Aesculus hippocastanum L. as a Stabilizer in Hemp Seed Oil Nanoemulsions for Potential Biomedical and Food Applications
Abstract
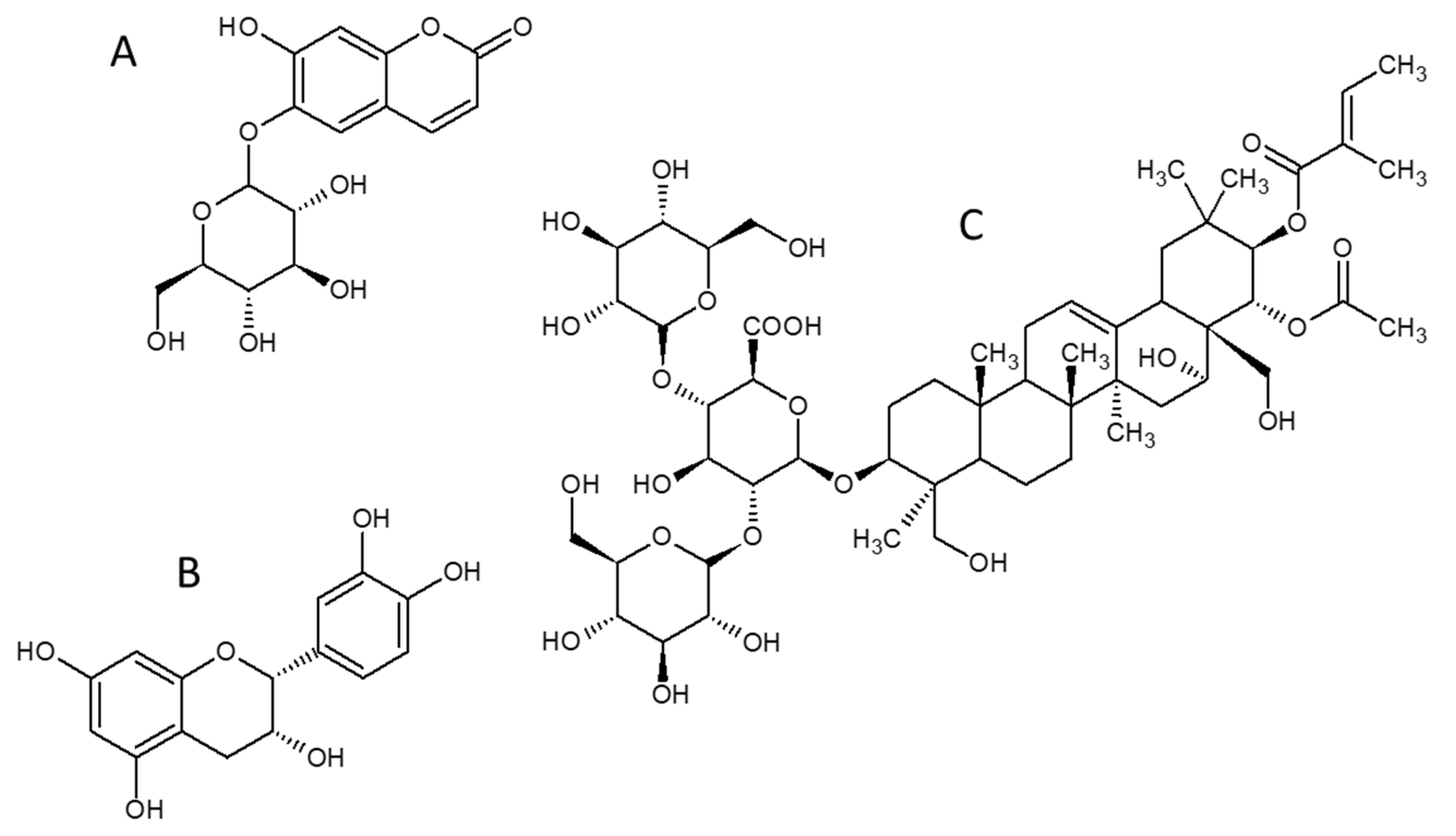
1. Introduction
2. Results and Discussion
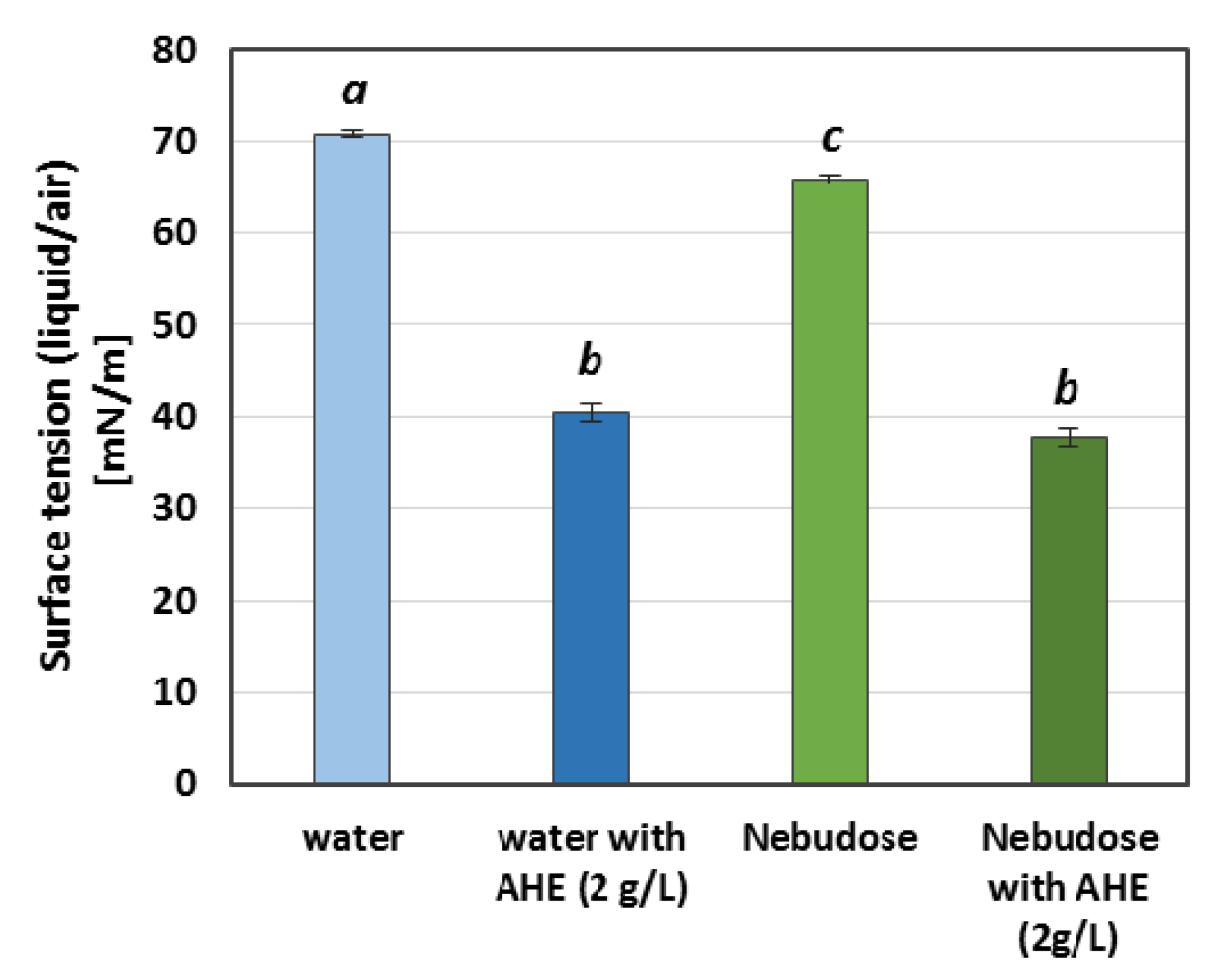
2.1. Emulsion Stability Tests
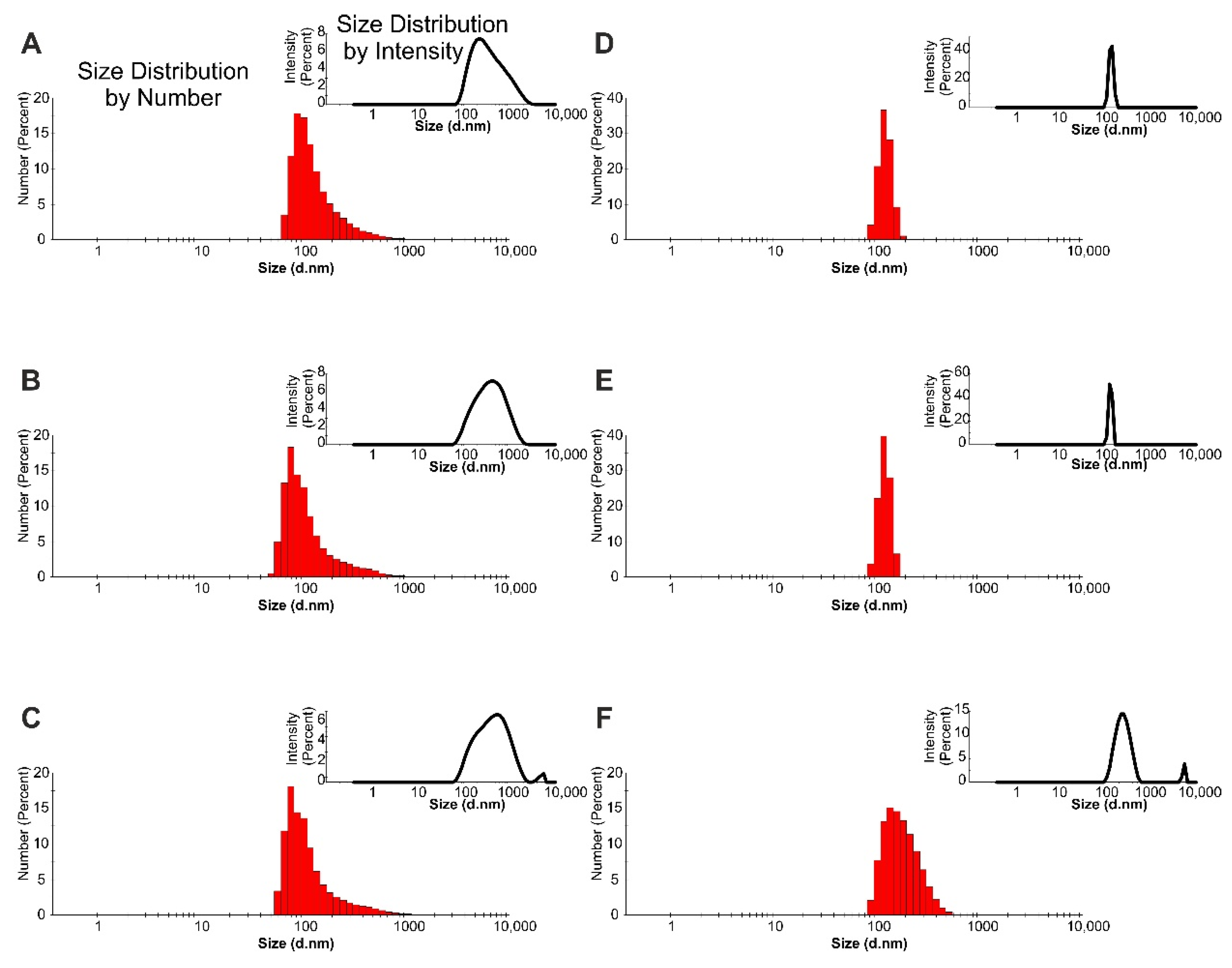
2.2. Droplet Size Determination
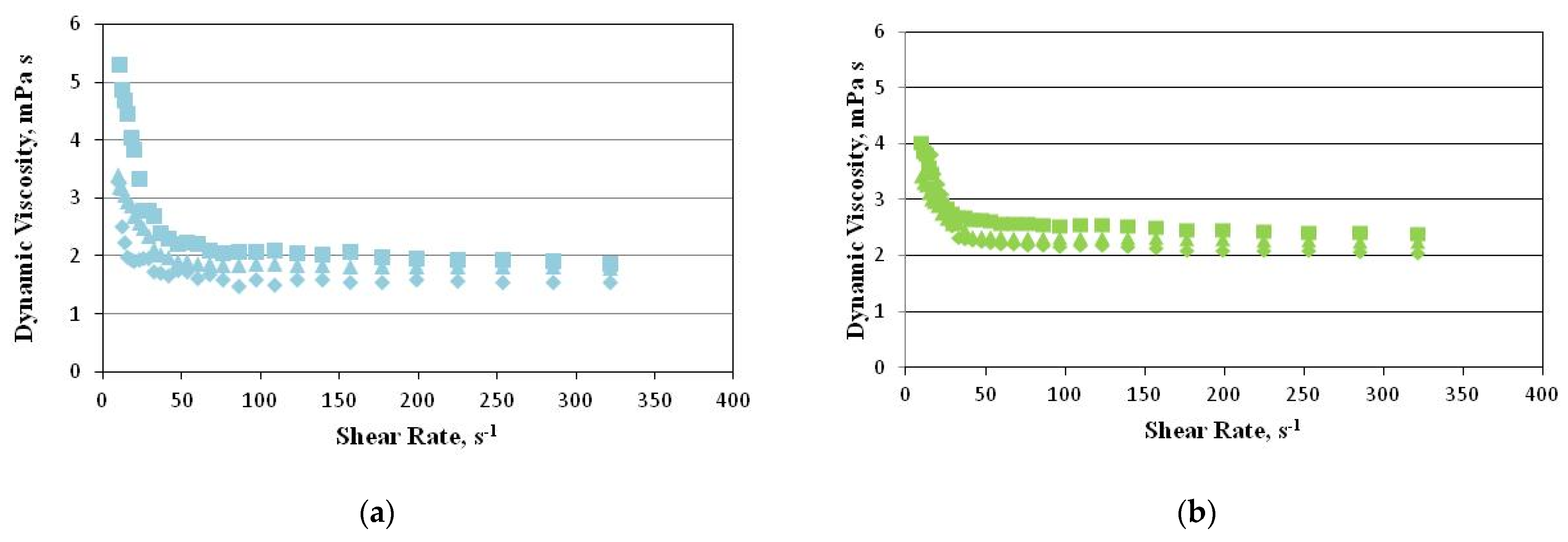
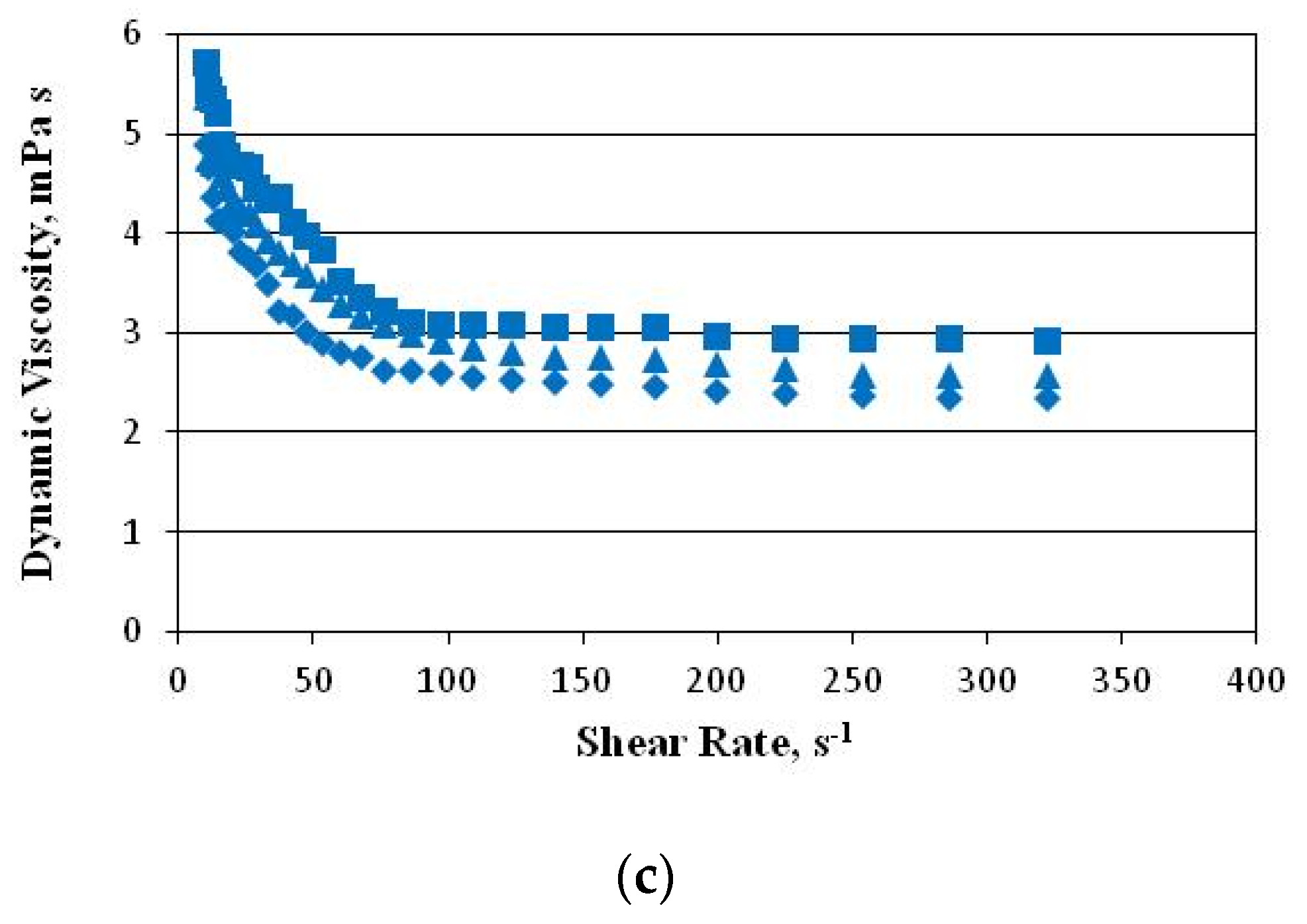
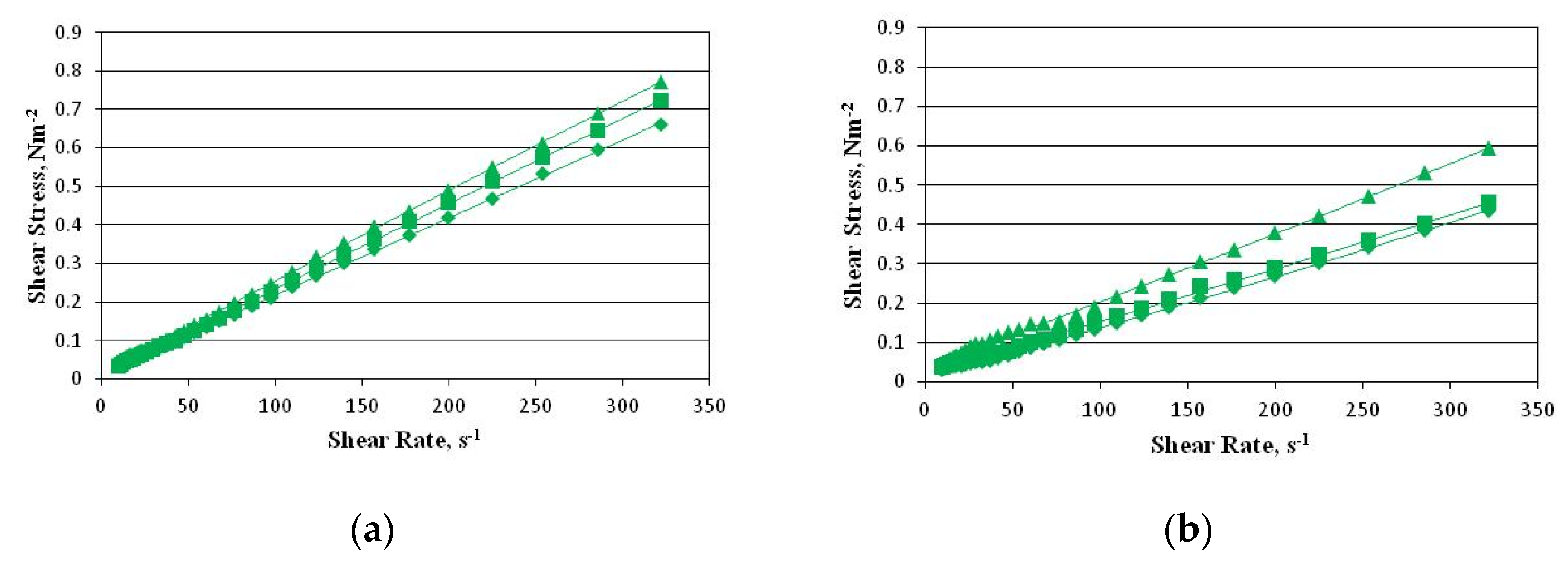
2.3. Rheological Properties
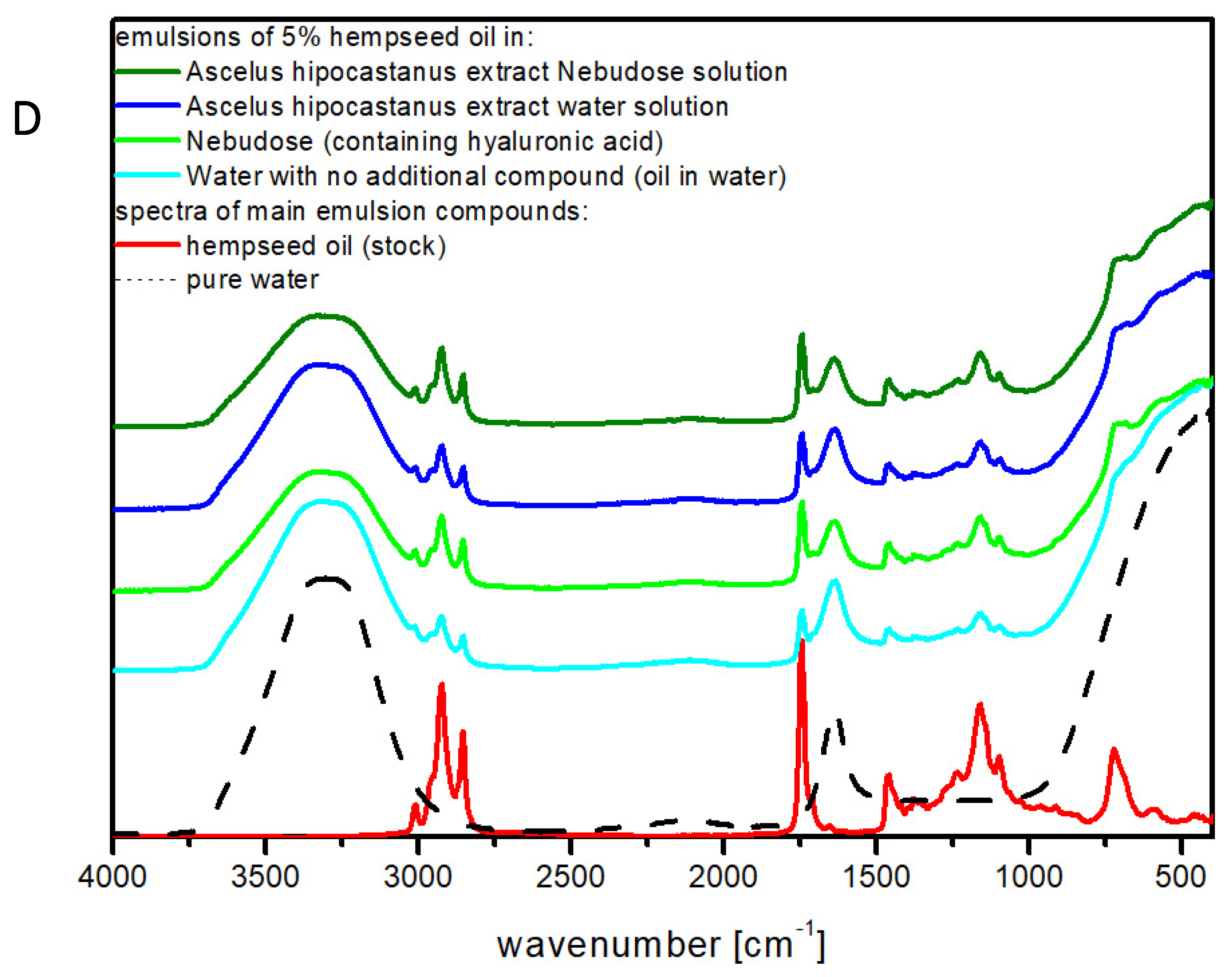
2.4. FTIR Studies of the Emulsion Systems
2.5. Microscopic Analysis
3. Materials and Methods
3.1. Reagents
3.2. Emulsion Preparation
3.3. Methods
3.3.1. Stability Tests
3.3.2. Surface Tension
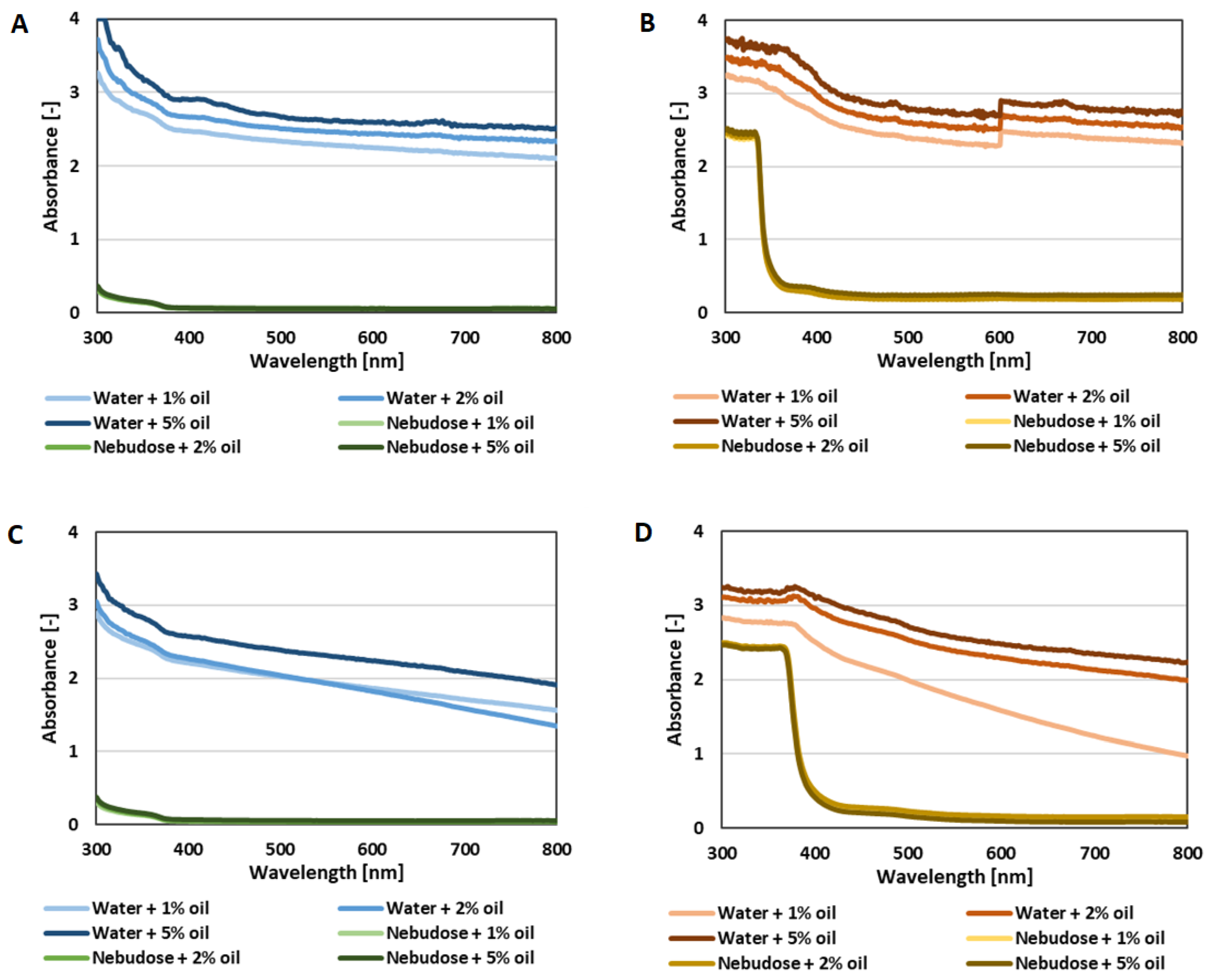
3.3.3. UV-Vis Test
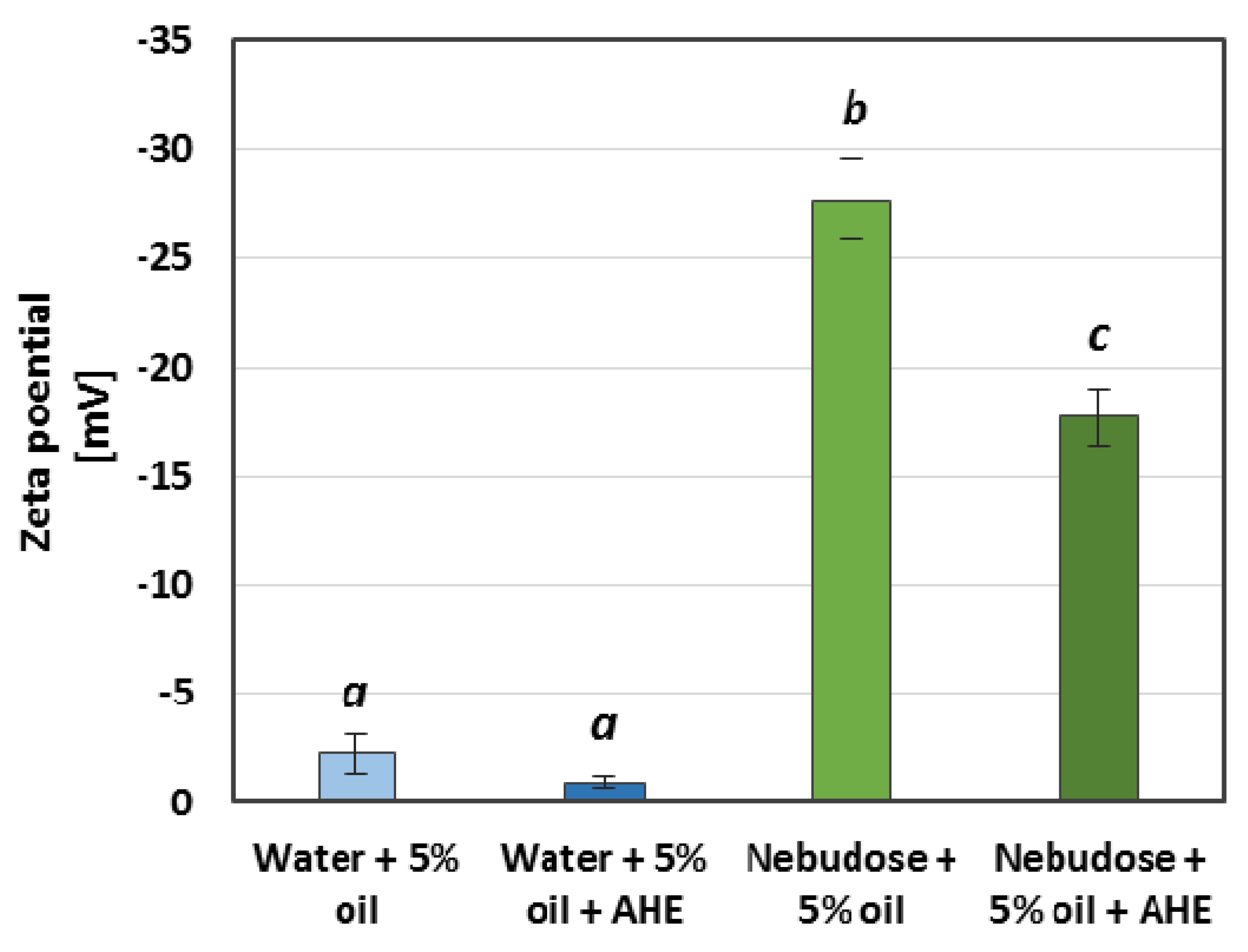
3.3.4. DLS and Zeta Potential
3.3.5. Viscosity
3.3.6. FTIR
3.3.7. Microscopic Investigations
3.3.8. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ravi, T.P.U.; Padma, T. Nanoemulsions for Drug Delivery through Different Routes. Res. Biotechnol. 2011, 2, 1–13. [Google Scholar]
- Lovelyn, C.; Attama, A.A. Current State of Nanoemulsions in Drug Delivery. J. Biomater. Nanobiotechnol. 2011, 2, 626–639. [Google Scholar] [CrossRef]
- Gupta, A.; Eral, H.B.; Hatton, T.A.; Doyle, P.S. Nanoemulsions: Formation, properties and applications. Soft Matter 2016, 12, 2826–2841. [Google Scholar] [CrossRef] [PubMed]
- Tamilvanan, S.; Benita, S. The potential of lipid emulsion for ocular delivery of lipophilic drugs. Eur. J. Pharm. Biopharm. 2004, 58, 357–368. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Appukuttan Aachary, A.; Thiyam-Holländer, U. Hemp seed oil: Minor components and oil quality. Lipid Technol. 2015, 27, 231–233. [Google Scholar] [CrossRef]
- Fathordoobady, F.; Singh, A.; Kitts, D.D.; Pratap Singh, A. Hemp (Cannabis Sativa L.) Extract: Anti-Microbial Properties, Methods of Extraction, and Potential Oral Delivery. Food Rev. Int. 2019, 35, 664–684. [Google Scholar] [CrossRef]
