Comparative Transcriptome Analysis of Human Adipose-Derived Stem Cells Undergoing Osteogenesis in 2D and 3D Culture Conditions
Abstract
:1. Introduction
2. Results
2.1. hADSCs Spheroid Formation in Microwell
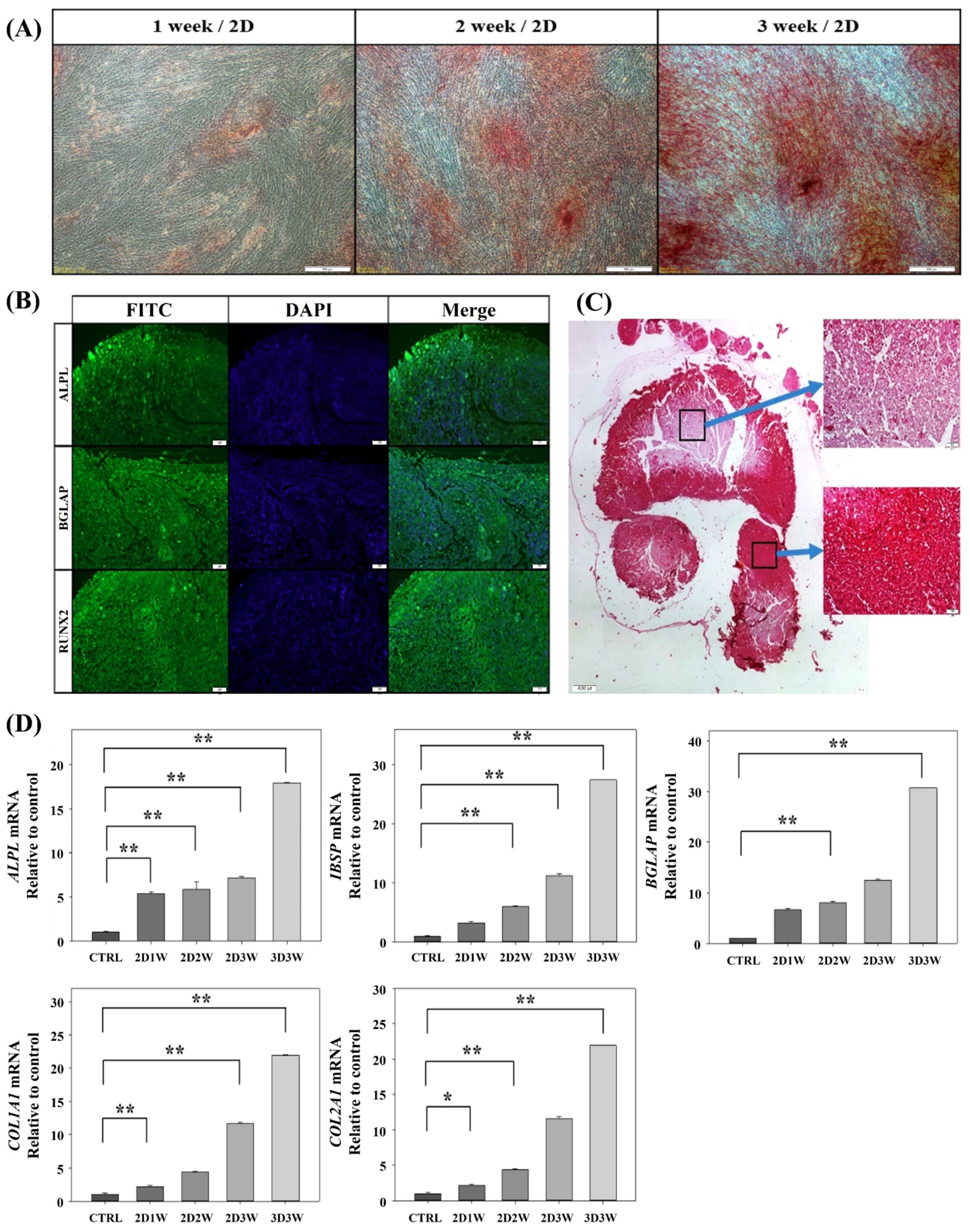
2.2. Osteogenic Differentiation of hADSCs in 2D and 3D Culture Conditions
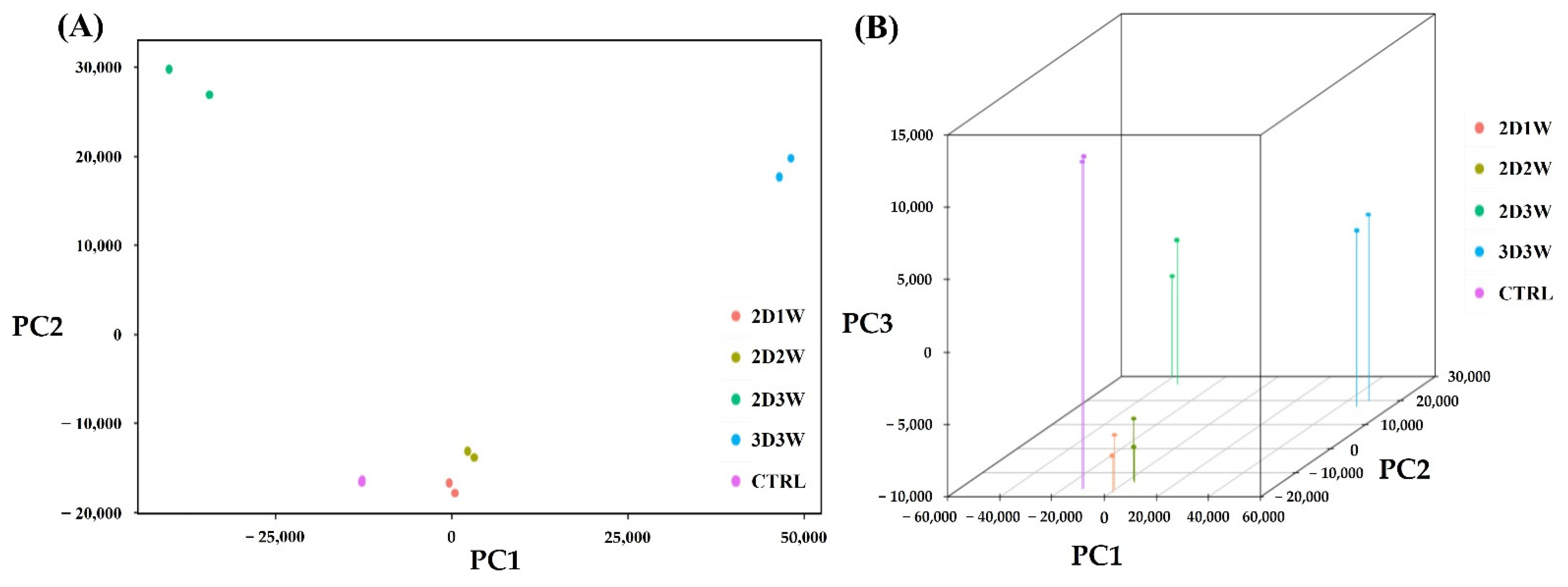
2.3. Gene Expression Profiles during Osteogenic Differentiation of hADSCs
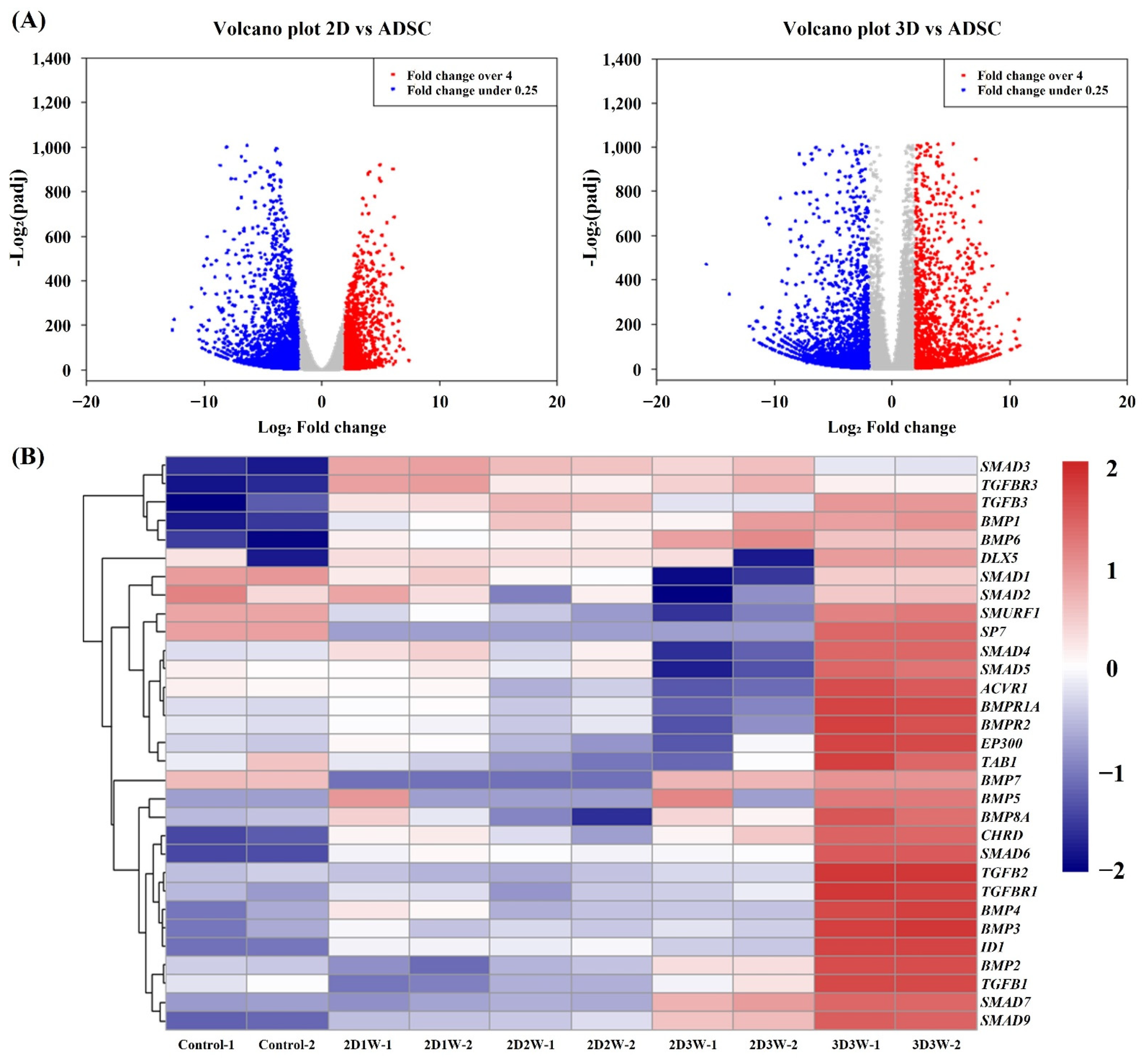
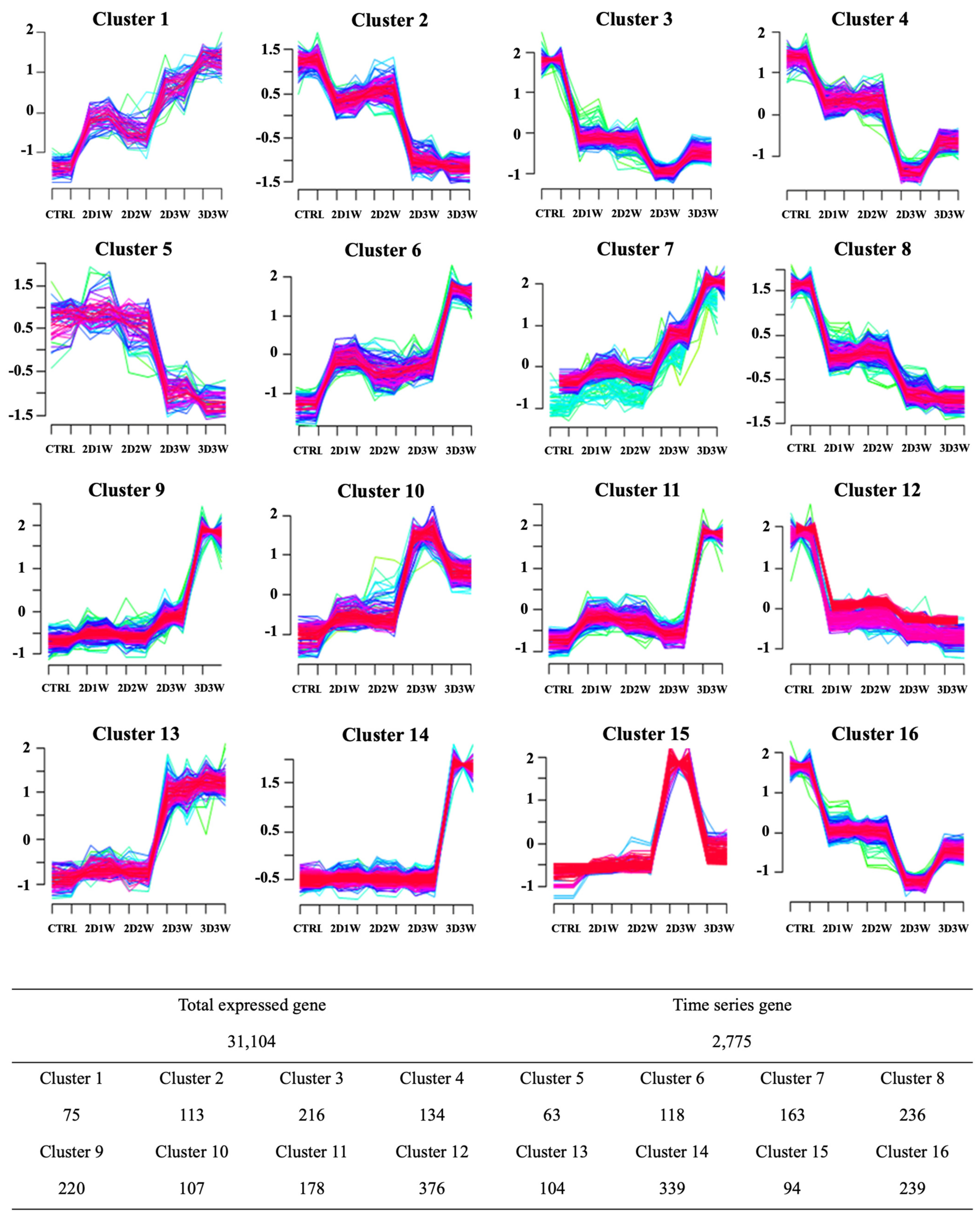
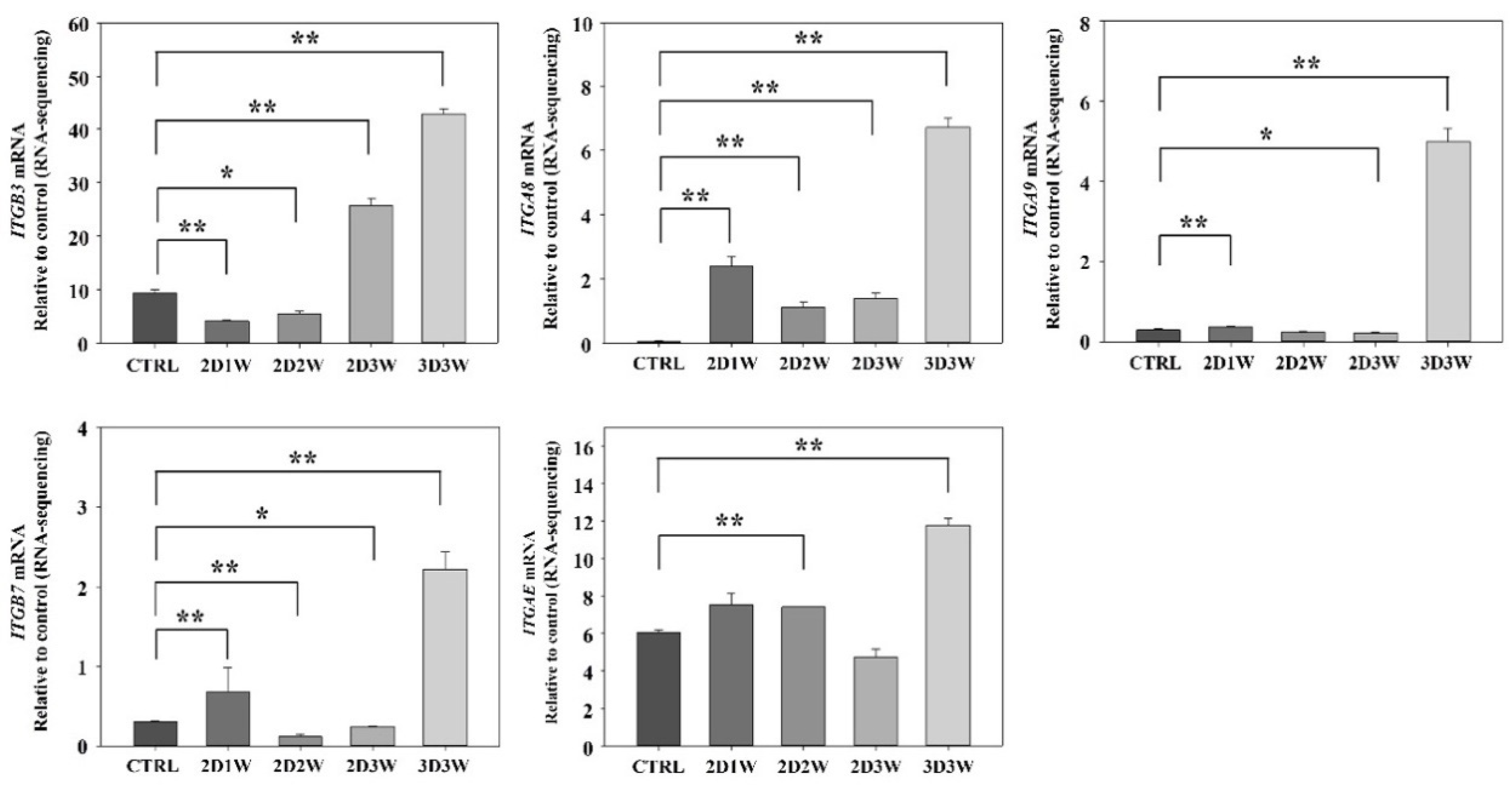
2.4. Identification of Differentially Expressed Genes during Osteogenic Differentiation
2.5. Identification of Molecular Signature Specific to Samples in 3D Culture Condition
3. Discussion
4. Materials and Methods
4.1. Cell Culture and Osteogenic Differentiation
4.2. Concave Microwell Preparation
4.3. Generation of hADSCs Spheroids and Osteogenic Differentiation in 3D Culture Condition
4.4. Quantitative Real-Time Polymerase Chain Reaction (qRT–PCR)
4.5. Histological Analysis of 3D Spheroid
4.6. Alizarin Red Staining
4.7. RNA Sequencing (RNA-Seq) and Read Mapping
4.8. Principal Component Analysis
4.9. Differential Gene Expression (DGE) Analysis and Functional Annotations for the Differentially Expressed Genes
4.10. Time-Series Analysis of Differential Gene Expression
4.11. RNA Sequencing Validation by qRT–PCR
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zuk, P.A.; Zhu, M.; Ashjian, P.; De Ugarte, D.A.; Huang, J.I.; Mizuno, H.; Alfonso, Z.C.; Fraser, J.K.; Benhaim, P.; Hedrick, M.H. Human adipose tissue is a source of multipotent stem cells. Mol. Biol. Cell 2002, 13, 4279–4295. [Google Scholar] [CrossRef]
- Caplan, A.I. Adult mesenchymal stem cells for tissue engineering versus regenerative medicine. J. Cell. Physiol. 2007, 213, 341–347. [Google Scholar] [CrossRef]
