Sperm Cohort-Specific Zinc Signature Acquisition and Capacitation-Induced Zinc Flux Regulate Sperm-Oviduct and Sperm-Zona Pellucida Interactions
Abstract
1. Introduction
2. Results
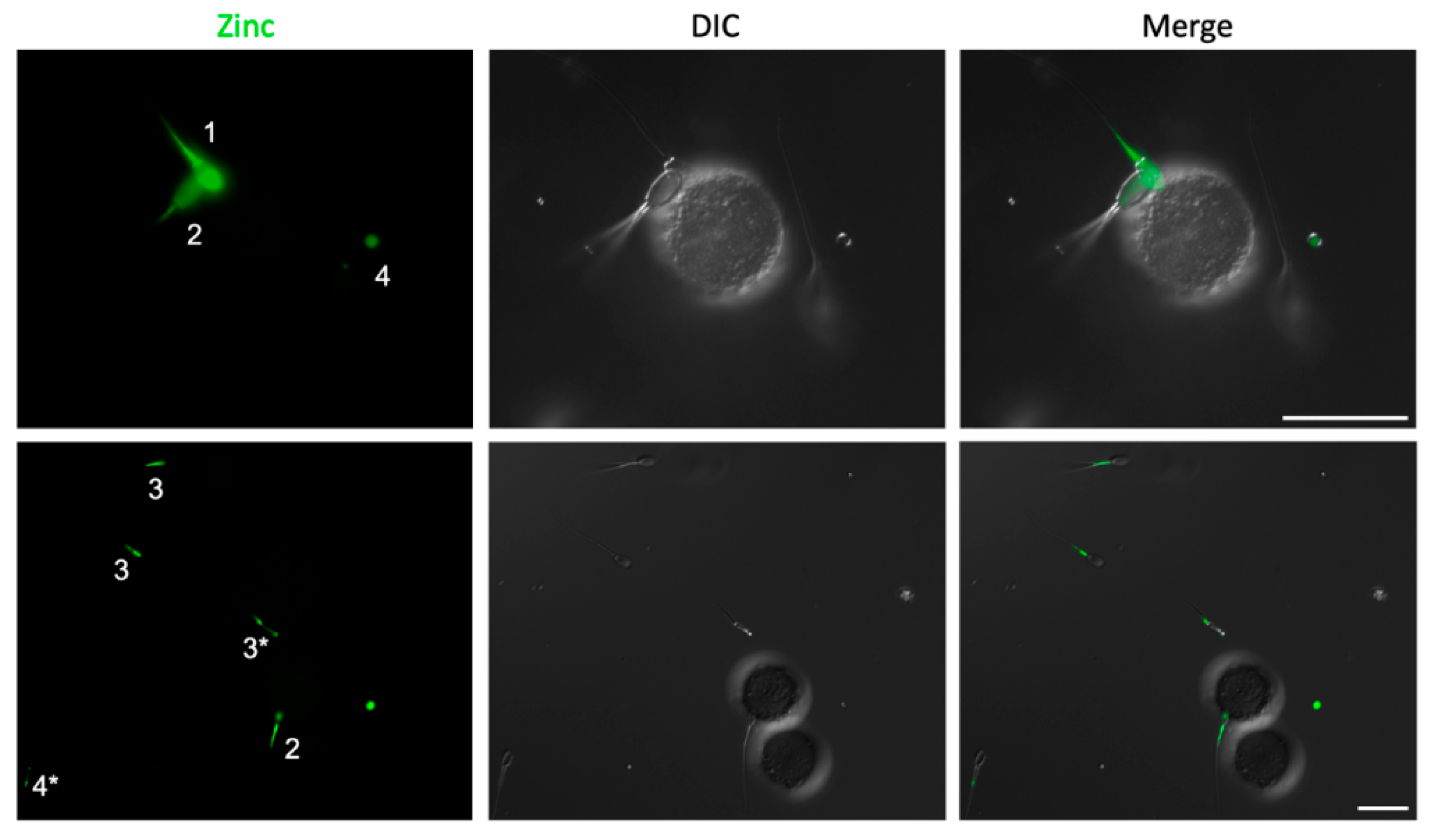
2.1. Sperm Zinc Signature is Established by Ejaculatory Sequence
2.2. Spermatozoa Posessing Zinc Signatures 1 and 2 Bind Glycans of the Oviductal Sperm Reservior
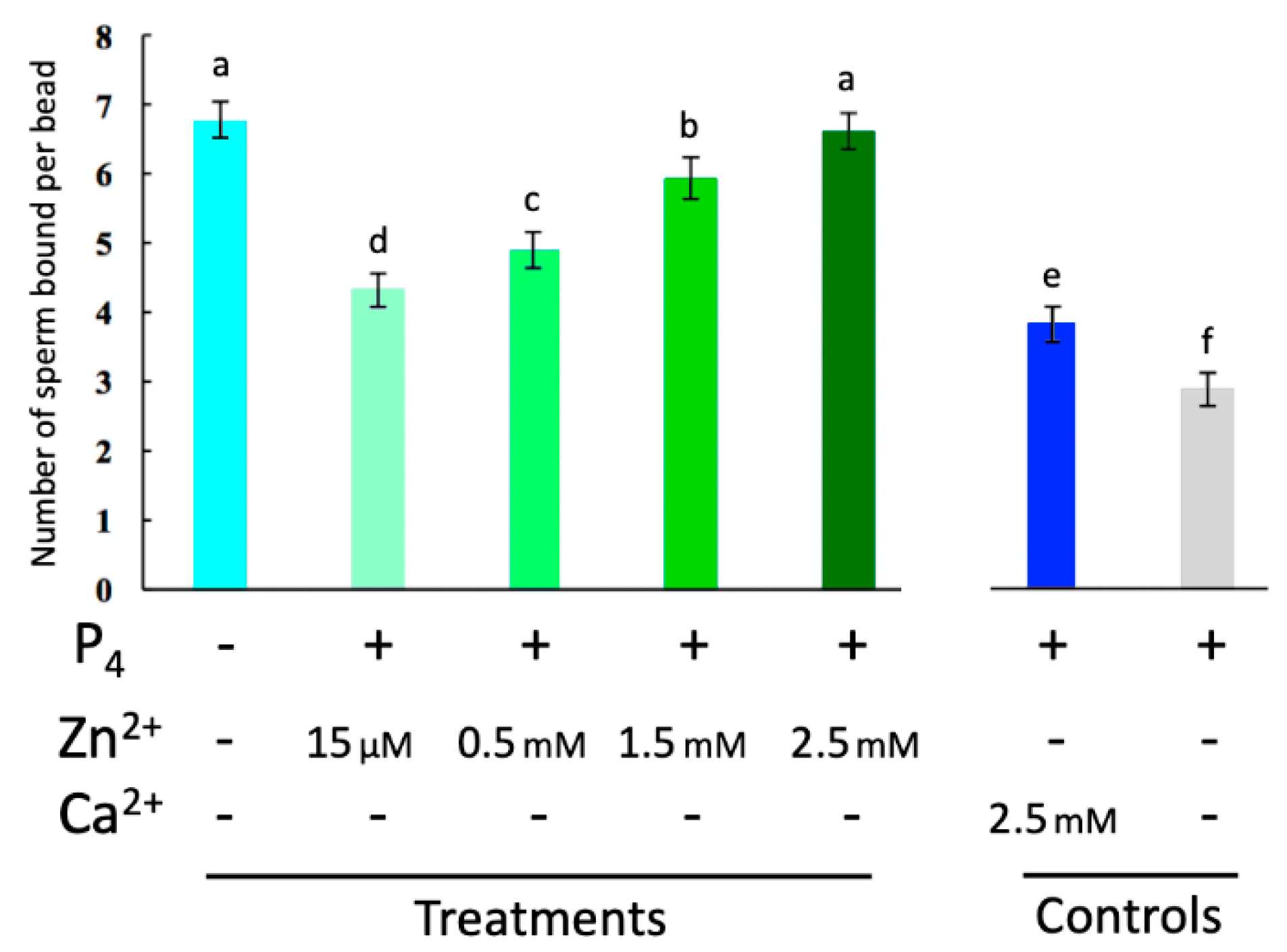
2.3. Externally Added Zinc Ions Inhibit Sperm Release from Glycans of the Oviductal Sperm Reservior
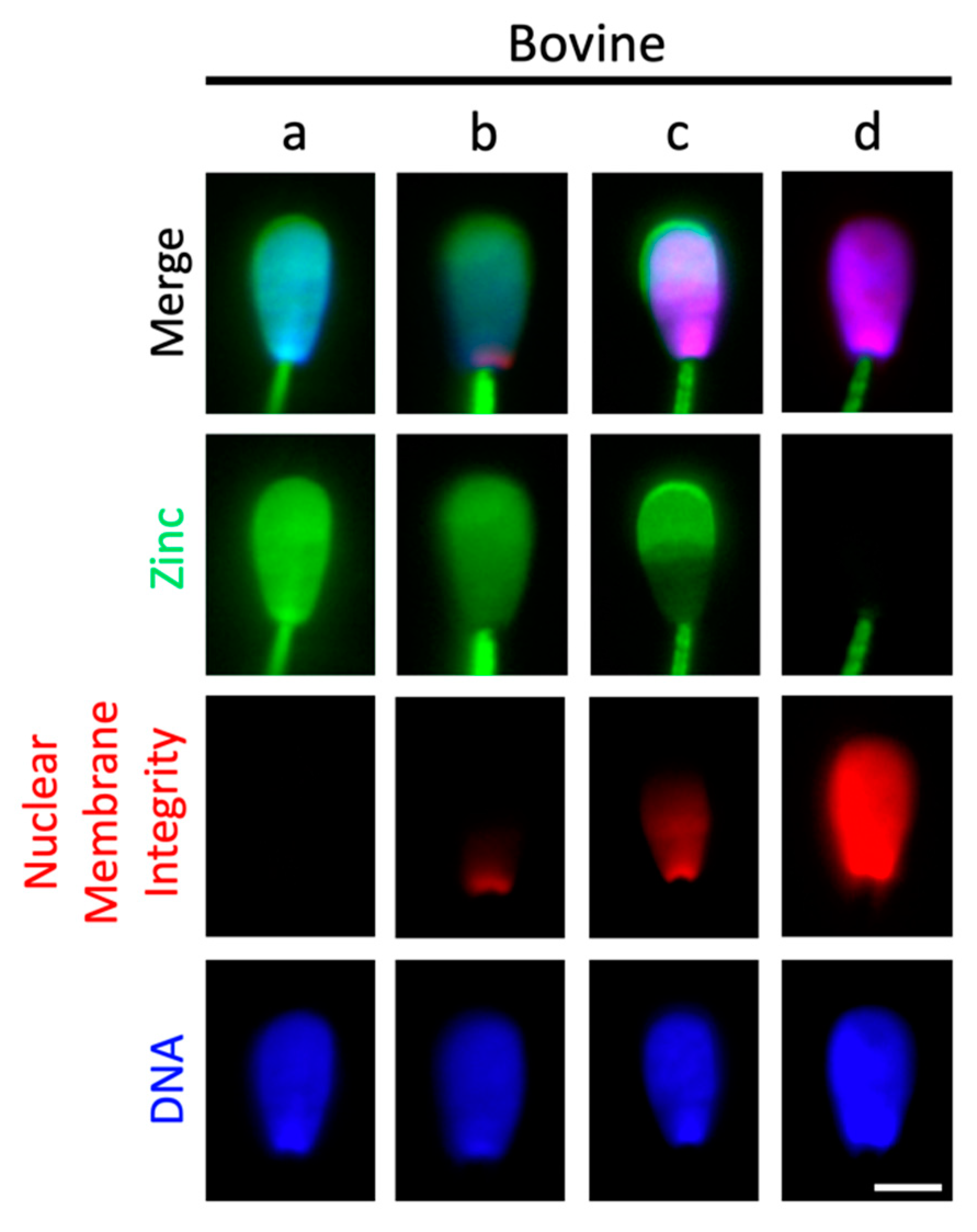
2.4. Sperm Head Zn-Efflux Occurs in a Posterior-to-Anterior Modification Wave and Localizes to Sperm Acrosome
2.5. External Zinc Inhibits the Activity of Sperm Proteinases
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Models, Semen Collection, and Processing
4.3. Collection and Processing of Spermatozoa for Zinc Signature Fraction Analysis
4.4. Multiplex Fluorescence Probing
4.5. Epifluorescence Microscopy Imaging
4.6. Image-Based Flow Cytometric Data Acquisition
4.7. IBFC Data Analysis
4.8. Fluorogenic Proteasomal Activity Assay
4.9. Sperm Oviductal Glycan Binding Assay
4.10. Zymography Sample Preparation and Assay
4.11. Statistics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| Zn2+ | Zinc ions |
| ZP | Zona pellucida |
| AI | Artificial insemination |
| MMP2 | Matrix metalloproteinase-2 |
| FZ3 | FluoZin ™-3 |
| bi-SiaLN | biantennary 6-sialylated lactosamine 11-oligosaccharide |
| suLeX | 3-O-sulfated Lewis X trisaccharide |
| DIC | differential interference contrast |
| P4 | Progesterone |
| PI | Propidium iodide |
| IAM | Inner acrosomal membrane |
| PNA | Lectin peanut agglutinin |
| ZnR | Zn-sensing receptor |
| HVCN1 | hydrogen voltage channel 1 |
| H33342 | Hoechst 33342 |
| Epox | Epoxomicin |
| CLBL | clasto-Lactacystin β-Lactone |
| IBFC | Image-based flow cytometry |
| FN | Fibronectin |
| NP-40 | Nonidet P-40 |
| TrX | Triton X-100 |
| SAS | Statistical Analysis Software |
| GLM | General Linear Model |
| GLIMMIX | Generalized linear mixed model |
| NIFA | National Institute of Food and Agriculture |
| USDA | U.S. Department of Agriculture |
| NIH | National Institute of Health |
| NSERC | Natural Sciences and Engineering Research Council |
