Identification of a Novel Link between the Intermediate Filament Organizer IFO-1 and Cholesterol Metabolism in the Caenorhabditis elegans Intestine
Abstract
1. Introduction
2. Results
2.1. Lipids Are Reduced in the Intestine of ifo-1 Mutants
2.2. ifo-1 Mutants Exhibit Increased Sensitivity to Cholesterol Depletion
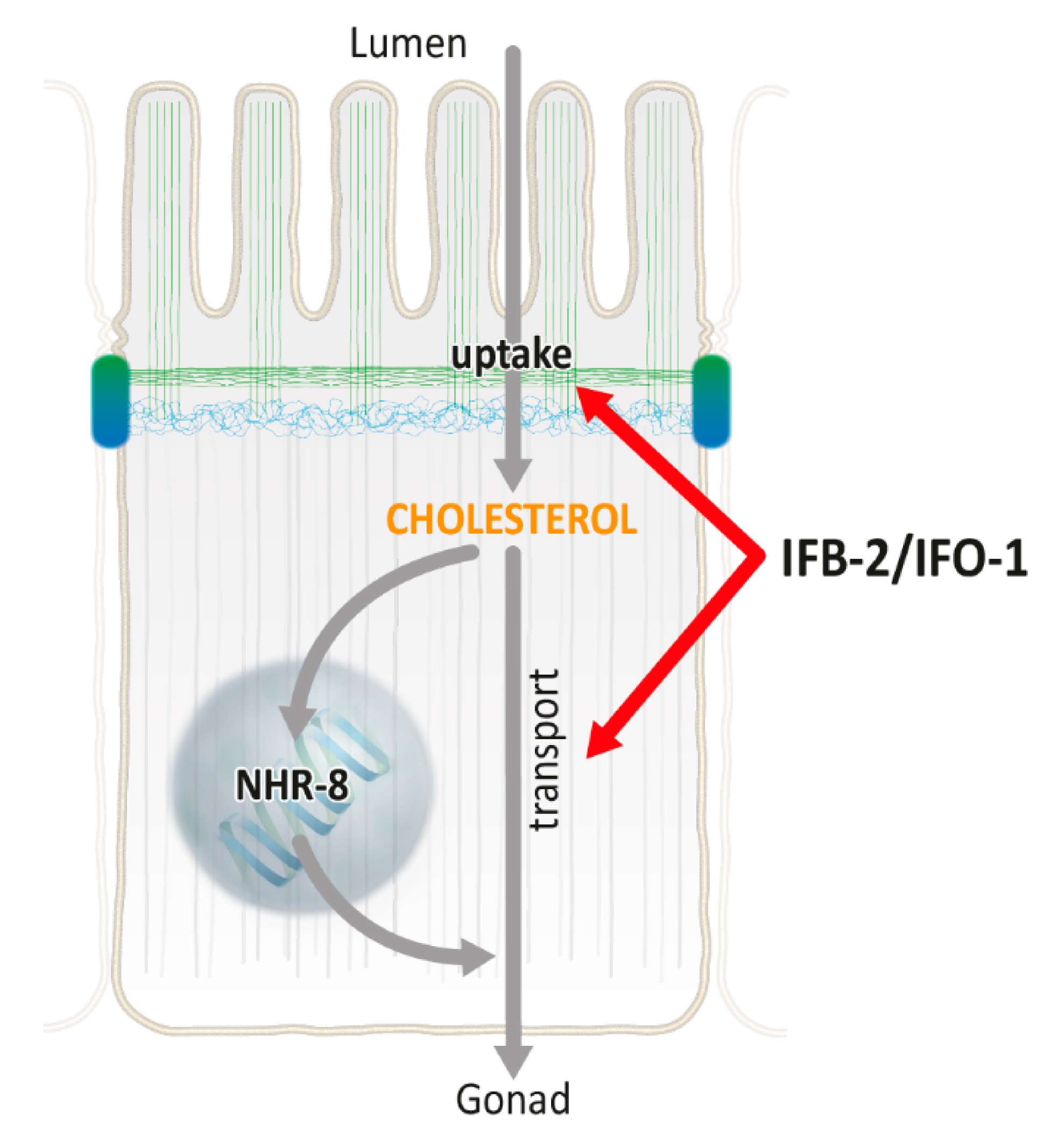
2.3. Cholesterol Uptake and Transport Is Reduced in ifo-1 and ifb-2 Mutants
2.4. Transcriptome Profiling Identifies Striking Similarities between ifo-1 and nhr-8 Mutants
2.5. Lipidomics Reveal Common Features of ifo-1, ifb-2 and nhr-8 Mutants
2.6. nhr-8(RNAi) Mimics the Effect of Cholesterol Depletion in ifo-1(kc2) and ifb-2(kc14)
2.7. High Cholesterol Concentration Partially Rescues ifo-1(kc2) Growth Retardation
3. Discussion
4. Material and Methods
4.1. Strains
4.2. Nile Red Staining
4.3. Electron Microscopy
4.4. RNAi-Induced Larval Arrest
4.5. Cholesterol Depletion Assay
4.6. Cholesterol Uptake and Transfer Assays
4.7. Cholesterol Rescue Experiment
4.8. Gene Ontology Analysis
4.9. Lipid Extraction and Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- McGhee, J.D. The C. elegans intestine. WormBook 2007, 1–36. [Google Scholar] [CrossRef]
- Carberry, K.; Wiesenfahrt, T.; Windoffer, R.; Bossinger, O.; Leube, R.E. Intermediate filaments in Caenorhabditis elegans. Cell Motil. Cytoskelet. 2009, 66, 852–864. [Google Scholar] [CrossRef]
- Geisler, F.; Coch, R.A.; Richardson, C.; Goldberg, M.; Bevilacqua, C.; Prevedel, R.; Leube, R.E. Intestinal intermediate filament polypeptides in C. elegans: Common and isotype-specific contributions to intestinal ultrastructure and function. Sci. Rep. 2020, 10, 3142. [Google Scholar] [CrossRef]
- Geisler, F.; Coch, R.A.; Richardson, C.; Goldberg, M.; Denecke, B.; Bossinger, O.; Leube, R.E. The intestinal intermediate filament network responds to and protects against microbial insults and toxins. Development 2019, 146. [Google Scholar] [CrossRef]
- Karabinos, A.; Schunemann, J.; Parry, D.A. Assembly studies of six intestinal intermediate filament (IF) proteins B2, C1, C2, D1, D2, and E1 in the nematode C. elegans. Cytoskeleton 2017, 74, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Carberry, K.; Wiesenfahrt, T.; Geisler, F.; Stocker, S.; Gerhardus, H.; Uberbach, D.; Davis, W.; Jorgensen, E.; Leube, R.E.; Bossinger, O. The novel intestinal filament organizer IFO-1 contributes to epithelial integrity in concert with ERM-1 and DLG-1. Development 2012, 139, 1851–1862. [Google Scholar] [CrossRef] [PubMed]
- Geisler, F.; Gerhardus, H.; Carberry, K.; Davis, W.; Jorgensen, E.; Richardson, C.; Bossinger, O.; Leube, R.E. A novel function for the MAP kinase SMA-5 in intestinal tube stability. Mol. Biol. Cell 2016, 27, 3855–3868. [Google Scholar] [CrossRef] [PubMed]