- Bianco, P.; Robey, P.G.; Simmons, P.J. Mesenchymal stem cells: Revisiting history, concepts, and assays. Cell Stem Cell 2008, 2, 313–319. [Google Scholar] [CrossRef] [Green Version]
- Burdick, J.A.; Vunjak-Novakovic, G. Engineered microenvironments for controlled stem cell differentiation. Tissue Eng. Part A 2009, 15, 205–219. [Google Scholar] [CrossRef]
- Kundu, A.K.; Khatiwala, C.B.; Putnam, A.J. Extracellular matrix remodeling, integrin expression, and downstream signaling pathways influence the osteogenic differentiation of mesenchymal stem cells on poly (lactide-co-glycolide) substrates. Tissue Eng. Part A 2009, 15, 273–283. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.; Itaka, K.; Ohba, S.; Nishiyama, N.; Chung, U.I.; Yamasaki, Y.; Kataoka, K. 3D spheroid culture system on micropatterned substrates for improved differentiation efficiency of multipotent mesenchymal stem cells. Biomaterials 2009, 30, 2705–2715. [Google Scholar] [CrossRef]
- Moeller, H.C.; Mian, M.K.; Shrivastava, S.; Chung, B.G.; Khademhosseini, A. A microwell array system for stem cell culture. Biomaterials 2007, 29, 752–763. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bhattacharya, I.; Ghayor, C.; Weber, F.E. The use of adipose tissue-derived progenitors in bone tissue engineering-a review. Transfus. Med. Hemother. 2016, 43, 336–343. [Google Scholar] [CrossRef] [Green Version]
- Hwang, Y.S.; Cho, J.; Tay, F.; Heng, J.Y.Y.; Ho, R.; Kazarian, S.G.; Williams, D.R.; Boccaccini, A.R.; Polak, J.M.; Mantalaris, A. The use of murine embryonic stem cells, alginate encapsulation, and rotary microgravity bioreactor in bone tissue engineering. Biomaterials 2008, 30, 499–507. [Google Scholar] [CrossRef] [PubMed]
- Sumanasinghe, R.D.; Bernacki, S.H.; Loboa, E.G. Osteogenic differentiation of human mesenchymal stem cells in collagen matrices: Effect of uniaxial cyclic tensile strain on bone morphogenetic protein (BMP-2) mRNA expression. Tissue Eng. 2006, 12, 3459–3465. [Google Scholar] [CrossRef]
