High-Density Lipoproteins as Homeostatic Nanoparticles of Blood Plasma
Abstract
1. Introduction
2. Composition and Structure of HDL
3. Functions of HDL
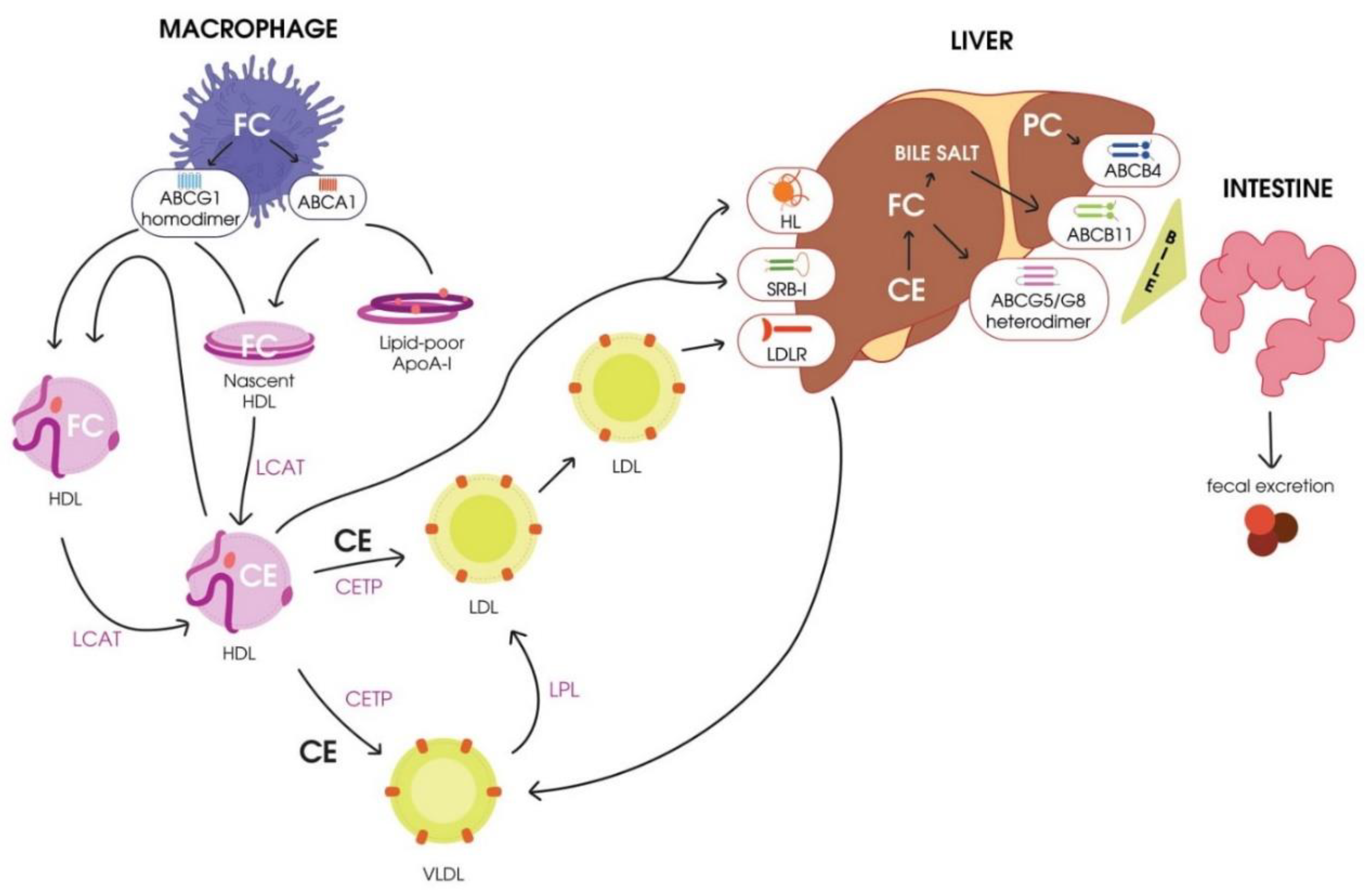
3.1. Reverse Cholesterol Transport (RCT)
- Cholesterol mobilization and transport to the plasmatic membrane of the cell.
- Esterification of free cholesterol bound to HDL by LCAT.
- Transport of cholesterol esters to hepatocytes by mature HDL.
- Engulfment of cholesterol esters from mature HDL particles mediated by the hepatocyte scavenger receptor class B type I (SR-BI) [38].
- Transport of cholesterol esters to hepatocytes by mature low-density lipoproteins (LDLs) due to cholesterol exchange to HDL mediated by CETP.
