Neurogranin and VILIP-1 as Molecular Indicators of Neurodegeneration in Alzheimer’s Disease: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Results
2.1. Dataset Characteristics and Groups
2.2. Ng and VILIP-1 Measurement
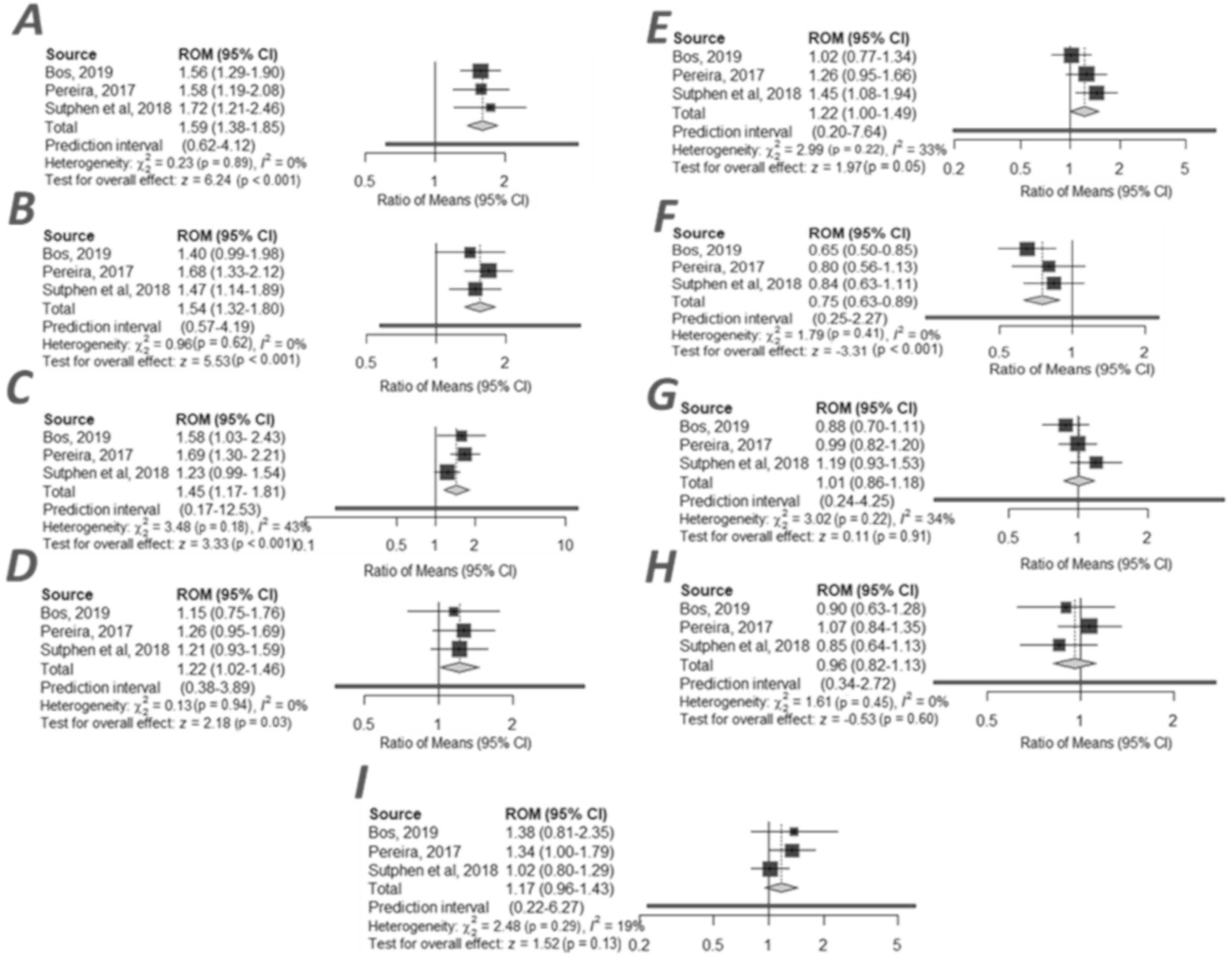
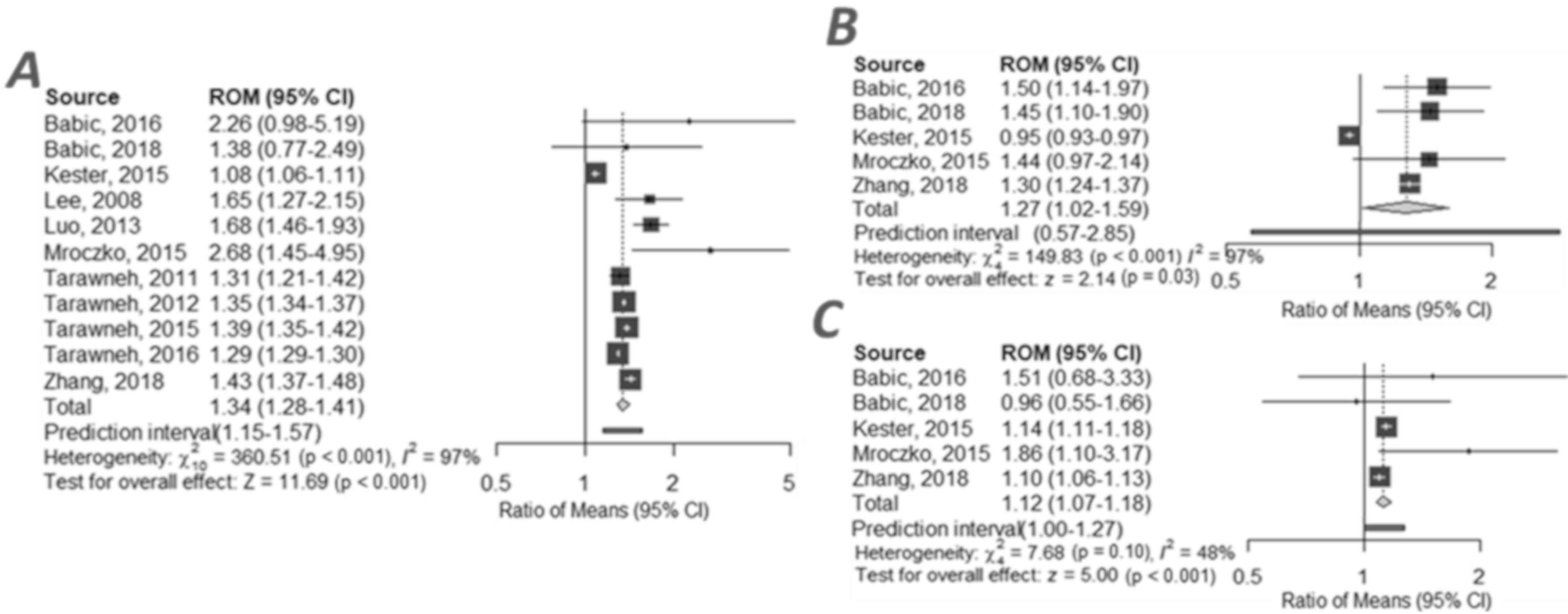
2.3. CSF Neurogranin in AD and MCI Groups
2.4. CSF Ng Levels Dependent on Aβ Status
2.5. CSF VILIP-1 in AD and MCI Group
3. Discussion
Limitation of the Study
4. Materials and Methods
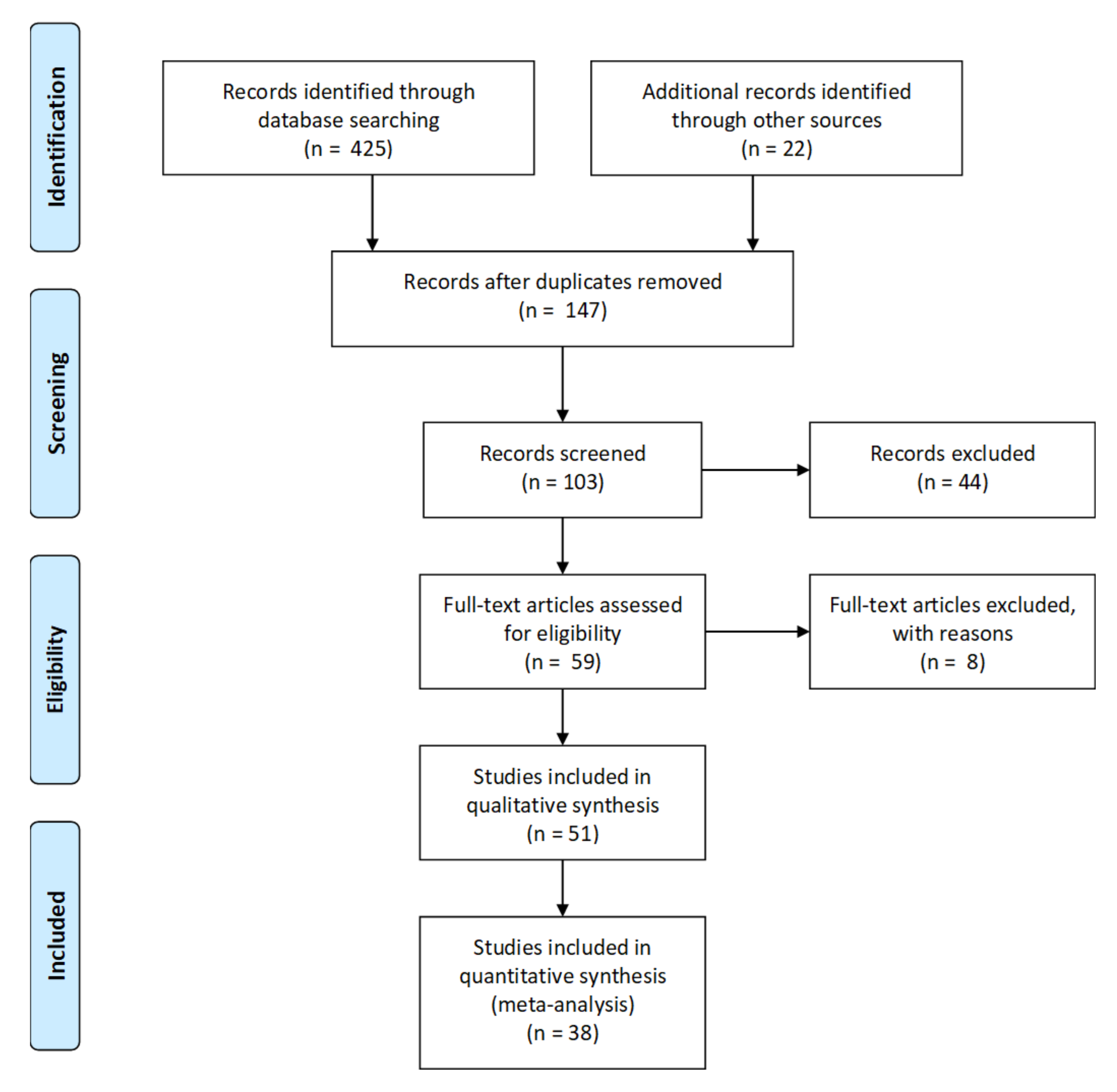
4.1. Search Strategy
4.2. Inclusion Criteria
4.3. Data Collection
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Blennow, K.; Zetterberg, H. Biomarkers for Alzheimer’s Disease: Current Status and Prospects for the Future; John Wiley & Sons, Ltd: Hoboken, NJ, USA, 2018; Volume 284, pp. 643–663. [Google Scholar]
- Hampel, H.; Toschi, N.; Baldacci, F.; Zetterberg, H.; Blennow, K.; Kilimann, I.; Teipel, S.J.; Cavedo, E.; Melo Dos Santos, A.; Epelbaum, S.; et al. Alzheimer’s disease biomarker-guided diagnostic workflow using the added value of six combined cerebrospinal fluid candidates: Aβ 1–42, total-tau, phosphorylated-tau, NFL, neurogranin, and YKL-40. Alzheimer’s Dement. 2018, 14, 492–501. [Google Scholar] [CrossRef] [PubMed]
- Höglund, K.; Kern, S.; Zettergren, A.; Börjesson-Hansson, A.; Zetterberg, H.; Skoog, I.; Blennow, K. Preclinical amyloid pathology biomarker positivity: Effects on tau pathology and neurodegeneration. Transl. Psychiatry 2017, 7, e995. [Google Scholar] [CrossRef] [PubMed]
- Headley, A.; De Leon-Benedetti, A.; Dong, C.; Levin, B.; Loewenstein, D.; Camargo, C.; Rundek, T.; Zetterberg, H.; Blennow, K.; Wright, C.B.; et al. Neurogranin as a predictor of memory and executive function decline in MCI patients. Neurology 2018, 90, e887–e895. [Google Scholar] [CrossRef] [PubMed]
- Kester, M.I.; Teunissen, C.E.; Crimmins, D.L.; Herries, E.M.; Ladenson, J.H.K.H.; Scheltens, P.; van der Flier, W.M.; Morris, J.C.; Holtzman, D.M.; Fagan, A.M. Neurogranin as a cerebrospinal fluid biomarker for synaptic loss in symptomatic Alzheimer disease. JAMA Neurol. 2015, 72, 1275–1280. [Google Scholar] [CrossRef]
- Petersen, A.; Gerges, N.Z. Neurogranin regulates CaM dynamics at dendritic spines. Sci. Rep. 2015, 5, 11135. [Google Scholar] [CrossRef]
- De Wilde, M.C.; Overk, C.R.; Sijben, J.W.; Masliah, E. Meta-analysis of synaptic pathology in Alzheimer’s disease reveals selective molecular vesicular machinery vulnerability. Alzheimer’s Dement. 2016, 12, 633–644. [Google Scholar] [CrossRef] [PubMed]
- Antonell, A.; Tort-Merino, A.; Ríos, J.; Balasa, M.; Borrego-Écija, S.; Auge, J.M.; Muñoz-García, C.; Bosch, B.; Falgàs, N.; Rami, L.; et al. Synaptic, axonal damage and inflammatory cerebrospinal fluid biomarkers in neurodegenerative dementias. Alzheimer’s Dement. 2020, 16, 262–272. [Google Scholar] [CrossRef]
- Janelidze, S.; Hertze, J.; Zetterberg, H.; Landqvist Waldö, M.; Santillo, A.; Blennow, K.; Hansson, O. Cerebrospinal fluid neurogranin and YKL-40 as biomarkers of Alzheimer’s disease. Ann. Clin. Transl. Neurol. 2016, 3, 12–20. [Google Scholar] [CrossRef]
- Kvartsberg, H.; Duits, F.H.; Ingelsson, M.; Andreasen, N.; Öhrfelt, A.; Andersson, K.; Brinkmalm, G.; Lannfelt, L.; Minthon, L.; Hansson, O.; et al. Cerebrospinal fluid levels of the synaptic protein neurogranin correlates with cognitive decline in prodromal Alzheimer’s disease. Alzheimer’s Dement. 2015, 11, 1180–1190. [Google Scholar] [CrossRef]
- Lista, S.; Toschi, N.; Baldacci, F.; Zetterberg, H.; Blennow, K.; Kilimann, I.; Teipel, S.J.; Cavedo, E.; Dos Santos, A.M.; Epelbaum, S.; et al. Cerebrospinal Fluid Neurogranin as a Biomarker of Neurodegenerative Diseases: A Cross-Sectional Study. J. Alzheimer’s Dis. 2017, 59, 1327–1334. [Google Scholar] [CrossRef]
