Cannabinoid-mediated Modulation of Oxidative Stress and Early Inflammatory Response after Hypoxia–Ischemia
Abstract
1. Introduction
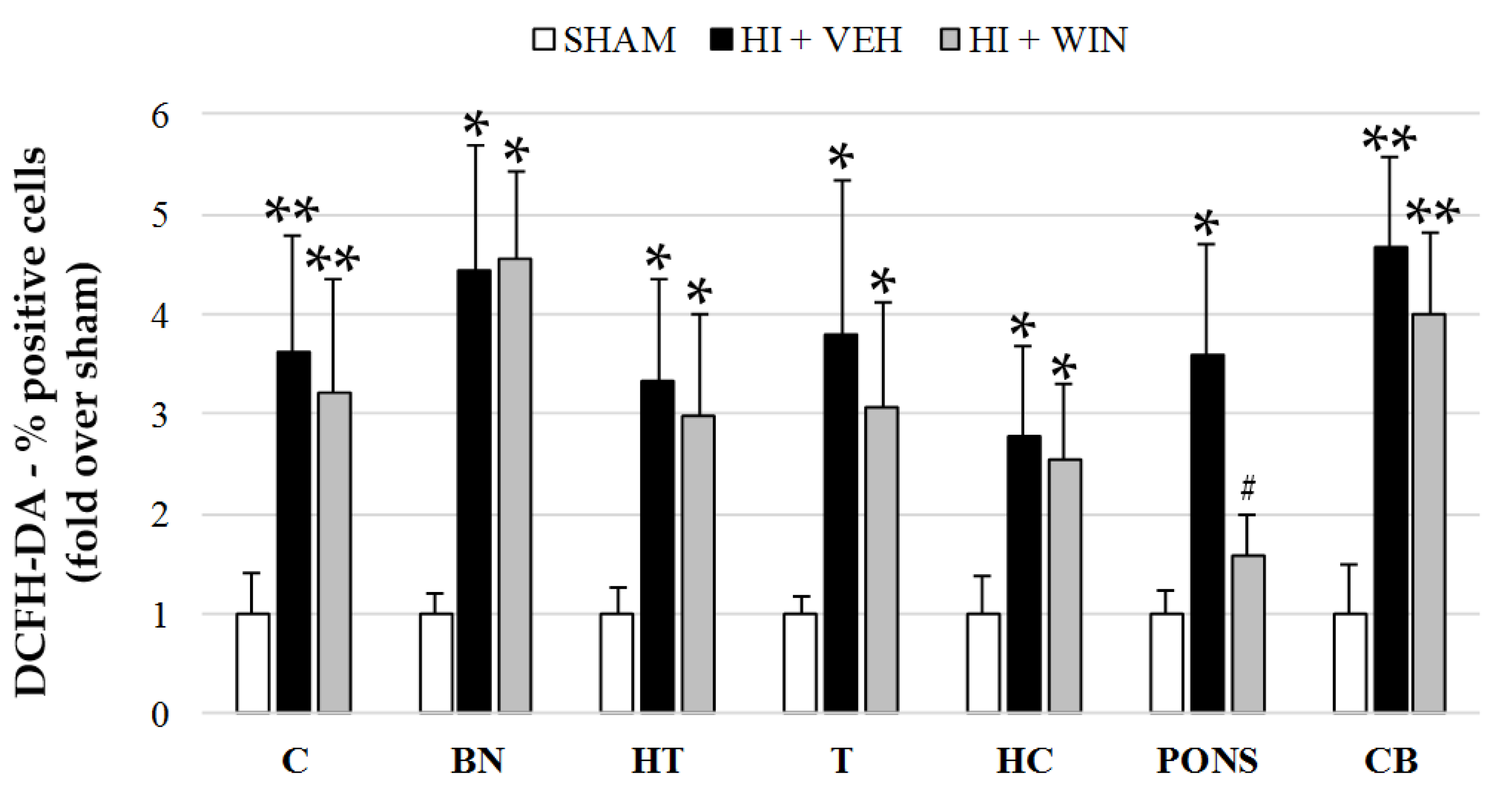
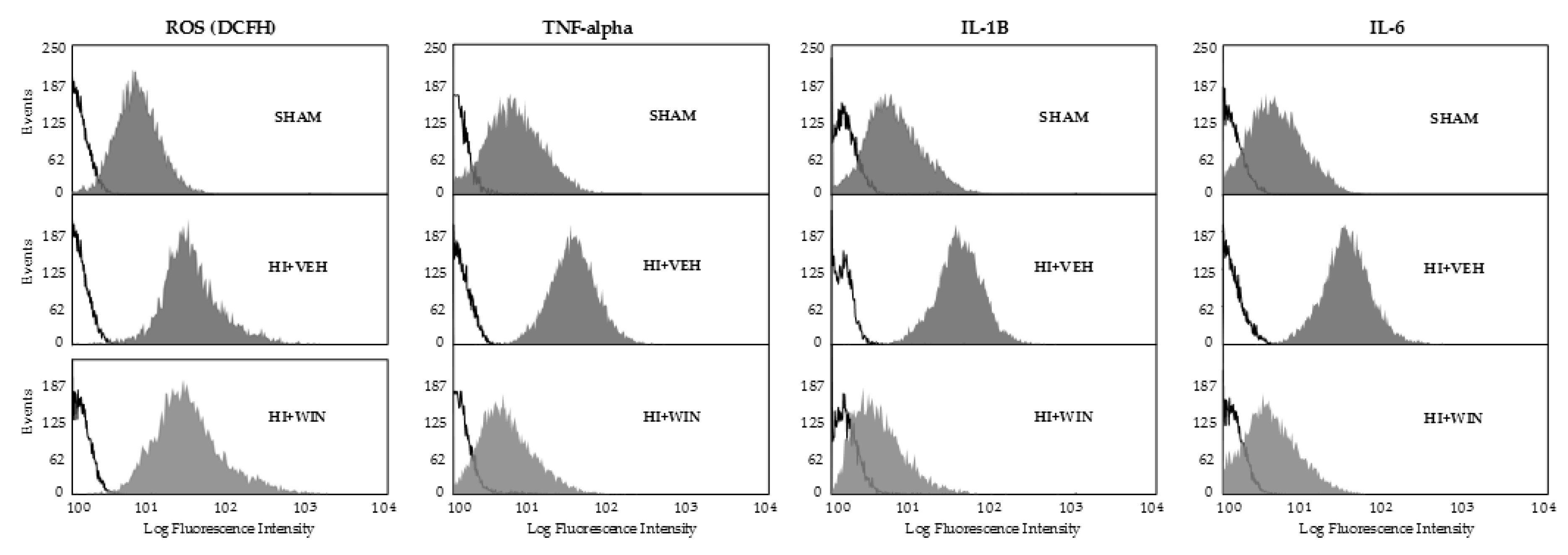
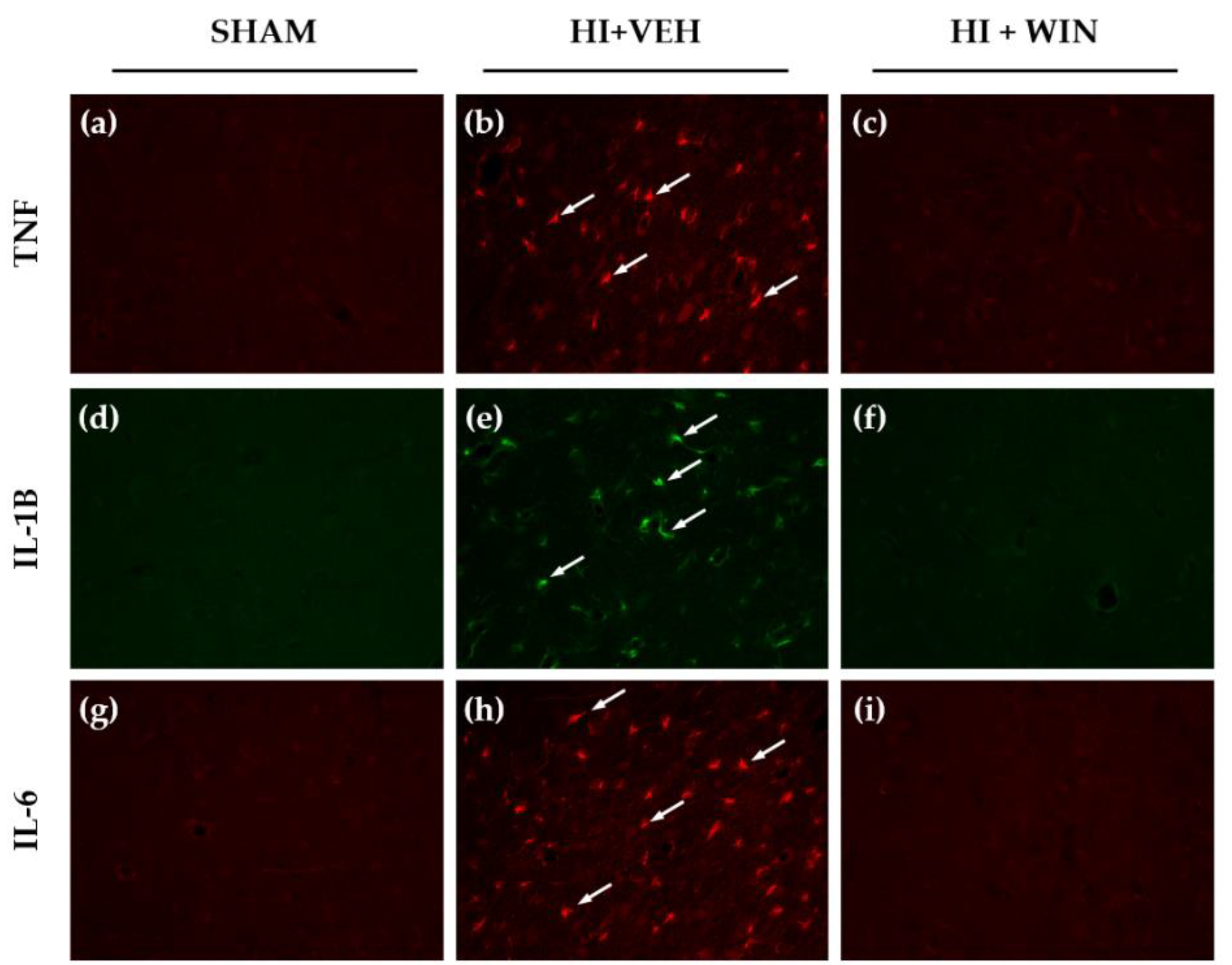
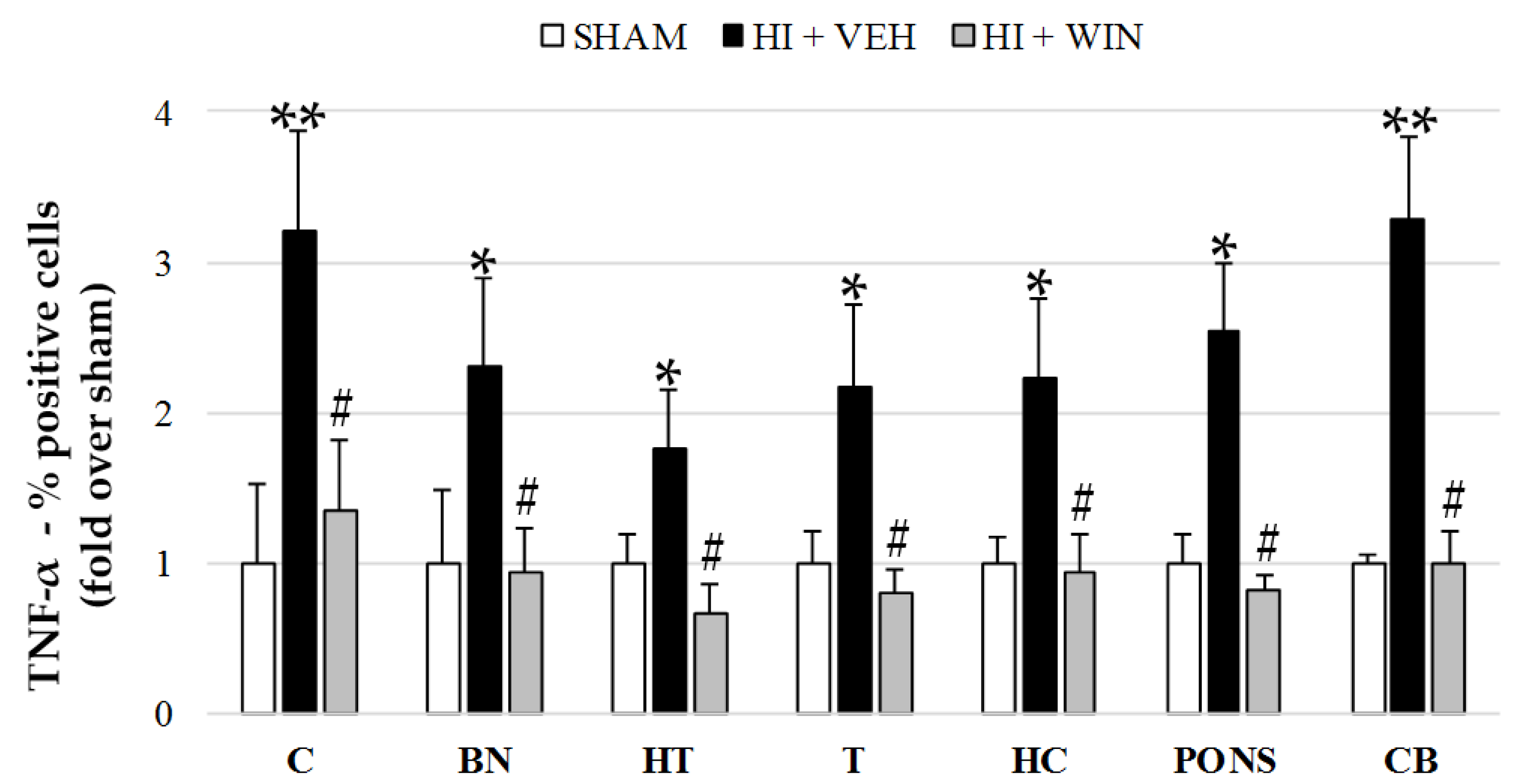
2. Results
3. Discussion
4. Materials and Methods
4.1. Surgery
4.2. Experimental Groups
Dose Selection
4.3. In vivo ROS Measurement
4.4. Immunohistochemistry
4.5. Cytokine Analyses
4.6. Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| WIN | WIN55,212-2 |
| HI | Hypoxia–ischemia |
| CB | Cannabinoid |
| CB1R | Cannabinoid type 1 receptor |
| CB2R | Cannabinoid type 1 receptor |
| CBRs | CB receptors |
| ROS | Reactive oxygen species |
| GSH/Cr | Glutathione/Creatine |
| TNF | Tumor necrosis factor |
| IL | Interleukin |
| LPS | Lipopolysaccharide |
| DMSO | Dimethyl sulfoxide |
References
- Liu, L.; Oza, S.; Hogan, D.; Chu, Y.; Perin, J.; Zhu, J.; Lawn, J.E.; Cousens, S.; Mathers, C.; Black, R.E. Global, regional, and national causes of under-5 mortality in 2000-15: An updated systematic analysis with implications for the Sustainable Development Goals. Lancet 2016, 388, 3027–3035. [Google Scholar] [CrossRef]
- Millar, L.J.; Shi, L.; Hoerder-Suabedissen, A.; Molnár, Z. Neonatal Hypoxia Ischaemia: Mechanisms, Models, and Therapeutic Challenges. Front. Cell Neurosci. 2017, 11, 78. [Google Scholar] [CrossRef] [PubMed]
- Ziemka-Nalecz, M.; Jaworska, J.; Zalewska, T. Insights Into the Neuroinflammatory Responses After Neonatal Hypoxia-Ischemia. J. Neuropathol. Exp. Neurol. 2017, 76, 644–654. [Google Scholar] [CrossRef] [PubMed]
- Panfoli, I.; Candiano, G.; Malova, M.; De Angelis, L.; Cardiello, V.; Buonocore, G.; Ramenghi, L.A. Oxidative Stress as a Primary Risk Factor for Brain Damage in Preterm Newborns. Front. Pediatr. 2018, 6, 369. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Alconada, D.; Hilario, E.; Alvarez, F.J.; Alvarez, A. Apoptotic cell death correlates with ROS overproduction and early cytokine expression after hypoxia-ischemia in fetal lambs. Reprod. Sci. 2012, 19, 754–763. [Google Scholar] [CrossRef] [PubMed]
