Curcumin Derivatives as Potential Mosquito Larvicidal Agents against Two Mosquito Vectors, Culex pipiens and Aedes albopictus
Abstract
:1. Introduction
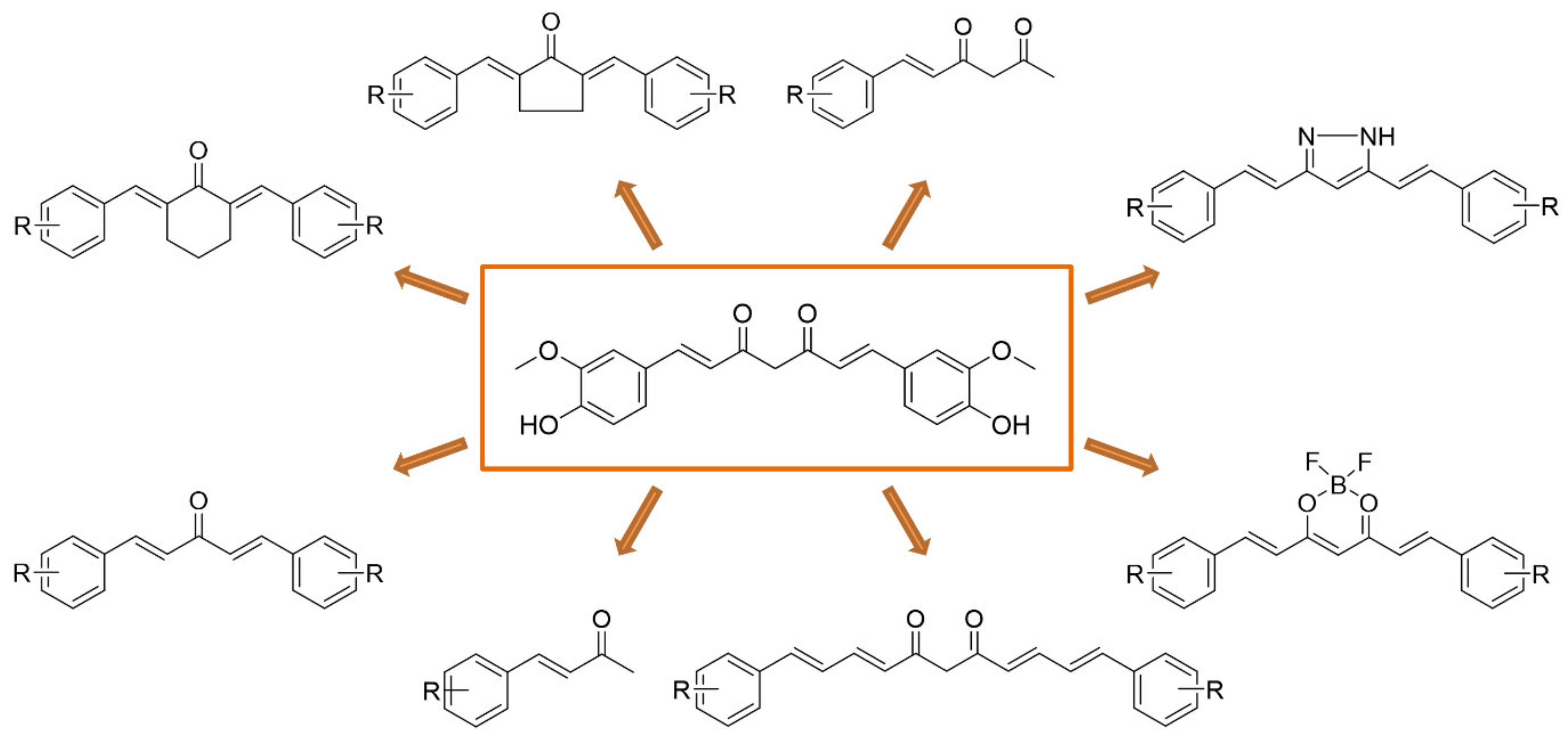
2. Results and Discussion
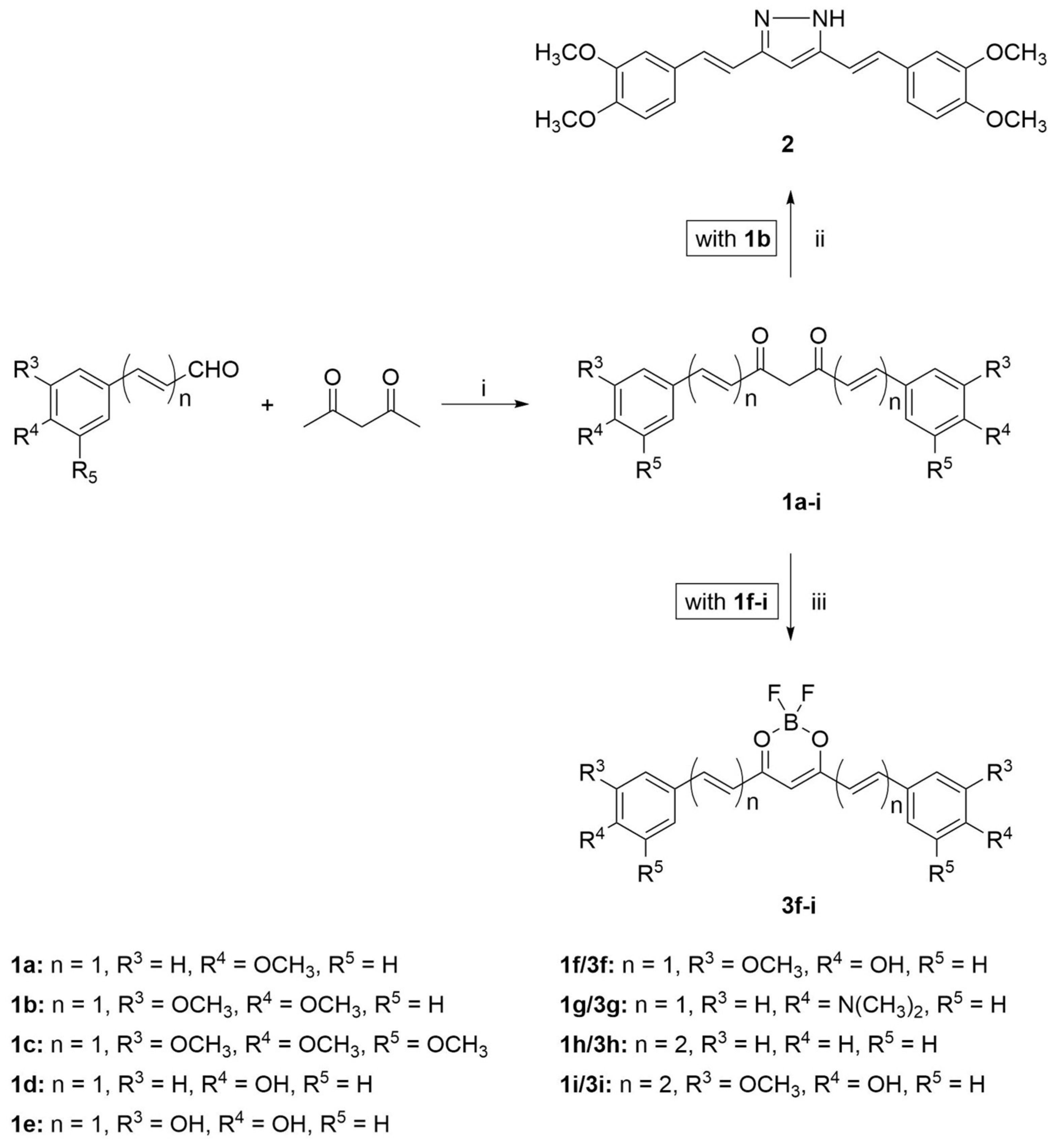
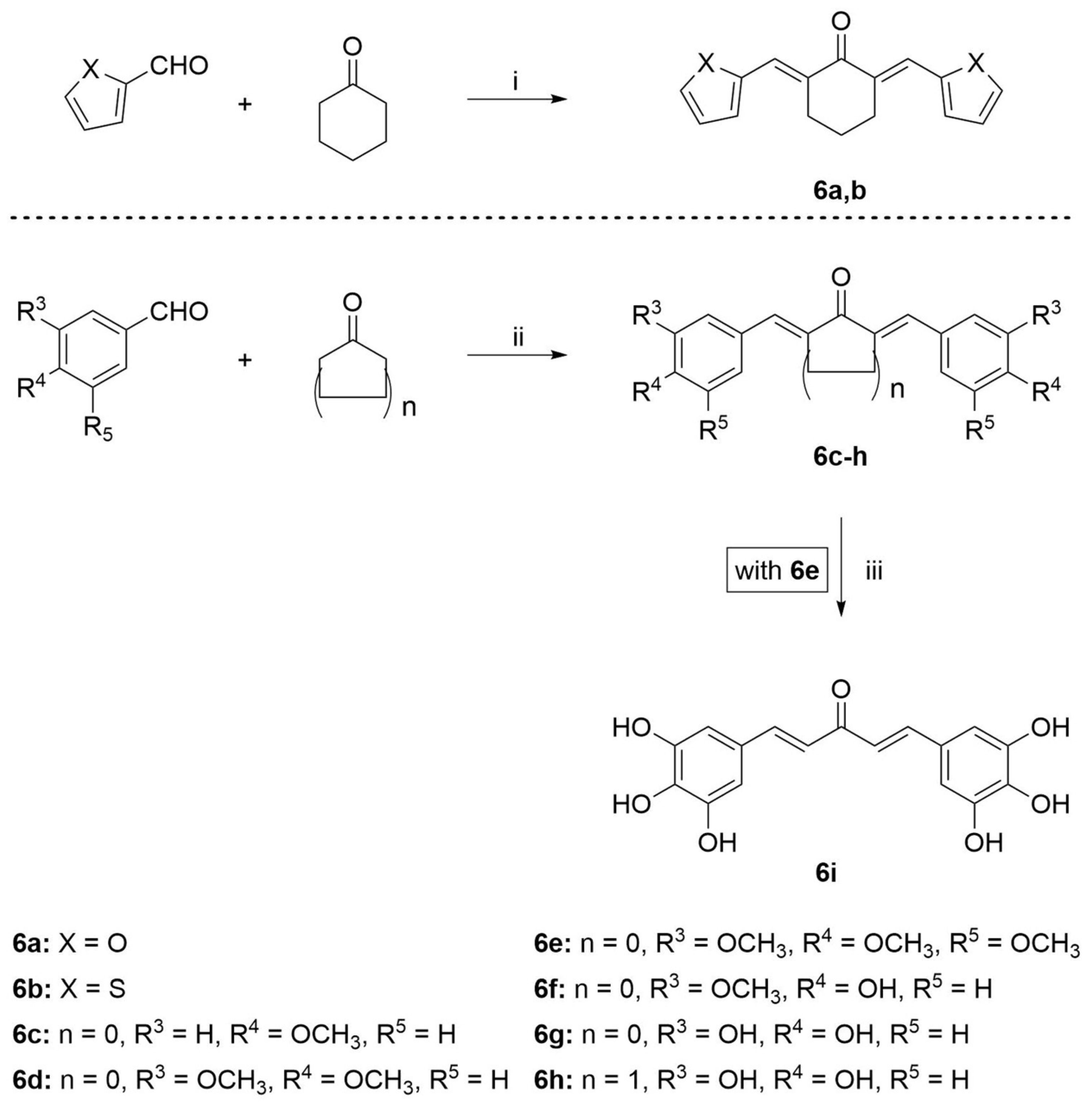
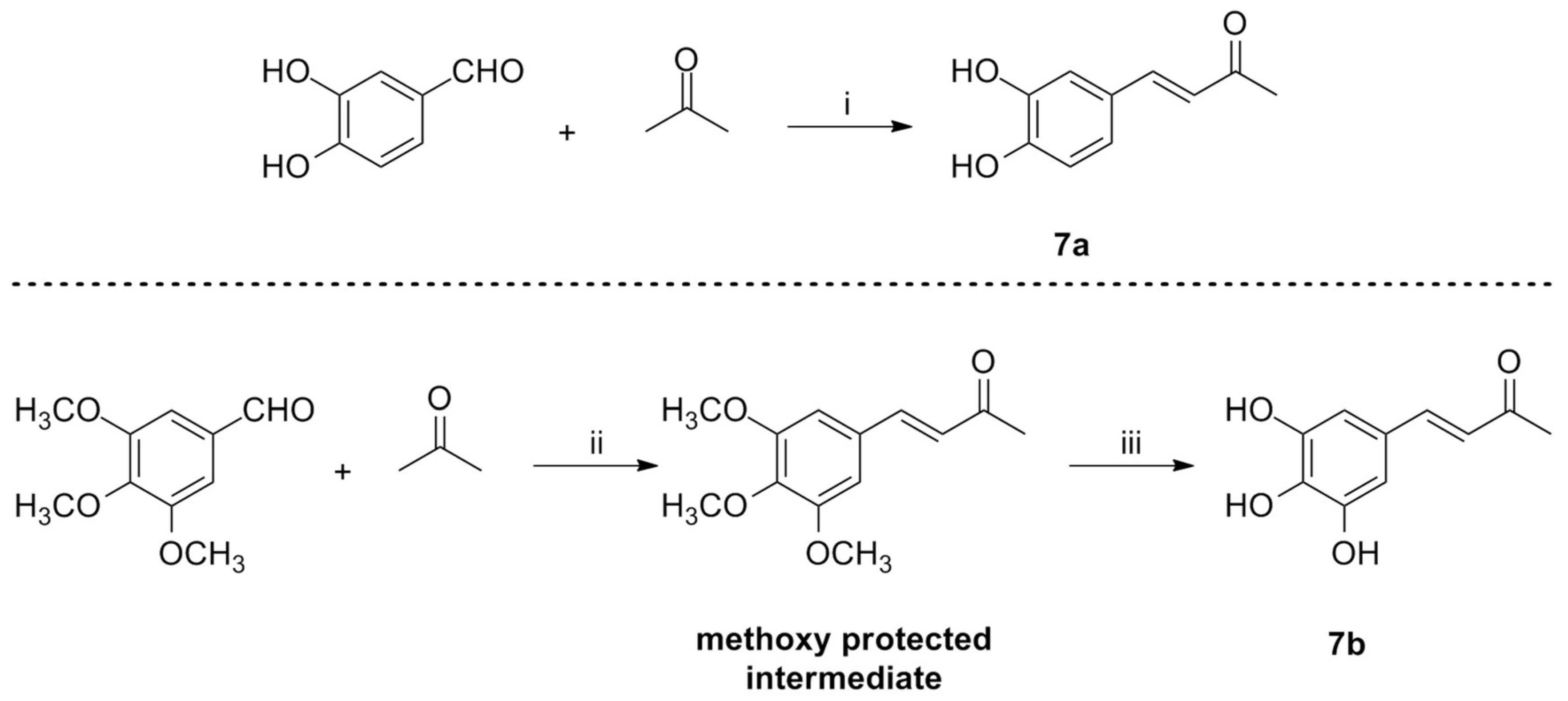
2.1. Synthesis
2.2. Larvicidal Activity
2.3. Calculations of Molecular Descriptors
3. Methods and Materials
3.1. General
3.2. Synthesis
3.2.1. Synthesis of Compounds 1a–i
- Orange solid, Yield: 1.46 g (44%); 1H NMR (500 MHz, DMSO-d6): δ 3.81 (s, 6H), 6.09 (s, 1H), 6.80 (d, 2H, J = 15.9 Hz), 7.01 (d, 4H, J = 8.6 Hz), 7.60 (d, 2H, J = 15.9 Hz), 7.69 (d, 4H, J = 8.5 Hz); Anal. Calcd for C21H20O4: C, 74.98, H, 5.99. Found C, 74.93, H, 6.05.