- Merluzzi, A.P.; Carlsson, C.M.; Johnson, S.C.; Schindler, S.E.; Asthana, S.; Blennow, K.; Zetterberg, H.; Bendlin, B.B. Neurodegeneration, synaptic dysfunction, and gliosis are phenotypic of Alzheimer dementia. Neurology 2018, 91, E436–E443. [Google Scholar] [CrossRef] [PubMed]
- Pereira, J.B.; Westman, E.; Hansson, O. Association between cerebrospinal fluid and plasma neurodegeneration biomarkers with brain atrophy in Alzheimer’s disease. Neurobiol. Aging 2017, 58, 14–29. [Google Scholar] [CrossRef]
- Portelius, E.; Zetterberg, H.; Skillbäck, T.; Törnqvist, U.; Andreasson, U.; Trojanowski, J.Q.; Weiner, M.W.; Shaw, L.M.; Mattsson, N.; Blennow, K.; et al. Cerebrospinal fluid neurogranin: Relation to cognition and neurodegeneration in Alzheimer’s disease. Brain 2015, 138, 3373–3385. [Google Scholar] [CrossRef]
- Portelius, E.; Olsson, B.; Höglund, K.; Cullen, N.C.; Kvartsberg, H.; Andreasson, U.; Zetterberg, H.; Sandelius, Å.; Shaw, L.M.; Lee, V.M.Y.; et al. Cerebrospinal fluid neurogranin concentration in neurodegeneration: Relation to clinical phenotypes and neuropathology. Acta Neuropathol. 2018, 136, 363–376. [Google Scholar] [CrossRef] [PubMed]
- Sanfilippo, C.; Forlenza, O.; Zetterberg, H.; Blennow, K. Increased neurogranin concentrations in cerebrospinal fluid of Alzheimer’s disease and in mild cognitive impairment due to AD. J. Neural Transm. 2016, 123, 1443–1447. [Google Scholar] [CrossRef]
- Sun, X.; Dong, C.; Levin, B.; Crocco, E.; Loewenstein, D.; Zetterberg, H.; Blennow, K.; Wright, C.B. APOE ε4 carriers may undergo synaptic damage conferring risk of Alzheimer’s disease. Alzheimer’s Dement. 2016, 12, 1159–1166. [Google Scholar] [CrossRef] [PubMed]
- Sutphen, C.L.; McCue, L.; Herries, E.M.; Xiong, C.; Ladenson, J.H.; Holtzman, D.M.; Fagan, A.M. ADNI Longitudinal decreases in multiple cerebrospinal fluid biomarkers of neuronal injury in symptomatic late onset Alzheimer’s disease. Alzheimer’s Dement. 2018, 14, 869–879. [Google Scholar] [CrossRef]
- Blennow, K.; Diaz-Lucena, D.; Zetterberg, H.; Villar-Pique, A.; Karch, A.; Vidal, E.; Hermann, P.; Schmitz, M.; Ferrer Abizanda, I.; Zerr, I.; et al. CSF neurogranin as a neuronal damage marker in CJD: A comparative study with AD. J. Neurol. Neurosurg. Psychiatry 2019, 90, 846–853. [Google Scholar] [CrossRef]
- Tarawneh, R.; D’Angelo, G.; Crimmins, D.; Herries, E.; Griest, T.; Fagan, A.M.; Zipfel, G.J.; Ladenson, J.H.; Morris, J.C.; Holtzman, D.M. Diagnostic and prognostic utility of the synaptic marker neurogranin in alzheimer disease. JAMA Neurol. 2016, 73, 561–571. [Google Scholar] [CrossRef]
- Vogt, N.M.; Romano, K.A.; Darst, B.F.; Engelman, C.D.; Johnson, S.C.; Carlsson, C.M.; Asthana, S.; Blennow, K.; Zetterberg, H.; Bendlin, B.B.; et al. The gut microbiota-derived metabolite trimethylamine N-oxide is elevated in Alzheimer’s disease. Alzheimers. Res. Ther. 2018, 10, 124. [Google Scholar] [CrossRef]
- Nichols, E.; Szoeke, C.E.I.; Vollset, S.E.; Abbasi, N.; Abd-Allah, F.; Abdela, J.; Aichour, M.T.E.; Akinyemi, R.O.; Alahdab, F.; Asgedom, S.W.; et al. Global, regional, and national burden of Alzheimer’s disease and other dementias, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2018, 18, 88–106. [Google Scholar] [CrossRef]
- Wellington, H.; Paterson, R.W.; Portelius, E.; Törnqvist, U.; Magdalinou, N.; Fox, N.C.; Blennow, K.; Schott, J.M.; Zetterberg, H. Increased CSF neurogranin concentration is specific to Alzheimer disease. Neurology 2016, 86, 829–835. [Google Scholar] [CrossRef]