- Pratap Singh, A.; Fathordoobady, F.; Guo, Y.; Singh, A.; Kitts, D.D. Antioxidants help favorably regulate the kinetics of lipid peroxidation, polyunsaturated fatty acids degradation and acidic cannabinoids decarboxylation in hempseed oil. Sci. Rep. 2020, 10, 10567. [Google Scholar] [CrossRef]
- Mikulcová, V.; Kašpárková, V.; Humpolíček, P.; Buňková, L. Formulation, characterization and properties of hemp seed oil and its emulsions. Molecules 2017, 22, 700. [Google Scholar] [CrossRef]
- Yin, J.; Xiang, C.; Wang, P.; Yin, Y.; Hou, Y. Biocompatible nanoemulsions based on hemp oil and less surfactants for oral delivery of baicalein with enhanced bioavailability. Int. J. Nanomed. 2017, 12, 2923–2931. [Google Scholar] [CrossRef]
- Nejadmansouri, M.; Hosseini, S.M.H.; Niakosari, M.; Yousefi, G.H.; Golmakani, M.T. Physicochemical properties and oxidative stability of fish oil nanoemulsions as affected by hydrophilic lipophilic balance, surfactant to oil ratio and storage temperature. Colloids Surfaces A Physicochem. Eng. Asp. 2016, 506, 821–832. [Google Scholar] [CrossRef]
- Bai, L.; Huan, S.; Gu, J.; McClements, D.J. Fabrication of oil-in-water nanoemulsions by dual-channel microfluidization using natural emulsifiers: Saponins, phospholipids, proteins, and polysaccharides. Food Hydrocoll. 2016, 61, 703–711. [Google Scholar] [CrossRef]
- Güçlü-Üstündağ, Ö.; Mazza, G. Saponins: Properties, Applications and Processing. Crit. Rev. Food Sci. Nutr. 2007, 47, 231–258. [Google Scholar] [CrossRef] [PubMed]
- Hajimohammadi, R.; Hosseini, M.; Amani, H.; Najafpour, G.D. Production of Saponin Biosurfactant from Glycyrrhiza glabra as an Agent for Upgrading Heavy Crude Oil. J. Surfactants Deterg. 2016, 19, 1251–1261. [Google Scholar] [CrossRef]
- Tsibranska, S.; Tcholakova, S.; Golemanov, K.; Denkov, N.; Pelan, E.; Stoyanov, S.D. Role of interfacial elasticity for the rheological properties of saponin-stabilized emulsions. J. Colloid Interface Sci. 2020, 564, 264–275. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Sun, Q.; McClements, D.J. Enhancing the formation and stability of emulsions using mixed natural emulsifiers: Hydrolyzed rice glutelin and quillaja saponin. Food Hydrocoll. 2019, 89, 396–405. [Google Scholar] [CrossRef]
