Pathogenesis and Molecular Mechanisms of Anderson–Fabry Disease and Possible New Molecular Addressed Therapeutic Strategies
Abstract
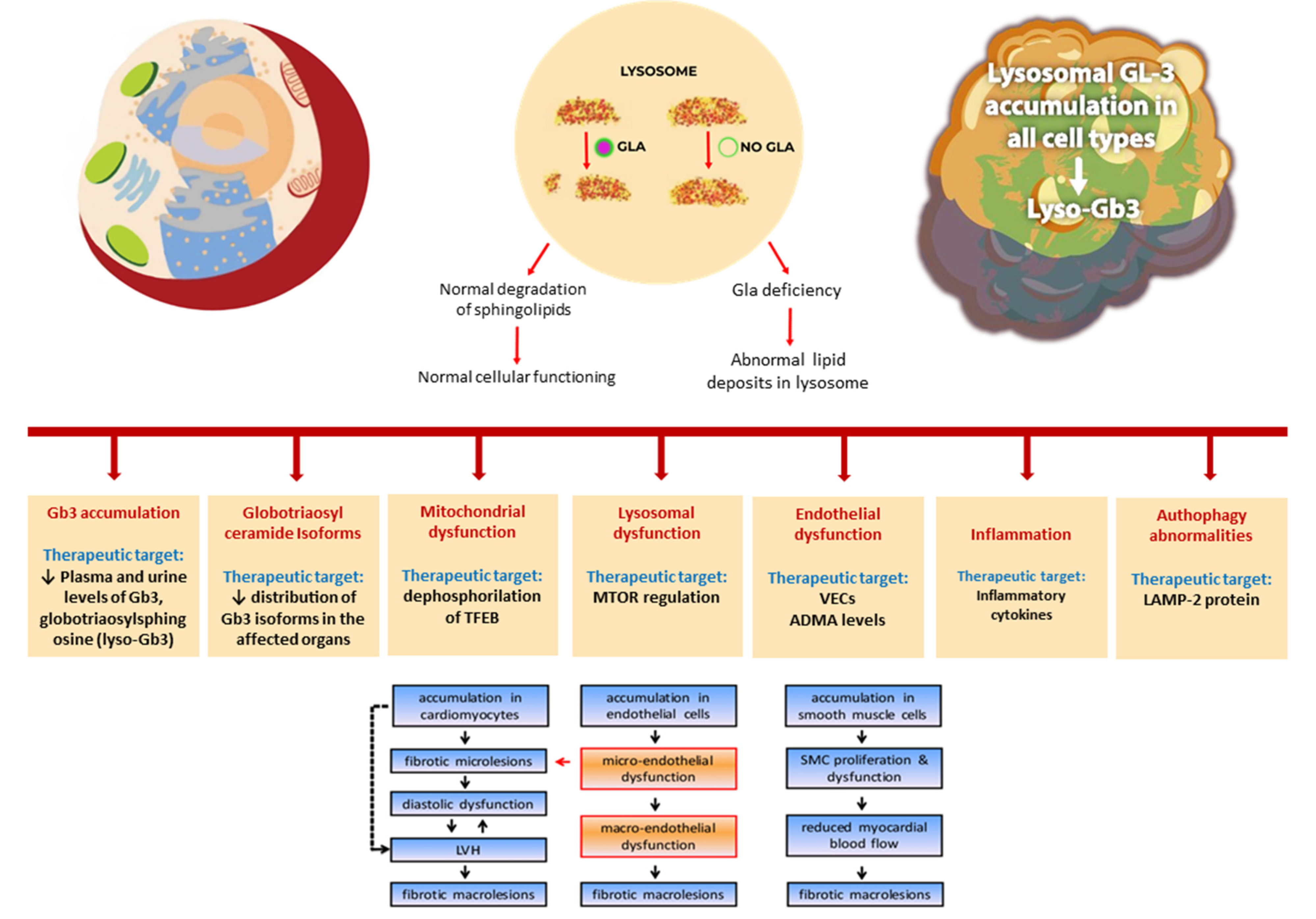
1. Background
2. Mitochondrial Dysfunction in Anderson–Fabry Disease
3. Neuropathological Aspects of Gb3 Accumulation in AA
4. Globotriaosylceramide Isoforms and Globotriaosylsphingosine in Organ Damage Related to Anderson–Fabry Disease
5. Pathogenesis of Endothelial Dysfunction Linked to Gb3 Accumulation
6. Pathogenesis of Lysosomal Damage due to Globotriaosylceramide (Gb3)
7. Molecular Pathogenesis of Renal Involvement in Anderson–Fabry Disease
8. Molecular Pathogenesis of Cardiac Involvement in Anderson–Fabry Disease
9. Molecular Mechanisms and Possible Therapeutic Targets
10. Autophagy Abnormalities in Molecular Pathogenesis of Anderson–Fabry Disease
11. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AFD | Anderson Fabry disease |
| Gb3 | Globotriaosylceramide |
| GLA | Alpha-galactosidase A |
| ESRD | End-stage renal disease |
| ADP | Adenosine-di-phosphate |
| AMP | Adenosin mono-phosphate |
| ATP | adenosine-tri-phosphate |
| LC3-II | Microtubule-associated protein 1A/1B-light chain 3 |
| MTOR | Mechanistic target of rapamycin kinase- |
| AMPK | AMP-activated protein kinase |
| TFEB | Transcription factor EB |
| Tfam | Mitochondrial transcription factor 1 |
| CoxI | Cytochrome c oxidase subunit 1 |
| Cox IV | Cytochrome c oxidase subunit IV |
| ECs | Endothelial cells |
| KCa3.1 | Ca2+ activated K+ channel |
| MAEC | Mouse aortic endothelial cells |
| PI(3)P | Phosphatidylinositol 3-phosphates |
| ROS | Reactive oxygen species |
| ERK 1/2 | Extracellular signal-regulated kinases |
| VECs | Endothelial cells |
| EDR | Endothelium–dependent relaxation |
| CRISPR)-Cas9 | Clustered Regularly Interspaced Short Palindromic Repeats Cas 9 |
| TSP-1 | Thrombospondin-1 |
| SMAD2 | p-mothers against decapentaplegic homolog 2 |
| SDMA | Symmetric Dimethylarginine |
| ADMA | l-arginine, asymmetric |
| hArg | l-homoarginine |
| MMP-9 | Metalloproteinase 9 Pathogenesis of lysosomal damage due to globotriaosylceramide (Gb3) |
| EDHF | Endothelium-derived hyperpolarizing factor |
| KCa3.1 | Ca2+-activated K+ channel |
| HEK cells | Human embryonic kidney cells |
| ACR | Albumin/creatinine ratio |
| RIPK3 | Receptor Interacting Serine/Threonine Kinase 3 |
| TGF-β1 | Transforming growth factor, beta 1 |
| CD74 | cluster differentiation 74 |
| ITGAV | Integrin Subunit Alpha V |
| ITGB3 | transforming growth factor beta 3, |
| PLAUR (UPAR) | Plasminogen Activator, Urokinase Receptor (rokinase plasminogen activator surface receptor) |
| MCP1 | Monocyte chemoattractant protein-1 |
| RANTES | Regulated upon Activation, Normal T Cell Expressed and Presumably Secreted |
| REST | Repressor element-1 silencing transcription factor |
| GWAS | genome-wide association studies |
| ADH4 | Alcohol Dehydrogenase 4 |
| ADH5 | Alcohol Dehydrogenase 5 |
| LAMP-2 | Lysosome-associated membrane protein 2 |
| VSMC | Vascular smooth muscle cells |
| GSI IX | γ-secretase inhibitor |
| PBMCs | Peripheral blood mononuclear cell |
| TLR4 | Toll-like Receptor 4 |
| SRT | substrate reduction therapy |
References
- Germain, D.P. Fabry disease. Orphanet J. Rare Dis. 2010, 5, 30. [Google Scholar] [CrossRef]
- Tuttolomondo, A.; Pecoraro, R.; Simonetta, I.; Miceli, S.; Pinto, A.; Licata, G. Anderson–Fabry Disease: A Multiorgan Disease. Curr. Pharm. Des. 2013, 19, 5974–5996. [Google Scholar] [CrossRef] [PubMed]
- Fabry, J. Ein Beitrag zur Kenntniss der Purpura haemorrhagica nodularis (Purpura papulosa haemorrhagica Hebrae). Arch. Dermatol. Res. 1898, 43, 187–200. [Google Scholar] [CrossRef]
- Anderson, W. A Case of “Angeio-Keratoma”. Br. J. Dermatol. 1898, 10, 113–117. [Google Scholar] [CrossRef]
- Branton, M.H.; Schiffmann, R.; Sabnis, S.G.; Murray, G.J.; Quirk, J.M.; Altarescu, G.; Goldfarb, L.; Brady, R.O.; Balow, J.E.; Austin, H.A.; et al. Natural History of Fabry Renal Disease. Medicine 2002, 81, 122–138. [Google Scholar] [CrossRef] [PubMed]
