Biodegradable Stent with mTOR Inhibitor-Eluting Reduces Progression of Ureteral Stricture
Abstract
1. Introduction
2. Results
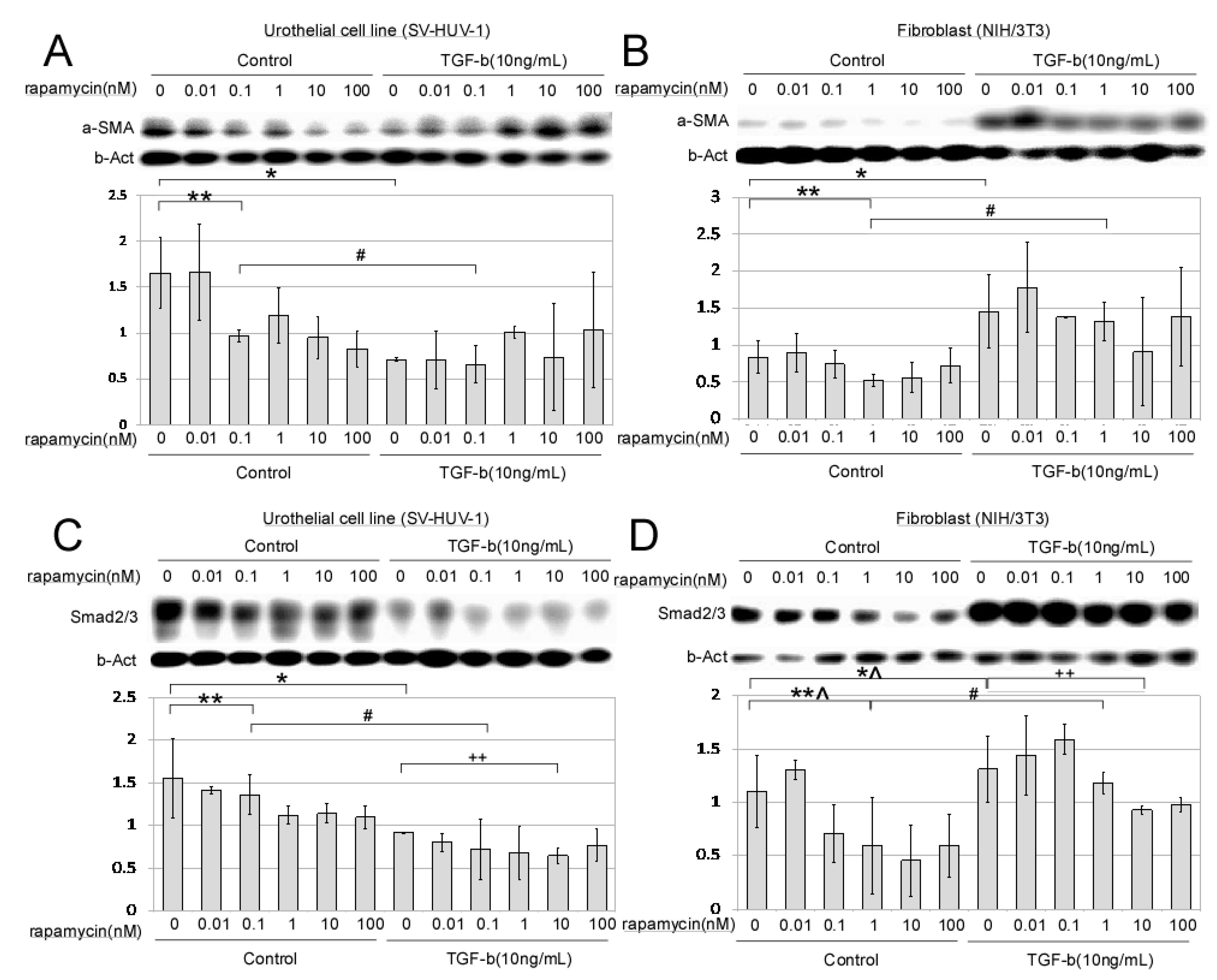
2.1. In Vitro Dose-Dependent Effect of mTORi on Fibroblast and Urothelial Cell Line
2.2. In Vivo Rabbit Segmental Ureteral Thermal Injury Model
2.2.1. Schematic Procedure and Gross Anatomy Changes in Upper Urinary Tract of Animal Model
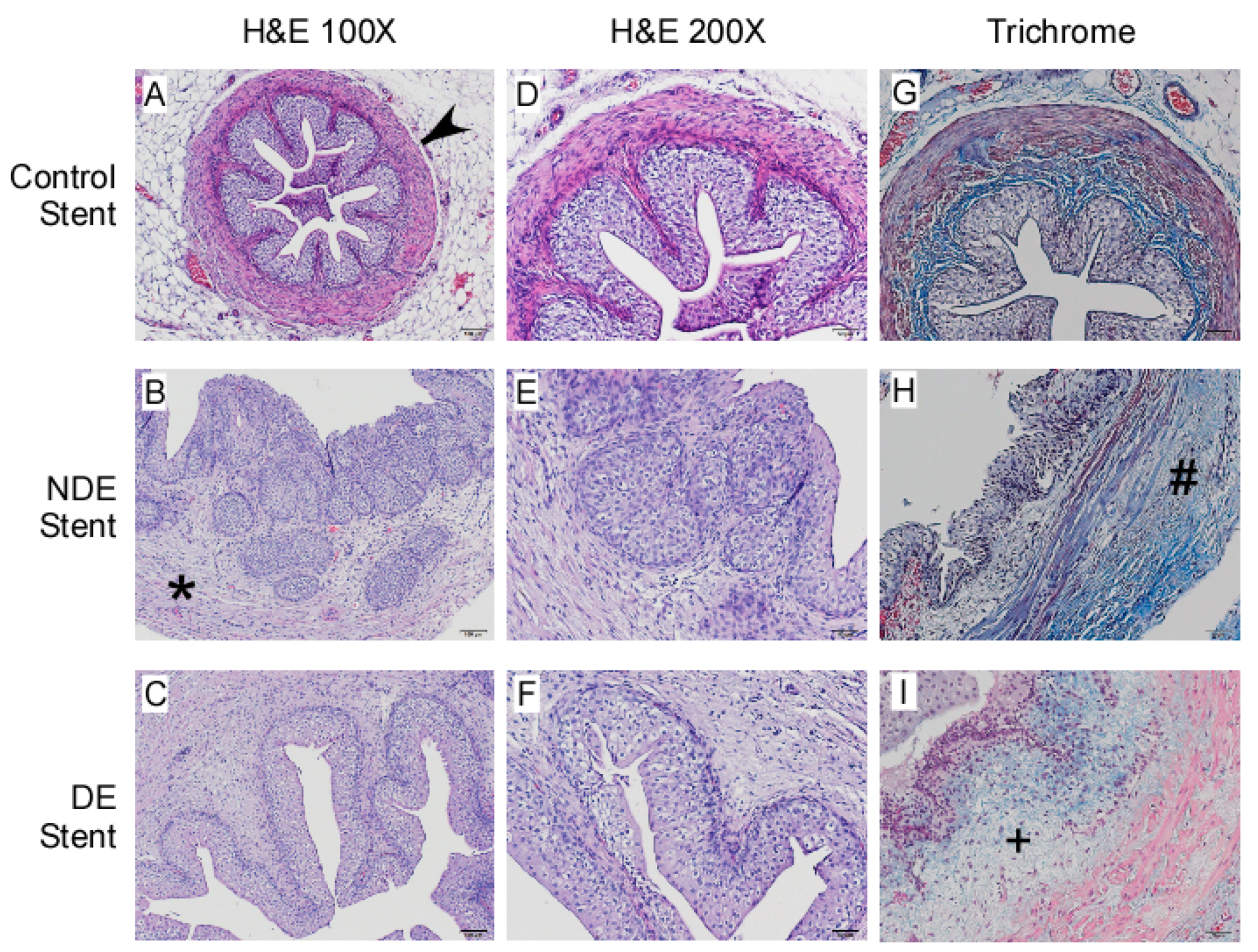
2.2.2. DE Stent Alleviate Fibrosis in Histopathology Examination
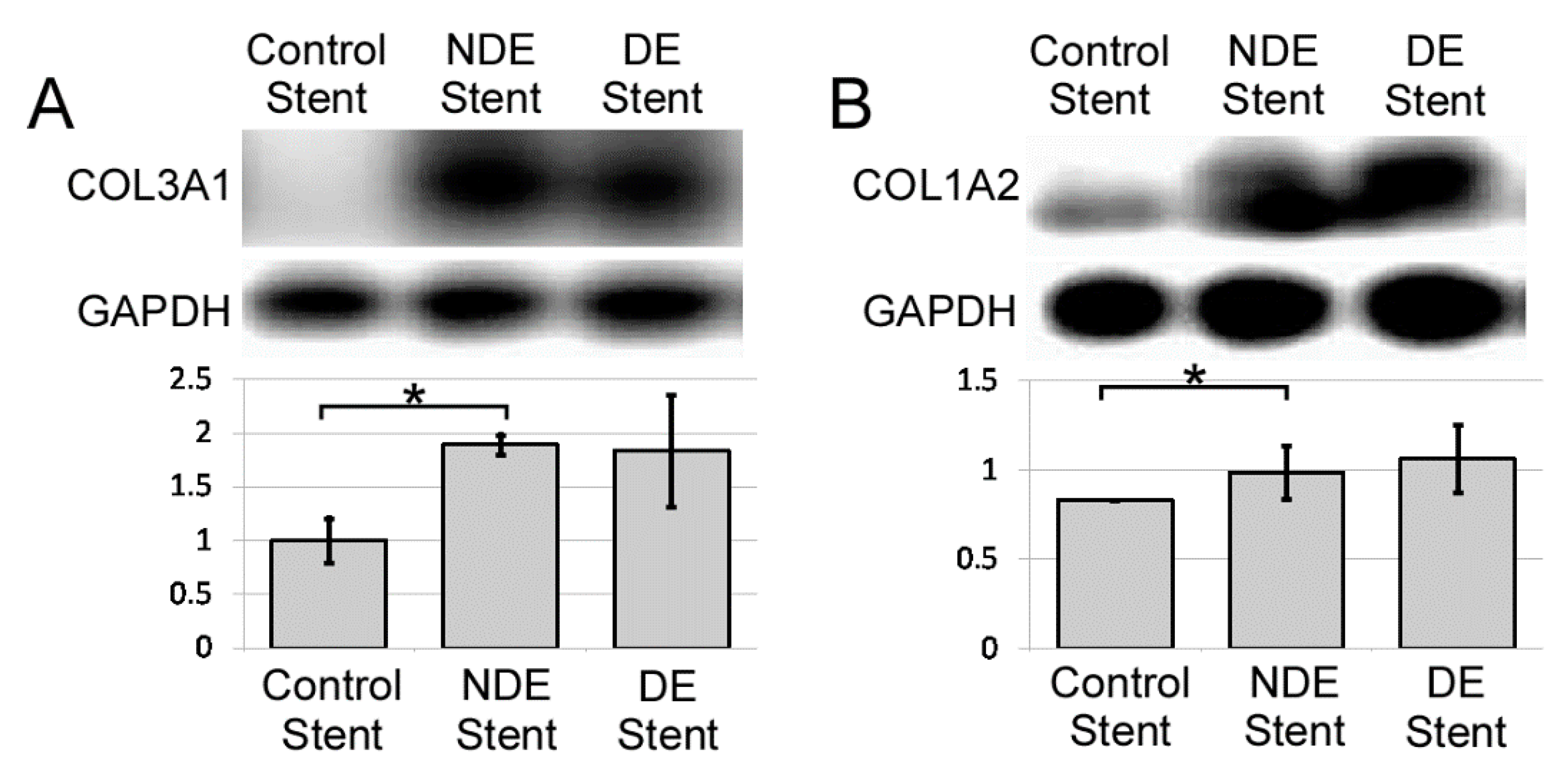
2.2.3. Thermal Injury Promotes Type I and III Collagen Expression in Ureter 4 Weeks after Treatment
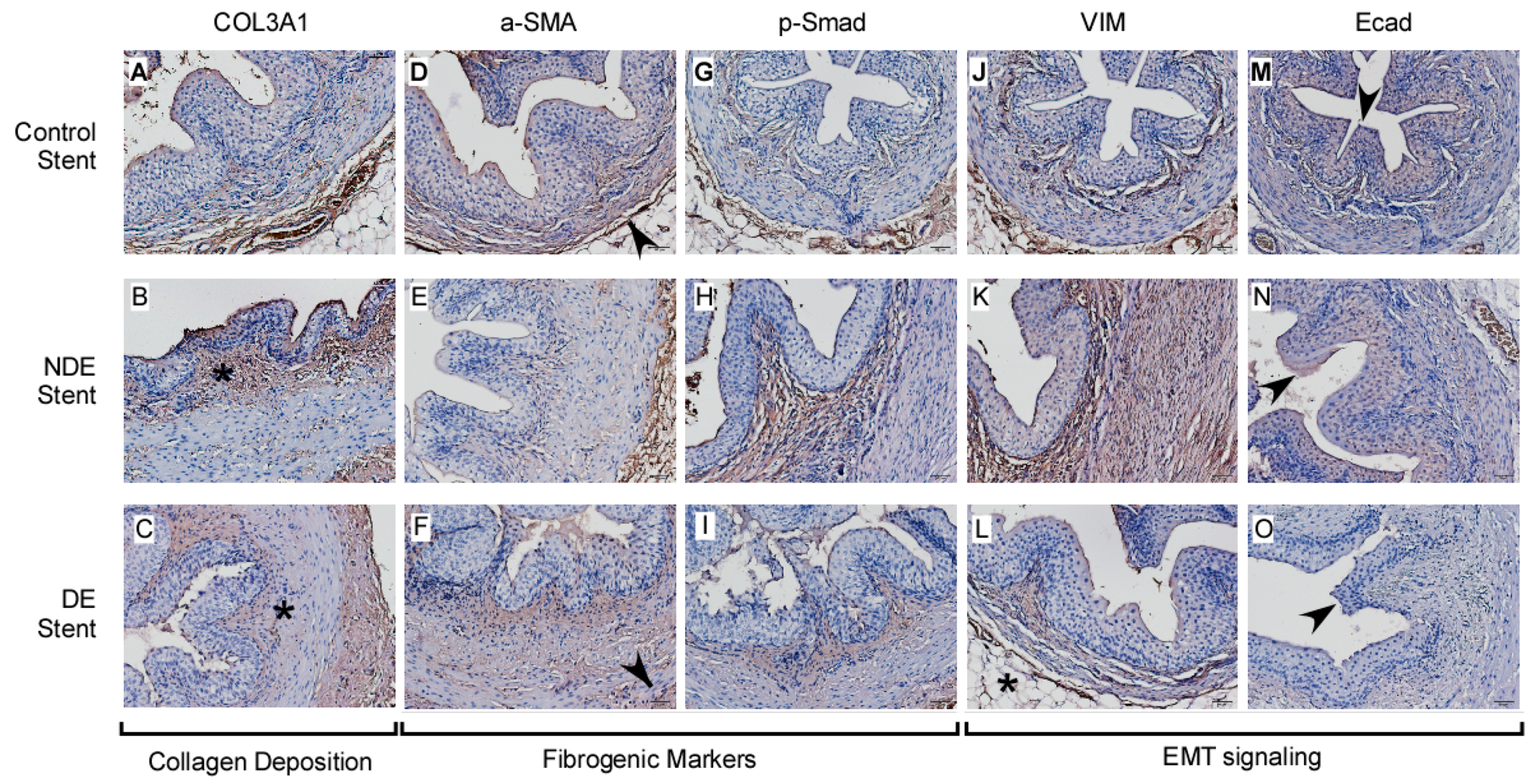
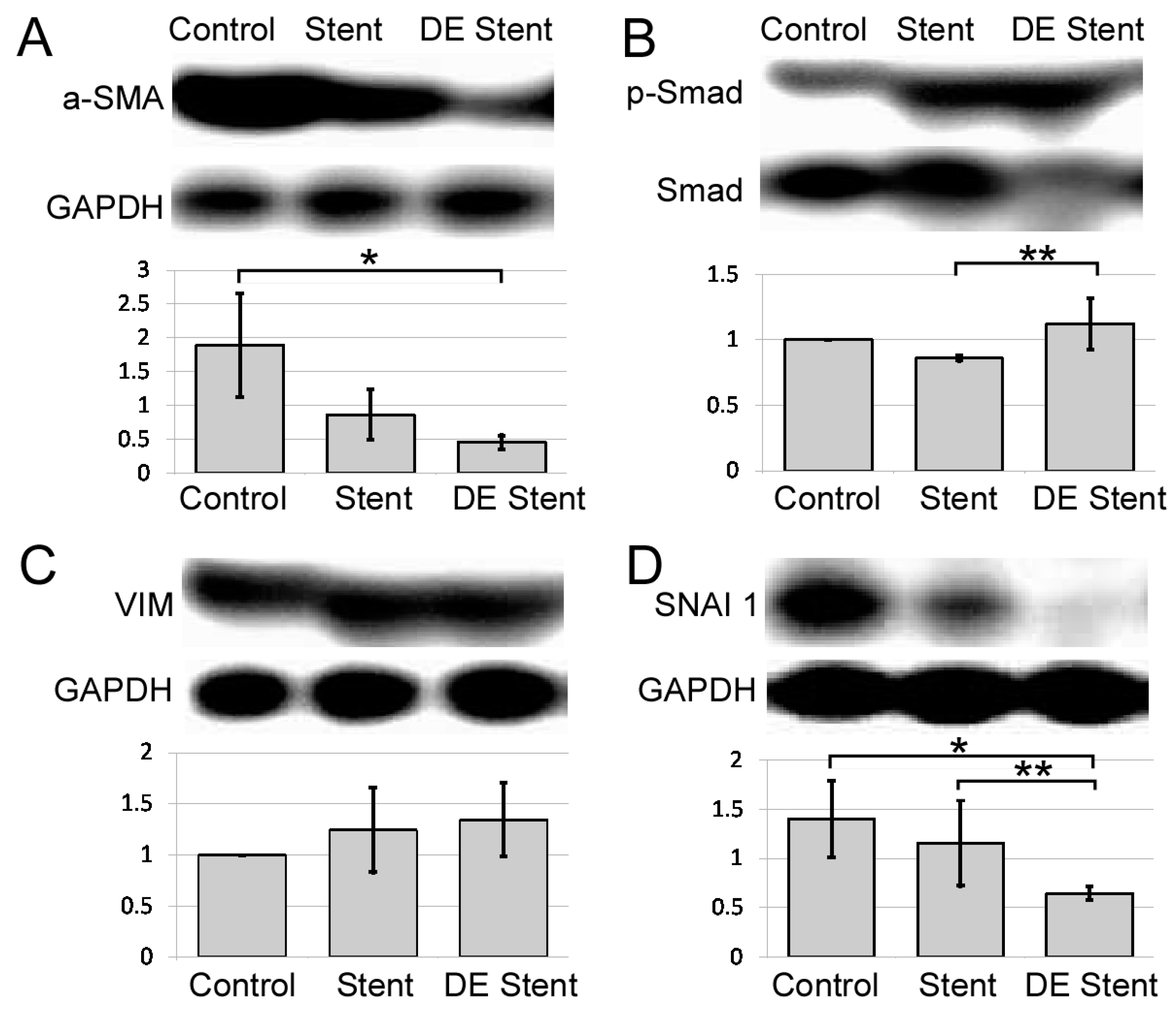
2.2.4. Eluted mTOR Inhibitor Downregulate Smooth Muscle Actin and Upregulate Epithelial-Mesenchymal Transition
2.2.5. Semi-Quantitative Analysis
3. Discussion
4. Materials and Methods
4.1. Cell Line
4.2. Drug-Eluting Biodegradable Stent
4.3. Animal Study
4.4. Histological Analysis
4.5. Western Blot Analysis
4.6. Statistics
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rotariu, P.; Yohannes, P.; Alexianu, M.; Rosner, D.; Lee, B.R.; Lucan, M.; Smith, A.D. Management of malignant extrinsic compression of the ureter by simultaneous placement of two ipsilateral ureteral stents. J. Endourol. 2001, 15, 979–983. [Google Scholar] [CrossRef] [PubMed]
- Demanes, D.J.; Banerjee, R.; Cahan, B.L.; Lee, S.P.; Park, S.J.; Fallon, J.M.; Reyes, P.; Van, T.Q.; Steinberg, M.L.; Kamrava, M.R. Ureteral stent insertion for gynecologic interstitial high-dose-rate brachytherapy. Brachytherapy 2015, 14, 245–251. [Google Scholar] [CrossRef]
- Lacquet, J.P.; Lacroix, H.; Nevelsteen, A.; Suy, R. Inflammatory abdominal aortic aneurysms. A retrospective study of 110 cases. Acta Chir. Belg. 1997, 97, 286–292. [Google Scholar] [PubMed]
- Roberts, W.W.; Cadeddu, J.A.; Micali, S.; Kavoussi, L.R.; Moore, R.G. Ureteral stricture formation after removal of impacted calculi. J. Urol. 1998, 159, 723–726. [Google Scholar] [CrossRef]
- Delvecchio, F.C.; Auge, B.K.; Brizuela, R.M.; Weizer, A.Z.; Silverstein, A.D.; Lallas, C.D.; Pietrow, P.K.; Albala, D.M.; Preminger, G.M. Assessment of stricture formation with the ureteral access sheath. Urology 2003, 61, 518–522, discussion 522. [Google Scholar] [CrossRef]
- dStern, K.L.; Loftus, C.J.; Doizi, S.; Traxer, O.; Monga, M. A prospective study analyzing the association between high-grade ureteral access sheath injuries and the formation of ureteral strictures. Urology 2019, 128, 38–41. [Google Scholar] [CrossRef] [PubMed]
- Vasudevan, V.P.; Johnson, E.U.; Wong, K.; Iskander, M.; Javed, S.; Gupta, N.; McCabe, J.E.; Kavoussi, L. Contemporary management of ureteral strictures. J. Clin. Urol. 2019, 12, 20–31. [Google Scholar] [CrossRef]
