The Antimicrobial Activity of Peripheral Blood Neutrophils Is Altered in Patients with Primary Ciliary Dyskinesia
Abstract
1. Introduction
2. Results
2.1. Patients and Controls
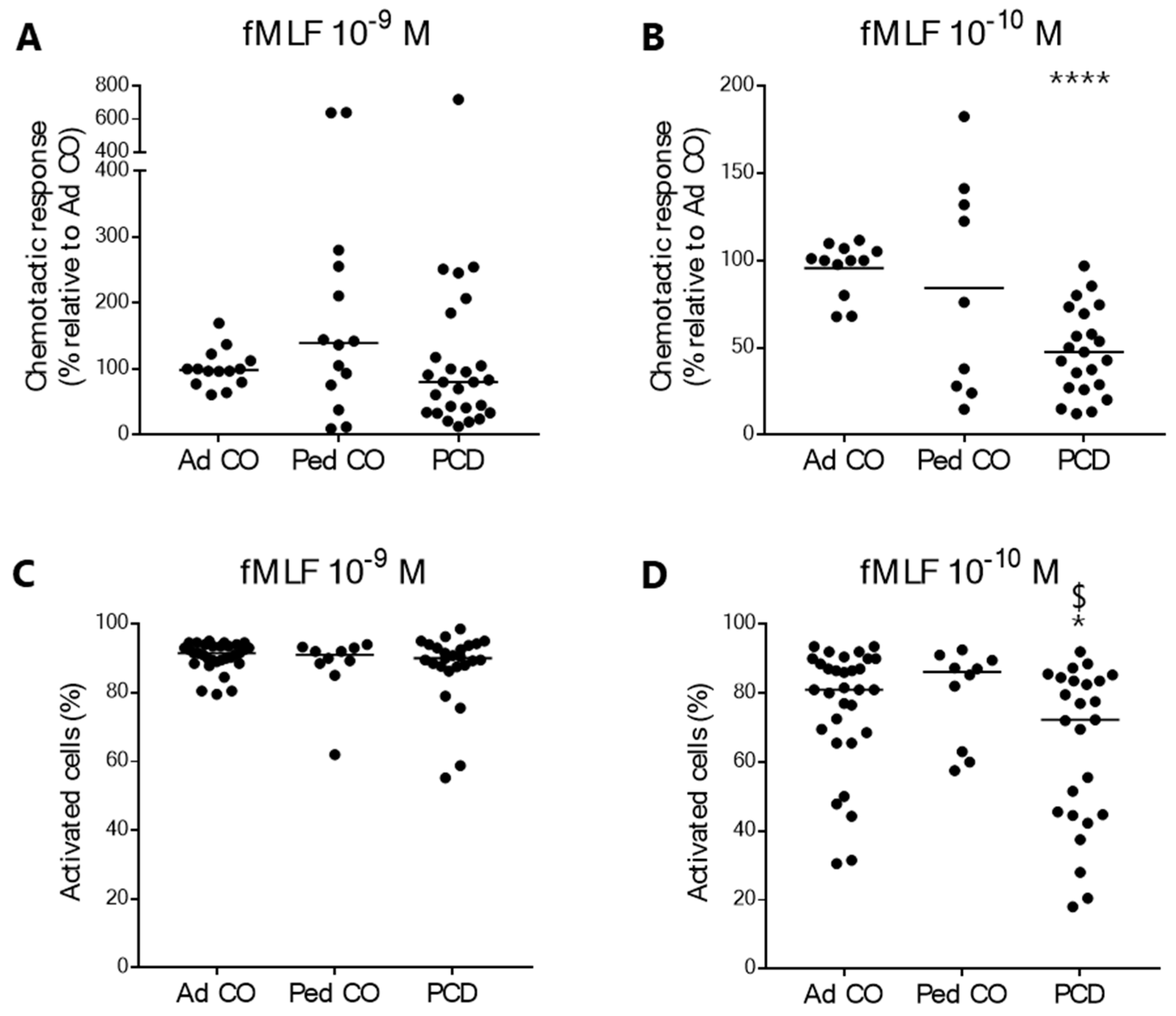
2.2. Peripheral Blood Neutrophils from Patients with PCD Show Reduced Chemotactic Response to fMLF in Three Chemotaxis Assays
2.3. Compared to Peripheral Blood Neutrophils of Healthy Individuals, Neutrophils from Patients with PCD Express Similar Adhesion Molecule Levels upon Stimulation
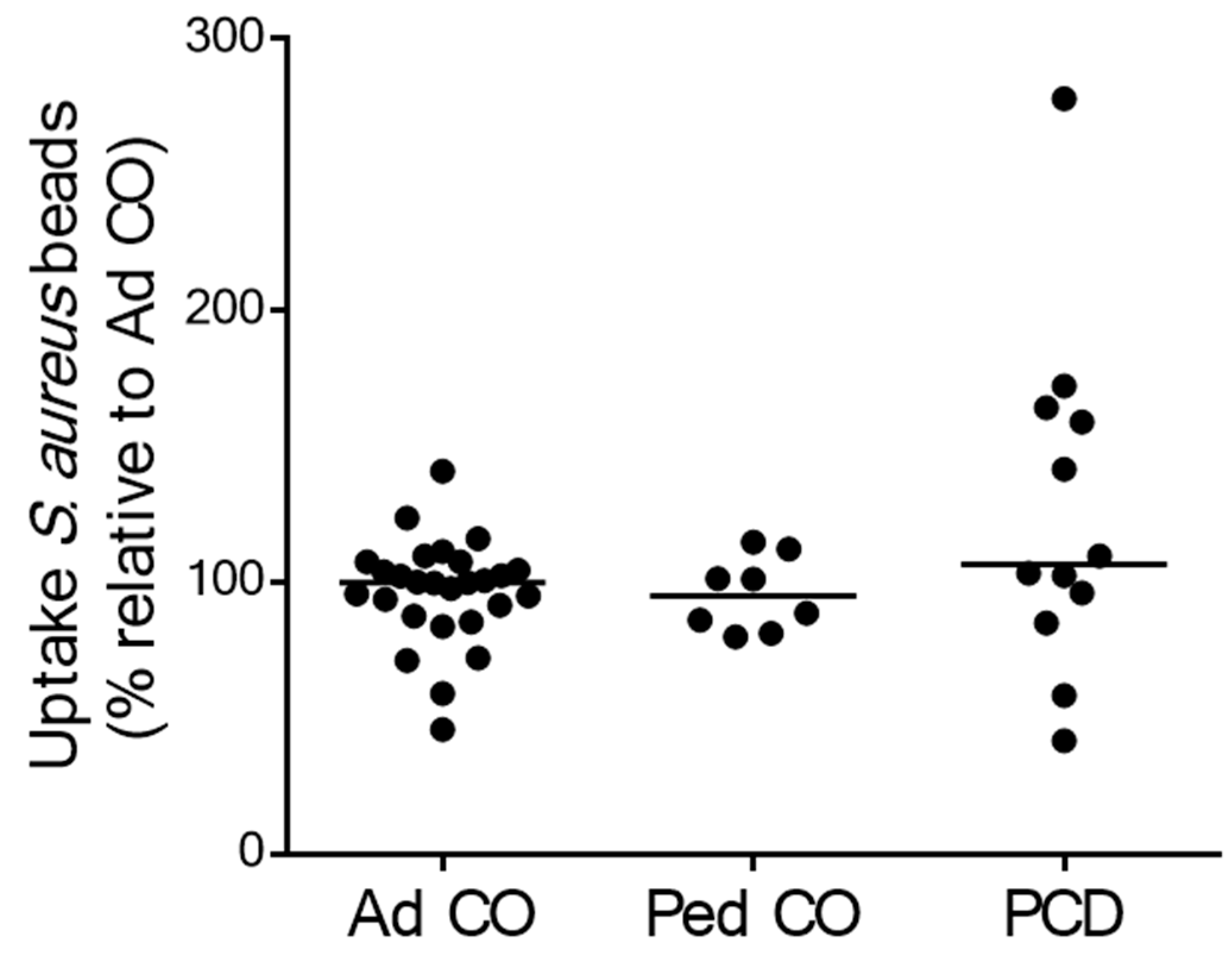
2.4. The Phagocytic Capacity of Peripheral Blood Neutrophils Is Not Affected in Patients with PCD
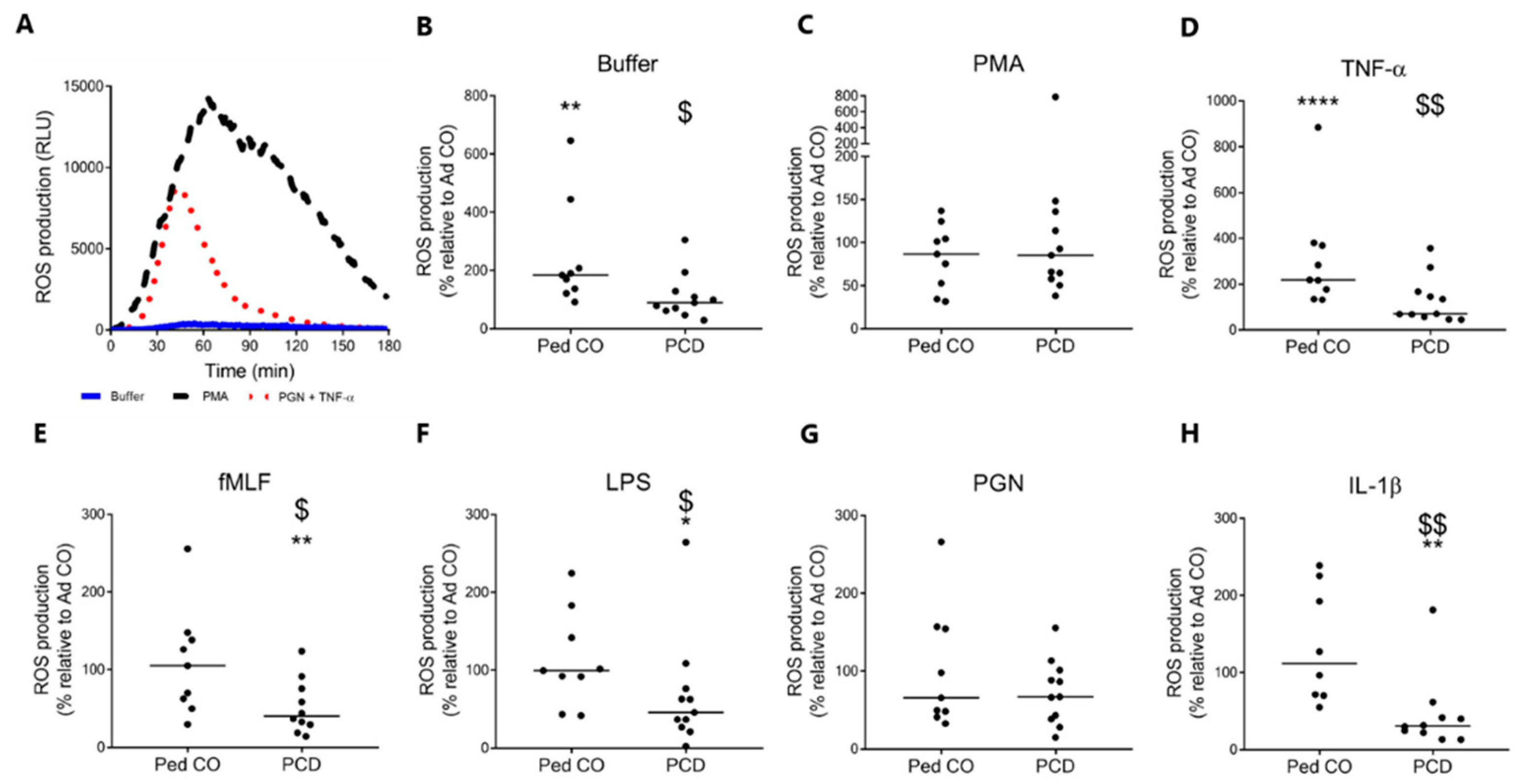
2.5. Peripheral Blood PMNs from Patients with PCD Produce Less Reactive Oxygen Species Compared to PMNs from Age-Matched Pediatric Controls
2.6. Circulating PMNs from Patients with PCD Tend to Release Less Neutrophil Extracellular Traps Compared to PMNs from Healthy Adults
2.7. Peripheral Blood Neutrophils from Patients with PCD Produce Less CXCL8 Compared to Neutrophils from Healthy Children
3. Discussion
4. Materials and Methods
4.1. Patients and Healthy Controls
4.2. Sample Collection and Processing
4.3. Reagents
4.4. Boyden Chamber Chemotaxis Assay
4.5. Shape Change Assay
4.6. µ-Slide 2D Chemotaxis Assay
4.7. Flow Cytometry
4.8. Adhesion Molecule Activity Assay
4.9. Phagocytosis Assay
4.10. Respiratory Burst Assay
4.11. Neutrophil Extracellular Trap (NET) Assay
4.12. Cell Induction
4.13. Statistics
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Boon, M.; Jorissen, M.; Proesmans, M.; De Boeck, K. Primary ciliary dyskinesia, an orphan disease. Eur. J. Pediatrc. 2013, 172, 151–162. [Google Scholar] [CrossRef]
- Bush, A.; Chodhari, R.; Collins, N.; Copeland, F.; Hall, P.; Harcourt, J.; Hariri, M.; Hogg, C.; Lucas, J.S.; Mitchison, H.M.; et al. Primary ciliary dyskinesia: Current state of the art. Arch. Dis. Child. 2007, 92, 1136–1140. [Google Scholar] [CrossRef]
- Knowles, M.R.; Daniels, L.A.; Davis, S.D.; Zariwala, M.A.; Leigh, M.W. Primary Ciliary Dyskinesia. Recent Advances in Diagnostics, Genetics, and Characterization of Clinical Disease. Am. J. Respir. Crit. Care Med. 2013, 188, 913–922. [Google Scholar] [CrossRef]
- Daniels, M.L.; Noone, P.G. Genetics, diagnosis and future treatment strategies for primary ciliary dyskinesia. Expert Opin. Orphan Drugs 2015, 3, 31–44. [Google Scholar] [CrossRef] [PubMed]
