A Glimpse into the Structural Properties of the Intermediate and Transition State in the Folding of Bromodomain 2 Domain 2 by Φ Value Analysis
Abstract
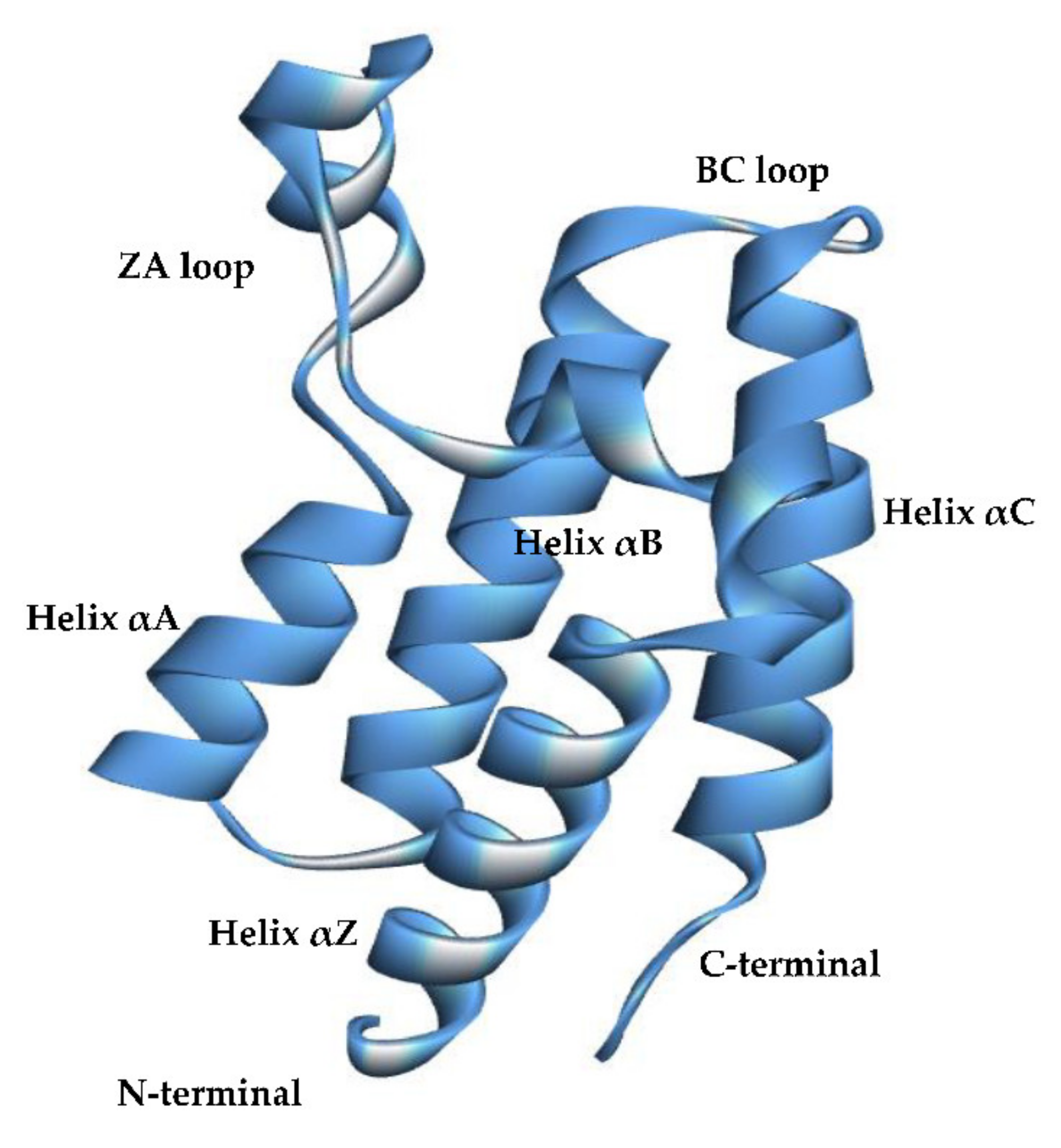
1. Introduction
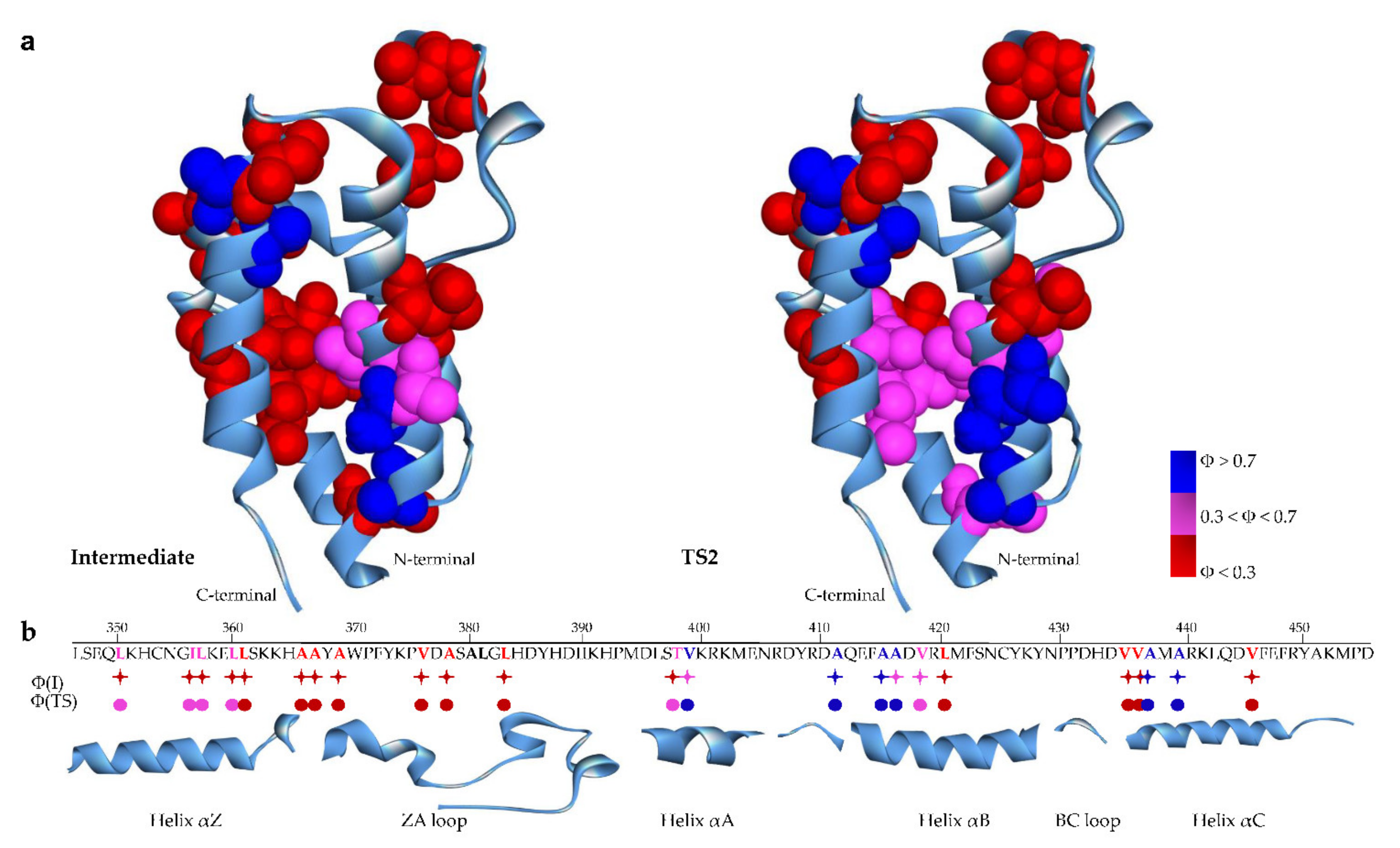
2. Results and Discussion
2.1. Urea-Induced Equilibrium Unfolding
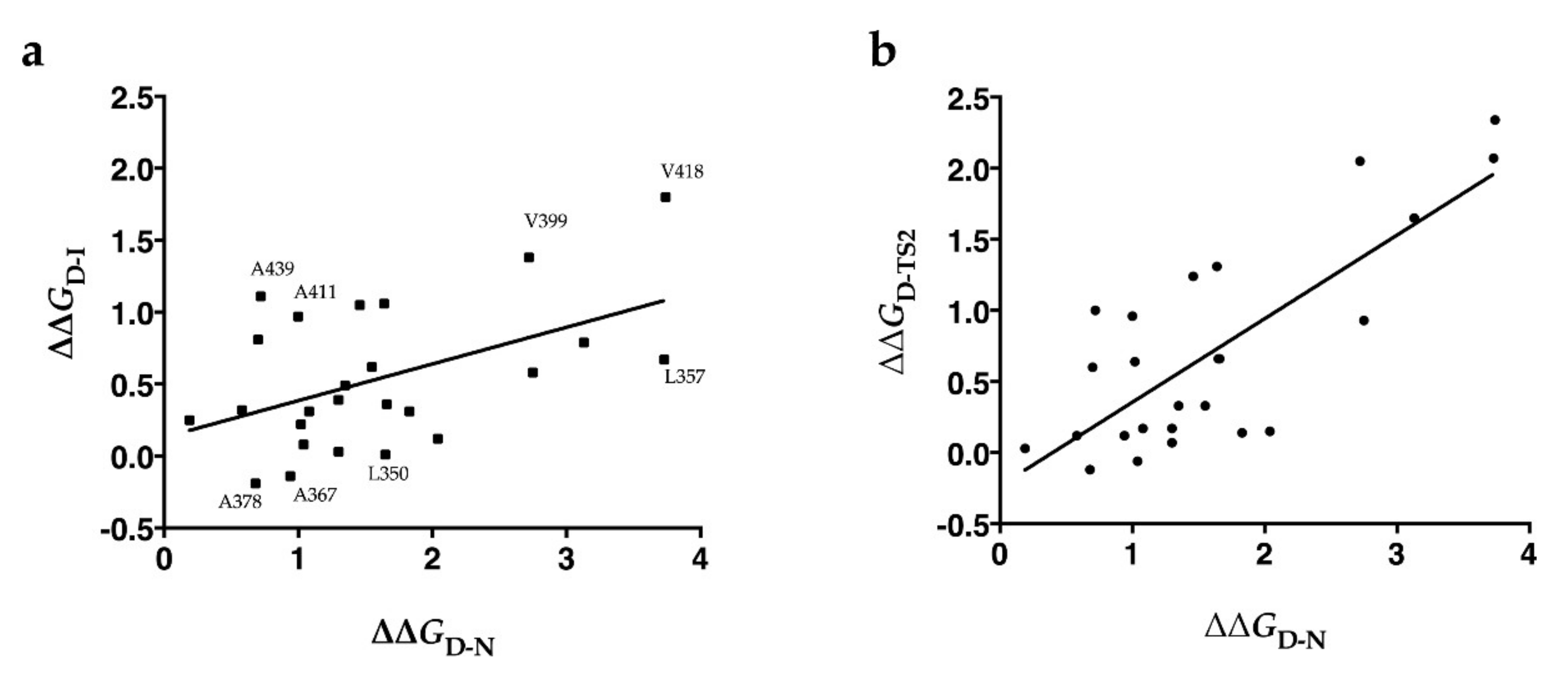
2.2. Folding Kinetics
3. Conclusions
4. Materials and Methods
4.1. Site-Directed Mutagenesis
4.2. Protein Expression and Purification
4.3. Equilibrium Experiments
4.4. Kinetic Experiments
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Gianni, S.; Ivarsson, Y.; Jemth, P.; Brunori, M.; Travaglini-Allocatelli, C. Identification and characterization of protein folding intermediates. Biophys. Chem. 2007, 128, 105–113. [Google Scholar] [CrossRef]
- Dill, K.A.; Maccallum, J.L. The Protein-Folding Problem, 50 Years On. Science 2012, 338, 1042–1046. [Google Scholar] [CrossRef]
- Dawson, M.A.; Kouzarides, T.; Huntly, B.J.P. Targeting Epigenetic Readers in Cancer. N. Engl. J. Med. 2012, 367, 647–657. [Google Scholar] [CrossRef]
- Muller, S.; Filippakopoulos, P.; Knapp, S. Bromodomains as therapeutic targets. Expert Rev. Mol. Med. 2011, 13, e29. [Google Scholar] [CrossRef]
- Qiao, Y.; Wang, X.-M.; Mannan, R.; Pitchiaya, S.; Zhang, Y.; Wotring, J.W.; Xiao, L.; Robinson, D.R.; Wu, Y.-M.; Tien, J.C.-Y.; et al. Targeting transcriptional regulation of SARS-CoV-2 entry factors ACE2 and TMPRSS2. Proc. Natl. Acad. Sci. USA 2021, 118, e2021450118. [Google Scholar] [CrossRef] [PubMed]
