Boosted Pro-Inflammatory Activity in Human PBMCs by Lipopolysaccharide and SARS-CoV-2 Spike Protein Is Regulated by α-1 Antitrypsin †
Abstract
1. Introduction
2. Results
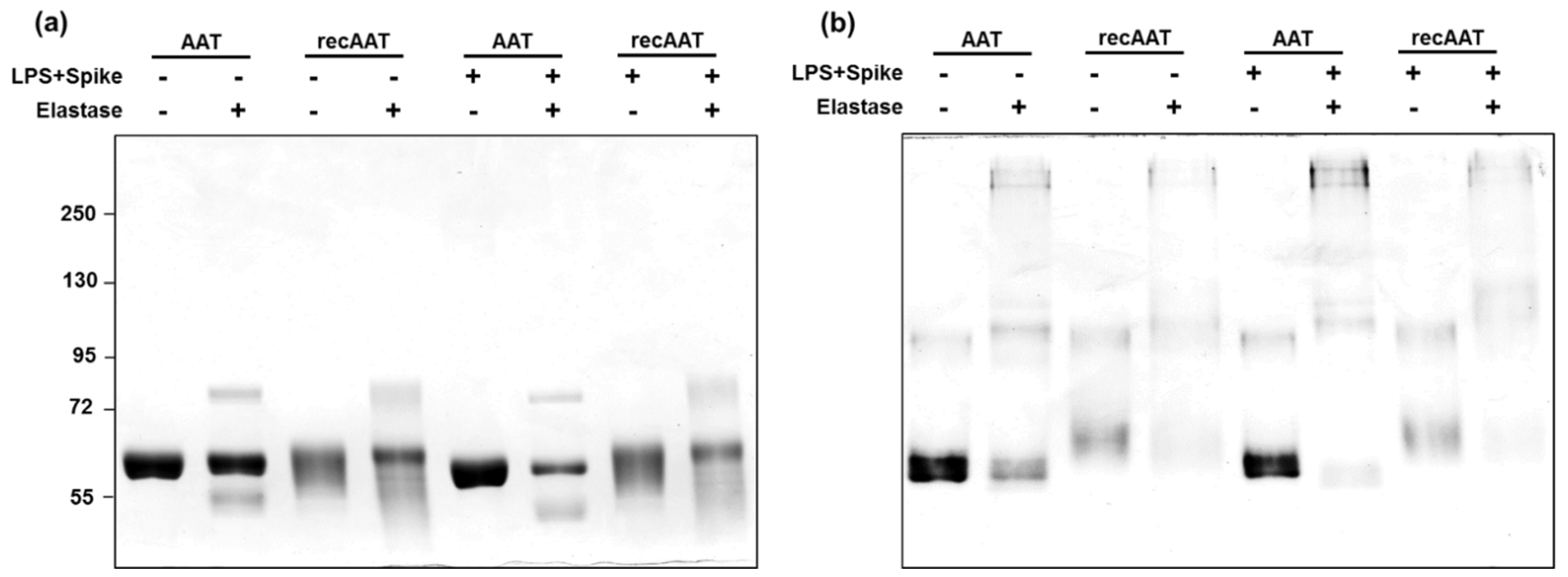
2.1. Characterization of AAT Proteins
2.2. Effects of AAT Proteins on Cytokine/Chemokine Expression
2.3. Effects of AAT Proteins on Cytokine/Chemokine Release after 6 Hours of PBMCs Culture
3. Discussion
4. Materials and Methods
4.1. Protein Preparations
4.1.1. Human Plasma Isolated AAT
4.1.2. Human Plasma Oxidized AAT
4.1.3. Recombinant AAT
4.1.4. Spike Protein
4.2. Characterization of AAT Proteins by Elastase Complex Formation
4.3. Isolation and Culture of Human PBMCs
4.4. Real-Time Polymerase Chain Reaction (RT-PCR) Analysis
4.5. Western Blot Analysis
4.6. ELISA Assays
4.7. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Sethi, A.; Bach, H. Evaluation of Current Therapies for COVID-19 Treatment. Microorganisms 2020, 8, 1097. [Google Scholar] [CrossRef]
- McElvaney, O.J.; McEvoy, N.L.; McElvaney, O.F.; Carroll, T.P.; Murphy, M.P.; Dunlea, D.M.; Ni Choileain, O.; Clarke, J.; O’Connor, E.; Hogan, G.; et al. Characterization of the Inflammatory Response to Severe COVID-19 Illness. Am. J. Respir. Crit. Care Med. 2020, 202, 812–821. [Google Scholar] [CrossRef]
- Letko, M.; Marzi, A.; Munster, V. Functional assessment of cell entry and receptor usage for SARS-CoV-2 and other lineage B betacoronaviruses. Nat. Microbiol. 2020, 5, 562–569. [Google Scholar] [CrossRef]
- Correa Giron, C.; Laaksonen, A.; Barroso da Silva, F.L. On the interactions of the receptor-binding domain of SARS-CoV-1 and SARS-CoV-2 spike proteins with monoclonal antibodies and the receptor ACE2. Virus Res. 2020, 285, 198021. [Google Scholar] [CrossRef]
- Walls, A.C.; Park, Y.J.; Tortorici, M.A.; Wall, A.; McGuire, A.T.; Veesler, D. Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein. Cell 2020, 181, 281–292 e286. [Google Scholar] [CrossRef]
- Bittmann, S.; Weissenstein, A.; Villalon, G.; Moschuring-Alieva, E.; Luchter, E. Simultaneous Treatment of COVID-19 With Serine Protease Inhibitor Camostat and/or Cathepsin L Inhibitor? J. Clin. Med. Res. 2020, 12, 320–322. [Google Scholar] [CrossRef]
- Gooz, M. ADAM-17: The enzyme that does it all. Crit. Rev. Biochem. Mol. Biol. 2010, 45, 146–169. [Google Scholar] [CrossRef] [PubMed]
- Palau, V.; Riera, M.; Soler, M.J. ADAM17 inhibition may exert a protective effect on COVID-19. Nephrol. Dial. Transplant. 2020, 35, 1071–1072. [Google Scholar] [CrossRef] [PubMed]