- Orange powder, Yield: 2.18 g (55%); 1H NMR (500 MHz, DMSO-d6): δ 3.81 (s, 6H), 3.83 (s, 6H), 6.11 (s, 1H), 6.84 (d, 2H, J = 15.9 Hz), 7.02 (d, 2H, J = 8.3 Hz), 7.27 (d, 2H, J = 8.3 Hz), 7.35 (s, 2H), 7.59 (d, 2H, J = 15.9 Hz); Anal. Calcd for C23H24O6: C, 69.68, H, 6.10. Found C, 69.62, H, 6.14.
- Orange solid, Yield: 1.87 mg (40%); 1H NMR (500 MHz, DMSO-d6): δ 3.71 (s, 6H), 3.84 (s, 12H), 6.18 (s, 1H), 6.94 (d, J = 16.0 Hz, 2H), 7.01 (s, 4H), 7.59 (d, J = 16.0 Hz, 2H); Anal. Calcd for C25H28O8: C, 65.78, H, 6.18. Found C, 65.84, H, 6.24.
- Orange solid, Yield: 1.61 g (52%); 1H NMR (500 MHz, DMSO-d6): δ 6.04 (s, 1H), 6.70 (d, J = 15.8 Hz, 2H), 6.82 (d, J = 8.3 Hz, 4H), 7.55–7.57 (m, 4H), 10.05 (br, 2H); Anal. Calcd for C19H16O4: C, 74.01, H, 5.23. Found C, 73.94, H, 5.26.
- Orange to red solid, Yield: 1.60 g (47%); 1H NMR (500 MHz, DMSO-d6): δ 6.04 (1H), 6.56 (J = 15.6 Hz, 2H), 6.77 (J = 8.3 Hz, 2H), 7.00 (J = 1.7 Hz, 2H), 7.07 (J = 8.3, 1.7 Hz, 2H), 7.46 (J = 15.6 Hz, 2H), 10.08 (br, 4H); Anal. Calcd for C19H16O6: C, 67.06, H, 4.74. Found C, 67.01, H, 4.71.
- Orange solid, Yield: 2.34 g (64%); 1H NMR (500 MHz, DMSO-d6): δ 3.83 (s, 6H), 6.06 (s, 2H), 6.76 (d, J = 15.8 Hz, 2H), 6.82 (d, J = 8.0 Hz, 2H), 7.15 (d, J = 8.0 Hz, 2H), 7.32 (s, 2H), 7.55 (d, J = 15.8 Hz, 2H), 9.65 (s, 4H); Anal. Calcd for C21H20O6: C, 68.47, H, 5.47. Found C, 68.41, H, 5.51.
- Dark red solid, Yield: 2.42g (67%); 1H NMR (500 MHz, DMSO-d6): 5.96 (s, 1H), 6.60 (d, J = 15.8 Hz, 2H), 6.73 (d, J = 8.8 Hz, 4H), 7.49–7.54 (m, 6H); Anal. Calcd for C23H26N2O2: C, 76.21, H, 7.23, N, 7.73. Found C, 76.23, H, 7.16, N, 7.69.
- Yellow to orange solid, Yield: 2.05 g (63%); 1H NMR (500 MHz, DMSO-d6): δ 6.09 (s, 2H), 6.38 (d, J = 15.1 Hz, 2H), 7.23–7.07 (m, 4H), 7.50–7.30 (m, 8H), 7.59 (d, J = 7.4 Hz, 4H); Anal. Calcd for C23H20O2: C, 84.12, H, 6.14. Found C, 84.16, H, 6.09.
- Deep red solid, Yield: 1.75 mg (42%); 1H NMR (500 MHz, DMSO-d6): δ 3.82 (s, 6H), 6.00 (s, 2H), 6.26 (d, J = 15.1 Hz, 2H), 6.78 (d, J = 7.8 Hz, 2H), 6.98–7.01 (m, 6H), 7.19 (s, 2H), 7.38–7.42 (m, 2H), 9.47 (s, 2H); Anal. Calcd for C25H24O6: C, 71.42, H, 5.75. Found C, 71.39, H, 5.80.
3.2.2. Synthesis of Compound 2
- 3,5-bis ((E)-3,4-dimethoxystyryl)-1H-pyrazole 2 [45]
- White to light pink solid, Yield: 905 mg (92%); 1H NMR (500 MHz, DMSO-d6): δ 3.77 (s, 3H), 3.82 (s, 3H), 6.65 (s, 1H), 6.91–6.99 (m, 3H), 7.03–7.09 (m, 5H), 7.15–7.21 (m, 2H), 12.88 (s, 1H); Anal. Calcd for C23H24N2O4: C, 70.39, H, 6.16, N, 7.14. Found C, 70.43, H, 6.10, N, 7.17.
3.2.3. Synthesis of Compounds 3f–i
- Dark red solid, Yield: 117 mg (47%); 1H NMR (500 MHz, DMSO-d6): δ 3.85 (s, 6H), 6.45 (s, 1H), 6.88 (d, J = 8.1 Hz, 2H), 7.02 (d, J = 15.6 Hz, 2H), 7.34 (d, J = 8.1 Hz 2H), 7.47 (s, 2H), 7.92 (d, J = 15.6 Hz, 2H), 10.09 (s, 2 H); Anal. Calcd for C21H19BF2O6: C, 60.61, H, 4.60. Found C, 60.63, H, 4.55.
- (1E,6E)-1,7-bis (4-(dimethylamino)phenyl) hepta-1,6-diene-3,5-dione-BF2 complex 3g [46] Black solid, Yield: 88 mg (36%); 1H NMR (500 MHz, DMSO-d6): δ 3.07 (s, 12H), 6.29 (s, 2H), 6.75 (d, J = 15.4 Hz, 2H), 6.79 (d, J = 8.9 Hz, 4H), 7.70 (d, J = 8.0 Hz, 4H), 8.01 (d, J = 15.4 Hz, 2H); Anal. Calcd for C23H25BF2N2O2: C, 67.33, H, 6.14, N, 6.83. Found C, 67.36, H, 6.10, N, 6.87.
- Black solid, Yield: 96 mg (42%); 1H NMR (500 MHz, DMSO-d6): δ 6.59 (d, J = 15.0 Hz, 2H), 6.62 (s, 1H), 7.30–7.46 (m, 11H), 8.21 (2 × d, J = 7.1 Hz, 4H), 7.84 (dd, J = 10.6 Hz, J = 15.0 Hz, 2H); Anal. Calcd for C23H19BF2O2: C, 73.43, H, 5.09. Found C, 73.39, H, 5.12.