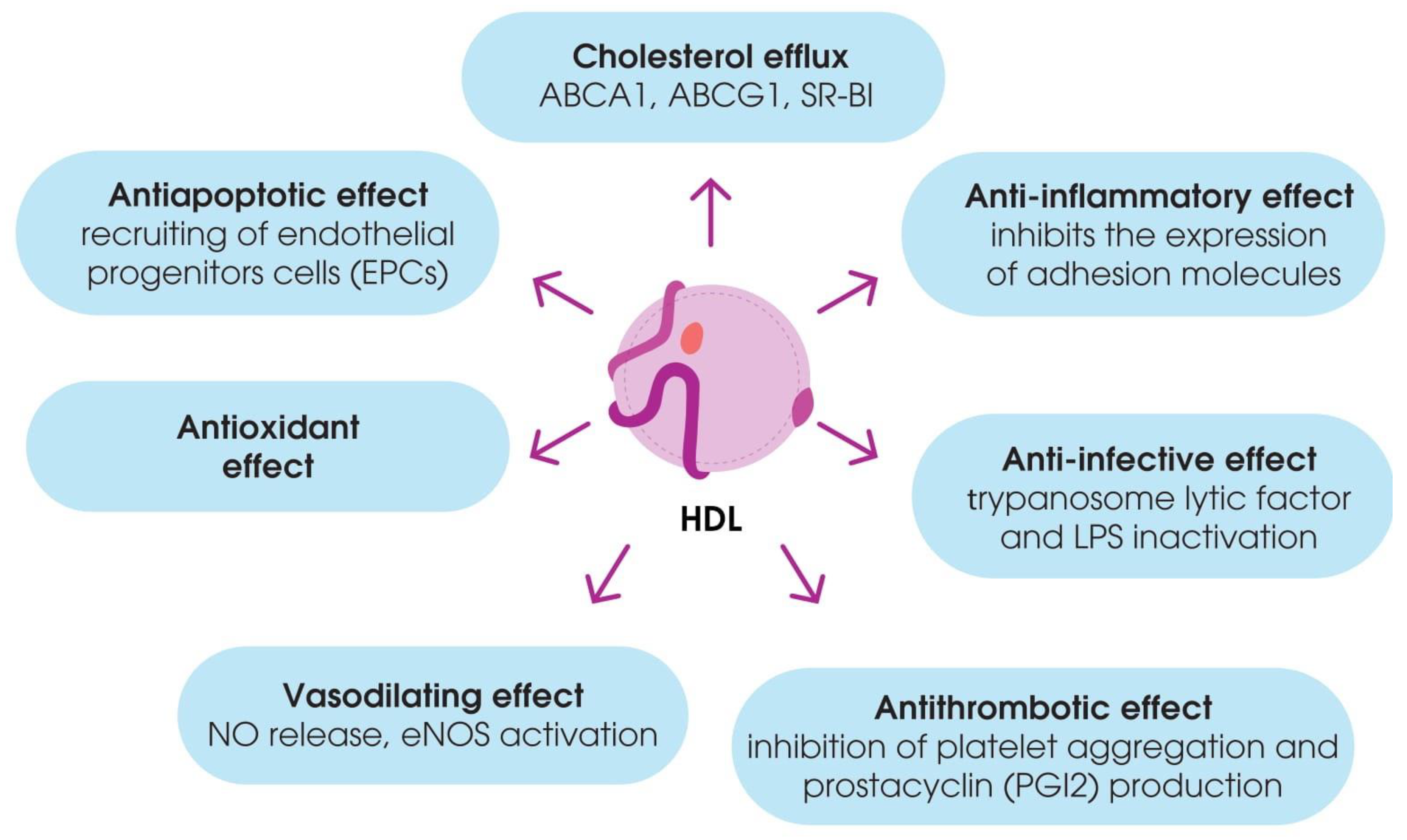
3.2. Non-Classical Functions of HDL
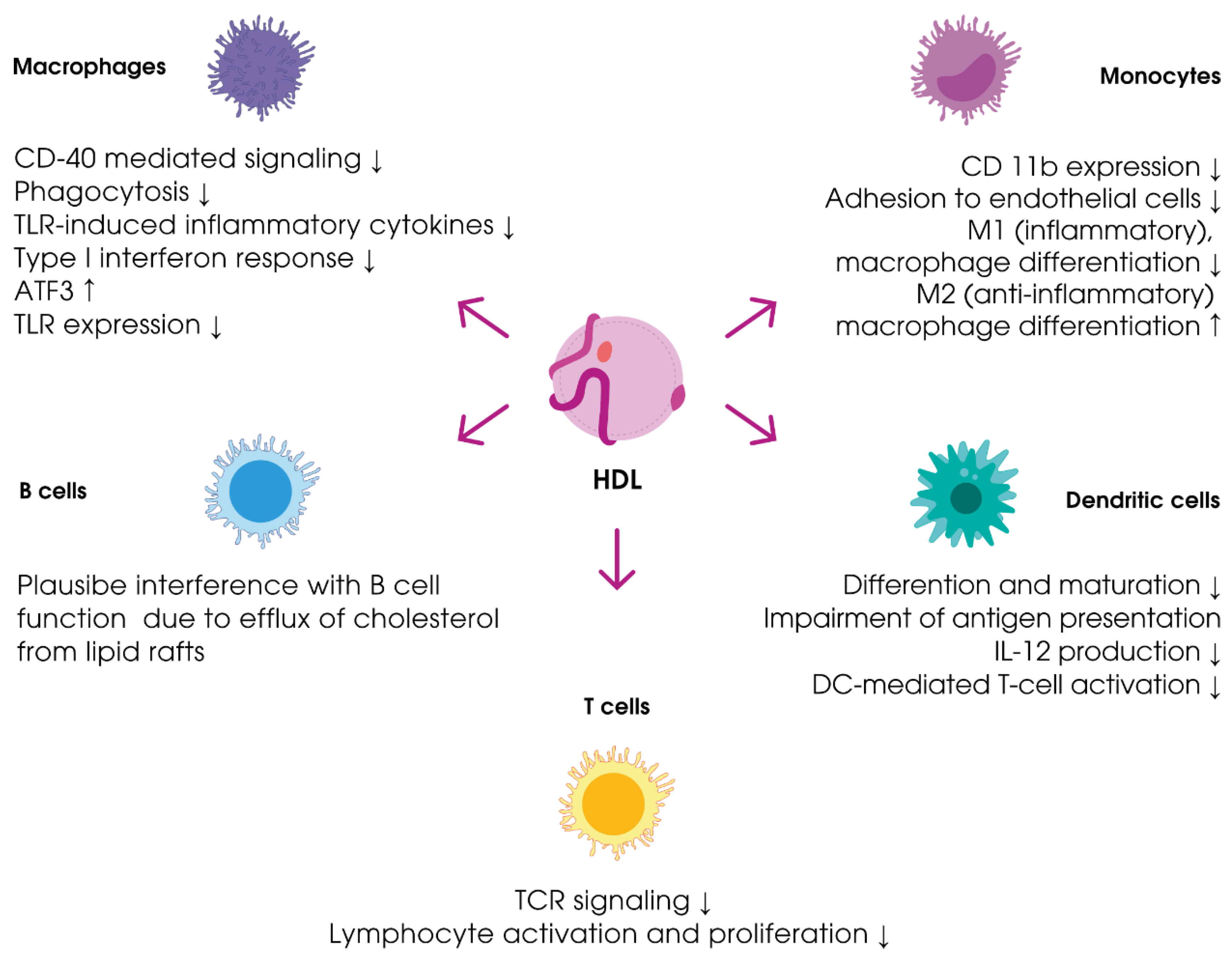
3.2.1. Immune Reactions
3.2.2. Antioxidant Properties
3.2.3. Anti-Inflammatory Properties
3.2.4. Vasodilating and Endothelial-Protecting Properties
3.2.5. Antithrombotic Properties
3.2.6. Contribution of HDL to MicroRNA Transport
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ABCA1 | ATP Binding Cassette Subfamily A Member 1 |
| ABCG1 | ATP Binding Cassette Subfamily G Member 1 |
| CETP | cholesteryl ester transfer protein |
| DC | dendritic cell |
| GGE | gradient non-denaturing polyacrylamide gel electrophoresis |
| GSPx-3 | glutathione selenoperoxidase 3 |
| HDL | high-density lipoproteins |
| Hrp | haptoglobin-related protein |
| ICAM-I | intercellular adhesion molecule 1 |
| IL | interleukin |
| LCAT | lecithin/cholesterol acyltransferase |
| LDL | low-density lipoproteins |
| LDLR | low-density lipoprotein receptor |
| LP | lipoprotein |
| LPL | lipoprotein lipase |
| LpPLA2 | lipoprotein-associated phospholipase A2 |
| LRP | LDL receptor-related protein |
| LPS | lipopolysaccharide |
| NETs | neutrophil extracellular traps |
| NF-kB | nuclear factor kappa B |
| NRF3 | NLR Family Pyrin Domain Containing 3 |
| MHC | major histocompatibility complex |
| PAF-AH | platelet-activating factor acetyl hydrolase |
| PL | phospholipid |
| PLTP | phospholipid transfer protein |
| PON1 | paraoxonase 1 |
| SAA | serum amyloid A |
| S1P | sphingosine-1-phosphate |
| SR-BI | scavenger receptor class B type I |
| TG | triglyceride |
| TLF-1 | trypanosome lytic factor-1 |
| TLR | Toll-like receptor |
| TNF-α | tumor necrosis factor α |
| TCR | T cell receptor |
| VCAM-1 | vascular cell adhesion molecule 1 |
| VLDL | very-low-density lipoproteins |
References
- Castelli, W.P.; Anderson, K.; Wilson, P.W.F.; Levy, D. Lipids and risk of coronary heart disease. The framingham study. Ann. Epidemiol. 1992, 2, 23–28. [Google Scholar] [CrossRef]
- Vitali, C.; Khetarpal, S.A.; Rader, D.J. HDL cholesterol metabolism and the risk of CHD: New insights from human genetics. Curr. Cardiol. Rep. 2017, 19, 132. [Google Scholar] [CrossRef] [PubMed]
- Kudinov, V.A.; Zakharova, T.S.; Torkhovskaya, T.I.; Ipatova, O.M.; Archakov, A.I. Pharmacological targets for correction of dyslipidemias. Opportunities and prospects of therapeutic use. Biochem. (Moscow) Suppl. Ser. B Biomed. Chem. 2018, 12, 96–113. [Google Scholar] [CrossRef]
- Ouimet, M.; Barrett, T.J.; Fisher, E.A. HDL and reverse cholesterol transport. Circ. Res. 2019, 124, 1505–1518. [Google Scholar] [CrossRef] [PubMed]
- Armitage, J.; Holmes, M.V.; Preiss, D. Cholesteryl ester transfer protein inhibition for preventing cardiovascular events: JACC review topic of the week. J. Am. Coll. Cardiol. 2019, 73, 477–487. [Google Scholar] [CrossRef] [PubMed]
- Annema, W.; von Eckardstein, A. Dysfunctional high-density lipoproteins in coronary heart disease: Implications for diagnostics and therapy. Transl. Res. 2016, 173, 30–57. [Google Scholar] [CrossRef] [PubMed]
- Kuai, R.; Li, D.; Chen, Y.E.; Moon, J.J.; Schwendeman, A. High-Density Lipoproteins (HDL)-nature’s multi-functional nanoparticles. ACS Nano 2016, 10, 3015–3041. [Google Scholar] [CrossRef]
- Bonacina, F.; Pirillo, A.; Catapano, A.L.; Norata, G.D. Cholesterol membrane content has a ubiquitous evolutionary function in immune cell activation: The role of HDL. Curr. Opin. Lipidol. 2019, 30, 462–469. [Google Scholar] [CrossRef]
- Primer, K.R.; Psaltis, P.J.; Tan, J.T.M.; Bursill, C.A. The role of high-density lipoproteins in endothelial cell metabolism and diabetes-impaired angiogenesis. Int. J. Mol. Sci. 2020, 21, 3633. [Google Scholar] [CrossRef]
- Haghikia, A.; Landmesser, U. High-density lipoproteins: Effects on vascular function and role in the immune response. Cardiol. Clin. 2018, 36, 317–327. [Google Scholar] [CrossRef]
- Jomard, A.; Osto, E. High density lipoproteins: Metabolism, function, and therapeutic potential. Front. Cardiovasc. Med. 2020, 7, 39. [Google Scholar] [CrossRef] [PubMed]
- Macheboeuf, M. Recherches sur les phosphoaminolipides du sérum sanguin. Nature des phospholipides liés aux albumines du sérum de Cheval à l’état de cenapses acido-précipitables. Bull. Soc. Chim. Biol. (Paris) 1929, 11, 485–503. [Google Scholar]
- Lund-Katz, S.; Liu, L.; Thuahnai, S.T.; Phillips, M.C. High density lipoprotein structure. Front. Biosci. 2003, 8, d1044–d1054. [Google Scholar] [CrossRef] [PubMed]