- Chitwood, D.J. Biochemistry and function of nematode steroids. Crit. Rev. Biochem. Mol. Biol. 1999, 34, 273–284. [Google Scholar] [CrossRef] [PubMed]
- Hieb, W.F.; Rothstein, M. Sterol requirement for reproduction of a free-living nematode. Science 1968, 160, 778–780. [Google Scholar] [CrossRef] [PubMed]
- Kurzchalia, T.V.; Ward, S. Why do worms need cholesterol? Nat. Cell Biol. 2003, 5, 684–688. [Google Scholar] [CrossRef] [PubMed]
- Merris, M.; Wadsworth, W.G.; Khamrai, U.; Bittman, R.; Chitwood, D.J.; Lenard, J. Sterol effects and sites of sterol accumulation in Caenorhabditis elegans: Developmental requirement for 4alpha-methyl sterols. J. Lipid. Res. 2003, 44, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Watts, J.L.; Ristow, M. Lipid and Carbohydrate Metabolism in Caenorhabditis elegans. Genetics 2017, 207, 413–446. [Google Scholar] [CrossRef]
- Matyash, V.; Entchev, E.V.; Mende, F.; Wilsch-Brauninger, M.; Thiele, C.; Schmidt, A.W.; Knolker, H.J.; Ward, S.; Kurzchalia, T.V. Sterol-derived hormone(s) controls entry into diapause in Caenorhabditis elegans by consecutive activation of DAF-12 and DAF-16. PLoS Biol. 2004, 2, e280. [Google Scholar] [CrossRef]
- Magner, D.B.; Wollam, J.; Shen, Y.; Hoppe, C.; Li, D.; Latza, C.; Rottiers, V.; Hutter, H.; Antebi, A. The NHR-8 nuclear receptor regulates cholesterol and bile acid homeostasis in C. elegans. Cell Metab. 2013, 18, 212–224. [Google Scholar] [CrossRef]
- Lemieux, G.A.; Ashrafi, K. Insights and challenges in using C. elegans for investigation of fat metabolism. Crit. Rev. Biochem. Mol. Biol. 2015, 50, 69–84. [Google Scholar] [CrossRef]
- Valdes, V.J.; Athie, A.; Salinas, L.S.; Navarro, R.E.; Vaca, L. CUP-1 is a novel protein involved in dietary cholesterol uptake in Caenorhabditis elegans. PLoS ONE 2012, 7, e33962. [Google Scholar] [CrossRef]
- Li, J.; Brown, G.; Ailion, M.; Lee, S.; Thomas, J.H. NCR-1 and NCR-2, the C. elegans homologs of the human Niemann-Pick type C1 disease protein, function upstream of DAF-9 in the dauer formation pathways. Development 2004, 131, 5741–5752. [Google Scholar] [CrossRef]
- Brock, T.J.; Browse, J.; Watts, J.L. Fatty acid desaturation and the regulation of adiposity in Caenorhabditis elegans. Genetics 2007, 176, 865–875. [Google Scholar] [CrossRef] [PubMed]
- Lyssenko, N.N.; Miteva, Y.; Gilroy, S.; Hanna-Rose, W.; Schlegel, R.A. An unexpectedly high degree of specialization and a widespread involvement in sterol metabolism among the C. elegans putative aminophospholipid translocases. BMC Dev. Biol. 2008, 8, 96. [Google Scholar] [CrossRef]