- Albay, D.; Adler, S.G.; Philipose, J.; Calescibetta, C.C.; Romansky, S.G.; Cohen, A.H. Chloroquine-induced lipidosis mimicking Fabry disease. Mod. Pathol. 2004, 18, 733–738. [Google Scholar] [CrossRef] [PubMed]
- Weidemann, F.; Sanchez-Niño, M.D.; Politei, J.; Oliveira, J.-P.; Wanner, C.; Warnock, D.G.; Ortiz, A. Fibrosis: A key feature of Fabry disease with potential therapeutic implications. Orphanet J. Rare Dis. 2013, 8, 116. [Google Scholar] [CrossRef]
- Sanchez-Niño, M.D.; Carpio, D.; Sanz, A.B.; Ruiz-Ortega, M.; Mezzano, S.; Ortiz, A. Lyso-Gb3 activates Notch1 in human podocytes. Hum. Mol. Genet. 2015, 24, 5720–5732. [Google Scholar] [CrossRef] [PubMed]
- Waldek, S.; Feriozzi, S. Fabry nephropathy: A review—How can we optimize the management of Fabry nephropathy? BMC Nephrol. 2014, 15, 72. [Google Scholar] [CrossRef]
- Sessa, A.; Toson, A.; Nebuloni, M. Renal ultrastructural findings in Anderson–Fabry disease. J. Nephrol. 2002, 15, 109–112. [Google Scholar]
- Boutin, M.; Menkovic, I.; Martineau, T.; Vaillancourt-Lavigueur, V.; Toupin, A.; Auray-Blais, C. Separation and Analysis of Lactosylceramide, Galabiosylceramide, and Globotriaosylceramide by LC-MS/MS in Urine of Fabry Disease Patients. Anal. Chem. 2017, 89, 13382–13390. [Google Scholar] [CrossRef] [PubMed]
- Kolter, T.; Sandhoff, K. Sphingolipid metabolism diseases. Biochim. Biophys. Acta 2006, 1758, 2057–2079. [Google Scholar] [CrossRef] [PubMed]
- Aerts, J.M.; Groener, J.E.; Kuiper, S.; Donker-Koopman, W.E.; Strijland, A.; Ottenhoff, R.; van Roomen, C.; Mirzaian, M.; Wijburg, F.A.; Linthorst, G.E.; et al. Elevated globotriaosylsphingosine is a hallmark of Fabry disease. Proc. Natl. Acad. Sci. USA 2008, 105, 2812–2817. [Google Scholar] [CrossRef] [PubMed]
- Das, A.; Naim, H. Chapter 3 Biochemical Basis of Fabry Disease with Emphasis on Mitochondrial Function and Protein Trafficking. Adv. Clin. Chem. 2009, 49, 57–71. [Google Scholar] [CrossRef] [PubMed]
- McLauchlan, H.; Newell, J.; Morrice, N.; Osborne, A.; West, M.; Smythe, E. A novel role for Rab5–GDI in ligand sequestration into clathrin-coated pits. Curr. Biol. 1998, 8, 34–45. [Google Scholar] [CrossRef]
- Platt, F.M.; Boland, B.; Van Der Spoel, A.C. Lysosomal storage disorders: The cellular impact of lysosomal dysfunction. J. Cell Biol. 2012, 199, 723–734. [Google Scholar] [CrossRef]
- Kiselyov, K.; Muallem, S. Mitochondrial Ca2+ homeostasis in lysosomal storage diseases. Cell Calcium 2008, 44, 103–111. [Google Scholar] [CrossRef] [PubMed]
- MacDermot, K.D.; Holmes, A.; Miners, A.H. Anderson–Fabry disease: Clinical manifestations and impact of disease in a cohort of 60 obligate carrier females. J. Med. Genet. 2001, 38, 769–775. [Google Scholar] [CrossRef]
- Machann, W.; Breunig, F.; Weidemann, F.; Sandstede, J.; Hahn, D.; Köstler, H.; Neubauer, S.; Wanner, C.; Beer, M. Cardiac energy metabolism is disturbed in Fabry disease and improves with enzyme replacement therapy using recombinant human galactosidase A. Eur. J. Hear. Fail. 2011, 13, 278–283. [Google Scholar] [CrossRef]
- Birket, M.J.; Raibaud, S.; Lettieri, M.; Adamson, A.D.; Letang, V.; Cervello, P.; Redon, N.; Ret, G.; Viale, S.; Wang, B.; et al. A Human Stem Cell Model of Fabry Disease Implicates LIMP-2 Accumulation in Cardiomyocyte Pathology. Stem Cell Rep. 2019, 13, 380–393. [Google Scholar] [CrossRef]
- Liebau, M.C.; Braun, F.; Höpker, K.; Weitbrecht, C.; Bartels, V.; Müller, R.-U.; Brodesser, S.; Saleem, M.A.; Benzing, T.; Schermer, B.; et al. Dysregulated Autophagy Contributes to Podocyte Damage in Fabry’s Disease. PLoS ONE 2013, 8, e63506. [Google Scholar] [CrossRef] [PubMed]
- Urine-Derived Cells: A Promising Diagnostic Tool in Fabry Disease Patients. Available online: https://europepmc.org/article/pmc/pmc6056427 (accessed on 26 March 2021).
