Boron and Gadolinium Loaded Fe3O4 Nanocarriers for Potential Application in Neutron Capture Therapy
Abstract
:1. Introduction
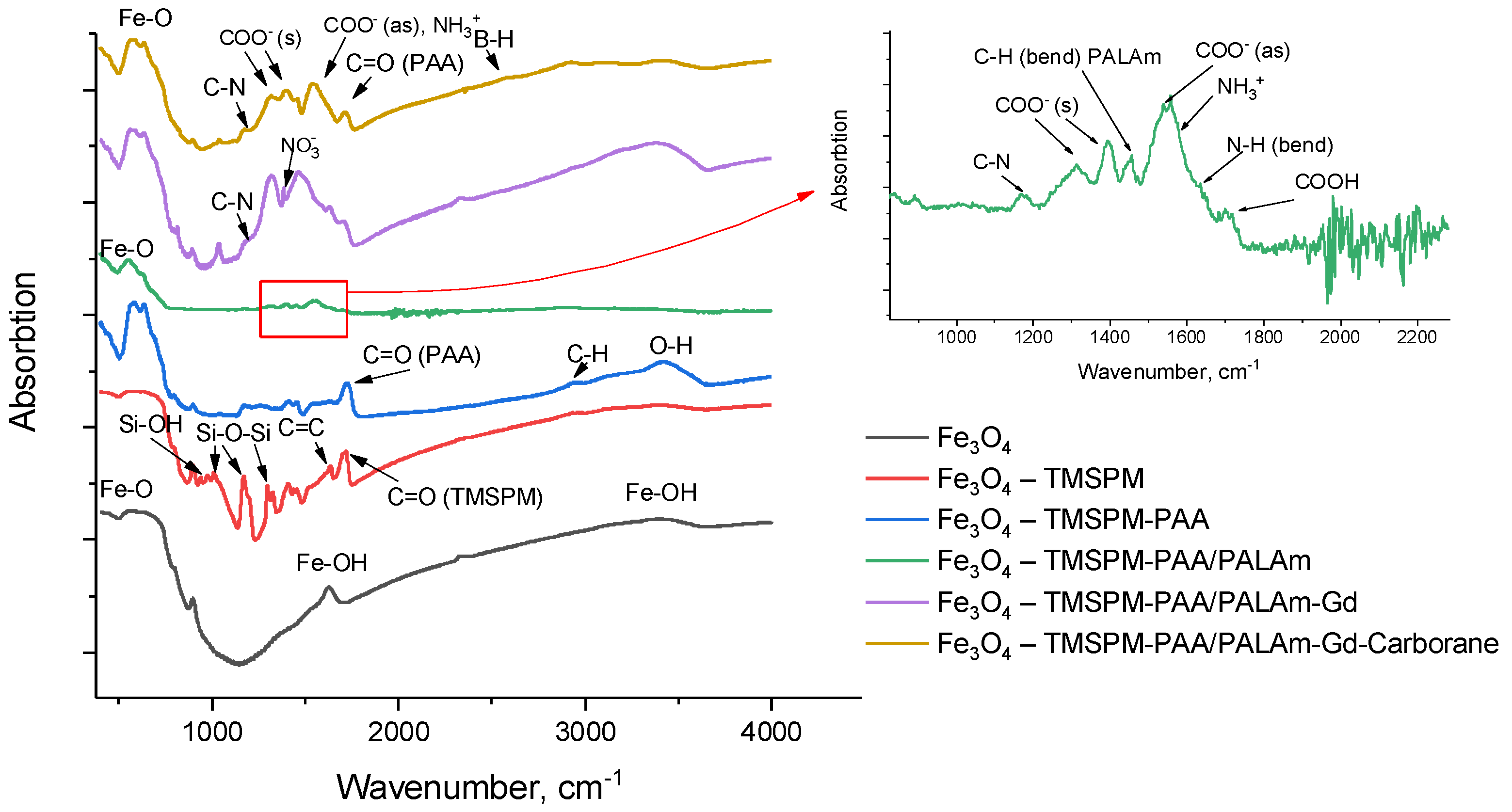
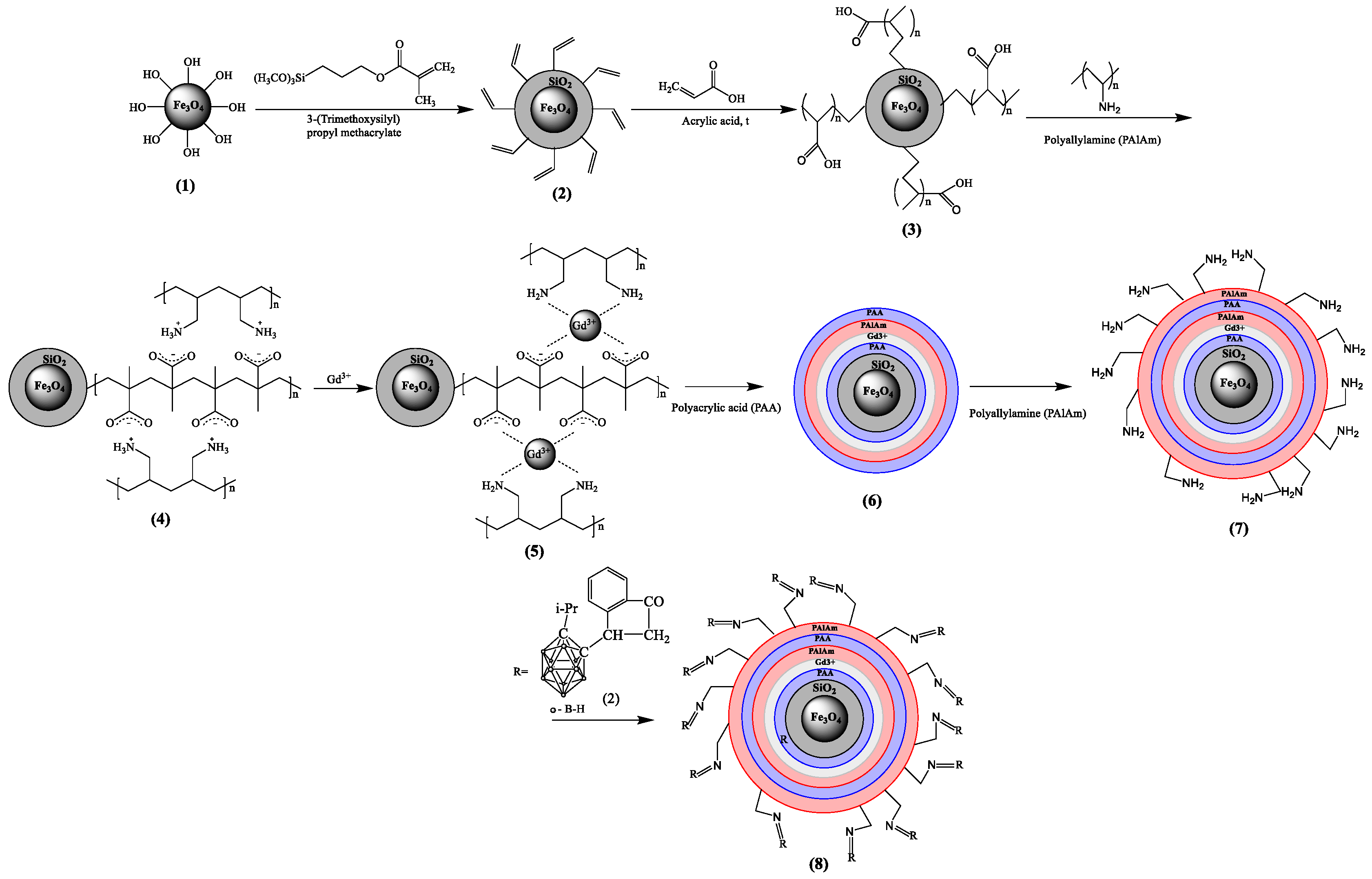
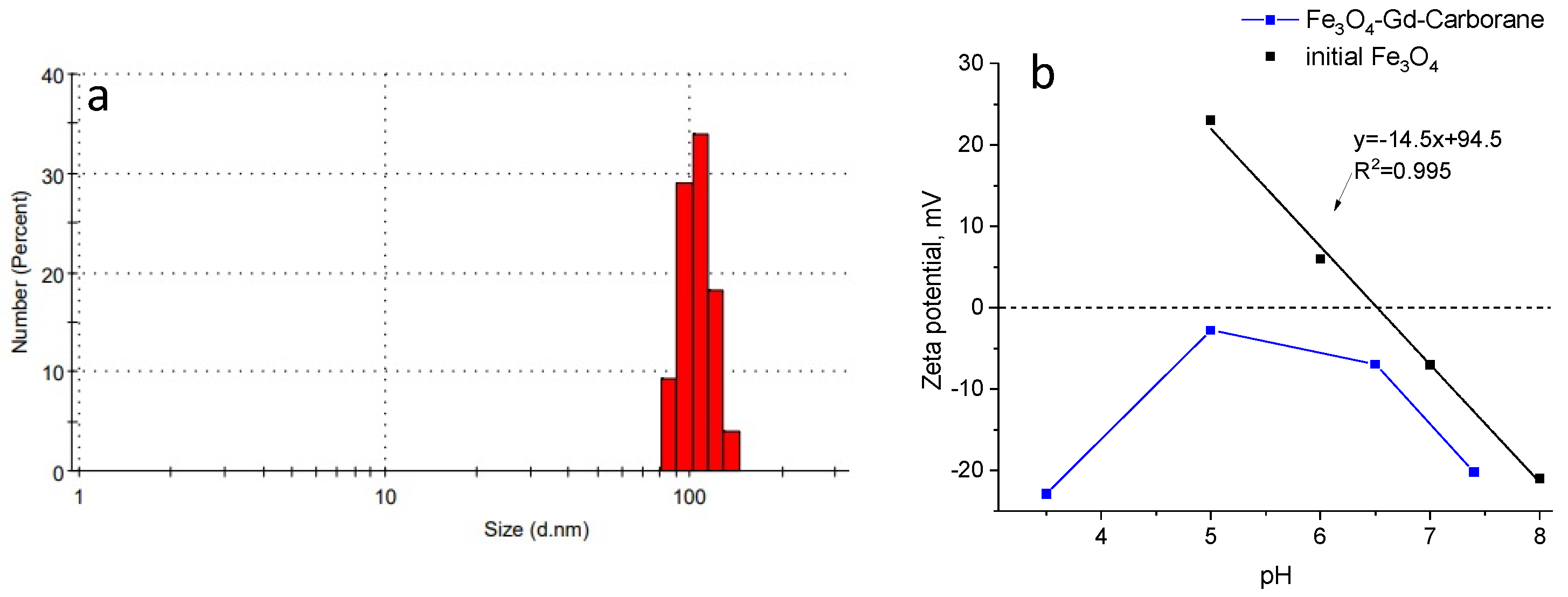
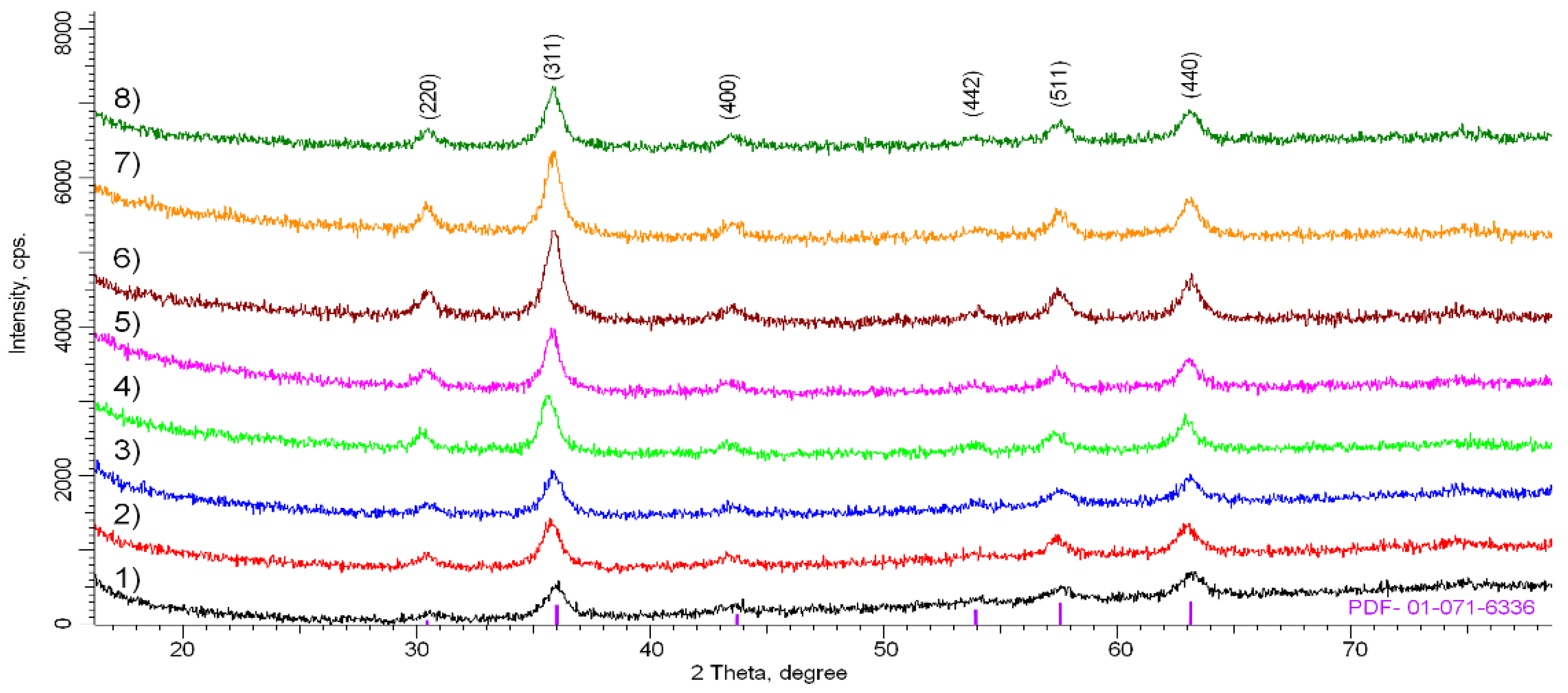
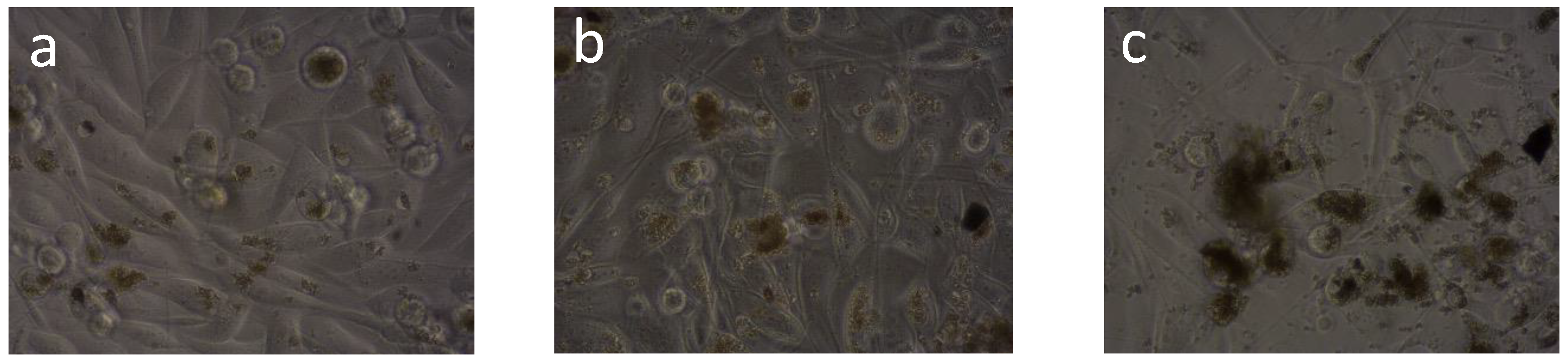
2. Results and Discussion
3. Materials and Methods