- Chen, W.W.; Yi, Y.H.; Chien, C.H.; Hsiung, K.C.; Ma, T.H.; Lin, Y.C.; Lo, S.J.; Chang, T.C. Specific polyunsaturated fatty acids modulate lipid delivery and oocyte development in C. elegans revealed by molecular-selective label-free imaging. Sci. Rep. 2016, 6, 32021. [Google Scholar] [CrossRef]
- Aguilaniu, H.; Fabrizio, P.; Witting, M. The Role of Dafachronic Acid Signaling in Development and Longevity in Caenorhabditis elegans: Digging Deeper Using Cutting-Edge Analytical Chemistry. Front Endocrinol. 2016, 7, 12. [Google Scholar] [CrossRef] [PubMed]
- Boland, S.; Schmidt, U.; Zagoriy, V.; Sampaio, J.L.; Fritsche, R.F.; Czerwonka, R.; Lubken, T.; Reimann, J.; Penkov, S.; Knolker, H.J.; et al. Phosphorylated glycosphingolipids essential for cholesterol mobilization in Caenorhabditis elegans. Nat. Chem. Biol. 2017, 13, 647–654. [Google Scholar] [CrossRef]
- Griffitts, J.S.; Haslam, S.M.; Yang, T.; Garczynski, S.F.; Mulloy, B.; Morris, H.; Cremer, P.S.; Dell, A.; Adang, M.J.; Aroian, R.V. Glycolipids as receptors for Bacillus thuringiensis crystal toxin. Science 2005, 307, 922–925. [Google Scholar] [CrossRef] [PubMed]
- Schweitzer, S.C.; Evans, R.M. Vimentin and lipid metabolism. Subcell. Biochem. 1998, 31, 437–462. [Google Scholar]
- Evans, R.M. Intermediate filaments and lipoprotein cholesterol. Trends Cell Biol. 1994, 4, 149–151. [Google Scholar] [CrossRef]
- Shen, W.J.; Zaidi, S.K.; Patel, S.; Cortez, Y.; Ueno, M.; Azhar, R.; Azhar, S.; Kraemer, F.B. Ablation of vimentin results in defective steroidogenesis. Endocrinology 2012, 153, 3249–3257. [Google Scholar] [CrossRef] [PubMed]
- Ko, C.W.; Qu, J.; Black, D.D.; Tso, P. Regulation of intestinal lipid metabolism: Current concepts and relevance to disease. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 169–183. [Google Scholar] [CrossRef]
- Wang, D.Q. New concepts of mechanisms of intestinal cholesterol absorption. Ann Hepatol 2003, 2, 113–121. [Google Scholar] [CrossRef]
- Matyash, V.; Geier, C.; Henske, A.; Mukherjee, S.; Hirsh, D.; Thiele, C.; Grant, B.; Maxfield, F.R.; Kurzchalia, T.V. Distribution and transport of cholesterol in Caenorhabditis elegans. Mol. Biol. Cell 2001, 12, 1725–1736. [Google Scholar] [CrossRef]
- Altmann, S.W.; Davis, H.R., Jr.; Zhu, L.J.; Yao, X.; Hoos, L.M.; Tetzloff, G.; Iyer, S.P.; Maguire, M.; Golovko, A.; Zeng, M.; et al. Niemann-Pick C1 Like 1 protein is critical for intestinal cholesterol absorption. Science 2004, 303, 1201–1204. [Google Scholar] [CrossRef]
- Asghar, M.N.; Priyamvada, S.; Nystrom, J.H.; Anbazhagan, A.N.; Dudeja, P.K.; Toivola, D.M. Keratin 8 knockdown leads to loss of the chloride transporter DRA in the colon. Am. J. Physiol. Gastrointest Liver. Physiol. 2016, 310, G1147–G1154. [Google Scholar] [CrossRef]