- Moes, A.D.; Van Der Lubbe, N.; Zietse, R.; Loffing, J.; Hoorn, E.J. The sodium chloride cotransporter SLC12A3: New roles in sodium, potassium, and blood pressure regulation. Pflüg. Arch. 2013, 466, 107–118. [Google Scholar] [CrossRef]
- Maruyama, H.; Taguchi, A.; Nishikawa, Y.; Guili, C.; Mikame, M.; Nameta, M.; Yamaguchi, Y.; Ueno, M.; Imai, N.; Ito, Y.; et al. Medullary thick ascending limb impairment in the Gla tm Tg(CAG-A4GALT) Fabry model mice. FASEB J. 2018, 32, 4544–4559. [Google Scholar] [CrossRef] [PubMed]
- Magalhaes, J.; Gegg, M.E.; Migdalska-Richards, A.; Doherty, M.K.; Whitfield, P.D.; Schapira, A.H. Autophagic lysosome reformation dysfunction in glucocerebrosidase deficient cells: Relevance to Parkinson disease. Hum. Mol. Genet. 2016, 25, 3432–3445. [Google Scholar] [CrossRef]
- Napolitano, G.; Ballabio, A. TFEB at a glance. J. Cell Sci. 2016, 129, 2475–2481. [Google Scholar] [CrossRef] [PubMed]
- Puertollano, R.; Ferguson, S.M.; Brugarolas, J.; Ballabio, A. The complex relationship between TFEB transcription factor phosphorylation and subcellular localization. EMBO J. 2018, 37, e98804. [Google Scholar] [CrossRef]
- Laplante, M.; Sabatini, D.M. mTOR signaling at a glance. J. Cell Sci. 2009, 122, 3589–3594. [Google Scholar] [CrossRef] [PubMed]
- Morita, M.; Gravel, S.-P.; Hulea, L.; Larsson, O.; Pollak, M.; St-Pierre, J.; Topisirovic, I. mTOR coordinates protein synthesis, mitochondrial activity and proliferation. Cell Cycle 2015, 14, 473–480. [Google Scholar] [CrossRef]
- Cabot, R.C.; Towne, V.W.; Castleman, B. Case 42502. N. Engl. J. Med. 1956, 255, 1153–1157. [Google Scholar] [CrossRef]
- Meschia, J.F.; Brott, T.G.; Brown, R.D. Genetics of Cerebrovascular Disorders. Mayo Clin. Proc. 2005, 80, 122–132. [Google Scholar] [CrossRef]
- Grewal, R.P. Stroke in Fabry’s disease. J. Neurol. 1993, 241, 153–156. [Google Scholar] [CrossRef]
- Kaye, E.M.; Kolodny, E.H.; Logigian, E.L.; Ullman, M.D. Nervous system involvement in Fabry’s disease: Clinicopathological and biochemical correlation. Ann. Neurol. 1988, 23, 505–509. [Google Scholar] [CrossRef]
- Menzies, D.G.; Campbell, I.W.; Kean, D.M. Magnetic resonance imaging in Fabry’s disease. J. Neurol. Neurosurg. Psychiatry 1988, 51, 1240–1241. [Google Scholar] [CrossRef][Green Version]
- Mendez, M.F.; Stanley, T.M.; Medel, N.M.; Li, Z.; Tedesco, D.T. The vascular dementia of Fabry’s disease. Dement. Geriatr. Cogn. Disord. 1997, 8, 252–257. [Google Scholar] [CrossRef]
- Mohanraj, R.; Leach, J.P.; Broome, J.C.; Smith, D.F. Neurological presentation of Fabry’s disease in a 52 year old man. J. Neurol. Neurosurg. Psychiatry 2002, 73, 340–342. [Google Scholar] [CrossRef] [PubMed]
- Moumdjian, R.; Tampieri, D.; Melanson, D.; Ethier, R. Anderson–Fabry Disease: A Case Report with MR, CT, and Cerebral Angiography. Am. J. Neuroradiol. 1989, 10, S69–S70. [Google Scholar]
- Morgan, S.H. The neurological complications of Anderson–Fabry disease (alpha-galactosidase A deficiency)--investigation of symptomatic and presymptomatic patients. Q. J. Med. 1990, 75, 491–507. [Google Scholar] [PubMed]
- Mitsias, P.; Levine, S.R. Cerebrovascular complications of Fabry’s disease. Ann. Neurol. 1996, 40, 8–17. [Google Scholar] [CrossRef]
- Crutchfield, K.E.; Patronas, N.J.; Dambrosia, J.M.; Frei, K.P.; Banerjee, T.K.; Barton, N.W.; Schiffmann, R. Quantitative analysis of cerebral vasculopathy in patients with Fabry disease. Neurology 1998, 50, 1746–1749. [Google Scholar] [CrossRef]
- Politei, J.M.; Capizzano, A.A. Magnetic Resonance Image Findings in 5 Young Patients with Fabry Disease. Neurologist 2006, 12, 103–105. [Google Scholar] [CrossRef] [PubMed]
- Rahman, A.N.; Lindenberg, R. The Neuropathology of Hereditary Dystopic Lipidosis. Arch. Neurol. 1963, 9, 373–385. [Google Scholar] [CrossRef]
- Schatzki, P.F.; Kipreos, B.; Payne, J. Fabry’s disease. Primary diagnosis by electron microscopy. Am. J. Surg. Pathol. 1979, 3, 211–219. [Google Scholar] [CrossRef] [PubMed]
