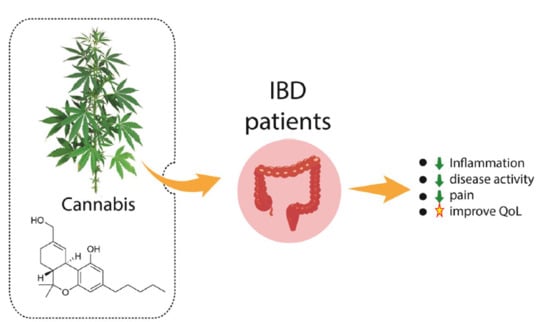
Cannabis and Canabidinoids on the Inflammatory Bowel Diseases: Going Beyond Misuse
Abstract
1. Introduction
2. Results
3. Discussion
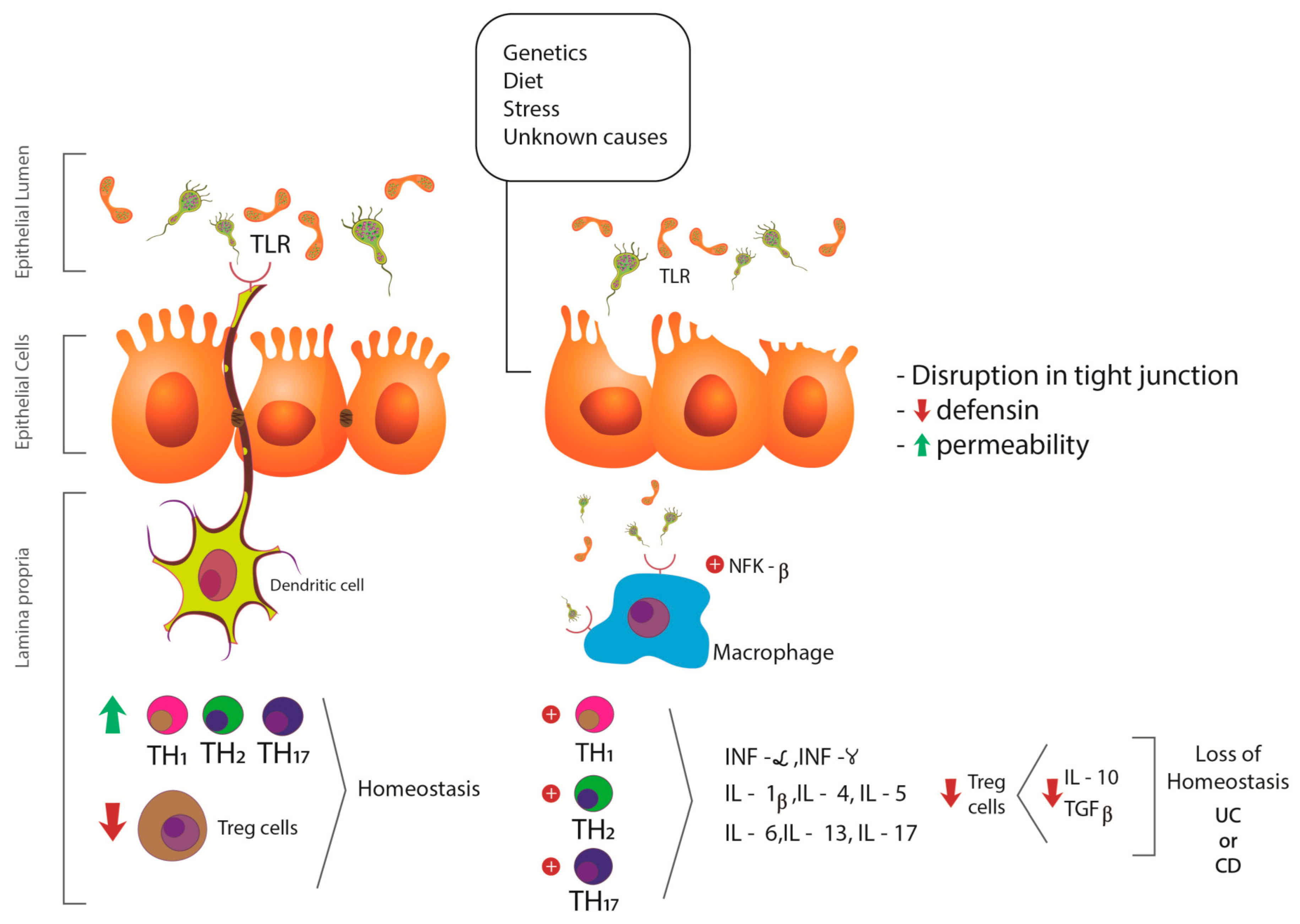
3.1. Inflammatory Bowel Diseases
3.2. Cannabis sativa and Endocannabinoid System
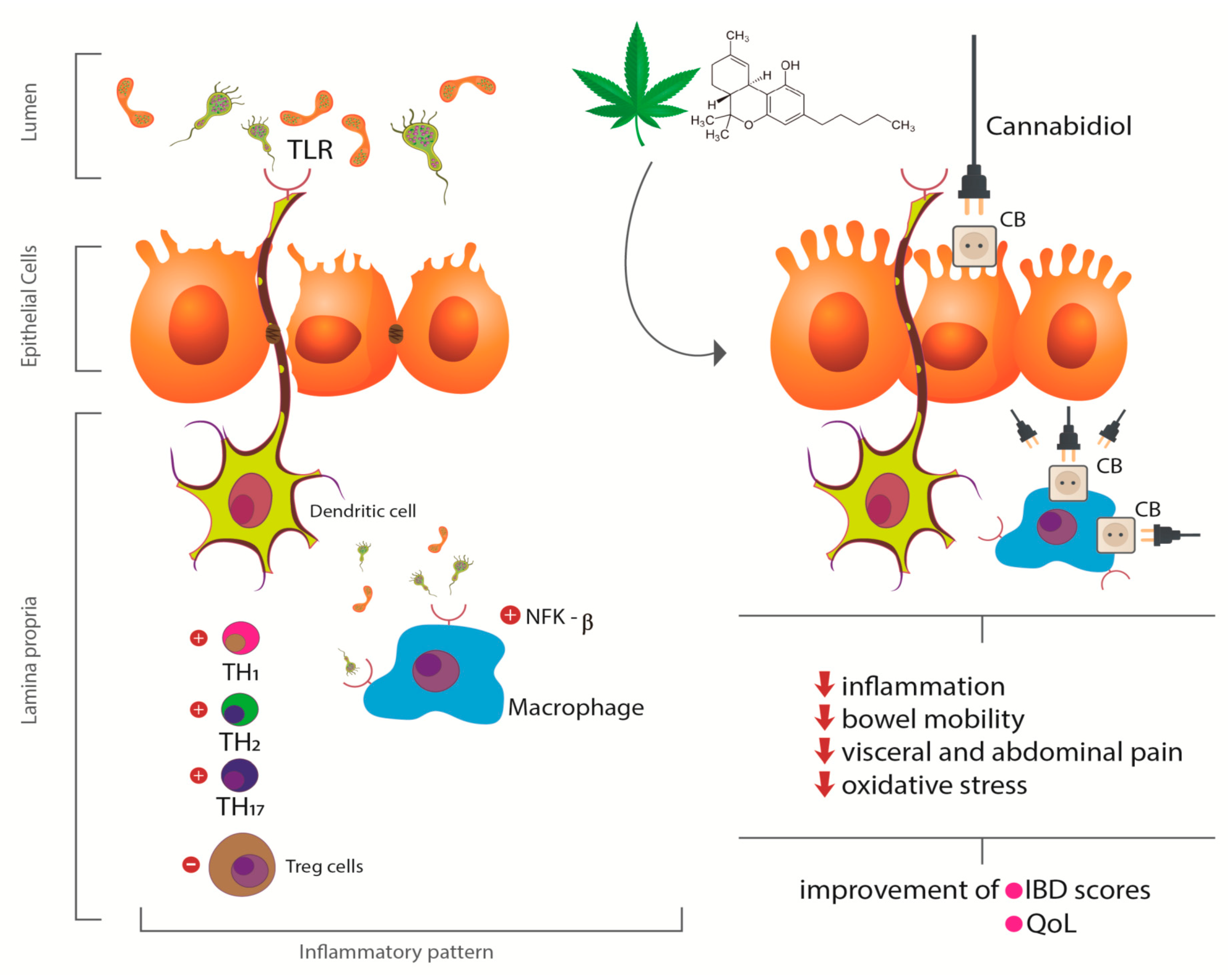
3.3. Inflammatory Bowel Diseases and Cannabis sativa
3.4. Ulcerative colitis and Cannabis sativa
3.5. Crohn´s Disease and Cannabis sativa
3.6. Cannabis sativa, Inflammatory Bowel Diseases, and Adverse Effects
3.7. The Use of Cannabis sativa per se
3.8. Final Comments
4. Material and Methods
4.1. Search Strategy
4.2. Focal Question
4.3. Eligibility criteria and PICO (Population, Intervention, Comparison, and Outcomes)
4.4. Data Extraction and Selection of the Studies
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pellino, G.; Keller, D.S.; Sampietro, G.M.; Carvello, M.; Celentano, V.; Coco, C.; Colombo, F.; Geccherle, A.; Luglio, G.; Rottoli, M.; et al. Inflammatory bowel disease position statement of the Italian Society of Colorectal Surgery (SICCR): Ulcerative colitis. Tech. Coloproctol. 2020. [Google Scholar] [CrossRef]
- Mak, J.W.Y.; So, J.; Tang, W.; Yip, T.C.F.; Leung, W.K.; Li, M.; Lo, F.H.; Ng, K.M.; Sze, S.F.; Leung, C.M.; et al. Cancer risk and chemoprevention in Chinese inflammatory bowel disease patients: A population-based cohort study. Scand. J. Gastroenterol. 2020, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Li, M.; Zheng, R.; Chen, X.; Xiang, J.; Wu, F.X.; Wang, J. Evaluation of pathway activation for a single sample toward inflammatory bowel disease classification. Front. Genet. 2019, 10, 1401. [Google Scholar] [CrossRef] [PubMed]
- Hansen, T.M.; Sabourin, B.C.; Oketola, B.; Bernstein, C.N.; Singh, H.; Targownik, L.E. Cannabis use in persons with inflammatory bowel disease and vulnerability to substance misuse. Inflamm. Bowel Dis. 2019. [Google Scholar] [CrossRef]
- Papamichael, K.; Cheifetz, A.S.; Irving, P.M. New role for azathioprine in case of switching anti-TNFs in IBD. Gut 2020. [Google Scholar] [CrossRef] [PubMed]
- Kafil, T.S.; Nguyen, T.M.; MacDonald, J.K.; Chande, N. Cannabis for the Treatment of Crohn’s Disease and Ulcerative Colitis: Evidence from Cochrane Reviews. Inflamm. Bowel Dis. 2019. [Google Scholar] [CrossRef]
- Downer, E.J. Anti-inflammatory Potential of Terpenes Present in Cannabis sativa L. ACS Chem. Neurosci. 2020. [Google Scholar] [CrossRef]
- Weinberger, A.H.; Zhu, J.; Lee, J.; Anastasiou, E.; Copeland, J.; Goodwin, R.D. Cannabis use among youth in the United States, 2004–2016: Faster rate of increase among youth with depression. Drug Alcohol Depend. 2020, 209, 107894. [Google Scholar] [CrossRef]
