Evolution of ASC Immunophenotypical Subsets During Expansion In Vitro
Abstract
1. Introduction
2. Results
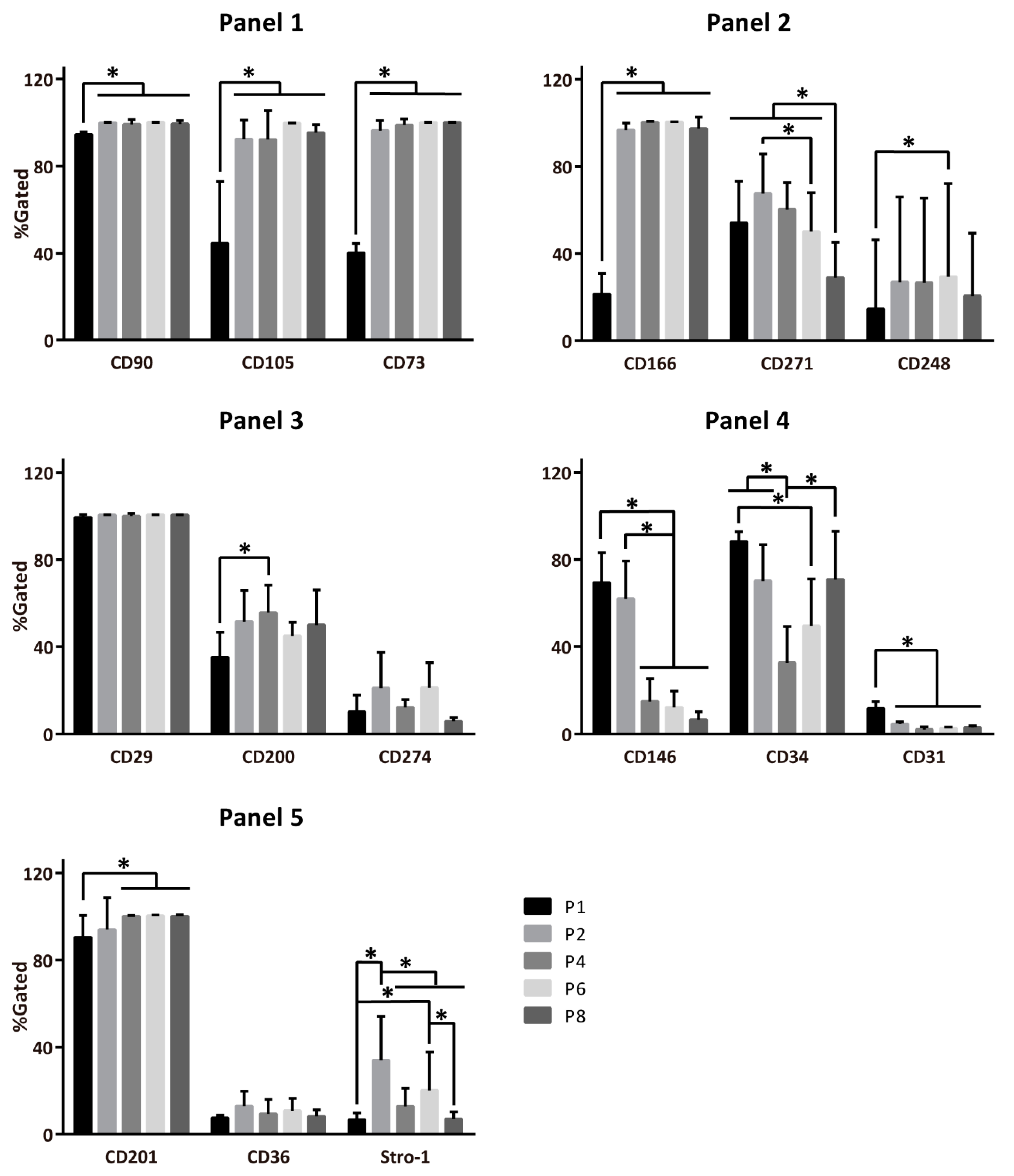
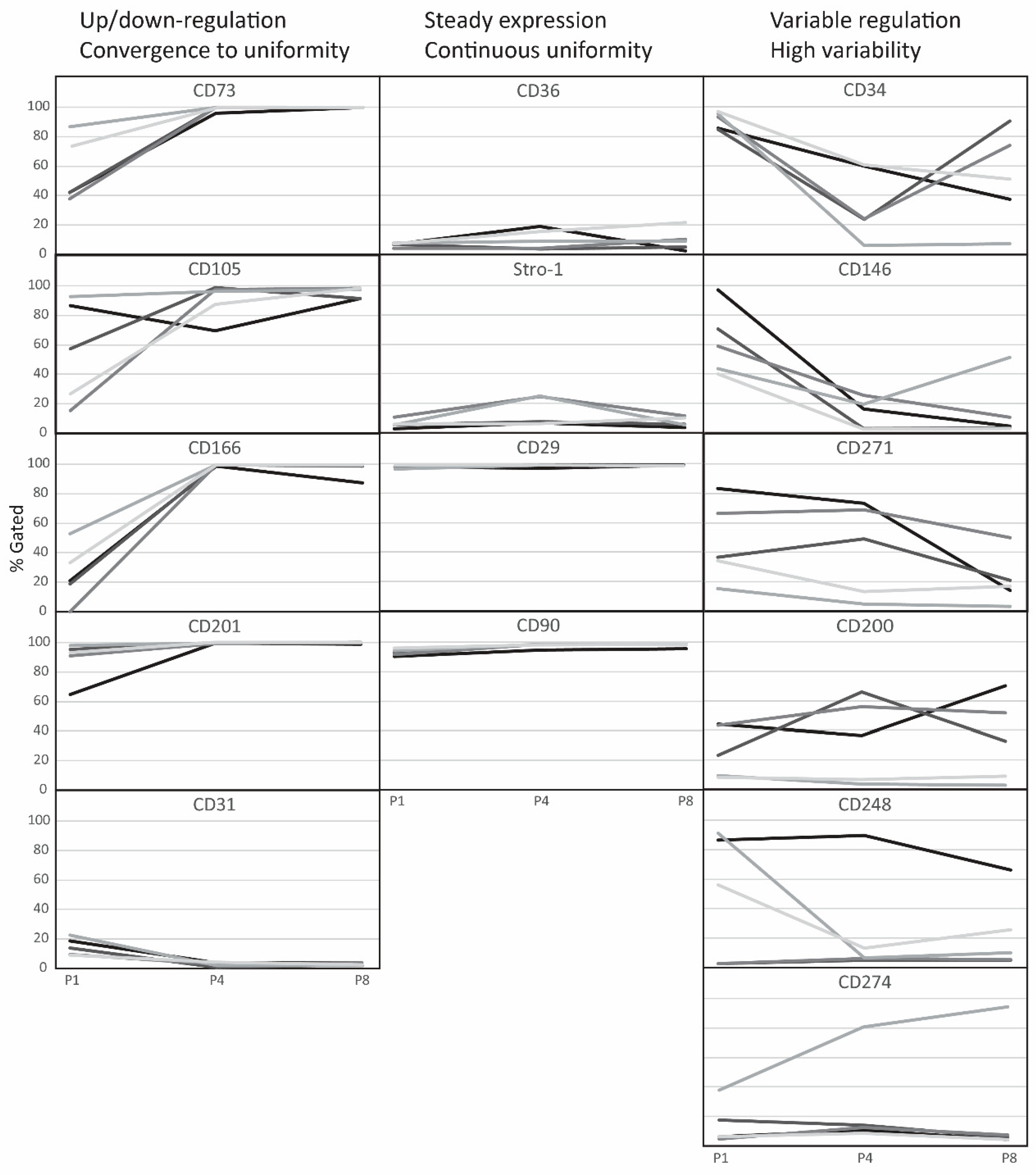
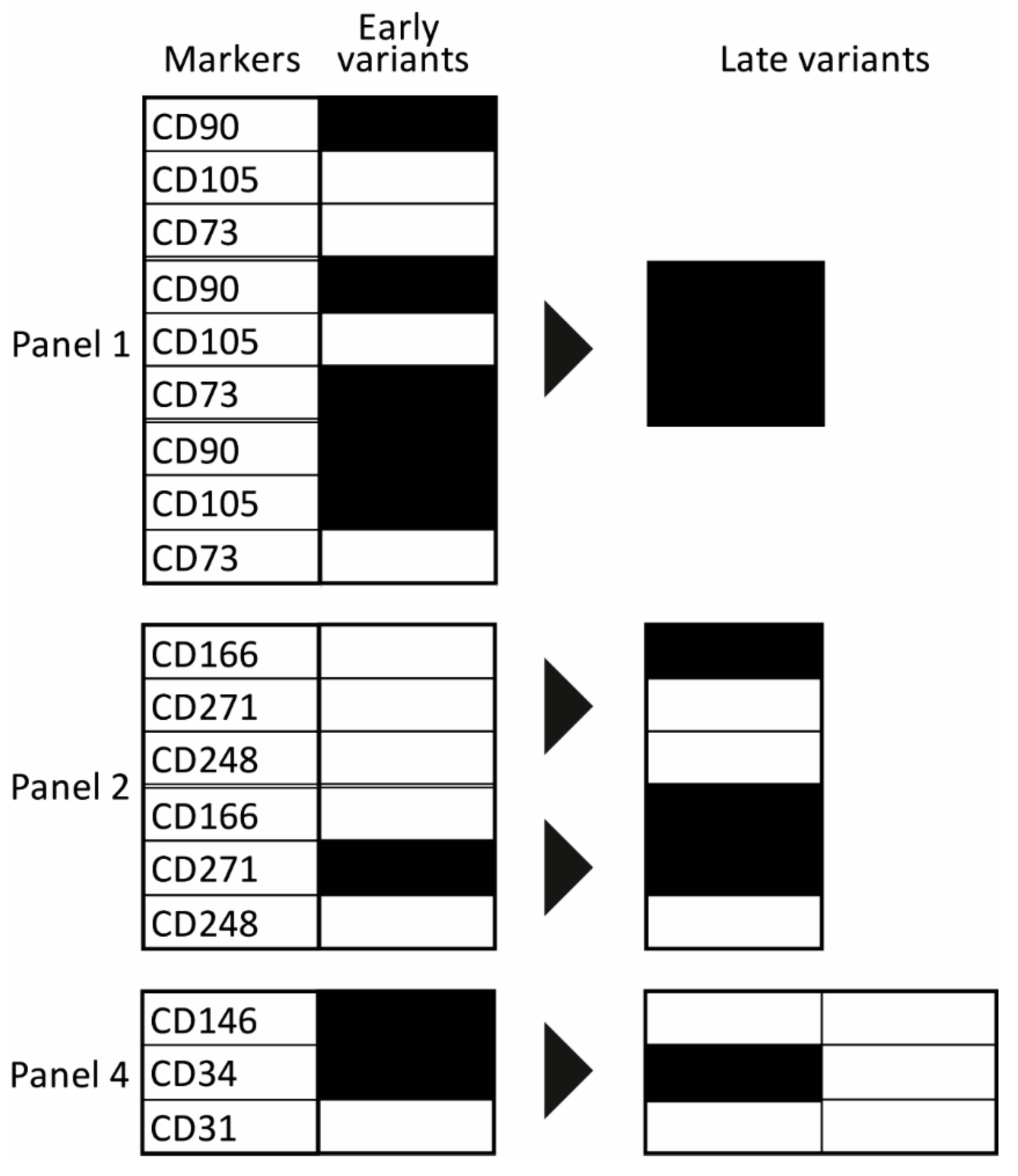
2.1. Temporal Changes of ASC Surface Markers during In Vitro Expansion
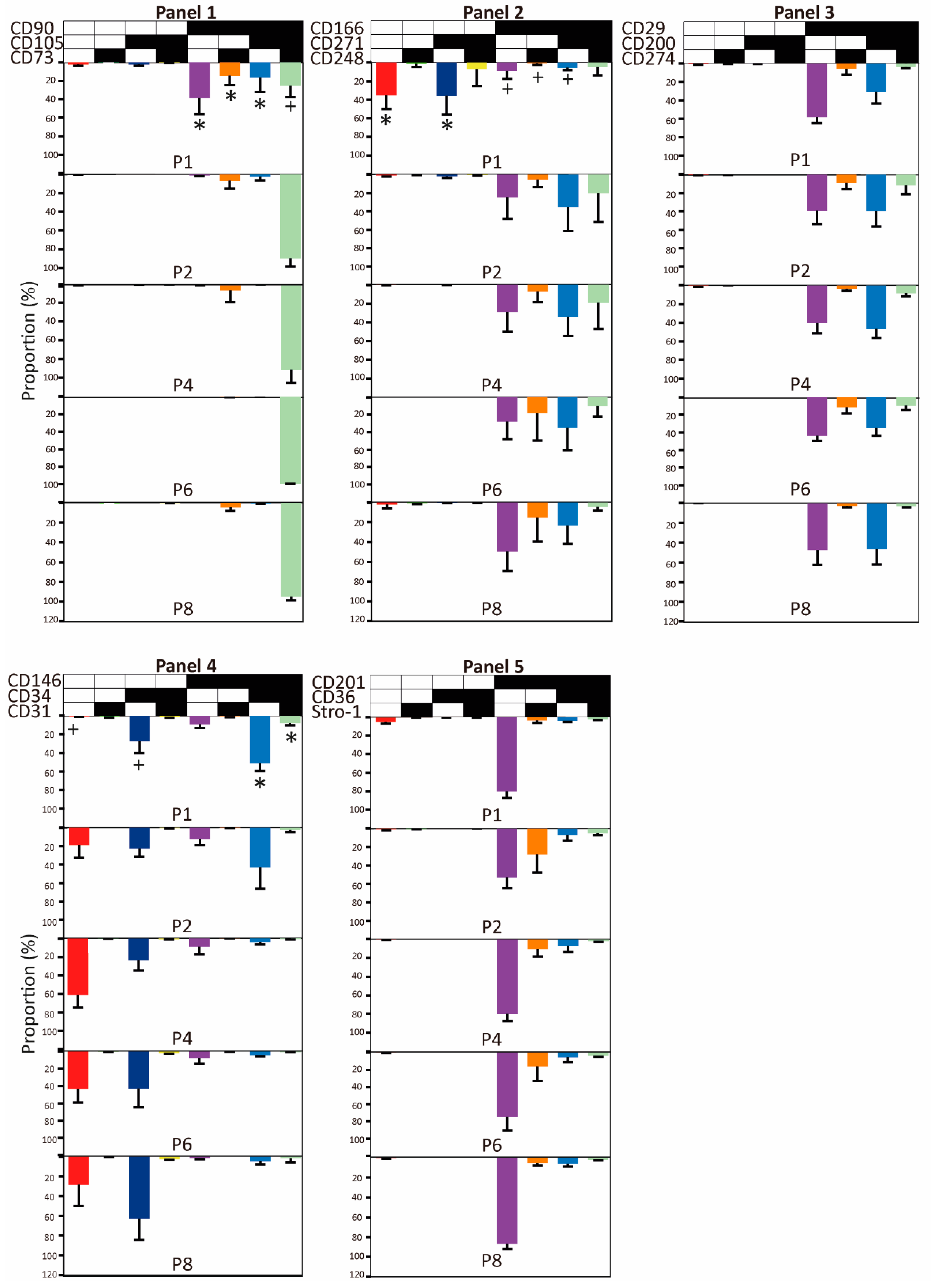
2.2. Evolution of Co-Expression Patterns
3. Discussion
4. Materials and Methods
4.1. ASC Isolation and Expansion
4.2. Multichromatic Flow Cytometry
4.3. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ASC | Adipose-derived stromal/stem cells |
| BP | Band pass |
| CD | Cluster of differentiation |
| FCS | Fetal calf serum |
| FMO | Fluorescence minus one |
| FSC-A | Forward scatter area |
| FSC-H | Forward scatter hight |
| FVS570 | Fixable viability stain 570 |
| GLM | General linear model |
| MSC | Mesenchymal stem cells |
| P | Passage |
| SD | Standard deviation |
| SSC-A | Side scatter area |
| SVF | Stromal vascular fraction |
Appendix A
| Fluorophore | Antigen | Host | Company | Catalog Number |
| BV421 | CD201 | rat | BD Biosciences | 743552 |
| BV510 | CD105 | mouse | BD Biosciences | 563264 |
| BV605 | CD166 | mouse | BD Biosciences | 742373 |
| APC-R700 | CD274 | mouse | BD Biosciences | 565188 |
| APC-Cy7 | CD31 | mouse | BD Biosciences | 563653 |
| FITC | CD73 | mouse | BD Biosciences | 561254 |
| BV605 | CD36 | mouse | BD Biosciences | 563518 |
| BV650 | CD29 | mouse | BD Biosciences | 743785 |
| PE-Cy 7 | CD200 | mouse | BD Biosciences | 562125 |
| PE-Cy7 | CD271 | mouse | BD Biosciences | 562122 |
| Alexa Fluor 647 | CD248 | mouse | BD Biosciences | 564994 |
| Alexa Fluor 647 | Stro-1 | mouse | R & D system | FAB1038R |
| Percp-Cy5.5 | CD90 | mouse | BD Biosciences | 561557 |
| PE-CF594 | CD146 | mouse | BD Biosciences | 564327 |
| PE-Cy7 | CD34 | mouse | BD Biosciences | 560710 |
| Product Name | Company | Catalog Number | ||
| BD Horizon™ Brilliant Stain Buffer | BD Biosciences | 563794 | ||
| Viability dye, FVS570 | BD Biosciences | 564995 | ||
| CompBeads Plus Set Anti-mouse Ig, κ | BD Biosciences | 560497 | ||
| CompBeads Plus Set Anti-rat Ig, κ | BD Biosciences | 560499 | ||
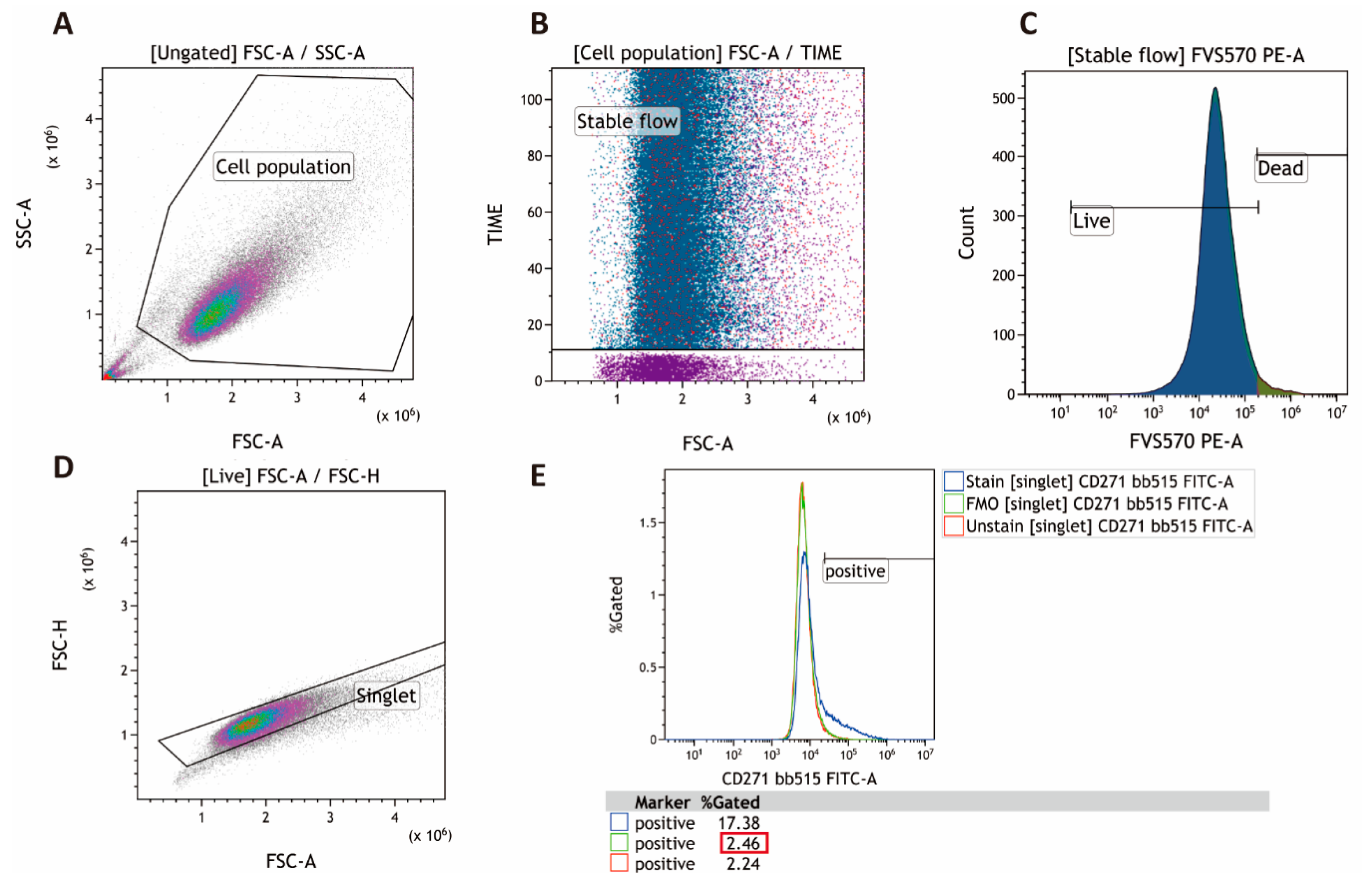
References
- Bajek, A.; Gurtowska, N.; Olkowska, J.; Kazmierski, L.; Maj, M.; Drewa, T. Adipose-Derived Stem Cells as a Tool in Cell-Based Therapies. Arch. Immunol. Ther. Exp. (Warsz.) 2016, 64, 443–454. [Google Scholar] [CrossRef] [PubMed]
- Strioga, M.; Viswanathan, S.; Darinskas, A.; Slaby, O.; Michalek, J. Same or not the same? Comparison of adipose tissue-derived versus bone marrow-derived mesenchymal stem and stromal cells. Stem Cells Dev. 2012, 21, 2724–2752. [Google Scholar] [CrossRef] [PubMed]
- Berry, D.C.; Jiang, Y.; Graff, J.M. Emerging Roles of Adipose Progenitor Cells in Tissue Development, Homeostasis, Expansion and Thermogenesis. Trends Endocrinol. Metab. 2016, 27, 574–585. [Google Scholar] [CrossRef] [PubMed]
