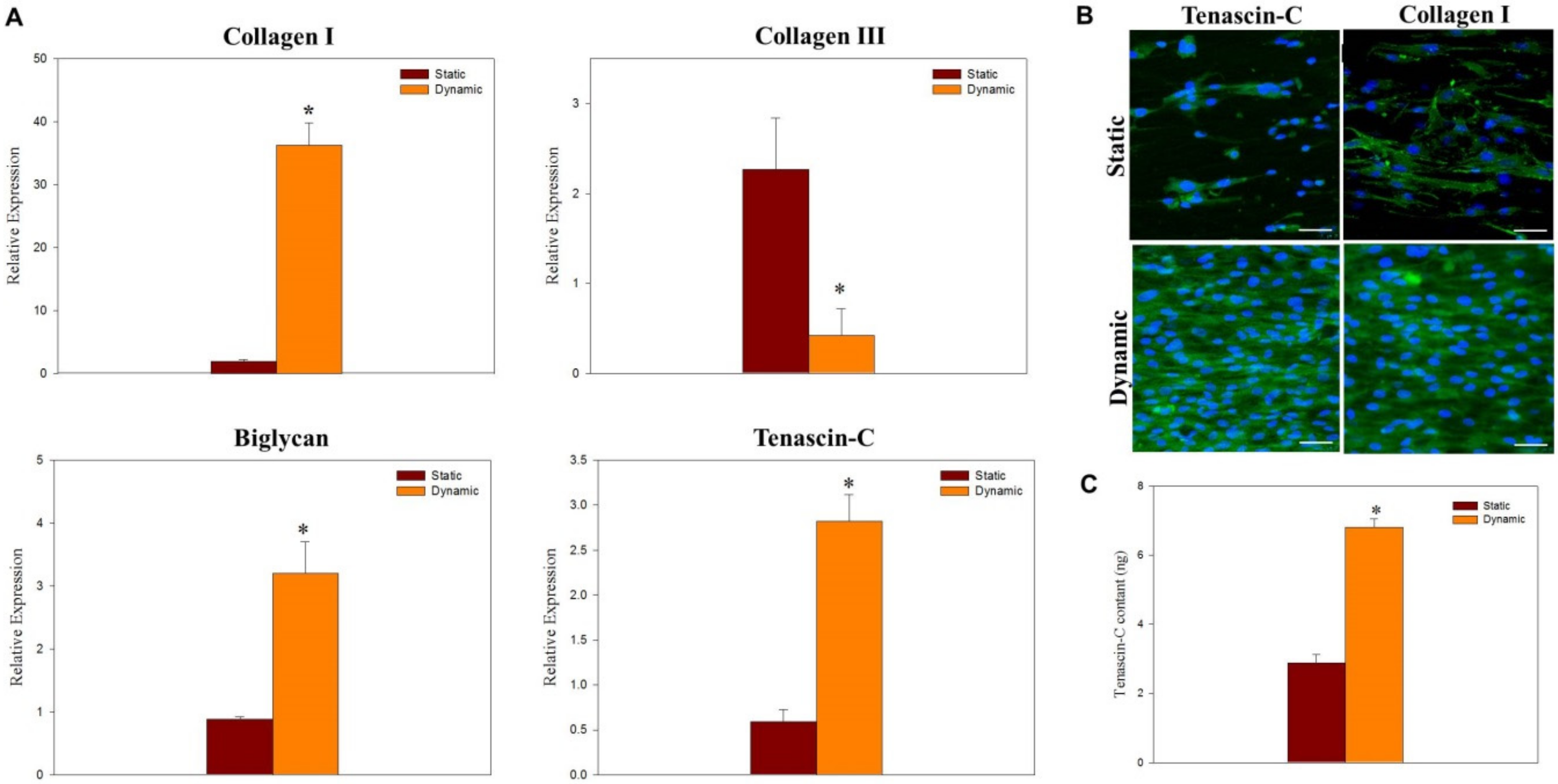
2.3. Dynamic Cell Culture
3. Materials and Methods
3.1. Materials
3.2. Preparation of Platelet-Rich Plasma (PRP)
3.3. Preparation of Core-Sheath Nanofiber Membrane Scaffold (CSNMS)
3.4. Characterization of Core-Sheath Nanofiber Membrane Scaffold (CSNMS)
3.5. In Vitro Cell Culture
3.5.1. Tenocyte Isolation
3.5.2. Cell Proliferation
3.5.3. Microscopy Observation
3.5.4. Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR)
3.6. Dynamic Cell Culture
3.7. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kaux, J.-F.; Bénédicte, F.; Le Goff, C.; Crielaard, J.-M.; Croisier, J.-L. Current opinions on tendinopathy. J. Sports Sci. Med. 2011, 10, 238–253. [Google Scholar]
- Barber, J.G.; Handorf, A.M.; Ailee, T.J.; Li, W.-J. Braided nanofibrous scaffold for tendon and ligament tissue engineering. Tissue Eng. Part A 2013, 19, 1265–1274. [Google Scholar] [CrossRef]
- Lipman, K.; Wang, C.; Ting, K.; Soo, C.; Zheng, Z. Tendinopathy: Injury, repair, and current exploration. Drug Des. Deliv. 2018, 12, 591–603. [Google Scholar] [CrossRef]
- Poggetti, A.; Novi, M.; Rosati, M.; Ciclamini, D.; Scaglione, M.; Battiston, B. Treatment of flexor tendon reconstruction failures: Multicentric experience with Brunelli active tendon implant. Eur. J. Orthop. Surg. Traumatol. 2018, 28, 877–883. [Google Scholar] [CrossRef]
- Wei, Z.; Reisdorf, R.L.; Thoreson, A.R.; Jay, G.D.; Moran, S.L.; An, K.-N.; Amadio, P.C.; Zhao, C. Comparison of Autograft and Allograft with Surface Modification for Flexor Tendon Reconstruction. J. Bone Jt. Surg. 2018, 100, 1–10. [Google Scholar] [CrossRef]
- Ouyang, H.W.; Goh, J.C.H.; Thambya, A.; Teoh, S.H.; Lee, E.H. Knitted poly-lactide-co-glycolide scaffold loaded with bone marrow stromal cells in repair and regeneration of rabbit Achilles tendon. Tissue Eng. 2003, 9, 431–439. [Google Scholar] [CrossRef]
- Snedeker, J.G.; Foolen, J. Tendon injury and repair—A perspective on the basic mechanisms of tendon disease and future clinical therapy. Acta Biomater. 2017, 63, 18–36. [Google Scholar] [CrossRef] [PubMed]
- Santoro, M.; Shah, S.R.; Walker, J.L.; Mikos, A.G. Poly(lactic acid) nanofibrous scaffolds for tissue engineering. Adv. Drug Del. Rev. 2016, 107, 206–212. [Google Scholar] [CrossRef]