References
- Chu, D.S. Zinc: A small molecule with a big impact on sperm function. PLoS Biol. 2018, 16, e2006204. [Google Scholar] [CrossRef] [PubMed]
- Andreychenko, S.V.; Klepko, A.V.; Gorban, L.V.; Motryna, O.A.; Grubska, L.V.; Trofimenko, O.V. Post-Chornobyl remote radiation effects on human sperm and seminal plasma characteristics. Exp. Oncol. 2016, 38, 245–251. [Google Scholar] [CrossRef]
- Nenkova, G.; Petrov, L.; Alexandrova, A. Role of Trace Elements for Oxidative Status and Quality of Human Sperm. Balk. Med. J. 2017, 34, 343. [Google Scholar] [CrossRef] [PubMed]
- Colagar, A.H.; Marzony, E.T.; Chaichi, M.J. Zinc levels in seminal plasma are associated with sperm quality in fertile and infertile men. Nutr. Res. 2009, 29, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Fallah, A.; Mohammad-Hasani, A.; Colagar, A.H. Zinc is an Essential Element for Male Fertility: A Review of Zn Roles in Men’s Health, Germination, Sperm Quality, and Fertilization. J. Reprod. Infertil. 2018, 19, 69–80. [Google Scholar]
- Zhao, J.; Dong, X.; Hu, X.; Long, Z.; Wang, L.; Liu, Q.; Sun, B.; Wang, Q.; Wu, Q.; Li, L. Zinc levels in seminal plasma and their correlation with male infertility: A systematic review and meta-analysis. Sci. Rep. 2016, 6, 22386. [Google Scholar] [CrossRef]
- Kerns, K.; Zigo, M.; Sutovsky, P. Zinc: A Necessary Ion for Mammalian Sperm Fertilization Competency. Int. J. Mol. Sci. 2018, 19, 4097. [Google Scholar] [CrossRef] [PubMed]
- Kerns, K.; Zigo, M.; Drobnis, E.Z.; Sutovsky, M.; Sutovsky, P. Zinc ion flux during mammalian sperm capacitation. Nat. Commun. 2018, 9, 2061. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.C. Fertilizing capacity of spermatozoa deposited into the fallopian tubes. Nature 1951, 168, 697–698. [Google Scholar] [CrossRef]
- Austin, C.R. Observations on the penetration of the sperm in the mammalian egg. Aust. J. Sci. Res. Ser. B Biol. Sci. 1951, 4, 581–596. [Google Scholar] [CrossRef]
- Jaiswal, B.S.; Eisenbach, M. Capacitation. In Fertilization; Hardy, D.M., Ed.; Academic Press: Cambridge, MA, USA, 2002; pp. 57–117. [Google Scholar]
- Knox, R.V. Semen Processing, Extending & Storage for Artificial Insemination in Swine; University of Illinois, Extension Publication: Urbana-Champaign, IL, USA, 2007. [Google Scholar]