- Badimon, L.; Cubedo, J. Adipose tissue depots and inflammation: Effects on plasticity and residentmesenchymal stem cell function. Cardiovasc. Res. 2017, 113, 1064–1073. [Google Scholar] [CrossRef]
- Cao, F.; Liu, T.; Xu, Y.; Xu, D.; Feng, S. Culture and properties of adipose-derived mesenchymal stem cells: Characteristics in vitro and immunosuppression in vivo. Int. J. Clin. Exp. Pathol. 2015, 8, 7694–7709. [Google Scholar]
- Larsen, L.; Tchanque-Fossuo, C.N.; Gorouhi, F.; Boudreault, D.; Nguyen, C.; Fuentes, J.J.; Crawford, R.W.; Dahle, S.E.; Whetzel, T.; Rivkah Isseroff, R. Combination therapy of autologous adipose mesenchymal stem cell-enriched, high-density lipoaspirate and topical timolol for healing chronic wounds. J. Tissue Eng. Regen. Med. 2018, 12, 186–190. [Google Scholar] [CrossRef]
- Moon, K.C.; Suh, H.S.; Kim, K.B.; Han, S.K.; Young, K.W.; Lee, J.W.; Kim, M.H. Potential of allogeneic adipose-derived stem cell–hydrogel complex for treating diabetic foot ulcers. Diabetes 2019, 68, 837–846. [Google Scholar] [CrossRef]
- Tang, Y.; Pan, Z.Y.; Zou, Y.; He, Y.; Yang, P.Y.; Tang, Q.Q.; Yin, F. A comparative assessment of adipose-derived stem cells from subcutaneous and visceral fat as a potential cell source for knee osteoarthritis treatment. J. Cell. Mol. Med. 2017, 21, 2153–2162. [Google Scholar] [CrossRef]
- Badimon, L.; Oñate, B.; Vilahur, G. Adipose-derived Mesenchymal Stem Cells and Their Reparative Potential in Ischemic Heart Disease. Rev. Española Cardiol. Engl. Ed. 2015, 68, 599–611. [Google Scholar] [CrossRef]
- Lin, H.P.; Chan, T.M.; Fu, R.H.; Chuu, C.P.; Chiu, S.C.; Tseng, Y.H.; Liu, S.P.; Lai, K.C.; Shih, M.C.; Lin, Z.S.; et al. Applicability of adipose-derived stem cells in type 1 diabetes mellitus. Cell Transplant. 2015, 24, 521–532. [Google Scholar] [CrossRef]
- Dominici, M.; Paolucci, P.; Conte, P.; Horwitz, E.M. Heterogeneity of Multipotent Mesenchymal Stromal Cells: From Stromal Cells to Stem Cells and Vice Versa. Transplantation 2009, 87, S36–S42. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, F.M.; Riis, S.E.; Andersen, J.I.; Lesage, R.; Fink, T.; Pennisi, C.P.; Zachar, V. Discrete adipose-derived stem cell subpopulations may display differential functionality after in vitro expansion despite convergence to a common phenotype distribution. Stem Cell Res. Ther. 2016, 7, 177. [Google Scholar] [CrossRef] [PubMed]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.; Horwitz, E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef] [PubMed]
- Maslova, O.O. Current view of mesenchymal stem cells biology (Brief review). Biopolym. Cell 2012, 28, 190–198. [Google Scholar] [CrossRef]