- Camont, L.; Chapman, M.J.; Kontush, A. Biological activities of HDL subpopulations and their relevance to cardiovascular disease. Trends Mol. Med. 2011, 17, 594–603. [Google Scholar] [CrossRef] [PubMed]
- Rosenson, R.S.; Brewer, H.; Bryan, J.; Chapman, M.J.; Fazio, S.; Hussain, M.M.; Kontush, A.; Krauss, R.M.; Otvos, J.D.; Remaley, A.T.; et al. HDL measures, particle heterogeneity, proposed nomenclature, and relation to atherosclerotic cardiovascular events. Clin. Chem. 2011, 57, 392–410. [Google Scholar] [CrossRef] [PubMed]
- Otvos, J.D. Measurement of lipoprotein subclass profiles by nuclear magnetic resonance spectroscopy. Clin. Lab. 2002, 48, 171–180. [Google Scholar] [PubMed]
- Vaisar, T.; Kanter, J.E.; Wimberger, J.; Irwin, A.D.; Gauthier, J.; Wolfson, E.; Bahnam, V.; Wu, I.-H.; Shah, H.; Keenan, H.A.; et al. High concentration of medium-sized HDL particles and enrichment in HDL paraoxonase 1 associate with protection from vascular complications in people with long-standing type 1 diabetes. Diabetes Care 2020, 43, 178–186. [Google Scholar] [CrossRef]
- Kontush, A.; Lindahl, M.; Lhomme, M.; Calabresi, L.; Chapman, M.J.; Davidson, W.S. Structure of HDL: Particle subclasses and molecular components. Handb. Exp. Pharmacol. 2015, 224, 3–51. [Google Scholar] [CrossRef]
- Otvos, J.D.; Jeyarajah, E.J.; Bennett, D.W.; Krauss, R.M. Development of a proton nuclear magnetic resonance spectroscopic method for determining plasma lipoprotein concentrations and subspecies distributions from a single, rapid measurement. Clin. Chem. 1992, 38, 1632–1638. [Google Scholar] [CrossRef]
- Fielding, C.J.; Fielding, P.E. Molecular physiology of reverse cholesterol transport. J. Lipid Res. 1995, 36, 211–228. [Google Scholar]
- Yamatani, K.; Hirayama, S.; Seino, U.; Hirayama, A.; Hori, A.; Suzuki, K.; Idei, M.; Kitahara, M.; Miida, T. Preβ1-high-density lipoprotein metabolism is delayed in patients with chronic kidney disease not on hemodialysis. J. Clin. Lipidol. 2020, 14, 730–739. [Google Scholar] [CrossRef] [PubMed]
- Cavigiolio, G.; Shao, B.; Geier, E.G.; Ren, G.; Heinecke, J.W.; Oda, M.N. The interplay between size, morphology, stability, and functionality of high-density lipoprotein subclasses. Biochemistry 2008, 47, 4770–4779. [Google Scholar] [CrossRef] [PubMed]
- Dergunov, A.D. Kinetic analysis of lecithin:cholesterol acyltransferase activity toward discoidal HDL. Lipids 2011, 46, 1075–1079. [Google Scholar] [CrossRef] [PubMed]
- Cedó, L.; Plana, N.; Metso, J.; Lee-Rueckert, M.; Sanchez-Quesada, J.L.; Kovanen, P.T.; Jauhiainen, M.; Masana, L.; Escolà-Gil, J.C.; Blanco-Vaca, F. Altered HDL remodeling and functionality in familial hypercholesterolemia. J. Am. Coll. Cardiol. 2018, 71, 466–468. [Google Scholar] [CrossRef] [PubMed]
- Asztalos, B.; Brunzell, J. The kinetics and remodeling of HDL particles: Lessons from inborn errors of lipid metabolism. In High Density Lipoproteins, Dyslipidemia, and Coronary Heart Disease; Springer: New York, NY, USA, 2010; pp. 33–44. [Google Scholar] [CrossRef]
- Thompson, G.R.; Sharp, M.; Limited, D. A Handbook of Hyperlipidaemia; Current Science: London, UK, 1990; ISBN 1870485165. [Google Scholar]
- Shah, A.; Tan, L.; Long, J.; Davidson, W. Proteomic diversity of high density lipoproteins: Our emerging understanding of its importance in lipid transport and beyond. J. Lipid Res. 2013, 54, 2575–2585. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.-H.; Zheng, L.; Wei, J.; Yu, M.-M.; Zhang, J.; Luo, G.-H.; Xu, N. Increased apolipoprotein M induced by lack of scavenger receptor BI is not activated via HDL-mediated cholesterol uptake in hepatocytes. Lipids Health Dis. 2018, 17, 200. [Google Scholar] [CrossRef]
- Vaisar, T.; Tang, C.; Babenko, I.; Hutchins, P.; Wimberger, J.; Suffredini, A.F.; Heinecke, J.W. Inflammatory remodeling of the HDL proteome impairs cholesterol efflux capacity. J. Lipid Res. 2015, 56, 1519–1530. [Google Scholar] [CrossRef]
- Salazar, J.; Olivar, L.C.; Ramos, E.; Chavez-Castillo, M.; Rojas, J.; Bermudez, V. Dysfunctional high-density lipoprotein: An innovative target for proteomics and lipidomics. Cholesterol 2015, 2015, 296417. [Google Scholar] [CrossRef]
- Torchovskaya, T.I.; Kudinov, V.A.; Zakharova, T.S.; Markin, S.S. Dysfunctional high-density lipoproteins: Role in atherogenesis and potential targets for phospholipid therapy. Kardiologiia 2018, 73–83. [Google Scholar] [CrossRef]
- Chadwick, A.C.; Holme, R.L.; Chen, Y.; Thomas, M.J.; Sorci-Thomas, M.G.; Silverstein, R.L.; Pritchard, K.A.J.; Sahoo, D. Acrolein impairs the cholesterol transport functions of high density lipoproteins. PLoS ONE 2015, 10, e0123138. [Google Scholar] [CrossRef]