- De Veber, G.A.; Schwarting, G.A.; Kolodny, E.H.; Kowall, N.W. Fabry disease: Immunocytochemical characterization of neuronal involvement. Ann. Neurol. 1992, 31, 409–415. [Google Scholar] [CrossRef] [PubMed]
- Del Pino, M.; Andrés, A.; Bernabéu, A.Á.; De Juan-Rivera, J.; Fernández, E.; Díaz, J.D.D.G.; Hernández, D.; Luño, J.; Fernández, I.M.; Paniagua, J.; et al. Fabry Nephropathy: An Evidence-Based Narrative Review. Kidney Blood Press. Res. 2018, 43, 406–421. [Google Scholar] [CrossRef]
- Park, S.; Kim, J.A.; Joo, K.Y.; Choi, S.; Choi, E.N.; Shin, J.A.; Han, K.H.; Jung, S.C.; Suh, S.H. Globotriaosylceramide leads to K(Ca)3.1 channel dysfunction: A new insight into endothelial dysfunction in Fabry disease. Cardiovasc. Res. 2011, 89, 290–299. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Balut, C.M.; Bailey, M.A.; Patino-Lopez, G.; Shaw, S.; Devor, D.C. Recycling of the Ca2+-activated K+ Channel, KCa2.3, Is Dependent upon RME-1, Rab35/EPI64C, and an N-terminal Domain. J. Biol. Chem. 2010, 285, 17938–17953. [Google Scholar] [CrossRef]
- Balut, C.M.; Gao, Y.; Murray, S.A.; Thibodeau, P.H.; Devor, D.C. ESCRT-dependent targeting of plasma membrane localized KCa3.1 to the lysosomes. Am. J. Physiol. Physiol. 2010, 299, C1015–C1027. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.; Kim, J.A.; Na, H.-Y.; Cho, S.-E.; Park, S.; Jung, S.-C.; Suh, S.H. Globotriaosylceramide Induces Lysosomal Degradation of Endothelial K Ca 3.1 in Fabry Disease. Arter. Thromb. Vasc. Biol. 2014, 34, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Si, H.; Heyken, W.-T.; Wölfle, S.E.; Tysiac, M.; Schubert, R.; Grgic, I.; Vilianovich, L.; Giebing, G.; Maier, T.; Gross, V.; et al. Impaired Endothelium-Derived Hyperpolarizing Factor-Mediated Dilations and Increased Blood Pressure in Mice Deficient of the Intermediate-Conductance Ca2+-Activated K+ Channel. Circ. Res. 2006, 99, 537–544. [Google Scholar] [CrossRef]
- Shu, L.; Shayman, J.A. Caveolin-associated Accumulation of Globotriaosylceramide in the Vascular Endothelium of α-Galactosidase A Null Mice. J. Biol. Chem. 2007, 282, 20960–20967. [Google Scholar] [CrossRef]
- Do, H.-S.; Park, S.-W.; Im, I.; Seo, D.; Yoo, H.-W.; Go, H.; Kim, Y.H.; Koh, G.Y.; Lee, B.-H.; Han, Y.-M. Enhanced thrombospondin-1 causes dysfunction of vascular endothelial cells derived from Fabry disease-induced pluripotent stem cells. EBioMedicine 2020, 52, 102633. [Google Scholar] [CrossRef] [PubMed]
- Loso, J.; Lund, N.; Avanesov, M.; Muschol, N.; Lezius, S.; Cordts, K.; Schwedhelm, E.; Patten, M. Serum Biomarkers of Endothelial Dysfunction in Fabry Associated Cardiomyopathy. Front. Cardiovasc. Med. 2018, 5, 108. [Google Scholar] [CrossRef]
- Brähler, S.; Kaistha, A.; Schmidt, V.J.; Wölfle, S.E.; Busch, C.; Kaistha, B.P.; Kacik, M.; Hasenau, A.-L.; Grgic, I.; Si, H.; et al. Genetic Deficit of SK3 and IK1 Channels Disrupts the Endothelium-Derived Hyperpolarizing Factor Vasodilator Pathway and Causes Hypertension. Circulation 2009, 119, 2323–2332. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-Y.; Park, S.; Lee, S.-W.; Lee, J.-H.; Lee, E.; Kim, M.; Kim, Y.; Kang, J.; Chung, C.; Moon, J.-S.; et al. RIPK3 Contributes to Lyso-Gb3-Induced Podocyte Death. Cells 2021, 10, 245. [Google Scholar] [CrossRef] [PubMed]
- Wanner, C.; Oliveira, J.P.; Ortiz, A.; Mauer, M.; Germain, D.; Linthorst, G.E.; Serra, A.L.; Maródi, L.; Mignani, R.; Cianciaruso, B.; et al. Prognostic Indicators of Renal Disease Progression in Adults with Fabry Disease: Natural History Data from the Fabry Registry. Clin. J. Am. Soc. Nephrol. 2010, 5, 2220–2228. [Google Scholar] [CrossRef] [PubMed]
- Taal, M.; Brenner, B. Predicting initiation and progression of chronic kidney disease: Developing renal risk scores. Kidney Int. 2006, 70, 1694–1705. [Google Scholar] [CrossRef]
- Verovnik, F.; Benko, D.; Vujkovac, B.; Linthorst, G.E. Remarkable variability in renal disease in a large Slovenian family with Fabry disease. Eur. J. Hum. Genet. 2004, 12, 678–681. [Google Scholar] [CrossRef] [PubMed]