- Luo, D.; Zhang, X.; Shahid, S.; Cattell, M.J.; Gould, D.J.; Sukhorukov, G.B. Electrospun poly(lactic acid) fibers containing novel chlorhexidine particles with sustained antibacterial activity. Biomater. Sci. 2017, 5, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Teh, T.K.H.; Toh, S.-L.; Goh, J.C.H. Aligned hybrid silk scaffold for enhanced differentiation of mesenchymal stem cells into ligament fibroblasts. Tissue Eng. Part C Methods 2011, 17, 687–703. [Google Scholar] [CrossRef]
- Teh, T.K.H.; Toh, S.-L.; Goh, J.C.H. Aligned fibrous scaffolds for enhanced mechanoresponse and tenogenesis of mesenchymal stem cells. Tissue Eng. Part A 2013, 19, 1360–1372. [Google Scholar] [CrossRef] [PubMed]
- Deepthi, S.; Nivedhitha Sundaram, M.; Deepti Kadavan, J.; Jayakumar, R. Layered chitosan-collagen hydrogel/aligned PLLA nanofiber construct for flexor tendon regeneration. Carbohydr. Polym. 2016, 153, 492–500. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, J.H.-C. The effects of mechanical loading on tendons—An in vivo and in vitro model study. PLoS ONE 2013, 8, e71740. [Google Scholar] [CrossRef] [PubMed]
- Yoshimi, R.; Yamada, Y.; Ito, K.; Nakamura, S.; Abe, A.; Nagasaka, T.; Okabe, K.; Kohgo, T.; Baba, S.; Ueda, M. Self-Assembling Peptide Nanofiber Scaffolds, Platelet-Rich Plasma, and Mesenchymal Stem Cells for Injectable Bone Regeneration with Tissue Engineering. J. Craniofac. Surg. 2009, 20, 1523–1530. [Google Scholar] [CrossRef]
- Wang, X.; Qiu, Y.; Triffitt, J.; Carr, A.; Xia, Z.; Sabokbar, A. Proliferation and differentiation of human tenocytes in response to platelet rich plasma: An in vitro and in vivo study. J. Orthop. Res. 2012, 30, 982–990. [Google Scholar] [CrossRef] [PubMed]
- Marx, R.E. Platelet-rich plasma (PRP): What is PRP and what is not PRP? Implant. Dent. 2001, 10, 225–228. [Google Scholar] [CrossRef]
- Andia, I.; Martin, J.I.; Maffulli, N. Advances with platelet rich plasma therapies for tendon regeneration. Expert Opin. Biol. Ther. 2018, 18, 389–398. [Google Scholar] [CrossRef]
- Docheva, D.; Müller, S.A.; Majewski, M.; Evans, C.H. Biologics for tendon repair. Adv. Drug Deliv. Rev. 2015, 84, 222–239. [Google Scholar] [CrossRef]
- Sell, S.A.; Wolfe, P.S.; Ericksen, J.J.; Simpson, D.G.; Bowlin, G.L. Incorporating platelet-rich plasma into electrospun scaffolds for tissue engineering applications. Tissue Eng. Part A 2011, 17, 2723–2737. [Google Scholar] [CrossRef]