- Levi, B.; Wan, D.C.; Glotzbach, J.P.; Hyun, J.; Januszyk, M.; Montoro, D.; Sorkin, M.; James, A.W.; Nelson, E.R.; Li, S.; et al. CD105 protein depletion enhances human adipose-derived stromal cell osteogenesis through reduction of transforming growth factor β1 (TGF-β1) signaling. J. Biol. Chem. 2011, 286, 39497–39509. [Google Scholar] [CrossRef]
- Chung, M.T.; Liu, C.; Hyun, J.S.; Lo, D.D.; Montoro, D.T.; Hasegawa, M.; Li, S.; Sorkin, M.; Rennert, R.; Keeney, M.; et al. CD90 (Thy-1)-Positive Selection Enhances Osteogenic Capacity of Human Adipose-Derived Stromal Cells. Tissue Eng. Part A 2013, 19, 989–997. [Google Scholar] [CrossRef]
- Yang, S.; Pilgaard, L.; Chase, L.G.; Boucher, S.; Vemuri, M.C.; Fink, T.; Zachar, V. Defined xenogeneic-free and hypoxic environment provides superior conditions for long-term expansion of human adipose-derived stem cells. Tissue Eng. Part C Methods 2012, 18, 593–602. [Google Scholar] [CrossRef]
- Baer, P.C.; Kuçi, S.; Krause, M.; Kuçi, Z.; Zielen, S.; Geiger, H.; Bader, P.; Schubert, R. Comprehensive Phenotypic Characterization of Human Adipose-Derived Stromal/Stem Cells and Their Subsets by a High Throughput Technology. Stem Cells Dev. 2012, 22, 330–339. [Google Scholar] [CrossRef]
- Mildmay-White, A.; Khan, W. Cell Surface Markers on Adipose-Derived Stem Cells: A Systematic Review. Curr. Stem Cell Res. Ther. 2017, 12, 484–492. [Google Scholar] [CrossRef]
- Gronthos, S.; Franklin, D.M.; Leddy, H.A.; Robey, P.G.; Storms, R.W.; Gimble, J.M. Surface protein characterization of human adipose tissue-derived stromal cells. J. Cell. Physiol. 2001, 189, 54–63. [Google Scholar] [CrossRef]
- Astori, G.; Vignati, F.; Bardelli, S.; Tubio, M.; Gola, M.; Albertini, V.; Bambi, F.; Scali, G.; Castelli, D.; Rasini, V.; et al. “In vitro” and multicolor phenotypic characterization of cell subpopulations identified in fresh human adipose tissue stromal vascular fraction and in the derived mesenchymal stem cells. J. Transl. Med. 2007, 5, 55. [Google Scholar] [CrossRef] [PubMed]
- Riis, S.; Zachar, V.; Boucher, S.; Vemuri, M.C.; Pennisi, C.P.; Fink, T. Critical steps in the isolation and expansion of adipose-derived stem cells for translational therapy. Expert Rev. Mol. Med. 2015, 17, e11. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Volat, F.; Sandhow, L.; Galitzky, J.; Nguyen, T.; Esteve, D.; Åström, G.; Mejhert, N.; Ledoux, S.; Thalamas, C.; et al. CD36 Is a Marker of Human Adipocyte Progenitors with Pronounced Adipogenic and Triglyceride Accumulation Potential. Stem Cells 2017, 35, 1799–1814. [Google Scholar] [CrossRef] [PubMed]
- Brett, E.; Zielins, E.R.; Chin, M.; Januszyk, M.; Blackshear, C.P.; Findlay, M.; Momeni, A.; Gurtner, G.C.; Longaker, M.T.; Wan, D.C. Isolation of CD248-expressing stromal vascular fraction for targeted improvement of wound healing. Wound Repair Regen. 2017, 25, 414–422. [Google Scholar] [CrossRef] [PubMed]