3.1. Materials
3.2. Synthesis of Fe3O4 Nanoparticles (NPs)
3.3. Functionalization of Fe3O4 NPs
3.3.1. Double-Bond Formation on Fe3O4 NPs
3.3.2. Thermoinitiated Graft Polymerization of Acrylic Acid
3.3.3. Immobilization of PALAm
3.3.4. Immobilization of Gd Ions
3.3.5. Immobilization of Polyelectrolytic Layer
3.4. Immobilization of Carborane Containing Agents
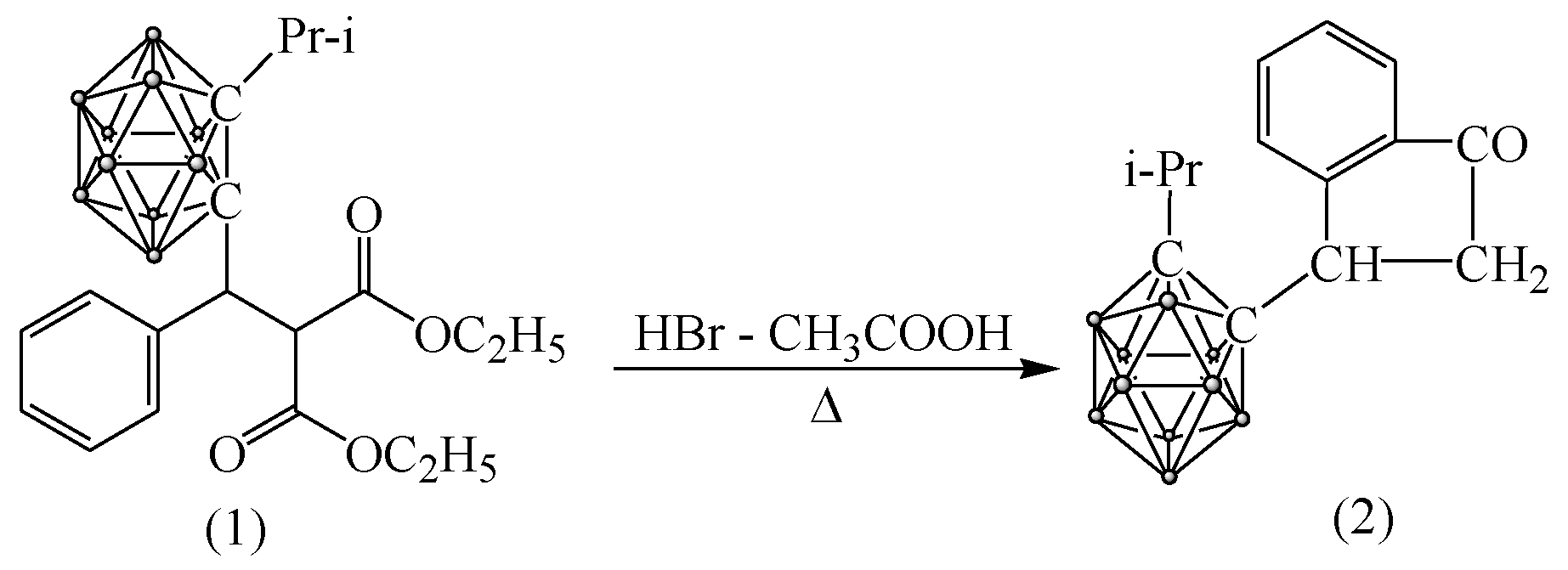
3.4.1. Synthesis of 3-(Isopropyl-o-carboranyl) Hydrindone
3.4.2. Immobilization of 3-(Isopropyl-o-carboranyl) Hydrindone
3.5. Methods of Characterization
3.6. Colorimetric Essay
3.7. Cytotoxicity Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- WHO Cancer. Available online: https://www.who.int/health-topics/cancer#tab=tab_1 (accessed on 12 August 2021).