- Gordon, D.E.; Jang, G.M.; Bouhaddou, M.; Xu, J.; Obernier, K.; White, K.M.; O’Meara, M.J.; Rezelj, V.V.; Guo, J.Z.; Swaney, D.L.; et al. A SARS-CoV-2 protein interaction map reveals targets for drug repurposing. Nature 2020, 583, 459–468. [Google Scholar] [CrossRef] [PubMed]
- Ferri, E.; Petosa, C.; McKenna, C.E. Bromodomains: Structure, function and pharmacology of inhibition. Biochem. Pharmacol. 2016, 106, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Josling, G.A.; Selvarajah, S.A.; Petter, M.; Duffy, M.F. The Role of Bromodomain Proteins in Regulating Gene Expression. Genes 2012, 3, 320–343. [Google Scholar] [CrossRef]
- Zeng, L.; Zhang, Q.; Gerona-Navarro, G.; Moshkina, N.; Zhou, M.-M. Structural Basis of Site-Specific Histone Recognition by the Bromodomains of Human Coactivators PCAF and CBP/p300. Structure 2008, 16, 643–652. [Google Scholar] [CrossRef] [PubMed]
- Mujtaba, S.; Zeng, L.; Zhou, M.-M. Structure and acetyl-lysine recognition of the bromodomain. Oncogene 2007, 26, 5521–5527. [Google Scholar] [CrossRef]
- Magno, A.; Steiner, S.; Caflisch, A. Mechanism and Kinetics of Acetyl-Lysine Binding to Bromodomains. J. Chem. Theory Comput. 2013, 9, 4225–4232. [Google Scholar] [CrossRef] [PubMed]
- Langini, C.; Caflisch, A.; Vitalis, A. The ATAD2 Bromodomain Binds Different Acetylation Marks on the Histone H4 in Similar Fuzzy Complexes. J. Biol. Chem. 2017, 292, 16734–16745. [Google Scholar] [CrossRef]
- Petrosino, M.; Bonetti, D.; Pasquo, A.; Lori, L.; Chiaraluce, R.; Consalvi, V.; Travaglini-Allocatelli, C. Unveiling the folding mechanism of the Bromodomains. Biochem. Biophys. Rep. 2017, 11, 99–104. [Google Scholar] [CrossRef]