- Wang, Q.; Lu, Y.; Hu, H.; Zhang, J.; Qin, B.; Zhu, J.; Dirie, N.I.; Zhang, Z.; Wang, S. Management of recurrent ureteral stricture: A retrospectively comparative study with robot-assisted laparoscopic surgery versus open approach. PeerJ 2019, 7, e8166. [Google Scholar] [CrossRef] [PubMed]
- Derrick, F.C., Jr.; Lynch, K.M., Jr.; Price, R., Jr.; Turner, W.R., Jr. Transureteroureterostomy. JAMA 1967, 200, 987–990. [Google Scholar] [CrossRef]
- Andrade, H.S.; Kaouk, J.H.; Zargar, H.; Caputo, P.A.; Akca, O.; Ramirez, D.; Autorino, R.; Noble, M.; Stein, R.J. Robotic ureteroureterostomy for treatment of a proximal ureteric stricture. Int. Braz. J. Urol. Off. J. Braz. Soc. Urol. 2016, 42, 1041–1042. [Google Scholar] [CrossRef]
- Stein, R.; Rubenwolf, P.; Ziesel, C.; Kamal, M.M.; Thüroff, J.W. Psoas hitch and boari flap ureteroneocystostomy. BJU Int. 2013, 112, 137–155. [Google Scholar] [CrossRef] [PubMed]
- Kuntz, N.J.; Neisius, A.; Tsivian, M.; Ghaffar, M.; Patel, N.; Ferrandino, M.N.; Sur, R.L.; Preminger, G.M.; Lipkin, M.E. Balloon dilation of the ureter: A contemporary review of outcomes and complications. J. Urol. 2015, 194, 413–417. [Google Scholar] [CrossRef] [PubMed]
- Slater, R.C.; Farber, N.J.; Riley, J.M.; Shilo, Y.; Ost, M.C. Contemporary series of robotic-assisted distal ureteral reconstruction utilizing side docking position. Int. Braz. J. Urol. Off. J. Braz. Soc. Urol. 2015, 41, 1154–1159. [Google Scholar] [CrossRef] [PubMed]
- Schiavina, R.; Zaramella, S.; Chessa, F.; Pultrone, C.V.; Borghesi, M.; Minervini, A.; Cocci, A.; Chindemi, A.; Antonelli, A.; Simeone, C.; et al. Laparoscopic and robotic ureteral stenosis repair: A multi-institutional experience with a long-term follow-up. J. Robot. Surg. 2016, 10, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Arora, S.; Campbell, L.; Tourojman, M.; Pucheril, D.; Jones, L.R.; Rogers, C. Robotic buccal mucosal graft ureteroplasty for complex ureteral stricture. Urology 2017, 110, 257–258. [Google Scholar] [CrossRef]
- Duty, B.D.; Kreshover, J.E.; Richstone, L.; Kavoussi, L.R. Review of appendiceal onlay flap in the management of complex ureteric strictures in six patients. BJU Int. 2015, 115, 282–287. [Google Scholar] [CrossRef]
- Alberts, V.P.; Idu, M.M.; Legemate, D.A.; Laguna Pes, M.P.; Minnee, R.C. Ureterovesical anastomotic techniques for kidney transplantation: A systematic review and meta-analysis. Transpl. Int. Off. J. Eur. Soc. Organ Transplant. 2014, 27, 593–605. [Google Scholar] [CrossRef]
- Ueshima, E.; Fujimori, M.; Kodama, H.; Felsen, D.; Chen, J.; Durack, J.C.; Solomon, S.B.; Coleman, J.A.; Srimathveeravalli, G. Macrophage-secreted tgf-beta1 contributes to fibroblast activation and ureteral stricture after ablation injury. Am. J. Physiol. Renal. Physiol. 2019, 317, F52–F64. [Google Scholar] [CrossRef] [PubMed]