- Damseh, N.; Quercia, N.; Rumman, N.; Dell, S.D.; Kim, R.H. Primary ciliary dyskinesia: Mechanisms and management. Appl. Clin. Genet. 2017, 10, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Polineni, D.; Davis, S.D.; Dell, S.D. Treatment recommendations in Primary Ciliary Dyskinesia. Paediatr. Respir. Rev. 2016, 18, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Mirra, V.; Werner, C.; Santamaria, F. Primary Ciliary Dyskinesia: An Update on Clinical Aspects, Genetics, Diagnosis, and Future Treatment Strategies. Front. Pediatr. 2017, 5, 135. [Google Scholar] [CrossRef]
- Behan, L.; Rubbo, B.; Lucas, J.S.; Galvin, A.D. The patient’s experience of primary ciliary dyskinesia: A systematic review. Qual. Life Res. 2017, 26, 2265–2285. [Google Scholar] [CrossRef]
- Bush, A.; Payne, D.; Pike, S.; Jenkins, G.; Henke, M.O.; Rubin, B.K. Mucus Properties in Children with Primary Ciliary Dyskinesia: Comparison with cystic fibrosis. Chest 2006, 129, 118–123. [Google Scholar] [CrossRef]
- Ratjen, F.; Waters, V.; Klingel, M.; McDonald, N.; Dell, S.; Leahy, T.R.; Yau, Y.; Grasemann, H. Changes in airway inflammation during pulmonary exacerbations in patients with cystic fibrosis and primary ciliary dyskinesia. Eur. Respir. J. 2016, 47, 829–836. [Google Scholar] [CrossRef]
- Juss, J.K.; House, D.; Amour, A.; Begg, M.; Herre, J.; Storisteanu, D.M.; Hoenderdos, K.; Bradley, G.; Lennon, M.; Summers, C.; et al. Acute Respiratory Distress Syndrome Neutrophils Have a Distinct Phenotype and Are Resistant to Phosphoinositide 3-Kinase Inhibition. Am. J. Respir. Crit. Care Med. 2016, 194, 961–973. [Google Scholar] [CrossRef]
- Hoenderdos, K.; Condliffe, A. The Neutrophil in Chronic Obstructive Pulmonary Disease. Too Little, Too Late or Too Much, Too Soon? Am. J. Respir. Cell Mol. Biol. 2013, 48, 531–539. [Google Scholar] [CrossRef] [PubMed]
- Baines, K.J.; Simpson, J.L.; Scott, R.J.; Gibson, P.G. Immune responses of airway neutrophils are impaired in asthma. Exp. Lung Res. 2009, 35, 554–569. [Google Scholar] [CrossRef] [PubMed]
- Cockx, M.; Gouwy, M.; Van Damme, J.; Struyf, S. Chemoattractants and cytokines in primary ciliary dyskinesia and cystic fibrosis: Key players in chronic respiratory diseases. Cell. Mol. Immunol. 2018, 15, 312–323. [Google Scholar] [CrossRef] [PubMed]
- Cantin, A.M.; Hartl, D.; Konstan, M.W.; Chmiel, J.F. Inflammation in cystic fibrosis lung disease: Pathogenesis and therapy. J. Cyst. Fibros. 2015, 14, 419–430. [Google Scholar] [CrossRef]