- Bidram, E.; Esmaeili, Y.; Ranji-Burachaloo, H.; Al-Zaubai, N.; Zarrabi, A.; Stewart, A.; Dunstan, D.E. A concise review on cancer treatment methods and delivery systems. J. Drug Deliv. Sci. Technol. 2019, 54, 101350. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2019. CA. Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef] [Green Version]
- Thomas, G.; Eisenhauer, E.; Bristow, R.G.; Grau, C.; Hurkmans, C.; Ost, P.; Guckenberger, M.; Deutsch, E.; Lacombe, D.; Weber, D.C. The European Organisation for Research and Treatment of Cancer, State of Science in radiation oncology and priorities for clinical trials meeting report. Eur. J. Cancer 2020, 131, 76–88. [Google Scholar] [CrossRef]
- Riveiro-Barciela, M.; Trallero-Araguás, E.; Martínez-Valle, F.; Roca-Herrera, M.; Zamora, E.; Barreira-Díaz, A.; Muñoz-Couselo, E. Toxicities from immunotherapy: From clinical trials to real-world clinical practice. Med. Clín. Engl. Ed. 2020, 155, 541–547. [Google Scholar] [CrossRef]
- Amini, S.M. Gold nanostructures absorption capacities of various energy forms for thermal therapy applications. J. Therm. Biol. 2019, 79, 81–84. [Google Scholar] [CrossRef]
- Kalia, V.C.; Patel, S.K.S.; Cho, B.K.; Wood, T.K.; Lee, J.K. Emerging applications of bacteria as antitumor agents. Semin. Cancer Biol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Hongxiu, J.; Xiaoming, Y. Microbial-based cancer therapy—Bugs as drugs: History & the essential role of medical imaging. Cancer Treat. Res. Commun. 2021, 28, 100436. [Google Scholar] [CrossRef]
- Cohen-Jonathan-Moyal; Vendrely, V.; Motte, L.; Balosso, J.; Thariat, J. Radioresistant tumours: From identification to targeting. Cancer/Radiotherapie 2020, 24, 699–705. [Google Scholar] [CrossRef]
- Lan, T.L.; Chou, F.I.; Lin, K.H.; Pan, P.S.; Lee, J.C.; Huang, W.S.; Liu, Y.M.; Chao, Y.; Chen, Y.W. Using salvage Boron Neutron Capture Therapy (BNCT) for recurrent malignant brain tumors in Taiwan. Appl. Radiat. Isot. 2020, 160, 109105. [Google Scholar] [CrossRef] [PubMed]
- Issa, F.; Ioppolo, J.A.; Rendina, L.M. Boron and Gadolinium Neutron Capture Therapy. In Comprehensive Inorganic Chemistry II (Second Edition): From Elements to Applications; Elsevier: Amsterdam, The Netherlands, 2013; Volume 3, pp. 877–900. ISBN 9780080965291. [Google Scholar]
- Vares, G.; Jallet, V.; Matsumoto, Y.; Rentier, C.; Takayama, K.; Sasaki, T.; Hayashi, Y.; Kumada, H.; Sugawara, H. Functionalized mesoporous silica nanoparticles for innovative boron-neutron capture therapy of resistant cancers. Nanomed. Nanotechnol. Biol. Med. 2020, 27, 102195. [Google Scholar] [CrossRef]
- Hu, K.; Yang, Z.; Zhang, L.; Xie, L.; Wang, L.; Xu, H.; Josephson, L.; Liang, S.H.; Zhang, M. Boron agents for neutron capture therapy. Coord. Chem. Rev. 2020, 405, 213139. [Google Scholar] [CrossRef]
- Barth, R.F.; Grecula, J.C. Boron neutron capture therapy at the crossroads—Where do we go from here? Appl. Radiat. Isot. 2020, 160, 109029. [Google Scholar] [CrossRef] [PubMed]
- Farhood, B.; Samadian, H.; Ghorbani, M.; Zakariaee, S.S.; Knaup, C. Physical, dosimetric and clinical aspects and delivery systems in neutron capture therapy. Rep. Pract. Oncol. Radiother. 2018, 23, 462–473. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, H.; Koganei, H.; Miyoshi, T.; Sakurai, Y.; Ono, K.; Suzuki, M. Antitumor effect of boron nitride nanotubes in combination with thermal neutron irradiation on BNCT. Bioorg. Med. Chem. Lett. 2015, 25, 172–174. [Google Scholar] [CrossRef]
- Sherje, A.P.; Jadhav, M.; Dravyakar, B.R.; Kadam, D. Dendrimers: A versatile nanocarrier for drug delivery and targeting. Int. J. Pharm. 2018, 548, 707–720. [Google Scholar] [CrossRef]
- Ueno, M.; Ban, H.S.; Nakai, K.; Inomata, R.; Kaneda, Y.; Matsumura, A.; Nakamura, H. Dodecaborate lipid liposomes as new vehicles for boron delivery system of neutron capture therapy. Bioorg. Med. Chem. 2010, 18, 3059–3065. [Google Scholar] [CrossRef]
- Andoh, T.; Nakatani, Y.; Suzuki, M.; Sakurai, Y.; Fujimoto, T.; Ichikawa, H. Influence of the particle size of gadolinium-loaded chitosan nanoparticles on their tumor-killing effect in neutron capture therapy in vitro. Appl. Radiat. Isot. 2020, 164, 109270. [Google Scholar] [CrossRef] [PubMed]
- Yew, Y.P.; Shameli, K.; Mohamad, S.E.; Lee, K.X.; Teow, S.Y. Green synthesized montmorillonite/carrageenan/ Fe3O4 nanocomposites for pH-responsive release of protocatechuic acid and its anticancer activity. Int. J. Mol. Sci. 2020, 21, 4851. [Google Scholar] [CrossRef]