- Alroy, J.; Sabnis, S.; Kopp, J. Renal Pathology in Fabry Disease. J. Am. Soc. Nephrol. 2002, 13, S134–S138. [Google Scholar] [CrossRef]
- Najafian, B.; Tøndel, C.; Svarstad, E.; Gubler, M.-C.; Oliveira, J.P.; Mauer, M. Accumulation of Globotriaosylceramide in Podocytes in Fabry Nephropathy Is Associated with Progressive Podocyte Loss. J. Am. Soc. Nephrol. 2020, 31, 865–875. [Google Scholar] [CrossRef]
- Fall, B.; Scott, C.R.; Mauer, M.; Shankland, S.; Pippin, J.; Jefferson, J.A.; Wallace, E.; Warnock, D.; Najafian, B. Urinary Podocyte Loss Is Increased in Patients with Fabry Disease and Correlates with Clinical Severity of Fabry Nephropathy. PLoS ONE 2016, 11, e0168346. [Google Scholar] [CrossRef]
- Trimarchi, H.; Canzonieri, R.; Costales-Collaguazo, C.; Politei, J.; Stern, A.; Paulero, M.; González-Hoyos, I.; Schiel, A.; Rengel, T.; Forrester, M.; et al. Early decrease in the podocalyxin to synaptopodin ratio in urinary Fabry podocytes. Clin. Kidney J. 2019, 12, 53–60. [Google Scholar] [CrossRef]
- Pereira, E.M.; Da Silva, A.S.; Labilloy, A.; Neto, J.T.D.M.; Monte, S.J.H.D. Podocyturia in Fabry disease. Braz. J. Nephrol. 2016, 38, 49–53. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Niño, M.D.; Sanz, A.B.; Carrasco, S.; Saleem, M.A.; Mathieson, P.W.; Valdivielso, J.M.; Ruiz-Ortega, M.; Egido, J.; Ortiz, A. Globotriaosylsphingosine actions on human glomerular podocytes: Implications for Fabry nephropathy. Nephrol. Dial. Transplant. 2011, 26, 1797–1802. [Google Scholar] [CrossRef] [PubMed]
- Trimarchi, H.; Ortiz, A.; Sánchez-Niño, M.D. Lyso-Gb3 Increases αvβ3 Integrin Gene Expression in Cultured Human Podocytes in Fabry Nephropathy. J. Clin. Med. 2020, 9, 3659. [Google Scholar] [CrossRef]
- Scionti, F.; Di Martino, M.T.; Sestito, S.; Nicoletti, A.; Falvo, F.; Roppa, K.; Arbitrio, M.; Guzzi, P.H.; Agapito, G.; Pisani, A.; et al. Genetic variants associated with Fabry disease progression despite enzyme replacement therapy. Oncotarget 2017, 8, 107558–107564. [Google Scholar] [CrossRef] [PubMed]
- Altarescu, G.; Moore, D.F.; Schiffmann, R. Effect of genetic modifiers on cerebral lesions in Fabry disease. Neurology 2005, 64, 2148–2150. [Google Scholar] [CrossRef] [PubMed]
- Altarescu, G.; Chicco, G.; Whybra, C.; Delgado-Sanchez, S.; Sharon, N.; Beck, M.; Elstein, D. Correlation between interleukin-6 promoter and C-reactive protein (CRP) polymorphisms and CRP levels with the Mainz Severity Score Index for Fabry disease. J. Inherit. Metab. Dis. 2008, 31, 117–123. [Google Scholar] [CrossRef]
- Jaurretche, S.; Perez, G.R.; Venera, G. High Lyso-Gb3 Plasma Levels Associated with Decreased miR-29 and miR-200 Urinary Excretion in Young Non-Albuminuric Male Patient with Classic Fabry Disease. Case Rep. Nephrol. 2019, 2019, 4980942. [Google Scholar] [CrossRef]
- Xiao, K.; Lu, D.; Hoepfner, J.; Santer, L.; Gupta, S.; Pfanne, A.; Thum, S.; Lenders, M.; Brand, E.; Nordbeck, P.; et al. Circulating microRNAs in Fabry Disease. Sci. Rep. 2019, 9, 15277. [Google Scholar] [CrossRef]
- Hübner, A.-C.; Metz, T.; Schanzer, A.; Greber-Platzer, S.; Item, C.B. Aberrant DNA methylation of calcitonin receptor in Fabry patients treated with enzyme replacement therapy. Mol. Genet. Metab. Rep. 2015, 5, 1–2. [Google Scholar] [CrossRef]
- Hossain, M.A.; Wu, C.; Yanagisawa, H.; Miyajima, T.; Akiyama, K.; Eto, Y. Future clinical and biochemical predictions of Fabry disease in females by methylation studies of the GLA gene. Mol. Genet. Metab. Rep. 2019, 20, 100497. [Google Scholar] [CrossRef]
- Boutin, M.; Auray-Blais, C. Multiplex Tandem Mass Spectrometry Analysis of Novel Plasma Lyso-Gb3-Related Analogues in Fabry Disease. Anal. Chem. 2014, 86, 3476–3483. [Google Scholar] [CrossRef]
- Abaoui, M.; Boutin, M.; Lavoie, P.; Auray-Blais, C. Tandem mass spectrometry multiplex analysis of methylated and non-methylated urinary Gb3 isoforms in Fabry disease patients. Clin. Chim. Acta 2016, 452, 191–198. [Google Scholar] [CrossRef]