- Hagberg, H.; Mallard, C.; Ferriero, D.M.; Vannucci, S.J.; Levison, S.W.; Vexler, Z.S.; Gressens, P. The role of inflammation in perinatal brain injury. Nat. Rev. Neurol. 2015, 11, 192–208. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Noori, H.; Burkovskiy, I.; Lafreniere, J.D.; Kelly, M.E.M.; Lehmann, C. Modulation of the Endocannabinoid System Following Central Nervous System Injury. Int. J. Mol. Sci. 2019, 20, 388. [Google Scholar] [CrossRef]
- Howlett, A.C.; Barth, F.; Bonner, T.I.; Cabral, G.; Casellas, P.; Devane, W.A.; Felder, C.C.; Herkenham, M.; Mackie, K.; Martin, B.R.; et al. International Union of Pharmacology. XXVII. Classification of cannabinoid receptors. Pharmacol. Rev. 2002, 54, 161–202. [Google Scholar] [CrossRef]
- Alonso-Alconada, D.; Alvarez, F.J.; Alvarez, A.; Mielgo, V.E.; Goni-de-Cerio, F.; Rey-Santano, M.C.; Caballero, A.; Martinez-Orgado, J.; Hilario, E. The cannabinoid receptor agonist WIN 55,212-2 reduces the initial cerebral damage after hypoxic-ischemic injury in fetal lambs. Brain Res. 2010, 1362, 150–159. [Google Scholar] [CrossRef]
- Alonso-Alconada, D.; Alvarez, A.; Alvarez, F.J.; Martinez-Orgado, J.A.; Hilario, E. The cannabinoid WIN 55212-2 mitigates apoptosis and mitochondrial dysfunction after hypoxia ischemia. Neurochem. Res. 2012, 37, 161–170. [Google Scholar] [CrossRef]
- Pellegrini-Giampietro, D.E.; Mannaioni, G.; Bagetta, G. Post-ischemic brain damage: The endocannabinoid system in the mechanisms of neuronal death. FEBS J. 2009, 276, 2–12. [Google Scholar] [CrossRef] [PubMed]
- Rivers-Auty, J.R.; Smith, P.F.; Ashton, J.C. The cannabinoid CB2 receptor agonist GW405833 does not ameliorate brain damage induced by hypoxia-ischemia in rats. Neurosci. Lett. 2014, 569, 104–109. [Google Scholar] [CrossRef]
- Hillard, C.J. Role of cannabinoids and endocannabinoids in cerebral ischemia. Curr. Pharm. Des. 2008, 14, 2347–2361. [Google Scholar] [CrossRef]