- Szentivanyi, A.; Chakradeo, T.; Zernetsch, H.; Glasmacher, B. Electrospun cellular microenvironments: Understanding controlled release and scaffold structure. Adv. Drug Deliv. Rev. 2011, 63, 209–220. [Google Scholar] [CrossRef]
- Diaz-Gomez, L.; Alvarez-Lorenzo, C.; Concheiro, A.; Silva, M.; Dominguez, F.; Sheikh, F.A.; Cantu, T.; Desai, R.; Garcia, V.L.; Macossay, J. Biodegradable electrospun nanofibers coated with platelet-rich plasma for cell adhesion and proliferation. Mater. Sci. Eng. C 2014, 40, 180–188. [Google Scholar] [CrossRef]
- Liu, J.; Nie, H.; Xu, Z.; Guo, F.; Guo, S.; Yin, J.; Wang, Y.; Zhang, C. Construction of PRP-containing nanofibrous scaffolds for controlled release and their application to cartilage regeneration. J. Mater. Chem. B 2015, 3, 581–591. [Google Scholar] [CrossRef]
- Xu, X.; Yang, L.; Xu, X.; Wang, X.; Chen, X.; Liang, Q.; Zeng, J.; Jing, X. Ultrafine medicated fibers electrospun from W/O emulsions. J. Control. Release 2005, 108, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Perez, R.A.; Kim, H.-W. Core–shell designed scaffolds for drug delivery and tissue engineering. Acta Biomater. 2015, 21, 2–19. [Google Scholar] [CrossRef]
- Cheng, G.; Ma, X.; Li, J.; Cheng, Y.; Cao, Y.; Wang, Z.; Shi, X.; Du, Y.; Deng, H.; Li, Z. Incorporating platelet-rich plasma into coaxial electrospun nanofibers for bone tissue engineering. Int. J. Pharm. 2018, 547, 656–666. [Google Scholar] [CrossRef]
- Kaux, J.F.; Samson, A.; Crielaard, J.M. Hyaluronic acid and tendon lesions. Muscles Ligaments Tendons J. 2015, 5, 264–269. [Google Scholar] [CrossRef]
- Osti, L.; Berardocco, M.; di Giacomo, V.; Di Bernardo, G.; Oliva, F.; Berardi, A.C. Hyaluronic acid increases tendon derived cell viability and collagen type I expression in vitro: Comparative study of four different Hyaluronic acid preparations by molecular weight. BMC Musculoskelet. Disord. 2015, 16, 284. [Google Scholar] [CrossRef]
- Waggett, A.D.; Benjamin, M.; Ralphs, J.R. Connexin 32 and 43 gap junctions differentially modulate tenocyte response to cyclic mechanical load. Eur. J. Cell Biol. 2006, 85, 1145–1154. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Liu, H.; Li, H.; Wang, F.; Cheng, K.; Zhou, G.; Zhang, W.; Ye, M.; Cao, Y.; Liu, W.; et al. A proteomic analysis of engineered tendon formation under dynamic mechanical loading in vitro. Biomaterials 2011, 32, 4085–4095. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Im, H.-J.; Wang, J.H.C. Repetitive mechanical stretching modulates IL-1β induced COX-2, MMP-1 expression, and PGE2 production in human patellar tendon fibroblasts. Gene 2005, 363, 166–172. [Google Scholar] [CrossRef] [PubMed]
- Tsuzaki, M.; Bynum, D.; Almekinders, L.; Yang, X.; Faber, J.; Banes, A.J. ATP modulates load-inducible IL-1β, COX 2, and MMP-3 gene expression in human tendon cells. J. Cell. Biochem. 2003, 89, 556–562. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Chen, P.; Zheng, M.; Wang, A.; Lloyd, D.; Leys, T.; Zheng, Q.; Zheng, M.H. In vitro loading models for tendon mechanobiology. J. Orthop. Res. 2018, 36, 566–575. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Li, J.; Wang, B.; Zhang, W.J.; Zhou, G.; Cao, Y.; Liu, W. The regulation of phenotype of cultured tenocytes by microgrooved surface structure. Biomaterials 2010, 31, 6952–6958. [Google Scholar] [CrossRef] [PubMed]
- Zhong, S.; Teo, W.E.; Zhu, X.; Beuerman, R.W.; Ramakrishna, S.; Yung, L.Y.L. An aligned nanofibrous collagen scaffold by electrospinning and its effects on in vitro fibroblast culture. J. Biomed. Mater. Res. Part A 2006, 79A, 456–463. [Google Scholar] [CrossRef] [PubMed]
- Erisken, C.; Zhang, X.; Moffat, K.L.; Levine, W.N.; Lu, H.H. Scaffold Fiber Diameter Regulates Human Tendon Fibroblast Growth and Differentiation. Tissue Eng. Part A 2013, 19, 519–528. [Google Scholar] [CrossRef] [PubMed]
- Jose, M.V.; Thomas, V.; Johnson, K.T.; Dean, D.R.; Nyairo, E. Aligned PLGA/HA nanofibrous nanocomposite scaffolds for bone tissue engineering. Acta Biomater. 2009, 5, 305–315. [Google Scholar] [CrossRef] [PubMed]
- Luong-Van, E.; Grøndahl, L.; Chua, K.N.; Leong, K.W.; Nurcombe, V.; Cool, S.M. Controlled release of heparin from poly(epsilon-caprolactone) electrospun fibers. Biomaterials 2006, 27, 2042–2050. [Google Scholar] [CrossRef]
- Liu, P.; He, J.-H. Geometric potential: An explanation of nanofiber’s wettability. Therm. Sci. 2017, 22, 33–38. [Google Scholar] [CrossRef]