- Ye, X.; Zhou, W.; Zhang, J.; Alzheimer’s Disease Neuroimaging Initiative. Association of CSF CD40 levels and synaptic degeneration across the Alzheimer’s disease spectrum. Neurosci. Lett. 2019, 694, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Bos, I.; Vos, S.; Verhey, F.; Scheltens, P.; Teunissen, C.; Engelborghs, S.; Sleegers, K.; Frisoni, G.; Blin, O.; Richardson, J.C.; et al. Cerebrospinal fluid biomarkers of neurodegeneration, synaptic integrity, and astroglial activation across the clinical Alzheimer’s disease spectrum. Alzheimer’s Dement. 2019, 15, 644–654. [Google Scholar] [CrossRef]
- Chatterjee, M.; Del Campo, M.; Morrema, T.H.J.J.; de Waal, M.; van der Flier, W.M.; Hoozemans, J.J.M.M.; Teunissen, C.E. Contactin-2, a synaptic and axonal protein, is reduced in cerebrospinal fluid and brain tissue in Alzheimer’s disease. Alzheimer’s Res. Ther. 2018, 10, 52. [Google Scholar] [CrossRef] [PubMed]
- De Vos, A.; Jacobs, D.; Struyfs, H.; Fransen, E.; Andersson, K.; Portelius, E.; Andreasson, U.; De Surgeloose, D.; Hernalsteen, D.; Sleegers, K.; et al. C-terminal neurogranin is increased in cerebrospinal fluid but unchanged in plasma in Alzheimer’s disease. Alzheimer’s Dement. 2015, 11, 1461–1469. [Google Scholar] [CrossRef]
- De Vos, A.; Struyfs, H.; Jacobs, D.; Fransen, E.; Klewansky, T.; De Roeck, E.; Robberecht, C.; Van Broeckhoven, C.; Duyckaerts, C.; Engelborghs, S.; et al. The cerebrospinal fluid neurogranin/BACE1 ratio is a potential correlate of cognitive decline in Alzheimer’s Disease. J. Alzheimer’s Dis. 2016, 53, 1523–1538. [Google Scholar] [CrossRef]
- Falgàs, N.; Ruiz-Peris, M.; Pérez-Millan, A.; Sala-Llonch, R.; Antonell, A.; Balasa, M.; Borrego-Écija, S.; Ramos-Campoy, O.; Augé, J.M.; Castellví, M.; et al. Contribution of CSF biomarkers to early-onset Alzheimer’s disease and frontotemporal dementia neuroimaging signatures. Hum. Brain Mapp. 2020, 41, 2004–2013. [Google Scholar] [CrossRef] [PubMed]
- Galasko, D.; Xiao, M.; Xu, D.; Smirnov, D.; Salmon, D.P.; Dewit, N.; Vanbrabant, J.; Jacobs, D.; Vanderstichele, H.; Vanmechelen, E.; et al. Synaptic biomarkers in CSF aid in diagnosis, correlate with cognition and predict progression in MCI and Alzheimer’s disease. Alzheimer’s Dement. 2019, 5, 871–882. [Google Scholar] [CrossRef]
- Hellwig, K.; Kvartsberg, H.; Portelius, E.; Andreasson, U.; Oberstein, T.J.; Lewczuk, P.; Blennow, K.; Kornhuber, J.; Maler, J.M.; Zetterberg, H.; et al. Neurogranin and YKL-40: Independent markers of synaptic degeneration and neuroinflammation in Alzheimer’s disease. Alzheimer’s Res. Ther. 2015, 7, 74. [Google Scholar] [CrossRef]
- Kvartsberg, H.; Lashley, T.; Murray, C.E.; Brinkmalm, G.; Cullen, N.C.; Höglund, K.; Zetterberg, H.; Blennow, K.; Portelius, E. The intact postsynaptic protein neurogranin is reduced in brain tissue from patients with familial and sporadic Alzheimer’s disease. Acta Neuropathol. 2018, 137, 89–102. [Google Scholar] [CrossRef]
- Kester, M.I.; Teunissen, C.E.; Sutphen, C.; Herries, E.M.; Ladenson, J.H.; Xiong, C.; Scheltens, P.; van der Flier, W.M.; Morris, J.C.; Holtzman, D.M.; et al. Cerebrospinal fluid VILIP-1 and YKL-40, candidate biomarkers to diagnose, predict and monitor Alzheimer’s disease in a memory clinic cohort. Alzheimers Res. Ther. 2015, 7, 59. [Google Scholar] [CrossRef]
- Lee, J.-M.; Blennow, K.; Andreasen, N.; Laterza, O.; Modur, V.; Olander, J.; Gao, F.; Ohlendorf, M.; Ladenson, J.H. The brain injury biomarker VLP-1 is increased in the cerebrospinal fluid of Alzheimer disease patients. Clin. Chem. 2008, 54, 1617–1623. [Google Scholar] [CrossRef]
- Babić Leko, M.; Willumsen, N.; Nikolac Perković, M.; Klepac, N.; Borovečki, F.; Hof, P.R.; Sonicki, Z.; Pivac, N.; de Silva, R.; Šimić, G. Association of MAPT haplotype-tagging polymorphisms with cerebrospinal fluid biomarkers of Alzheimer’s disease: A preliminary study in a Croatian cohort. Brain Behav. 2018, 8, e01128. [Google Scholar] [CrossRef]