- Ashton, J.C.; Glass, M. The cannabinoid CB2 receptor as a target for inflammation-dependent neurodegeneration. Curr. Neuropharmacol. 2007, 5, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Hassell, K.J.; Ezzati, M.; Alonso-Alconada, D.; Hausenloy, D.J.; Robertson, N.J. New horizons for newborn brain protection: Enhancing endogenous neuroprotection. Arch. Dis. Child. Fetal Neonatal Ed. 2015, 100, F541–F552. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.Y.Z.; Chakranon, P.; Lee, S.W.H. Comparative efficacy and safety of neuroprotective therapies for neonates with hypoxic ischemic encephalopathy: A network meta-analysis. Front. Pharmacol. 2019, 10, 1221. [Google Scholar] [CrossRef]
- Zhao, M.; Zhu, P.; Fujino, M.; Zhuang, J.; Guo, H.; Sheikh, I.; Zhao, L.; Li, X.K. Oxidative Stress in Hypoxic-Ischemic Encephalopathy: Molecular Mechanisms and Therapeutic Strategies. Int. J. Mol. Sci. 2016, 17, 2078. [Google Scholar] [CrossRef]
- Torres-Cuevas, I.; Corral-Debrinski, M.; Gressens, P. Brain oxidative damage in murine models of neonatal hypoxia/ischemia and reoxygenation. Free Radic. Biol. Med. 2019, 142, 3–15. [Google Scholar] [CrossRef]
- Kim, S.H.; Won, S.J.; Mao, X.O.; Jin, K.; Greenberg, D.A. Involvement of protein kinase A in cannabinoid receptor-mediated protection from oxidative neuronal injury. J. Pharmacol. Exp. Ther. 2005, 313, 88–94. [Google Scholar] [CrossRef]
- Chung, E.S.; Bok, E.; Chung, Y.C.; Baik, H.H.; Jin, B.K. Cannabinoids prevent lipopolysaccharide-induced neurodegeneration in the rat substantia nigra in vivo through inhibition of microglial activation and NADPH oxidase. Brain Res. 2012, 1451, 110–116. [Google Scholar] [CrossRef]
- Solberg, R.; Andresen, J.H.; Escrig, R.; Vento, M.; Saugstad, O.D. Resuscitation of hypoxic newborn piglets with oxygen induces a dose-dependent increase in markers of oxidation. Pediatr. Res. 2007, 62, 559–563. [Google Scholar] [CrossRef] [PubMed]
- Mukhopadhyay, P.; Rajesh, M.; Pan, H.; Patel, V.; Mukhopadhyay, B.; Batkai, S.; Gao, B.; Hasko, G.; Pacher, P. Cannabinoid-2 receptor limits inflammation, oxidative/nitrosative stress, and cell death in nephropathy. Free Radic. Biol. Med. 2010, 48, 457–467. [Google Scholar] [CrossRef] [PubMed]