- Fersht, A.R.; Sato, S. Φ value analysis and the nature of protein-folding transition states. Proc. Natl. Acad. Sci. USA 2004, 101, 7976–7981. [Google Scholar] [CrossRef] [PubMed]
- Serrano, L.; Matouschek, A.; Fersht, A.R. The folding of an enzyme: III. Structure of the transition state for unfolding of barnase analysed by a protein engineering procedure. J. Mol. Biol. 1992, 224, 805–818. [Google Scholar] [CrossRef]
- Fersht, A. Structure and Mechanism in Protein Science: A Guide to Enzyme Catalysis and Protein Folding; Freeman WH and Co.: New York, NY, USA, 1999. [Google Scholar]
- Tanford, C. Protein denaturation. C. Theoretical models for the mechanism of denaturation. Adv. Protein. Chem. 1970, 24, 1–95. [Google Scholar]
- Friel, C.T.; Smith, D.A.; Vendruscolo, M.; Gsponer, J.; Radford, S.E. The mechanism of folding of Im7 reveals competition between functional and kinetic evolutionary constraints. Nat. Struct. Mol. Biol. 2009, 16, 318–324. [Google Scholar] [CrossRef]
- Baxa, M.C.; Yu, W.; Adhikari, A.N.; Ge, L.; Xia, Z.; Zhou, R.; Freed, K.F.; Sosnick, T.R. Even with nonnative interactions, the updated folding transition states of the homologs Proteins G & L are extensive and similar. Proc. Natl. Acad. Sci. USA 2015, 112, 8302–8307. [Google Scholar] [CrossRef]
- Gianni, S.; Freiberger, M.I.; Jemth, P.; Ferreiro, D.U.; Wolynes, P.G.; Fuxreiter, M. Fuzziness and Frustration in the Energy Landscape of Protein Folding, Function, and Assembly. Accounts Chem. Res. 2021, 54, 1251–1259. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Unzue, A.; Dong, J.; Spiliotopoulos, D.; Nevado, C.; Caflisch, A. Discovery of CREBBP Bromodomain Inhibitors by High-Throughput Docking and Hit Optimization Guided by Molecular Dynamics. J. Med. Chem. 2015, 59, 1340–1349. [Google Scholar] [CrossRef]