- Tarawneh, R.; D’Angelo, G.; Macy, E.; Xiong, C.; Carter, D.; Cairns, N.J.; Fagan, A.M.; Head, D.; Mintun, M.A.; Ladenson, J.H.; et al. Visinin-like protein-1: Diagnostic and prognostic biomarker in Alzheimer disease. Ann. Neurol. 2011, 70, 274–285. [Google Scholar] [CrossRef]
- Tarawneh, R.; Lee, J.-M.; Ladenson, J.H.; Morris, J.C.; Holtzman, D.M. CSF VILIP-1 predicts rates of cognitive decline in early Alzheimer disease. Neurology 2012, 78, 709–719. [Google Scholar] [CrossRef]
- Luo, X.; Hou, L.; Shi, H.; Zhong, X.; Zhang, Y.; Zheng, D.; Tan, Y.; Hu, G.; Mu, N.; Chan, J.; et al. CSF levels of the neuronal injury biomarker visinin-like protein-1 in Alzheimer’s disease and dementia with Lewy bodies. J. Neurochem. 2013, 127, 681–690. [Google Scholar] [CrossRef]
- Tarawneh, R.; Head, D.; Allison, S.; Buckles, V.; Fagan, A.M.; Ladenson, J.H.; Morris, J.C.; Holtzman, D.M. Cerebrospinal Fluid markers of neurodegeneration and rates of brain atrophy in early Alzheimer Disease. JAMA Neurol. 2015, 72, 656–665. [Google Scholar] [CrossRef] [PubMed]
- Mroczko, B.; Groblewska, M.; Zboch, M.; Muszyński, P.; Zajkowska, A.; Borawska, R.; Szmitkowski, M.; Kornhuber, J.; Lewczuk, P. Evaluation of visinin-like protein 1 concentrations in the cerebrospinal fluid of patients with mild cognitive impairment as a dynamic biomarker of Alzheimer’s disease. J. Alzheimers. Dis. 2015, 43, 1031–1037. [Google Scholar] [CrossRef]
- Babić Leko, M.; Borovečki, F.; Dejanović, N.; Hof, P.R.; Šimić, G. Predictive value of cerebrospinal fluid visinin-like protein-1 levels for Alzheimer’s Disease early detection and differential diagnosis in patients with mild cognitive impairment. J. Alzheimer’s Dis. 2016, 50, 765–778. [Google Scholar] [CrossRef]
- Zhang, H.; Ng, K.P.; Therriault, J.; Kang, M.S.; Pascoal, T.A.; Rosa-Neto, P.; Gauthier, S. Cerebrospinal fluid phosphorylated tau, visinin-like protein-1, and chitinase-3-like protein 1 in mild cognitive impairment and Alzheimer’s disease. Transl. Neurodegener. 2018, 7, 23. [Google Scholar] [CrossRef]
- Kvartsberg, H.; Portelius, E.; Andreasson, U.; Brinkmalm, G.; Hellwig, K.; Lelental, N.; Kornhuber, J.; Hansson, O.; Minthon, L.; Spitzer, P.; et al. Characterization of the postsynaptic protein neurogranin in paired cerebrospinal fluid and plasma samples from Alzheimer’s disease patients and healthy controls. Alzheimer’s Res. Ther. 2015, 7, 1–9. [Google Scholar] [CrossRef]
- McKhann, G.M.; Knopman, D.S.; Chertkow, H.; Hyman, B.T.; Jack, C.R.; Kawas, C.H.; Klunk, W.E.; Koroshetz, W.J.; Manly, J.J.; Mayeux, R.; et al. The diagnosis of dementia due to Alzheimer’s disease: Recommendations from the national institute on aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimer’s Dement. 2011, 7, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Albert, M.S.; DeKosky, S.T.; Dickson, D.; Dubois, B.; Feldman, H.H.; Fox, N.C.; Gamst, A.; Holtzman, D.M.; Jagust, W.J.; Petersen, R.C.; et al. The diagnosis of mild cognitive impairment due to Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimer’s Dement. 2011, 7, 270–279. [Google Scholar] [CrossRef]
- McKhann, G.; Drachman, D.; Folstein, M.; Katzman, R.; Price, D.; Stadlan, E.M. Clinical diagnosis of Alzheimer’s disease: Report of the NINCDS-ADRDA Work Group* under the auspices of Department of Health and Human Services Task Force on Alzheimer’s Disease. Neurology 1984, 34, 939. [Google Scholar] [CrossRef]
- Petersen, R.C. Mild cognitive impairment as a diagnostic entity. J. Intern. Med. 2004, 256, 183–194. [Google Scholar] [CrossRef]