- Rozen-Zvi, B.; Hayashida, T.; Hubchak, S.C.; Hanna, C.; Platanias, L.C.; Schnaper, H.W. Tgf-beta/smad3 activates mammalian target of rapamycin complex-1 to promote collagen production by increasing hif-1alpha expression. Am. J. Physiol. Renal. Physiol. 2013, 305, F485–F494. [Google Scholar] [CrossRef] [PubMed]
- Milani, B.Y.; Milani, F.Y.; Park, D.W.; Namavari, A.; Shah, J.; Amirjamshidi, H.; Ying, H.; Djalilian, A.R. Rapamycin inhibits the production of myofibroblasts and reduces corneal scarring after photorefractive keratectomy. Invest. Ophthalmol. Vis. Sci. 2013, 54, 7424–7430. [Google Scholar] [CrossRef]
- Wang, B.; Ding, W.; Zhang, M.; Li, H.; Gu, Y. Rapamycin attenuates aldosterone-induced tubulointerstitial inflammation and fibrosis. Cell. Physiol. Biochem. 2015, 35, 116–125. [Google Scholar] [CrossRef]
- Xiang, S.; Li, M.; Xie, X.; Xie, Z.; Zhou, Q.; Tian, Y.; Lin, W.; Zhang, X.; Jiang, H.; Shou, Z.; et al. Rapamycin inhibits epithelial-to-mesenchymal transition of peritoneal mesothelium cells through regulation of rho gtpases. FEBS J. 2016, 283, 2309–2325. [Google Scholar] [CrossRef]
- Chen, X.; Yang, Y.; Liu, C.; Chen, Z.; Wang, D. Astragaloside iv ameliorates high glucoseinduced renal tubular epithelialmesenchymal transition by blocking mtorc1/p70s6k signaling in hk2 cells. Int. J. Mol. Med. 2019, 43, 709–716. [Google Scholar]
- Takahashi, H.; Wang, Y.; Grainger, D.W. Device-based local delivery of sirna against mammalian target of rapamycin (mtor) in a murine subcutaneous implant model to inhibit fibrous encapsulation. J. Control Release 2010, 147, 400–407. [Google Scholar] [CrossRef]
- Ong, C.T.; Khoo, Y.T.; Mukhopadhyay, A.; Do, D.V.; Lim, I.J.; Aalami, O.; Phan, T.T. Mtor as a potential therapeutic target for treatment of keloids and excessive scars. Exp. Dermatol. 2007, 16, 394–404. [Google Scholar] [CrossRef]
- Liatsikos, E.N.; Karnabatidis, D.; Kagadis, G.C.; Rokkas, K.; Constantinides, C.; Christeas, N.; Flaris, N.; Voudoukis, T.; Scopa, C.D.; Perimenis, P.; et al. Application of paclitaxel-eluting metal mesh stents within the pig ureter: An experimental study. Eur. Urol. 2007, 51, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Minardi, D.; Ghiselli, R.; Cirioni, O.; Giacometti, A.; Kamysz, W.; Orlando, F.; Silvestri, C.; Parri, G.; Kamysz, E.; Scalise, G.; et al. The antimicrobial peptide tachyplesin iii coated alone and in combination with intraperitoneal piperacillin-tazobactam prevents ureteral stent pseudomonas infection in a rat subcutaneous pouch model. Peptides 2007, 28, 2293–2298. [Google Scholar] [CrossRef]
- Cadieux, P.A.; Chew, B.H.; Nott, L.; Seney, S.; Elwood, C.N.; Wignall, G.R.; Goneau, L.W.; Denstedt, J.D. Use of triclosan-eluting ureteral stents in patients with long-term stents. J. Endourol. 2009, 23, 1187–1194. [Google Scholar] [CrossRef] [PubMed]