- Calabrese, G.; Giuffrida, R.; Fabbi, C.; Figallo, E.; Furno, D.L.; Gulino, R.; Colarossi, C.; Fullone, F.; Giuffrida, R.; Parenti, R.; et al. Collagen-Hydroxyapatite Scaffolds Induce Human Adipose Derived Stem Cells Osteogenic Differentiation In Vitro. PLoS ONE 2016, 11, e0151181. [Google Scholar] [CrossRef] [Green Version]
- Jeong, C.S.; Chakrabarty, D.; Hahn, E.J.; Lee, H.L.; Paek, K.Y. Effects of oxygen, carbon dioxide and ethylene on growth and bioactive compound production in bioreactor culture of ginseng adventitious roots. Biochem. Eng. J. 2006, 27, 252–263. [Google Scholar] [CrossRef]
- Kempf, H.; Kropp, C.; Olmer, R.; Martin, U.; Zweigerdt, R. Cardiac differentiation of human pluripotent stem cells in scalable suspension culture. Nat. Protoc. 2015, 10, 1345–1361. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, Y.; Sato, S.; Kurashima, Y.; Yamamoto, T.; Kurokawa, S.; Yuki, Y.; Takemura, N.; Uematsu, S.; Lai, C.Y.; Otsu, M.; et al. A Refined Culture System for Human Induced Pluripotent Stem Cell-Derived Intestinal Epithelial Organoids. Stem Cell Rep. 2018, 10, 314–328. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pekor, C.; Gerlach, J.C.; Nettleship, I.; Schmelzer, E. Induction of hepatic and endothelial differentiation by perfusion in a three-dimensional cell culture model of human fetal liver. Tissue Eng. Part C Methods 2015, 21, 705–715. [Google Scholar] [CrossRef] [Green Version]
- Mellor, L.F.; Mohiti-Asli, M.; Williams, J.; Kannan, A.; Dent, M.R.; Guilak, F.; Loboa, E. G Extracellular calcium modulates chondrogenic and osteogenic differentiation of human adipose-derived stem cells: A novel approach for osteochondral tissue engineering using a single stem cell source. Tissue Eng. Part A 2015, 21, 2323–2333. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tung, Y.C.; Hsiao, A.Y.; Allen, S.G.; Torisawa, Y.S.; Ho, M.; Takayama, S. High-throughput 3D spheroid culture and drug testing using a 384 hanging drop array. Analyst 2011, 136, 473–478. [Google Scholar] [CrossRef] [PubMed]
- Mortazavi, A.; Wold, B.; Williams, B.A.; Schaeffer, L.; McCue, K. Mapping and quantifying mammalian transcriptomes by RNA-Seq. Nat. Methods 2008, 5, 621–628. [Google Scholar] [CrossRef]
- Meyer, M.B.; Benkusky, N.A.; Sen, B.; Rubin, J.; Pike, J.W. Epigenetic plasticity drives adipogenic and osteogenic differentiation of marrow-derived mesenchymal stem cells. J. Biol. Chem. 2016, 291, 17829–17847. [Google Scholar] [CrossRef] [Green Version]
- Twine, N.A.; Chen, L.; Pang, C.N.; Wilkins, M.R.; Kassem, M. Identification of differentiation-stage specific markers that define the ex vivo osteoblastic phenotype. Bone 2014, 67, 23–32. [Google Scholar] [CrossRef]
- Kim, M.J.; Yu, Y.S.; Moon, J.H.; Koh, I.S.; Lee, J.H. Differential Expression Profiling of Long Noncoding RNA and mRNA during Osteoblast Differentiation in Mouse. Int. J. Genom. 2018, 2018, 7691794. [Google Scholar] [CrossRef] [Green Version]
- Tannock, I.F.; Kopelyan, I. Influence of glucose concentration on growth and formation of necrosis in spheroids derived from a human bladder cancer cell line. Cancer Res. 1986, 46, 3105–3110. [Google Scholar]
- Strem, B.M.; Kevin, C.H.; Min, Z.; Isabella, W.; Zeni, A.; Ronda, E.S.; John, K.F.; Marc, H.H. Multipotential differentiation of adipose tissue-derived stem cells. Keio J. Med. 2005, 54, 132–141. [Google Scholar] [CrossRef] [Green Version]
- McCoy, R.J.; O’Brien, F.J. Influence of shear stress in perfusion bioreactor cultures for the development of three-dimensional bone tissue constructs: A review. Tissue Eng. Part B Rev. 2010, 16, 587–601. [Google Scholar] [CrossRef]