- Tiyerili, V.; Zimmer, S.; Jung, S.; Wassmann, K.; Naehle, C.P.; Lutjohann, D.; Zimmer, A.; Nickenig, G.; Wassmann, S. CB1 receptor inhibition leads to decreased vascular AT1 receptor expression, inhibition of oxidative stress and improved endothelial function. Basic Res. Cardiol. 2010, 105, 465–477. [Google Scholar] [CrossRef] [PubMed]
- Shen, M.; Thayer, S.A. Cannabinoid receptor agonists protect cultured rat hippocampal neurons from excitotoxicity. Mol. Pharmacol. 1998, 54, 459–462. [Google Scholar] [CrossRef] [PubMed]
- Marsicano, G.; Moosmann, B.; Hermann, H.; Lutz, B.; Behl, C. Neuroprotective properties of cannabinoids against oxidative stress: Role of the cannabinoid receptor CB1. J. Neurochem. 2002, 80, 448–456. [Google Scholar] [CrossRef] [PubMed]
- Tauber, S.; Paulsen, K.; Wolf, S.; Synwoldt, P.; Pahl, A.; Schneider-Stock, R.; Ullrich, O. Regulation of MMP-9 by a WIN-binding site in the monocyte-macrophage system independent from cannabinoid receptors. PLoS ONE 2012, 7, e48272. [Google Scholar]
- Garberg, H.T.; Huun, M.U.; Escobar, J.; Martinez-Orgado, J.; Loberg, E.M.; Solberg, R.; Didrik Saugstad, O. Short-term effects of cannabidiol after global hypoxia-ischemia in newborn piglets. Pediatr. Res. 2016, 80, 710–718. [Google Scholar] [CrossRef]
- Pazos, M.R.; Cinquina, V.; Gomez, A.; Layunta, R.; Santos, M.; Fernandez-Ruiz, J.; Martinez-Orgado, J. Cannabidiol administration after hypoxia-ischemia to newborn rats reduces long-term brain injury and restores neurobehavioral function. Neuropharmacology 2012, 63, 776–783. [Google Scholar] [CrossRef]
- Satoh, T.; Yoshioka, Y. Contribution of reduced and oxidized glutathione to signals detected by magnetic resonance spectroscopy as indicators of local brain redox state. Neurosci. Res. 2006, 55, 34–39. [Google Scholar] [CrossRef]
- Tuttolomondo, A.; Di Raimondo, D.; di Sciacca, R.; Pinto, A.; Licata, G. Inflammatory cytokines in acute ischemic stroke. Curr. Pharm. Des. 2008, 14, 3574–3589. [Google Scholar] [CrossRef]