- Krambeck, A.E.; Walsh, R.S.; Denstedt, J.D.; Preminger, G.M.; Li, J.; Evans, J.C.; Lingeman, J.E.; Lexington Trial Study, G. A novel drug eluting ureteral stent: A prospective, randomized, multicenter clinical trial to evaluate the safety and effectiveness of a ketorolac loaded ureteral stent. J. Urol. 2010, 183, 1037–1042. [Google Scholar] [CrossRef] [PubMed]
- Kallidonis, P.; Kitrou, P.; Karnabatidis, D.; Kyriazis, I.; Kalogeropoulou, C.; Tsamandas, A.; Apostolopoulos, D.J.; Vrettos, T.; Liourdi, D.; Spiliopoulos, S.; et al. Evaluation of zotarolimus-eluting metal stent in animal ureters. J. Endourol. 2011, 25, 1661–1667. [Google Scholar] [CrossRef] [PubMed]
- Schlick, R.W.; Planz, K. Potentially useful materials for biodegradable ureteric stents. Br. J. Urol. 1997, 80, 908–910. [Google Scholar] [CrossRef]
- Li, G.; Wang, Z.X.; Fu, W.J.; Hong, B.F.; Wang, X.X.; Cao, L.; Xu, F.Q.; Song, Q.; Cui, F.Z.; Zhang, X. Introduction to biodegradable polylactic acid ureteral stent application for treatment of ureteral war injury. BJU Int. 2011, 108, 901–906. [Google Scholar] [CrossRef] [PubMed]
- Soria, F.; Morcillo, E.; Lopez de Alda, A.; Pastor, T.; Sanchez-Margallo, F.M. Biodegradable catheters and urinary stents. When? Arch. Esp. Urol. 2016, 69, 553–564. [Google Scholar] [PubMed]
- Barros, A.A.; Oliveira, C.; Reis, R.L.; Lima, E.; Duarte, A.R. Ketoprofen-eluting biodegradable ureteral stents by co2 impregnation: In vitro study. Int. J. Pharm. 2015, 495, 651–659. [Google Scholar] [CrossRef] [PubMed]
- Barros, A.A.; Browne, S.; Oliveira, C.; Lima, E.; Duarte, A.R.; Healy, K.E.; Reis, R.L. Drug-eluting biodegradable ureteral stent: New approach for urothelial tumors of upper urinary tract cancer. Int. J. Pharm. 2016, 513, 227–237. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Xiao, Y.; Xu, H.; Lei, K.; Lang, M. Preparation, degradation and in vitro release of ciprofloxacin-eluting ureteral stents for potential antibacterial application. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 66, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Barros, A.A.; Oliveira, C.; Reis, R.L.; Lima, E.; Duarte, A.R.C. In vitro and ex vivo permeability studies of paclitaxel and doxorubicin from drug-eluting biodegradable ureteral stents. J. Pharm. Sci. 2017, 106, 1466–1474. [Google Scholar] [CrossRef] [PubMed]
- Byrne, R.A.; Kufner, S.; Tiroch, K.; Massberg, S.; Laugwitz, K.L.; Birkmeier, A.; Schulz, S.; Mehilli, J.; Investigators, I.-T. Randomised trial of three rapamycin-eluting stents with different coating strategies for the reduction of coronary restenosis: 2-year follow-up results. Heart 2009, 95, 1489–1494. [Google Scholar] [CrossRef]