- Elleder, M.; Bradová, V.; Šmíd, F.; Buděšínský, M.; Harzer, K.; Kustermann-Kuhn, B.; Ledvinova, J.; Kral, V.; Dorazilová, V. Belohlavek Cardiocyte storage and hypertrophy as a sole manifestation of Fabry’s disease. Virchows Arch. A Pathol. Anat. Histopathol. 1990, 417, 449–455. [Google Scholar] [CrossRef] [PubMed]
- Hypertrophic Cardiomyopathy: A Paradigm for Myocardial Energy Depletion. Available online: https://pubmed.ncbi.nlm.nih.gov/12711218/ (accessed on 31 March 2021).
- Lücke, T. Fabry disease: Reduced activities of respiratory chain enzymes with decreased levels of energy-rich phosphates in fibroblasts. Mol. Genet. Metab. 2004, 82, 93–97. [Google Scholar] [CrossRef]
- Niemann, M.; Breunig, F.; Beer, M.; Herrmann, S.; Strotmann, J.; Hu, K.; Emmert, A.; Voelker, W.; Ertl, G.; Wanner, C.; et al. The right ventricle in Fabry disease: Natural history and impact of enzyme replacement therapy. Heart 2010, 96, 1915–1919. [Google Scholar] [CrossRef] [PubMed]
- Chimenti, C.; Scopelliti, F.; Vulpis, E.; Tafani, M.; Villanova, L.; Verardo, R.; De Paulis, R.; Russo, M.A.; Frustaci, A. Increased oxidative stress contributes to cardiomyocyte dysfunction and death in patients with Fabry disease cardiomyopathy. Hum. Pathol. 2015, 46, 1760–1768. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.-H.; Chien, Y.; Wang, K.-L.; Leu, H.-B.; Hsiao, C.-Y.; Lai, Y.-H.; Wang, C.-Y.; Chang, Y.-L.; Lin, S.-J.; Niu, D.-M.; et al. Evaluation of Proinflammatory Prognostic Biomarkers for Fabry Cardiomyopathy With Enzyme Replacement Therapy. Can. J. Cardiol. 2016, 32, 1221–e1. [Google Scholar] [CrossRef]
- Sheppard, M.N.; Cane, P.; Florio, R.; Kavantzas, N.; Close, L.; Shah, J.; Lee, P.; Elliott, P. A detailed pathologic examination of heart tissue from three older patients with Anderson–Fabry disease on enzyme replacement therapy. Cardiovasc. Pathol. 2010, 19, 293–301. [Google Scholar] [CrossRef] [PubMed]
- Pastores, G.M.; Lien, Y.-H.H. Biochemical and Molecular Genetic Basis of Fabry Disease. J. Am. Soc. Nephrol. 2002, 13, S130–S133. [Google Scholar] [CrossRef]
- Schiffmann, R.; Kopp, J.B.; Iii, H.A.A.; Sabnis, S.; Moore, D.F.; Weibel, T.; Balow, J.E.; Brady, R.O. Enzyme Replacement Therapy in Fabry Disease. JAMA 2001, 285, 2743–2749. [Google Scholar] [CrossRef]
- Hughes, A.D.; Elliott, P.; Shah, J.; Zuckerman, J.; Coghlan, G.; Brookes, J.; Mehta, A.B. Effects of enzyme replacement therapy on the cardiomyopathy of Anderson Fabry disease: A randomised, double-blind, placebo-controlled clinical trial of agalsidase alfa. Heart 2008, 94, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Haneef, S.S.; Doss, C.G.P. Personalized Pharmacoperones for Lysosomal Storage Disorder. Adv. Protein Chem. Struct. Biol. 2016, 102, 225–265. [Google Scholar] [CrossRef] [PubMed]
- Parenti, G.; Andria, G.; Valenzano, K.J. Pharmacological Chaperone Therapy: Preclinical Development, Clinical Translation, and Prospects for the Treatment of Lysosomal Storage Disorders. Mol. Ther. 2015, 23, 1138–1148. [Google Scholar] [CrossRef]
- Asano, N.; Ishii, S.; Kizu, H.; Ikeda, K.; Yasuda, K.; Kato, A.; Martin, O.R.; Fan, J.-Q. In vitroinhibition and intracellular enhancement of lysosomal α-galactosidase A activity in Fabry lymphoblasts by 1-deoxygalactonojirimycin and its derivatives. JBIC J. Biol. Inorg. Chem. 2000, 267, 4179–4186. [Google Scholar] [CrossRef]
- Germain, D.; Hughes, D.; Nicholls, K.; Bichet, D.-G.; Giugliani, R.; Wilcox, W.R.; Feliciani, C.; Shankar, S.P.; Ezgu, F.; Amartino, H.; et al. Treatment of Fabry’s Disease with the Pharmacologic Chaperone Migalastat. N. Engl. J. Med. 2016, 375, 545–555. [Google Scholar] [CrossRef] [PubMed]
- Oral Pharmacological Chaperone Migalastat Compared with Enzyme Replacement Therapy in Fabry Disease: 18-Month Results from the Randomised Phase III ATTRACT Study. Available online: https://pubmed.ncbi.nlm.nih.gov/27834756/ (accessed on 31 March 2021).