- Tietze, R.; Unterweger, H.; Dürr, S.; Lyer, S.; Canella, L.; Kudejova, P.; Wagner, F.M.; Petry, W.; Taccardi, N.; Alexiou, C. Boron containing magnetic nanoparticles for neutron capture therapy—An innovative approach for specifically targeting tumors. Appl. Radiat. Isot. 2015, 106, 151–155. [Google Scholar] [CrossRef]
- Oleshkevich, E.; Morancho, A.; Saha, A.; Galenkamp, K.M.O.; Grayston, A.; Crich, S.G.; Alberti, D.; Protti, N.; Comella, J.X.; Teixidor, F.; et al. Combining magnetic nanoparticles and icosahedral boron clusters in biocompatible inorganic nanohybrids for cancer therapy. Nanomed. Nanotechnol. Biol. Med. 2019, 20, 101986. [Google Scholar] [CrossRef]
- Salem, M.M.; Panina, L.V.; Trukhanova, E.L.; Darwish, M.A.; Morchenko, A.T.; Zubar, T.I.; Trukhanov, S.V.; Trukhanov, A.V. Structural, electric and magnetic properties of (BaFe11.9Al0.1O19)1−x-(BaTiO3)x composites. Compos. Part B Eng. 2019, 174, 107054. [Google Scholar] [CrossRef]
- Ketsko, V.A.; Beresnev, E.N.; Kop’eva, M.A.; Elesina, L.V.; Baranchikov, A.I.; Stognii, A.I.; Trukhanov, A.V.; Kuznetsov, N.T. Specifics of pyrohydrolytic and solid-phase syntheses of solid solutions in the (MgGa2O4)x(MgFe2O4)1−x system. Russ. J. Inorg. Chem. 2010, 55, 427–429. [Google Scholar] [CrossRef]
- Stefanescu, M.; Stoia, M.; Dippong, T.; Stefanescu, O.; Barvinschi, P. Preparation of CoXFe3-XO4 oxydic system starting from metal nitrates and propanediol. Acta Chim. Slov. 2009, 56, 379–385. [Google Scholar]
- Stefanescu, M.; Stoia, M.; Caizer, C.; Dippong, T.; Barvinschi, P. Preparation of CoxFe3-xO4 nanoparticles by thermal decomposition of some organo-metallic precursors. J. Therm. Anal. Calorim. 2009, 97, 245–250. [Google Scholar] [CrossRef]
- Dippong, T.; Cadar, O.; Deac, I.G.; Lazar, M.; Borodi, G.; Levei, E.A. Influence of ferrite to silica ratio and thermal treatment on porosity, surface, microstructure and magnetic properties of Zn0.5Ni0.5Fe2O4/SiO2 nanocomposites. J. Alloys Compd. 2020, 828, 154409. [Google Scholar] [CrossRef]
- Zhu, Y.; Lin, Y.; Zhu, Y.Z.; Lu, J.; Maguire, J.A.; Hosmane, N.S. Boron Drug Delivery via Encapsulated Magnetic Nanocomposites: A New Approach for BNCT in Cancer Treatment. J. Nanomater. 2010, 2010, 409320. [Google Scholar] [CrossRef] [Green Version]
- Korolkov, I.V.; Ludzik, K.; Kozlovskiy, A.L.; Fadeev, M.S.; Shumskaya, A.E.; Gorin, Y.G.; Jazdzewska, M.; Anisovich, M.; Rusakov, V.S.; Zdorovets, M.V. Immobilization of carboranes on Fe3O4-polymer nanocomposites for potential application in boron neutron cancer therapy. Colloids Surf. A Physicochem. Eng. Asp. 2020, 601, 125035. [Google Scholar] [CrossRef]
- Santra, S.; Jativa, S.D.; Kaittanis, C.; Normand, G.; Grimm, J.; Perez, J.M. Gadolinium-encapsulating iron oxide nanoprobe as activatable NMR/MRI contrast agent. ACS Nano 2012, 6, 7281–7294. [Google Scholar] [CrossRef]
- da Silva, W.M.; de Andrade Alves e Silva, R.H.; Cipreste, M.F.; Andrade, G.F.; Gastelois, P.L.; de Almeida Macedo, W.A.; de Sousa, E.M.B. Boron nitride nanotubes radiolabeled with 153Sm and 159Gd: Potential application in nanomedicine. Appl. Radiat. Isot. 2020, 157, 109032. [Google Scholar] [CrossRef]
- Yoshida, F.; Yamamoto, T.; Nakai, K.; Zaboronok, A.; Matsumura, A. Additive effect of BPA and Gd-DTPA for application in accelerator-based neutron source. Appl. Radiat. Isot. 2015, 106, 247–250. [Google Scholar] [CrossRef]
- Yang, K.; Peng, H.; Wen, Y.; Li, N. Applied Surface Science Re-examination of characteristic FTIR spectrum of secondary layer in bilayer oleic acid-coated Fe3O4 nanoparticles. Appl. Surface Sci. 2010, 256, 3093–3097. [Google Scholar] [CrossRef]
- Shahbazi, M.; Bahari, A.; Ghasemi, S. Studying saturation mobility, threshold voltage, and stability of PMMA-SiO2-TMSPM nano-hybrid as OFET gate dielectric. Synth. Met. 2016, 32, 100–108. [Google Scholar] [CrossRef]
- Korolkov, I.V.; Ludzik, K.; Lisovskaya, L.I.; Zibert, A.V.; Yeszhanov, A.B.; Zdorovets, M.V. Modification of magnetic Fe3O4 nanoparticles for targeted delivery of payloads. Bull. Karaganda Univ. “Chem.” Ser. 2021, 101, 99–108. [Google Scholar] [CrossRef]
- Billingham, J.; Breen, C.; Yarwood, J. Adsorption of polyamine, polyacrylic acid and polyethylene glycol on montmorillonite: An in situ study using ATR-FTIR. Vib. Spectrosc. 1997, 14, 19–34. [Google Scholar] [CrossRef]
- Alghool, S.; Zoromba, M.S.; El-halim, H.F.A. Lanthanide amino acid Schiff base complexes: Synthesis, spectroscopic characterization, physical properties and in vitro antimicrobial studies. J. Rare Earths 2013, 31, 715–721. [Google Scholar] [CrossRef]