- Buszman, P.P.; Orlik, B.; Pajak, J.; Jelonek, M.; Krauze, A.; Janas, A.; Legutko, J.; Wojakowski, W.; Buszman, P.E.; Milewski, K. Dose-dependent vascular response following delivery of sirolimus via fast releasing, biodegradable polymer stent matrix: An experimental study in the porcine coronary model of restenosis. Kardiol. Pol. 2015, 73, 916–923. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Tai, W.; Qu, X.; Wu, W.; Li, Z.; Deng, S.; Vongphouttha, C.; Dong, Z. Rapamycin protects against paraquat-induced pulmonary fibrosis: Activation of nrf2 signaling pathway. Biochem. Biophys. Res. Commun. 2017, 490, 535–540. [Google Scholar] [CrossRef]
- Mehilli, J.; Byrne, R.A.; Wieczorek, A.; Iijima, R.; Schulz, S.; Bruskina, O.; Pache, J.; Wessely, R.; Schomig, A.; Kastrati, A.; et al. Randomized trial of three rapamycin-eluting stents with different coating strategies for the reduction of coronary restenosis. Eur. Heart J. 2008, 29, 1975–1982. [Google Scholar] [CrossRef]
- Roleder, T.; Smolka, G.; Podolecka, E.; Chudek, J.; Dworowy, S.; Zelazowska, K.; Wojakowski, W.; Ochala, A. Outcomes of biodegradable polymer sirolimus-eluting prolim stent in patients with coronary artery disease. Results of 12-month follow-up of prospective registry. Kardiol. Pol. 2016, 74, 411–417. [Google Scholar] [CrossRef] [PubMed]
- Anidjar, M.; Mongiat-Artus, P.; Brouland, J.P.; Meria, P.; Teillac, P.; Le Duc, A.; Berthon, P.; Cussenot, O. Thermal radiofrequency induced porcine ureteral stricture: A convenient endourologic model. J. Urol. 1999, 161, 298–303. [Google Scholar] [CrossRef]
- Soria, F.; Delgado, M.I.; Rioja, L.A.; Blas, M.; Pamplona, M.; Duran, E.; Uson, J.; Sanchez, F.M. Ureteral double-j wire stent effectiveness after endopyelotomy: An animal model study. Urol. Int. 2010, 85, 314–319. [Google Scholar] [CrossRef] [PubMed]
- Lumiaho, J.; Heino, A.; Aaltomaa, S.; Valimaa, T.; Talja, M. A short biodegradable helical spiral ureteric stent provides better antireflux and drainage properties than a double-j stent. Scand. J. Urol. Nephrol. 2011, 45, 129–133. [Google Scholar] [CrossRef]
- Soria, F.; Morcillo, E.; Serrano, A.; Rioja, J.; Budia, A.; Moreno, J.; Sanchez-Margallo, F.M. Preliminary assessment of a new antireflux ureteral stent design in swine model. Urology 2015, 86, 417–422. [Google Scholar] [CrossRef]
- Kim, H.W.; Park, C.J.; Seo, S.; Park, Y.; Lee, J.Z.; Shin, D.G.; Moon, H.S.; Lee, J.H. Evaluation of a polymeric flap valve-attached ureteral stent for preventing vesicoureteral reflux in elevated intravesical pressure conditions: A pilot study using a porcine model. J. Endourol. 2016, 30, 428–432. [Google Scholar] [CrossRef] [PubMed]
- Wainstein, M.; Anderson, J.; Elder, J.S. Comparison of effects of suture materials on wound healing in a rabbit pyeloplasty model. Urology 1997, 49, 261–264. [Google Scholar] [CrossRef]