- von Eckardstein, A.; Nofer, J.-R.; Assmann, G. High density lipoproteins and arteriosclerosis. Arterioscler. Thromb. Vasc. Biol. 2001, 21, 13–27. [Google Scholar] [CrossRef] [PubMed]
- Glomset, J.A. The plasma lecithin: Cholesterol acyltransferase reaction. J. Lipid Res. 1968, 9, 155–167. [Google Scholar] [PubMed]
- Small, D.M.; Shipley, G.G. Physical-Chemical Basis of Lipid Deposition in Atherosclerosis. Science 1974, 185, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Havel, R.J.; Hunninghake, D.B.; ILlingworth, D.R.; Lees, R.S.; Stein, E.A.; Tobert, J.A.; Bacon, S.R.; Bolognese, J.A.; Frost, P.H.; Lamkin, G.E.; et al. Lovastatin (mevinolin) in the treatment of heterozygous familial hypercholesterolemia. A multicenter study. Ann. Intern. Med. 1987, 107, 609–615. [Google Scholar] [CrossRef] [PubMed]
- Phillips, M.C. Molecular mechanisms of cellular cholesterol efflux. J. Biol. Chem. 2014, 289, 24020–24029. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Gu, H.-M.; Zhang, D.-W. ATP-binding cassette transporters and cholesterol translocation. IUBMB Life 2013, 65, 505–512. [Google Scholar] [CrossRef]
- Trajkovska, K.T. High-density lipoprotein metabolism and reverse cholesterol transport: Strategies for raising HDL cholesterol. Anatol. J. Cardiol. 2017, 18, 149–154. [Google Scholar] [CrossRef]
- Riggs, K.A.; Rohatgi, A. HDL and Reverse cholesterol transport biomarkers. Methodist Debakey Cardiovasc. J. 2019, 15, 39–46. [Google Scholar] [CrossRef]
- Dergunov, A.D.; Garaeva, E.A.; Savushkin, E.V.; Litvinov, D.Y. Significance of lipid-free and lipid-associated ApoA-I in cellular cho-lesterol efflux. Curr. Protein Pept. Sci. 2017, 18, 92–99. [Google Scholar] [CrossRef]
- Torkhovskaya, T.I.; Kudinov, V.A.; Zakharova, T.S.; Ipatova, O.M.; Markin, S.S. High density lipoproteins phosphatidylcholine as a regulator of reverse cholesterol transport. Russ. J. Bioorganic Chem. 2018, 44, 608–618. [Google Scholar] [CrossRef]
- Hafiane, A.; Genest, J. HDL, atherosclerosis, and emerging therapies. Cholesterol 2013, 2013, 891403. [Google Scholar] [CrossRef] [PubMed]
- Catapano, A.L.; Pirillo, A.; Bonacina, F.; Norata, G.D. HDL in innate and adaptive immunity. Cardiovasc. Res. 2014, 103, 372–383. [Google Scholar] [CrossRef] [PubMed]
- Jozefczuk, E.; Guzik, T.J.; Siedlinski, M. Significance of sphingosine-1-phosphate in cardiovascular physiology and pathology. Pharmacol. Res. 2020, 156, 104793. [Google Scholar] [CrossRef] [PubMed]
- Meilhac, O.; Tanaka, S.; Couret, D. High-density lipoproteins are bug scavengers. Biomolecules 2020, 10, 598. [Google Scholar] [CrossRef]
- Thompson, P.A.; Berbée, J.F.P.; Rensen, P.C.N.; Kitchens, R.L. Apolipoprotein A-II augments monocyte responses to LPS by suppressing the inhibitory activity of LPS-binding protein. Innate Immun. 2008, 14, 365–374. [Google Scholar] [CrossRef]
- Kumaraswamy, S.B.; Linder, A.; Åkesson, P.; Dahlbäck, B. Decreased plasma concentrations of apolipoprotein M in sepsis and systemic inflammatory response syndromes. Crit. Care 2012, 16, R60. [Google Scholar] [CrossRef]
- Kurano, M.; Tsuneyama, K.; Morimoto, Y.; Shimizu, T.; Jona, M.; Kassai, H.; Nakao, K.; Aiba, A.; Yatomi, Y. Apolipoprotein M protects lipopolysaccharide-treated mice from death and organ injury. Thromb. Haemost. 2018, 118, 1021–1035. [Google Scholar] [CrossRef]
- Shiflett, A.M.; Bishop, J.R.; Pahwa, A.; Hajduk, S.L. Human high density lipoproteins are platforms for the assembly of multi-component innate immune complexes. J. Biol. Chem. 2005, 280, 32578–32585. [Google Scholar] [CrossRef]
- Bricarello, D.A.; Mills, E.J.; Petrlova, J.; Voss, J.C.; Parikh, A.N. Ganglioside embedded in reconstituted lipoprotein binds cholera toxin with elevated affinity. J. Lipid Res. 2010, 51, 2731–2738. [Google Scholar] [CrossRef]
- Whitehead, A.S.; De Beerd, M.C.; Steel, D.M.; Rits, M.; Leliasp, J.M.; Lane, W.S.; De Beer, C. Identification of novel members of the serum amyloid a protein superfamily as constitutive apolipoproteins of high density lipoprotein. J. Biol. Chem. 1992, 267, 3862–3867. [Google Scholar]
- Wroblewski, J.M.; Jahangiri, A.; Ji, A.; de Beer, F.C.; van der Westhuyzen, D.R.; Webb, N.R. Nascent HDL formation by hepatocytes is reduced by the concerted action of serum amyloid A and endothelial lipase. J. Lipid Res. 2011, 52, 2255–2261. [Google Scholar] [CrossRef] [PubMed]
- Gordon, S.M.; Deng, J.; Lu, L.J.; Davidson, W.S. Proteomic characterization of human plasma high density lipoprotein fractionated by gel filtration chromatography. J. Proteome Res. 2010, 9, 5239–5249. [Google Scholar] [CrossRef] [PubMed]