- Andersen, A.M.; Lei, M.K.; Beach, S.R.H.; Philibert, R.A.; Sinha, S.; Colgan, J.D. Cigarette and Cannabis Smoking Effects on GPR15+ Helper T Cell Levels in Peripheral Blood: Relationships with Epigenetic Biomarkers. Genes 2020, 11, 149. [Google Scholar] [CrossRef]
- Naftali, T.; Dor, M. Cannabis for the treatment of inflammatory bowel disease: A true medicine or a false promise? Rambam Maimonides Med. J. 2020, 11. [Google Scholar] [CrossRef]
- Cassano, T.; Villani, R.; Pace, L.; Carbone, A.; Bukke, V.N.; Orkisz, S.; Avolio, C.; Serviddio, G. From Cannabis sativa to Cannabidiol: Promising Therapeutic Candidate for the Treatment of Neurodegenerative Diseases. Front. Pharmacol. 2020, 11, 124. [Google Scholar] [CrossRef]
- Maayah, Z.H.; Takahara, S.; Ferdaoussi, M.; Dyck, J.R.B. The molecular mechanisms that underpin the biological benefit of full spectrum cannabis extract in the treatment of neuropathic pain and inflammation. Biochim. Biophys. Acta Mol. Basis Dis. 2020, 165771. [Google Scholar] [CrossRef] [PubMed]
- Arboleda, M.F.; Prosk, E.; Cyr, C.; Gamaoun, R.; Vigano, A. Medical cannabis in supportive cancer care: Lessons from Canada. Supportive Care Cancer 2020. [Google Scholar] [CrossRef]
- Kienzl, M.; Storr, M.; Schicho, R. Cannabinoids and opioids in the treatment of inflammatory bowel diseases. Clin. Transl. Gastroenterol. 2020, 11, e00120. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Ann. Intern. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef]
- Naftali, T.; Lev, L.B.; Yablecovitch, D.; Half, E.; Konikoff, F.M. Treatment of Crohn’s disease with cannabis: An observational study. Isr. Med Assoc. J. IMAJ 2011, 13, 455–458. [Google Scholar] [PubMed]
- Lahat, A.; Lang, A.; Ben-Horin, S. Impact of cannabis treatment on the quality of life, weight and clinical disease activity in inflammatory bowel disease patients: A pilot prospective study. Digestion 2012, 85, 1–8. [Google Scholar] [CrossRef]
- Naftali, T.; Bar-Lev Schleider, L.; Dotan, I.; Lansky, E.P.; Sklerovsky Benjaminov, F.; Konikoff, F.M. Cannabis induces a clinical response in patients with Crohn’s disease: A prospective placebo-controlled study. Clin. Gastroenterol. Hepatol. 2013, 11, 1276–1280. [Google Scholar] [CrossRef] [PubMed]
- Naftali, T.; Mechulam, R.; Marii, A.; Gabay, G.; Stein, A.; Bronshtain, M.; Laish, I.; Benjaminov, F.; Konikoff, F.M. Low-Dose Cannabidiol Is Safe but Not Effective in the Treatment for Crohn’s Disease, a Randomized Controlled Trial. Dig. Dis. Sci. 2017, 62, 1615–1620. [Google Scholar] [CrossRef]
- Kerlin, A.M.; Long, M.; Kappelman, M.; Martin, C.; Sandler, R.S. Profiles of Patients Who Use Marijuana for Inflammatory Bowel Disease. Dig. Dis. Sci. 2018, 63, 1600–1604. [Google Scholar] [CrossRef]
- Irving, P.M.; Iqbal, T.; Nwokolo, C.; Subramanian, S.; Bloom, S.; Prasad, N.; Hart, A.; Murray, C.; Lindsay, J.O.; Taylor, A.; et al. A Randomized, Double-blind, Placebo-controlled, Parallel-group, Pilot Study of Cannabidiol-rich Botanical Extract in the Symptomatic Treatment of Ulcerative Colitis. Inflamm. Bowel Dis. 2018, 24, 714–724. [Google Scholar] [CrossRef]