- Zhu, J.; Xu, X.; Cosgrove, J.R.; Foxcroft, G.R. Effects of semen plasma from different fractions of individual ejaculates on IVF in pigs. Theriogenology 2000, 54, 1443–1452. [Google Scholar] [CrossRef]
- Hebles, M.; Dorado, M.; Gallardo, M.; González-Martínez, M.; Sánchez-Martín, P. Seminal quality in the first fraction of ejaculate. Syst. Biol. Reprod. Med. 2015, 61, 113–116. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Martinez, H.; Saravia, F.; Wallgren, M.; Roca, J.; Pena, F.J. Influence of seminal plasma on the kinematics of boar spermatozoa during freezing. Theriogenology 2008, 70, 1242–1250. [Google Scholar] [CrossRef] [PubMed]
- Wallgren, M.; Saravia, F.; Rodriguez-Martinez, H. The vanguard sperm cohort of the boar ejaculate is overrepresented in the tubal sperm reservoir in vivo. J. Reprod. Dev. 2010, 56, 68–72. [Google Scholar] [CrossRef]
- Winters, R.A.; Nettenstrom, L.M.; Lopez, D.G.; Willenburg, K.L.; Vishwanath, R.; Bovin, N.V.; Miller, D.J. Effect of sorting boar spermatozoa by sex chromosomes on oviduct cell binding. Theriogenology 2018, 108, 22–28. [Google Scholar] [CrossRef]
- Gervasi, M.G.; Visconti, P.E. Chang’s meaning of capacitation: A molecular perspective. Mol. Reprod. Dev. 2016, 83, 860–874. [Google Scholar] [CrossRef]
- Backstrom, J.R.; Miller, C.A.; Tokes, Z.A. Characterization of neutral proteinases from Alzheimer-affected and control brain specimens: Identification of calcium-dependent metalloproteinases from the hippocampus. J. Neurochem. 1992, 58, 983–992. [Google Scholar] [CrossRef]
- Kerns, K.; Morales, P.; Sutovsky, P. Regulation of Sperm Capacitation by the 26S Proteasome: An Emerging New Paradigm in Spermatology. Biol. Reprod. 2016, 94, 117. [Google Scholar] [CrossRef]
- Sutovsky, P. Sperm proteasome and fertilization. Reproduction 2011, 142, 1–14. [Google Scholar] [CrossRef]
- Machado, S.A.; Kadirvel, G.; Daigneault, B.W.; Korneli, C.; Miller, P.; Bovin, N.; Miller, D.J. LewisX-containing glycans on the porcine oviductal epithelium contribute to formation of the sperm reservoir. Biol. Reprod. 2014, 91, 140. [Google Scholar] [CrossRef]
- Machado, S.A.; Sharif, M.; Wang, H.; Bovin, N.; Miller, D.J. Release of Porcine Sperm from Oviduct Cells is Stimulated by Progesterone and Requires CatSper. Sci. Rep. 2019, 9, 19546. [Google Scholar] [CrossRef] [PubMed]