- Paoloni-Giacobino, A.; Chen, H.; Peitsch, M.C.; Rossier, C.; Antonarakis, S.E. Cloning of the TMPRSS2 gene, which encodes a novel serine protease with transmembrane, LDLRA, and SRCR domains and maps to 21q22.3. Genomics 1997, 44, 309–320. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Hippensteel, J.; Leavitt, A.; Maloney, J.P.; Beckham, D.; Garcia, C.; Li, Q.; Freed, B.M.; Ordway, D.; Sandhaus, R.A.; et al. Hypothesis: Alpha-1-antitrypsin is a promising treatment option for COVID-19. Med. Hypotheses 2021, 146, 110394. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Kruger, N.; Herrler, T.; Erichsen, S.; Schiergens, T.S.; Herrler, G.; Wu, N.H.; Nitsche, A.; et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 2020, 181, 271–280 e278. [Google Scholar] [CrossRef]
- Janciauskiene, S.M.; Bals, R.; Koczulla, R.; Vogelmeier, C.; Kohnlein, T.; Welte, T. The discovery of alpha1-antitrypsin and its role in health and disease. Respir. Med. 2011, 105, 1129–1139. [Google Scholar] [CrossRef] [PubMed]
- Janciauskiene, S.; Larsson, S.; Larsson, P.; Virtala, R.; Jansson, L.; Stevens, T. Inhibition of lipopolysaccharide-mediated human monocyte activation, in vitro, by alpha1-antitrypsin. Biochem. Biophys. Res. Commun. 2004, 321, 592–600. [Google Scholar] [CrossRef]
- Bergin, D.A.; Reeves, E.P.; Meleady, P.; Henry, M.; McElvaney, O.J.; Carroll, T.P.; Condron, C.; Chotirmall, S.H.; Clynes, M.; O’Neill, S.J.; et al. alpha-1 Antitrypsin regulates human neutrophil chemotaxis induced by soluble immune complexes and IL-8. J. Clin. Investig. 2010, 120, 4236–4250. [Google Scholar] [CrossRef] [PubMed]
- Janciauskiene, S.; Tumpara, S.; Wiese, M.; Wrenger, S.; Vijayan, V.; Gueler, F.; Chen, R.; Madyaningrana, K.; Mahadeva, R.; Welte, T.; et al. Alpha1-antitrypsin binds hemin and prevents oxidative activation of human neutrophils: Putative pathophysiological significance. J. Leukoc. Biol. 2017, 102, 1127–1141. [Google Scholar] [CrossRef]
- Siebers, K.; Fink, B.; Zakrzewicz, A.; Agne, A.; Richter, K.; Konzok, S.; Hecker, A.; Zukunft, S.; Kullmar, M.; Klein, J.; et al. Alpha-1 Antitrypsin Inhibits ATP-Mediated Release of Interleukin-1beta via CD36 and Nicotinic Acetylcholine Receptors. Front. Immunol. 2018, 9, 877. [Google Scholar] [CrossRef]
- Cantin, A.M.; Woods, D.E. Aerosolized prolastin suppresses bacterial proliferation in a model of chronic Pseudomonas aeruginosa lung infection. Am. J. Respir. Crit. Care Med. 1999, 160, 1130–1135. [Google Scholar] [CrossRef] [PubMed]
- Libert, C.; Van Molle, W.; Brouckaert, P.; Fiers, W. alpha1-Antitrypsin inhibits the lethal response to TNF in mice. J. Immunol. 1996, 157, 5126–5129. [Google Scholar]
- Marcondes, A.M.; Karoopongse, E.; Lesnikova, M.; Margineantu, D.; Welte, T.; Dinarello, C.A.; Hockenbery, D.; Janciauskiene, S.; Deeg, H.J. alpha-1-Antitrypsin (AAT)-modified donor cells suppress GVHD but enhance the GVL effect: A role for mitochondrial bioenergetics. Blood 2014, 124, 2881–2891. [Google Scholar] [CrossRef] [PubMed]