- Müntze, J.; Gensler, D.; Maniuc, O.; Liu, D.; Cairns, T.; Oder, D.; Hu, K.; Lorenz, K.; Frantz, S.; Wanner, C.; et al. Oral Chaperone Therapy Migalastat for Treating Fabry Disease: Enzymatic Response and Serum Biomarker Changes After 1 Year. Clin. Pharmacol. Ther. 2019, 105, 1224–1233. [Google Scholar] [CrossRef]
- Lenders, M.; Stappers, F.; Brand, E. In Vitro and In Vivo Amenability to Migalastat in Fabry Disease. Mol. Ther.-Methods Clin. Dev. 2020, 19, 24–34. [Google Scholar] [CrossRef]
- Platt, F. Emptying the stores: Lysosomal diseases and therapeutic strategies. Nat. Rev. Drug Discov. 2018, 17, 133–150. [Google Scholar] [CrossRef]
- Guérard, N.; Oder, D.; Nordbeck, P.; Zwingelstein, C.; Morand, O.; Welford, R.W.; Dingemanse, J.; Wanner, C. Lucerastat, an Iminosugar for Substrate Reduction Therapy: Tolerability, Pharmacodynamics, and Pharmacokinetics in Patients With Fabry Disease on Enzyme Replacement. Clin. Pharmacol. Ther. 2018, 103, 703–711. [Google Scholar] [CrossRef]
- Guérard, N.; Morand, O.; Dingemanse, J. Lucerastat, an iminosugar with potential as substrate reduction therapy for glycolipid storage disorders: Safety, tolerability, and pharmacokinetics in healthy subjects. Orphanet J. Rare Dis. 2017, 12, 9. [Google Scholar] [CrossRef]
- Lavandero, S.; Chiong, M.; Rothermel, B.A.; Hill, J.A. Autophagy in cardiovascular biology. J. Clin. Investig. 2015, 125, 55–64. [Google Scholar] [CrossRef]
- Nguyen, H.T.; Noguchi, S.; Sugie, K.; Matsuo, Y.; Nguyen, C.; Koito, H.; Shiojima, I.; Nishino, I.; Tsukaguchi, H. Small-Vessel Vasculopathy Due to Aberrant Autophagy in LAMP-2 Deficiency. Sci. Rep. 2018, 8, 3326. [Google Scholar] [CrossRef]
- Autophagy-Lysosome Pathway Associated Neuropathology and axonal Degeneration in the Brains of Alpha-Galactosidase A-Deficient Mice. Available online: https://pubmed.ncbi.nlm.nih.gov/24529306/ (accessed on 28 March 2021).
- Ferraz, M.J.; Kallemeijn, W.W.; Mirzaian, M.; Moro, D.H.; Marques, A.; Wisse, P.; Boot, R.G.; Willems, L.I.; Overkleeft, H.; Aerts, J. Gaucher disease and Fabry disease: New markers and insights in pathophysiology for two distinct glycosphingolipidoses. Biochim. Biophys. Acta 2014, 1841, 811–825. [Google Scholar] [CrossRef] [PubMed]
- Barbey, F.; Brakch, N.; Linhart, A.; Rosenblatt, N.; Jeanrenaud, X.; Qanadli, S.; Steinmann, B.; Burnier, M.; Palecek, T.; Bultas, J.; et al. Cardiac and Vascular Hypertrophy in Fabry Disease. Arter. Thromb. Vasc. Biol. 2006, 26, 839–844. [Google Scholar] [CrossRef] [PubMed]
- Kaissarian, N.; Kang, J.; Shu, L.; Ferraz, M.J.; Aerts, J.M.; Shayman, J.A. Dissociation of globotriaosylceramide and impaired endothelial function in α-galactosidase-A deficient EA.hy926 cells. Mol. Genet. Metab. 2018, 125, 338–344. [Google Scholar] [CrossRef] [PubMed]