- Peng, N.-B.; He, J.-H. Insight into the Wetting Property of a Nanofiber Membrane by the Geometrical Potential. Recent Pat. Nanotechnol. 2020, 14, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.-A.; Ma, Y.-H.; Hsu, T.-Y.; Chen, J.-P. Preparation of Peptide and Recombinant Tissue Plasminogen Activator Conjugated Poly(Lactic-Co-Glycolic Acid) (PLGA) Magnetic Nanoparticles for Dual Targeted Thrombolytic Therapy. Int. J. Mol. Sci. 2020, 21, 2690. [Google Scholar] [CrossRef]
- Milleret, V.; Simona, B.; Neuenschwander, P.; Hall, H. Tuning electrospinning parameters for production of 3D-fiber-fleeces with increased porosity for soft tissue engineering applications. Eur. Cells Mater. 2011, 21, 286–303. [Google Scholar] [CrossRef]
- Chen, C.-H.; Chen, S.-H.; Kuo, C.-Y.; Li, M.-L.; Chen, J.-P. Response of Dermal Fibroblasts to Biochemical and Physical Cues in Aligned Polycaprolactone/Silk Fibroin Nanofiber Scaffolds for Application in Tendon Tissue Engineering. Nanomaterials 2017, 7, 219. [Google Scholar] [CrossRef]
- Wang, X.; Zhao, H.; Turng, L.-S.; Li, Q. Crystalline Morphology of Electrospun Poly(ε-caprolactone) (PCL) Nanofibers. Ind. Eng. Chem. Res. 2013, 52, 4939–4949. [Google Scholar] [CrossRef]
- Xu, K.; Al-ani, M.K.; Sun, Y.; Xu, W.; Pan, L.; Song, Y.; Xu, Z.; Pan, X.; Yang, L. Platelet-rich plasma activates tendon-derived stem cells to promote regeneration of Achilles tendon rupture in rats. J. Tissue Eng. Regen. Med. 2017, 11, 1173–1184. [Google Scholar] [CrossRef] [PubMed]
- Jo, C.H.; Kim, J.E.; Yoon, K.S.; Shin, S. Platelet-Rich Plasma Stimulates Cell Proliferation and Enhances Matrix Gene Expression and Synthesis in Tenocytes from Human Rotator Cuff Tendons with Degenerative Tears. Am. J. Sports Med. 2012, 40, 1035–1045. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; Zhang, Y.; Liu, Y.; Wang, Y.; Cao, F.; Yang, Q.; Tian, F. Explanation of the cell orientation in a nanofiber membrane by the geometric potential theory. Results Phys. 2019, 15, 102537. [Google Scholar] [CrossRef]
- Kapoor, A.; Caporali, E.H.G.; Kenis, P.J.A.; Stewart, M.C. Microtopographically patterned surfaces promote the alignment of tenocytes and extracellular collagen. Acta Biomater. 2010, 6, 2580–2589. [Google Scholar] [CrossRef]
- Riley, G.P.; Harrall, R.L.; Constant, C.R.; Chard, M.D.; Cawston, T.E.; Hazleman, B.L. Tendon degeneration and chronic shoulder pain: Changes in the collagen composition of the human rotator cuff tendons in rotator cuff tendinitis. Ann. Rheum. Dis. 1994, 53, 359–366. [Google Scholar] [CrossRef]
