Japan’s Practical Guidelines for Zinc Deficiency with a Particular Focus on Taste Disorders, Inflammatory Bowel Disease, and Liver Cirrhosis
Abstract
1. Introduction
2. Japan’s Practical Guideline for Zinc Deficiency
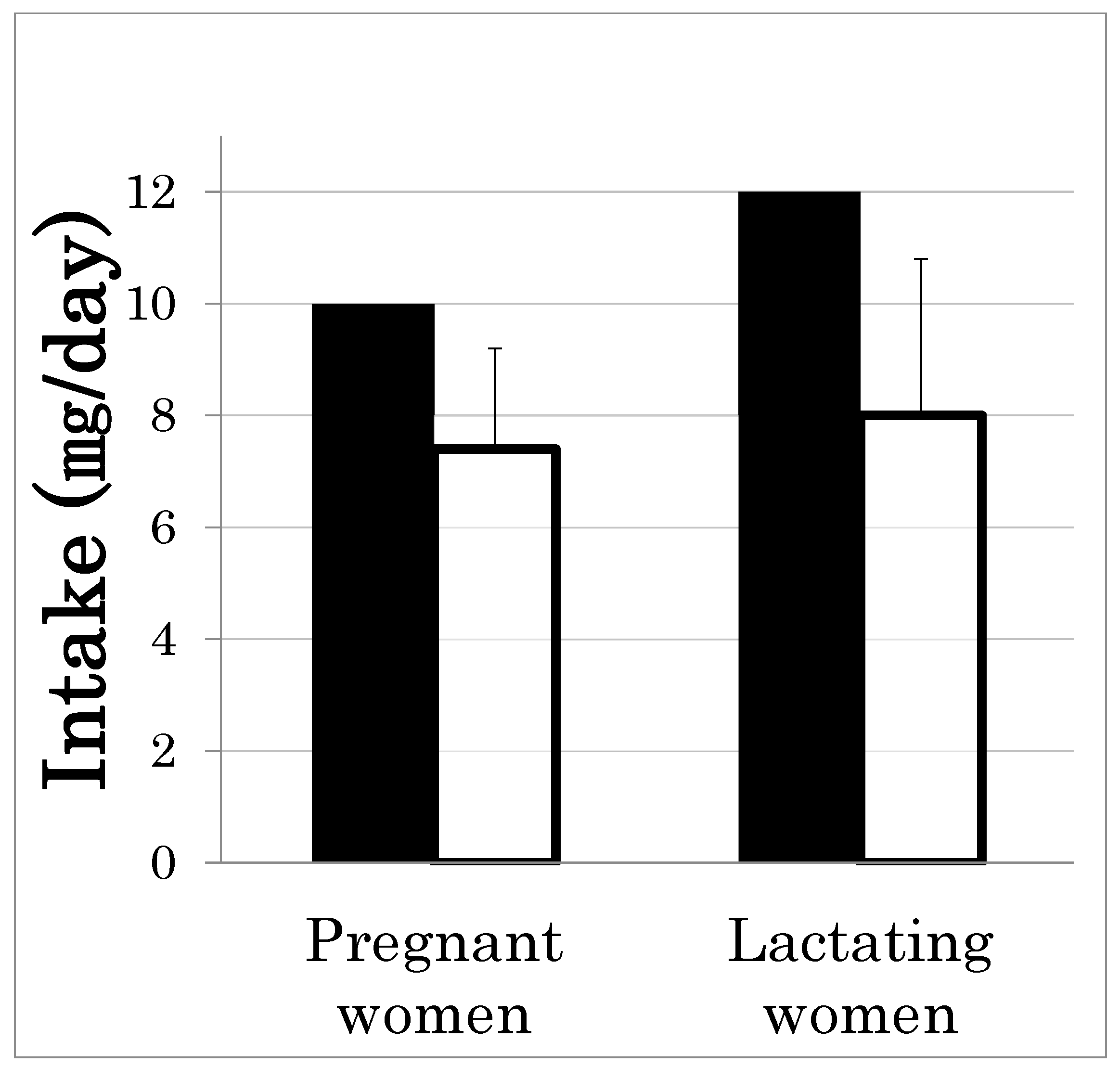
2.1. Prevalence of Insufficient Zinc Intake and Zinc Deficiency in Japan
2.2. Causes of Zinc Deficiency in Japan
Causes of Zinc Deficiency Can Be Divided into Four Major Categories
2.3. Diagnostic Guideline for Zinc Deficiency
2.4. Treatment of Zinc Deficiency
3. Zinc Deficiency and Taste Disorders
3.1. Taste Perception System
3.2. Effects of Zinc Deficiency on Taste Function in Rats
3.3. Taste Disorders
3.4. Zinc Replacement Therapy
3.5. Nutritional Guidance
4. Zinc Deficiency Enhances Intestinal Inflammation via Macrophage Activation
4.1. Role of Metallothioneins (MTs) in Intestinal Inflammation
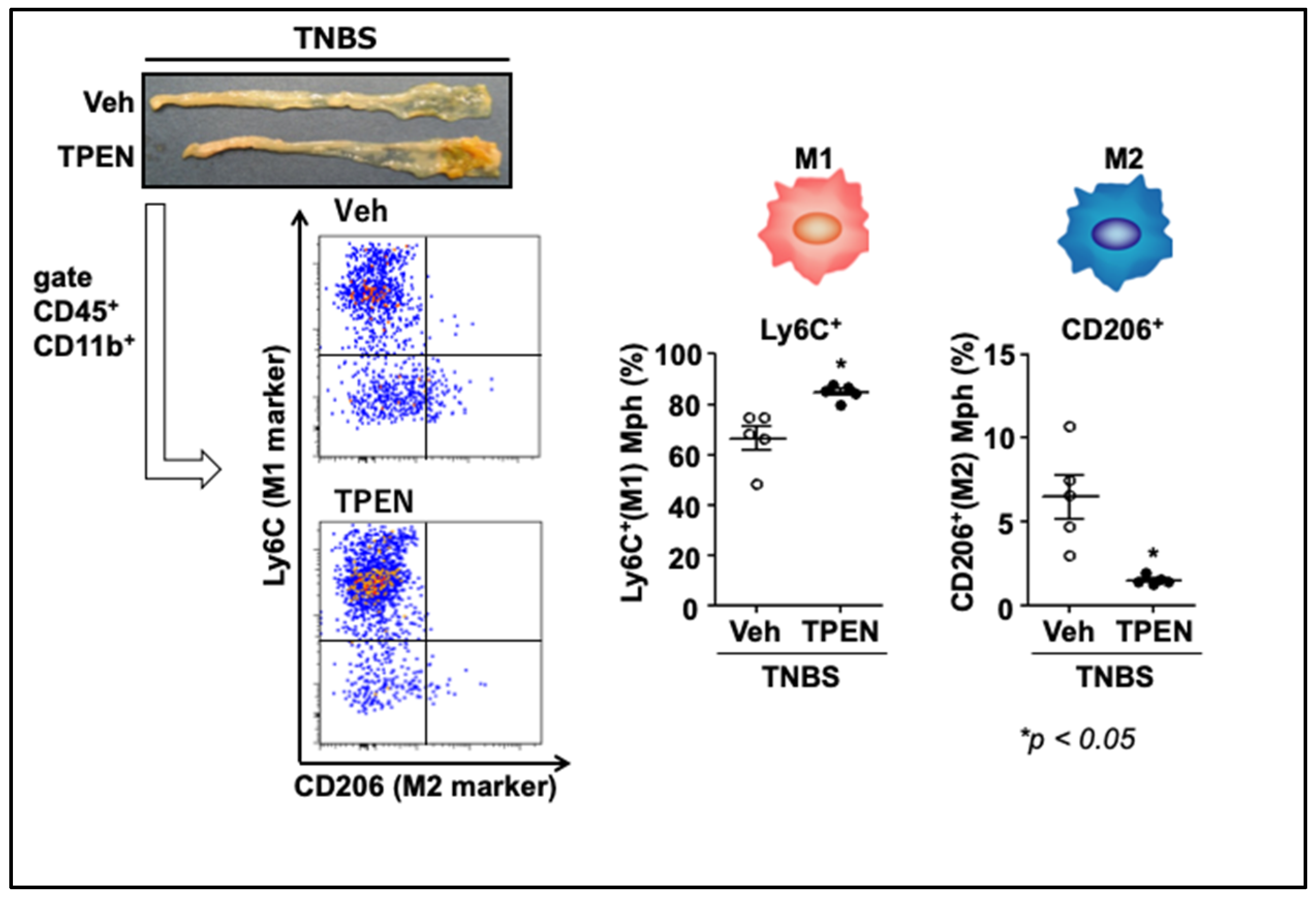
4.2. Zinc Deficiency Aggravates Trinitrobenzene Sulfonic Acid (TNBS)-Induced Colitis
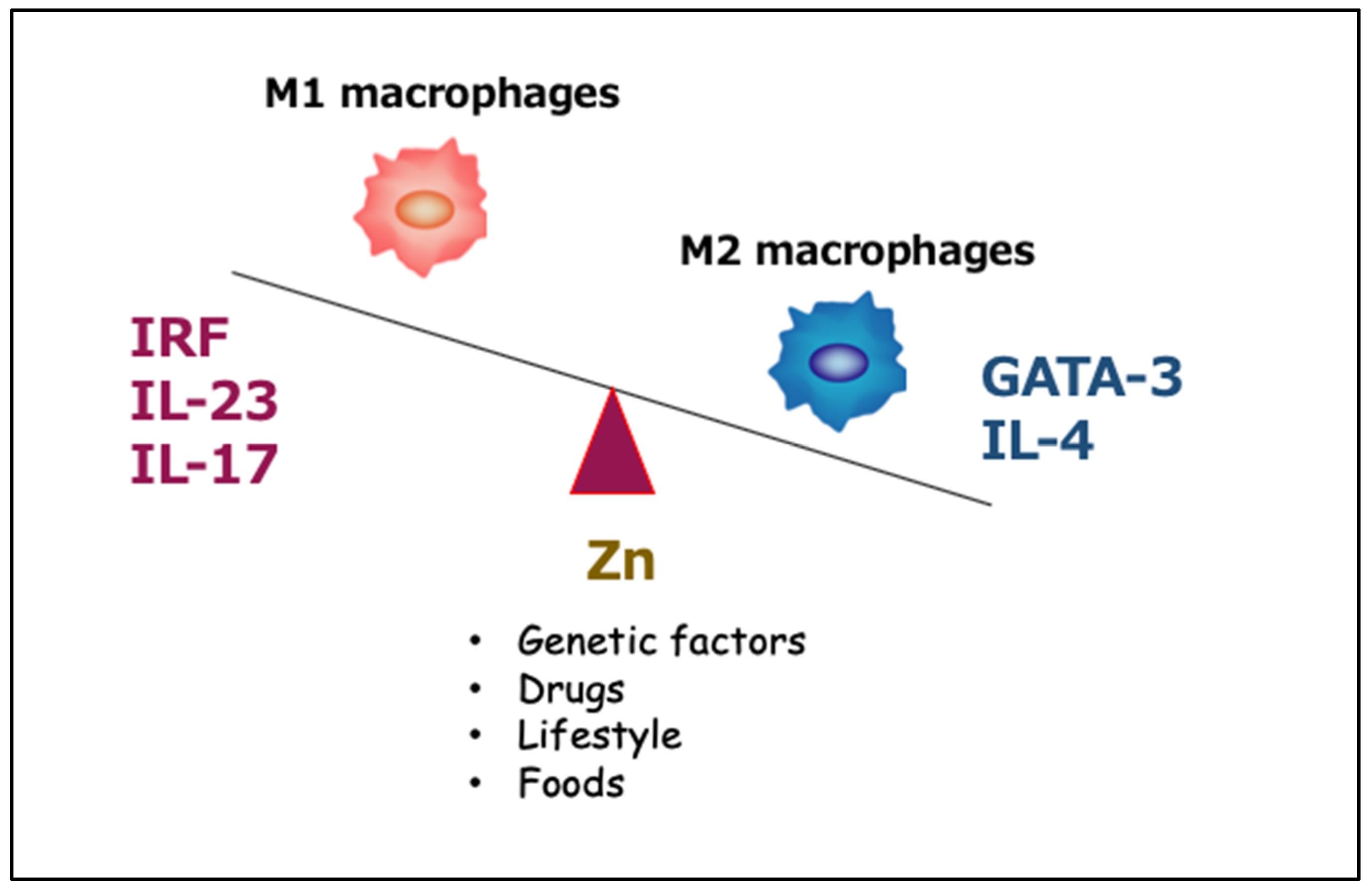
4.3. Effects of Zinc Deficiency on Macrophage Phenotype under Inflammatory Conditions
5. Zinc and Protein Metabolism in Chronic Liver Disease
5.1. Mechanism Underlying Reduced Capacity to Detoxify Ammonia in Patients with Liver Cirrhosis
5.2. Does Zinc Supplementation in Patients with Liver Cirrhosis Improve Protein Metabolism and Prognosis?
6. Conclusions and Future Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Prasad, A.S. Discovery of human zinc deficiency: 50 years later. J. Trace Elem. Med. Biol. 2012, 26, 66–69. [Google Scholar] [CrossRef] [PubMed]
- Wessells, K.R.; Brown, K.H. Estimating the grobal prevalence of zinc deficiency: Results based on zinc availability in national food supplies and the prevalence of stunting. PLoS ONE 2012, 7, e50567. [Google Scholar] [CrossRef] [PubMed]
- Kodama, H.; Itakura, H.; Ohmori, H.; Sasaki, M.; Sando, K.; Takamura, T.; Fuse, Y.; Hosoi, T.; Yoshida, H. Practice Guideline for Zinc Deficiency. J. Jpn. Soc. Clin. Nutr. 2018, 40, 120–167. (In Japanese) [Google Scholar]
- Ministry of Health, Labor and Welfare of Japan. Dietary Reference Intakes for Japanese 2020. Available online: https://www.mhlw.go.jp/stf/newpage_08517.html (accessed on 18 February 2020).
- Ministry of Health, Labor and Welfare of Japan. National Health and Nutrition Survey. Available online: https://www.mhlw.go.jp/stf/newpage_08789.html (accessed on 18 February 2020).