- Silveira, R.C.; Procianoy, R.S. Interleukin-6 and tumor necrosis factor-alpha levels in plasma and cerebrospinal fluid of term newborn infants with hypoxic-ischemic encephalopathy. J. Pediatr. 2003, 143, 625–629. [Google Scholar] [CrossRef]
- Aly, H.; Khashaba, M.T.; El-Ayouty, M.; El-Sayed, O.; Hasanein, B.M. IL-1beta, IL-6 and TNF-alpha and outcomes of neonatal hypoxic ischemic encephalopathy. Brain Dev. 2006, 28, 178–182. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Feng, Z.C. Increased umbilical cord plasma interleukin-1 beta levels was correlated with adverse outcomes of neonatal hypoxic-ischemic encephalopathy. J. Trop. Pediatr. 2010, 56, 178–182. [Google Scholar] [CrossRef]
- Girard, S.; Sebire, H.; Brochu, M.E.; Briota, S.; Sarret, P.; Sebire, G. Postnatal administration of IL-1Ra exerts neuroprotective effects following perinatal inflammation and/or hypoxic-ischemic injuries. Brain Behav. Immun. 2012, 26, 1331–1339. [Google Scholar] [CrossRef] [PubMed]
- Stigger, F.; Lovatel, G.; Marques, M.; Bertoldi, K.; Moyses, F.; Elsner, V.; Siqueira, I.R.; Achaval, M.; Marcuzzo, S. Inflammatory response and oxidative stress in developing rat brain and its consequences on motor behavior following maternal administration of LPS and perinatal anoxia. Int. J. Dev. Neurosci. 2013, 31, 820–827. [Google Scholar] [CrossRef]
- Li, S.J.; Liu, W.; Wang, J.L.; Zhang, Y.; Zhao, D.J.; Wang, T.J.; Li, Y.Y. The role of TNF-alpha, IL-6, IL-10, and GDNF in neuronal apoptosis in neonatal rat with hypoxic-ischemic encephalopathy. Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 905–909. [Google Scholar]
- Wajant, H.; Pfizenmaier, K.; Scheurich, P. Tumor necrosis factor signaling. Cell Death Differ. 2003, 10, 45–65. [Google Scholar] [CrossRef]
- Wang, Y.; Cao, M.; Liu, A.; Di, W.; Zhao, F.; Tian, Y.; Jia, J. Changes of inflammatory cytokines and neurotrophins emphasized their roles in hypoxic-ischemic brain damage. Int. J. Neurosci. 2013, 123, 191–195. [Google Scholar] [CrossRef]