- Anada, T.; Fukuda, J.; Sai, Y.; Suzuki, O. An oxygen-permeable spheroid culture system for the prevention of central hypoxia and necrosis of spheroids. Biomaterials 2012, 33, 8430–8441. [Google Scholar] [CrossRef] [PubMed]
- Saarinen, M.A.; Reece, J.S.; Arnold, M.A.; Murhammer, D.W. Monitoring and controlling the dissolved oxygen (DO) concentration within the high aspect ratio vessel (HARV). Biotechnol. Prog. 2003, 19, 1335–1341. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Tu, Q.; Meng, S.; Zhang, L.; Yu, L.; Song, J.; Hu, Y.; Sui, L.; Zhang, J.; Dard, M.; et al. Runx2/DICER/miRNA pathway in regulating osteogenesis. J. Cell. Physiol. 2017, 232, 182–191. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fisher, L.W.; McBride, O.W.; Termine, J.D.; Young, M.F. Human bone sialoprotein. Deduced protein sequence and chromosomal localization. J. Biol. Chem. 1990, 265, 2347–2351. [Google Scholar] [CrossRef]
- Gao, G.; Schilling, A.F.; Hubbell, K.; Yonezawa, T.; Truong, D.; Hong, Y.; Dai, G.; Cui, X. Improved properties of bone and cartilage tissue from 3D inkjet-bioprinted human mesenchymal stem cells by simultaneous deposition and photocrosslinking in PEG-GelMA. Biotechnol. Lett. 2015, 37, 2349–2355. [Google Scholar] [CrossRef]
- Golub, E.E.; Boesze-Battaglia, K. The role of alkaline phosphatase in mineralization. Curr. Opin. Orthop. 2007, 18, 444–448. [Google Scholar] [CrossRef]
- Moss, D.W. Perspectives in alkaline phosphatase research. Clin. Chem. 1992, 38, 2486–2492. [Google Scholar] [CrossRef]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef] [Green Version]
- Chen, G.; Deng, C.; Li, Y.P. TGF-β and BMP signaling in osteoblast differentiation and bone formation. Int. J. Biol. Sci. 2012, 8, 272–288. [Google Scholar] [CrossRef] [Green Version]