- Muguruma, T.; Ohmori, H.; Takeda, I.; Yamasaki, M.; Murata, Y.; Fukuba, H.; Agari, M.; Maruyama, H.; Matsumoto, M.; Miyachi, T. Nutritional evaluation in patients with intractable neurological diseases undergoing long-term tube feeding- in point of trace elements and carnitine. J. Jpn. Sco. Clin. Nutr. 2015, 37, 36–43. [Google Scholar]
- Nishikawa, H.; Enomoto, H.; Yoh, K.; Iwata, Y.; Sakai, Y.; Kishino, K.; Ikeda, N.; Takashima, T.; Aizawa, N.; Takata, R.; et al. Serum zinc concentration and sarcopenia: A close linkage in chronic liver diseases. J. Clin. Med. 2019, 8, 336. [Google Scholar] [CrossRef]
- Sava, S.; Rubin, D.T.; Gulotta, G.; Wroblewaki, K.; Pekow, J. Zinc deficiency is associated with poor clinical outcomes in patients with inflammatory bowel disease. Inflamm. Bowel Dis. 2017, 23, 152–157. [Google Scholar] [CrossRef] [PubMed]
- Ishihara, J.; Sakaguchi, H.; Arai, K. Serum zinc and selenium status in pediatric patients with inflammatory bowel disease: A retrospective multicenter study in Japan. JPGN 2019, 69, 142. [Google Scholar]
- Sagara, M.; Kudo, M.; Yashiro, H.; Makino, I.; Takabe, K. Urinary excretion and serum levels of zinc in diabetics, and its relationship to renal damage. J. Jpn. Diab. Soc. 1982, 25, 697–704. [Google Scholar]
- Tsutsui, T.; Ito, K.; Tsunoda, C.; Nakajima, H.; Sunaga, S.; Nohara, T.; Otaka, Y.; Hoshi, A.; Takemi, N.; Nohara, A.; et al. Zinc is more strongly related to ESA-resistance than iron in chronic kidney disease patients. Jpn. J. Nephrol. 2018, 60, 609–618. [Google Scholar]
- Nagano, N.; Ito, K.; Oishi, Y.; Minami, M.; Hayashi, H.; Tsunoda, C.; Nakajima, H.; Sunaga, S.; Nohara, T.; Otaka, Y.; et al. Distribution of serum zinc levels in hemodialysis patients: Factors related to hypozincemia. J. Jpn. Soc. Dial. Ther. 2018, 51, 369–377. [Google Scholar] [CrossRef][Green Version]
- Hirao, Y.; Shiraishi, Y.; Sato, Y.; Urade, H.; Nakanishi, N.; Zen, K.; Nakamura, T.; Yamano, T.; Shirayama, T.; Matoba, S. Clinical Significance of Serum Zinc Level in Heart Failure Patients. In Proceedings of the 67th Scientific Session together with ACC Asia Conference, Nagoya city, Japan, 13–14 September 2019. [Google Scholar]
- Yagisawa, H.; Shono, T.; Tokaibayashi, H.; Obinata, K.; Shimizu, T. Evaluation of Serum Zinc Level with Growth Gailure and Short Stature Children. Jpn. J. Pediatr. Gastroenterol. Nutr. 2012, 26, 256. [Google Scholar]