- Gifford, A.M.; Chalmers, J.D. The role of neutrophils in cystic fibrosis. Curr. Opin. Hematol. 2014, 21, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Mócsai, A. Diverse novel functions of neutrophils in immunity, inflammation, and beyond. J. Exp. Med. 2013, 210, 1283–1299. [Google Scholar] [CrossRef]
- Wang, J.; Arase, H. Regulation of immune responses by neutrophils. Ann. N. Y. Acad. Sci. 2014, 1319, 66–81. [Google Scholar] [CrossRef]
- Sørensen, O.E.; Borregaard, N. Neutrophil extracellular traps—The dark side of neutrophils. J. Clin. Investig. 2016, 126, 1612–1620. [Google Scholar] [CrossRef]
- Jaillon, S.; Galdiero, M.R.; Del Prete, D.; Cassatella, M.A.; Garlanda, C.; Mantovani, A. Neutrophils in innate and adaptive immunity. Semin. Immunopathol. 2013, 35, 377–394. [Google Scholar] [CrossRef]
- Spoor, J.; Farajifard, H.F.; Rezaei, N. Congenital neutropenia and primary immunodeficiency diseases. Crit. Rev. Oncol. Hematol. 2019, 133, 149–162. [Google Scholar] [CrossRef]
- Dinauer, M.C. Primary immune deficiencies with defects in neutrophil function. Hematol. Am. Soc. Hematol. Educ. Program. 2016, 2016, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Lucas, J.S.; Barbato, A.; Collins, S.A.; Goutaki, M.; Behan, L.; Caudri, D.; Dell, S.; Eber, E.; Escudier, E.; Hirst, R.A.; et al. European Respiratory Society guidelines for the diagnosis of primary ciliary dyskinesia. Eur. Respir. J. 2017, 49, 1601090. [Google Scholar] [CrossRef] [PubMed]
- Dwyer, M.; Shan, Q.; D’Ortona, S.; Maurer, R.; Mitchell, R.; Olesen, H.; Thiel, S.; Huebner, J.; Gadjeva, M. Cystic Fibrosis Sputum DNA Has NETosis Characteristics and Neutrophil Extracellular Trap Release Is Regulated by Macrophage Migration-Inhibitory Factor. J. Innate Immun. 2014, 6, 765–779. [Google Scholar] [CrossRef]
- Liu, J.; Pang, Z.; Wang, G.; Guan, X.; Fang, K.; Wang, Z.; Wang, F. Advanced Role of Neutrophils in Common Respiratory Diseases. J. Immunol. Res. 2017, 2017, 1–21. [Google Scholar] [CrossRef]
- Paone, G.; Conti, V.; Vestri, A.; Leone, A.; Puglisi, G.; Benassi, F.; Brunetti, G.; Schmid, G.; Cammarella, I.; Terzano, C. Analysis of Sputum Markers in the Evaluation of Lung Inflammation and Functional Impairment in Symptomatic Smokers and COPD Patients. Dis. Markers 2011, 31, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Brockbank, S.; Downey, D.; Elborn, J.S.; Ennis, M. Effect of cystic fibrosis exacerbations on neutrophil function. Int. Immunopharmacol. 2005, 5, 601–608. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Burnett, D.; Chamba, A.; Hill, S.; Stockley, R.A. Neutrophils from subjects with chronic obstructive lung disease show enhanced chemotaxis and extracellular proteolysis. Lancet 1987, 2, 1043–1046. [Google Scholar] [CrossRef]