- Miao, X.; Ho, S.L.; Tegafaw, T.; Cha, H.; Chang, Y.; Oh, I.T.; Yaseen, A.M.; Marasini, S.; Ghazanfari, A.; Yue, H.; et al. Stable and non-Toxic ultrasmall gadolinium oxide nanoparticle colloids (coating material = polyacrylic acid) as high-performance: T1 magnetic resonance imaging contrast agents. RSC Adv. 2018, 8, 3189–3197. [Google Scholar] [CrossRef] [Green Version]
- He, N.; Ni, Y.; Teng, J.; Li, H.; Yao, L.; Zhao, P. Identification of inorganic oxidizing salts in homemade explosives using Fourier transform infrared spectroscopy. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2019, 221, 117164. [Google Scholar] [CrossRef] [PubMed]
- Yue, J.; Li, Y.; Zhao, Y.; Xiang, D.; Dai, Y. Thermal degradation behavior of carborane-containing phenylethynyl terminated imide systems. Polym. Degrad. Stab. 2016, 129, 286–295. [Google Scholar] [CrossRef]
- Eaton, P.; Quaresma, P.; Soares, C.; Neves, C.; de Almeida, M.P.; Pereira, E.; West, P. A direct comparison of experimental methods to measure dimensions of synthetic nanoparticles. Ultramicroscopy 2017, 182, 179–190. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.K.S.; Choi, S.H.; Kang, Y.C.; Lee, J.K. Eco-friendly composite of Fe3O4-reduced graphene oxide particles for efficient enzyme immobilization. ACS Appl. Mater. Interfaces 2017, 9, 2213–2222. [Google Scholar] [CrossRef]
- Kozlovskiy, A.L.; Ermekova, A.E.; Korolkov, I.V.; Chudoba, D.; Jazdzewska, M.; Ludzik, K.; Nazarova, A.; Marciniak, B.; Kontek, R.; Shumskaya, A.E.; et al. Study of phase transformations, structural, corrosion properties and cytotoxicity of magnetite-based nanoparticles. Vacuum 2019, 163, 236–247. [Google Scholar] [CrossRef]
- Fadeev, M.; Kozlovskiy, A.; Korolkov, I.; Egizbek, K.; Nazarova, A.; Chudoba, D.; Rusakov, V.; Zdorovets, M. Iron oxide @ gold nanoparticles: Synthesis, properties and potential use as anode materials for lithium-ion batteries. Colloids Surf. A Physicochem. Eng. Asp. 2020, 603, 125178. [Google Scholar] [CrossRef]
- Bondarenko, L.S.; Kovel, E.S.; Kydralieva, K.A.; Dzhardimalieva, G.I.; Illés, E.; Tombácz, E. Effects of Modified Magnetite Nanoparticles on Bacterial Cells and Enzyme Reactions. Polymers 2020, 10, 1499. [Google Scholar] [CrossRef] [PubMed]
- Bondarenko, L.; Kahru, A.; Terekhova, V.; Dzhardimalieva, G.; Uchanov, P.; Kydralieva, K. Effects of Humic Acids on the Ecotoxicity of Fe3O4 Nanoparticles and Fe-Ions: Impact of Oxidation and Aging. Nanomaterials 2020, 10, 2011. [Google Scholar] [CrossRef] [PubMed]
- Lu, T.; Wang, J.; Yin, J.; Wang, A.; Wang, X.; Zhang, T. Surfactant effects on the microstructures of Fe3O4 nanoparticles synthesized by microemulsion method. Colloids Surf. A Physicochem. Eng. Asp. 2013, 436, 675–683. [Google Scholar] [CrossRef]
- Gangwar, A.; Varghese, S.S.; Sharma, A.; Meena, S.S.; Prajapat, C.L.; Viswanadh, M.K.; Neogi, K.; Muthu, M.S.; Prasad, N.K. Physical and in-vitro evaluation of ϵ-Fe3N@Fe3O4 nanoparticles for bioapplications. Ceram. Int. 2020, 46, 10952–10962. [Google Scholar] [CrossRef]
- Voleník, K.; Seberíni, M.; Neid, J. A Mössbauer and X-ray diffraction study of nonstoichiometry in magnetite. Czechoslov. J. Phys. 1975, 25, 1063–1071. [Google Scholar] [CrossRef]
- Wei, Y.; Han, B.; Hu, X.; Lin, Y.; Wang, X.; Deng, X. Synthesis of Fe3O4 nanoparticles and their magnetic properties. Procedia Eng. 2012, 27, 632–637. [Google Scholar] [CrossRef] [Green Version]
- Huan, W.; Cheng, C.; Yang, Y.; Yuan, H.; Li, Y. A study on the magnetic and photoluminescence properties of Eun+ and Sm3+ doped Fe3O4 nanoparticles. J. Nanosci. Nanotechnol. 2012, 12, 4621–4634. [Google Scholar] [CrossRef]
- Vlazan, P.; Stoia, M. Structural and magnetic properties of CoFe2O4 nanopowders, prepared using a modified Pechini method. Ceram. Int. 2018, 44, 530–536. [Google Scholar] [CrossRef]
- Kharat, S.P.; Darvade, T.C.; Gaikwad, S.K.; Baraskar, B.G.; Kakade, S.G.; Kambale, R.C.; Kolekar, Y.D. Magnetic properties of CoFe2O4 and ZnFe2O4 nanoparticles synthesized by novel chemical route. AIP Conf. Proc. 2016, 1731, 130056. [Google Scholar] [CrossRef]
- Staruch, M.; Hires, D.; Violette, D.; Navarathne, D.; Sotzing, G.A.; Jain, M. Structural and Magnetic Properties of of CoFe2O4 and Co0.5Zn0.5Fe2O4 Nanoparticles for the Magnetoelectric Composite Films. Integr. Ferroelectr. 2011, 131, 102–109. [Google Scholar] [CrossRef]
- Ramanujan, R.V. Magnetic particles for biomedical application. In Biomedical Materials, 1st ed.; Narayan, R., Ed.; Springer Science+Business Media: New York, NY, USA, 2009; Volume 17, pp. 477–491. [Google Scholar]