- Wan, Z.; Sun, Y.; Ma, L.; Guo, J.; Wang, J.; Yin, S.; Yang, X. Thermoresponsive structured emulsions based on the fibrillar self-assembly of natural saponin glycyrrhizic acid. Food Funct. 2017, 8, 75–85. [Google Scholar] [CrossRef]
- Chung, C.; Sher, A.; Rousset, P.; Decker, E.A.; McClements, D.J. Formulation of food emulsions using natural emulsifiers: Utilization of quillaja saponin and soy lecithin to fabricate liquid coffee whiteners. J. Food Eng. 2017, 209, 1–11. [Google Scholar] [CrossRef]
- Ralla, T.; Salminen, H.; Tuosto, J.; Weiss, J. Formation and stability of emulsions stabilised by Yucca saponin extract. Int. J. Food Sci. Technol. 2018, 53, 1381–1388. [Google Scholar] [CrossRef]
- Góral, I.; Wojciechowski, K. Surface activity and foaming properties of saponin-rich plants extracts. Adv. Colloid Interface Sci. 2020, 279, 102145. [Google Scholar] [CrossRef]
- Rojewska, M.; Smułek, W.; Prochaska, K.; Kaczorek, E. Combined Effect of Nitrofurantoin and Plant Surfactant on Bacteria Phospholipid Membrane. Molecules 2020, 25, 2527. [Google Scholar] [CrossRef]
- Jarzębski, M.; Smułek, W.; Siejak, P.; Kobus-Cisowska, J.; Pieczyrak, D.; Baranowska, H.M.; Jakubowicz, J.; Sopata, M.; Białopiotrowicz, T.; Kaczorek, E. Aesculus hippocastanum L. extract as a potential emulsion stabilizer. Food Hydrocoll. 2019, 97, 105237. [Google Scholar] [CrossRef]
- Wilkinson, J.A.; Brown, A.M.G. Horse Chestnut—Aesculus Hippocastanum: Potential Applications in Cosmetic Skin-care Products. Int. J. Cosmet. Sci. 1999, 21, 437–447. [Google Scholar] [CrossRef] [PubMed]
- Shinoda, R.; Uchimura, T. Evaluating the Creaming of an Emulsion via Mass Spectrometry and UV-Vis Spectrophotometry. ACS Omega 2018, 3, 13752–13756. [Google Scholar] [CrossRef] [PubMed]
- Jarzębski, M.; Siejak, P.; Smułek, W.; Fathordoobady, F.; Guo, Y.; Pawlicz, J.; Trzeciak, T.; Kowalczewski, P.Ł.; Kitts, D.D.; Singh, A.; et al. Plant Extracts Containing Saponins Affects the Stability and Biological Activity of Hempseed Oil Emulsion System. Molecules 2020, 25, 2696. [Google Scholar] [CrossRef] [PubMed]