- Tang, F.S.; Van Ly, D.; Spann, K.; Reading, P.C.; Burgess, J.K.; Hartl, D.; Baines, K.J.; Oliver, B.G. Differential neutrophil activation in viral infections: Enhanced TLR-7/8-mediated CXCL8 release in asthma. Respirology 2016, 21, 172–179. [Google Scholar] [CrossRef] [PubMed]
- Konstan, M.W. Clinical Trials Gov 2018-02-23 2006; NCT00322868. Available online: https://clinicaltrials.gov/ct2/search (accessed on 28 February 2019).
- Cockx, M.; Gouwy, M.; Godding, V.; De Boeck, K.; Van Damme, J.; Boon, M.; Struyf, S. Neutrophils from Patients with Primary Ciliary Dyskinesia Display Reduced Chemotaxis to CXCR2 Ligands. Front. Immunol. 2017, 8. [Google Scholar] [CrossRef]
- Koh, Y.Y.; Sun, Y.H.; Min, Y.-G.; Chi, J.-G.; Kim, C.K. Chemotaxis of Blood Neutrophils from Patients with Primary Ciliary Dyskinesia. J. Korean Med. Sci. 2003, 18, 36–41. [Google Scholar] [CrossRef]
- Corkey, C.W.; Minta, J.O.; Turner, J.A.; Biggar, W.D. Neutrophil function in the immotile cilia syndrome. J. Lab. Clin. Med. 1982, 99, 838–844. [Google Scholar]
- Montecucco, F.; Steffens, S.; Burger, F.; Da Costa, A.; Bianchi, G.; Bertolotto, M.B.; Mach, F.; Dallegri, F.; Ottonello, L. Tumor necrosis factor-alpha (TNF-α) induces integrin CD11b/CD18 (Mac-1) up-regulation and migration to the CC chemokine CCL3 (MIP-1α) on human neutrophils through defined signalling pathways. Cell. Signal. 2008, 20, 557–568. [Google Scholar] [CrossRef] [PubMed]
- Ley, K.; Laudanna, C.; Cybulsky, M.I.; Nourshargh, S. Getting to the site of inflammation: The leukocyte adhesion cascade updated. Nat. Rev. Immunol. 2007, 7, 678–689. [Google Scholar] [CrossRef]
- Antonelli, M.; Moschese, V.; Bertasi, S.; Galli, E.; Stancati, M.; Lombardi, V.R. Leukocyte locomotory function in children with the immotile cilia syndrome. Helv. Paediatr. Acta 1986, 41, 409–414. [Google Scholar] [PubMed]
- Walter, R.J.; Danielson, J.R.; Reyes, H.M. Characterization of a Chemotactic Defect in Patients with Kartagener Syndrome. Arch. Otolaryngol. Head Neck Surg. 1990, 116, 465–469. [Google Scholar] [CrossRef]