- Donnellan, S.M.; Ryan, A.J.; Bolton, D.M. Gastric patch pyeloplasty: Development of an animal model to produce upper tract urinary acidification for treating struvite urinary calculi. J. Urol. 2001, 166, 684–687. [Google Scholar] [CrossRef]
- Srimathveeravalli, G.; Cornelis, F.; Wimmer, T.; Monette, S.; Kimm, S.Y.; Maybody, M.; Solomon, S.B.; Coleman, J.A.; Durack, J.C. Normal porcine ureter retains lumen wall integrity but not patency following catheter-directed irreversible electroporation: Imaging and histologic assessment over 28 days. J. Vasc. Interv. Radiol. 2017, 28, 913–919.e911. [Google Scholar] [CrossRef]
- Honma, S.; Shinohara, M.; Takahashi, N.; Nakamura, K.; Hamano, S.; Mitazaki, S.; Abe, S.; Yoshida, M. Effect of cyclooxygenase (cox)-2 inhibition on mouse renal interstitial fibrosis. Eur. J. Pharmacol. 2014, 740, 578–583. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Chen, D.Q.; Chen, L.; Cao, G.; Zhao, H.; Liu, D.; Vaziri, N.D.; Guo, Y.; Zhao, Y.Y. Novel inhibitors of the cellular renin-angiotensin system components, poricoic acids, target smad3 phosphorylation and wnt/β-catenin pathway against renal fibrosis. Br. J. Pharmacol. 2018, 175, 2689–2708. [Google Scholar] [CrossRef] [PubMed]
- Baba, I.; Egi, Y.; Utsumi, H.; Kakimoto, T.; Suzuki, K. Inhibitory effects of fasudil on renal interstitial fibrosis induced by unilateral ureteral obstruction. Mol. Med. Rep. 2015, 12, 8010–8020. [Google Scholar] [CrossRef] [PubMed]
- Anorga, S.; Overstreet, J.M.; Falke, L.L.; Tang, J.; Goldschmeding, R.G.; Higgins, P.J.; Samarakoon, R. Deregulation of hippo-taz pathway during renal injury confers a fibrotic maladaptive phenotype. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2018, 32, 2644–2657. [Google Scholar] [CrossRef] [PubMed]
- Tasanarong, A.; Kongkham, S.; Khositseth, S. Dual inhibiting senescence and epithelial-to-mesenchymal transition by erythropoietin preserve tubular epithelial cell regeneration and ameliorate renal fibrosis in unilateral ureteral obstruction. Biomed Res. Int. 2013, 2013, 308130. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Xia, Z.; Abe, K.; Furusu, A.; Miyazaki, M.; Obata, Y.; Tabata, Y.; Koji, T.; Kohno, S. Suppression of renal tubulointerstitial fibrosis by small interfering rna targeting heat shock protein 47. Am. J. Nephrol. 2008, 28, 34–46. [Google Scholar] [CrossRef]
- Martínez-Klimova, E.; Aparicio-Trejo, O.E.; Tapia, E.; Pedraza-Chaverri, J. Unilateral ureteral obstruction as a model to investigate fibrosis-attenuating treatments. Biomolecules 2019, 9, 141. [Google Scholar] [CrossRef]
- Xianyuan, L.; Wei, Z.; Yaqian, D.; Dan, Z.; Xueli, T.; Zhanglu, D.; Guanyi, L.; Lan, T.; Menghua, L. Anti-renal fibrosis effect of asperulosidic acid via tgf-β1/smad2/smad3 and nf-κb signaling pathways in a rat model of unilateral ureteral obstruction. Phytomedicine 2019, 53, 274–285. [Google Scholar] [CrossRef]
