Hepatitis C Virus p7 Induces Membrane Permeabilization by Interacting with Phosphatidylserine
Abstract
1. Introduction
2. Results
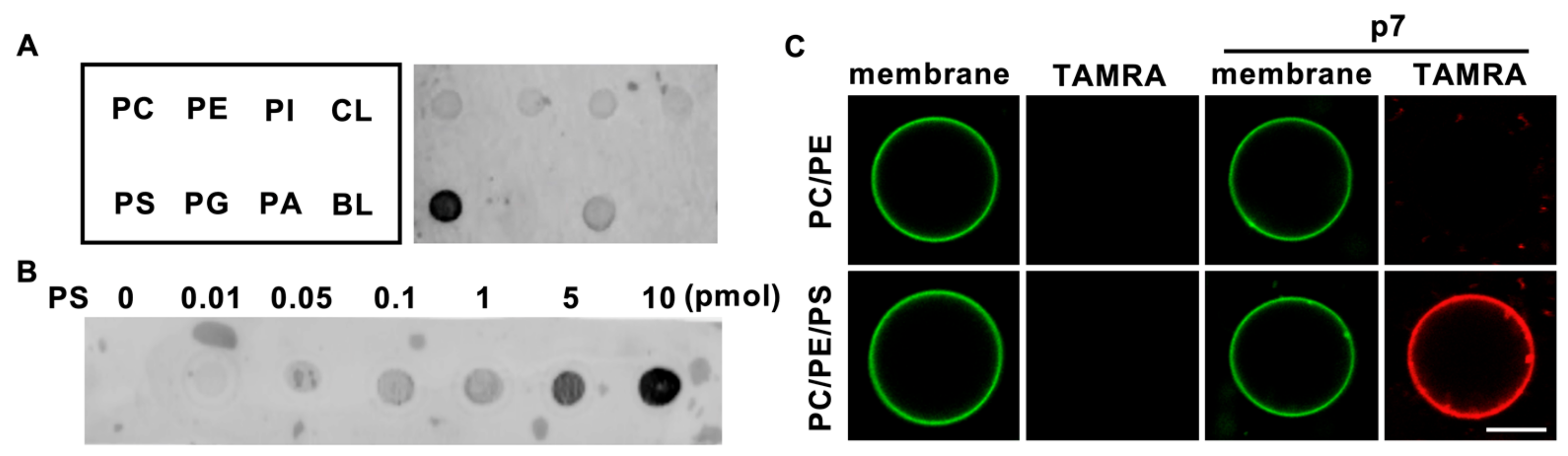
2.1. HCV p7 Specifically Interacts with PS
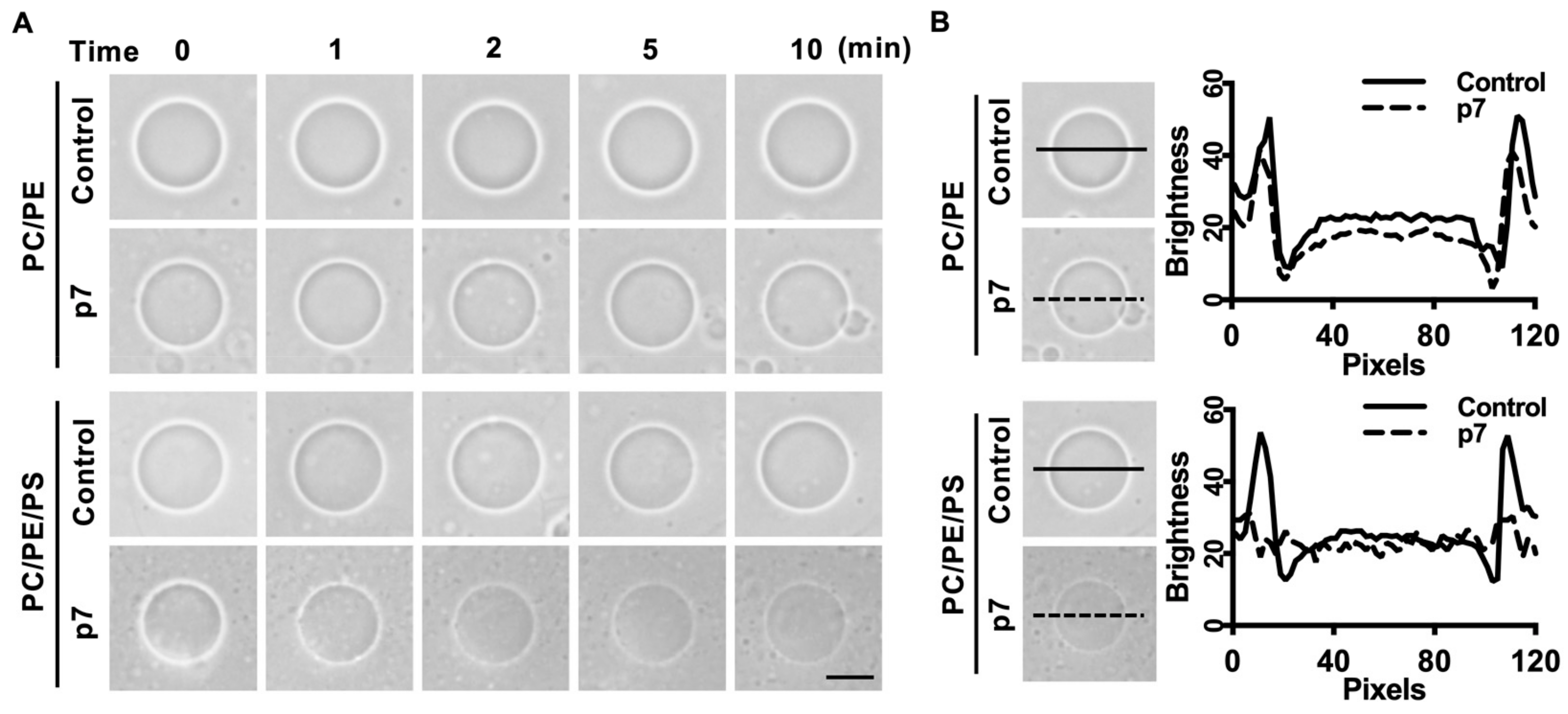
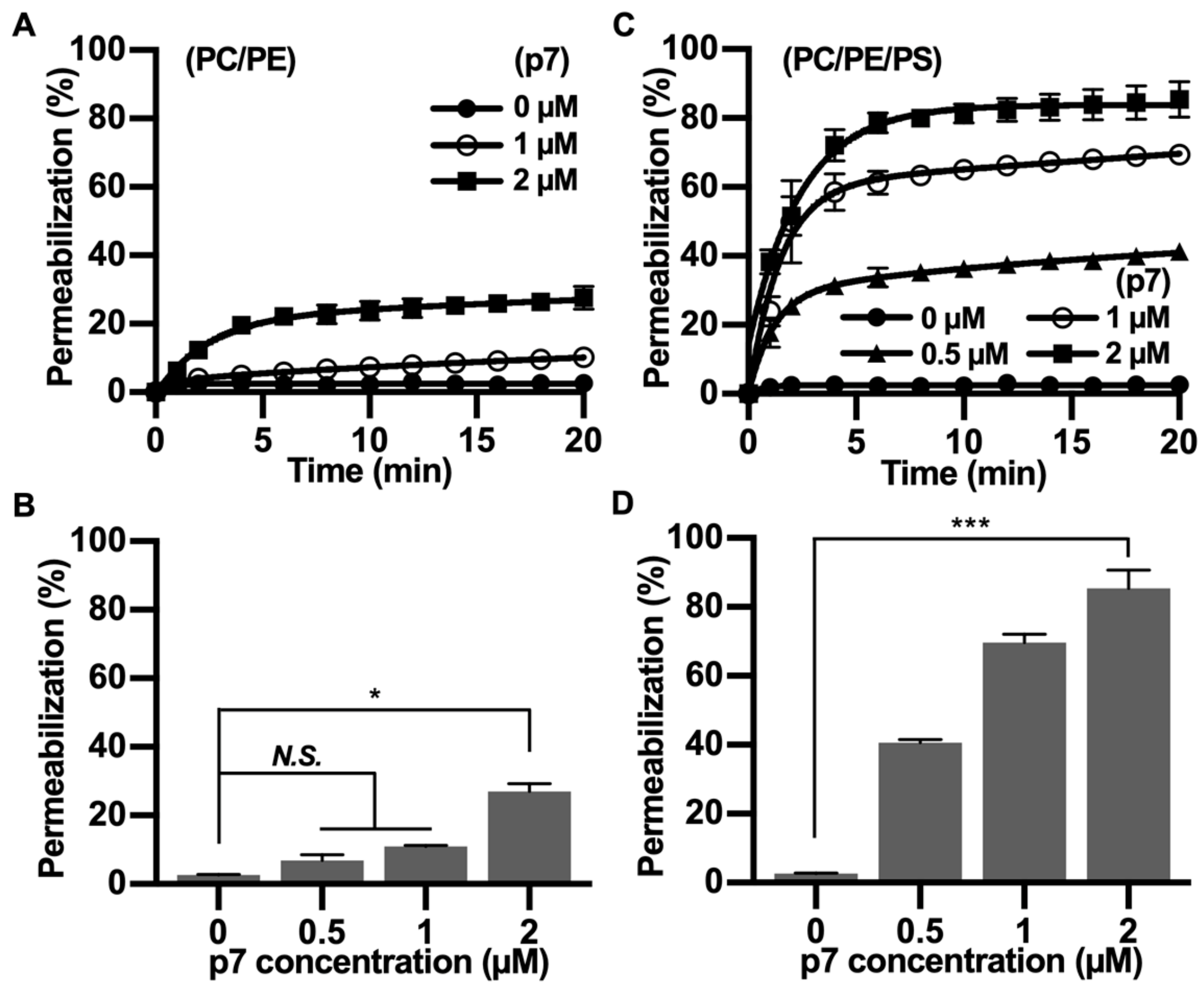
2.2. HCV p7 Enhances Membrane Permeabilization in the Presence of PS
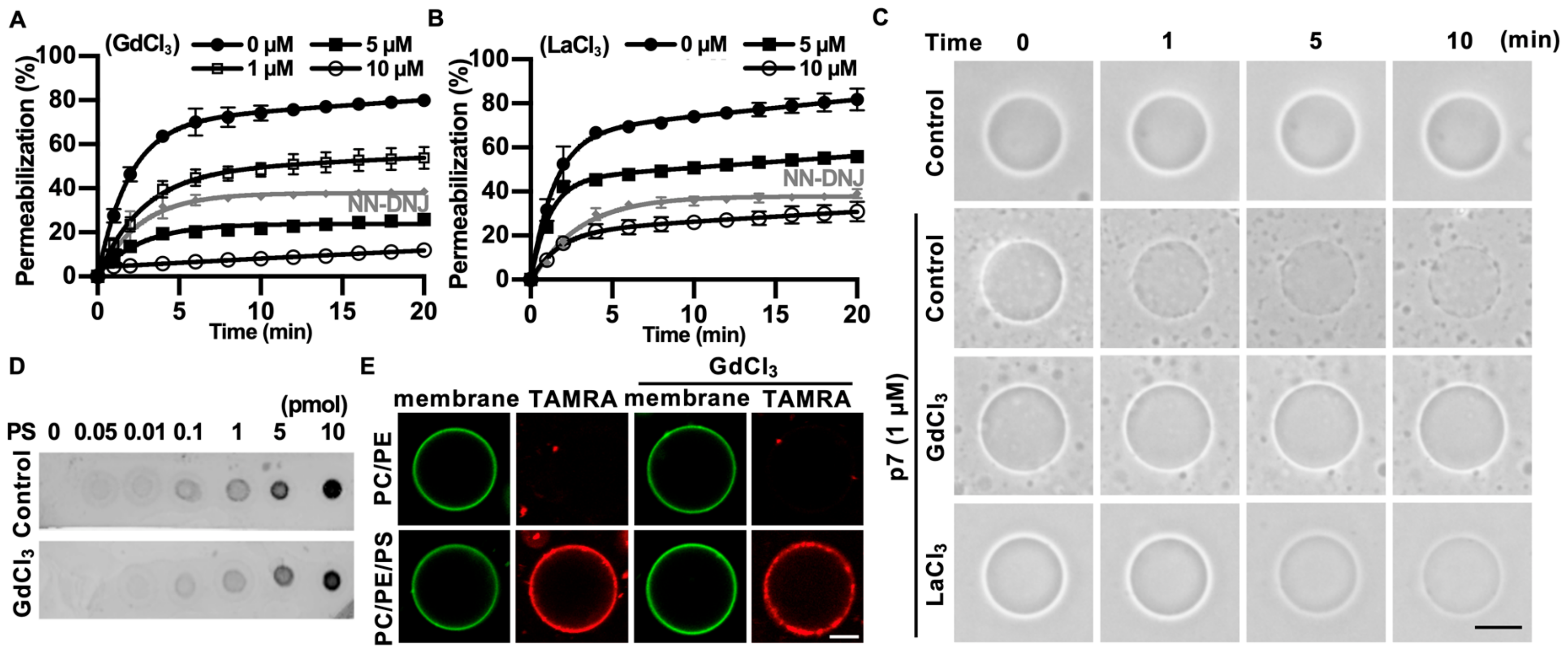
2.3. Gd3+ Ions Block p7-Induced Membrane Permeabilization
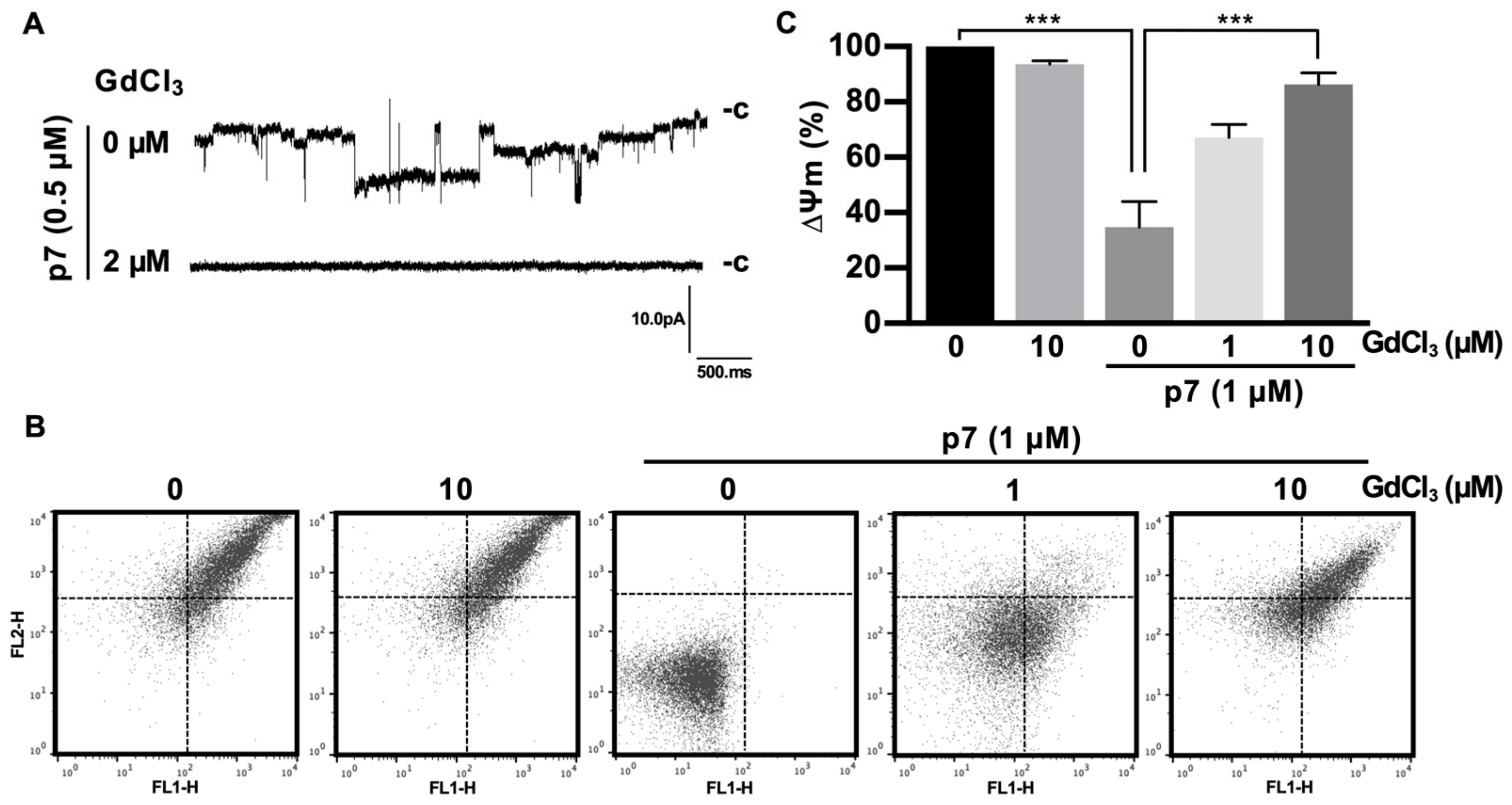
2.4. Gd3+ Ions Block the Channel Activity of p7 in the Bilayer and Inhibit p7-Induced Mitochondrial Depolarization
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. PLO Assay
4.3. GUV Preparation
4.4. LUV Preparation
4.5. Flow Cytometric Assay
4.6. Planar Lipid Bilayer Recordings
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| HCV | Hepatitis C virus |
| ER | Endoplasmic reticulum |
| LDs | Lipid droplets |
| PI4KA | Phosphatidylinositol 4-kinase alpha |
| PI4P | Phosphatidylinositol 4-phosphate |
| PI(4,5)P2 | Phosphatidylinositol 4,5-bisphosphate |
| LUVs | Large unilamellar vesicles |
| PC | Phosphatidylcholine |
| PE | Phosphatidylethanolamine |
| PI | Phosphatidylinositol |
| PS | Phosphatidylserine |
| PG | Phosphatidylglycerol |
| PA | Phosphatidic acid |
| CL | Cardiolipin |
| TAMRA | 5-carboxytetramethylrhodamine |
| TFE | Trifluoroethanol |