- Omelyanchik, A.; da Silva, F.G.; Gomide, G.; Kozenkov, I.; Depeyrot, J.; Aquino, R.; Campos, A.F.C.; Fiorani, D.; Peddis, D.; Rodionova, V.; et al. Effect of citric acid on the morpho-structural and magnetic properties of ultrasmall iron oxide nanoparticles. J. Alloys Compd. 2021, 883, 160779. [Google Scholar] [CrossRef]
- van de Loosdrecht, M.M.; Draack, S.; Waanders, S.; Schlief, J.G.L.; Krooshoop, H.J.G.; Viereck, T.; Ludwig, F.; ten Haken, B. A novel characterization technique for superparamagnetic iron oxide nanoparticles: The superparamagnetic quantifier, compared with magnetic particle spectroscopy. Rev. Sci. Instrum. 2019, 90, 024101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dallet, L.; Stanicki, D.; Voisin, P.; Miraux, S.; Ribot, E.J. Micron-sized iron oxide particles for both MRI cell tracking and magnetic fluid hyperthermia treatment. Sci. Rep. 2021, 11, 3286. [Google Scholar] [CrossRef]
- Wu, K.; Su, D.; Liu, J.; Saha, R.; Wang, J.-P. Magnetic nanoparticles in nanomedicine: A review of recent advances. Nanotechnology 2019, 30, 502003. [Google Scholar] [CrossRef] [Green Version]
- Myrovali, E.; Maniotis, N.; Makridis, A.; Terzopoulou, A.; Ntomprougkidis, V.; Simeonidis, K.; Sakellari, D.; Kalogirou, O.; Samaras, T.; Salikhov, R.; et al. Arrangement at the nanoscale: Effect on magnetic particle hyperthermia. Sci. Rep. 2016, 6, 37934. [Google Scholar] [CrossRef] [PubMed]
- Mansell, R.; Vemulkar, T.; Petit, D.C.M.C.; Cheng, Y.; Murphy, J.; Lesniak, M.S.; Cowburn, R.P. Magnetic particles with perpendicular anisotropy for mechanical cancer cell destruction. Sci. Rep. 2017, 7, 4257. [Google Scholar] [CrossRef]
- Moghimi, H.; Zohdiaghdam, R.; Riahialam, N.; Behrouzkia, Z. The assessment of toxicity characteristics of cellular uptake of paramagnetic nanoparticles as a new magnetic resonance imaging contrast agent. Iran. J. Pharm. Res. 2019, 18, 2083–2092. [Google Scholar] [CrossRef]
- Wörle-Knirsch, J.M.; Pulskamp, K.; Krug, H.F. Oops they did it again! Carbon nanotubes hoax scientists in viability assays. Nano Lett. 2006, 6, 1261–1268. [Google Scholar] [CrossRef]
- Dong, X.; Wu, Z.; Li, X.; Xiao, L.; Yang, M.; Li, Y.; Duan, J.; Sun, Z. The Size-dependent Cytotoxicity of Amorphous Silica Nanoparticles: A Systematic Review of in vitro Studies. Int. J. Nanomed. 2020, 15, 9089–9113. [Google Scholar] [CrossRef] [PubMed]
- Shundo, C.; Zhang, H.; Nakanishi, T.; Osaka, T. Cytotoxicity evaluation of magnetite (Fe3O4) nanoparticles in mouse embryonic stem cells. Colloids Surf. B Biointerfaces 2012, 97, 221–225. [Google Scholar] [CrossRef] [PubMed]
- Alberti, D.; Michelotti, A.; Lanfranco, A.; Protti, N.; Altieri, S.; Deagostino, A.; Crich, S.G. In vitro and in vivo BNCT investigations using a carborane containing sulfonamide targeting CAIX epitopes on malignant pleural mesothelioma and breast cancer cells. Sci. Rep. 2020, 10, 19274. [Google Scholar] [CrossRef]
- Wang, W.; Lin, J.; Xing, C.; Chai, R.; Abbas, S.; Song, T.; Tang, C.; Huang, Y. Fe3O4 nanoparticle-coated boron nitride nanospheres: Synthesis, magnetic property and biocompatibility study. Ceram. Int. 2017, 43, 6371–6376. [Google Scholar] [CrossRef]
- Zheng, S.; Jin, S.; Jiao, M.; Wang, W.; Zhou, X.; Xu, J.; Wang, Y.; Dou, P.; Jin, Z.; Wu, C.; et al. Tumor-targeted Gd-doped mesoporous Fe3O4 nanoparticles for T1/T2 MR imaging guided synergistic cancer therapy. Drug Deliv. 2021, 28, 787–799. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Xiang, H.H.; Zhao, Z.Z.; Wu, Y.K.; Fei, M.Y.; Song, M.M. An ultra-sensitive T2-weighted MR contrast agent based on Gd3+ ion chelated Fe3O4 nanoparticles. RSC Adv. 2020, 10, 18054–18061. [Google Scholar] [CrossRef]
- Drake, P.; Cho, H.J.; Shih, P.S.; Kao, C.H.; Lee, K.F.; Kuo, C.H.; Lin, X.Z.; Lin, Y.J. Gd-doped iron-oxide nanoparticles for tumour therapy via magnetic field hyperthermia. J. Mater. Chem. 2007, 17, 4914–4918. [Google Scholar] [CrossRef]
- Sena, N.C.; Castro, T.J.; Garg, V.K.; Oliveira, A.C.; Morais, P.C.; da Silva, S.W. Gadolinium ferrite nanoparticles: Synthesis and morphological, structural and magnetic properties. Ceram. Int. 2017, 43, 4042–4047. [Google Scholar] [CrossRef]
- Zhang, X.; Hao, L.; Wang, H.; Zhu, X.; Zhang, Z.; Hu, X.; Jiang, W. Preparation and characterization of superparamagnetic Fe3O4/CNTs nanocomposites dual-drug carrier. J. Wuhan Univ. Technol. Mater. Sci. Ed. 2017, 32, 42–46. [Google Scholar] [CrossRef]
- Hennig, A.; Borcherding, H.; Jaeger, C.; Hatami, S.; Würth, C.; Hoffmann, A.; Hoffmann, K.; Thiele, T.; Schedler, U.; Resch-Genger, U. Scope and limitations of surface functional group quantification methods: Exploratory study with poly(acrylic acid)-grafted micro- and nanoparticles. J. Am. Chem. Soc. 2012, 134, 8268–8276. [Google Scholar] [CrossRef]