- Wongmekiat, O.; Leelarungrayub, D.; Thamprasert, K. Alpha-lipoic acid attenuates renal injury in rats with obstructive nephropathy. Biomed Res. Int. 2013, 2013, 138719. [Google Scholar] [CrossRef]
- Hosseinian, S.; Rad, A.K.; Bideskan, A.E.; Soukhtanloo, M.; Sadeghnia, H.; Shafei, M.N.; Motejadded, F.; Mohebbati, R.; Shahraki, S.; Beheshti, F. Thymoquinone ameliorates renal damage in unilateral ureteral obstruction in rats. Pharmacol. Rep. PR 2017, 69, 648–657. [Google Scholar] [CrossRef]
- Hammad, F.T.; Lubbad, L. Does curcumin protect against renal dysfunction following reversible unilateral ureteric obstruction in the rat? Eur. Surg. Res. 2011, 46, 188–193. [Google Scholar] [CrossRef]
- Nakada, S.Y.; Soble, J.J.; Gardner, S.M.; Wolf, J.S., Jr.; Figenshau, R.S.; Pearle, M.S.; Humphrey, P.A.; Clayman, R.V. Comparison of acucise endopyelotomy and endoballoon rupture for management of secondary proximal ureteral stricture in the porcine model. J. Endourol. 1996, 10, 311–318. [Google Scholar] [CrossRef] [PubMed]
- Chiu, A.W.; Lin, C.H.; Huan, S.K.; Liu, C.J.; Lin, C.C.; Huang, Y.L.; Lin, W.L.; Huang, S.H.; Lee, P.S.; Lin, C.N. Creation of ureteropelvic junction obstruction and its correction by chemical glue-assisted laparoscopic dismembered pyeloplasty. J. Endourol. 2003, 17, 23–28. [Google Scholar] [CrossRef]
- Luo, J.; Zhao, S.; Wang, J.; Luo, L.; Li, E.; Zhu, Z.; Liu, Y.; Kang, R.; Zhao, Z. Bone marrow mesenchymal stem cells reduce ureteral stricture formation in a rat model via the paracrine effect of extracellular vesicles. J. Cell. Mol. Med. 2018, 22, 4449–4459. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Wang, X.; Sun, K.-H.; Zhou, L. A-smooth muscle actin is not a marker of fibrogenic cell activity in skeletal muscle fibrosis. PLoS ONE 2018, 13, e0191031. [Google Scholar] [CrossRef]
- Rosner, D.; McCarthy, N.; Bennett, M. Rapamycin inhibits human in stent restenosis vascular smooth muscle cells independently of prb phosphorylation and p53. Cardiovasc. Res. 2005, 66, 601–610. [Google Scholar] [CrossRef] [PubMed]
- Buellesfeld, L.; Grube, E. Abt-578-eluting stents. The promising successor of sirolimus- and paclitaxel-eluting stent concepts? Herz 2004, 29, 167–170. [Google Scholar] [CrossRef] [PubMed]
- Kern, G.; Mair, S.M.; Noppert, S.J.; Jennings, P.; Schramek, H.; Rudnicki, M.; Mueller, G.A.; Mayer, G.; Koppelstaetter, C. Tacrolimus increases nox4 expression in human renal fibroblasts and induces fibrosis-related genes by aberrant tgf-beta receptor signalling. PLoS ONE 2014, 9, e96377. [Google Scholar]