- (1E,3E,8E,10E)-1,11-bis (4-hydroxy-3-methoxyphenyl) undeca-1,3,8,10-tetraene-5,7-dione-BF2 complex 3i
- Black solid, Yield: 128 mg (45%); 1H NMR (500 MHz, DMSO-d6): δ 3.84 (s, 6H), 6.43 (d, J = 14.8 Hz, 2H), 6.49 (s, 1H), 8.10 (d, J = 8.1 Hz, 2H), 6.97 (s, 2H), 7.07–7.30 (m, 12H), 7.77 (dd, J = 11.2 Hz, J = 15.8 Hz, 2H); 13C NMR (125 MHz, DMSO-d6): δ 55.7, 110.8, 115.8, 122.3, 123.2, 124.5, 127.5, 127.6, 127.5, 127.6, 146.1, 147.9, 148.0, 149.5, 178.0. Anal. Calcd for C25H23BF2O6: C, 64.13, H, 4.95. Found C, 64.15, H, 4.90.
3.2.4. Synthesis of Compound 4
- (E)-6-(4-hydroxy-3-methoxyphenyl) hex-5-ene-2,4-dione 4 [47]
- Pale yellow solid, Yield: 730 mg (35%); 1H NMR (500 MHz, DMSO-d6): δ 2.12 (s, 3H), 3.82 (s, 3H), 5.84 (s, 1H), 6.65 (d, J = 15.8 Hz, 1H), 6.80 (d, J = 8.1 Hz, 1H), 7.11 (d, J = 8.1 Hz, 1H), 7.29 (s, 1H), 7.49 (d, J = 15.9 Hz, 1H), 9.61 (s, 1H); Anal. Calcd for C13H14O4: C, 66.66, H, 6.02. Found C, 66.71, H, 5.97.
3.2.5. Synthesis of Compound 5
- Orange solid, Yield: 245 mg (42%); 1H NMR (500 MHz, DMSO-d6): δ 3.83 (s, 3H), 6.04 (s, 1H), 6.70 (d, J = 15.8 Hz, 1H), 6.75 (d, J = 15.8 Hz, 1H), 6.82 (m, 3H), 7.14 (d, J = 7.8 Hz, 1H), 7.32 (s, 1H), 7.52–7.57 (m, 4H), 9.68 (br, 1H), 10.05 (br, 1H); Anal. Calcd for C20H18O5: C, 71.00, H, 5.36. Found C, 70.91, H, 5.42.
3.2.6. Synthesis of Compounds 6a,b
- Orange needles, Yield: 2.26 g (89%); 1H NMR (500 MHz, DMSO-d6): δ 1.81 (quintet, J = 6.2 Hz, 2H), 2.95 (t, J = 6.2 Hz, 4H), 6.69 (dd, J = 1.8 Hz, J = 3.6 Hz, 2H), 6.95 (d, J = 3.6 Hz, 2H), 7.40 (s, 2H), 7.92 (d, J = 1.8 Hz, 2H); Anal. Calcd for C16H14O3: C, 75.58, H, 5.55. Found C, 75.68, H, 5.49.
- Yellow needles, Yield: 2.10 g (73%); 1H NMR (500 MHz, DMSO-d6): δ 1.90 (quintet, J = 6.2 Hz, 2H), 2.88 (t, J = 6.2 Hz, 4H), 7.15 (dd, J = 3.6 Hz, J = 5.0 Hz, 2H), 7.60 (d, J = 3.6 Hz, 2H), 7.86 (s, 2H), 7.90 (d, J = 5.0, 2H); Anal. Calcd for C16H14OS2: C, 67.10, H, 4.93, S, 22.39. Found C, 67.17, H, 4.99, S, 22.34.
3.2.7. Synthesis of Compounds 6c–e
- Light yellow needles, Yield: 1.84 mg (63%); 1H NMR (500 MHz, DMSO-d6): δ; 3.82 (s, 6H), 7.02 (d, J = 8.6 Hz, 4H), 7.19 (d, J = 15.8 Hz, 2H), 7.73 (d, J = 15.8, 2H), 7.74 (d, J = 8.6 Hz, 4H); Anal. Calcd for C19H18O3: C, 77.53, H, 6.16. Found C, 77.45, H, 6.14.
- Yellow neeedles, Yield: 2.84 mg (72%); 1H NMR (500 MHz, DMSO-d6): δ 3.82 (s, 6H), 3.84 (s, 6H), 7.03 (d, J = 8.3 Hz, 2H), 7.23 (d, J = 15.9 Hz, 2H), 7.33 (d, J = 8.3 Hz, 2H), 7.41 (s, 2H), 7.70 (d, J = 15.9 Hz, 2H); Anal. Calcd for C21H22O5: C, 71.17, H, 6.26. Found C, 71.11, H, 6.31.
- Yellow needles, Yield: 1.92 mg (46%); 1H NMR (500 MHz, DMSO-d6): δ 3.71 (s, 6H), 3.85 (s, 12H), 7.13 (s, 4H), 7.31 (d, J = 16.0 Hz, 2H), 7.71 (d, J = 16.0 Hz, 4H); Anal. Calcd for C23H26O7: C, 66.65, H, 6.32. Found C, 66.71, H, 6.27.
3.2.8. Synthesis of Compounds 6f–h
- Orange solid, Yield: 2.15 g (66%), 1H NMR (500 MHz, DMSO-d6): 3.85 (s, 6H), 6.83 (d, J = 8.2 Hz, 2H), 7.15 (d, J = 16.0 Hz, 2H), 7.20 (d, J = 8.2 Hz, 2H), 7.37 (s, 2H), 7.65 (d, J = 16.0 Hz, 2H), 9.65 (2H, s, OH); Anal. Calc. for C19H18O5: C 69.93, H 5.56. Found: C 69.82, H 5.50.
- Dark green solid, Yield: 1.45 g (49%); 1H NMR (500 MHz, DMSO-d6): δ 6.79 (d, J = 8.2 Hz, 2H), 6.99 (d, J = 16.0 Hz, 2H), 7.07 (d, J = 8.2 Hz, 2H), 7.14 (s, 2H), 7.56 (d, J = 16.0 Hz, 2H), 9.38 (br, 4H); Anal. Calcd for C17H14O5: C, 68.45, H, 4.73. Found C, 68.38, H, 4.79.
- 2,5-bis ((E)-3,4-dihydroxybenzylidene) cyclopentan-1-one 6h [53]
- Brown solid, Yield: 1.75 g (54%); 1H NMR (500 MHz, DMSO-d6): δ 3.00 (s, 4H), 6.83 (d, J = 8.2 Hz, 2H), 7.01 (d, J = 8.2 Hz, 2H), 7.11 (s, 2H), 7.24 (d, 2H), 9.39 (brs, 4H); Anal. Calcd for C19H16O5: C, 70.36, H, 4.97. Found C, 70.30, H, 5.06.
3.2.9. Synthesis of Compound 6i
- Dark green solid, Yield: 74 mg (45%); 1H NMR (500 MHz, DMSO-d6): δ 6.69 (s, 4H), 6.91 (d, J = 8.4 Hz, 2H), 7.45 (d, J = 8.4 Hz, 2H), 8.86 (br, 2H), 9.09 (br, 4H); Anal. Calcd for C17H14O7: C, 61.82, H, 4.27. Found C, 61.75, H, 4.33.
3.2.10. Synthesis of Compound 7a
- (E)-4-(3,4-dihydroxyphenyl) but-3-en-2-one 7a [54]
- Yellow solid, Yield: 1.18 g (66%); 1H NMR (500 MHz, DMSO-d6): δ 2.27 (s, 3H), 6.48 (d, J = 16.4 Hz, 1H), 6.77 (d, J = 8.2 Hz, 1H), 7.01 (dd, J = 1.5, Hz, J = 8.2 Hz, 1H), 7.06 (d, J = 1.5 Hz, 1H), 7.45 (d, J = 16.4 Hz, 1H), 9.37 (s, 1H, OH); Anal. Calcd for C10H10O3: C, 67.41, H, 5.66. Found C, 67.46, H, 5.60.