- Berdyshev, E.; Boichot, E.; Corbel, M.; Germain, N.; Lagente, V. Effects of cannabinoid receptor ligands on LPS-induced pulmonary inflammation in mice. Life Sci. 1998, 63, PL125–PL129. [Google Scholar] [CrossRef]
- Smith, S.R.; Terminelli, C.; Denhardt, G. Modulation of cytokine responses in Corynebacterium parvum-primed endotoxemic mice by centrally administered cannabinoid ligands. Eur. J. Pharmacol. 2001, 425, 73–83. [Google Scholar] [CrossRef]
- Fernandez-Lopez, D.; Martinez-Orgado, J.; Nunez, E.; Romero, J.; Lorenzo, P.; Moro, M.A.; Lizasoain, I. Characterization of the neuroprotective effect of the cannabinoid agonist WIN-55212 in an in vitro model of hypoxic-ischemic brain damage in newborn rats. Pediatr. Res. 2006, 60, 169–173. [Google Scholar] [CrossRef] [PubMed]
- Szaflarski, J.; Burtrum, D.; Silverstein, F.S. Cerebral hypoxia-ischemia stimulates cytokine gene expression in perinatal rats. Stroke 1995, 26, 1093–1100. [Google Scholar] [CrossRef] [PubMed]
- Hagberg, H.; Gilland, E.; Bona, E.; Hanson, L.A.; Hahin-Zoric, M.; Blennow, M.; Holst, M.; McRae, A.; Soder, O. Enhanced expression of interleukin (IL)-1 and IL-6 messenger RNA and bioactive protein after hypoxia-ischemia in neonatal rats. Pediatr. Res. 1996, 40, 603–609. [Google Scholar] [CrossRef] [PubMed]
- Su, S.H.; Wu, Y.F.; Lin, Q.; Hai, J. Cannabinoid receptor agonist WIN55,212-2 and fatty acid amide hydrolase inhibitor URB597 ameliorate neuroinflammatory responses in chronic cerebral hypoperfusion model by blocking NF-kappaB pathways. Naunyn Schmiedebergs Arch. Pharmacol. 2017, 390, 1189–1200. [Google Scholar] [CrossRef]
- Marchalant, Y.; Brothers, H.M.; Norman, G.J.; Karelina, K.; DeVries, A.C.; Wenk, G.L. Cannabinoids attenuate the effects of aging upon neuroinflammation and neurogenesis. Neurobiol. Dis. 2009, 34, 300–307. [Google Scholar] [CrossRef]
- Hasseldam, H.; Johansen, F.F. Cannabinoid treatment renders neurons less vulnerable than oligodendrocytes in Experimental Autoimmune Encephalomyelitis. Int. J. Neurosci. 2011, 121, 510–520. [Google Scholar] [CrossRef]