- Maciej, J.; Przemysław, S.; Arkadiusz, S.; Maksymilian, S.; Kamil, R.; Łukasz, M.; Krzysztof, P.; Farahnaz, F.; Yigong, G. Nanoparticles Size Determination by Dynamic Light Scattering in Real (Non-standard) Conditions Regulators—Design, Tests and Applications. In Practical Aspects of Chemical Engineering; Ochowiak, M., Woziwodzki, S., Mitkowski, P.T., Doligalski, M., Eds.; Springer Nature Switzerland AG: Cham, Switzerland, 2020. [Google Scholar]
- Jarzębski, M.; Smułek, W.; Baranowska, H.M.; Masewicz, Ł.; Kobus-Cisowska, J.; Ligaj, M.; Kaczorek, E. Characterization of St. John’s wort (Hypericum perforatum L.) and the impact of filtration process on bioactive extracts incorporated into carbohydrate-based hydrogels. Food Hydrocoll. 2020, 104, 105748. [Google Scholar] [CrossRef]
- Jarzębski, M.; Bellich, B.; Białopiotrowicz, T.; Śliwa, T.; Kościński, J.; Cesàro, A. Particle tracking analysis in food and hydrocolloids investigations. Food Hydrocoll. 2016. [Google Scholar] [CrossRef]
- de Faria, J.T.; de Oliveira, E.B.; Minim, V.P.R.; Minim, L.A. Emulsifying properties of β-lactoglobulin and Quillaja bark saponin mixtures: Effects of number of homogenization passes, pH, and NaCl concentration. Int. J. Food Prop. 2017, 20, 1643–1654. [Google Scholar] [CrossRef]
- Prichapan, N.; McClements, D.J.; Klinkesorn, U. Encapsulation of Iron within W1/O/W2 Emulsions Formulated Using a Natural Hydrophilic Surfactant (Saponin): Impact of Surfactant Level and Oil Phase Crystallization. Food Biophys. 2020, 15, 346–354. [Google Scholar] [CrossRef]
- Zhu, L.; Xu, Q.; Liu, X.; Xu, Y.; Yang, L.; Wang, S.; Li, J.; He, Y.; Liu, H. Soy glycinin-soyasaponin mixtures at oil–water interface: Interfacial behavior and O/W emulsion stability. Food Chem. 2020, 327, 127062. [Google Scholar] [CrossRef]
- Rha, C. Rheology of fluid foods. Food Technol. 1978, 32, 77–81. [Google Scholar]
- Rave, M.C.; Echeverri, J.D.; Salamanca, C.H. Improvement of the physical stability of oil-in-water nanoemulsions elaborated with Sacha inchi oil employing ultra-high-pressure homogenization. J. Food Eng. 2020, 273, 109801. [Google Scholar] [CrossRef]
- Zheng, H. Introduction: Measuring Rheological Properties of Foods. In Rheology of Semisolid Foods; Joyner, H., Ed.; Springer Nature Switzerland AG: Cham, Switzerland, 2019. [Google Scholar]