3.2.11. Synthesis of Compound 7b
- (E)-4-(3,4,5-trihydroxyphenyl) but-3-en-2-one 7b [52]
- Yellow solid, Yield: 52 mg (54% calculated from 3,4,5-trimethoxy intermediate); 1H NMR (500 MHz, DMSO-d6): δ 2.27 (s, 3H), 6.38 (d, J = 16.2 Hz, 1H), 6.61 (s, 2H), 7.36 (d, J = 16.2 Hz, 1H), 8.95 (br, 3H); Anal. Calcd for C10H10O4: C, 61.85, H, 5.19. Found C, 61.89, H, 5.12.
3.3. Larvicidal Evaluation
3.3.1. Mosquito Rearing
3.3.2. Larvicidal Bioassays
3.3.3. Data Analysis
3.4. In Silico Molecular Properties and Descriptors Predictions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- U.S. Department of Health and Human Services: Centers for Disease Control and Prevention (CDC). Illnesses on the Rise: From Mosquito, Tick and Flea Bites. Available online: https://www.cdc.gov/vitalsigns/vector-borne/index.html (accessed on 10 June 2021).
- Gascon, J.; Bern, C.; Pinazo, M.J. Chagas disease in Spain, the United States and other non-endemic countries. Acta Trop. 2010, 115, 22–27. [Google Scholar] [CrossRef]
- Reiter, P.; Sprenger, D. The used tire trade: A mechanism for the worldwide dispersal of container breeding mosquitoes. J. Am. Mosq. Control. Assoc. 1987, 3, 494–501. [Google Scholar]
- World Health Organization: Dengue and Severe Dengue. Available online: https://www.who.int/news-room/fact-sheets/detail/dengue-and-severe-dengue (accessed on 10 June 2021).
- European Centre for Disease Prevention and Control: Zika Virus Disease. Available online: https://www.ecdc.europa.eu/en/zika-virus-disease (accessed on 10 June 2021).
- World Health Organization: West Nile Virus. Available online: https://www.who.int/news-room/fact-sheets/detail/west-nile-virus (accessed on 10 June 2021).
- Franklinos, L.H.V.; Jones, K.E.; Redding, D.W.; Abubakar, I. The effect of global change on mosquito-borne disease. Lancet Infect. Dis. 2019, 19, e302–e312. [Google Scholar] [CrossRef]
- Bellini, R.; Michaelakis, A.; Petrić, D.; Schaffner, F.; Alten, B.; Angelini, P.; Aranda, C.; Becker, N.; Carrieri, M.; di Luca, M.; et al. Practical management plan for invasive mosquito species in Europe: I. Asian tiger mosquito (Aedes albopictus). Travel Med. Infect. Dis. 2020, 35, 101691. [Google Scholar] [CrossRef]
- Stefopoulou, A.; LaDeau, S.L.; Syrigou, N.; Balatsos, G.; Karras, V.; Lytra, Ι.; Boukouvala, E.; Papachristos, D.P.; Milonas, P.G.; Kapranas, A.; et al. Knowledge, attitude and practices survey in greece before the implementation of sterile insect technique against Aedes albopictus. Insects 2021, 12, 212. [Google Scholar] [CrossRef]
- Manica, M.; Cobre, P.; Rosà, R.; Caputo, B. Not in my backyard: Effectiveness of outdoor residual spraying from hand-held sprayers against the mosquito Aedes albopictus in Rome, Italy. Pest Manag. Sci. 2017, 73, 138–145. [Google Scholar] [CrossRef]
- Vinogradova, E.B. Culex pipiens Pipiens Mosquitoes: Taxonomy, Distribution, Ecology, Physiology, Genetics, Applied Importance and Control; Pensoft: Sofia, Bulgaria, 2000. [Google Scholar]
- Barr, A.R. The distribution of Culex p. pipiens and Culex p. quinquefasciatus in North America. Am. J. Trop. Med. Hyg. 1957, 6, 153–165. [Google Scholar] [CrossRef]
- European Centre for Disease Prevention and Control: Culex pipiens Group—Current Known Distribution: March 2021. Available online: https://www.ecdc.europa.eu/en/publications-data/culex-pipiens-group-current-known-distribution-march-2021 (accessed on 10 June 2021).
- Oliva, C.F.; Benedict, M.Q.; Collins, C.M.; Baldet, T.; Bellini, R.; Bossin, H.; Bouyer, J.; Corbel, V.; Facchinelli, L.; Fouque, F.; et al. Sterile Insect Technique (SIT) against Aedes species mosquitoes: A roadmap and good practice framework for designing, implementing and evaluating pilot field trials. Insects 2021, 12, 191. [Google Scholar] [CrossRef]
- Marcombe, S.; Farajollahi, A.; Healy, S.P.; Clark, G.G.; Fonseca, D.M. Insecticide resistance status of United States populations of Aedes albopictus and mechanisms involved. PLoS ONE 2014, 9, e101992. [Google Scholar] [CrossRef] [Green Version]
- Bedini, S.; Flamini, G.; Ascrizzi, R.; Venturi, F.; Bader, A.; Girardi, J.; Conti, B. Essential oils sensory quality and their bioactivity against the mosquito Aedes albopictus. Sci. Rep. 2018, 8, 17857. [Google Scholar] [CrossRef] [Green Version]
- Li, M.-X.; Ma, Y.-P.; Zhang, H.-X.; Sun, H.-Z.; Su, H.-H.; Pei, S.-J.; Du, Z.-Z. Repellent, larvicidal and adulticidal activities of essential oil from Dai medicinal plant Zingiber cassumunar against Aedes albopictus. Plant Divers. 2020, 2. [Google Scholar] [CrossRef]
- Zhu, J.; Zeng, X.; O’neal, M.; Schultz, G.; Tucker, B.; Coats, J.; Bartholomay, L.; Xue, R.-D. Mosquito larvicidal activity of botanical-based mosquito repellents. J. Am. Mosq. Control Assoc. 2008, 24, 161–168. [Google Scholar] [CrossRef]