- Mbachi, C.; Attar, B.; Oyenubi, O.; Yuchen, W.; Efesomwan, A.; Paintsil, I.; Madhu, M.; Ajiboye, O.; Simons-Linares, C.R.; Trick, W.E.; et al. Association between cannabis use and complications related to ulcerative colitis in hospitalized patients: A propensity matched retrospective cohort study. Medicine 2019, 98, e16551. [Google Scholar] [CrossRef] [PubMed]
- Marton, L.T.; Goulart, R.A.; Carvalho, A.C.A.; Barbalho, S.M. Omega Fatty Acids and Inflammatory Bowel Diseases: An Overview. Int. J. Mol. Sci. 2019, 20, 4851. [Google Scholar] [CrossRef] [PubMed]
- Andreou, N.P.; Legaki, E.; Gazouli, M. Inflammatory bowel disease pathobiology: The role of the interferon signature. Ann. Gastroenterol. 2020, 33, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Asto, E.; Mendez, I.; Audivert, S.; Farran-Codina, A.; Espadaler, J. The efficacy of probiotics, prebiotic inulin-type fructans, and synbiotics in human ulcerative colitis: A systematic review and meta-analysis. Nutrients 2019, 11, 293. [Google Scholar] [CrossRef] [PubMed]
- Olivera, P.; Danese, S.; Pouillon, L.; Bonovas, S.; Peyrin-Biroulet, L. Effectiveness of golimumab in ulcerative colitis: A review of the real world evidence. Dig. Liver Dis. 2019, 51, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Mazieiro, R.; Frizon, R.R.; Barbalho, S.M.; Goulart, R.A. Is curcumin a possibility to treat inflammatory bowel diseases? J. Med. Food 2018, 21, 1077–1085. [Google Scholar] [CrossRef]
- Takenaka, K.; Kitazume, Y.; Fujii, T.; Tsuchiya, K.; Watanabe, M.; Ohtsuka, K. Objective evaluation for treat to target in Crohn’s disease. J. Gastroenterol. 2020. [Google Scholar] [CrossRef]
- Jangi, S.; Ruan, A.; Korzenik, J.; de Silva, P. South Asian patients with inflammatory bowel disease in the United States demonstrate more fistulizing and perianal crohn phenotype. Inflamm. Bowel Dis. 2020. [Google Scholar] [CrossRef]
- Markovic, B.S.; Kanjevac, T.; Harrell, C.R.; Gazdic, M.; Fellabaum, C.; Arsenijevic, N.; Volarevic, V. Molecular and Cellular Mechanisms Involved in Mesenchymal Stem Cell-Based Therapy of Inflammatory Bowel Diseases. Stem Cell Rev. Rep. 2018, 14, 153–165. [Google Scholar] [CrossRef]
- Skok, P.; Skok, K. Acute febrile neutrophilic dermatosis in a patient with Crohn’s disease: Case report and review of the literature. Acta Dermatovenerol. Alp. Pannonica Adriat. 2018, 27, 161–163. [Google Scholar] [CrossRef] [PubMed]
- Ambrose, T.; Simmons, A. Cannabis, Cannabinoids, and the Endocannabinoid System-Is there Therapeutic Potential for Inflammatory Bowel Disease? J. Crohn’s Colitis 2019, 13, 525–535. [Google Scholar] [CrossRef] [PubMed]