- Zhu, Y.; Gao, H.; Liu, W.; Zou, L.; McClements, D.J. A review of the rheological properties of dilute and concentrated food emulsions. J. Texture Stud. 2019, 12444. [Google Scholar] [CrossRef] [PubMed]
- Domian, E.; Szczepaniak, M. Rheological behavior of concentrated emulsions containing carotenoids with different polarity. J. Food Eng. 2020, 274, 109827. [Google Scholar] [CrossRef]
- Olsen, E.F.; Rukke, E.-O.; Egelandsdal, B.; Isaksson, T. Determination of Omega-6 and Omega-3 Fatty Acids in Pork Adipose Tissue with Nondestructive Raman and Fourier Transform Infrared Spectroscopy. Appl. Spectrosc. 2008, 62, 968–974. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, M.; Khanmohammadi Khorrami, M.; Vatani, A.; Ghasemzadeh, H.; Vatanparast, H.; Bahramian, A.; Fallah, A. Rapid determination and classification of crude oils by ATR-FTIR spectroscopy and chemometric methods. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2020, 232, 118157. [Google Scholar] [CrossRef] [PubMed]
- Owczarek, A.; Kłys, A.; Olszewska, M.A. A validated 1H qNMR method for direct and simultaneous quantification of esculin, fraxin and (–)-epicatechin in Hippocastani cortex. Talanta 2019, 192, 263–269. [Google Scholar] [CrossRef]
- Geisler, R.; Dargel, C.; Hellweg, T. The Biosurfactant β-Aescin: A Review on the Physico-Chemical Properties and Its Interaction with Lipid Model Membranes and Langmuir Monolayers. Molecules 2019, 25, 117. [Google Scholar] [CrossRef]
- Camacho-Chab, J.C.; Guézennec, J.; Chan-Bacab, M.J.; Ríos-Leal, E.; Sinquin, C.; Muñiz-Salazar, R.; De la Rosa-García, S.D.C.; Reyes-Estebanez, M.; Ortega-Morales, B.O. Emulsifying activity and stability of a non-toxic bioemulsifier synthesized by Microbacterium sp. MC3B-10. Int. J. Mol. Sci. 2013, 14, 18959–18972. [Google Scholar] [CrossRef]

| Sample No. | Hemp Seed Oil Content (%) | Liquid Phase | AHE Concentration(g/L) |
|---|---|---|---|
| 1 | 1 | water | 0 |
| 2 | 0.5 | ||
| 3 | 1.0 | ||
| 4 | 2.0 | ||
| 5 | 2 | water | 0 |
| 6 | 0.5 | ||
| 7 | 1.0 | ||
| 8 | 2.0 | ||
| 9 | 5 | water | 0 |
| 10 | 0.5 | ||
| 11 | 1.0 | ||
| 12 | 2.0 | ||
| 13 | 1 | Nebudose | 0 |
| 14 | 0.5 | ||
| 15 | 1.0 | ||
| 16 | 2.0 | ||
| 17 | 2 | Nebudose | 0 |