- Kishore, N.; Mishra, B.B.; Tiwari, V.K.; Tripathi, V.; Lall, N. Natural products as leads to potential mosquitocides. Phytochem. Rev. 2014, 13, 587–627. [Google Scholar] [CrossRef] [Green Version]
- Khattak, S.; ur-Rehman, S.; Shah, H.U.; Ahmad, W.; Ahmad, M. Biological effects of indigenous medicinal plants Curcuma longa and Alpinia galanga. Fitoterapia 2005, 76, 254–257. [Google Scholar] [CrossRef] [PubMed]
- Ajaiyeoba, E.O.; Sama, W.; Essien, E.E.; Olayemi, J.O.; Ekundayo, O.; Walker, T.M.; Setzer, W.N. Larvicidal activity of turmerone-rich essential oils of Curcuma longa. Leaf and rhizome from Nigeria on Anopheles gambiae. Pharm. Biol. 2008, 46, 279–282. [Google Scholar] [CrossRef]
- Lacoma, A.; Garcia-Sierra, N.; Prat, C.; Ruiz-Manzano, J.; Haba, L.; Rosés, S.; Domínguez, J. Synergistic mosquito-repellent activity of Curcuma longa, Pogostemon heyneanus and Zanthoxylum limonella essential oils. J. Infect. Public Health 2015, 8, 323–328. [Google Scholar] [CrossRef] [Green Version]
- Madhu, S.K.; Shaukath, A.K.; Vijayan, V.A. Efficacy of bioactive compounds from Curcuma aromatica against mosquito larvae. Acta Trop. 2010, 113, 7–11. [Google Scholar] [CrossRef]
- Singha, S.; Chandra, G. Mosquito larvicidal activity of some common spices and vegetable waste on Culex quinquefasciatus and Anopheles stephensi. Asian Pac. J. Trop. Med. 2011, 4, 288–293. [Google Scholar] [CrossRef] [Green Version]
- Liu, J.; Zhang, M.; Fu, W.-J.; Hu, J.-F.; Dai, G.-H. Efficacy of bioactive compounds from Curcuma longa L. against mosquito larvae. J. Appl. Entomol. 2018, 142, 1–8. [Google Scholar] [CrossRef]
- Sagnou, M.; Mitsopoulou, K.P.; Koliopoulos, G.; Pelecanou, M.; Couladouros, E.A.; Michaelakis, A. Evaluation of naturally occurring curcuminoids and related compounds against mosquito larvae. Acta Trop. 2012, 123, 190–195. [Google Scholar] [CrossRef]
- Shaalan, E.A.; Canyon, D.; Younes, M.W.; Abdel-Wahab, H.; Mansour, A.H. A review of botanical phytochemicals with mosquitocidal potential. Environ. Int. 2005, 31, 1149–1166. [Google Scholar] [CrossRef]
- Larson, R.T.; Wessely, V.; Jiang, Z.; Lan, Q. Larvicidal activity of sterol carrier protein-2 inhibitor in four species of mosquitoes. J. Med. Entomol. 2008, 45, 439–444. [Google Scholar] [CrossRef]
- Chellasamy, P.; Kadarkarai, M.; Kalimuthu, K.; Mahesh, K.P. Mosquito larvicidal, pupicidal, adulticidal, and repellent activity of Artemisia nilagirica (Family: Compositae) against Anopheles stephensi and Aedes aegypti. Parasitol. Res. 2012, 111, 2241–2251. [Google Scholar] [CrossRef]
- Perumalsamy, H.; Jang, M.J.; Kim, J.R.; Kadarkarai, M.; Ahn, Y.-J. Larvicidal activity and possible mode of action of four flavonoids and two fatty acids identified in Millettia pinnata seed toward three mosquito species. Parasites Vectors 2015, 8, 237. [Google Scholar] [CrossRef] [Green Version]
- Singh, A.; Singh, J.V.; Rana, A.; Bhagat, K.; Gulati, H.V.; Kumar, R.; Salwan, R.; Bhagat, K.; Kaur, G.; Singh, N.; et al. Monocarbonyl curcumin-based molecular hybrids as potent antibacterial agents. ACS Omega 2019, 4, 11673–11684. [Google Scholar] [CrossRef]
- Wiggers, H.J.; Zaioncz, S.; Cheleski, J.; Mainardes, R.M.; Khalil, N.M. Chapter 7-Curcumin, a multitarget phytochemical: Challenges and perspectives. Stud. Nat. Prod. Chem. 2017, 53, 243–276. [Google Scholar] [CrossRef]
- Hotsumi, M.; Tajiri, M.; Nikaido, Y.; Sato, T.; Makabe, K.; Konno, H. Design, synthesis, and evaluation of a water soluble C5-monoketone type curcumin analogue as a potent amyloid β aggregation inhibitor. Bioorg. Med. Chem. Lett. 2019, 29, 2157–2161. [Google Scholar] [CrossRef] [PubMed]
- Qudjani, E.; Iman, M.; Davood, A.; Ramandi, M.F.; Shafiee, A. Design and synthesis of curcumin-like diarylpentanoid analogues as potential anticancer agents. Recent Pat. Anticancer Drug Discov. 2016, 11, 342–351. [Google Scholar] [CrossRef] [PubMed]
- Anstrom, D.M.; Zhou, X.; Kalk, C.N.; Song, B.; Lan, Q. Mosquitocidal properties of natural product compounds isolated from Chinese herbs and synthetic analogs of curcumin. J. Med. Entomol. 2012, 49, 350–355. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Targanski, S.K.; Sousa, J.R.; de Pádua, G.M.; de Sousa, J.M.; Vieira, L.C.; Soares, M.A. Larvicidal activity of substituted chalcones against Aedes aegypti (Diptera: Culicidae) and non-target organisms. Pest Manag. Sci. 2021, 77, 325–334. [Google Scholar] [CrossRef] [PubMed]
- Begum, N.A.; Roy, N.; Laskar, R.A.; Roy, K. Mosquito larvicidal studies of some chalcone analogues and their derived products: Structure–activity relationship analysis. Med. Chem. Res. 2011, 20, 184–191. [Google Scholar] [CrossRef]
- Lee, S.-H.; Choi, J.Y.; Lee, B.R.; Fang, Y.; Kim, J.H.; Park, D.H.; Park, M.G.; Woo, R.M.; Kim, W.J.; Je, Y.H. Insect growth regulatory and larvicidal activity of chalcones against Aedes albopictus. Entomol. Res. 2018, 48, 55–59. [Google Scholar] [CrossRef] [Green Version]