- Beckenkamp, L.R.; Souza, L.E.B.; Melo, F.U.F.; Thomé, C.H.; Magalhães, D.A.R.; Palma, P.V.B.; Covas, D.T. Comparative characterization of CD271+ and CD271− subpopulations of CD34+ human adipose-derived stromal cells. J. Cell. Biochem. 2018, 119, 3873–3884. [Google Scholar] [CrossRef]
- Mihaila, S.M.; Frias, A.M.; Pirraco, R.P.; Rada, T.; Reis, R.L.; Gomes, M.E.; Marques, A.P. Human adipose tissue-derived SSEA-4 subpopulation multi-differentiation potential towards the endothelial and osteogenic lineages. Tissue Eng. Part A 2013, 19, 235–246. [Google Scholar] [CrossRef]
- Rada, T.; Santos, T.C.; Marques, A.P.; Correlo, V.M.; Frias, A.M.; Castro, A.G.; Neves, N.M.; Gomes, M.E.; Reis, R.L. Osteogenic differentiation of two distinct subpopulations of human adipose-derived stem cells: An in vitro and in vivo study. J. Tissue Eng. Regen. Med. 2012, 6, 1–11. [Google Scholar] [CrossRef]
- Li, H.; Zimmerlin, L.; Marra, K.G.; Donnenberg, V.S.; Donnenberg, A.D.; Rubin, J.P. Adipogenic potential of adipose stem cell subpopulations. Plast. Reconstr. Surg. 2011, 128, 663–672. [Google Scholar] [CrossRef]
- Najar, M.; Raicevic, G.; Jebbawi, F.; De Bruyn, C.; Meuleman, N.; Bron, D.; Toungouz, M.; Lagneaux, L. Characterization and functionality of the CD200-CD200R system during mesenchymal stromal cell interactions with T-lymphocytes. Immunol. Lett. 2012, 146, 50–56. [Google Scholar] [CrossRef]
- Psaltis, P.J.; Paton, S.; See, F.; Arthur, A.; Martin, S.; Itescu, S.; Worthley, S.G.; Gronthos, S.; Zannettino, A.C.W. Enrichment for STRO-1 expression enhances the cardiovascular paracrine activity of human bone marrow-derived mesenchymal cell populations. J. Cell. Physiol. 2010, 223, 530–540. [Google Scholar] [CrossRef]
- Bruder, S.P.; Ricalton, N.S.; Boynton, R.E.; Connolly, T.J.; Jaiswal, N.; Zaia, J.; Barry, F.P. Mesenchymal stem cell surface antigen SB-10 corresponds to activated leukocyte cell adhesion molecule and is involved in osteogenic differentiation. J. Bone Miner. Res. 1998, 13, 655–663. [Google Scholar] [CrossRef] [PubMed]
- Sanders, A.J.; Jiang, D.G.; Jiang, W.G.; Harding, K.G.; Patel, G.K. Activated leukocyte cell adhesion molecule impacts on clinical wound healing and inhibits HaCaT migration. Int. Wound J. 2011, 8, 500–507. [Google Scholar] [CrossRef]
- Luz-Crawford, P.; Noël, D.; Fernandez, X.; Khoury, M.; Figueroa, F.; Carrión, F.; Jorgensen, C.; Djouad, F. Mesenchymal Stem Cells Repress Th17 Molecular Program through the PD-1 Pathway. PLoS ONE 2012, 7. [Google Scholar] [CrossRef]
- Camilleri, E.T.; Gustafson, M.P.; Dudakovic, A.; Riester, S.M.; Garces, C.G.; Paradise, C.R.; Takai, H.; Karperien, M.; Cool, S.; Sampen, H.J.I.; et al. Identification and validation of multiple cell surface markers of clinical-grade adipose-derived mesenchymal stromal cells as novel release criteria for good manufacturing practice-compliant production. Stem Cell Res. Ther. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Miyata, Y.; Otsuki, M.; Kita, S.; Shimomura, I. Identification of Mouse Mesenteric and Subcutaneous in vitro Adipogenic Cells. Sci. Rep. 2016, 6, 21041. [Google Scholar] [CrossRef] [PubMed]