- Rombach, S.; Twickler, T.; Aerts, J.; Linthorst, G.; Wijburg, F.; Hollak, C. Vasculopathy in patients with Fabry disease: Current controversies and research directions. Mol. Genet. Metab. 2010, 99, 99–108. [Google Scholar] [CrossRef] [PubMed]
- De Francesco, P.N.; Mucci, J.M.; Ceci, R.; Fossati, C.A.; Rozenfeld, P.A. Fabry disease peripheral blood immune cells release inflammatory cytokines: Role of globotriaosylceramide. Mol. Genet. Metab. 2013, 109, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Rozenfeld, P.; Feriozzi, S. Contribution of inflammatory pathways to Fabry disease pathogenesis. Mol. Genet. Metab. 2017, 122, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Viana-Baptista, M. Stroke and Fabry disease. J. Neurol. 2011, 259, 1019–1028. [Google Scholar] [CrossRef] [PubMed]
- Platt, F.M.; Jeyakumar, M.; Andersson-Hall, U.; Priestman, D.A.; Dwek, R.A.; Butters, T.D.; Cox, T.M.; Lachmann, R.; Hollak, C.; Aerts, J.; et al. Inhibition of substrate synthesis as a strategy for glycolipid lysosomal storage disease therapy. J. Inherit. Metab. Dis. 2001, 24, 275–290. [Google Scholar] [CrossRef] [PubMed]
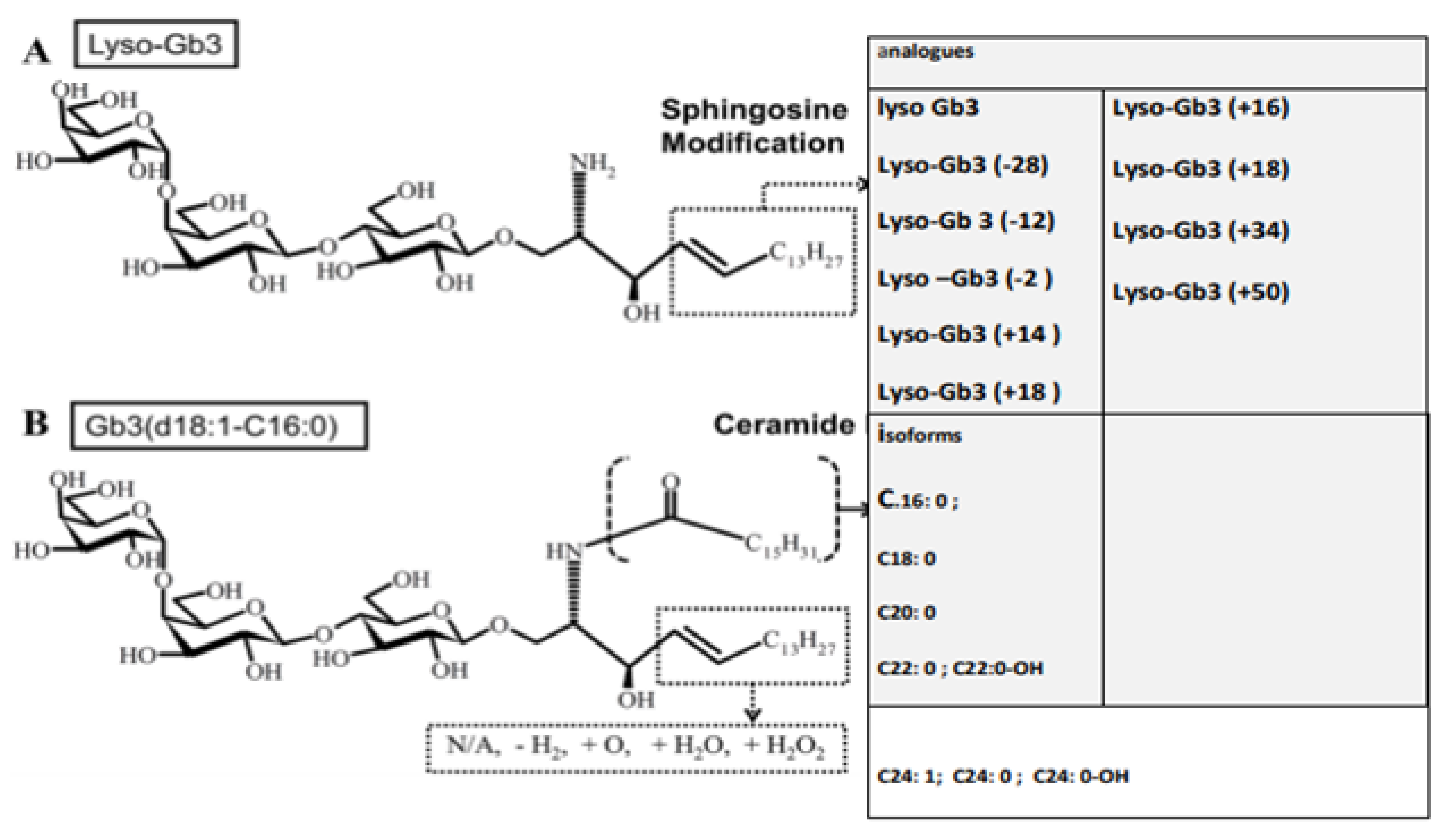

| Kidney-Specific Gb3 (Globotriaosylceramide) Isoforms | Cardiac Specifics Gb3 Isoforms | Lyso-Gb3 Analougues |
|---|---|---|
| Gb3 (d18: 1–C24: 0) | Gb3 (d18: 1–C16: 0) | Lyso-Gb3 (−2) |
| Gb3 (d18: 1–C16): 0) | Lyso-Gb3 (+16) | |
| Lyso-Gb3 (+18) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tuttolomondo, A.; Simonetta, I.; Riolo, R.; Todaro, F.; Di Chiara, T.; Miceli, S.; Pinto, A. Pathogenesis and Molecular Mechanisms of Anderson–Fabry Disease and Possible New Molecular Addressed Therapeutic Strategies. Int. J. Mol. Sci. 2021, 22, 10088. https://doi.org/10.3390/ijms221810088
Tuttolomondo A, Simonetta I, Riolo R, Todaro F, Di Chiara T, Miceli S, Pinto A. Pathogenesis and Molecular Mechanisms of Anderson–Fabry Disease and Possible New Molecular Addressed Therapeutic Strategies. International Journal of Molecular Sciences. 2021; 22(18):10088. https://doi.org/10.3390/ijms221810088
Chicago/Turabian StyleTuttolomondo, Antonino, Irene Simonetta, Renata Riolo, Federica Todaro, Tiziana Di Chiara, Salvatore Miceli, and Antonio Pinto. 2021. "Pathogenesis and Molecular Mechanisms of Anderson–Fabry Disease and Possible New Molecular Addressed Therapeutic Strategies" International Journal of Molecular Sciences 22, no. 18: 10088. https://doi.org/10.3390/ijms221810088
APA StyleTuttolomondo, A., Simonetta, I., Riolo, R., Todaro, F., Di Chiara, T., Miceli, S., & Pinto, A. (2021). Pathogenesis and Molecular Mechanisms of Anderson–Fabry Disease and Possible New Molecular Addressed Therapeutic Strategies. International Journal of Molecular Sciences, 22(18), 10088. https://doi.org/10.3390/ijms221810088