- Goda, F.; Inukai, M.; Okuyama, H.; Himoto, T.; Masugata, H.; Senda, S. The feasibility study of the necessity of supplementations of trace elements in healthy elderly subjects. J. JSPEN 2010, 25, 823–829. [Google Scholar]
- Irsumura, N.; Kibihara, Y.; Fukue, K.; Miyata, A.; Fukushima, K.; Tamagawa-Mineoka, R.; Katoh, N.; Nishino, Y.; Ishida, R.; Narita, H.; et al. Novel mutations in SLC30A2 involved in the pathogenesis of transient neonatal zinc deficiency. Pediatr. Res. 2016, 80, 586–594. [Google Scholar] [CrossRef] [PubMed]
- Salimi, S.; Yaghmaei, M.; Joshaghani, H.R.; Mansourian, A.R. Study of Zinc Deficiency in Pregnant Women. Iranian J. Publ. Health 2004, 33, 15–18. [Google Scholar]
- Nishiyama, S.; Inomoto, T.; Nakamura, T.; Higashi, A.; Matsuda, I. Zinc status relates to hematological deficits in women endurance runners. J. Am. Coll Nutr. 1996, 15, 359–363. [Google Scholar] [CrossRef]
- Sutex, P.M.; Russell, R.M. Vitamin and trace mineral deficiency and excess. In Harrison’s Principles of Internal Medicine, 17th ed.; The McGraw-Hill Companies: New York, NY, USA, 2015; pp. 2309–2319. [Google Scholar]
- Nakamura, T.; Higashi, A.; Nishiyama, S.; Fujimoto, S.; Matsuda, I. Kinetics of zinc status in children with insulin dependent diabetes mellitus (IDDMI). Diabetes Care 1991, 14, 553–557. [Google Scholar] [CrossRef]
- Nakamura, T.; Higashi, A.; Takano, S.; Akagi, M.; Matsuda, I. Zinc clearance correlates with clinical severity of Crohn’s disease. A kinetic study. Dig. Dis. Sci. 1988, 33, 1520–1524. [Google Scholar] [CrossRef]
- Nakamura, T.; Nishiyama, S.; Futagoishi-Suginohara, Y.; Matsuda, I.; Higashi, A. Mild to moderate zinc deficiency in short children: Effect of zinc supplementation on linear growth velocity. J. Pediatr. 1993, 123, 65–69. [Google Scholar] [CrossRef]
- Kaji, M.; Gotoh, M.; Takagi, Y.; Masuda, H.; Kimura, Y.; Uenoyama, Y. Studies to determine the usefulness of the zinc clearance test to diagnose marginal zinc deficiency and the effects of oral zinc supplementation for short children. J. Am. Coll. Nutr. 1998, 17, 388–391. [Google Scholar] [CrossRef]
- Tomita, H.; Tanaka, M.; Ikui, A. Clinical standard value for diagnosis of zinc deficiency by the serum zinc value on the basis of evidence. Biomed. Res. Trace Elements 2007, 18, 54–62. [Google Scholar]
- Singh, P.; Nee, J. BMJ Best Practice: Zinc Deficiency. Available online: https://bestpractice.bmj.com/topics/en-gb/1195 (accessed on 22 February 2018).