- Boursnell, J.C.; Baronos, S.; Briggs, P.A.; Butler, E.J. The concentrations of zinc in boar seminal plasma and vesicular secretion in relation to those of nitrogenous substances, citrate, galactose and fructose. J. Reprod. Fertil. 1972, 29, 215–227. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Liu, D.-Y.; Sie, B.-S.; Liu, M.-L.; Agresta, F.; Baker, H.G. Relationship between seminal plasma zinc concentration and spermatozoa–zona pellucida binding and the ZP-induced acrosome reaction in subfertile men. Asian J. Androl. 2009, 11, 499–507. [Google Scholar] [CrossRef] [PubMed]
- Patek, E.; Hagenfeldt, K. Trace elements in the human fallopian tube epithelium. Copper, zinc, manganese and potassium in the menstrual cycle. Int. J. Fertil. 1974, 19, 85–88. [Google Scholar] [PubMed]
- Ménézo, Y.; Pluntz, L.; Chouteau, J.; Gurgan, T.; Demirol, A.; Dalleac, A.; Benkhalifa, M. Zinc concentrations in serum and follicular fluid during ovarian stimulation and expression of Zn2+ transporters in human oocytes and cumulus cells. Reprod. BioMed. Online 2011, 22, 647–652. [Google Scholar] [CrossRef]
- Brssow, K.P.; Rtky, J.; Rodriguez-Martinez, H. Fertilization and Early Embryonic Development in the Porcine Fallopian Tube. Reprod. Domest. Anim. 2008, 43, 245–251. [Google Scholar] [CrossRef]
- Steven, F.S.; Griffin, M.M.; Chantler, E.N. Inhibition of human and bovine sperm acrosin by divalent metal ions. Possible role of zinc as a regulator of acrosin activity. Int. J. Androl. 1982, 5, 401–412. [Google Scholar] [CrossRef]
- Baba, T.; Azuma, S.; Kashiwabara, S.; Toyoda, Y. Sperm from mice carrying a targeted mutation of the acrosin gene can penetrate the oocyte zona pellucida and effect fertilization. J. Biol. Chem. 1994, 269, 31845–31849. [Google Scholar]
- Zimmerman, S.W.; Manandhar, G.; Yi, Y.J.; Gupta, S.K.; Sutovsky, M.; Odhiambo, J.F.; Powell, M.D.; Miller, D.J.; Sutovsky, P. Sperm proteasomes degrade sperm receptor on the egg zona pellucida during mammalian fertilization. PLoS ONE 2011, 6, e17256. [Google Scholar] [CrossRef]