| PLO | Protein lipid overlay |
| GUV | Giant unilamellar vesicle |
| CF | Carboxyfluorescein |
| NN-DNJ | N-nonyl-deoxynojirimycin |
| Aman. | Amantadine |
| DphPC | 2-diphytanoyl-sn-glycero-3-phosphatidylcholine |
| DphPS | 2-diphytanoyl-sn-glycero-3-phosphatidylserine |
| DOPC | 1,2-dioleoyl-sn-glycero-3-phosphatidylcholine |
| DOPE | 1,2-dioleoyl-sn-glycero-3-phosphatidylethanolamine |
| DOPS | 1,2-dioleoyl-sn-glycero-3-phosphatidylserine |
| TF-CHOL | TopFluor-cholesterol |
| TBS | Tris-buffered saline |
| EGTA | Ethylene glycol tetra-acetic acid |
References
- Moradpour, D.; Penin, F.; Rice, C.M. Replication of hepatitis C virus. Nat. Rev. Microbiol. 2007, 5, 453. [Google Scholar] [CrossRef] [PubMed]
- Feld, J.J.; Jacobson, I.M.; Hézode, C.; Asselah, T.; Ruane, P.J.; Gruener, N.; Abergel, A.; Mangia, A.; Lai, C.-L.; Chan, H.L. Sofosbuvir and velpatasvir for HCV genotype 1, 2, 4, 5, and 6 infection. N. Engl. J. Med. 2015, 373, 2599–2607. [Google Scholar] [CrossRef] [PubMed]
- Chevaliez, S.; Pawlotsky, J.-M. HCV genome and life cycle. In Hepatitis C Viruses: Genomes and Molecular Biology; Tan, S.L., Ed.; Horizon Bioscience: Norfolk, UK, 2006; Volume 1, pp. 5–47. [Google Scholar]
- Vescovo, T.; Romagnoli, A.; Perdomo, A.B.; Corazzari, M.; Ciccosanti, F.; Alonzi, T.; Nardacci, R.; Ippolito, G.; Tripodi, M.; Garcia–Monzon, C. Autophagy protects cells from HCV-induced defects in lipid metabolism. Gastroenterology 2012, 142, 644–653. [Google Scholar] [CrossRef] [PubMed]