- Takuma, Y.; Nouso, K.; Makino, Y.; Hayashi, M.; Takahashi, H. Clinical trial: Oral zinc in hepatic encephalopathy. Aliment. Pharmacol Ther. 2010, 32, 1080–1090. [Google Scholar] [CrossRef] [PubMed]
- Sturniolo, G.C.; Di Leo, V.; Ferronato, A.; D’Odorico, A.; D’Inca, R. Zinc supplementation tightens “Leaky gut” in Crohn’s disease. Imflamm. Bowel Dis. 2001, 7, 94–98. [Google Scholar] [CrossRef] [PubMed]
- Parham, M.; Amini, M.; Aminorroaya, A.; Heidarian, E. Effect of zinc supplementation on microalbuminuria in patients with type 2 diabetes: A double blind, randomized, placebo-controlled, cross-over trial. Rev. Diabet. Stud. 2008, 5, 102–109. [Google Scholar] [CrossRef]
- Roussel, A.M.; Kerkeni, A.; Zouari, N.; Mahjoub, S.; Matheau, J.M.; Anderson, R.A. Antioxidant effects of zinc supplementation in Tunisians with type 2 diabetes mellitus. J. Am. Coll. Nutr. 2003, 22, 316–321. [Google Scholar] [CrossRef] [PubMed]
- El-Shazly, A.N.; El-Hady Ibrahim, S.A.; El-Mashad, G.M.; Sabry, J.H.; Sherbini, N.S. Effect of zinc supplementation on body mass index and serum levels of zinc and leptin in pediatric hemodialysis patients. Int. J. Nephrol. Renovasc. Dis. 2015, 8, 159–163. [Google Scholar]
- Pakfetrat, M.; Shahroodi, J.R.; Zolgadr, A.A.; Larie, H.A.; Nikoo, M.H.; Malekmakan, L. Effects of zinc supplement on plasma homocysteine level in end-stage renal disease patients: A double-blind randomized clinical trial. Biol. Trace Elem. Res. 2013, 153, 11–15. [Google Scholar] [CrossRef]
- Willis, M.S.; Monaghan, S.A.; Miller, M.L.; McKenna, R.W.; Perkins, W.D.; Levinson, B.S.; Bhushan, V.; Kroft, S.H. Zinc-induced copper deficiency. A report of three cases initially recognized on bone marrow examination. Am. J. Clin. Pathol. 2005, 123, 125–131. [Google Scholar] [CrossRef]
- Duncan, A.; Yacoubian, C.; Watson, N.; Morrison, I. The risk of copper deficiency in patients prescribed zinc supplements. J Clin. Pathol. 2015, 68, 723–725. [Google Scholar] [CrossRef]
- Hamano, H.; Yoshinaga, K.; Eta, R.; Emori, Y.; Kawasaki, D.; Iino, Y.; Sawada, M.; Kuroda, H.; Takei, M. Effect of polaprezinc on taste disorders in zinc-deficient rats. Biofactors 2006, 28, 185–193. [Google Scholar] [CrossRef]
- Naganuma, M.; Ikeda, M.; Tomita, H. Changes in soft palate taste buds of rats due to aging and zinc deficiency-scanning electron microscopic observation. Auris Nasus Larynx. 1988, 15, 117–127. [Google Scholar] [CrossRef]