- Yokota, N.; Sawada, H. Sperm proteasomes are responsible for the acrosome reaction and sperm penetration of the vitelline envelope during fertilization of the sea urchin Pseudocentrotus depressus. Dev. Biol. 2007, 308, 222–231. [Google Scholar] [CrossRef]
- Ferrer, M.; Rodriguez, H.; Zara, L.; Yu, Y.; Xu, W.; Oko, R. MMP2 and acrosin are major proteinases associated with the inner acrosomal membrane and may cooperate in sperm penetration of the zona pellucida during fertilization. Cell Tissue Res. 2012, 349, 881–895. [Google Scholar] [CrossRef] [PubMed]
- Bjorndahl, L.; Kvist, U. Sequence of ejaculation affects the spermatozoon as a carrier and its message. Reprod. BioMed. Online 2003, 7, 440–448. [Google Scholar] [CrossRef]
- Huggins, C.; Johnson, A. Chemical observation on fluids of the seminal tract. Am. J. Physiol. 1933, 1933, 574–581. [Google Scholar] [CrossRef]
- Lavon, U.; Boursnel, J.C. The split ejaculate of the boar: Contributions of the epidiymides and seminal vesicles. J. Reprod. Fertil. 1975, 42, 541. [Google Scholar] [CrossRef] [PubMed]
- Kvist, U.; Kjellberg, S.; Björndahl, L.; Soufir, J.C.; Arver, S. Seminal fluid from men with agenesis of the Wolffian ducts: Zinc-binding properties and effects on sperm chromatin stability. Int. J. Androl. 1990, 13, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Campanero-Rhodes, M.A.; Menéndez, M.; Sáiz, J.L.; Sanz, L.; Calvete, J.J.; Solís, D. Zinc Ions Induce the Unfolding and Self-Association of Boar Spermadhesin PSP-I, a Protein with a Single CUB Domain Architecture, and Promote Its Binding to Heparin. Biochemistry 2006, 45, 8227–8235. [Google Scholar] [CrossRef]
- Rodriguez-Martinez, H.; Kvist, U.; Saravia, F.; Wallgren, M.; Johannisson, A.; Sanz, L.; Pena, F.J.; Martinez, E.A.; Roca, J.; Vazquez, J.M.; et al. The physiological roles of the boar ejaculate. Soc. Reprod. Fertil. Suppl. 2009, 66, 1–21. [Google Scholar]
- Robertson, S.A. Seminal plasma and male factor signalling in the female reproductive tract. Cell Tissue Res. 2005, 322, 43–52. [Google Scholar] [CrossRef]
- Crawford, G.; Ray, A.; Gudi, A.; Shah, A.; Homburg, R. The role of seminal plasma for improved outcomes during in vitro fertilization treatment: Review of the literature and meta-analysis. Hum. Reprod. Update 2015, 21, 275–284. [Google Scholar] [CrossRef]