- Ong, W.K.; Tan, C.S.; Chan, K.L.; Goesantoso, G.G.; Chan, X.H.D.; Chan, E.; Yin, J.; Yeo, C.R.; Khoo, C.M.; So, J.B.Y.; et al. Identification of Specific Cell-Surface Markers of Adipose-Derived Stem Cells from Subcutaneous and Visceral Fat Depots. Stem Cell Rep. 2014, 2, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Durandt, C.; van Vollenstee, F.A.; Dessels, C.; Kallmeyer, K.; de Villiers, D.; Murdoch, C.; Potgieter, M.; Pepper, M.S. Novel flow cytometric approach for the detection of adipocyte subpopulations during adipogenesis. J. Lipid Res. 2016, 57, 729–742. [Google Scholar] [CrossRef]
- Bourin, P.; Bunnell, B.A.; Casteilla, L.; Dominici, M.; Katz, A.J.; March, K.L.; Redl, H.; Rubin, J.P.; Yoshimura, K.; Gimble, J.M. Stromal cells from the adipose tissue-derived stromal vascular fraction and culture expanded adipose tissue-derived stromal/stem cells: A joint statement of the International Federation for Adipose Therapeutics and Science (IFATS) and the International So. Cytotherapy 2013, 15, 641–648. [Google Scholar] [CrossRef]
- Baer, P.C. Adipose-derived mesenchymal stromal/stem cells: An update on their phenotype in vivo and in vitro. World J. Stem Cells 2014, 6, 256–265. [Google Scholar] [CrossRef]
- Lee, N.E.; Kim, S.J.; Yang, S.J.; Joo, S.Y.; Park, H.; Lee, K.W.; Yang, H.M.; Park, J.B. Comparative characterization of mesenchymal stromal cells from multiple abdominal adipose tissues and enrichment of angiogenic ability via CD146 molecule. Cytotherapy 2017, 19, 170–180. [Google Scholar] [CrossRef]
- Davies, O.G.; Cooper, P.R.; Shelton, R.M.; Smith, A.J.; Scheven, B.A. Isolation of adipose and bone marrow mesenchymal stem cells using CD29 and CD90 modifies their capacity for osteogenic and adipogenic differentiation. J. Tissue Eng. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Rada, T.; Reis, R.L.; Gomes, M.E. Distinct stem cells subpopulations isolated from human adipose tissue exhibit different chondrogenic and osteogenic differentiation potential. Stem Cell Rev. Rep. 2011, 7, 64–76. [Google Scholar] [CrossRef] [PubMed]
- Ip, J.E.; Wu, Y.; Huang, J.; Zhang, L.; Pratt, R.E.; Dzau, V.J. Mesenchymal stem cells use integrin β1 not CXC chemokine receptor 4 for myocardial migration and engraftment. Mol. Biol. Cell 2007, 18, 2873–2882. [Google Scholar] [CrossRef]
- Calabrese, G.; Giuffrida, R.; Lo Furno, D.; Parrinello, N.L.; Forte, S.; Gulino, R.; Colarossi, C.; Schinocca, L.R.; Giuffrida, R.; Cardile, V.; et al. Potential effect of CD271 on human mesenchymal stromal cell proliferation and differentiation. Int. J. Mol. Sci. 2015, 16, 15609–15624. [Google Scholar] [CrossRef] [PubMed]