- Lorizate, M.; Kräusslich, H.-G. Role of lipids in virus replication. CSH Perspect. Biol. 2011, 3, a004820. [Google Scholar] [CrossRef] [PubMed]
- Simons, K.; Ikonen, E. Functional rafts in cell membranes. Nature 1997, 387, 569. [Google Scholar] [CrossRef] [PubMed]
- Miyanari, Y.; Atsuzawa, K.; Usuda, N.; Watashi, K.; Hishiki, T.; Zayas, M.; Bartenschlager, R.; Wakita, T.; Hijikata, M.; Shimotohno, K. The lipid droplet is an important organelle for hepatitis C virus production. Nat. Cell Biol. 2007, 9, 1089. [Google Scholar] [CrossRef]
- Tai, A.W.; Salloum, S. The role of the phosphatidylinositol 4-kinase PI4KA in hepatitis C virus-induced host membrane rearrangement. PLoS ONE 2011, 6, e26300. [Google Scholar] [CrossRef]
- Rodenhuis-Zybert, I.A.; Wilschut, J.; Smit, J.M. Dengue virus life cycle: viral and host factors modulating infectivity. Cell. Mol. Life Sci. 2010, 67, 2773–2786. [Google Scholar] [CrossRef]
- Pavlović, D.; Neville, D.C.; Argaud, O.; Blumberg, B.; Dwek, R.A.; Fischer, W.B.; Zitzmann, N. The hepatitis C virus p7 protein forms an ion channel that is inhibited by long-alkyl-chain iminosugar derivatives. Proc. Natl. Acad. Sci. USA 2003, 100, 6104–6108. [Google Scholar] [CrossRef]