| 18 | 0.5 | ||
| 19 | 1.0 | ||
| 20 | 2.0 | ||
| 21 | 5 | Nebudose | 0 |
| 22 | 0.5 | ||
| 23 | 1.0 | ||
| 24 | 2.0 |
| Liquid Phase | HSO Content (%) | AHE Concentration(g/L) | Emulsification Index [%] | |
|---|---|---|---|---|
| 24 h | 7 days | |||
| water | 1 | 0 | 99 ± 1 (a) | 98 ± 1 (a) |
| 0.5 | 99 ± 1 (a) | 96 ± 2 (ab) | ||
| 1.0 | 99 ± 1 (a) | 96 ± 3 (ab) | ||
| 2.0 | 99 ± 1 (a) | 95 ± 3 (ab) | ||
| 2 | 0 | 98 ± 2 (a) | 98 ± 2 (ab) | |
| 0.5 | 96 ± 2 (ab) | 93 ± 2 (b) | ||
| 1.0 | 98 ± 2 (ab) | 95 ± 2 (b) | ||
| 2.0 | 96 ± 3 (ab) | 96 ± 3 (ab) | ||
| 5 | 0 | 98 ± 2 (ab) | 98 ± 2 (ab) | |
| 0.5 | 98 ± 1 (ab) | 95 ± 2 (b) | ||
| 1.0 | 96 ± 2 (ab) | 93 ± 3 (b) | ||
| 2.0 | 94 ± 4 (ab) | 94 ± 4 (b) | ||
| Nebudose | 1 | 0 | 1 ± 1 (c) | 0 ± 0 (e) |
| 0.5 | 2 ± 1 (c) | 0 ± 0 (e) | ||
| 1.0 | 3 ± 2 (cd) | 1 ± 1 (cd) | ||
| 2.0 | 6 ± 2 (d) | 5 ± 1 (d) | ||
| 2 | 0 | 1 ± 1 (c) | 0 ± 0 (e) | |
| 0.5 | 2 ± 1 (c) | 0 ± 0 (e) | ||
| 1.0 | 3 ± 1 (cd) | 1 ± 1 (c) | ||
| 2.0 | 6 ± 2 (d) | 5 ± 1 (d) | ||
| 5 | 0 | 1 ± 1 (c) | 0 ± 0 (e) | |
| 0.5 | 3 ± 2 (cd) | 0 ± 0 (e) | ||
| 1.0 | 5 ± 2 (d) | 2 ± 1 (cd) | ||
| 2.0 | 8 ± 3 (d) | 6 ± 1 (d) | ||
| Hemp Seed Oil Content (%) | Liquid Phase | AHE Conc. (g/L) | d-ave (nm) | PDI | 1st Peak Max (nm) | 2nd Peak Max (nm) |
|---|---|---|---|---|---|---|
| 1 | water | 0 | 296 ± 3 (a) | 0.398 ± 0.035 (a) | 459 ± 47 (a) | 2935 ± 2574 (a) |
| 2.0 | 275 ± 1 (b) | 0.283 ± 0.010 (b) | 452 ± 37 (a) | 81 ± 47 (b) | ||
| 2 | 0 | 357 ± 8 (c) | 0.434 ± 0.010 (c) | 661 ± 62 (a) | 32 ± 28 (bc) | |
| 2.0 | 298 ± 4 (a) | 0.306 ± 0.045 (d) | 445 ± 33 (a) | 35 ± 20 (bc) | ||
| 5 | 0 | 393 ± 8 (d) | 0.516 ± 0.008 (e) | 750 ± 89 (a) | 3427 ± 2968 (a) | |
| 2.0 | 306 ± 5 (e) | 0.468 ± 0.033 (c) | 570 ± 86 (a) | 2783 ± 1703 (a) | ||
| 1 | Nebudose | 0 | 1527 ± 409 (f) | 0.812 ± 0.174 (f) | 1087 ± 222 (a) | 98 ± 42 (b) |
| 2.0 | 2533 ± 923 (g) | 1.000 (g) | 148 ± 19 (a) | 10 ± 16 (c) | ||
| 2 | 0 | 2099 ± 483 (g) | 0.601 ± 0.192 (e) | 843 ± 219 (a) | 47 ± 27 (bc) | |
| 2.0 | 1512 ± 239 (f) | 1.000 (g) | 124 ± 21 (a) | --- | ||
| 5 | 0 | 4301 ± 314 (h) | 0.442 ± 0.009 (c) | 2758 ± 1418 (a) | --- | |
| 2.0 | 283 ± 10 (a) | 0.606 ± 0.112 (e) | 313 ± 104 (a) | 3569 ± 2976 (a) |