- Petersen, R.C.; Smith, G.E.; Waring, S.C.; Ivnik, R.J.; Tangalos, E.G.; Kokmen, E. Mild cognitive impairment: Clinical characterization and outcome. Arch. Neurol. 1999, 56, 303–308. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, X.; Zhu, B.; Fu, P. Association of clusterin levels in cerebrospinal fluid with synaptic degeneration across the Alzheimer’s Disease continuum. Neuropsychiatr. Dis. Treat. 2020, 16, 183–190. [Google Scholar] [CrossRef]
- Wang, L. Association of cerebrospinal fluid Neurogranin with Alzheimer’s disease. Aging Clin. Exp. Res. 2019, 31, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Dubois, B.; Feldman, H.H.; Jacova, C.; Hampel, H.; Molinuevo, J.L.; Blennow, K.; DeKosky, S.T.; Gauthier, S.; Selkoe, D.; Bateman, R.; et al. Advancing research diagnostic criteria for Alzheimer’s disease: The IWG-2 criteria. Lancet Neurol. 2014, 13, 614–629. [Google Scholar] [CrossRef]
- Dubois, B.; Feldman, H.H.; Jacova, C.; DeKosky, S.T.; Barberger-Gateau, P.; Cummings, J.; Delacourte, A.; Galasko, D.; Gauthier, S.; Jicha, G.; et al. Research criteria for the diagnosis of Alzheimer’s disease: Revising the NINCDS–ADRDA criteria. Lancet Neurol. 2007, 6, 734–746. [Google Scholar] [CrossRef]
- Morris, M.C.; Evans, D.A.; Tangney, C.C.; Bienias, J.L.; Wilson, R.S. Associations of vegetable and fruit consumption with age-related cognitive change. Neurology 2006, 67, 1370–1376. [Google Scholar] [CrossRef] [PubMed]
- Berg, L.; McKeel, D.W.; Miller, J.P.; Storandt, M.; Rubin, E.H.; Morris, J.C.; Baty, J.; Coats, M.; Norton, J.; Goate, A.M.; et al. Clinicopathologic Studies in Cognitively Healthy Aging and Alzheimer Disease. Arch. Neurol. 1998, 55, 326. [Google Scholar] [CrossRef]
- Zhong, L.; Gerges, N.Z. Neurogranin targets calmodulin and lowers the threshold for the induction of long-term potentiation. PLoS ONE 2012, 7, e41275. [Google Scholar] [CrossRef]
- Khachaturian, Z.S. Calcium Hypothesis of Alzheimer’s disease and brain aging: A framework for integrating new evidence into a comprehensive theory of pathogenesis. Alzheimer’s Dement. 2017, 13, 178–182. [Google Scholar]
- Lleó, A.; Núñez-Llaves, R.; Alcolea, D.; Chiva, C.; Balateu-Paños, D.; Colom-Cadena, M.; Gomez-Giro, G.; Muñoz, L.; Querol-Vilaseca, M.; Pegueroles, J.; et al. Changes in synaptic proteins precede neurodegeneration markers in preclinical Alzheimer’s Disease cerebrospinal fluid. Mol. Cell. Proteom. 2019, 18, 546–560. [Google Scholar] [CrossRef]
- Vagnoni, A.; Perkinton, M.S.; Gray, E.H.; Francis, P.T.; Noble, W.; Miller, C.C.J. Calsyntenin-1 mediates axonal transport of the amyloid precursor protein and regulates A production. Hum. Mol. Genet. 2012, 21, 2845–2854. [Google Scholar] [CrossRef]
- Brinkmalm, G.; Sjödin, S.; Simonsen, A.H.; Hasselbalch, S.G.; Zetterberg, H.; Brinkmalm, A.; Blennow, K. A Parallel reaction monitoring mass spectrometric method for analysis of potential CSF biomarkers for Alzheimer’s Disease. Proteom. Clin. Appl. 2018, 12, 1875–1881. [Google Scholar] [CrossRef]
- Oeckl, P.; Metzger, F.; Nagl, M.; von Arnim, C.A.F.; Halbgebauer, S.; Steinacker, P.; Ludolph, A.C.; Otto, M. Alpha-, Beta-, and Gamma-synuclein quantification in cerebrospinal fluid by multiple reaction monitoring reveals increased concentrations in Alzheimer′s and Creutzfeldt-Jakob Disease but no alteration in synucleinopathies. Mol. Cell. Proteom. 2016, 15, 3126–3138. [Google Scholar] [CrossRef]
- Spellman, D.S.; Wildsmith, K.R.; Honigberg, L.A.; Tuefferd, M.; Baker, D.; Raghavan, N.; Nairn, A.C.; Croteau, P.; Schirm, M.; Allard, R.; et al. Development and evaluation of a multiplexed mass spectrometry based assay for measuring candidate peptide biomarkers in Alzheimer’s Disease Neuroimaging Initiative (ADNI) CSF. Proteom. Clin. Appl. 2015, 9, 715–731. [Google Scholar] [CrossRef] [PubMed]