- Kumar, L.; Futschik, M.E. Mfuzz: A software package for soft clustering of microarray data. Bioinformation 2007, 2, 5–7. [Google Scholar] [CrossRef] [PubMed]
- Olivares-Navarrete, R.; Rodil, S.E.; Hyzy, S.L.; Dunn, G.R.; Almaguer-Flores, A.; Schwartz, Z.; Boyan, B.D. Role of integrin subunits in mesenchymal stem cell differentiation and osteoblast maturation on graphitic carbon-coated microstructured surfaces. Biomaterials 2015, 51, 69–79. [Google Scholar] [CrossRef] [Green Version]
- Drury, J.L.; Mooney, D.J. Hydrogels for tissue engineering: Scaffold design variables and applications. Biomaterials 2003, 24, 4337–4351. [Google Scholar] [CrossRef]
- Ratner, B.D. Replacing and renewing: Synthetic materials, biomimetics, and tissue engineering in implant dentistry. J. Dent. Educ. 2001, 65, 1340–1347. [Google Scholar] [CrossRef] [PubMed]
- Bose, S.; Roy, M.; Bandyopadhyay, A. Recent advances in bone tissue engineering scaffolds. Trends Biotechnol. 2012, 30, 546–554. [Google Scholar] [CrossRef] [Green Version]
- Dozio, S.M.; Montesi, M.; Campodoni, E.; Sandri, M.; Piattelli, A.; Tampieri, A.; Panseri, S. Differences in osteogenic induction of human mesenchymal stem cells between a tailored 3D hybrid scaffold and a 2D standard culture. J. Mater. Sci. Mater. Med. 2019, 30, 136. [Google Scholar] [CrossRef]
- Bicer, M.; Cottrell, G.S.; Widera, D. Impact of 3D cell culture on bone regeneration potential of mesenchymal stromal cells. Stem Cell Res. Ther. 2021, 12, 31. [Google Scholar] [CrossRef] [PubMed]
- Asagiri, M.; Koga, T.; de Crombrugghe, B.; Kodama, T.; Matsui, Y.; Takayanagi, H.; Nakashima, K. NFAT and Osterix cooperatively regulate bone formation. Nat. Med. 2005, 11, 880–885. [Google Scholar]