- Karplus, M.; Weaver, D.L. Protein folding dynamics: The diffusion-collision model and experimental data. Protein Sci. 1994, 3, 650–668. [Google Scholar] [CrossRef]
- Gianni, S.; Geierhaas, C.D.; Calosci, N.; Jemth, P.; Vuister, G.; Travaglini-Allocatelli, C.; Vendruscolo, M.; Brunori, M. A PDZ domain recapitulates a unifying mechanism for protein folding. Proc. Natl. Acad. Sci. USA 2007, 104, 128–133. [Google Scholar] [CrossRef]
- Onuchic, J.N.; Wolynes, P.G. Theory of protein folding. Curr. Opin. Struct. Biol. 2004, 14, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Lori, L.; Pasquo, A.; Lori, C.; Petrosino, M.; Chiaraluce, R.; Tallant, C.; Knapp, S.; Consalvi, V. Effect of BET Missense Mutations on Bromodomain Function, Inhibitor Binding and Stability. PLoS ONE 2016, 11, e0159180. [Google Scholar] [CrossRef] [PubMed]
- Gill, S.C.; von Hippel, P.H. Calculation of protein extinction coefficients from amino acid sequence data. Anal. Biochem. 1989, 182, 319–326. [Google Scholar] [CrossRef]
- Santoro, M.M.; Bolen, D.W. Unfolding Free Energy Changes Determined by the Linear Extrapolation Method. 1. Un-folding of Phenylmethanesulfonyl. Alpha.-Chymotrypsin Using Different Denaturants. Biochemistry 1988, 27, 8063–8068. [Google Scholar] [CrossRef] [PubMed]