- Latifi-Pupovci, H.; Kuçi, Z.; Wehner, S.; Bönig, H.; Lieberz, R.; Klingebiel, T.; Bader, P.; Kuçi, S. In vitro migration and proliferation (“wound healing”) potential of mesenchymal stromal cells generated from human CD271(+) bone marrow mononuclear cells. J. Transl. Med. 2015, 13, 315. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, J.B.; McIntosh, K.; Zvonic, S.; Garrett, S.; Floyd, Z.E.; Kloster, A.; Di Halvorsen, Y.; Storms, R.W.; Goh, B.; Kilroy, G.; et al. Immunophenotype of Human Adipose-Derived Cells: Temporal Changes in Stromal-Associated and Stem Cell-Associated Markers. Stem Cells 2006, 24, 376–385. [Google Scholar] [CrossRef] [PubMed]
- Prieto González, E.A. Heterogeneity in Adipose Stem Cells. In Advances in Experimental Medicine and Biology; Springer: New York, NY, USA, 2019; Volume 1123, pp. 119–150. [Google Scholar]
- Yang, H.J.; Kim, K.J.; Kim, M.K.; Lee, S.J.; Ryu, Y.H.; Seo, B.F.; Oh, D.Y.; Ahn, S.T.; Lee, H.Y.; Rhie, J.W. The stem cell potential and multipotency of human adipose tissue-derived stem cells vary by cell donor and are different from those of other types of stem cells. Cells Tissues Organs 2014, 199, 373–383. [Google Scholar] [CrossRef]
- Reumann, M.K.; Linnemann, C.; Aspera-Werz, R.H.; Arnold, S.; Held, M.; Seeliger, C.; Nussler, A.K.; Ehnert, S. Donor site location is critical for proliferation, stem cell capacity, and osteogenic differentiation of adipose mesenchymal stem/stromal cells: Implications for bone tissue engineering. Int. J. Mol. Sci. 2018, 19, 1868. [Google Scholar] [CrossRef]
- Chaker, D.; Mouawad, C.; Azar, A.; Quilliot, D.; Achkar, I.; Fajloun, Z.; Makdissy, N. Inhibition of the RhoGTPase Cdc42 by ML141 enhances hepatocyte differentiation from human adipose-derived mesenchymal stem cells via the Wnt5a/PI3K/miR-122 pathway: Impact of the age of the donor. Stem Cell Res. Ther. 2018, 9. [Google Scholar] [CrossRef]
- Liu, M.; Lei, H.; Dong, P.; Fu, X.; Yang, Z.; Yang, Y.; Ma, J.; Liu, X.; Cao, Y.; Xiao, R. Adipose-Derived Mesenchymal Stem Cells from the Elderly Exhibit Decreased Migration and Differentiation Abilities with Senescent Properties. Cell Transplant. 2017, 26, 1505–1519. [Google Scholar] [CrossRef]
- Guneta, V.; Tan, N.S.; Sugii, S.; Lim, T.C.; Wong, T.C.M.; Choong, C. Comparative study of adipose-derived stem cells from abdomen and breast. Ann. Plast. Surg. 2016, 76, 569–575. [Google Scholar] [CrossRef] [PubMed]
- Donnenberg, A.D.; Meyer, E.M.; Rubin, J.P.; Donnenberg, V.S. The cell-surface proteome of cultured adipose stromal cells. Cytom. Part A 2015, 87. [Google Scholar] [CrossRef] [PubMed]
- Walmsley, G.G.; Atashroo, D.A.; Maan, Z.N.; Hu, M.S.; Zielins, E.R.; Tsai, J.M.; Duscher, D.; Paik, K.; Tevlin, R.; Marecic, O.; et al. High-Throughput Screening of Surface Marker Expression on Undifferentiated and Differentiated Human Adipose-Derived Stromal Cells. Tissue Eng. Part A 2015. [Google Scholar] [CrossRef] [PubMed]