| HSO Content (%) | Liquid Phase | AHE Concentration (g/L) | 24 h | 7 days | ||
|---|---|---|---|---|---|---|
| K (Pa·s) | n | K (Pa·s) | n | |||
| 1 | water | 0 | 0.0053 ± 0.0004 (a) | 0.79 ± 0.05 (a) | 0.0037 ± 0.0004 (c) | 0.70 ± 0.05 (a) |
| 2.0 | 0.0079 ± 0.0006 (b) | 0.76 ± 0.05 (ab) | 0.0070 ± 0.0003 (ab) | 0.75 ± 0.05 (a) | ||
| 2 | 0 | 0.0059 ± 0.0004 (a) | 0.78 ± 0.05 (a) | 0.0045 ± 0.0003 (d) | 0.71 ± 0.05 (ab) | |
| 2.0 | 0.0084 ± 0.0007 (b) | 0.78 ± 0.05 (a) | 0.0079 ± 0.0004 (b) | 0.74 ± 0.05 (a) | ||
| 5 | 0 | 0.0064 ± 0.0006 (ae) | 0.68 ± 0.04 (b) | 0.0056 ± 0.0006 (a) | 0.62 ± 0.04 (b) | |
| 2.0 | 0.0092 ± 0.0008 (b) | 0.79 ± 0.05 (a) | 0.0087 ± 0.0005 (b) | 0.78 ± 0.05 (a) | ||
| 1 | Nebudose | 0 | 0.0054 ± 0.0005 (a) | 0.77 ± 0.03 (a) | 0.0044 ± 0.0005 (cd) | 0.72 ± 0.05 (ab) |
| 2.0 | --- | --- | --- | --- | ||
| 2 | 0 | 0.0061 ± 0.0005 (a) | 0.83 ± 0.06 (a) | 0.0057 ± 0.0002 (a) | 0.81 ± 0.06 (a) | |
| 2.0 | --- | --- | --- | --- | ||
| 5 | 0 | 0.0070 ± 0.0006 (ab) | 0.85 ± 0.07 (a) | 0.0068 ± 0.0003 (e) | 0.80 ± 0.06 (a) | |
| 2.0 | 0.0093 ± 0.0008 (b) | 0.80 ±0.06 (a) | 0.0089± 0.0005 (b) | 0.80 ± 0.05 (a) | ||
| HSO Content (%) | Liquid Phase | AHE Concentration (g/L) | 24 h | 7 days | ||||
|---|---|---|---|---|---|---|---|---|
| τo, (Pa) | K (Pa·s) | n | τo, (Pa) | K, (Pa·s) | n | |||
| 1 | water | 0 | 0.018 ± 0.004 (ag) | 0.0025 ± 0.0001 (a) | 0.89 ± 0.09 (a) | 0.011 ± 0.006 (g) | 0.0016 ± 0.0001 (f) | 0.73 ± 0.05 (b) |
| 2.0 | 0.050 ± 0.001 (b) | 0.0038 ± 0.0001 (b) | 0.94 ± 0.05 (a) | 0.032 ± 0.005 (a) | 0.032 ± 0.005 (b) | 0.79 ± 0.03 (b) | ||
| 2 | 0 | 0.042 ± 0.004 (c) | 0.0027 ± 0.0002 (a) | 0.89 ± 0.16 (a) | 0.021 ± 0.006 (ag) | 0.0018 ± 0.0001 (g) | 0.89 ± 0.05 (a) | |
| 2.0 | 0.066 ±0.002 (d) | 0.0041 ± 0.0002 (b) | 0.94 ± 0.06 (a) | 0.049 ± 0.006 (c) | 0.0033 ± 0.0003 (b) | 0.75 ± 0.18 (b) | ||
| 5 | 0 | 0.050 ±0.001 (b) | 0.0029 ± 0.0003 (a) | 0.89 ± 0.21 (a) | 0.031 ± 0.001 (a) | 0.0023 ± 0.0001 (h) | 0.79 ± 0.07 (ab) | |
| 2.0 | 0.070 ± 0.001 (e) | 0.0046 ± 0.0002 (c) | 0.96 ± 0.09 (a) | 0.052 ± 0.003 (b) | 0.0040 ±0.0008 (bc) | 0.68 ± 0.08 (bc) | ||