- Vaisar, T.; Pennathur, S.; Green, P.S.; Gharib, S.A.; Hoofnagle, A.N.; Cheung, M.C.; Byun, J.; Vuletic, S.; Kassim, S.; Singh, P.; et al. Shotgun proteomics implicates protease inhibition and complement activation in the antiinflammatory properties of HDL. J. Clin. Investig. 2007, 117, 746–756. [Google Scholar] [CrossRef] [PubMed]
- Rosenfeld, S.I.; Packman, C.H.; Leddy, J.P. Inhibition of the lytic action of cell-bound terminal complement components by human high density lipoproteins and apoproteins. J. Clin. Investig. 1983, 71, 795–808. [Google Scholar] [CrossRef]
- Hamilton, K.K.; Zhao, J.; Sims, P.J. Interaction between apolipoproteins A-I and A-I1 and the membrane attack complex of complement. J. Biol. Chem. 1993, 268, 3632–3638. [Google Scholar]
- Pasqui, A.L.; Puccetti, L.; Bova, G.; Di Renzo, M.; Bruni, F.; Pastorelli, M.; Palazzuoli, A.; Auteri, A. Relationship between serum complement and different lipid disorders. Clin. Exp. Med. 2002, 2, 33–38. [Google Scholar] [CrossRef]
- Wendel, M.; Paul, R.; Heller, A.R. Lipoproteins in inflammation and sepsis. II. Clinical aspects. Intensive Care Med. 2007, 33, 25–35. [Google Scholar] [CrossRef]
- Birjmohun, R.S.; van Leuven, S.I.; Levels, J.H.M.; van ’t Veer, C.; Kuivenhoven, J.A.; Meijers, J.C.M.; Levi, M.; Kastelein, J.J.P.; van der Poll, T.; Stroes, E.S.G. High-density lipoprotein attenuates inflammation and coagulation response on endotoxin challenge in humans. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 1153–1158. [Google Scholar] [CrossRef]
- Han, J.; Ulevitch, R.J. Limiting inflammatory responses during activation of innate immunity. Nat. Immunol. 2005, 6, 1198–1205. [Google Scholar] [CrossRef]
- Grunfeld, C.; Marshall, M.; Shigenaga, J.K.; Moser, A.H.; Tobias, P.; Feingold, K.R. Lipoproteins inhibit macrophage activation by lipoteichoic acid. J. Lipid Res. 1999, 40, 245–252. [Google Scholar]
- Murch, O.; Collin, M.; Thiemermann, C. Lysophosphatidic acid reduces the organ injury caused by endotoxemia-a role for G-protein-coupled receptors and peroxisome proliferator-activated receptor-gamma. Shock 2007, 27, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Levine, D.M.; Parker, T.S.; Donnelly, T.M.; Walsh, A.; Rubin, A.L. In vivo protection against endotoxin by plasma high density lipoprotein. Proc. Natl. Acad. Sci. USA 1993, 90, 12040–12044. [Google Scholar] [CrossRef] [PubMed]
- Lee, R.-P.; Lin, N.-T.; Chao, Y.-F.C.; Lin, C.-C.; Harn, H.-J.; Chen, H.-I. High-density lipoprotein prevents organ damage in endotoxemia. Res. Nurs. Health 2007, 30, 250–260. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Ai, J.; Zheng, Z.; Howatt, D.A.; Daugherty, A.; Huang, B.; Li, X.-A. High density lipoprotein protects against polymicrobe-induced sepsis in mice. J. Biol. Chem. 2013, 288, 17947–17953. [Google Scholar] [CrossRef] [PubMed]
- Vishnyakova, T.G.; Bocharov, A.V.; Baranova, I.N.; Chen, Z.; Remaley, A.T.; Csako, G.; Eggerman, T.L.; Patterson, A.P. Binding and internalization of lipopolysaccharide by Cla-1, a human orthologue of rodent scavenger receptor B1. J. Biol. Chem. 2003, 278, 22771–22780. [Google Scholar] [CrossRef] [PubMed]
- Duchateau, P.N.; Pullinger, C.R.; Orellana, R.E.; Kunitake, S.T.; Naya-Vigne, J.; O’Connor, P.M.; Malloy, M.J.; Kane, J.P. Apolipoprotein L, a new human high density lipoprotein apolipoprotein expressed by the pancreas. Identification, cloning, characterization, and plasma distribution of apolipoprotein L. J. Biol. Chem. 1997, 272, 25576–25582. [Google Scholar] [CrossRef] [PubMed]
- Simons, K.; Ikonen, E. Functional rafts in cell membranes. Nature 1997, 387, 569–572. [Google Scholar] [CrossRef]
- Brown, D.A.; London, E. Functions of lipid rafts in biological membranes. Annu. Rev. Cell Dev. Biol. 1998, 14, 111–136. [Google Scholar] [CrossRef]
- McIntosh, T.J.; Vidal, A.; Simon, S.A. Sorting of lipids and transmembrane peptides between detergent-soluble bilayers and detergent-resistant rafts. Biophys. J. 2003, 85, 1656–1666. [Google Scholar] [CrossRef]
- Kabouridis, P.S.; Janzen, J.; Magee, A.L.; Ley, S.C. Cholesterol depletion disrupts lipid rafts and modulates the activity of multiple signaling pathways in T lymphocytes. Eur. J. Immunol. 2000, 30, 954–963. [Google Scholar] [CrossRef]
- Anderson, H.A.; Hiltbold, E.M.; Roche, P.A. Concentration of MHC class II molecules in lipid rafts facilitates antigen presentation. Nat. Immunol. 2000, 1, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Yin, K.; Chen, W.-J.; Zhou, Z.-G.; Zhao, G.-J.; Lv, Y.-C.; Ouyang, X.-P.; Yu, X.-H.; Fu, Y.; Jiang, Z.-S.; Tang, C.-K. Apolipoprotein A-I inhibits CD40 proinflammatory signaling via ATP-binding cassette transporter A1-mediated modulation of lipid raft in macrophages. J. Atheroscler. Thromb. 2012, 19, 823–836. [Google Scholar] [CrossRef] [PubMed]