- Kobayashi, T.; Tomita, H. Electron microscopic observation of vallate taste buds of zinc deficient rats with taste disturbance. Auris Nasus Larynx. 1986, 13, S25–S31. [Google Scholar] [CrossRef]
- Sekine, H.; Takao, H.; Yoshinaga, K.; Kokubun, S.; Ikeda, M. Effects of zinc deficiency and supplementation on gene expression of bitter taste receptors (TAS2Rs) on the tongue in rats. Laryngoscope 2012, 122, 2411–2417. [Google Scholar] [CrossRef] [PubMed]
- Henkin, R.I.; Bradley, D.F. Hypogeusia corrected by N++ and Zn++. Life Sci. 1970, 9, 701–709. [Google Scholar] [CrossRef]
- Yoshida, S.; Endo, S.; Tomita, H. A double-blind study of the therapeutic efficacy of zinc gluconate on taste disorder. Auris Nasus Larynx 1991, 18, 153–161. [Google Scholar] [CrossRef]
- Sakai, F.; Yoshida, S.; Endo, S.; Tomita, H. Double-blind, pracebo, controlled trial of zinc picolinate for taste disorders. Acta Otolaryngol. 2002, 546, 129–133. [Google Scholar] [CrossRef]
- Heckmann, S.M.; Hujoel, P.; Habiger, S.; Friess, W.; Wichmann, M.; Heckmann, J.G.; Hummel, T. Zinc gluconate in the treatment of dysgeusia–a randomized clinical trial. J. Dent. Res. 2005, 84, 35–38. [Google Scholar] [CrossRef]
- Sakagami, M.; Ikeda, M.; Tomita, H.; Ikui, A.; Aiba, T.; Takeda, N.; Inokuchi, A.; Kurorno, Y.; Nakashima, M.; Shibasaki, Y.; et al. A zinc-containing compound, Polaprezinc, is effective for patients with taste disorders: Randomized, double-blind, placebo-controlled, multi-center study. Acta Otolaryngol. 2009, 129, 1115–1120. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, Y.; Ohfuji, S.; Kondo, K.; Fukushima, W.; Sasaki, S.; Kamata, N.; Yamagami, H.; Fujiwara, Y.; Suzuki, Y.; Hirota, Y. Association between dietary iron and zinc intake and development of ulcerative colitis: A case-control study in Japan. J. Gastroenterol. Hepatol. 2019, 34, 1703–1710. [Google Scholar] [CrossRef] [PubMed]
- Higashimura, Y.; Takagi, T.; Naito, Y.; Uchiyama, K.; Mizushima, K.; Tanaka, M.; Hamaguchi, M.; Itoh, Y. Zinc deficiency activates IL-23/Th17 axis to aggravate experimental colitis in mice. J. Crohns Colitis 2019. [Google Scholar] [CrossRef] [PubMed]
- Tsuji, T.; Naito, Y.; Takagi, T.; Kugai, M.; Yoriki, H.; Horie, R.; Fukui, A.; Mizushima, K.; Hirai, Y.; Katada, K.; et al. Role of metallothionein in murine experimental colitis. Int. J. Mol. Med. 2013, 31, 1037–1046. [Google Scholar] [CrossRef]