- Begcevic, I.; Tsolaki, M.; Brinc, D.; Brown, M.; Martinez-Morillo, E.; Lazarou, I.; Kozori, M.; Tagaraki, F.; Nenopoulou, S.; Gkioka, M.; et al. Neuronal pentraxin receptor-1 is a new cerebrospinal fluid biomarker of Alzheimer’s disease progression. F1000Research 2018, 7, 1012. [Google Scholar] [CrossRef]
- Clarke, M.T.M.; Brinkmalm, A.; Foiani, M.S.; Woollacott, I.O.C.; Heller, C.; Heslegrave, A.; Keshavan, A.; Fox, N.C.; Schott, J.M.; Warren, J.D.; et al. CSF synaptic protein concentrations are raised in those with atypical Alzheimer’s disease but not frontotemporal dementia. Alzheimer’s Res. Ther. 2019, 11, 105. [Google Scholar] [CrossRef]
- Mecca, A.P.; Chen, M.; O’Dell, R.S.; Naganawa, M.; Toyonaga, T.; Godek, T.A.; Harris, J.E.; Bartlett, H.H.; Zhao, W.; Nabulsi, N.B.; et al. In vivo measurement of widespread synaptic loss in Alzheimer’s disease with SV2A PET. Alzheimer’s Dement. 2020, 16, 974–982. [Google Scholar] [CrossRef] [PubMed]
- Davidsson, P.; Puchades, M.; Blennow, K. Identification of synaptic vesicle, pre- and postsynaptic proteins in human cerebrospinal fluid using liquid-phase isoelectric focusing. Electrophoresis 1999, 20, 431–437. [Google Scholar] [CrossRef]
- Goetzl, E.J.; Kapogiannis, D.; Schwartz, J.B.; Lobach, I.V.; Goetzl, L.; Abner, E.L.; Jicha, G.A.; Karydas, A.M.; Boxer, A.; Miller, B.L. Decreased synaptic proteins in neuronal exosomes of frontotemporal dementia and Alzheimer’s disease. FASEB J. 2016, 30, 4141–4148. [Google Scholar] [CrossRef]
- He, M.F.; Sun, L.; Cao, W.; Yin, C.; Sun, W.; Liu, P.; Tan, L.; Xu, Z.; Zhao, W. Association between plasma exosome neurogranin and brain structure in patients with Alzheimer’s disease: A protocol study. BMJ Open 2020, 10, e036990. [Google Scholar] [CrossRef]
- Braunewell, K.H. The visinin-like proteins VILIP-1 and VILIP-3 in Alzheimer’s disease—Old wine in new bottles. Front. Mol. Neurosci. 2012, 5, 1–12. [Google Scholar] [CrossRef]
- Willemse, E.A.J.J.; De Vos, A.; Herries, E.M.; Andreasson, U.; Engelborghs, S.; van der Flier, W.M.; Scheltens, P.; Crimmins, D.; Ladenson, J.H.; Vanmechelen, E.; et al. Neurogranin as cerebrospinal fluid biomarker for Alzheimer disease: An assay comparison study. Clin. Chem. 2018, 64, 927–937. [Google Scholar] [CrossRef]
- Li, L.; Lai, M.; Cole, S.; Le Novère, N.; Edelstein, S.J. Neurogranin stimulates Ca2+/calmodulin-dependent kinase II by suppressing calcineurin activity at specific calcium spike frequencies. PLoS Comput. Biol. 2020, 16, 1–29. [Google Scholar] [CrossRef]
- Zhong, L.; Gerges, N.Z. Neurogranin Regulates Metaplasticity. Front. Mol. Neurosci. 2020, 12, 1–9. [Google Scholar] [CrossRef]
- Wan, X.; Wang, W.; Liu, J.; Tong, T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med. Res. Methodol. 2014, 14, 135. [Google Scholar] [CrossRef]
- DerSimonian, R.; Laird, N. Meta-analysis in clinical trials. Control. Clin. Trials 1986, 7, 177–188. [Google Scholar] [CrossRef]
- Friedrich, J.O.; Adhikari, N.K.J.; Beyene, J. The ratio of means method as an alternative to mean differences for analyzing continuous outcome variables in meta-analysis: A simulation study. BMC Med. Res. Methodol. 2008, 8, 32. [Google Scholar] [CrossRef] [PubMed]

| Neurogranin (Ng) | |||||||
|---|---|---|---|---|---|---|---|
| N. | Source | Diagnostic Categories | Controls (CTRL) | Diagnostic Criteria | Method | Type of Capture Antibody | PMID |
| 1 | Antonell et al., 2019 [8] | AD (n = 102); MCI-AD (n = 56) | (n = 47) | McKhann et al., 2011 [44]; Albert et al., 2011 [45] | ELISA In-house | Ng7 (G52–G65) | 31668967 |
| 2 | Blennow et al., 2019 [19] | AD (n = 46) | (n = 64) | McKhann et al., 1984 [46] | ECL In-house (MSD) | Ng7 (G52–65) | 31097472 |
| 3 | Bos et al., 2019 [25] | AD+ (n = 157); MCI+ (n = 263); MCI- (n = 187) | Aβ+ (n = 45); Aβ- (n = 95) | McKhann et al., 1984 [46]; Petersen, 2004 [47] | ECL In-house (MSD) | Ng7 (G52–G65) | 30853464 |