- Murphy, A.J.; Woollard, K.J.; Hoang, A.; Mukhamedova, N.; Stirzaker, R.A.; McCormick, S.P.A.; Remaley, A.T.; Sviridov, D.; Chin-Dusting, J. High-density lipoprotein reduces the human monocyte inflammatory response. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 2071–2077. [Google Scholar] [CrossRef] [PubMed]
- Whitmore, M.M.; Iparraguirre, A.; Kubelka, L.; Weninger, W.; Hai, T.; Williams, B.R.G. Negative regulation of TLR-signaling pathways by activating transcription factor-3. J. Immunol. 2007, 179, 3622–3630. [Google Scholar] [CrossRef]
- De Nardo, D.; Labzin, L.I.; Kono, H.; Seki, R.; Schmidt, S.V.; Beyer, M.; Xu, D.; Zimmer, S.; Lahrmann, C.; Schildberg, F.A.; et al. High-density lipoprotein mediates anti-inflammatory reprogramming of macrophages via the transcriptional regulator ATF3. Nat. Immunol. 2014, 15, 152–160. [Google Scholar] [CrossRef]
- Cheng, A.M.; Handa, P.; Tateya, S.; Schwartz, J.; Tang, C.; Mitra, P.; Oram, J.F.; Chait, A.; Kim, F. Apolipoprotein A-I attenuates palmitate-mediated NF-κB activation by reducing toll-like receptor-4 recruitment into lipid rafts. PLoS ONE 2012, 7, e33917. [Google Scholar] [CrossRef]
- Wang, S.-H.; Yuan, S.-G.; Peng, D.-Q.; Zhao, S.-P. HDL and ApoA-I inhibit antigen presentation-mediated T cell activation by disrupting lipid rafts in antigen presenting cells. Atherosclerosis 2012, 225, 105–114. [Google Scholar] [CrossRef]
- Kim, K.D.; Lim, H.Y.; Lee, H.G.; Yoon, D.-Y.; Choe, Y.-K.; Choi, I.; Paik, S.-G.; Kim, Y.-S.; Yang, Y.; Lim, J.-S. Apolipoprotein A-I induces IL-10 and PGE2 production in human monocytes and inhibits dendritic cell differentiation and maturation. Biochem. Biophys. Res. Commun. 2005, 338, 1126–1136. [Google Scholar] [CrossRef]
- Besler, C.; Luscher, T.F.; Landmesser, U. Molecular mechanisms of vascular effects of high-density lipoprotein: Alterations in cardiovascular disease. EMBO Mol. Med. 2012, 4, 251–268. [Google Scholar] [CrossRef]
- Kontush, A.; Chapman, M.J. Antiatherogenic function of HDL particle subpopulations: Focus on antioxidative activities. Curr. Opin. Lipidol. 2010, 21, 312–318. [Google Scholar] [CrossRef]
- Xepapadaki, E.; Zvintzou, E.; Kalogeropoulou, C.; Filou, S.; Kypreos, K.E. Τhe antioxidant function of HDL in atherosclerosis. Angiology 2020, 71, 112–121. [Google Scholar] [CrossRef] [PubMed]
- Qin, S. LDL and HDL oxidative modification and atherosclerosis. Adv. Exp. Med. Biol. 2020, 1276, 157–169. [Google Scholar] [CrossRef] [PubMed]
- Navab, M.; Hama, S.Y.; Hough, G.P.; Subbanagounder, G.; Reddy, S.T.; Fogelman, A.M. A cell-free assay for detecting HDL that is dysfunctional in preventing the formation of or inactivating oxidized phospholipids. J. Lipid Res. 2001, 42, 1308–1317. [Google Scholar] [PubMed]
- Thacker, S.G.; Zarzour, A.; Chen, Y.; Alcicek, M.S.; Freeman, L.A.; Sviridov, D.O.; Demosky, S.J.; Remaley, A.T. High-density lipoprotein reduces inflammation from cholesterol crystals by inhibiting inflammasome activation. Immunology 2016, 149, 306–319. [Google Scholar] [CrossRef] [PubMed]
- Alwaili, K.; Bailey, D.; Awan, Z.; Bailey, S.D.; Ruel, I.; Hafiane, A.; Krimbou, L.; Laboissiere, S.; Genest, J. The HDL proteome in acute coronary syndromes shifts to an inflammatory profile. Biochim. Biophys. Acta 2012, 1821, 405–415. [Google Scholar] [CrossRef]
- Wilkerson, B.A.; Grass, G.D.; Wing, S.B.; Argraves, W.S.; Argraves, K.M. Sphingosine 1-Phosphate (S1P) carrier-dependent regulation of endothelial barrier: High density lipoprotein (HDL)-S1P prolongs endothelial barrier enhancement as compared with albumin- S1P via effects on levels, trafficking, and signaling of S1P1. J. Biol. Chem. 2012, 287, 44645–44653. [Google Scholar] [CrossRef]
- Argraves, K.M.; Gazzolo, P.J.; Groh, E.M.; Wilkerson, B.A.; Matsuura, B.S.; Twal, W.O.; Hammad, S.M.; Argraves, W.S. High density lipoprotein-associated Sphingosine 1-Phosphate promotes endothelial barrier function. J. Biol. Chem. 2008, 283, 25074–25081. [Google Scholar] [CrossRef]
- Nofer, J.-R.; Brodde, M.F.; Kehrel, B.E. High-density lipoproteins, platelets and the pathogenesis of atherosclerosis. Clin. Exp. Pharmacol. Physiol. 2010, 37, 726–735. [Google Scholar] [CrossRef]