- Gotzfried, J.; Smirnova, N.F.; Morrone, C.; Korkmaz, B.; Yildirim, A.O.; Eickelberg, O.; Jenne, D.E. Preservation with alpha1-antitrypsin improves primary graft function of murine lung transplants. J. Heart Lung Transplant. 2018, 37, 1021–1028. [Google Scholar] [CrossRef]
- Lior, Y.; Jasevitch, M.; Ochayon, D.E.; Zaretsky, M.; Lewis, E.C.; Aharoni, A. Application of directed evolution and back-to-consensus algorithms to human alpha1-antitrypsin leads to diminished anti-protease activity and augmented anti-inflammatory activities. Cell Immunol. 2020, 355, 104135. [Google Scholar] [CrossRef]
- Petruk, G.; Puthia, M.; Petrlova, J.; Samsudin, F.; Stromdahl, A.C.; Cerps, S.; Uller, L.; Kjellstrom, S.; Bond, P.J.; Schmidtchen, A.A. SARS-CoV-2 spike protein binds to bacterial lipopolysaccharide and boosts proinflammatory activity. J. Mol. Cell Biol. 2021, 12, 916–932. [Google Scholar] [CrossRef]
- Pino, P.; Kint, J.; Kiseljak, D.; Agnolon, V.; Corradin, G.; Kajava, A.V.; Rovero, P.; Dijkman, R.; den Hartog, G.; McLellan, J.S.; et al. Trimeric SARS-CoV-2 Spike Proteins Produced from CHO Cells in Bioreactors Are High-Quality Antigens. Processes 2020, 8, 1539. [Google Scholar] [CrossRef]
- Tumpara, S.; Martinez-Delgado, B.; Gomez-Mariano, G.; Liu, B.; DeLuca, D.S.; Korenbaum, E.; Jonigk, D.; Jugert, F.; Wurm, F.M.; Wurm, M.J.; et al. The Delivery of alpha1-Antitrypsin Therapy Through Transepidermal Route: Worthwhile to Explore. Front. Pharmacol. 2020, 11, 983. [Google Scholar] [CrossRef]
- Liu, J.; Li, S.; Liu, J.; Liang, B.; Wang, X.; Wang, H.; Li, W.; Tong, Q.; Yi, J.; Zhao, L.; et al. Longitudinal characteristics of lymphocyte responses and cytokine profiles in the peripheral blood of SARS-CoV-2 infected patients. EBioMedicine 2020, 55, 102763. [Google Scholar] [CrossRef] [PubMed]
- Del Valle, D.M.; Kim-Schulze, S.; Huang, H.H.; Beckmann, N.D.; Nirenberg, S.; Wang, B.; Lavin, Y.; Swartz, T.H.; Madduri, D.; Stock, A.; et al. An inflammatory cytokine signature predicts COVID-19 severity and survival. Nat. Med. 2020, 26, 1636–1643. [Google Scholar] [CrossRef]
- Xu, X.; Han, M.; Li, T.; Sun, W.; Wang, D.; Fu, B.; Zhou, Y.; Zheng, X.; Yang, Y.; Li, X.; et al. Effective treatment of severe COVID-19 patients with tocilizumab. Proc. Natl. Acad. Sci. USA 2020, 117, 10970–10975. [Google Scholar] [CrossRef] [PubMed]
- Ostermann, L.; Maus, R.; Stolper, J.; Schutte, L.; Katsarou, K.; Tumpara, S.; Pich, A.; Mueller, C.; Janciauskiene, S.; Welte, T.; et al. Alpha-1 antitrypsin deficiency impairs lung antibacterial immunity in mice. JCI Insight 2021, 6. [Google Scholar] [CrossRef] [PubMed]