- Parker, M.J.; Spencer, J.; Clarke, A.R. An Integrated Kinetic Analysis of Intermediates and Transition States in Protein Folding Reactions. J. Mol. Biol. 1995, 253, 771–786. [Google Scholar] [CrossRef] [PubMed]

| Protein | ΔGH2O (kcal/mol) | [Urea]0.5 |
|---|---|---|
| wild type | 8.94 ± 0.06 | 4.63 |
| L350A | 6.93 ± 0.06 | 3.59 |
| I356V | 8.24 ± 0.09 | 4.27 |
| L357A | 5.16 ± 0.09 | 2.67 |
| L360V | 6.37 ± 0.08 | 3.30 |
| L361A | 6.09 ± 0.05 | 3.15 |
| A366G | 8.31 ± 0.08 | 4.30 |
| A367G | 8.19 ± 0.06 | 4.24 |
| A369G | 7.88 ± 0.10 | 4.08 |
| V376A | 7.37 ± 0.08 | 3.82 |
| A378G | 8.49 ± 0.07 | 4.40 |
| A380G | 8.75 ± 0.10 | 4.53 |
| L381A | 9.23 ± 0.09 | 4.78 |
| L383A | 8.50 ± 0.08 | 4.40 |
| T398S | 7.57 ± 0.07 | 3.92 |
| V399A | 6.69 ± 0.07 | 3.47 |
| A411G | 8.44 ± 0.05 | 4.37 |
| A415G | 7.60 ± 0.11 | 3.94 |
| A416G | 7.77 ± 0.10 | 4.02 |
| V418A | 5.44 ± 0.04 | 2.82 |
| L420A | 6.97 ± 0.08 | 3.61 |
| V435A | 7.31 ± 0.08 | 3.79 |
| V436A | 7.81 ± 0.10 | 4.05 |
| A437G | 8.56 ± 0.11 | 4.43 |
| A439G | 7.54 ± 0.12 | 3.91 |
| V445A | 8.00 ± 0.08 | 4.14 |
| kI-N (s−1) | kN-I (s−1) | KD-I | ∆GD-N (kcal/mol) | ∆∆GD-N (kcal/mol) | ∆∆GTS2-N (kcal/mol) | ∆∆GD-I (kcal/mol) | Φ (I) | Φ (TS2) | |
|---|---|---|---|---|---|---|---|---|---|
| wild type | 362 ± 32 | 7.44 × 10−4 ± 4.61 × 10−5 | 21 ± 2.7 | 9.37 ± 0.10 | |||||
| L350A | 118 ± 13 | 4.13 × 10−3 ± 2.59 × 10−4 | 21 ± 3.1 | 7.72 ± 0.11 | 1.65 ± 0.15 | 0.99 ± 0.05 | 0.01 ± 0.11 | 0.01 ± 0.11 | 0.40 ± 0.04 |
| I356V | 176± 21 | 1.43 × 10−3 ± 1.01 × 10−4 | 15 ± 2.5 | 8.36 ± 0.13 | 1.02 ± 0.16 | 0.38 ± 0.05 | 0.22 ± 0.12 | 0.22 ± 0.13 | 0.63 ± 0.13 |