- Michailov, Y.; Ickowicz, D.; Breitbart, H. Zn2+-stimulation of sperm capacitation and of the acrosome reaction is mediated by EGFR activation. Dev. Biol. 2014, 396, 246–255. [Google Scholar] [CrossRef]
- Aitken, R.J.; Baker, M.A.; Nixon, B. Are sperm capacitation and apoptosis the opposite ends of a continuum driven by oxidative stress? Asian J. Androl. 2015, 17, 633–639. [Google Scholar] [CrossRef] [PubMed]
- Lishko, P.V.; Kirichok, Y. The role of Hv1 and CatSper channels in sperm activation. J. Physiol. 2010, 588, 4667–4672. [Google Scholar] [CrossRef]
- Suarez, S.S. Regulation of sperm storage and movement in the mammalian oviduct. Int. J. Dev. Biol. 2008, 52, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.; Kim, C.H.; Kim, J.H.; Lee, J.; Choi, J.J.; Chen, Z.A.; Lee, M.G.; Chung, K.C.; Hsu, C.Y.; Ahn, Y.S. Pyrrolidine dithiocarbamate and zinc inhibit proteasome-dependent proteolysis. Exp. Cell Res. 2004, 298, 229–238. [Google Scholar] [CrossRef]
- Amici, M.; Forti, K.; Nobili, C.; Lupidi, G.; Angeletti, M.; Fioretti, E.; Eleuteri, A. Effect of neurotoxic metal ions on the proteolytic activities of the 20S proteasome from bovine brain. JBIC J. Biol. Inorg. Chem. 2002, 7, 750–756. [Google Scholar] [CrossRef]
- Zigo, M.; Jonakova, V.; Manaskova-Postlerova, P.; Kerns, K.; Sutovsky, P. Ubiquitin-proteasome system participates in the de-aggregation of spermadhesins and DQH protein during boar sperm capacitation. Reproduction 2019, 157, 283–295. [Google Scholar] [CrossRef]
- McCall, K.A.; Huang, C.-C.; Fierke, C.A. Function and Mechanism of Zinc Metalloenzymes. J. Nutr. 2000, 130, 1437S–1446S. [Google Scholar] [CrossRef] [PubMed]
- The UniProt Consortium. UniProt: The universal protein knowledgebase. Nucleic Acids Res. 2018, 46, 2699. [Google Scholar] [CrossRef] [PubMed]
- Vettakkorumakankav, N.N.; Ananthanarayanan, V.S. Ca2+ and Zn2+ binding properties of peptide substrates of vertebrate collagenase, MMP-1. Biochim. Biophys. Acta Protein Struct. Mol. Enzymol. 1999, 1432, 356–370. [Google Scholar] [CrossRef]
- Lippa, E. The eye: Topical carbonic anhydrase inhibitors. In The Carbonic Anhydrases: Cellular Physiology and Molecular Genetics; Dodgson, S., Tashian, R., Gros, G., Carter, N., Eds.; Plenum Press: New York, NY, USA, 1991; pp. 171–182. [Google Scholar]