- Besler, C.; Heinrich, K.; Rohrer, L.; Doerries, C.; Riwanto, M.; Shih, D.M.; Chroni, A.; Yonekawa, K.; Stein, S.; Schaefer, N.; et al. Mechanisms underlying adverse effects of HDL on eNOS-activating pathways in patients with coronary artery disease. J. Clin. Investig. 2011, 121, 2693–2708. [Google Scholar] [CrossRef]
- Lee, R.C.; Feinbaum, R.L.; Ambros, V. The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell 1993, 75, 843–854. [Google Scholar] [CrossRef]
- Bartel, D.P.; Chen, C.-Z. Micromanagers of gene expression: The potentially widespread influence of metazoan microRNAs. Nat. Rev. Genet. 2004, 5, 396–400. [Google Scholar] [CrossRef] [PubMed]
- Rajewsky, N. microRNA target predictions in animals. Nat. Genet. 2006, 38 (Suppl. S8–S13). [Google Scholar] [CrossRef] [PubMed]
- Ludwig, N.; Leidinger, P.; Becker, K.; Backes, C.; Fehlmann, T.; Pallasch, C.; Rheinheimer, S.; Meder, B.; Stahler, C.; Meese, E.; et al. Distribution of miRNA expression across human tissues. Nucleic Acids Res. 2016, 44, 3865–3877. [Google Scholar] [CrossRef] [PubMed]
- Sohel, M.H. Extracellular/circulating microRNAs: Release mechanisms, functions and challenges. Achiev. Life Sci. 2016, 10, 175–186. [Google Scholar] [CrossRef]
- Vickers, K.C.; Palmisano, B.T.; Shoucri, B.M.; Shamburek, R.D.; Remaley, A.T. MicroRNAs are transported in plasma and delivered to recipient cells by high-density lipoproteins. Nat. Cell Biol. 2011, 13, 423–435. [Google Scholar] [CrossRef]
- Lee, H.; Kim, S.I.; Shin, D.; Yoon, Y.; Choi, T.H.; Cheon, G.-J.; Kim, M. Hepatic siRNA delivery using recombinant human apolipoprotein A-I in mice. Biochem. Biophys. Res. Commun. 2009, 378, 192–196. [Google Scholar] [CrossRef]
- Tabet, F.; Vickers, K.C.; Cuesta Torres, L.F.; Wiese, C.B.; Shoucri, B.M.; Lambert, G.; Catherinet, C.; Prado-Lourenco, L.; Levin, M.G.; Thacker, S.; et al. HDL-transferred microRNA-223 regulates ICAM-1 expression in endothelial cells. Nat. Commun. 2014, 5, 3292. [Google Scholar] [CrossRef]
- Niculescu, L.S.; Simionescu, N.; Sanda, G.M.; Carnuta, M.G.; Stancu, C.S.; Popescu, A.C.; Popescu, M.R.; Vlad, A.; Dimulescu, D.R.; Simionescu, M.; et al. MiR-486 and miR-92a identified in circulating HDL discriminate between stable and vulnerable coronary artery disease patients. PLoS ONE 2015, 10, e0140958. [Google Scholar] [CrossRef]
- Wagner, J.; Riwanto, M.; Besler, C.; Knau, A.; Fichtlscherer, S.; Roxe, T.; Zeiher, A.M.; Landmesser, U.; Dimmeler, S. Characterization of levels and cellular transfer of circulating lipoprotein-bound microRNAs. Arterioscler. Thromb. Vasc. Biol. 2013, 33, 1392–1400. [Google Scholar] [CrossRef]
- Kosaka, N.; Iguchi, H.; Yoshioka, Y.; Takeshita, F.; Matsuki, Y.; Ochiya, T. Secretory mechanisms and intercellular transfer of microRNAs in living cells. J. Biol. Chem. 2010, 285, 17442–17452. [Google Scholar] [CrossRef]
| Density, g/mL | HDL Separation by Ultracentrifugation | HDL Fractions Separation by Electrophoresis | ||
|---|---|---|---|---|
| HDL Fractions | Diameter, nm | HDL Subfractions | Diameter, nm | |
| 1.063–1.125 | HDL2 | 8.8–12 | HDL2b * | 9.7–12 |
| HDL2a * | 8.8–9.7 | |||
| 1.125–1.21 | HDL3 | 7.2–8.8 | HDL3a * | 8.2–8.8 |
| HDL3b * | 7.8–8.2 | |||
| HDL3c * | 7.2–7.8 | |||
| >1.21 | preβ-HDL | preβ-HDL ** | ||
| Protein | Origin and Biological Function |
|---|---|
| ApoA-I | The main structural and functional apolipoprotein, which interacts with cellular receptors, activates lecithin–cholesterol acyltransferase (LCAT) and exhibits antiatherogenic activity. The main sites for ApoAI synthesis and secretion are the liver and small intestine. |
| ApoA-II | Structural and functional apolipoprotein, predominantly synthesized in the liver. |
| ApoA-IV | Structural and functional apolipoprotein, synthesized in the intestine. |
| ApoC-I | Possesses a high positive charge and, thus, can bind free fatty acids, can modulate the activity of some of the proteins involved in HDL metabolism, can activate LCAT, and can inhibit hepatic lipase and cholesterol ester transport protein (CETP). |
| ApoC-II | Activates lipoprotein lipase (LPL). |
| ApoC-III | LPL and hepatic lipase inhibitor. |
| ApoC-IV | Regulator of triglyceride (TG) metabolism. |
| ApoD | Responsible for the binding and transport of small hydrophobic molecules. Expressed in many tissues, including the liver and the intestines. |
| ApoE | Structural and functional apolipoprotein, a ligand for low-density lipoprotein (LDL) receptors and LDL receptor-associated protein (LRP), and binds to glycosaminoglycans on cells. Synthesized in several tissues and cell types, including the liver, endocrine tissues, central nervous system, and macrophages. |
| ApoF | Inhibitor of cholesterol ester transport protein (CETP). It is synthesized in the liver. |