- Guttman, O.; Freixo-Lima, G.S.; Kaner, Z.; Lior, Y.; Rider, P.; Lewis, E.C. Context-Specific and Immune Cell-Dependent Antitumor Activities of alpha1-Antitrypsin. Front. Immunol. 2016, 7, 559. [Google Scholar] [CrossRef] [PubMed]
- Sirivongrangson, P.; Kulvichit, W.; Payungporn, S.; Pisitkun, T.; Chindamporn, A.; Peerapornratana, S.; Pisitkun, P.; Chitcharoen, S.; Sawaswong, V.; Worasilchai, N.; et al. Endotoxemia and circulating bacteriome in severe COVID-19 patients. Intensive Care Med. Exp. 2020, 8, 72. [Google Scholar] [CrossRef] [PubMed]
- Kruglikov, I.L.; Scherer, P.E. Preexisting and inducible endotoxemia as crucial contributors to the severity of COVID-19 outcomes. PLoS Pathog. 2021, 17, e1009306. [Google Scholar] [CrossRef]
- Brenchley, J.M.; Price, D.A.; Schacker, T.W.; Asher, T.E.; Silvestri, G.; Rao, S.; Kazzaz, Z.; Bornstein, E.; Lambotte, O.; Altmann, D.; et al. Microbial translocation is a cause of systemic immune activation in chronic HIV infection. Nat. Med. 2006, 12, 1365–1371. [Google Scholar] [CrossRef]
- van Reeth, K.; Nauwynck, H.; Pensaert, M. A potential role for tumour necrosis factor-alpha in synergy between porcine respiratory coronavirus and bacterial lipopolysaccharide in the induction of respiratory disease in pigs. J. Med. Microbiol. 2000, 49, 613–620. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, P.A.; Fraser, J.D.; Pridmore, A.C.; Russell, H.H.; Read, R.C.; Sriskandan, S. Superantigen recognition by HLA class II on monocytes up-regulates toll-like receptor 4 and enhances proinflammatory responses to endotoxin. Blood 2005, 105, 3655–3662. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.; Bhardwaj, N. Super(antigen) target for SARS-CoV-2. Nat. Rev. Immunol. 2021, 21, 72. [Google Scholar] [CrossRef] [PubMed]
- Cheng, M.H.; Porritt, R.A.; Rivas, M.N.; Krieger, J.M.; Ozdemir, A.B.; Garcia, G., Jr.; Arumugaswami, V.; Fries, B.C.; Arditi, M.; Bahar, I. A monoclonal antibody against staphylococcal enterotoxin B superantigen inhibits SARS-CoV-2 entry in vitro. bioRxiv 2020. [Google Scholar] [CrossRef]
- Choudhury, A.; Mukherjee, S. In silico studies on the comparative characterization of the interactions of SARS-CoV-2 spike glycoprotein with ACE-2 receptor homologs and human TLRs. J. Med. Virol. 2020, 92, 2105–2113. [Google Scholar] [CrossRef]
- Bhattacharya, M.; Sharma, A.R.; Mallick, B.; Sharma, G.; Lee, S.S.; Chakraborty, C. Immunoinformatics approach to understand molecular interaction between multi-epitopic regions of SARS-CoV-2 spike-protein with TLR4/MD-2 complex. Infect. Genet. Evol. 2020, 85, 104587. [Google Scholar] [CrossRef]
- Subramaniyam, D.; Steele, C.; Kohnlein, T.; Welte, T.; Grip, O.; Matalon, S.; Janciauskiene, S. Effects of alpha 1-antitrypsin on endotoxin-induced lung inflammation in vivo. Inflamm. Res. 2010, 59, 571–578. [Google Scholar] [CrossRef]