- Hou, X.; Wu, Q.; Rajagopalan, C.; Zhang, C.; Bouhamdan, M.; Wei, H.; Chen, X.; Zaman, K.; Li, C.; Sun, X.; et al. CK19 stabilizes CFTR at the cell surface by limiting its endocytic pathway degradation. FASEB J. 2019, 33, 12602–12615. [Google Scholar] [CrossRef]
- Toivola, D.M.; Krishnan, S.; Binder, H.J.; Singh, S.K.; Omary, M.B. Keratins modulate colonocyte electrolyte transport via protein mistargeting. J. Cell Biol. 2004, 164, 911–921. [Google Scholar] [CrossRef] [PubMed]
- Duan, Y.; Sun, Y.; Zhang, F.; Zhang, W.K.; Wang, D.; Wang, Y.; Cao, X.; Hu, W.; Xie, C.; Cuppoletti, J.; et al. Keratin K18 increases cystic fibrosis transmembrane conductance regulator (CFTR) surface expression by binding to its C-terminal hydrophobic patch. J. Biol. Chem. 2012, 287, 40547–40559. [Google Scholar] [CrossRef]
- Watts, J.L.; Browse, J. Genetic dissection of polyunsaturated fatty acid synthesis in Caenorhabditis elegans. Proc. Natl. Acad. Sci. USA 2002, 99, 5854–5859. [Google Scholar] [CrossRef]
- Watts, J.L. Fat synthesis and adiposity regulation in Caenorhabditis elegans. Trends Endocrinol Metab 2009, 20, 58–65. [Google Scholar] [CrossRef]
- Marza, E.; Simonsen, K.T.; Faergeman, N.J.; Lesa, G.M. Expression of ceramide glucosyltransferases, which are essential for glycosphingolipid synthesis, is only required in a small subset of C. elegans cells. J. Cell Sci. 2009, 122, 822–833. [Google Scholar] [CrossRef]
- Nomura, K.H.; Murata, D.; Hayashi, Y.; Dejima, K.; Mizuguchi, S.; Kage-Nakadai, E.; Gengyo-Ando, K.; Mitani, S.; Hirabayashi, Y.; Ito, M.; et al. Ceramide glucosyltransferase of the nematode Caenorhabditis elegans is involved in oocyte formation and in early embryonic cell division. Glycobiology 2011, 21, 834–848. [Google Scholar] [CrossRef] [PubMed]
- Gerisch, B.; Antebi, A. Hormonal signals produced by DAF-9/cytochrome P450 regulate C. elegans dauer diapause in response to environmental cues. Development 2004, 131, 1765–1776. [Google Scholar] [CrossRef]
- Motola, D.L.; Cummins, C.L.; Rottiers, V.; Sharma, K.K.; Li, T.; Li, Y.; Suino-Powell, K.; Xu, H.E.; Auchus, R.J.; Antebi, A.; et al. Identification of ligands for DAF-12 that govern dauer formation and reproduction in C. elegans. Cell 2006, 124, 1209–1223. [Google Scholar] [CrossRef]
- Sarria, A.J.; Panini, S.R.; Evans, R.M. A functional role for vimentin intermediate filaments in the metabolism of lipoprotein-derived cholesterol in human SW-13 cells. J. Biol. Chem. 1992, 267, 19455–19463. [Google Scholar] [PubMed]
- Styers, M.L.; Salazar, G.; Love, R.; Peden, A.A.; Kowalczyk, A.P.; Faundez, V. The endo-lysosomal sorting machinery interacts with the intermediate filament cytoskeleton. Mol. Biol. Cell 2004, 15, 5369–5382. [Google Scholar] [CrossRef]
- Peake, K.B.; Vance, J.E. Defective cholesterol trafficking in Niemann-Pick C-deficient cells. FEBS Lett. 2010, 584, 2731–2739. [Google Scholar] [CrossRef]