- Forsman, H.; Önnheim, K.; Andréasson, E.; Christenson, K.; Karlsson, A.; Bylund, J.; Dahlgren, C. Reactivation of Desensitized Formyl Peptide Receptors by Platelet Activating Factor: A Novel Receptor Cross Talk Mechanism Regulating Neutrophil Superoxide Anion Production. PLoS ONE 2013, 8, e60169. [Google Scholar] [CrossRef]
- Mortier, A.; Loos, T.; Gouwy, M.; Ronsse, I.; Van Damme, J.; Proost, P. Posttranslational Modification of the NH2-terminal Region of CXCL5 by Proteases or Peptidylarginine Deiminases (PAD) Differently Affects Its Biological Activity. J. Biol. Chem. 2010, 285, 29750–29759. [Google Scholar] [CrossRef]
- Fong, O.N.; Chan, K.Y.; Leung, K.T.; Lam, H.S.; Cheung, H.M.; Leung, T.Y.; Li, K.; Ng, P.C. Expression profile of cord blood neutrophils and dysregulation of HSPA1A and OLR1 upon challenge by bacterial peptidoglycan. J. Leukoc. Biol. 2014, 95, 169–178. [Google Scholar] [CrossRef] [PubMed]
- Pizurki, L.; Morris, M.A.; Chanson, M.; Solomon, M.; Pavirani, A.; Bouchardy, I.; Suter, S. Cystic Fibrosis Transmembrane Conductance Regulator Does Not Affect Neutrophil Migration across Cystic Fibrosis Airway Epithelial Monolayers. Am. J. Pathol. 2000, 156, 1407–1416. [Google Scholar] [CrossRef]
- Russell, K.J.; McRedmond, J.; Mukherji, N.; Costello, C.; Keatings, V.; Linnane, S.; Henry, M.; Fitzgerald, M.X.; O’Connor, C.M. Neutrophil Adhesion Molecule Surface Expression and Responsiveness in Cystic Fibrosis. Am. J. Respir. Crit. Care Med. 1998, 157, 756–761. [Google Scholar] [CrossRef] [PubMed]
- Noguera, A.; Batle, S.; Miralles, C.; Iglesias, J.; Busquets, X.; MacNee, W.; Agustí, A.G.N. Enhanced neutrophil response in chronic obstructive pulmonary disease. Thorax 2001, 56, 432–437. [Google Scholar] [CrossRef]
- Noguera, A.; Busquets, X.; Sauleda, J.; Villaverde, J.M.; MacNee, W.; Agustí, A.G.N. Expression of Adhesion Molecules and G Proteins in Circulating Neutrophils in Chronic Obstructive Pulmonary Disease. Am. J. Respir. Crit. Care Med. 1998, 158, 1664–1668. [Google Scholar] [CrossRef] [PubMed]
- Mann, B.S.; Chung, K.F. Blood neutrophil activation markers in severe asthma: Lack of inhibition by prednisolone therapy. Respir. Res. 2006, 7, 59. [Google Scholar] [CrossRef] [PubMed]
- Morris, M.R.; Doull, I.J.M.; Dewitt, S.; Hallett, M.B. Reduced iC3b-mediated phagocytotic capacity of pulmonary neutrophils in cystic fibrosis. Clin. Exp. Immunol. 2005, 142, 68–75. [Google Scholar] [CrossRef]
- Gray, R.D.; Hardisty, G.; Regan, K.H.; Smith, M.; Robb, C.T.; Duffin, R.; Mackellar, A.; Felton, J.M.; Paemka, L.; McCullagh, B.N.; et al. Delayed neutrophil apoptosis enhances NET formation in cystic fibrosis. Thorax 2018, 73, 134–144. [Google Scholar] [CrossRef]