- Aldonyte, R.; Jansson, L.; Janciauskiene, S. Concentration-dependent effects of native and polymerised alpha1-antitrypsin on primary human monocytes, in vitro. BMC Cell Biol. 2004, 5, 11. [Google Scholar] [CrossRef]
- Sohrab, S.; Petrusca, D.N.; Lockett, A.D.; Schweitzer, K.S.; Rush, N.I.; Gu, Y.; Kamocki, K.; Garrison, J.; Petrache, I. Mechanism of alpha-1 antitrypsin endocytosis by lung endothelium. FASEB J. 2009, 23, 3149–3158. [Google Scholar] [CrossRef]
- Serban, K.A.; Petrache, I. Alpha-1 Antitrypsin and Lung Cell Apoptosis. Ann. Am. Thorac. Soc. 2016, 13 (Suppl. 2), S146–S149. [Google Scholar] [CrossRef]
- Tumpara, S.; Ballmaier, M.; Wrenger, S.; Konig, M.; Lehmann, M.; Lichtinghagen, R.; Martinez-Delgado, B.; Korenbaum, E.; DeLuca, D.; Jedicke, N.; et al. Polymerization of misfolded Z alpha-1 antitrypsin protein lowers CX3CR1 expression in human PBMCs. Elife 2021, 10. [Google Scholar] [CrossRef]
- Subramaniyam, D.; Zhou, H.; Liang, M.; Welte, T.; Mahadeva, R.; Janciauskiene, S. Cholesterol rich lipid raft microdomains are gateway for acute phase protein, SERPINA1. Int. J. Biochem. Cell Biol. 2010, 42, 1562–1570. [Google Scholar] [CrossRef] [PubMed]
- Lechowicz, U.; Rudzinski, S.; Jezela-Stanek, A.; Janciauskiene, S.; Chorostowska-Wynimko, J. Post-Translational Modifications of Circulating Alpha-1-Antitrypsin Protein. Int. J. Mol. Sci. 2020, 21, 9187. [Google Scholar] [CrossRef]
- Shental-Bechor, D.; Levy, Y. Folding of glycoproteins: Toward understanding the biophysics of the glycosylation code. Curr. Opin. Struct. Biol. 2009, 19, 524–533. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, C.; Dunlea, D.M.; Saldova, R.; Henry, M.; Meleady, P.; McElvaney, O.J.; Marsh, B.; Rudd, P.M.; Reeves, E.P.; McElvaney, N.G. Glycosylation Repurposes Alpha-1 Antitrypsin for Resolution of Community-acquired Pneumonia. Am. J. Respir. Crit. Care Med. 2018, 197, 1346–1349. [Google Scholar] [CrossRef]
- Baker, A.N.; Richards, S.J.; Guy, C.S.; Congdon, T.R.; Hasan, M.; Zwetsloot, A.J.; Gallo, A.; Lewandowski, J.R.; Stansfeld, P.J.; Straube, A.; et al. The SARS-CoV-2 Spike Protein Binds Sialic Acids and Enables Rapid Detection in a Lateral Flow Point of Care Diagnostic Device. ACS Cent. Sci. 2020, 6, 2046–2052. [Google Scholar] [CrossRef] [PubMed]
- Lewis, N.E.; Liu, X.; Li, Y.; Nagarajan, H.; Yerganian, G.; O’Brien, E.; Bordbar, A.; Roth, A.M.; Rosenbloom, J.; Bian, C.; et al. Genomic landscapes of Chinese hamster ovary cell lines as revealed by the Cricetulus griseus draft genome. Nat. Biotechnol. 2013, 31, 759–765. [Google Scholar] [CrossRef]