- Schmidt, C.; Giese, T.; Ludwig, B.; Mueller-Molaian, I.; Marth, T.; Zeuzem, S.; Meuer, S.C.; Stallmach, A. Expression of interleukin-12-related cytokine transcripts in inflammatory bowel disease: Elevated interleukin-23p19 and interleukin-27p28 in Crohn’s disease but not in ulcerative colitis. Inflamm. Bowel Dis. 2005, 11, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Sandborn, W.J.; Feagan, B.G.; Fedorak, R.N.; Scherl, E.; Fleisher, M.R.; Katz, S.; Johanns, J.; Blank, M.; Rutgeerts, P. A randomized trial of Ustekinumab, a human interleukin-12/23 monoclonal antibody, in patients with moderate-to-severe Crohn’s disease. Gastroenterology 2008, 135, 1130–1141. [Google Scholar] [CrossRef] [PubMed]
- Kido, T.; Ishiwata, K.; Suka, M.; Yanagisawa, H. Inflammatory response under zinc deficiency is exacerbated by dysfunction of the T helper type 2 lymphocyte-M2 macrophage pathway. Immunology 2019, 156, 356–372. [Google Scholar] [CrossRef] [PubMed]
- Prasad, A.S. Zinc: An overview. Nutrition 1995, 11, 93–95. [Google Scholar]
- Riggio, O.; Merli, M.; Capocaccia, L.; Cashera, M.; Zullo, A.; Pinto, G.; Gaudio, E.; Franchitto, A.; Spagnolo, R.; D’Aquilino, E.; et al. Zinc supplementation reduces blood ammonia and increases liver ornithine transcarbamylase activity in experimental cirrhosis. Hepatology 1992, 16, 785–789. [Google Scholar] [CrossRef]
- Stamoulis, I.; Kouraklis, G.; Theocharis, S. Zinc and the liver: An active interaction. Dig. Dis. Sci. 2007, 52, 1595–1612. [Google Scholar] [CrossRef]
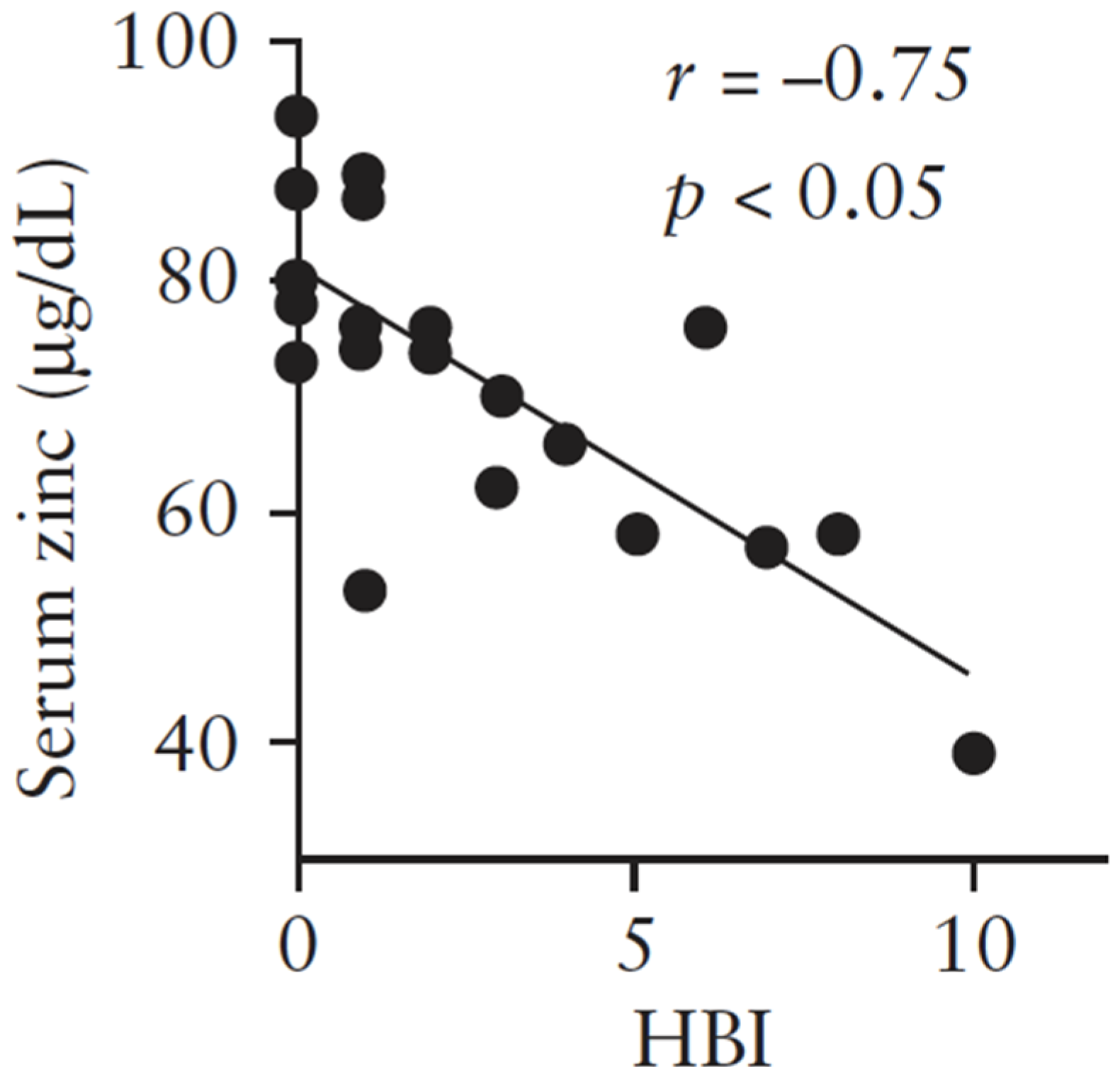
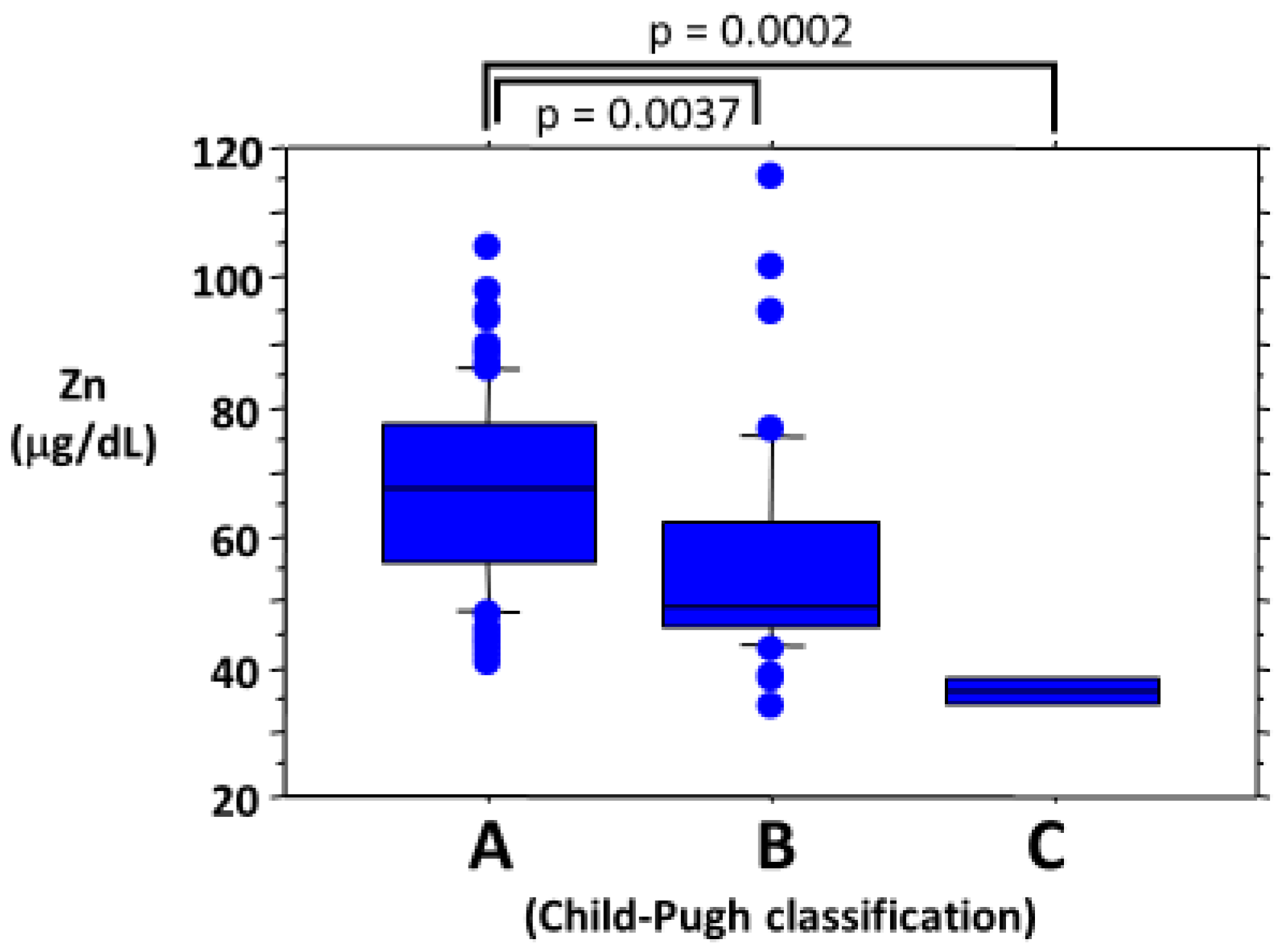
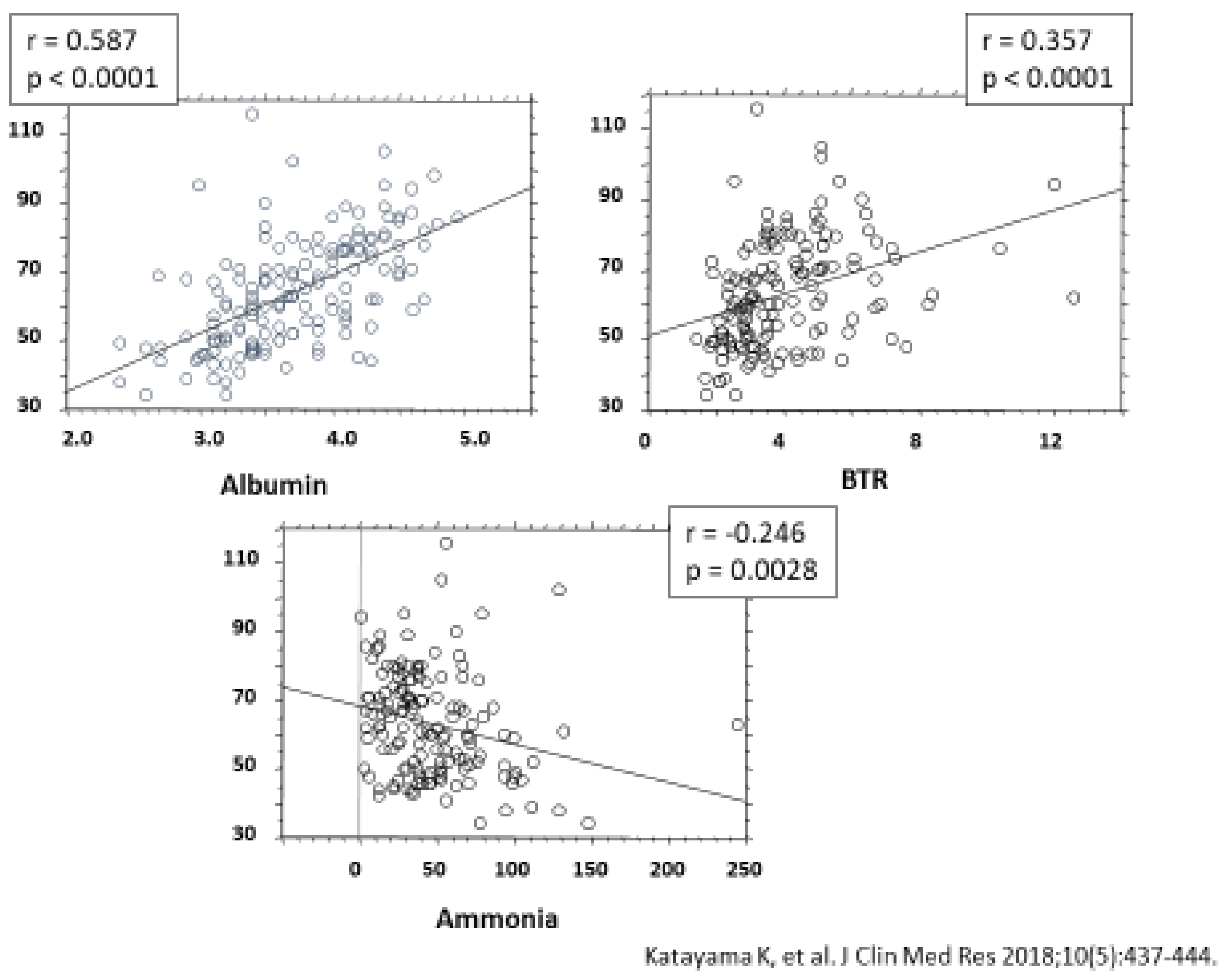
- Katayama, K.; Kawaguchi, T.; Shiraishi, K.; Ito, T. The prevalence and implication of zinc deficiency in patients with chronic liver disease. J. Clin. Med. Res. 2018, 10, 437–444. [Google Scholar] [CrossRef]
- Katayama, K. Zinc and protein metabolism in chronic liver diseases. Nutr. Res. 2020, 74, 1–9. [Google Scholar] [CrossRef]
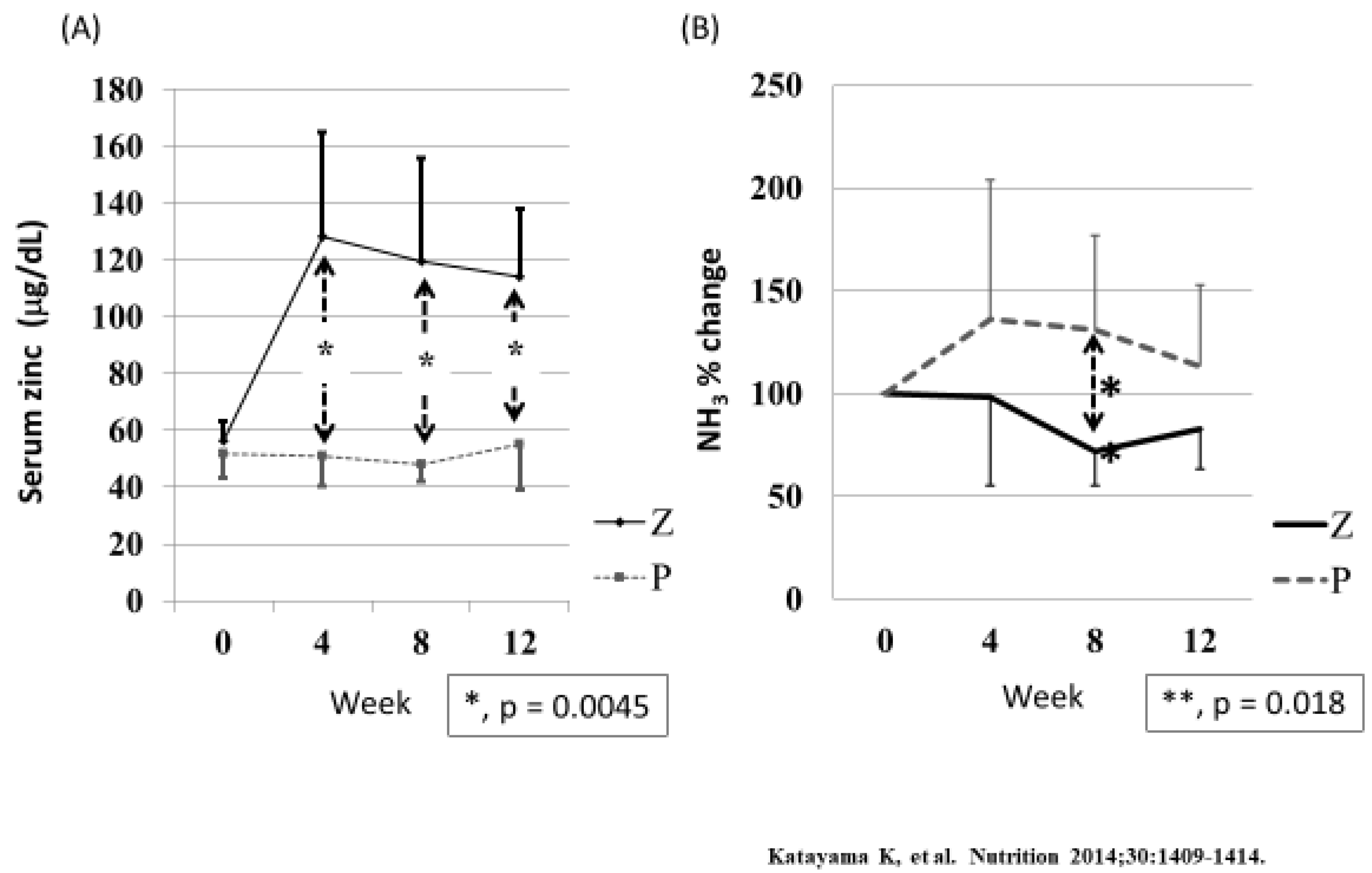
- Katayama, K.; Saito, M.; Kawaguchi, T.; Endo, R.; Sawara, K.; Nishiguchi, S.; Kato, A.; Kohgo, H.; Suzuki, K.; Sakaida, I.; et al. Effects of zinc on liver cirrhosis with hyperammonemia: A preliminary randomized, placebo-controlled double-blind trial. Nutrition 2014, 30, 1409–1414. [Google Scholar] [CrossRef]
- Reding, P.; Duchateau, J.; Bataille, C. Oral zinc supplementation improves hepatic encephalopathy. Results of a randomized controlled trial. Lancet 1984, 2, 493–495. [Google Scholar] [CrossRef]