- Perisetti, A.; Rimu, A.H.; Khan, S.A.; Bansal, P.; Goyal, H. Role of cannabis in inflammatory bowel diseases. Ann. Gastroenterol. 2020, 33, 134–144. [Google Scholar] [CrossRef]
- Johal, H.; Devji, T.; Chang, Y.; Simone, J.; Vannabouathong, C.; Bhandari, M. Cannabinoids in Chronic Non-Cancer Pain: A Systematic Review and Meta-Analysis. Clinical medicine insights. Arthritis Musculoskelet. Disord. 2020, 13. [Google Scholar] [CrossRef]
- Paunescu, H.; Dima, L.; Ghita, I.; Coman, L.; Ifteni, P.I.; Fulga, I.; Coman, O.A. A Systematic Review of Clinical Studies on the Effect of Psychoactive Cannabinoids in Psychiatric Conditions in Alzheimer Dementia. Am. J. Ther. 2020. [Google Scholar] [CrossRef]
- Li, H.; Liu, Y.; Tian, D.; Tian, L.; Ju, X.; Qi, L.; Wang, Y.; Liang, C. Overview of cannabidiol (CBD) and its analogues: Structures, biological activities, and neuroprotective mechanisms in epilepsy and Alzheimer’s disease. Eur. J. Med. Chem. 2020, 192, 112163. [Google Scholar] [CrossRef]
- Kim, T.E.; Townsend, R.K.; Branch, C.L.; Romero-Sandoval, E.A.; Hsu, W. Cannabinoids in the Treatment of Back Pain. Neurosurgery 2020. [Google Scholar] [CrossRef]
- Capasso, R.; Orlando, P.; Pagano, E.; Aveta, T.; Buono, L.; Borrelli, F.; Di Marzo, V.; Izzo, A.A. Palmitoylethanolamide normalizes intestinal motility in a model of post-inflammatory accelerated transit: Involvement of CB(1) receptors and TRPV1 channels. Br. J. Pharmacol. 2014, 171, 4026–4037. [Google Scholar] [CrossRef]
- Massa, F.; Marsicano, G.; Hermann, H.; Cannich, A.; Monory, K.; Cravatt, B.F.; Ferri, G.L.; Sibaev, A.; Storr, M.; Lutz, B. The endogenous cannabinoid system protects against colonic inflammation. J. Clin. Investig. 2004, 113, 1202–1209. [Google Scholar] [CrossRef]
- Fichna, J.; Bawa, M.; Thakur, G.A.; Tichkule, R.; Makriyannis, A.; McCafferty, D.M.; Sharkey, K.A.; Storr, M. Cannabinoids alleviate experimentally induced intestinal inflammation by acting at central and peripheral receptors. PLoS ONE 2014, 9, e109115. [Google Scholar] [CrossRef]
- Nallathambi, R.; Mazuz, M.; Ion, A.; Selvaraj, G.; Weininger, S.; Fridlender, M.; Nasser, A.; Sagee, O.; Kumari, P.; Nemichenizer, D.; et al. Anti-Inflammatory Activity in Colon Models Is Derived from Delta9-Tetrahydrocannabinolic Acid That Interacts with Additional Compounds in Cannabis Extracts. Cannabis Cannabinoid Res. 2017, 2, 167–182. [Google Scholar] [CrossRef] [PubMed]
- Couch, D.G.; Tasker, C.; Theophilidou, E.; Lund, J.N.; O’Sullivan, S.E. Cannabidiol and palmitoylethanolamide are anti-inflammatory in the acutely inflamed human colon. Clin. Sci. (Lond. UK 1979) 2017, 131, 2611–2626. [Google Scholar] [CrossRef]
- Quezada, S.M.; Cross, R.K. Cannabis and Turmeric as Complementary Treatments for IBD and Other Digestive Diseases. Curr. Gastroenterol. Rep. 2019, 21, 2. [Google Scholar] [CrossRef] [PubMed]