- Arshia, H.; Yee, L.S.; Lim, C.W.; Sofian, A.M.; Sanghiran, L.V.; Khalijah, A. Inhibition and larvicidal activity of phenylpropanoids from piper sarmentosum on acetylcholinesterase against mosquito vectors and their binding mode of interaction. PLoS ONE 2016, 11, e0155265. [Google Scholar]
- Liu, J.; Fernandez, D.; Gao, Y.; Silvie, P.; Gao, Y.; Dai, G. Enzymology, histological and ultrastructural effects of ar-turmerone on Culex pipiens pallens larvae. Insects 2020, 11, 336. [Google Scholar] [CrossRef] [PubMed]
- Mazumder, A.; Neamati, N.; Sunder, S.; Schulz, J.; Pertz, H.; Eich, E.; Pommier, Y. Curcumin analogs with altered potencies against HIV-1 integrase as probes for biochemical mechanisms of drug action. J. Med. Chem. 1997, 40, 3057–3063. [Google Scholar] [CrossRef]
- Laali, K.K.; Greves, W.J.; Correa-Smits, S.J.; Zwarycz, A.T.; Bunge, S.D.; Borosky, G.L.; Manna, A.; Paulus, A.; Chanan-Khan, A. Novel fluorinated curcuminoids and their pyrazole and isoxazole derivatives: Synthesis, structural studies, Computational/Docking and in-vitro bioassay. J. Fluor. Chem. 2018, 206, 82–98. [Google Scholar] [CrossRef]
- Kazantzis, K.T.; Koutsonikoli, K.; Mavroidi, B.; Zachariadis, M.; Alexiou, P.; Pelecanou, M.; Politopoulos, K.; Alexandratou, E.; Sagnou, M. Curcumin derivatives as photosensitizers in photodynamic therapy: Photophysical properties and in vitro studies with prostate cancer cells. Photochem. Photobiol. Sci. 2020, 19, 193–206. [Google Scholar] [CrossRef]
- Lin, L.; Nyarko, A.K.; Bastow, K.F.; Wu, C.-C.; Su, C.-Y.; Shih, C.C.-Y.; Lee, K.-H. Antitumor agents. 250. Design and synthesis of new curcumin analogues as potential anti-prostate cancer agents. J. Med. Chem. 2006, 49, 3963–3972. [Google Scholar] [CrossRef] [Green Version]
- Dong, H.-H.; Wang, Y.-H.; Peng, X.-M.; Zhou, H.-Y.; Zhao, F.; Jiang, Y.-Y.; Zhang, D.-Z.; Jin, Y.-S. Synergistic antifungal effects of curcumin derivatives as fungal biofilm inhibitors with fluconazole. Chem. Biol. Drug Des. 2021, 97, 1079–1088. [Google Scholar] [CrossRef]
- Liu, K.; Chen, J.; Chojnacki, J.; Zhang, S. BF3·OEt2-promoted concise synthesis of difluoroboron-derivatized curcumins from aldehydes and 2,4-pentanedione. Tetrahedron Lett. 2013, 54, 2070–2073. [Google Scholar] [CrossRef] [Green Version]
- Feng, J.-Y.; Liu, Z.-Q. Feruloylacetone as the model compound of half-curcumin: Synthesis and antioxidant properties. Eur. J. Med. Chem. 2011, 46, 1198–1206. [Google Scholar] [CrossRef]
- Kar, S.; Ramamoorthy, G.; Sinha, S.; Ramanan, M.; Pola, J.K.; Golakoti, N.R.; Nanubolu, J.B.; Sahoo, S.K.; Dandamudi, R.B.; Doble, M. Synthesis of diarylidenecyclohexanone derivatives as potential anti-inflammatory leads against COX-2/mPGES1 and 5-LOX. New J. Chem. 2019, 43, 9012–9020. [Google Scholar] [CrossRef]
- Youssef, K.M.; El-Sherbeny, M.A.; El-Shafie, F.S.; Farag, H.A.; Al-Deeb, O.A.; Awadalla, S.A.A. Synthesis of curcumin analogues as potential antioxidant, cancer chemopreventive agents. Arch. Pharm. 2004, 337, 42–54. [Google Scholar] [CrossRef]
- Matiadis, D.; Mavroidi, B.; Panagiotopoulou, A.; Methenitis, C.; Pelecanou, M.; Sagnou, M. (E)-(1-(4-Ethoxycarbonylphenyl)-5-(3,4-dimethoxyphenyl)-3-(3,4-dimethoxystyryl)-2-pyrazoline: Synthesis, characterization, DNA-interaction, and evaluation of activity against drug-resistant cell lines. Molbank 2020, 2020, M1114. [Google Scholar] [CrossRef] [Green Version]
- Yamakoshi, H.; Ohori, H.; Kudo, C.; Sato, A.; Kanoh, N.; Ishioka, C.; Shibata, H.; Iwabuchi, Y. Structure–activity relationship of C5-curcuminoids and synthesis of their molecular probes thereof. Bioorg. Med. Chem. 2010, 18, 1083–1092. [Google Scholar] [CrossRef]
- Matiadis, D.; Ng, S.-T.; Chen, E.H.-L.; Nigianni, G.; Vidali, V.P.; Canko, A.; Chen, R.P.-Y.; Sagnou, M. Synthesis and biological evaluation of hydroxylated monocarbonyl curcumin derivatives as potential inducers of neprilysin activity. Biomedicines 2021, 9, 955. [Google Scholar] [CrossRef]
- Du, Z.-Y.; Liu, R.-R.; Shao, W.-Y.; Mao, Z.-P.; Ma, L.; Gu, L.-P.; Huang, Z.-S.; Chan, A.S.C. α-Glucosidase inhibition of natural curcuminoids and curcumin analogues. Eur. J. Med. Chem. 2006, 41, 213–218. [Google Scholar] [CrossRef]
- Chao, W.; Deng, J.-S.; Li, P.-Y.; Liang, Y.-C.; Huang, G.-J. 3,4-Dihydroxybenzalactone suppresses human non-small cell lung carcinoma cells metastasis via suppression of epithelial to mesenchymal transition, ROS-mediated PI3K/AKT/MAPK/MMP and NFκB signaling pathways. Molecules 2017, 22, 537. [Google Scholar] [CrossRef] [Green Version]
- Giatropoulos, A.; Papachristos, D.P.; Kimbaris, A.; Koliopoulos, G.; Polissiou, M.G.; Emmanouel, N.; Michaelakis, A. Evaluation of bioefficacy of three Citrus essential oils against the dengue vector Aedes albopictus (Diptera: Culicidae) in correlation to their components enantiomeric distribution. Parasitol. Res. 2012, 111, 2253–2263. [Google Scholar] [CrossRef]