| L357A | 32 ± 4.5 | 1.30 × 10−2 ± 7.01 × 10−4 | 6.7 ± 1.4 | 5.64 ± 0.14 | 3.73 ± 0.17 | 1.66 ± 0.05 | 0.67 ± 0.14 | 0.18 ± 0.04 | 0.55 ± 0.03 |
| L360V | 81 ± 14 | 9.53 × 10−3 ± 5.93 × 10−4 | 5.5 ± 1.4 | 6.24 ± 0.19 | 3.13 ± 0.21 | 1.48 ± 0.05 | 0.79 ± 0.17 | 0.25 ± 0.06 | 0.53 ± 0.04 |
| L361A | 200 ± 30 | 1.71 × 10−2 ± 1.09 × 10−3 | 7.8 ± 1.7 | 6.63 ± 0.16 | 2.75 ± 0.19 | 1.82 ± 0.05 | 0.58 ± 0.15 | 0.21 ± 0.06 | 0.34 ± 0.03 |
| A366G | 458 ± 54 | 4.92 × 10−3 ± 3.46 × 10−4 | 19 ± 3.1 | 8.33 ± 0.12 | 1.04 ± 0.16 | 1.10 ± 0.05 | 0.08 ± 0.12 | 0.08 ± 0.12 | −0.05 ± 0.01 |
| A367G | 232 ± 26 | 3.07 × 10−3 ± 2.18 × 10−4 | 27 ± 3.5 | 8.43 ± 0.12 | 0.94 ± 0.16 | 0.82 ± 0.05 | −0.14 ± 0.12 | −0.15 ± 0.13 | 0.12 ± 0.02 |
| A369G | 340 ± 30 | 6.22 × 10−3 ± 4.02 × 10−4 | 20 ± 3.1 | 8.08 ± 0.12 | 1.30 ± 0.15 | 1.23 ± 0.05 | 0.03 ± 0.12 | 0.02 ± 0.08 | 0.05 ± 0.01 |
| V376A | 496 ± 42 | 1.38 × 10−2 ± 9.25 × 10−4 | 12 ± 2.3 | 7.54 ± 0.13 | 1.83 ± 0.16 | 1.69 ± 0.05 | 0.31 ± 0.13 | 0.17 ± 0.07 | 0.07 ± 0.01 |
| A378G | 319 ± 32 | 2.97 × 10−3 ± 1.95 × 10−4 | 30 ± 4.1 | 8.69 ± 0.11 | 0.68 ± 0.14 | 0.80 ± 0.05 | −0.19 ± 0.11 | −0.28 ± 0.17 | −0.17 ± 0.04 |
| A380G | 502 ± 51 | 1.63 × 10−3 ± 1.19 × 10−4 | 12 ± 2.2 | 8.79 ± 0.13 | 0.58 ± 0.16 | 0.46 ± 0.06 | 0.32 ± 0.13 | 0.54 a ± 0.07 | 0.22 a ± 0.26 |
| L381A | 537 ± 56 | 9.83 × 10−4 ± 7.35 × 10−5 | 14 ± 2.4 | 9.19 ± 0.13 | 0.19 ± 0.16 | 0.16 ± 0.06 | 0.25 ± 0.12 | 1.36 a ± 0.12 | 0.13 a ± 1.28 |
| L383A | 461 ± 34 | 3.57 × 10−3 ± 2.23 × 10−4 | 12 ± 1.5 | 8.29 ± 0.10 | 1.08 ± 0.14 | 0.91 ± 0.05 | 0.31 ± 0.10 | 0.29 ± 0.05 | 0.16 ± 0.02 |