- Mandelbaum, D.E.; de la Monte, S.M. Adverse Structural and Functional Effects of Marijuana on the Brain: Evidence Reviewed. Pediatric Neurol. 2017, 66, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Yanai, H.; Salomon, N.; Lahat, A. Complementary Therapies in Inflammatory Bowel Diseases. Curr. Gastroenterol. Rep. 2016, 18, 62. [Google Scholar] [CrossRef] [PubMed]
- Cheifetz, A.S.; Gianotti, R.; Luber, R.; Gibson, P.R. Complementary and Alternative Medicines Used by Patients With Inflammatory Bowel Diseases. Gastroenterology 2017, 152, 415–429. [Google Scholar] [CrossRef] [PubMed]
- Gentili, M.; Ronchetti, S.; Ricci, E.; Di Paola, R.; Gugliandolo, E.; Cuzzocrea, S.; Bereshchenko, O.; Migliorati, G.; Riccardi, C. Selective CB2 inverse agonist JTE907 drives T cell differentiation towards a Treg cell phenotype and ameliorates inflammation in a mouse model of inflammatory bowel disease. Pharmacol. Res. 2019, 141, 21–31. [Google Scholar] [CrossRef]
- Grof, C.P.L. Cannabis, from plant to pill. Br. J. Clin. Pharmacol. 2018, 84, 2463–2467. [Google Scholar] [CrossRef]
- Ben-Shabat, S.; Fride, E.; Sheskin, T.; Tamiri, T.; Rhee, M.H.; Vogel, Z.; Bisogno, T.; De Petrocellis, L.; Di Marzo, V.; Mechoulam, R. An entourage effect: Inactive endogenous fatty acid glycerol esters enhance 2-arachidonoyl-glycerol cannabinoid activity. Eur. J. Pharmacol. 1998, 353, 23–31. [Google Scholar] [CrossRef]
- Russo, E.B. Taming THC: Potential cannabis synergy and phytocannabinoid-terpenoid entourage effects. Br. J. Clin. Pharmacol. 2011, 163, 1344–1364. [Google Scholar] [CrossRef]
- Bergamaschi, M.M.; Queiroz, R.H.; Zuardi, A.W.; Crippa, J.A. Safety and side effects of cannabidiol, a Cannabis sativa constituent. Curr. Drug Saf. 2011, 6, 237–249. [Google Scholar] [CrossRef] [PubMed]
- Swaminath, A.; Berlin, E.P.; Cheifetz, A.; Hoffenberg, E.; Kinnucan, J.; Wingate, L.; Buchanan, S.; Zmeter, N.; Rubin, D.T. The Role of Cannabis in the Management of Inflammatory Bowel Disease: A Review of Clinical, Scientific, and Regulatory Information. Inflamm. Bowel Dis. 2019, 25, 427–435. [Google Scholar] [CrossRef] [PubMed]
- Picardo, S.; Kaplan, G.G.; Sharkey, K.A.; Seow, C.H. Insights into the role of cannabis in the management of inflammatory bowel disease. Ther. Adv. Gastroenterol. 2019, 12, 1756284819870977. [Google Scholar] [CrossRef] [PubMed]
- Jamal, N.; Korman, J.; Musing, M.; Malavade, A.; Coleman, B.L.; Siddiqui, N.; Friedman, Z. Effects of pre-operative recreational smoked cannabis use on opioid consumption following inflammatory bowel disease surgery: A historical cohort study. Eur. J. Anaesthesiol. 2019, 36, 705–706. [Google Scholar] [CrossRef]
- Lachenmeier, D.W.; Habel, S.; Fischer, B.; Herbi, F.; Zerbe, Y.; Bock, V.; Rajcic de Rezende, T.; Walch, S.G.; Sproll, C. Are side effects of cannabidiol (CBD) products caused by tetrahydrocannabinol (THC) contamination? F1000Research 2019, 8, 1394. [Google Scholar] [CrossRef]