- Tedbury, P.; Welbourn, S.; Pause, A.; King, B.; Griffin, S.; Harris, M. The subcellular localization of the hepatitis C virus non-structural protein NS2 is regulated by an ion channel-independent function of the p7 protein. J. Gen. Virol. 2011, 92, 819–830. [Google Scholar] [CrossRef]
- Lee, G.Y.; Lee, S.; Lee, H.-R.; Do Yoo, Y. Hepatitis C virus p7 mediates membrane-to-membrane adhesion. Biochim. Biophys. Acta 2016, 1861, 1096–1101. [Google Scholar] [CrossRef] [PubMed]
- Van Meer, G.; de Kroon, A.I. Lipid map of the mammalian cell. J. Cell. Sci. 2011, 124, 5–8. [Google Scholar] [CrossRef] [PubMed]
- Friedrich, C.; Scott, M.G.; Karunaratne, N.; Yan, H.; Hancock, R.E. Salt-resistant alpha-helical cationic antimicrobial peptides. Antimicrob. Agents Chemother. 1999, 43, 1542–1548. [Google Scholar] [CrossRef] [PubMed]
- Mazzon, M.; Mercer, J. Lipid interactions during virus entry and infection. Cell. Microbiol. 2014, 16, 1493–1502. [Google Scholar] [CrossRef] [PubMed]
- Largo, E.; Gladue, D.P.; Huarte, N.; Borca, M.V.; Nieva, J.L. Pore-forming activity of pestivirus p7 in a minimal model system supports genus-specific viroporin function. Antiviral Res. 2014, 101, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Premkumar, A.; Wilson, L.; Ewart, G.; Gage, P. Cation-selective ion channels formed by p7 of hepatitis C virus are blocked by hexamethylene amiloride. FEBS Lett. 2004, 557, 99–103. [Google Scholar] [CrossRef]