- Marchesini, G.; Fabbri, A.; Bianchi, G.; Brizi, M.; Zoli, M. Zinc supplementation and amino acid-nitrogen metabolism in patients with advanced cirrhosis. Hepatology 1996, 23, 1084–1092. [Google Scholar] [CrossRef] [PubMed]

| Zinc deficiency can be reliably diagnosed by the four criteria below: |
| I. One or more symptoms and signs of zinc deficiency (dermatitis, aphthous stomatitis, hair loss, loss of appetite, taste disorder, hypogonadism in males, anemia, increased susceptibility to infection, growth disturbances in terms of weight and height in children, and low levels of serum alkaline phosphatase (ALP). However, serum ALP levels are not always low in patients with liver disease, osteoporosis, chronic kidney disease, or diabetes mellitus. |
| II. Ruling out of other diseases associated with the above symptoms or low serum ALP levels. For example, conditions such as contact dermatitis, atopic dermatitis, dermatitis due to deficiencies in vitamin A, biotin, or essential fatty acids, alopecia areata, hair-pulling, short stature due to growth hormone deficiency, familial short stature, Turner syndrome, and congenital hypophosphatasia should be ruled out. |
| III. Low serum zinc levels |
| III-1: <60 µg/dL: zinc deficiency |
| III-2: 60–80 µg/dL: marginal zinc deficiency |
| (Blood sampling is recommended in the morning under fasting conditions) |
| IV. Zinc treatment can be performed on patients who meet criteria I, II, and III. Symptoms in these patients can be improved with zinc treatment. |
| Food | Zinc Concentration (mg/100 g) | Amount of Zinc/Dish | |
|---|---|---|---|
| Weight | Zinc Concentration (mg) | ||
| Oyster | 13.2 | 5 pieces (60 g) | 7.9 |
| Pig liver | 6.9 | 70 g | 4.8 |
| Flat iron beef steak | 5.8 | 70 g | 4.1 |
| Beef liver | 3.8 | 70 g | 2.7 |
| Chicken liver | 3.3 | 1 meal (70 g) | 2.3 |
| Beef flank | 3.0 | 1 meal (70 g) | 2.1 |
| Scallops | 2.7 | 3 pieces (60 g) | 1.6 |
| Rice | 0.8 | 150 g | 1.2 |
| Grilled eel | 1.4 | Half of an eel (80 g) | 1.1 |
| Firm tofu | 0.6 | Half (150 g) | 0.9 |
| Natto | 1.9 | 1 piece (40 g) | 0.8 |
| Egg yolk | 4.2 | 1 piece (16 g) | 0.7 |
| Japanese noodles | 0.4 | 1 dish (180 g) | 0.7 |
| Processed cheese | 3.2 | 1 piece (20 g) | 0.6 |
| Powdered green tea | 6.3 | 1 spoonful (6 g) | 0.4 |
| Cocoa | 7.0 | 1 spoonful (6 g) | 0.4 |
| Quantitative Dysgeusia |