| T398S | 215 ± 27 | 4.18 × 10−3 ± 2.83 × 10−4 | 11 ± 2.2 | 7.71 ± 0.14 | 1.66 ± 0.17 | 1.00 ± 0.05 | 0.36 ± 0.13 | 0.21 ± 0.08 | 0.45 ± 0.05 |
| V399A | 114 ± 36 | 2.37 × 10−3 ± 1.51 × 10−4 | 1.9 ± 0.8 | 6.65 ± 0.11 | 2.72 ± 0.15 | 0.67 ± 0.05 | 1.38 ± 0.10 | 0.51 ± 0.05 | 0.75 ± 0.07 |
| A411G | 373 ± 30 | 7.97 × 10−4 ± 5.64 × 10−5 | 3.9 ± 0.7 | 8.37 ± 0.08 | 1.00 ± 0.12 | 0.04 * ± 0.05 | 0.97 ± 0.09 | 0.98 ± 0.15 | 0.96 ± 1.19 |
| A415G | 260 ± 24 | 1.08 × 10−3 ± 7.43 × 10−5 | 3.4 ± 0.7 | 7.91 ± 0.10 | 1.46 ± 0.14 | 0.22 ± 0.04 | 1.05 ± 0.11 | 0.72 ± 0.10 | 0.85 ± 0.16 |
| A416G | 237 ± 19 | 1.32 × 10−3 ± 8.96 × 10−5 | 3.4 ± 0.7 | 7.73 ± 0.10 | 1.64 ± 0.14 | 0.33 ± 0.05 | 1.06 ± 0.11 | 0.65 ± 0.09 | 0.80 ± 0.15 |
| V418A | 143 ± 13 | 8.38 × 10−3 ± 3.05 × 10−4 | 1.0 ± 0.4 | 5.63 ± 0.17 | 3.74 ± 0.19 | 1.40 ± 0.05 | 1.80 ± 0.17 | 0.48 ± 0.05 | 0.62 ± 0.04 |
| L420A | 348 ± 41 | 1.93 × 10−2 ± 1.28 × 10−3 | 17 ± 3.1 | 7.33 ± 0.13 | 2.04 ± 0.16 | 1.89 ± 0.05 | 0.12 ± 0.13 | 0.06 ± 0.06 | 0.07 ± 0.01 |
| V435A | 594 ± 56 | 6.07 × 10−3 ± 4.14 × 10−4 | 7.3 ± 1.6 | 7.82 ± 0.15 | 1.55 ± 0.18 | 1.22 ± 0.05 | 0.62 ± 0.14 | 0.40 ± 0.10 | 0.21 ± 0.03 |
| V436A | 479 ± 48 | 4.31 × 10−3 ± 2.66E × 10−4 | 9.2 ± 1.4 | 8.03 ± 0.11 | 1.35 ± 0.15 | 1.02 ± 0.05 | 0.49 ± 0.12 | 0.36 ± 0.09 | 0.24 ± 0.03 |
| A437G | 519 ± 35 | 8.83 × 10−4 ± 6.38 × 10−5 | 5.2 ± 1.2 | 8.67 ± 0.07 | 0.70 ± 0.12 | 0.10 ± 0.02 | 0.81 ± 0.10 | 1.16 ± 0.24 | 0.86 ± 0.17 |
| A439G | 436 ± 42 | 4.60 × 10−4 ± 3.33 × 10−5 | 3.2 ± 0.7 | 8.65 ± 0.11 | 0.72 ± 0.15 | −0.28 ± 0.06 | 1.11 ± 0.11 | 1.54 ± 0.35 | 1.39 ± 0.39 |