- Caldwell, R.A.; Clemo, H.F.; Baumgarten, C.M. Using gadolinium to identify stretch-activated channels: technical considerations. Am. J. Physiol. Cell Physiol. 1998, 275, C619–CC621. [Google Scholar] [CrossRef]
- You, D.G.; Lee, H.R.; Kim, W.K.; Kim, H.J.; Lee, G.Y.; Yoo, Y.D. Hepatitis C virus p7 induces mitochondrial depolarization of isolated liver mitochondria. Mol. Med. Rep. 2017, 16, 9533–9538. [Google Scholar] [CrossRef][Green Version]
- Ingólfsson, H.I.; Melo, M.N.; Van Eerden, F.J.; Arnarez, C.; Lopez, C.A.; Wassenaar, T.A.; Periole, X.; De Vries, A.H.; Tieleman, D.P.; Marrink, S.J. Lipid organization of the plasma membrane. J. Am. Chem. Soc. 2014, 136, 14554–14559. [Google Scholar] [CrossRef]
- Marsh, D. Lipid-protein interactions in membranes. FEBS Lett. 1990, 268, 371–375. [Google Scholar] [CrossRef]
- Sengupta, T.; Manoj, N. Phosphatidylserine and phosphatidylethanolamine bind to protein Z cooperatively and with equal affinity. PLoS ONE 2016, 11, e0161896. [Google Scholar] [CrossRef] [PubMed]
- Von Schwedler, U.K.; Stuchell, M.; Müller, B.; Ward, D.M.; Chung, H.-Y.; Morita, E.; Wang, H.E.; Davis, T.; He, G.-P.; Cimbora, D.M. The protein network of HIV budding. Cell 2003, 114, 701–713. [Google Scholar] [CrossRef]
- Pedersen, U.R.; Leidy, C.; Westh, P.; Peters, G.H. The effect of calcium on the properties of charged phospholipid bilayers. Biochim. Biophys. Acta 2006, 1758, 573–582. [Google Scholar] [CrossRef] [PubMed]