- Millan-Guerrero, R.O.; Isais-Millan, S. Cannabis and the exocannabinoid and endocannabinoid systems. Their use and controversies. Gac. Med. Mex. 2019, 155, 471–474. [Google Scholar] [CrossRef]
- Naftali, T. Cannabis for Inflammatory Bowel Diseases: Should We Follow the Wisdom of the Crowd? Isr. Med Assoc. J. IMAJ 2019, 21, 756–758. [Google Scholar]
- Atalay, S.; Jarocka-Karpowicz, I.; Skrzydlewska, E. Antioxidative and Anti-Inflammatory Properties of Cannabidiol. Antioxidants 2019, 9, 21. [Google Scholar] [CrossRef]
- Maroon, J.; Bost, J. Review of the neurological benefits of phytocannabinoids. Surg. Neurol. Int. 2018, 9, 91. [Google Scholar] [CrossRef]
- ElSohly, M.A.; Radwan, M.M.; Gul, W.; Chandra, S.; Galal, A. Phytochemistry of Cannabis sativa L. Prog. Chem. Org. Nat. Prod. 2017, 103, 1–36. [Google Scholar] [CrossRef]
- Meccariello, R.; Santoro, A.; D’Angelo, S.; Morrone, R.; Fasano, S.; Viggiano, A.; Pierantoni, R. The Epigenetics of the Endocannabinoid System. Int. J. Mol. Sci. 2020, 21, 1113. [Google Scholar] [CrossRef] [PubMed]
- Turner, S.E.; Williams, C.M.; Iversen, L.; Whalley, B.J. Molecular Pharmacology of Phytocannabinoids. Prog. Chem. Org. Nat. Prod. 2017, 103, 61–101. [Google Scholar] [CrossRef] [PubMed]

| Reference | Type and Country of the Study | Patients/Intervention | Outcomes | Side Effects | Main Conclusions |
|---|---|---|---|---|---|
| Mbachi et al. 2019 [22] | Population-based cohort study/United States) | 39,806 individuals with UC (23–69 y; 26,391 men). Cannabis users: 298; non-users: 39,508 | Evaluation of clinical end-points showed lower bowel obstruction (6.4% versus 12.0%), and partial or total colectomy (4.4% versus 9.6%). Cannabis users presented shorter hospital lengths of stay. | Not reported | Cannabis may mitigate some of the well-described complications of UC among hospitalized patients. |
| Kerlin et al. 2018 [20] | Longitudinal, internet-based cohort study/Israel | 1666 Individuals (CD: 1045; UC: 121; 116 women; 503 men) who completed a baseline health survey with updates every 6 m. Cannabis users (recreational or prescription): 114; non-users: 1552. The patients who complete a survey on marijuana were included. | The majority of marijuana users (80.7%) perceived improvement in pain (68%), appetite (49%), anxiety (48%), fatigue (26%), stool frequency (23%), weight gain (20%), and blood in the stool (5%)). | Anxiety, pain, depression, and lower social satisfaction. | Users reported clinical improvement of IBD symptoms, but they reported more anxiety, depression, and pain. Marijuana use may be higher in patients with IBD symptoms not well treated by conventional medical approaches. |