- Fernandez-Lopez, D.; Pradillo, J.M.; Garcia-Yebenes, I.; Martinez-Orgado, J.A.; Moro, M.A.; Lizasoain, I. The cannabinoid WIN55212-2 promotes neural repair after neonatal hypoxia-ischemia. Stroke 2010, 41, 2956–2964. [Google Scholar] [CrossRef]
- Zhang, M.; Martin, B.R.; Adler, M.W.; Razdan, R.K.; Jallo, J.I.; Tuma, R.F. Cannabinoid CB(2) receptor activation decreases cerebral infarction in a mouse focal ischemia/reperfusion model. J. Cereb. Blood Flow Metab. 2007, 27, 1387–1396. [Google Scholar] [CrossRef]
- Rivers, J.R.; Ashton, J.C. The development of cannabinoid CBII receptor agonists for the treatment of central neuropathies. Cent. Nerv. Syst. Agents Med. Chem. 2010, 10, 47–64. [Google Scholar] [CrossRef] [PubMed]
- de Lago, E.; Moreno-Martet, M.; Cabranes, A.; Ramos, J.A.; Fernandez-Ruiz, J. Cannabinoids ameliorate disease progression in a model of multiple sclerosis in mice, acting preferentially through CB1 receptor-mediated anti-inflammatory effects. Neuropharmacology 2012, 62, 2299–2308. [Google Scholar] [CrossRef]
- Shimizu, S.; Simon, R.P.; Graham, S.H. Dimethylsulfoxide (DMSO) treatment reduces infarction volume after permanent focal cerebral ischemia in rats. Neurosci. Lett. 1997, 239, 125–127. [Google Scholar] [CrossRef]
- Di Giorgio, A.M.; Hou, Y.; Zhao, X.; Zhang, B.; Lyeth, B.G.; Russell, M.J. Dimethyl sulfoxide provides neuroprotection in a traumatic brain injury model. Restor. Neurol. Neurosci. 2008, 26, 501–507. [Google Scholar]
- Hanslick, J.L.; Lau, K.; Noguchi, K.K.; Olney, J.W.; Zorumski, C.F.; Mennerick, S.; Farber, N.B. Dimethyl sulfoxide (DMSO) produces widespread apoptosis in the developing central nervous system. Neurobiol. Dis. 2009, 34, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Huizenga, M.N.; Forcelli, P.A. Neuroprotective Action of the CB1/2 Receptor Agonist, WIN 55,212-2, against DMSO but Not Phenobarbital-Induced Neurotoxicity in Immature Rats. Neurotox. Res. 2019, 35, 173–182. [Google Scholar] [CrossRef]