- Boyd, D.; Beckwith, J. Positively charged amino acid residues can act as topogenic determinants in membrane proteins. Proc. Natl. Acad. Sci. USA 1989, 86, 9446–9450. [Google Scholar] [CrossRef]
- Griffin, S.D.; Harvey, R.; Clarke, D.S.; Barclay, W.S.; Harris, M.; Rowlands, D.J. A conserved basic loop in hepatitis C virus p7 protein is required for amantadine-sensitive ion channel activity in mammalian cells but is dispensable for localization to mitochondria. J. Gen. Virol. 2004, 85, 451–461. [Google Scholar] [CrossRef]
- Li, C.; Ullrich, B.; Zhang, J.Z.; Anderson, R.G.; Brose, N.; Südhof, T.C. Ca2+-dependent and-independent activities of neural and non-neural synaptotagmins. Nature 1995, 375, 594–599. [Google Scholar] [CrossRef]
- Krasznai, Z.; Morisawa, M.; Krasznai, Z.T.; Morisawa, S.; Inaba, K.; Bazsáné, Z.K.; Rubovszky, B.; Bodnár, B.; Borsos, A.; Márián, T. Gadolinium, a mechano-sensitive channel blocker, inhibits osmosis-initiated motility of sea-and freshwater fish sperm, but does not affect human or ascidian sperm motility. Cell Motil. Cytoskeleton 2003, 55, 232–243. [Google Scholar] [CrossRef]
- Chew, C.F.; Vijayan, R.; Chang, J.; Zitzmann, N.; Biggin, P.C. Determination of pore-lining residues in the hepatitis C virus p7 protein. Biophys. J. 2009, 96, L10–L12. [Google Scholar] [CrossRef]