| Irving et al. 2018 [18] | Multicenter, randomized, double-blind, placebo-controlled study/12 weeks/United Kingdom | 60 mild to moderate UC patients refractory to 5-ASA (16 women; 44 men). Placebo group n = 31 (42.8 ± 12.9 y) and treated group (n = 29; 44.8 ± 15.1 y) that received oral hard gelatin capsules with 50 mg CBD-RBE, 2xd, 30 min before morning and evening meals. Patients entered a 2-week dose-escalation period and were required to reach their maximum tolerated dose of up to 250 mg, 2xd/6 weeks. | Remission was observed in both groups at about equal levels. Treated group reported a reduction in the severity of the disease, abdominal pain, and reported feeling better. NO differences were found for stool, bleeding, and levels of IL-2, IL-6, and TNF-α. | Dizziness and somnolence | Treated group showed clinical remission but without statistical significance. Patients treated with cannabis reported improvement in the quality of life, showing potential to treated UC. |
| Naftali et al. 2017 [16] | Double-blind, randomized placebo-controlled trial/8 weeks/Israel | 19 patients with active DC (8women; 11men). Placebo group: n = 9 (20–50 y), and treated group: n = 10 (18–75 y) that received oral CBD oil (05 mg/about 0.3 mg/kg) or placebo 2xd. | No clinical improvement of CDAI was observed after oral CBD. Hemoglobin, albumin, CRP, and kidney and liver function tests remained unchanged with the treatment. | Side effects did not differ between the groups. | Patients showed clinical remission without statistical significance, and any other beneficial effect was reported. |
| Naftali et al. 2013 [15] | Double-blind, randomized, placebo-controlled trial/8 weeks/Israel | 21 patients with active CD (9 women; 12men). Placebo group: n = 10 (26–48 y); and treated group: n = 11 (29–63 y). The recommendation was inhalation of cannabis, 2xd, in the form of cigarettes containing 11.5 mg of THC. The cigarettes of the placebo group contained cannabis flowers. | Complete remission (CDAI score <150) was achieved by 5/11 subjects in the cannabis group and 1/10 in the placebo group. A decrease in CDAI score of >100 was observed in 10/11 subjects in the cannabis group and 4/10 in the placebo group. Moreover, it improved appetite and sleep. | No significant differences in side effects (confusion, sleepiness, and nausea) for both groups. | Patients showed clinical remission without statistical significance. However, the administration of inhaled cannabis provided benefits in clinical response and steroid dependence. |
| Lahat et al. 2012 [14] | Open-label, prospective and single-arm trial/3 months/Israel | 13 patients (4 women; 9 men); CD: 11 (28–62 y), UC: 2 (28–31 y) were instructed to use cigarettes with 50 g of dry processed cannabis (inhaled) whenever they observed pain. They were guided to take up to 3 inhalations from the prepared cigarettes each time for 3 months. | After treatment, patients reported improvement of daily activities, decreased pain, improvement of general health perception, patients’ ability to work and to maintain social activities. Patients also presented weight gain. | Not reported | Administration of inhaled cannabis can promote clinical improvement in patients with IBD. |