- Rahman, M.S.; Akhtar, N.; Jamil, H.M.; Banik, R.S.; Asaduzzaman, S.M. TGF-β/BMP signaling and other molecular events: Regulation of osteoblastogenesis and bone formation. Bone Res. 2015, 3, 15005. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, M.; Chen, G.; Li, Y.P. TGF-β and BMP signaling in osteoblast, skeletal development, and bone formation, homeostasis and disease. Bone Res. 2016, 4, 16009. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Patil, S.; Gao, Y.G.; Qian, A. The Bone Extracellular Matrix in Bone Formation and Regeneration. Front. Pharmacol. 2020, 11, 757. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Tang, Y.; Lozito, T.P.; Oyster, N.; Wang, B.; Tuan, R.S. Efficient in vivo bone formation by BMP-2 engineered human mesenchymal stem cells encapsulated in a projection stereolithographically fabricated hydrogel scaffold. Stem Cell Res. Ther. 2019, 10, 254. [Google Scholar] [CrossRef] [Green Version]
- Campbell, I.D.; Humphries, M.J. Integrin structure, activation, and interactions. Cold Spring Harb. Perspect. Biol. 2011, 3, a004994. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lehmann, J.; Huehn, J.; de la Rosa, M.; Maszyna, F.; Kretschmer, U.; Krenn, V.; Brunner, M.; Scheffold, A.; Hamann, A. Expression of the integrin αEβ7 identifies unique subsets of CD25+ as well as CD25− regulatory T cells. Proc. Natl. Acad. Sci. USA 2002, 99, 13031–13036. [Google Scholar] [CrossRef] [Green Version]
- Dobin, A.; Davis, C.A.; Schlesinger, F.; Drenkow, J.; Zaleski, C.; Jha, S.; Batut, P.; Chaisson, M.; Gingeras, T.R. STAR: Ultrafast universal RNA-seq aligner. Bioinformatics 2013, 29, 15–21. [Google Scholar] [CrossRef]
- Li, B.; Ruotti, V.; Stewart, R.M.; Thomson, J.A.; Dewey, C.N. RNA-Seq gene expression estimation with read mapping uncertainty. Bioinformatics 2009, 26, 493–500. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.H.; Gao, C.; Peng, G.; Greer, C.; Ren, S.; Wang, Y.; Xiao, X. Analysis of transcriptome complexity through RNA sequencing in normal and failing murine hearts. Circ. Res. 2011, 109, 1332–1341. [Google Scholar] [CrossRef] [Green Version]