|---|
| taste reduction |
| taste loss |
| loss of a particular taste |
| Qualitative Dysgeusia |
| spontaneous dysgeusia |
| feeling that food tastes different |
| bad taste regardless of what is eaten |
| Mean Age | n | Zinc Prepararion Dosage (mg/Day) | Duration (Months) | Serum Zinc (μg/dL) | Improvement Rate (%) | Year & Reference | ||
|---|---|---|---|---|---|---|---|---|
| Before | After | |||||||
| Zinc gluconate | 55.1 | 35 | 67.5 | 3 | 80.5 ± 13.1 | 94.0 ± 24.6 | 77.1 | 1991 |
| Plabebo | 59.2 | 30 | ― | 76.8 ± 10.8 | 71.9 ± 10.9 | 60.0 | [39] | |
| Zinc picolinate | 55.2 | 37 | 87 | 3 | 71.0 | 81.6 | 75.6 | 2002 |
| Plabebo | 50.4 | 36 | ― | 71.5 | 72.3 | 44.4 | [40] | |
| Zinc gluconate | 61.1 | 24 | 20 | 3 | 72.28 ± 18.38 | 81.53 ± 19.61 | 50.0 | 2005 |
| Plabebo | 61.0 | 26 | ― | 67.90 ± 14.64 | 72.01 ± 10.22 | 25 | [41] | |
| Poraprezinc | 47.1 | 27 | 17 | 3 | 69.7 ± 12.1 | Δ 5.7 ± 13.5 | 63.0 | |
| 43.7 | 25 | 34 | 72.6 ± 13.9 | Δ 11.4 ± 16.6 | 51.9 | 2009 | ||
| 44.7 | 28 | 68 | 70.2 ± 11.7 | Δ 20.6 ± 21.3 | 80.0 | [42] | ||
| Plabebo | 44.9 | 27 | ― | 71.7 ± 13.6 | Δ 1.8 ± 12.7 | 89.3 | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kodama, H.; Tanaka, M.; Naito, Y.; Katayama, K.; Moriyama, M. Japan’s Practical Guidelines for Zinc Deficiency with a Particular Focus on Taste Disorders, Inflammatory Bowel Disease, and Liver Cirrhosis. Int. J. Mol. Sci. 2020, 21, 2941. https://doi.org/10.3390/ijms21082941
Kodama H, Tanaka M, Naito Y, Katayama K, Moriyama M. Japan’s Practical Guidelines for Zinc Deficiency with a Particular Focus on Taste Disorders, Inflammatory Bowel Disease, and Liver Cirrhosis. International Journal of Molecular Sciences. 2020; 21(8):2941. https://doi.org/10.3390/ijms21082941
Chicago/Turabian StyleKodama, Hiroko, Makoto Tanaka, Yuji Naito, Kazuhiro Katayama, and Mitsuhiko Moriyama. 2020. "Japan’s Practical Guidelines for Zinc Deficiency with a Particular Focus on Taste Disorders, Inflammatory Bowel Disease, and Liver Cirrhosis" International Journal of Molecular Sciences 21, no. 8: 2941. https://doi.org/10.3390/ijms21082941
APA StyleKodama, H., Tanaka, M., Naito, Y., Katayama, K., & Moriyama, M. (2020). Japan’s Practical Guidelines for Zinc Deficiency with a Particular Focus on Taste Disorders, Inflammatory Bowel Disease, and Liver Cirrhosis. International Journal of Molecular Sciences, 21(8), 2941. https://doi.org/10.3390/ijms21082941