- Vanier, M.T. Niemann-Pick disease type C. Orphanet J. Rare Dis. 2010, 5, 16. [Google Scholar] [CrossRef]
- Vanier, M.T. Complex lipid trafficking in Niemann-Pick disease type C. J. Inherit. Metab. Dis. 2015, 38, 187–199. [Google Scholar] [CrossRef]
- Otarigho, B.; Aballay, A. Cholesterol Regulates Innate Immunity via Nuclear Hormone Receptor NHR-8. iScience 2020, 23, 101068. [Google Scholar] [CrossRef]
- Greenspan, P.; Mayer, E.P.; Fowler, S.D. Nile red: A selective fluorescent stain for intracellular lipid droplets. J. Cell Biol. 1985, 100, 965–973. [Google Scholar] [CrossRef]
- Timmons, L.; Fire, A. Specific interference by ingested dsRNA. Nature 1998, 395, 854. [Google Scholar] [CrossRef] [PubMed]
- Huang da, W.; Sherman, B.T.; Lempicki, R.A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 2009, 4, 44–57. [Google Scholar] [CrossRef]
- Tharyan, R.G.; Annibal, A.; Schiffer, I.; Laboy, R.; Atanassov, I.; Weber, A.L.; Gerisch, B.; Antebi, A. NFYB-1 regulates mitochondrial function and longevity via lysosomal prosaposin. Nat. Metab. 2020, 2, 387–396. [Google Scholar] [CrossRef]
- Dabrowski, R.; Ripa, R.; Latza, C.; Annibal, A.; Antebi, A. Optimization of mass spectrometry settings for steroidomic analysis in young and old killifish. Anal. Bioanal. Chem. 2020, 412, 4089–4099. [Google Scholar] [CrossRef]







Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Coch, R.A.; Geisler, F.; Annibal, A.; Antebi, A.; Leube, R.E. Identification of a Novel Link between the Intermediate Filament Organizer IFO-1 and Cholesterol Metabolism in the Caenorhabditis elegans Intestine. Int. J. Mol. Sci. 2020, 21, 8219. https://doi.org/10.3390/ijms21218219
Coch RA, Geisler F, Annibal A, Antebi A, Leube RE. Identification of a Novel Link between the Intermediate Filament Organizer IFO-1 and Cholesterol Metabolism in the Caenorhabditis elegans Intestine. International Journal of Molecular Sciences. 2020; 21(21):8219. https://doi.org/10.3390/ijms21218219
Chicago/Turabian StyleCoch, Richard A., Florian Geisler, Andrea Annibal, Adam Antebi, and Rudolf E. Leube. 2020. "Identification of a Novel Link between the Intermediate Filament Organizer IFO-1 and Cholesterol Metabolism in the Caenorhabditis elegans Intestine" International Journal of Molecular Sciences 21, no. 21: 8219. https://doi.org/10.3390/ijms21218219
APA StyleCoch, R. A., Geisler, F., Annibal, A., Antebi, A., & Leube, R. E. (2020). Identification of a Novel Link between the Intermediate Filament Organizer IFO-1 and Cholesterol Metabolism in the Caenorhabditis elegans Intestine. International Journal of Molecular Sciences, 21(21), 8219. https://doi.org/10.3390/ijms21218219