| GO Term | GO Term ID | Gene Count | p-Value |
|---|---|---|---|
| extracellular matrix organization | GO:0030198 | 23 | <0.0001 |
| skeletal system development | GO:0001501 | 17 | <0.0001 |
| angiogenesis | GO:0001525 | 16 | 0.0003 |
| osteoblast differentiation | GO:0001649 | 11 | <0.0001 |
| BMP signaling pathway | GO:0030509 | 10 | <0.0001 |
| embryonic limb morphogenesis | GO:0030326 | 10 | <0.0001 |
| cartilage development | GO:0051216 | 10 | <0.0001 |
| collagen fibril organization | GO:0030199 | 9 | <0.0001 |
| embryonic skeletal system morphogenesis | GO:0048704 | 9 | <0.0001 |
| odontogenesis of dentin-containing tooth | GO:0042475 | 9 | 0.0001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, B.-C.; Kwack, K.H.; Chun, J.; Lee, J.-H. Comparative Transcriptome Analysis of Human Adipose-Derived Stem Cells Undergoing Osteogenesis in 2D and 3D Culture Conditions. Int. J. Mol. Sci. 2021, 22, 7939. https://doi.org/10.3390/ijms22157939
Kim B-C, Kwack KH, Chun J, Lee J-H. Comparative Transcriptome Analysis of Human Adipose-Derived Stem Cells Undergoing Osteogenesis in 2D and 3D Culture Conditions. International Journal of Molecular Sciences. 2021; 22(15):7939. https://doi.org/10.3390/ijms22157939
Chicago/Turabian StyleKim, Byung-Chul, Kyu Hwan Kwack, Jeewan Chun, and Jae-Hyung Lee. 2021. "Comparative Transcriptome Analysis of Human Adipose-Derived Stem Cells Undergoing Osteogenesis in 2D and 3D Culture Conditions" International Journal of Molecular Sciences 22, no. 15: 7939. https://doi.org/10.3390/ijms22157939
APA StyleKim, B.-C., Kwack, K. H., Chun, J., & Lee, J.-H. (2021). Comparative Transcriptome Analysis of Human Adipose-Derived Stem Cells Undergoing Osteogenesis in 2D and 3D Culture Conditions. International Journal of Molecular Sciences, 22(15), 7939. https://doi.org/10.3390/ijms22157939