- Theochari, I.; Giatropoulos, A.; Papadimitriou, V.; Karras, V.; Balatsos, G.; Papachristos, D.; Michaelakis, A. Physicochemical characteristics of four limonene-based nanoemulsions and their larvicidal properties against two mosquito species, Aedes albopictus and Culex pipiens molestus. Insects 2020, 11, 740. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization: Guidelines for Laboratory and Field Testing of Mosquito Larvicides. Available online: https://apps.who.int/iris/handle/10665/69101 (accessed on 10 June 2021).
- SPSS Inc. SPSS 14 for Windows Users Guide; SPSS Inc.: Chicago, IL, USA, 2004. [Google Scholar]
- Schrödinger LLC. Schrödinger Release 2020-3: Canvas; Schrödinger, LLC: New York, NY, USA, 2020. [Google Scholar]
- Schrödinger LLC. Schrödinger Release 2020-3: MacroModel; Schrödinger, LLC: New York, NY, USA, 2020. [Google Scholar]

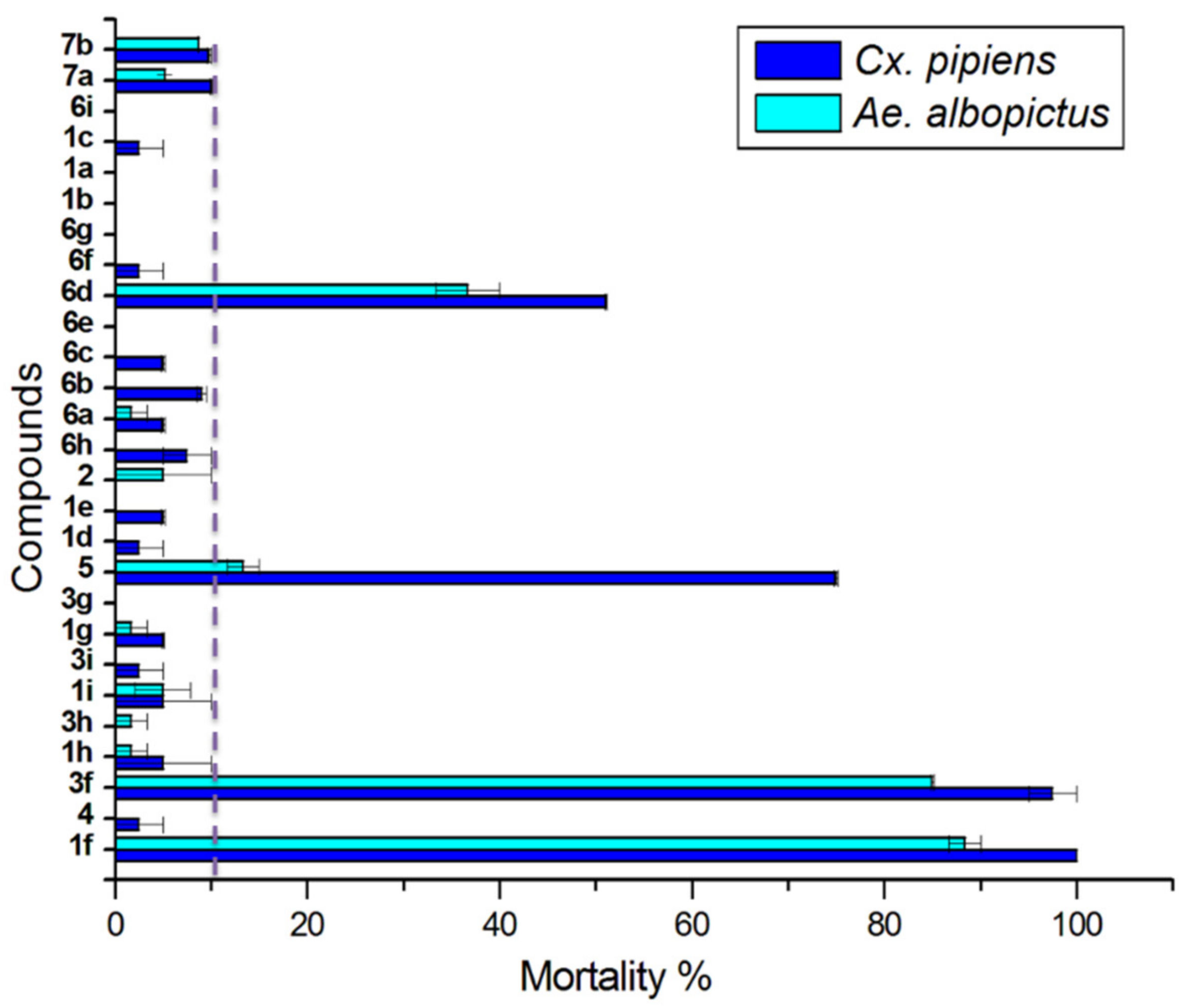
| Compound | Slope (±SE) | LC50 (95% CL) a | LC90 (95% CL) a | x2 | df | |
|---|---|---|---|---|---|---|
| Aedes albopictus | 1f | 6.4 ± 0.6 | 9.2 8.5–9.9 | 14.5 12.8–17.8 | 37.064 | 20 |
| 5 | 9.7 ± 1.1 | 36.0 32.6–39.5 | 48.8 43.6–60.9 | 26.174 b | 10 | |
| 3f | 2.3 ± 0.2 | 5.5 4.6–6.3 | 20.2 16.4–26.9 | 14.302 | 22 | |
| 6d | 4.2 ± 0.5 | 23.6 21.3–26.2 | 47.38 40.5–59.8 | 14.013 | 10 | |
| Culex pipiens | 1f | 4.0 ± 0.4 | 6.0 5.4–6.6 | 12.5 11.0–15.0 | 10.395 | 14 |
| 5 | 3.1 ± 0.3 | 9.4 8.2–10.6 | 24.0 20.2–30.5 | 10.194 b | 13 | |
| 3f | 2.4 ± 0.2 | 5.0 4.4–5.7 | 17.0 14.3–21.2 | 15.977 | 24 | |
| 6d | 10.6 ± 0.9. | 32.5 31.4–33.7 | 42.93 40.8–45.9 | 12.193 | 17 |
| Compound | AlogP | HBA | HBD | RB | PSA | Estate | MR | Polar |
|---|---|---|---|---|---|---|---|---|
| 1a | 3.54 | 4 | 0 | 8 | 52.6 | 60.50 | 99.85 | 44.20 |
| 1b | 3.51 | 6 | 0 | 10 | 71.06 | 70.84 | 112.77 | 49.14 |
| 1c | 3.48 | 8 | 0 | 12 | 89.52 | 81.17 | 125.70 | 54.09 |
| 1d | 3.04 | 4 | 2 | 6 | 74.6 | 61.50 | 90.31 | 40.53 |
| 1e | 2.51 | 6 | 4 | 6 | 115.06 | 72.84 | 93.69 | 41.80 |
| 1f | 3.01 | 6 | 2 | 8 | 93.06 | 71.84 | 103.2 | 45.47 |
| 1g | 3.90 | 2 | 0 | 8 | 40.62 | 61.50 | 115.78 | 49.30 |
| 1h | 4.51 | 2 | 0 | 8 | 34.14 | 58.17 | 107.56 | 46.21 |
| 1i | 3.94 | 6 | 2 | 10 | 93.06 | 79.84 | 123.87 | 52.43 |
| 3f | 4.53 | 6 | 2 | 6 | 85.22 | 78.84 | 107.61 | 45.74 |
| 3g | 5.42 | 2 | 0 | 6 | 32.78 | 68.50 | 120.15 | 49.57 |
| 3h | 6.03 | 2 | 0 | 6 | 26.3 | 65.17 | 111.93 | 46.48 |
| 3i | 5.46 | 6 | 2 | 8 | 85.22 | 86.87 | 128.25 | 52.70 |
| 2 | 3.85 | 5 | 3 | 6 | 87.6 | 63.84 | 106.70 | 48.32 |
| 4 | 1.27 | 4 | 1 | 5 | 63.6 | 47.33 | 64.66 | 27.80 |
| 5 | 3.03 | 5 | 2 | 7 | 83.83 | 66.67 | 96.77 | 43.00 |
| 6a | 3.12 | 3 | 0 | 2 | 43.35 | 42.83 | 69.08 | 31.65 |
| 6b | 4.24 | 1 | 0 | 2 | 73.55 | 39.50 | 81.97 | 37.10 |
| 6c | 3.77 | 3 | 0 | 6 | 35.53 | 50.33 | 89.95 | 40.44 |
| 6d | 3.74 | 5 | 0 | 8 | 53.99 | 60.67 | 102.88 | 45.39 |
| 6e | 3.71 | 7 | 0 | 10 | 72.45 | 71.00 | 115.81 | 50.33 |
| 6f | 3.24 | 5 | 2 | 6 | 75.99 | 61.67 | 93.34 | 41.72 |
| 6g | 2.74 | 5 | 4 | 4 | 97.99 | 66.67 | 83.80 | 38.05 |
| 6h | 3.47 | 5 | 4 | 2 | 97.99 | 65.00 | 90.72 | 40.94 |
| 6i | 2.20 | 7 | 6 | 4 | 138.45 | 74.00 | 87.19 | 39.32 |
| 7a | 1.25 | 3 | 2 | 2 | 57.53 | 37.67 | 49.99 | 22.21 |
| 7b | 0.98 | 4 | 3 | 2 | 77.76 | 43.33 | 51.69 | 22.84 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matiadis, D.; Liggri, P.G.V.; Kritsi, E.; Tzioumaki, N.; Zoumpoulakis, P.; Papachristos, D.P.; Balatsos, G.; Sagnou, M.; Michaelakis, A. Curcumin Derivatives as Potential Mosquito Larvicidal Agents against Two Mosquito Vectors, Culex pipiens and Aedes albopictus. Int. J. Mol. Sci. 2021, 22, 8915. https://doi.org/10.3390/ijms22168915
Matiadis D, Liggri PGV, Kritsi E, Tzioumaki N, Zoumpoulakis P, Papachristos DP, Balatsos G, Sagnou M, Michaelakis A. Curcumin Derivatives as Potential Mosquito Larvicidal Agents against Two Mosquito Vectors, Culex pipiens and Aedes albopictus. International Journal of Molecular Sciences. 2021; 22(16):8915. https://doi.org/10.3390/ijms22168915
Chicago/Turabian StyleMatiadis, Dimitris, Panagiota G. V. Liggri, Eftichia Kritsi, Niki Tzioumaki, Panagiotis Zoumpoulakis, Dimitrios P. Papachristos, George Balatsos, Marina Sagnou, and Antonios Michaelakis. 2021. "Curcumin Derivatives as Potential Mosquito Larvicidal Agents against Two Mosquito Vectors, Culex pipiens and Aedes albopictus" International Journal of Molecular Sciences 22, no. 16: 8915. https://doi.org/10.3390/ijms22168915
APA StyleMatiadis, D., Liggri, P. G. V., Kritsi, E., Tzioumaki, N., Zoumpoulakis, P., Papachristos, D. P., Balatsos, G., Sagnou, M., & Michaelakis, A. (2021). Curcumin Derivatives as Potential Mosquito Larvicidal Agents against Two Mosquito Vectors, Culex pipiens and Aedes albopictus. International Journal of Molecular Sciences, 22(16), 8915. https://doi.org/10.3390/ijms22168915