- Goh, J.C.H.; Ouyang, H.W.; Teoh, S.H.; Chan, C.K.C.; Lee, E.-H. Tissue-engineering approach to the repair and regeneration of tendons and ligaments. Tissue Eng. 2003, 9, 31–44. [Google Scholar] [CrossRef]
- Citeroni, M.R.; Ciardulli, M.C.; Russo, V.; Della Porta, G.; Mauro, A.; El Khatib, M.; Di Mattia, M.; Galesso, D.; Barbera, C.; Forsyth, N.R.; et al. In Vitro Innovation of Tendon Tissue Engineering Strategies. Int. J. Mol. Sci. 2020, 21, 6726. [Google Scholar] [CrossRef]
- Martin, J.A.; Mehr, D.; Pardubsky, P.D.; Buckwalter, J.A. The role of tenascin-C in adaptation of tendons to compressive loading. Biorheology 2003, 40, 321–329. [Google Scholar]
- Sawaguchi, N.; Majima, T.; Funakoshi, T.; Shimode, K.; Harada, K.; Minami, A.; Nishimura, S.-I. Effect of cyclic three-dimensional strain on cell proliferation and collagen synthesis of fibroblast-seeded chitosan-hyaluronan hybrid polymer fiber. J. Orthop. Sci. 2010, 15, 569–577. [Google Scholar] [CrossRef] [PubMed]
- Ferraris, S.; Spriano, S.; Scalia, A.C.; Cochis, A.; Rimondini, L.; Cruz-Maya, I.; Guarino, V.; Varesano, A.; Vineis, C. Topographical and Biomechanical Guidance of Electrospun Fibers for Biomedical Applications. Polymers 2020, 12, 2896. [Google Scholar] [CrossRef]
- Wang, Y.; Jin, S.; Luo, D.; He, D.; Shi, C.; Zhu, L.; Guan, B.; Li, Z.; Zhang, T.; Zhou, Y.; et al. Functional regeneration and repair of tendons using biomimetic scaffolds loaded with recombinant periostin. Nat. Commun. 2021, 12, 1293. [Google Scholar] [CrossRef] [PubMed]
- Schenk, S.; Chiquet-Ehrismann, R.; Battegay, E.J. The fibrinogen globe of tenascin-C promotes basic fibroblast growth factor-induced endothelial cell elongation. Mol. Biol. Cell 1999, 10, 2933–2943. [Google Scholar] [CrossRef][Green Version]
- Wenk, M.B.; Midwood, K.S.; Schwarzbauer, J.E. Tenascin-C suppresses Rho activation. J. Cell Biol. 2000, 150, 913–920. [Google Scholar] [CrossRef] [PubMed]
- Yao, L.; Bestwick, C.S.; Bestwick, L.A.; Maffulli, N.; Aspden, R.M. Phenotypic drift in human tenocyte culture. Tissue Eng. 2006, 12, 1843–1849. [Google Scholar] [CrossRef]
- Vermeulen, S.; Vasilevich, A.; Tsiapalis, D.; Roumans, N.; Vroemen, P.; Beijer, N.R.M.; Dede Eren, A.; Zeugolis, D.; de Boer, J. Identification of topographical architectures supporting the phenotype of rat tenocytes. Acta Biomater. 2019, 83, 277–290. [Google Scholar] [CrossRef]