- Hilario, E.; Rey-Santano, M.C.; Goni-de-Cerio, F.; Alvarez, F.J.; Gastiasoro, E.; Mielgo, V.E.; Caballero, A.; Valls-i-Soler, A.; Gomez-Urquijo, S.; Alvarez, A. Cerebral blood flow and morphological changes after hypoxic-ischaemic injury in preterm lambs. Acta Paediatr. 2005, 94, 903–911. [Google Scholar] [CrossRef] [PubMed]
- Back, S.A.; Riddle, A.; Dean, J.; Hohimer, A.R. The instrumented fetal sheep as a model of cerebral white matter injury in the premature infant. Neurotherapeutics 2012, 9, 359–370. [Google Scholar] [CrossRef] [PubMed]
- Rey-Santano, C.; Mielgo, V.E.; Gastiasoro, E.; Murgia, X.; Lafuente, H.; Ruiz-Del-Yerro, E.; Valls-I-Soler, A.; Hilario, E.; Alvarez, F.J. Early cerebral hemodynamic, metabolic, and histological changes in hypoxic-ischemic fetal lambs during postnatal life. Front. Neurosci. 2011, 5, 111. [Google Scholar] [CrossRef] [PubMed][Green Version]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alonso-Alconada, D.; Álvarez, F.J.; Goñi-de-Cerio, F.; Hilario, E.; Álvarez, A. Cannabinoid-mediated Modulation of Oxidative Stress and Early Inflammatory Response after Hypoxia–Ischemia. Int. J. Mol. Sci. 2020, 21, 1283. https://doi.org/10.3390/ijms21041283
Alonso-Alconada D, Álvarez FJ, Goñi-de-Cerio F, Hilario E, Álvarez A. Cannabinoid-mediated Modulation of Oxidative Stress and Early Inflammatory Response after Hypoxia–Ischemia. International Journal of Molecular Sciences. 2020; 21(4):1283. https://doi.org/10.3390/ijms21041283
Chicago/Turabian StyleAlonso-Alconada, Daniel, Francisco José Álvarez, Felipe Goñi-de-Cerio, Enrique Hilario, and Antonia Álvarez. 2020. "Cannabinoid-mediated Modulation of Oxidative Stress and Early Inflammatory Response after Hypoxia–Ischemia" International Journal of Molecular Sciences 21, no. 4: 1283. https://doi.org/10.3390/ijms21041283
APA StyleAlonso-Alconada, D., Álvarez, F. J., Goñi-de-Cerio, F., Hilario, E., & Álvarez, A. (2020). Cannabinoid-mediated Modulation of Oxidative Stress and Early Inflammatory Response after Hypoxia–Ischemia. International Journal of Molecular Sciences, 21(4), 1283. https://doi.org/10.3390/ijms21041283