- Masola, V.; Carraro, A.; Zaza, G.; Bellin, G.; Montin, U.; Violi, P.; Lupo, A.; Tedeschi, U. Epithelial to mesenchymal transition in the liver field: The double face of everolimus in vitro. BMC Gastroenterol. 2015, 15, 118. [Google Scholar] [CrossRef]
- Ko, D.Y.; Shin, J.M.; Um, J.Y.; Kang, B.; Park, I.H.; Lee, H.M. Rapamycin inhibits transforming growth factor beta 1 induced myofibroblast differentiation via the phosphorylated-phosphatidylinositol 3-kinase mammalian target of rapamycin signal pathways in nasal polyp-derived fibroblasts. Am. J. Rhinol. Allergy 2016, 30, 211–217. [Google Scholar] [CrossRef]
- Shan, Z.; Wei, L.; Yu, S.; Jiang, S.; Ma, Y.; Zhang, C.; Wang, J.; Gao, Z.; Wan, F.; Zhuang, G.; et al. Ketamine induces reactive oxygen species and enhances autophagy in sv-huc-1 human uroepithelial cells. J. Cell. Physiol. 2019, 234, 2778–2787. [Google Scholar] [CrossRef] [PubMed]
- Tenekecioglu, E.; Serruys, P.W.; Onuma, Y.; Costa, R.; Chamié, D.; Sotomi, Y.; Yu, T.-B.; Abizaid, A.; Liew, H.-B.; Santoso, T. Randomized comparison of absorb bioresorbable vascular scaffold and mirage microfiber sirolimus-eluting scaffold using multimodality imaging. JACC Cardiovasc. Interv. 2017, 10, 1115–1130. [Google Scholar] [CrossRef] [PubMed]
- Costa, R.A.; Liew, H.-B.; Abizaid, A.; de Ribamar Costa, J.; Chamié, D.; Abizaid, A.; Castro, J.P.; Serruys, P.W.; Santoso, T. Tct-546 6-month angiographic results of the novel mirage microfiber sirolimus-eluting bioresorbable vascular scaffold—A quantitative coronary angiography analysis from the prospective, randomized mirage clinical trial. J. Am. Coll. Cardiol. 2015, 66, B223. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ho, D.-R.; Su, S.-H.; Chang, P.-J.; Lin, W.-Y.; Huang, Y.-C.; Lin, J.-H.; Huang, K.-T.; Chan, W.-N.; Chen, C.-S. Biodegradable Stent with mTOR Inhibitor-Eluting Reduces Progression of Ureteral Stricture. Int. J. Mol. Sci. 2021, 22, 5664. https://doi.org/10.3390/ijms22115664
Ho D-R, Su S-H, Chang P-J, Lin W-Y, Huang Y-C, Lin J-H, Huang K-T, Chan W-N, Chen C-S. Biodegradable Stent with mTOR Inhibitor-Eluting Reduces Progression of Ureteral Stricture. International Journal of Molecular Sciences. 2021; 22(11):5664. https://doi.org/10.3390/ijms22115664
Chicago/Turabian StyleHo, Dong-Ru, Shih-Horng Su, Pey-Jium Chang, Wei-Yu Lin, Yun-Ching Huang, Jian-Hui Lin, Kuo-Tsai Huang, Wai-Nga Chan, and Chih-Shou Chen. 2021. "Biodegradable Stent with mTOR Inhibitor-Eluting Reduces Progression of Ureteral Stricture" International Journal of Molecular Sciences 22, no. 11: 5664. https://doi.org/10.3390/ijms22115664
APA StyleHo, D.-R., Su, S.-H., Chang, P.-J., Lin, W.-Y., Huang, Y.-C., Lin, J.-H., Huang, K.-T., Chan, W.-N., & Chen, C.-S. (2021). Biodegradable Stent with mTOR Inhibitor-Eluting Reduces Progression of Ureteral Stricture. International Journal of Molecular Sciences, 22(11), 5664. https://doi.org/10.3390/ijms22115664