- Caliari, S.R.; Harley, B.A. Composite growth factor supplementation strategies to enhance tenocyte bioactivity in aligned collagen-GAG scaffolds. Tissue Eng. Part A 2013, 19, 1100–1112. [Google Scholar] [CrossRef]
- Domingues, R.M.; Chiera, S.; Gershovich, P.; Motta, A.; Reis, R.L.; Gomes, M.E. Enhancing the Biomechanical Performance of Anisotropic Nanofibrous Scaffolds in Tendon Tissue Engineering: Reinforcement with Cellulose Nanocrystals. Adv. Healthc. Mater. 2016, 5, 1364–1375. [Google Scholar] [CrossRef]
- Martino, F.; Perestrelo, A.R.; Vinarský, V.; Pagliari, S.; Forte, G. Cellular Mechanotransduction: From Tension to Function. Front. Physiol. 2018, 9, 824. [Google Scholar] [CrossRef] [PubMed]
- Kreja, L.; Liedert, A.; Schlenker, H.; Brenner, R.E.; Fiedler, J.; Friemert, B.; Dürselen, L.; Ignatius, A. Effects of mechanical strain on human mesenchymal stem cells and ligament fibroblasts in a textured poly(L-lactide) scaffold for ligament tissue engineering. J. Mater. Sci. Mater. Med. 2012, 23, 2575–2582. [Google Scholar] [CrossRef] [PubMed]
- Liao, H.T.; Tsai, M.-J.; Brahmayya, M.; Chen, J.-P. Bone Regeneration Using Adipose-Derived Stem Cells in Injectable Thermo-Gelling Hydrogel Scaffold Containing Platelet-Rich Plasma and Biphasic Calcium Phosphate. Int. J. Mol. Sci. 2018, 19, 2537. [Google Scholar] [CrossRef] [PubMed]
- Liao, H.T.; Lai, Y.-T.; Kuo, C.-Y.; Chen, J.-P. A bioactive multi-functional heparin-grafted aligned poly(lactide-co-glycolide)/curcumin nanofiber membrane to accelerate diabetic wound healing. Mater. Sci. Eng. C 2021, 120, 111689. [Google Scholar] [CrossRef] [PubMed]
- Yew, C.H.T.; Azari, P.; Choi, J.R.; Muhamad, F.; Pingguan-Murphy, B. Electrospun Polycaprolactone Nanofibers as a Reaction Membrane for Lateral Flow Assay. Polymers 2018, 10, 1387. [Google Scholar] [CrossRef]
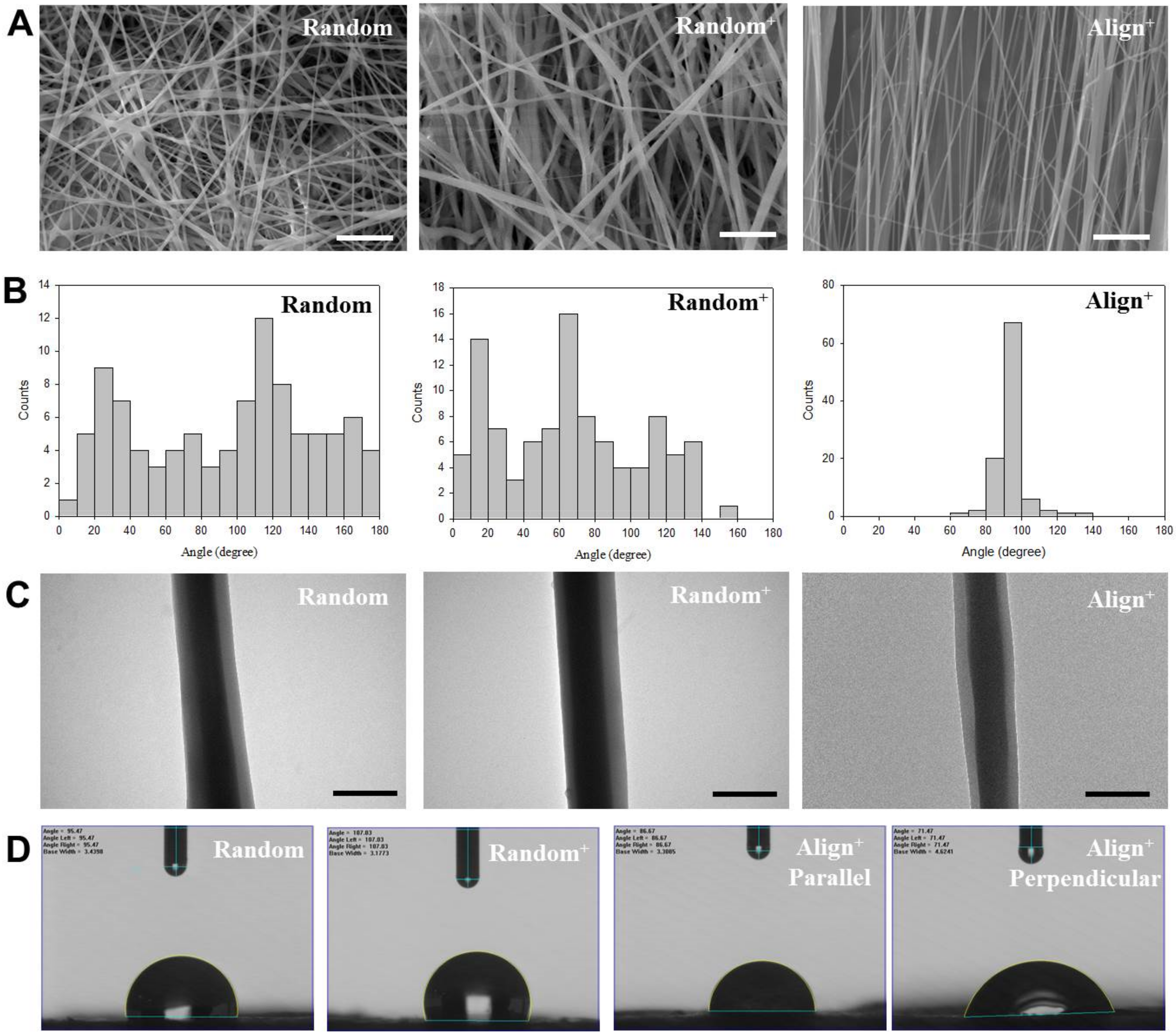


| Properties | Random | Random+ | Align+ |
|---|---|---|---|
| Core width (nm) | 164 ± 50 | 242 ± 61 | 241 ± 107 |
| Sheath width (nm) | 228 ± 98 | 286 ± 106 | 163 ± 134 |
| BET surface area (m2/g) | 22.2 | 17.9 | 28.5 |
| Porosity (%) | 83.1 ± 5.6 | 86.8 ± 1.8 | 69.4 ± 3.6 *,# |
| Density (g/cm3) | 0.28 ± 0.01 | 0.27 ± 0.02 | 0.35 ± 0.01 *,# |