| 4A | Chatterjeet et al., 2018 [26] | AD (n = 70) | (n = 20) | McKhann et al., 2011 [44] | ELISA kit Euroimmun | Ng (G62-P75) | 29859129 |
| 4B | Chatterjeet et al., 2018 [26] | AD (n = 36) | (n = 28) | McKhann et al., 2011 [44] | ELISA kit Euroimmun | Ng (G62-P75) | 29859129 |
| 5 | De Vos et al., 2015 [27] | AD (n = 20) | (n = 29) | McKhann et al., 2011 [44] | ELISA In-house | Ng7 (G53–64) | 26092348 |
| 6 | De Vos et al., 2016 [28] | AD (n = 50); MCI (n = 38) | (n = 20) | McKhann et al., 2011 [44] | ELISA In-house | Ng (G62-P75) | 27392859 |
| 7 | Falgàs et al., 2020 [29] | AD (n = 23); MCI-AD (n = 26) | (n = 37) | McKhann et al., 2011 [44]; Albert et al., 2011 [45] | ELISA In-house | Ng7 (G52–G65) | 31944489 |
| 8 | Galasko et al., 2019 [30] | AD (n = 46); MCI (n = 57) | (n = 90) | McKhann et al., 2011 [44]; Albert et al., 2011 [45] | ELISA kit Euroimmun | Ng (G62-P75) | 31853477 |
| 9 | Headley et al., 2018 [4] | MCI (n = 193) | (n = 111) | McKhann et al., 1984 [46] | ECL In-house (MSD) | Ng7 (G53–G64) | 29429972 |
| 10 | Hellwig et al., 2015 [31] | AD (n = 39); MCI-AD (n = 13) | (n = 21) | McKhann et al., 2011 [44] | ECL In-house (MSD) | Ng7 (G52–G65) | 26698298 |
| 11 | Janelidze et al., 2016 [9] | AD (n = 74); MCI-AD (n = 35); sMCI (n = 62) | (n = 53) | McKhann et al., 1984 [46]; Petersen, 2004 [47] | ELISA In-house | Ng7 (G52–G65) | 26783546 |
| 12 | Kester et al., 2015 [5] | AD (n = 65); MCI-AD (n = 36); sMCI (n = 17) | (n = 37) | McKhann et al., 1984 [46]; Petersen et al., 1999 [48] | Erenna® Singulex | Ng G49-G60(P-4793) | 26366630 |
| 13A | Kvartsberg et al., 2015 [10] | AD (n = 16) | (n = 10) | McKhann et al., 1984 [46] | ELISA In-house | Ng7 (G52–G65) | 25533203 |
| 13B | Kvartsberg et al., 2015 [10] | AD (n = 44) | (n = 30) | McKhann et al., 1984 [46] | ELISA In-house | Ng7 (G52–G65) | 25533203 |
| 13C | Kvartsberg et al., 2015 [10] | AD (n = 40); MCI (n = 40) | (n = 40) | McKhann et al., 1984 [46] | ELISA In-house | Ng7 (G52–G65) | 25533203 |
| 13D | Kvartsberg et al., 2015 [10] | sMCI (n = 23); MCI-AD (n = 14) | (n = 0) | McKhann et al., 1984 [46] Petersen et al., 1999 [48] Petersen, 2004 [47] | ELISA In-house | Ng7 (G52–G65) | 25533203 |
| 14 | Kvartsberg et al., 2015 [43] | AD (n = 25) | (n = 20) | McKhann et al., 1984 [46] | ECL In-house (MSD) | Ng7 (G52–G65) | 26136856 |
| 15 * | Lista et al., 2017 [11] | AD (n = 35); MCI (n = 41) | (n = 21) | McKhann et al., 2011 [44]; Albert et al., 2011 [45] | ELISA In-house | Ng7 (G52–G65) | 28731449 |
| 16A | Merluzzi et al., 2018 [12] | AD (n = 40) | (n = 25) | McKhann et al., 2011 [44] | ECL In-house (MSD) | Ng7 (G52–G65) | 29959263 |
| 16B | Merluzzi et al., 2018 [12] | AD (n = 61) | (n = 291) | McKhann et al., 2011 [44] | ECL In-house (MSD) | Ng7 (G52–G65) | 29959263 |
| 17 | Pereira et al., 2017 [13] | AD+ (n = 65); MCI+ (n = 109); MCI- (n = 36) | Aβ+ (n = 37); Aβ- (n = 57) | McKhann et al., 1984 [46]; Petersen, 2004 [47] | ECL In-house (MSD) | Ng7 (G52–G65) | 28692877 |
| 18 | Portelius et al., 2015 [14] | AD (n = 95); MCI-AD (n = 105); sMCI (n = 68) | (n = 110) | McKhann et al., 1984 [46]; Petersen, 2004 [47] | ECL In-house (MSD) | Ng7 (G52–G65) | 26373605 |
| 19 * | Portelius et al., 2018 [15] | AD (n = 397); MCI (n = 114) | (n = 75) | McKhann et al., 2011 [44]; McKhann et al., 1984 [46] | ELISA In-house | Ng22 (epitope 63–75) | 29700597 |
| 20 * | Sanfillipo et al., 2016 [16] | AD (n = 25); MCI (n = 50) | (n = 44) | McKhann et al., 2011 [44] | ELISA In-house | Ng7 (G52–G65) | 27531278 |
| 21 | Sun et al., 2016 [17] | AD (n = 95); MCI (n = 193) | (n = 111) | McKhann et al., 1984 [46] | ECL In-house (MSD) | Ng7 (G52–G65) | 27321472 |