| 1 | Nebudose | 0 | 0.021 ± 0.005 (ag) | 0.0030 ± 0.0001 (a) | 0.94 ± 0.02 (a) | 0.010 ± 0.008 (g) | 0.0020 ± 0.0001 (g) | 0.59 ± 0.05 (c) |
| 2.0 | --- | --- | --- | --- | --- | --- | ||
| 2 | 0 | 0.027 ± 0.005 (a) | 0.0032 ± 0.0001 (a) | 0.94 ± 0.05 (a) | 0.024 ± 0.008 (ag) | 0.0022 ± 0.0001 (g) | 0.67 ± 0.05 (bc) | |
| 2.0 | --- | --- | --- | --- | --- | --- | ||
| 5 | 0 | 0.046 ± 0.009 (b) | 0.0035 ± 0.0002 (d) | 0.94 ± 0.09 (a) | 0.049 ± 0.003 (b) | 0.0025 ± 0.0002 (g) | 0.69 ± 0.16 (bc) | |
| 2.0 | 0.076 ± 0.003 (f) | 0.0050 ± 0.0002 (e) | 0.97 ± 0.07 (a) | 0.053 ± 0.003 (b) | 0.0042 ± 0.0005 (b) | 0.69 ± 0.08 (bc) | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jarzębski, M.; Smułek, W.; Siejak, P.; Rezler, R.; Pawlicz, J.; Trzeciak, T.; Jarzębska, M.; Majchrzak, O.; Kaczorek, E.; Kazemian, P.; et al. Aesculus hippocastanum L. as a Stabilizer in Hemp Seed Oil Nanoemulsions for Potential Biomedical and Food Applications. Int. J. Mol. Sci. 2021, 22, 887. https://doi.org/10.3390/ijms22020887
Jarzębski M, Smułek W, Siejak P, Rezler R, Pawlicz J, Trzeciak T, Jarzębska M, Majchrzak O, Kaczorek E, Kazemian P, et al. Aesculus hippocastanum L. as a Stabilizer in Hemp Seed Oil Nanoemulsions for Potential Biomedical and Food Applications. International Journal of Molecular Sciences. 2021; 22(2):887. https://doi.org/10.3390/ijms22020887
Chicago/Turabian StyleJarzębski, Maciej, Wojciech Smułek, Przemysław Siejak, Ryszard Rezler, Jarosław Pawlicz, Tomasz Trzeciak, Małgorzata Jarzębska, Oliwia Majchrzak, Ewa Kaczorek, Pardis Kazemian, and et al. 2021. "Aesculus hippocastanum L. as a Stabilizer in Hemp Seed Oil Nanoemulsions for Potential Biomedical and Food Applications" International Journal of Molecular Sciences 22, no. 2: 887. https://doi.org/10.3390/ijms22020887
APA StyleJarzębski, M., Smułek, W., Siejak, P., Rezler, R., Pawlicz, J., Trzeciak, T., Jarzębska, M., Majchrzak, O., Kaczorek, E., Kazemian, P., Ponieważ-Pawlicz, M., & Fathordoobady, F. (2021). Aesculus hippocastanum L. as a Stabilizer in Hemp Seed Oil Nanoemulsions for Potential Biomedical and Food Applications. International Journal of Molecular Sciences, 22(2), 887. https://doi.org/10.3390/ijms22020887