- Stockley, J.A.; Walton, G.M.; Lord, J.M.; Sapey, E. Aberrant neutrophil functions in stable chronic obstructive pulmonary disease: The neutrophil as an immunotherapeutic target. Int. Immunopharmacol. 2013, 17, 1211–1217. [Google Scholar] [CrossRef]
- Mosca, T.; Menezes, M.C.S.; Silva, A.V.; Stirbulov, R.; Forte, W.C.N. Chemotactic and Phagocytic Activity of Blood Neutrophils in Allergic Asthma. Immunol. Investig. 2015, 44, 509–520. [Google Scholar] [CrossRef]
- Witko-Sarsat, V.; Delacourt, C.; Rabier, D.; Bardet, J.; Nguyen, A.T.; Descamps-Latscha, B. Neutrophil-derived long-lived oxidants in cystic fibrosis sputum. Am. J. Respir. Crit. Care Med. 1995, 152, 1910–1916. [Google Scholar] [CrossRef]
- Teramoto, S.; Shu, C.Y.; Ouchi, Y.; Fukuchi, Y. Increased Spontaneous Production and Generation of Superoxide Anion by Blood Neutrophils in Patients with Asthma. J. Asthma 1996, 33, 149–155. [Google Scholar] [CrossRef]
- Corvol, H.; Fitting, C.; Chadelat, K.; Jacquot, J.; Tabary, O.; Boule, M.; Cavaillon, J.-M.; Clement, A. Distinct cytokine production by lung and blood neutrophils from children with cystic fibrosis. Am. J. Physiol. Cell. Mol. Physiol. 2003, 284, L997–L1003. [Google Scholar] [CrossRef] [PubMed]
- Montemurro, P.; Mariggiò, M.A.; Barbuti, G.; Cassano, A.; Vincenti, A.; Serio, G.; Guerra, L.; Diana, A.; Santostasi, T.; Polizzi, A.; et al. Increase in interleukin-8 production from circulating neutrophils upon antibiotic therapy in cystic fibrosis patients. J. Cyst. Fibros. 2012, 11, 518–524. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Baines, K.J.; Simpson, J.L.; Bowden, N.A.; Scott, R.J.; Gibson, P.G. Differential gene expression and cytokine production from neutrophils in asthma phenotypes. Eur. Respir. J. 2010, 35, 522–531. [Google Scholar] [CrossRef]
- Lawrence, R.H.; Sorrelli, T.C. Decreased polymorphonuclear leucocyte chemotactic response to leukotriene B4 in cystic fibrosis. Clin. Exp. Immunol. 1992, 89, 321–324. [Google Scholar] [CrossRef]
- Dai, Y.; Dean, T.P.; Church, M.K.; Warner, J.O.; Shute, J.K. Desensitisation of neutrophil responses by systemic interleukin 8 in cystic fibrosis. Thorax 1994, 49, 867–871. [Google Scholar] [CrossRef] [PubMed]
- Brennan, S.; Cooper, D.; Sly, P.D. Directed neutrophil migration to IL-8 is increased in cystic fibrosis: A study of the effect of erythromycin. Thorax 2001, 56, 62–64. [Google Scholar] [CrossRef][Green Version]