| V445A | 527 ± 51 | 5.21 × 10−3 ± 3.23 × 10−4 | 11 ± 1.6 | 8.07 ± 0.11 | 1.30 ± 0.15 | 1.13 ± 0.05 | 0.39 ± 0.11 | 0.30 ± 0.09 | 0.13 ± 0.02 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Novak, L.; Petrosino, M.; Santorelli, D.; Chiaraluce, R.; Consalvi, V.; Pasquo, A.; Travaglini-Allocatelli, C. A Glimpse into the Structural Properties of the Intermediate and Transition State in the Folding of Bromodomain 2 Domain 2 by Φ Value Analysis. Int. J. Mol. Sci. 2021, 22, 5953. https://doi.org/10.3390/ijms22115953
Novak L, Petrosino M, Santorelli D, Chiaraluce R, Consalvi V, Pasquo A, Travaglini-Allocatelli C. A Glimpse into the Structural Properties of the Intermediate and Transition State in the Folding of Bromodomain 2 Domain 2 by Φ Value Analysis. International Journal of Molecular Sciences. 2021; 22(11):5953. https://doi.org/10.3390/ijms22115953
Chicago/Turabian StyleNovak, Leonore, Maria Petrosino, Daniele Santorelli, Roberta Chiaraluce, Valerio Consalvi, Alessandra Pasquo, and Carlo Travaglini-Allocatelli. 2021. "A Glimpse into the Structural Properties of the Intermediate and Transition State in the Folding of Bromodomain 2 Domain 2 by Φ Value Analysis" International Journal of Molecular Sciences 22, no. 11: 5953. https://doi.org/10.3390/ijms22115953
APA StyleNovak, L., Petrosino, M., Santorelli, D., Chiaraluce, R., Consalvi, V., Pasquo, A., & Travaglini-Allocatelli, C. (2021). A Glimpse into the Structural Properties of the Intermediate and Transition State in the Folding of Bromodomain 2 Domain 2 by Φ Value Analysis. International Journal of Molecular Sciences, 22(11), 5953. https://doi.org/10.3390/ijms22115953