- Zuk, P. Regenerative Medicine and Tissue Engineering; Andrades, J.A., Ed.; InTech: London, UK, 2013; ISBN 978-953-51-1108-5. [Google Scholar]
- Mafi, P. Adult Mesenchymal Stem Cells and Cell Surface Characterization-A Systematic Review of the Literature. Open Orthop. J. 2011, 5, 253–260. [Google Scholar] [CrossRef]
- Yoshimura, K.; Shigeura, T.; Matsumoto, D.; Sato, T.; Takaki, Y.; Aiba-Kojima, E.; Sato, K.; Inoue, K.; Nagase, T.; Koshima, I.; et al. Characterization of freshly isolated and cultured cells derived from the fatty and fluid portions of liposuction aspirates. J. Cell. Physiol. 2006, 208, 64–76. [Google Scholar] [CrossRef]
- Gimble, J.M.; Katz, A.J.; Bunnell, B.A. Adipose-derived stem cells for regenerative medicine. Circ. Res. 2007, 100, 1249–1260. [Google Scholar] [CrossRef]
- Ning, H.; Lin, G.; Lue, T.F.; Lin, C.-S. Mesenchymal Stem Cell Marker Stro-1 is a 75kd Endothelial Antigen. Biochem. Biophys. Res. Commun. 2011, 413. [Google Scholar] [CrossRef]
- Zuk, P.A.; Zhu, M.; Ashjian, P.; De Ugarte, D.A.; Huang, J.I.; Mizuno, H.; Alfonso, Z.C.; Fraser, J.K.; Benhaim, P.; Hedrick, M.H. Human adipose tissue is a source of multipotent stem cells. Mol. Biol. Cell 2002, 13, 4279–4295. [Google Scholar] [CrossRef]
- Riis, S.; Nielsen, F.M.; Pennisi, C.P.; Zachar, V.; Fink, T. Comparative Analysis of Media and Supplements on Initiation and Expansion of Adipose-Derived Stem Cells. Stem Cells Transl. Med. 2016, 5, 314–324. [Google Scholar] [CrossRef]
- Nepali, S.; Park, M.; Lew, H.; Kim, O. Comparative Analysis of Human Adipose-Derived Mesenchymal Stem Cells from Orbital and Abdominal Fat. Stem Cells Int. 2018, 2018. [Google Scholar] [CrossRef]
- Dizaji Asl, K.; Shafaei, H.; Soleimani Rad, J.; Ollah Nozad, H. Comparison of Characteristics of Human Amniotic Membrane and Human Adipose Tissue Derived Mesenchymal Stem Cells. World J. Plast. Surg. 2017, 6, 33–39. [Google Scholar] [PubMed]
- Zimmerlin, L.; Donnenberg, V.S.; Rubin, J.P.; Donnenberg, A.D. Mesenchymal markers on human adipose stem/progenitor cells. Cytom. Part A 2013, 83A, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Varma, M.J.O.; Breuls, R.G.M.; Schouten, T.E.; Jurgens, W.J.F.M.; Bontkes, H.J.; Schuurhuis, G.J.; Ham, S.M.V.; Milligen, F.J.V. Phenotypical and Functional Characterization of Freshly Isolated Adipose Tissue-Derived Stem Cells. Stem Cells Dev. 2007, 16, 91–104. [Google Scholar] [CrossRef] [PubMed]
- Schachtele, S.; Clouser, C.; Aho, J. Markers and Methods to Verify Mesenchymal Stem Cell Identity, Potency, and Quality. Available online: https://www.rndsystems.com/cn/resources/articles/markers-and-methods-verify-mesenchymal-stem-cell-identity-potency-and-quality (accessed on 29 January 2020).