| 22 | Sutphen et al., 2018 [18] | AD+ (n = 16); MCI+ (n = 58); MCI- (n = 18) | Aβ+ (n = 21); Aβ- (n = 35) | McKhann et al., 1984 [46]; | Erenna® Singulex | Ng G49-G60(P-4793) | 29580670 |
| 23 | Tarawneh et al., 2016 [20] | AD (n = 95) | (n = 207) | McKhann et al., 1984 [46] | Erenna® Singulex | Ng G49-G60(P-4793) | 27018940 |
| 24 | Vogt et al., 2018 [21] | AD (n = 40); MCI (n = 35) | (n = 335) | McKhann et al., 1984 [46]; Albert et al., 2011 [45] | ECL In-house (MSD) | Ng7 (G52–G65) | 30579367 |
| 25 | Wang et al., 2020 [49] | AD (n = 67); MCI (n = 143) | (n = 47) | McKhann et al., 1984 [46] | ECL In-house (MSD) | Ng7 (G52–G65) | 32021212 |
| 26 * | Wang, et al., 2019 [50] | AD (n = 81); MCI (n = 171) | (n = 99) | McKhann et al., 1984 [46] | ECL In-house (MSD) | Ng7 (G52–G65) | 29667155 |
| 27 | Wellington et al., 2016 [23] | AD (n = 100) | (n = 19) | McKhann et al., 1984 [46] | ELISA In-house | Ng7 (G52–G65) | 26826204 |
| 28 | Ye et al., 2019 [24] | AD (n = 67); MCI (n = 143) | (n = 84) | IWG-2 [51] | ECL In-house (MSD) | Ng7 (G52–G65) | 30447377 |
| Visinin-like protein 1 (VILIP-1) | |||||||
| 1. | Babic et al., 2016 [41] | AD (n = 109); MCI (n = 43) | (n = 9) | McKhann et al., 1984 [46] Petersen et al., 1999 [48] | ELISA kit | 26836160 | |
| 2. | Babic et al., 2018 [35] | AD (n = 111); MCI (n = 50) | (n = 9) | McKhann et al., 1984 [46] Petersen et al., 1999 [48] Albert et al., 2011 [45] | ELISA kit | 30329219 | |
| 3. | Kester et al., 2015 [33] | AD (n = 65); MCI (n = 61) | (n = 37) | McKhann et al., 1984 [46] | ELISA kit | 26383836 | |
| 4. | Lee et al., 2008 [34] | AD (n = 33) | (n = 24) | McKhann et al., 1984 [46] | ECL In-house (MSD) | 18703769 | |
| 5. | Luo et al., 2013 [38] | AD (n = 61) | (n = 40) | Dubois et al., 2007 [52] | ELISA kit | 23800322 | |
| 6. | Mroczko et al., 2015 [40] | AD (n = 33); MCI (n = 15) | (n = 18) | McKhann et al., 2011 [44] | ELISA kit | 25159667 | |
| 7. | Tarawneh et al., 2011 [36] | AD (n = 98) | (n = 211) | Morris et al., 2006 [53]; Berg et al., 1998 [54] | MBI Erenna® Singulex | 21823155 | |
| 8. | Tarawneh et al., 2012 [37] | AD (n = 60) | (n = 211) | Morris et al., 2006 [53]; Berg et al., 1998 [54] | MBI Erenna® Singulex | 22357717 | |
| 9. | Tarawneh et al., 2015 [39] | AD (n = 23) | (n = 64) | Morris et al., 2006 [53]; Berg et al., 1998 [54] | MBI Erenna® Singulex | 25867677 | |
| 10. | Tarawneh et al., 2016 [20] | AD (n = 95) | (n = 207) | Albert et al., 2011 [45] | MBI Erenna® Singulex | 27018940 | |
| 11. | Zhang et al., 2018 [42] | AD (n = 18); MCI (n = 24) | (n = 32) | McKhann et al., 1984 [46] | ELISA kit | 30311914 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dulewicz, M.; Kulczyńska-Przybik, A.; Mroczko, B. Neurogranin and VILIP-1 as Molecular Indicators of Neurodegeneration in Alzheimer’s Disease: A Systematic Review and Meta-Analysis. Int. J. Mol. Sci. 2020, 21, 8335. https://doi.org/10.3390/ijms21218335
Dulewicz M, Kulczyńska-Przybik A, Mroczko B. Neurogranin and VILIP-1 as Molecular Indicators of Neurodegeneration in Alzheimer’s Disease: A Systematic Review and Meta-Analysis. International Journal of Molecular Sciences. 2020; 21(21):8335. https://doi.org/10.3390/ijms21218335
Chicago/Turabian StyleDulewicz, Maciej, Agnieszka Kulczyńska-Przybik, and Barbara Mroczko. 2020. "Neurogranin and VILIP-1 as Molecular Indicators of Neurodegeneration in Alzheimer’s Disease: A Systematic Review and Meta-Analysis" International Journal of Molecular Sciences 21, no. 21: 8335. https://doi.org/10.3390/ijms21218335
APA StyleDulewicz, M., Kulczyńska-Przybik, A., & Mroczko, B. (2020). Neurogranin and VILIP-1 as Molecular Indicators of Neurodegeneration in Alzheimer’s Disease: A Systematic Review and Meta-Analysis. International Journal of Molecular Sciences, 21(21), 8335. https://doi.org/10.3390/ijms21218335