- Amann, T.; Hansen, A.H.; Kol, S.; Hansen, H.G.; Arnsdorf, J.; Nallapareddy, S.; Voldborg, B.; Lee, G.M.; Andersen, M.R.; Kildegaard, H.F. Glyco-engineered CHO cell lines producing alpha-1-antitrypsin and C1 esterase inhibitor with fully humanized N-glycosylation profiles. Metab. Eng. 2019, 52, 143–152. [Google Scholar] [CrossRef] [PubMed]
- de Loyola, M.B.; Dos Reis, T.T.A.; de Oliveira, G.; da Fonseca Palmeira, J.; Arganaraz, G.A.; Arganaraz, E.R. Alpha-1-antitrypsin: A possible host protective factor against COVID-19. Rev. Med. Virol. 2021, 31, e2157. [Google Scholar] [CrossRef] [PubMed]
- Hubbard, R.C.; Sellers, S.; Czerski, D.; Stephens, L.; Crystal, R.G. Biochemical efficacy and safety of monthly augmentation therapy for alpha 1-antitrypsin deficiency. JAMA 1988, 260, 1259–1264. [Google Scholar] [CrossRef]
- Ritzmann, F.; Chitirala, P.; Kruger, N.; Hoffmann, M.; Zuo, W.; Lammert, F.; Smola, S.; Tov, N.; Alagem, N.; Lepper, P.M.; et al. Therapeutic Application of alpha-1-antitrypsin in COVID-19. Am. J. Respir. Crit. Care Med. 2021. [Google Scholar] [CrossRef] [PubMed]
- Wrapp, D.; Wang, N.; Corbett, K.S.; Goldsmith, J.A.; Hsieh, C.L.; Abiona, O.; Graham, B.S.; McLellan, J.S. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science 2020, 367, 1260–1263. [Google Scholar] [CrossRef] [PubMed]
- Frenzel, E.; Wrenger, S.; Immenschuh, S.; Koczulla, R.; Mahadeva, R.; Deeg, H.J.; Dinarello, C.A.; Welte, T.; Marcondes, A.M.; Janciauskiene, S. Acute-phase protein alpha1-antitrypsin—A novel regulator of angiopoietin-like protein 4 transcription and secretion. J. Immunol. 2014, 192, 5354–5362. [Google Scholar] [CrossRef] [PubMed]


| Gene Expression (Relative to Housekeeping Gene HPRT1) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Sample, N = 7 donors | TNFA | IL1B | IL6 | CXCL8 | ||||
| Control | 3.1 | (1.2–4.8) | 43.2 | (34.4–55.4) | 0.07 | (0.03–0.10) | 75.6 | (46.2–95.3) |
| LPS | 31.5 | (38.4–59.9) | 559.2 | (452.8–686.0) | 63.0 | (40.8–105.7) | 965.5 | (852.4–1394.4) |
| LPS + AAT | 28.1 | (8.3–63.0) | 511.6 | (490.8–625.3) | 62.8 | (43.7–95.6) | 986.6 | (809.5–1126.6) |
| LPS + oxAAT | 28.6 | (9.4–34.6) | 574.2 | (447.3–733.7) | 47.7 | (40.7–83.4) | 1077.9 | (833.5–1247.1) |
| LPS + recAAT | 14.6 | (8.0–32.7) | 637.3 | (489.4–725.4) | 50.3 | (35.5–68.0) | 1208.9 | (1001.2–1323.7) |
| LPS + Spike | 33.7 | (24.4–163.1) | 853.2 | (729.1–903.9) | 110.4 | (83.8–127.2) | 1540.2 | (1248.2–1983.4) |
| LPS + Spike + AAT | 34.5 | (25.7–109.7) | 751.5 | (639.2–903.6) | 102.6 | (56.5–112.7) | 1735.2 | (1319.5–1841.3) |
| LPS + Spike + oxAAT | 33.3 | (20.7–129.1) | 668.4 | (517.2–868.8) | 104.6 | (49.2–124.5) | 1610.6 | (846.7–2190.2) |
| LPS + Spike + recAAT | 16.8 | (13.4–38.8) * | 472.5 | (320–754.1) ** | 49.7 | (32.3–73.8) | 1183.9 | (1012.7–1597.4) |