- Uhlén, M.; Fagerberg, L.; Hallström, B.M.; Lindskog, C.; Oksvold, P.; Mardinoglu, A.; Sivertsson, Å.; Kampf, C.; Sjöstedt, E.; Asplund, A.; et al. Tissue-based map of the human proteome. Science 2015, 347, 1260419. [Google Scholar] [CrossRef] [PubMed]
- Gouwy, M.; Struyf, S.; Catusse, J.; Proost, P.; Van Damme, J. Synergy between proinflammatory ligands of G protein-coupled receptors in neutrophil activation and migration. J. Leukoc. Biol. 2004, 76, 185–194. [Google Scholar] [CrossRef]
- Gouwy, M.; De Buck, M.; Pörtner, N.; Opdenakker, G.; Proost, P.; Struyf, S.; Van Damme, J. Serum amyloid A chemoattracts immature dendritic cells and indirectly provokes monocyte chemotaxis by induction of cooperating CC and CXC chemokines. Eur. J. Immunol. 2015, 45, 101–112. [Google Scholar] [CrossRef]
- Gupta, S.; Chan, D.W.; Zaal, K.J.; Kaplan, M.J. A High-Throughput Real-Time Imaging Technique to Quantify NETosis and Distinguish Mechanisms of Cell Death in Human Neutrophils. J. Immunol. 2018, 200, 869–879. [Google Scholar] [CrossRef]
- Cockx, M.; Gouwy, M.; Ruytinx, P.; Lodewijckx, I.; Van Hout, A.; Knoops, S.; Pörtner, N.; Ronsse, I.; Vanbrabant, L.; Godding, V.; et al. Monocytes from patients with Primary Ciliary Dyskinesia show enhanced inflammatory properties and produce higher levels of pro-inflammatory cytokines. Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef] [PubMed]





| PCD | Healthy Adult Controls (Ad CO) | Healthy Pediatric Controls (Ped CO) | |
|---|---|---|---|
| Female/Male No. | 21/27 (44%/56%) | 16/5 (76%/24%) | 23/10 (70%/30%) |
| Age, median (range) in years | 14 (3–69) | 27.0 (23–44) | 11.1 (4–18) |
| Pediatric/Adult No. | 32/16 (67%/33%) | 0/21 (0%/100%) | 33/0 (100%/0%) |
| Ultrastructural defect | |||
| Normal ultrastructure | 20/48 (42%) | ||
| ODA deficiency | 20/48 (42%) | ||
| IDA deficiency + CP abnormalities | 3/48 (6%) | ||
| CP deficiency | 4/48 (8%) | ||
| Ciliary aplasia | 1/48 (2%) | ||
| Genetic abnormalities | |||
| Unknown | 8/48 (17%) | ||
| DNAH11 | 13/48 (27%) | ||
| DNAH5 | 11/48(23%) | ||
| DNAAF1 | 3/48 (6%) | ||
| SPAG1 | 2/48 (4%) | ||
| CCDC39 | 1/48 (2%) | ||
| CCDC40 | 2/48 (4%) | ||
| CCDC65 | 1/48 (2%) | ||
| CCDC103 | 2/48 (4%) | ||
| HYDIN | 1/48 (2%) | ||
| RSPH1 | 1/48 (2%) | ||
| RSPH4 | 1/48 (2%) | ||
| RSPH9 | 1/48 (2%) | ||
| OFD1 | 1/48 (2%) | ||
| Clinical features | |||
| Bronchiectasis | 33/48 (69%) | ||