| Water contact angle (degree) | 94.4 ± 5.1 | 96.7 ± 6.1 | 85.7 ± 8.9 (parallel) 73.1 ± 3.0 *,# (perpendicular) |
| CSNMS | Ultimate Stress (MPa) | Ultimate Strain (%) | Young’s Modulus (MPa) |
|---|---|---|---|
| Random | 2.21 ± 0.99 | 31.3 ± 1.4 | 8.90 ± 0.88 |
| Random+ | 1.61 ± 0.51 | 23.63 ± 1.1 * | 18.64 ± 4.08 * |
| Align+ | 6.62 ± 0.21 *,# | 45.65 ± 1.0 *,# | 76.02 ± 9.21 *,# |
| Gene | Sequence (5′ → 3′) | Size (Base Pairs) | |
|---|---|---|---|
| GADPH | Forward | GACATCAAGAAGGTGGTGAAGC | 22 |
| Reverse | CTTCACAAAGTGGTCATTGAGG | 22 | |
| Collagen I | Forward | GCATGTCTGGTTAGGAGAAACC | 22 |
| Reverse | ATGTATGCAATGCTGTTCTTGC | 21 | |
| Collagen III | Forward | AAGCCCCAGCAGAAAATTC | 19 |
| Reverse | TGGTGGAACAGCAAAAATCA | 20 | |
| Biglycan | Forward | AGATCTGCCAGAGACCCTGA | 20 |
| Reverse | ACCCTGGACAGCTTGTTGTT | 20 | |
| Tenascin-C | Forward | CTCTGCACATAGTGAAAAACAATACC | 27 |
| Reverse | TCAAGGCAGTGGTGTCTGTGA | 21 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, C.-H.; Li, D.-L.; Chuang, A.D.-C.; Dash, B.S.; Chen, J.-P. Tension Stimulation of Tenocytes in Aligned Hyaluronic Acid/Platelet-Rich Plasma-Polycaprolactone Core-Sheath Nanofiber Membrane Scaffold for Tendon Tissue Engineering. Int. J. Mol. Sci. 2021, 22, 11215. https://doi.org/10.3390/ijms222011215
Chen C-H, Li D-L, Chuang AD-C, Dash BS, Chen J-P. Tension Stimulation of Tenocytes in Aligned Hyaluronic Acid/Platelet-Rich Plasma-Polycaprolactone Core-Sheath Nanofiber Membrane Scaffold for Tendon Tissue Engineering. International Journal of Molecular Sciences. 2021; 22(20):11215. https://doi.org/10.3390/ijms222011215
Chicago/Turabian StyleChen, Chih-Hao, Dai-Ling Li, Andy Deng-Chi Chuang, Banendu Sunder Dash, and Jyh-Ping Chen. 2021. "Tension Stimulation of Tenocytes in Aligned Hyaluronic Acid/Platelet-Rich Plasma-Polycaprolactone Core-Sheath Nanofiber Membrane Scaffold for Tendon Tissue Engineering" International Journal of Molecular Sciences 22, no. 20: 11215. https://doi.org/10.3390/ijms222011215
APA StyleChen, C.-H., Li, D.-L., Chuang, A. D.-C., Dash, B. S., & Chen, J.-P. (2021). Tension Stimulation of Tenocytes in Aligned Hyaluronic Acid/Platelet-Rich Plasma-Polycaprolactone Core-Sheath Nanofiber Membrane Scaffold for Tendon Tissue Engineering. International Journal of Molecular Sciences, 22(20), 11215. https://doi.org/10.3390/ijms222011215