| ApoH | Binds negatively-charged molecules, primarily cardiolipin, and prevents the activation of the blood coagulation cascade by binding to phospholipids on the surface of damaged cells. Regulates platelet aggregation and is expressed in the liver. |
| ApoJ | Binds hydrophobic molecules and interacts with cell receptors |
| ApoL-I | The main component of the serum trypanolytic factor. It is expressed in the pancreas, lungs, prostate, liver, placenta, and spleen. |
| ApoM | Binds small hydrophobic molecules, primarily sphingosine-1-phosphate (S1P), as well as oxidized phospholipids. It is synthesized in the liver and kidneys. |
| PON1 (paraoxonase 1) | Ca2+- dependent lactonase with antioxidant properties, mainly synthesized in the liver, but also in the kidneys and colon. |
| Protein | Biological Function |
|---|---|
| Enzymes | |
| LCAT (lecithin–cholesterol acyltransferase) | Esterifies cholesterol to cholesterol esters. LCAT is mainly expressed in the liver and, to a lesser extent, in the brain and testes. |
| PAF-AH (platelet-activating factor acetyl hydrolase; lipoprotein-associated phospholipase A2 (LpPLA2)) | Hydrolyzes short-chain oxidized phospholipids. Synthesized in the brain, white adipose tissue, and placenta. Macrophages are the most important source of the circulating enzyme. |
| GSPx-3 (glutathione selenoperoxidase 3) | A component of the system of protection against the oxidative damage of molecules. Catalyzes the redox reaction of peroxides (hydrogen peroxide to water or lipid peroxides to the corresponding alcohols) with glutathione. It is synthesized in the liver, kidneys, heart, lungs, mammary glands, and placenta. |
| Lipid transport proteins | |
| PLTP (phospholipid transfer protein) | Remodels HDL into large and small particles and binds and transports bacterial lipopolysaccharide. It is synthesized in the placenta, pancreas, lungs, kidneys, heart, liver, skeletal muscles, and brain. It is also a positive marker of the acute phase of inflammation. |
| CETP (cholesterol ester transport protein) | Provides heteroexchange of cholesteryl ester (CE) and TG and homoexchange of phospholipid (PL) between HDL and ApoB-containing lipoproteins. It is synthesized in the liver and adipose tissue. |
| Acute-phase proteins | |
| SAA1 (serum amyloid A1) | Major acute-phase reactant. Formed preferably in the liver. |
| SAA4 (serum amyloid A4) | Minor acute-phase reactant. Formed preferably in the liver. |
| Alpha-2-HS glycoprotein | Negative acute-phase reactant, which promotes endocytosis and opsonization. It is synthesized in the liver. |
| Fibrinogen alpha chain | Fibrin precursor, main component of blood clots and platelet aggregation. |
| Complement system proteins | |
| C3 | One of the main activators of the complement system through classical and alternative paths. |
| Proteinase inhibitors | |
| α-1-antitrypsin | Inhibits serine proteases, especially neutrophil elastase. |
| Hrp (haptoglobin-related protein) | Decoy substrate to prevent proteolysis. |
| Other proteins | |
| Transthyretin | Thyroid hormone binding and transport. |
| Serotransferin | Iron binding and transport. |
| Vitamin D-binding protein | Vitamin D binding and transport. |
| α-1B-glycoprotein | Unknown. |
| Hemopexin | Heme binding and transport. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kudinov, V.A.; Alekseeva, O.Y.; Torkhovskaya, T.I.; Baskaev, K.K.; Artyushev, R.I.; Saburina, I.N.; Markin, S.S. High-Density Lipoproteins as Homeostatic Nanoparticles of Blood Plasma. Int. J. Mol. Sci. 2020, 21, 8737. https://doi.org/10.3390/ijms21228737
Kudinov VA, Alekseeva OY, Torkhovskaya TI, Baskaev KK, Artyushev RI, Saburina IN, Markin SS. High-Density Lipoproteins as Homeostatic Nanoparticles of Blood Plasma. International Journal of Molecular Sciences. 2020; 21(22):8737. https://doi.org/10.3390/ijms21228737
Chicago/Turabian StyleKudinov, Vasily A., Olga Yu. Alekseeva, Tatiana I. Torkhovskaya, Konstantin K. Baskaev, Rafael I. Artyushev, Irina N. Saburina, and Sergey S. Markin. 2020. "High-Density Lipoproteins as Homeostatic Nanoparticles of Blood Plasma" International Journal of Molecular Sciences 21, no. 22: 8737. https://doi.org/10.3390/ijms21228737
APA StyleKudinov, V. A., Alekseeva, O. Y., Torkhovskaya, T. I., Baskaev, K. K., Artyushev, R. I., Saburina, I. N., & Markin, S. S. (2020). High-Density Lipoproteins as Homeostatic Nanoparticles of Blood Plasma. International Journal of Molecular Sciences, 21(22), 8737. https://doi.org/10.3390/ijms21228737