| Situs inversus | 17/48 (35%) | ||
| FEV1, median (range) (%) | 92 (43–113) | ||
| FVC, median (range) (%) | 100 (76–125) | ||
| Laboratory features, median (range) | |||
| WBC count (/µL) | 7415 (3900–17,240) | ||
| CRP (mg/dL) | 1.1 (<0.3–10.2) | ||
| Sputum bacteria | |||
| H. influenzae | 10/48 (21%) | ||
| P. aeruginosa | 7/48 (15%) | ||
| S. aureus | 10/48 (21%) | ||
| S. pneumoniae | 4/48 (8%) | ||
| Treatment | |||
| Azithromycin | 6/48 (13%) | ||
| Oral antibiotics | 3/48 (6%) | ||
| Inhaled antibiotics | 2/48 (4%) | ||
| Inhaled steroids | 1/48 (2%) | ||
| Neutrophil Function | CF | COPD | Asthma | PCD |
|---|---|---|---|---|
| Phagocytosis | =[46] | =/↓[48] | ↑[25,49] | = |
| ROS production | ≠[27,50] | ↑[43] | =/↑[25,45,51] | ↓ |
| NET formation | ↑[24,47] | NA | NA | ↓ |
| CXCL8 production | ↑[52,53] | NA | =/↑[29,54] | ↓ |
| Chemotaxis | ≠[55,56,57] | ↑[28] | ↑[49] | ↓ |
| Adhesion molecules | =[41,42] | ↑[43,44] | ↑[49] | = |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cockx, M.; Blanter, M.; Gouwy, M.; Ruytinx, P.; Abouelasrar Salama, S.; Knoops, S.; Pörtner, N.; Vanbrabant, L.; Lorent, N.; Boon, M.; et al. The Antimicrobial Activity of Peripheral Blood Neutrophils Is Altered in Patients with Primary Ciliary Dyskinesia. Int. J. Mol. Sci. 2021, 22, 6172. https://doi.org/10.3390/ijms22126172
Cockx M, Blanter M, Gouwy M, Ruytinx P, Abouelasrar Salama S, Knoops S, Pörtner N, Vanbrabant L, Lorent N, Boon M, et al. The Antimicrobial Activity of Peripheral Blood Neutrophils Is Altered in Patients with Primary Ciliary Dyskinesia. International Journal of Molecular Sciences. 2021; 22(12):6172. https://doi.org/10.3390/ijms22126172
Chicago/Turabian StyleCockx, Maaike, Marfa Blanter, Mieke Gouwy, Pieter Ruytinx, Sara Abouelasrar Salama, Sofie Knoops, Noëmie Pörtner, Lotte Vanbrabant, Natalie Lorent, Mieke Boon, and et al. 2021. "The Antimicrobial Activity of Peripheral Blood Neutrophils Is Altered in Patients with Primary Ciliary Dyskinesia" International Journal of Molecular Sciences 22, no. 12: 6172. https://doi.org/10.3390/ijms22126172
APA StyleCockx, M., Blanter, M., Gouwy, M., Ruytinx, P., Abouelasrar Salama, S., Knoops, S., Pörtner, N., Vanbrabant, L., Lorent, N., Boon, M., & Struyf, S. (2021). The Antimicrobial Activity of Peripheral Blood Neutrophils Is Altered in Patients with Primary Ciliary Dyskinesia. International Journal of Molecular Sciences, 22(12), 6172. https://doi.org/10.3390/ijms22126172