- Lacorte, L.M.; Rinaldi, J.C.; Justulin, L.A., Jr.; Delella, F.K.; Moroz, A.; Felisbino, S.L. Cadmium exposure inhibits MMP2 and MMP9 activities in the prostate and testis. Biochem. Biophys. Res. Commun. 2015, 457, 538–541. [Google Scholar] [CrossRef]
- Guidobaldi, H.A.; Cubilla, M.; Moreno, A.; Molino, M.V.; Bahamondes, L.; Giojalas, L.C. Sperm chemorepulsion, a supplementary mechanism to regulate fertilization. Hum. Reprod. 2017, 32, 1560–1573. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.T.; Yang, R.C.; Wu, H.T.; Wang, C.N.; Pang, J.H. Zinc-chelation contributes to the anti-angiogenic effect of ellagic acid on inhibiting MMP-2 activity, cell migration and tube formation. PLoS ONE 2011, 6, e18986. [Google Scholar] [CrossRef] [PubMed]
- Yi, Y.J.; Zimmerman, S.W.; Manandhar, G.; Odhiambo, J.F.; Kennedy, C.; Jonáková, V.; Maňásková-Postlerová, P.; Sutovsky, M.; Park, C.S.; Sutovsky, P. Ubiquitin-activating enzyme (UBA1) is required for sperm capacitation, acrosomal exocytosis and sperm-egg coat penetration during porcine fertilization. Int. J. Androl. 2012, 35, 196–210. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, C.E.; Krieger, K.B.; Sutovsky, M.; Xu, W.; Vargovic, P.; Didion, B.A.; Ellersieck, M.R.; Hennessy, M.E.; Verstegen, J.; Oko, R.; et al. Protein expression pattern of PAWP in bull spermatozoa is associated with sperm quality and fertility following artificial insemination. Mol. Reprod. Dev. 2014, 81, 436–449. [Google Scholar] [CrossRef]
- Kisselev, A.F.; Goldberg, A.L. Monitoring activity and inhibition of 26S proteasomes with fluorogenic peptide substrates. Methods Enzymol. 2005, 398, 364–378. [Google Scholar]
- Yi, Y.J.; Manandhar, G.; Sutovsky, M.; Li, R.; Jonakova, V.; Oko, R.; Park, C.S.; Prather, R.S.; Sutovsky, P. Ubiquitin C-terminal hydrolase-activity is involved in sperm acrosomal function and anti-polyspermy defense during porcine fertilization. Biol. Reprod. 2007, 77, 780–793. [Google Scholar] [CrossRef]
- Bovin, N.V.; Korchagina, E.Y.; Zemlyanukhina, T.V.; Byramova, N.E.; Galanina, O.E.; Zemlyakov, A.E.; Ivanov, A.E.; Zubov, V.P.; Mochalova, L.V. Synthesis of polymeric neoglycoconjugates based on N-substituted polyacrylamide. Glycoconj. J. 1993, 10, 142–151. [Google Scholar] [CrossRef]