| Cytokine/Chemokine Concentration (pg/mL Supernatant) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Sample, N = 7 donors | TNF-α | IL-1β | IL-6 | IL-8 | ||||
| Control | 12.4 | (8.7–34.5) | 4.9 | (1.9–5.9) | 2.3 | (1.53–3.0) | 167.0 | (57.4–456.4) |
| LPS | 1684 | (1359–2762) | 1271 | (675–2204) | 4844 | (3330–7304) | 18,323 | (15,095–27,513) |
| LPS + AAT | 1967 | (1452–3050) | 1582 | (980–2501) | 6944 | (6118–11,008) | 18,691 | (16,899–25,970) |
| LPS + oxAAT | 1926 | (1390–3605) | 1165 | (729–1652) | 9857 | (6744–14,231) | 21,283 | (17,394–32,982) |
| LPS + recAAT | 1555 | (1225–2538) | 1151 | (491–1699) | 6683 | (4976–9377) | 19,365 | (16,595–32,256) |
| LPS + Spike | 2326 | (2078–3373) | 1222 | (697–1791) | 5136 | (3446–9070) | 20,376 | (14,231–27,923) |
| LPS + Spike + AAT | 2103 | (1531–2840) | 1116 | (685–1756) | 5211 | (3177–9524) | 18,984 | (14,155–25,301) |
| LPS + Spike + oxAAT | 2575 | (1736–4020) | 922 | (799–1585) | 8041 | (7184–11,013) | 20,236 | (17,831–32,659) |
| LPS + Spike + recAAT | 1266 | (984–1899) ** | 505 | (252–916) * | 4753 | (2399–8200) | 17,072 | (14,359–21,857) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tumpara, S.; Gründing, A.R.; Sivaraman, K.; Wrenger, S.; Olejnicka, B.; Welte, T.; Wurm, M.J.; Pino, P.; Kiseljak, D.; Wurm, F.M.; et al. Boosted Pro-Inflammatory Activity in Human PBMCs by Lipopolysaccharide and SARS-CoV-2 Spike Protein Is Regulated by α-1 Antitrypsin. Int. J. Mol. Sci. 2021, 22, 7941. https://doi.org/10.3390/ijms22157941
Tumpara S, Gründing AR, Sivaraman K, Wrenger S, Olejnicka B, Welte T, Wurm MJ, Pino P, Kiseljak D, Wurm FM, et al. Boosted Pro-Inflammatory Activity in Human PBMCs by Lipopolysaccharide and SARS-CoV-2 Spike Protein Is Regulated by α-1 Antitrypsin. International Journal of Molecular Sciences. 2021; 22(15):7941. https://doi.org/10.3390/ijms22157941
Chicago/Turabian StyleTumpara, Srinu, Anna R. Gründing, Kokilavani Sivaraman, Sabine Wrenger, Beata Olejnicka, Tobias Welte, Maria J. Wurm, Paco Pino, Divor Kiseljak, Florian M. Wurm, and et al. 2021. "Boosted Pro-Inflammatory Activity in Human PBMCs by Lipopolysaccharide and SARS-CoV-2 Spike Protein Is Regulated by α-1 Antitrypsin" International Journal of Molecular Sciences 22, no. 15: 7941. https://doi.org/10.3390/ijms22157941
APA StyleTumpara, S., Gründing, A. R., Sivaraman, K., Wrenger, S., Olejnicka, B., Welte, T., Wurm, M. J., Pino, P., Kiseljak, D., Wurm, F. M., & Janciauskiene, S. (2021). Boosted Pro-Inflammatory Activity in Human PBMCs by Lipopolysaccharide and SARS-CoV-2 Spike Protein Is Regulated by α-1 Antitrypsin. International Journal of Molecular Sciences, 22(15), 7941. https://doi.org/10.3390/ijms22157941