- Lv, F.-J.; Tuan, R.S.; Cheung, K.M.C.; Leung, V.Y.L. Concise Review: The Surface Markers and Identity of Human Mesenchymal Stem Cells. Stem Cells 2014, 32, 1408–1419. [Google Scholar] [CrossRef]
- Hörl, S.; Ejaz, A.; Ernst, S.; Mattesich, M.; Kaiser, A.; Jenewein, B.; Zwierzina, M.E.; Hammerle, S.; Miggitsch, C.; Mitterberger-Vogt, M.C.; et al. CD146 (MCAM) in human cs-DLK1−/cs-CD34+ adipose stromal/progenitor cells. Stem Cell Res. 2017, 22, 1–12. [Google Scholar] [CrossRef]
- Crisan, M.; Yap, S.; Casteilla, L.; Chen, C.-W.; Corselli, M.; Park, T.S.; Andriolo, G.; Sun, B.; Zheng, B.; Zhang, L.; et al. A perivascular origin for mesenchymal stem cells in multiple human organs. Cell Stem Cell 2008, 3, 301–313. [Google Scholar] [CrossRef]
- Li, X.; Guo, W.; Zha, K.; Jing, X.; Wang, M.; Zhang, Y.; Hao, C.; Gao, S.; Chen, M.; Yuan, Z.; et al. Enrichment of CD146+ adipose-derived stem cells in combination with articular cartilage extracellular matrix scaffold promotes cartilage regeneration. Theranostics 2019, 9, 5105–5121. [Google Scholar] [CrossRef]
- Su, X.; Zuo, W.; Wu, Z.; Chen, J.; Wu, N.; Ma, P.; Xia, Z.; Jiang, C.; Ye, Z.; Liu, S.; et al. CD146 as a new marker for an increased chondroprogenitor cell sub-population in the later stages of osteoarthritis. J. Orthop. Res. 2015, 33, 84–91. [Google Scholar] [CrossRef]
- Patel, R.S.; Carter, G.; El Bassit, G.; Patel, A.A.; Cooper, D.R.; Murr, M.; Patel, N.A. Adipose-derived stem cells from lean and obese humans show depot specific differences in their stem cell markers, exosome contents and senescence: Role of protein kinase C delta (PKCδ) in adipose stem cell niche. Stem Cell Investig. 2016, 2016. [Google Scholar] [CrossRef]
- Nugraha Setyawan, E.M.; Oh, H.J.; Kim, M.J.; Kim, G.A.; Lee, S.H.; Choi, Y.B.; Ra, K.; Lee, B.C. Despite the donor’s age, human adipose-derived stem cells enhance the maturation and development rates of porcine oocytes in a co-culture system. Theriogenology 2018, 115, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Erickson, I.E.; Huang, A.H.; Garrity, S.T.; Mauck, R.L.; Steinberg, D.R. Donor Variation and Optimization of Human Mesenchymal Stem Cell Chondrogenesis in Hyaluronic Acid. Tissue Eng. Part A 2018, 24, 1693–1703. [Google Scholar] [CrossRef] [PubMed]
- De Francesco, F.; Tirino, V.; Desiderio, V.; Ferraro, G.; D’andrea, F.; Giuliano, M.; Libondi, G.; Pirozzi, G.; De Rosa, A.; Papaccio, G. Human CD34 +/CD90 + ASCs Are Capable of Growing as Sphere Clusters, Producing High Levels of VEGF and Forming Capillaries. PLoS ONE 2009, 4. [Google Scholar] [CrossRef] [PubMed]
- Ferraro, G.A.; De Francesco, F.; Nicoletti, G.; Paino, F.; Desiderio, V.; Tirino, V.; D’Andrea, F. Human adipose CD34+CD90+ stem cells and collagen scaffold constructs grafted in vivo fabricate loose connective and adipose tissues. J. Cell. Biochem. 2013, 114, 1039–1049. [Google Scholar] [CrossRef] [PubMed]
- D’Andrea, F.; De Francesco, F.; Ferraro, G.A.; Desiderio, V.; Tirino, V.; De Rosa, A.; Papaccio, G. Large-scale production of human adipose tissue from stem cells: A new tool for regenerative medicine and tissue banking. Tissue Eng. Part C Methods 2008, 14, 233–242. [Google Scholar] [CrossRef]
- Zachar, V.; Rasmussen, J.G.; Fink, T.; Zachar, V.; Pennisi, C.P.; Zachar, V.; Fink, T.; Gurevich, L.; Fojan, P. Isolation and growth of adipose tissue-derived stem cells. Methods Mol. Biol. 2011, 698, 243–251. [Google Scholar]

| Panels | Cytometer Setup | Fluorochrome | |||||
|---|---|---|---|---|---|---|---|
| 1. MSC Markers | 2. Wound Healing | 3. Immune Regulation | 4. ASC Marker | 5. Differentiation Capacity | Laser | Emission Channel | |
| CD201 | 405 nm | 450/45BP | BV421 | ||||
| CD105 | 525/40BP | BV510 | |||||
| CD166 | CD36 | 610/20BP | BV605 | ||||
| CD29 | 660/20BP | BV650 | |||||
| CD146 | 561 nm | 610/20BP | PE-CF594 | ||||
| FVS570 | FVS570 | FVS570 | FVS570 | FVS570 | 585/42BP | Viability dye | |
| CD271 | CD200 | CD34 | 780/60BP | PE-Cy7 | |||
| CD248 | Stro-1 | 638 nm | 660/20BP | AF647 | |||
| CD274 | 712/25BP | APC-R700 | |||||
| CD31 | 780/60BP | APC-Cy7 | |||||
| CD73 | 488 nm | 525/40BP | FITC | ||||
| CD90 | 690/50BP | PerCP-Cy5.5 | |||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peng, Q.; Alipour, H.; Porsborg, S.; Fink, T.; Zachar, V. Evolution of ASC Immunophenotypical Subsets During Expansion In Vitro. Int. J. Mol. Sci. 2020, 21, 1408. https://doi.org/10.3390/ijms21041408
Peng Q, Alipour H, Porsborg S, Fink T, Zachar V. Evolution of ASC Immunophenotypical Subsets During Expansion In Vitro. International Journal of Molecular Sciences. 2020; 21(4):1408. https://doi.org/10.3390/ijms21041408
Chicago/Turabian StylePeng, Qiuyue, Hiva Alipour, Simone Porsborg, Trine Fink, and Vladimir Zachar. 2020. "Evolution of ASC Immunophenotypical Subsets During Expansion In Vitro" International Journal of Molecular Sciences 21, no. 4: 1408. https://doi.org/10.3390/ijms21041408
APA StylePeng, Q., Alipour, H., Porsborg, S., Fink, T., & Zachar, V. (2020). Evolution of ASC Immunophenotypical Subsets During Expansion In Vitro. International Journal of Molecular Sciences, 21(4), 1408. https://doi.org/10.3390/ijms21041408