- Uchida, E.; Uyama, Y.; Ikada, Y. Sorption of Low-Molecular-Weight Anions into Thin Polycation Layers Grafted onto a Film. Langmuir 1993, 9, 1121–1124. [Google Scholar] [CrossRef]
- Stockert, J.C.; Horobin, R.W.; Colombo, L.L.; Blázquez-Castro, A. Tetrazolium salts and formazan products in Cell Biology: Viability assessment, fluorescence imaging, and labeling perspectives. Acta Histochem. 2018, 120, 159–167. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Otari, S.V.; Kumar, M.; Anwar, M.Z.; Thorat, N.D.; Patel, S.K.; Lee, D.; Lee, J.H.; Lee, J.K.; Kang, Y.C.; Zhang, L. Rapid synthesis and decoration of reduced graphene oxide with gold nanoparticles by thermostable peptides for memory device and photothermal applications. Sci. Rep. 2017, 7, 10980. [Google Scholar] [CrossRef] [PubMed]

| Concentration of Initiator, % | Concentration of Monomer, % | Time of Reaction, h | |||||||
|---|---|---|---|---|---|---|---|---|---|
| 0.0125 | 0.03125 | 0.05 | 10 | 20 | 30 | 2 | 6 | 24 | |
| IC=O/IFe-O-Fe | 0.15 | 0.184 | 0.181 | 0.184 | 0.181 | 0.103 | 0.162 | 0.181 | 0.184 |
| Sample | Lattice Parameter, Å | Crystalline Size, nm | Degree of Structural Ordering, % | Stoichiometry |
|---|---|---|---|---|
| Fe3O4 | a = 8.29402 | 10.5 | 53.6 | Fe2.88O4 |
| Fe3O4-TMSPM | a = 8.31243 | 14.3 | 61.2 | Fe2.85O4 |
| Fe3O4-TMSPM-PAA | a = 8.29124 | 12.4 | 69.3 | Fe2.77O4 |
| Fe3O4-TMSPM-PAA/PALAm | a = 8.33838 | 18.7 | 63.1 | Fe2.76O4 |
| Fe3O4-TMSPM-PAA/PALAm-Gd | a = 8.33172 | 13.2 | 66.3 | Fe2.79O4 |
| Fe3O4-TMSPM-PAA/PALAm-Gd-PAA | a = 8.31174 | 11.4 | 66.1 | Fe2.79O4 |
| Fe3O4-TMSPM-PAA/PALAm-Gd-PAA/PALAm | a = 8.29846 | 12.6 | 66.2 | Fe2.69O4 |
| Fe3O4-TMSPM-PAA/PALAm-Gd-PAA/PALAm—Carborane | a = 8.30508 | 16.5 | 61.7 | Fe2.71O4 |
| Sample | A-Sublattice Hn, kOe | B-Sublattice Hn, kOe | Intensity of Doublet, % | IB-sublattice/IA-sublattice | δ (Fe3-δ)O4 | χ2 |
|---|---|---|---|---|---|---|
| Fe3O4 | 475.34 ± 3.24 | 425.84 ± 4.53 | 0.7 | 74.9/24.4 | 0.327 ± 0.031 | 0.94 ± 0.03 |
| Fe3O4-TMSPM | 479.57 ± 2.32 | 427.07 ± 3.43 | 0.6 | 75.8/23.6 | 0.331 ± 0.025 | 1.09 ± 0.05 |
| Fe3O4-TMSPM-PAA | 469.42 ± 3.25 | 422.65 ± 4.31 | 1.2 | 84.5/14.3 | 0.330 ± 0.027 | 1.10 ± 0.06 |
| Fe3O4-TMSPM-PAA/PALAm | 465.75 ± 4.21 | 440.54 ± 3.64 | 2.5 | 87.4/10.1 | 0.332 ± 0.025 | 1.08 ± 0.04 |
| Fe3O4-TMSPM-PAA/PALAm-Gd | 465.32 ± 4.14 | 435.17 ± 5.42 | 1.6 | 86.5/11.9 | 0.322 ± 0.016 | 1.04 ± 0.10 |
| Fe3O4-TMSPM-PAA/PALAm-Gd -PAA | 459.65 ± 3.52 | 437.13 ± 2.31 | 1.1 | 87.2/11.7 | 0.304 ± 0.014 | 1.01 ± 0.09 |
| Fe3O4-TMSPM-PAA/PALAm-Gd- PAA/PALAm | 469.64 ± 2.45 | 435.65 ± 2.56 | 0.9 | 87.7/11.4 | 0.323 ± 0.021 | 1.05 ± 0.07 |
| Fe3O4-TMSPM-PAA/PALAm-Gd- PAA/PALAm—Carborane | 468.42 ± 2.63 | 437.39 ± 2.35 | 0.7 | 86.2/13.1 | 0.317 ± 0.027 | 1.21 ± 0.04 |
| Cell Line | IC50 Mean Values and 95%-Confidence Intervals (Parentheses) μg/mL |
|---|---|
| L 929 | 83.56 (72.36–96.42) |
| PC-3 | 80.63 (69.59–93.06) |
| BxPC3 | 22.23 (18.11–26.85) |
| MCF-7 | 55.32 (47.94–63.84) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Korolkov, I.V.; Zibert, A.V.; Lissovskaya, L.I.; Ludzik, K.; Anisovich, M.; Kozlovskiy, A.L.; Shumskaya, A.E.; Vasilyeva, M.; Shlimas, D.I.; Jażdżewska, M.; et al. Boron and Gadolinium Loaded Fe3O4 Nanocarriers for Potential Application in Neutron Capture Therapy. Int. J. Mol. Sci. 2021, 22, 8687. https://doi.org/10.3390/ijms22168687
Korolkov IV, Zibert AV, Lissovskaya LI, Ludzik K, Anisovich M, Kozlovskiy AL, Shumskaya AE, Vasilyeva M, Shlimas DI, Jażdżewska M, et al. Boron and Gadolinium Loaded Fe3O4 Nanocarriers for Potential Application in Neutron Capture Therapy. International Journal of Molecular Sciences. 2021; 22(16):8687. https://doi.org/10.3390/ijms22168687
Chicago/Turabian StyleKorolkov, Ilya V., Alexandr V. Zibert, Lana I. Lissovskaya, K. Ludzik, M. Anisovich, Artem L. Kozlovskiy, A. E. Shumskaya, M. Vasilyeva, Dmitriy I. Shlimas, Monika Jażdżewska, and et al. 2021. "Boron and Gadolinium Loaded Fe3O4 Nanocarriers for Potential Application in Neutron Capture Therapy" International Journal of Molecular Sciences 22, no. 16: 8687. https://doi.org/10.3390/ijms22168687
APA StyleKorolkov, I. V., Zibert, A. V., Lissovskaya, L. I., Ludzik, K., Anisovich, M., Kozlovskiy, A. L., Shumskaya, A. E., Vasilyeva, M., Shlimas, D. I., Jażdżewska, M., Marciniak, B., Kontek, R., Chudoba, D., & Zdorovets, M. V. (2021). Boron and Gadolinium Loaded Fe3O4 Nanocarriers for Potential Application in Neutron Capture Therapy. International Journal of Molecular Sciences, 22(16), 8687. https://doi.org/10.3390/ijms22168687