- Kim, Y.-J.; Jaramillo, D.; Millis, M.B.; Gray, M.L.; Burstein, D. Assessment of early osteoarthritis in hip dysplasia with delayed gadolinium-enhanced magnetic resonance imaging of cartilage. JBJS 2003, 85, 1987–1992. [Google Scholar] [CrossRef]
- Dowler, S.; Kular, G.; Alessi, D.R. Protein lipid overlay assay. Sci. STKE 2002, 2002, pl6. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, H.-R.; Lee, G.Y.; You, D.-G.; Kim, H.K.; Yoo, Y.D. Hepatitis C Virus p7 Induces Membrane Permeabilization by Interacting with Phosphatidylserine. Int. J. Mol. Sci. 2020, 21, 897. https://doi.org/10.3390/ijms21030897
Lee H-R, Lee GY, You D-G, Kim HK, Yoo YD. Hepatitis C Virus p7 Induces Membrane Permeabilization by Interacting with Phosphatidylserine. International Journal of Molecular Sciences. 2020; 21(3):897. https://doi.org/10.3390/ijms21030897
Chicago/Turabian StyleLee, Hye-Ra, Gi Young Lee, Deok-Gyun You, Hong Kyu Kim, and Young Do Yoo. 2020. "Hepatitis C Virus p7 Induces Membrane Permeabilization by Interacting with Phosphatidylserine" International Journal of Molecular Sciences 21, no. 3: 897. https://doi.org/10.3390/ijms21030897
APA StyleLee, H.-R., Lee, G. Y., You, D.-G., Kim, H. K., & Yoo, Y. D. (2020). Hepatitis C Virus p7 Induces Membrane Permeabilization by Interacting with Phosphatidylserine. International Journal of Molecular Sciences, 21(3), 897. https://doi.org/10.3390/ijms21030897