- Littell, R.C.; Henry, P.R.; Ammerman, C.B. Statistical analysis of repeated measures data using SAS procedures. J. Anim. Sci. 1998, 76, 1216–1231. [Google Scholar] [CrossRef]
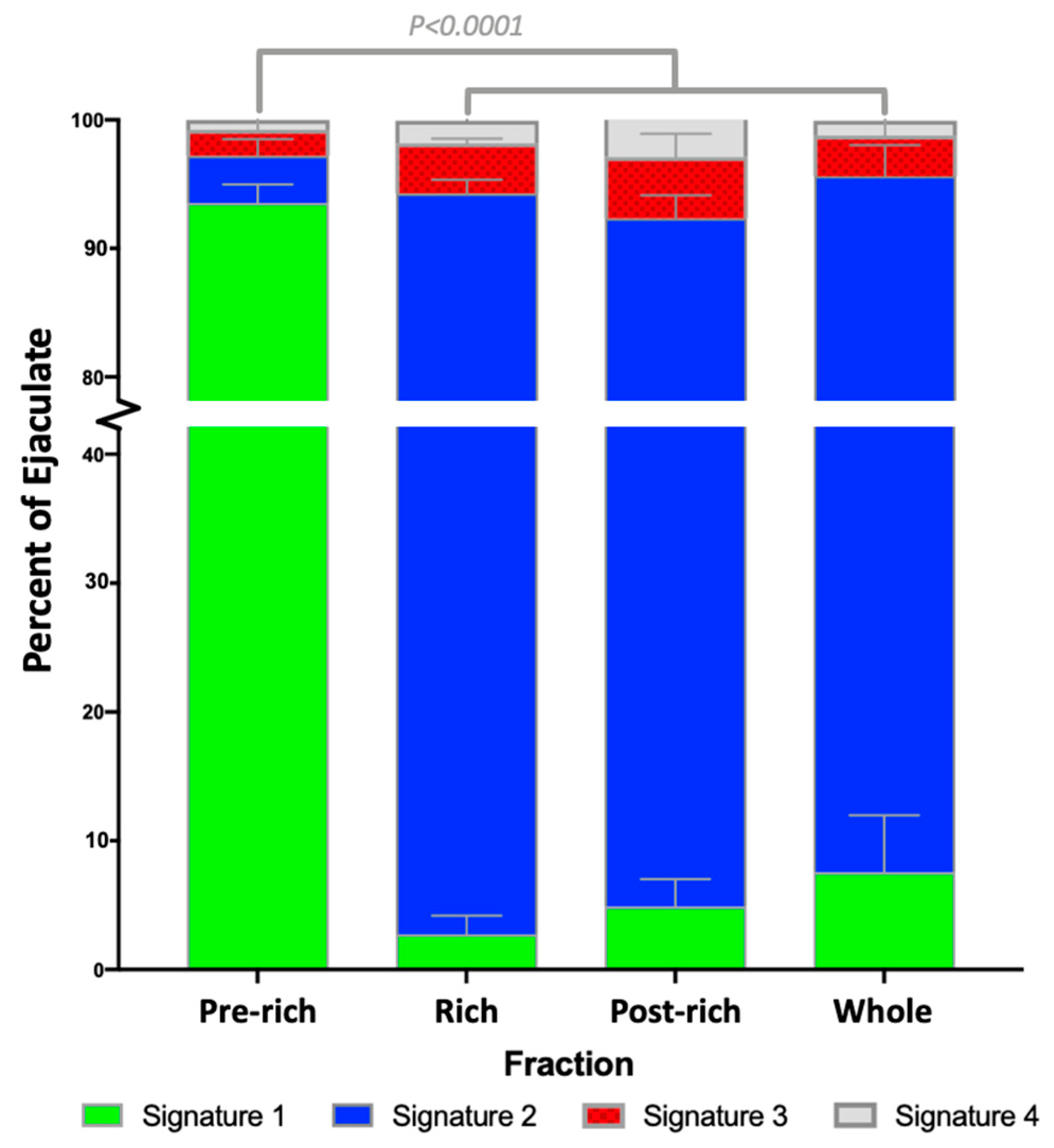
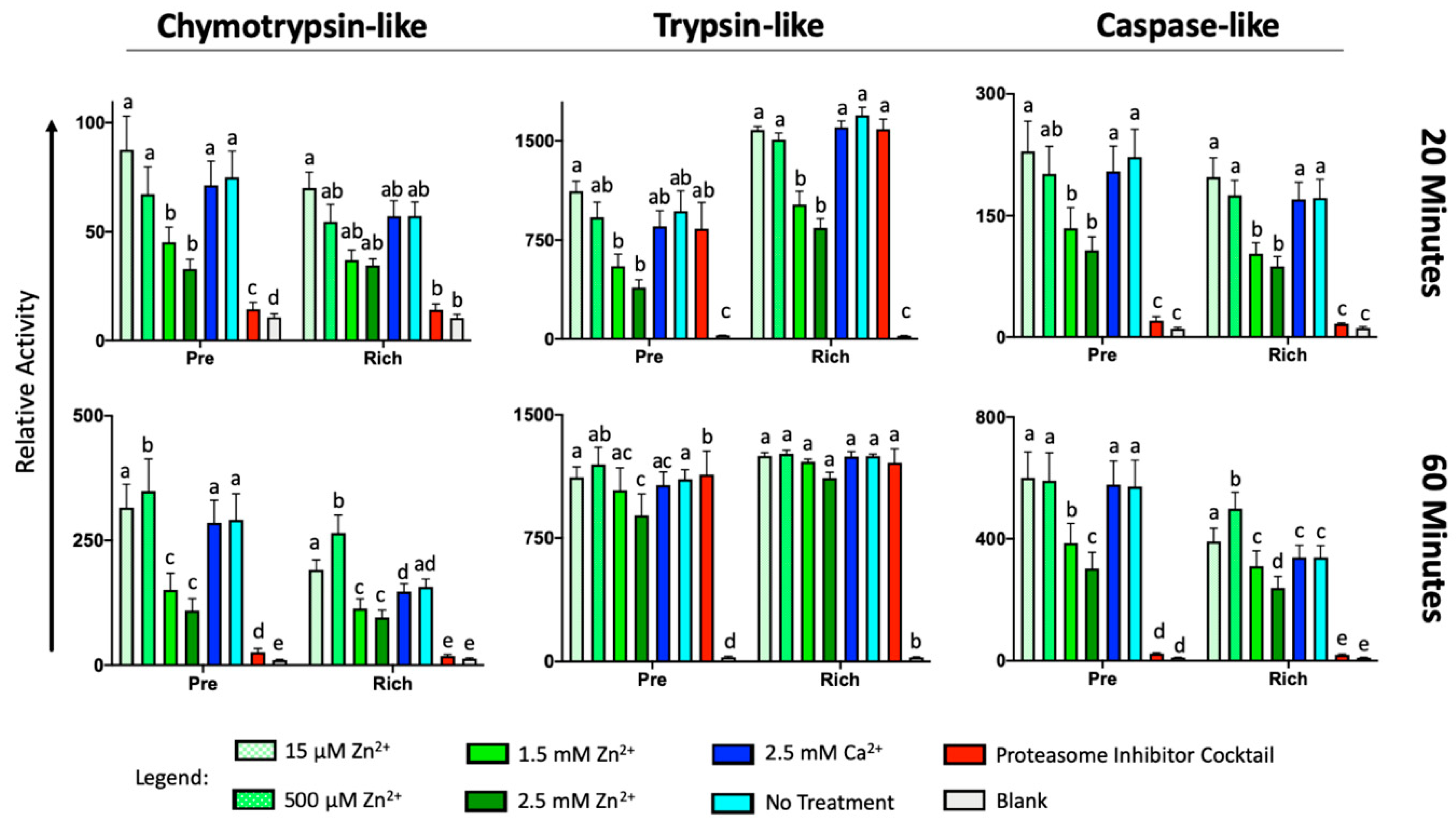
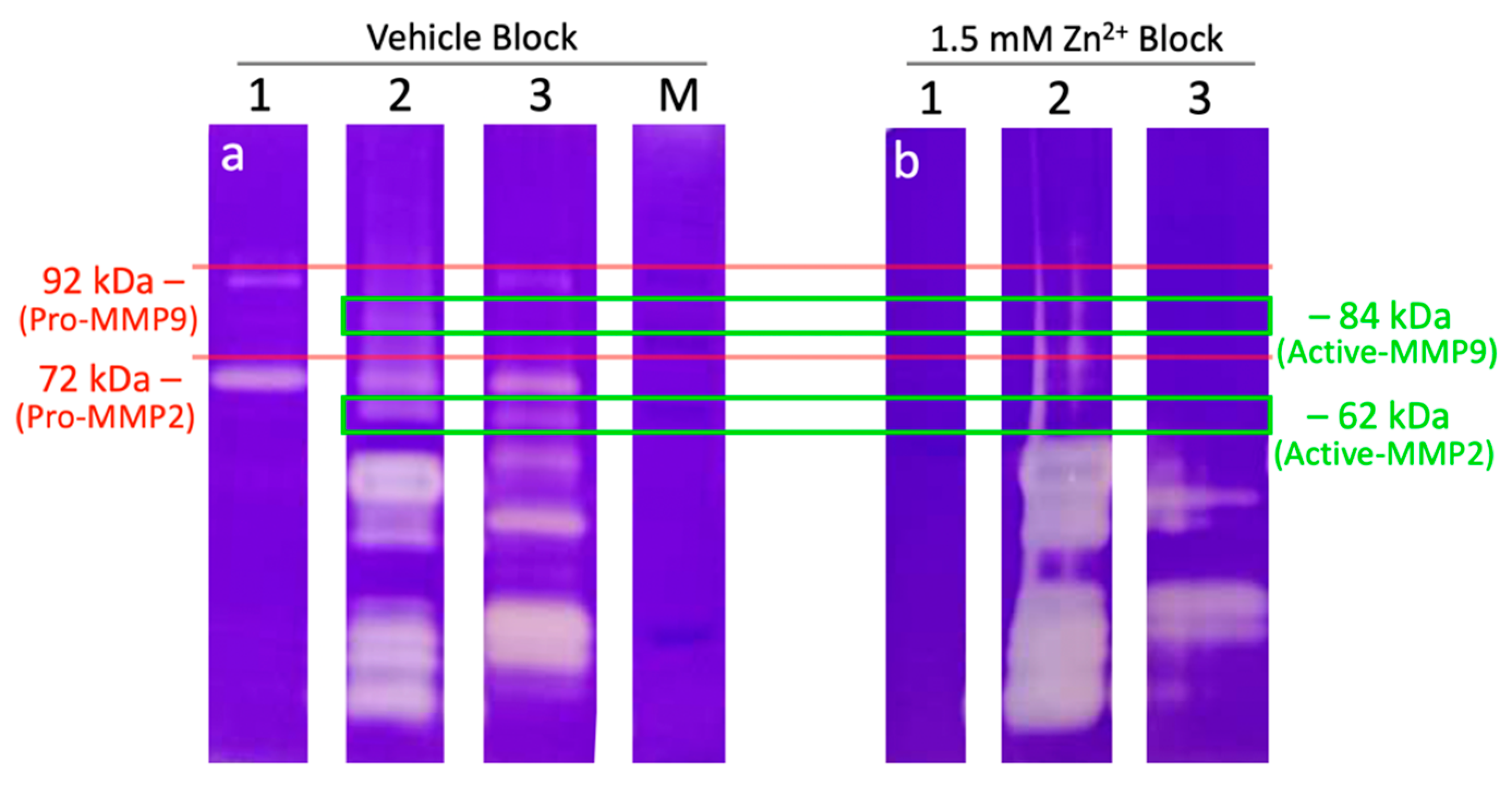
| Fraction | Signature 1 | Signature 2 | Signature 3 | Signature 4 |
|---|---|---|---|---|
| Pre | 93.5 ± 1.4% Aa | 3.7 ± 1.3% Ab | 2.0 ± 0.8% b | 0.8 ± 0.4% b |
| Rich | 2.8 ± 1.5% Ba | 91.5 ± 1.1% Bb | 3.9 ± 0.4% a | 1.8 ± 0.5% a |
| Post | 4.9 ± 2.1% Ba | 87.5 ± 1.8% Bb | 4.8 ± 1.8% c | 2.9 ± 0.6% c |
| Whole | 7.6 ± 4.4% Ba | 88.0 ± 2.4% Bb | 3.2 ± 1.5% c | 1.2 ± 0.6% a |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kerns, K.; Sharif, M.; Zigo, M.; Xu, W.; Hamilton, L.E.; Sutovsky, M.; Ellersieck, M.; Drobnis, E.Z.; Bovin, N.; Oko, R.; et al. Sperm Cohort-Specific Zinc Signature Acquisition and Capacitation-Induced Zinc Flux Regulate Sperm-Oviduct and Sperm-Zona Pellucida Interactions. Int. J. Mol. Sci. 2020, 21, 2121. https://doi.org/10.3390/ijms21062121
Kerns K, Sharif M, Zigo M, Xu W, Hamilton LE, Sutovsky M, Ellersieck M, Drobnis EZ, Bovin N, Oko R, et al. Sperm Cohort-Specific Zinc Signature Acquisition and Capacitation-Induced Zinc Flux Regulate Sperm-Oviduct and Sperm-Zona Pellucida Interactions. International Journal of Molecular Sciences. 2020; 21(6):2121. https://doi.org/10.3390/ijms21062121
Chicago/Turabian StyleKerns, Karl, Momal Sharif, Michal Zigo, Wei Xu, Lauren E. Hamilton, Miriam Sutovsky, Mark Ellersieck, Erma Z. Drobnis, Nicolai Bovin, Richard Oko, and et al. 2020. "Sperm Cohort-Specific Zinc Signature Acquisition and Capacitation-Induced Zinc Flux Regulate Sperm-Oviduct and Sperm-Zona Pellucida Interactions" International Journal of Molecular Sciences 21, no. 6: 2121. https://doi.org/10.3390/ijms21062121
APA StyleKerns, K., Sharif, M., Zigo, M., Xu, W., Hamilton, L. E., Sutovsky, M., Ellersieck, M., Drobnis, E. Z., Bovin, N., Oko, R., Miller, D., & Sutovsky, P. (2020). Sperm Cohort-Specific Zinc Signature Acquisition and Capacitation-Induced Zinc Flux Regulate Sperm-Oviduct and Sperm-Zona Pellucida Interactions. International Journal of Molecular Sciences, 21(6), 2121. https://doi.org/10.3390/ijms21062121