| Naftali et al. 2011 [13] | Retrospective observational study/Israel | 30 patients (26 men and 4 women; 21–65 y) with CD using cannabis (because of lack of response to conventional therapy) were interviewed. Four patients used recreational cannabis. | Most patients used inhaled cannabis (joints) or through water (bongs), and all of them reported that the use of cannabis reduced disease activity (Harvey–Bradshaw scale) and the use of other medications. | Not reported | The use of cannabis shows positive effects on CD activity. |
| Phytocompound | CB1 | CB2 | Others | ||
|---|---|---|---|---|---|
 9δ-tetrahydrocannabinol | ++ | + |
| References [10,33,53,59,60,61,62] | CB1 receptor: Central, peripheral and enteric nervous system
Immune tissues (macrophages, neutrophils, epithelial cells, B cells and T cells)
|
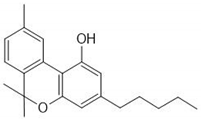 Cannabinol | + | ++ |
| ||
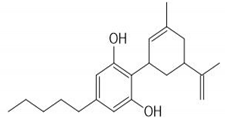 Cannabidiol | + | + |
| ||
 Cannabigerol | + | NR |
| ||
 Cannabicrhomene | I | I |
|
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carvalho, A.C.A.d.; Souza, G.A.d.; Marqui, S.V.d.; Guiguer, É.L.; Araújo, A.C.; Rubira, C.J.; Goulart, R.d.A.; Flato, U.A.P.; Bueno, P.C.d.S.; Buchaim, R.L.; et al. Cannabis and Canabidinoids on the Inflammatory Bowel Diseases: Going Beyond Misuse. Int. J. Mol. Sci. 2020, 21, 2940. https://doi.org/10.3390/ijms21082940
Carvalho ACAd, Souza GAd, Marqui SVd, Guiguer ÉL, Araújo AC, Rubira CJ, Goulart RdA, Flato UAP, Bueno PCdS, Buchaim RL, et al. Cannabis and Canabidinoids on the Inflammatory Bowel Diseases: Going Beyond Misuse. International Journal of Molecular Sciences. 2020; 21(8):2940. https://doi.org/10.3390/ijms21082940
Chicago/Turabian StyleCarvalho, Antonelly Cassio Alves de, Gabriela Achete de Souza, Samylla Vaz de Marqui, Élen Landgraf Guiguer, Adriano Cressoni Araújo, Claudio José Rubira, Ricardo de Alvares Goulart, Uri Adrian Prync Flato, Patricia Cincotto dos Santos Bueno, Rogério Leone Buchaim, and et al. 2020. "Cannabis and Canabidinoids on the Inflammatory Bowel Diseases: Going Beyond Misuse" International Journal of Molecular Sciences 21, no. 8: 2940. https://doi.org/10.3390/ijms21082940
APA StyleCarvalho, A. C. A. d., Souza, G. A. d., Marqui, S. V. d., Guiguer, É. L., Araújo, A. C., Rubira, C. J., Goulart, R. d. A., Flato, U. A. P., Bueno, P. C. d. S., Buchaim, R. L., & Barbalho, S. M. (2020). Cannabis and Canabidinoids on the Inflammatory Bowel Diseases: Going Beyond Misuse. International Journal of Molecular Sciences, 21(8), 2940. https://doi.org/10.3390/ijms21082940