Role of AT1G72910, AT1G72940, and ADR1-LIKE 2 in Plant Immunity under Nonsense-Mediated mRNA Decay-Compromised Conditions at Low Temperatures
Abstract
:1. Introduction
2. Results
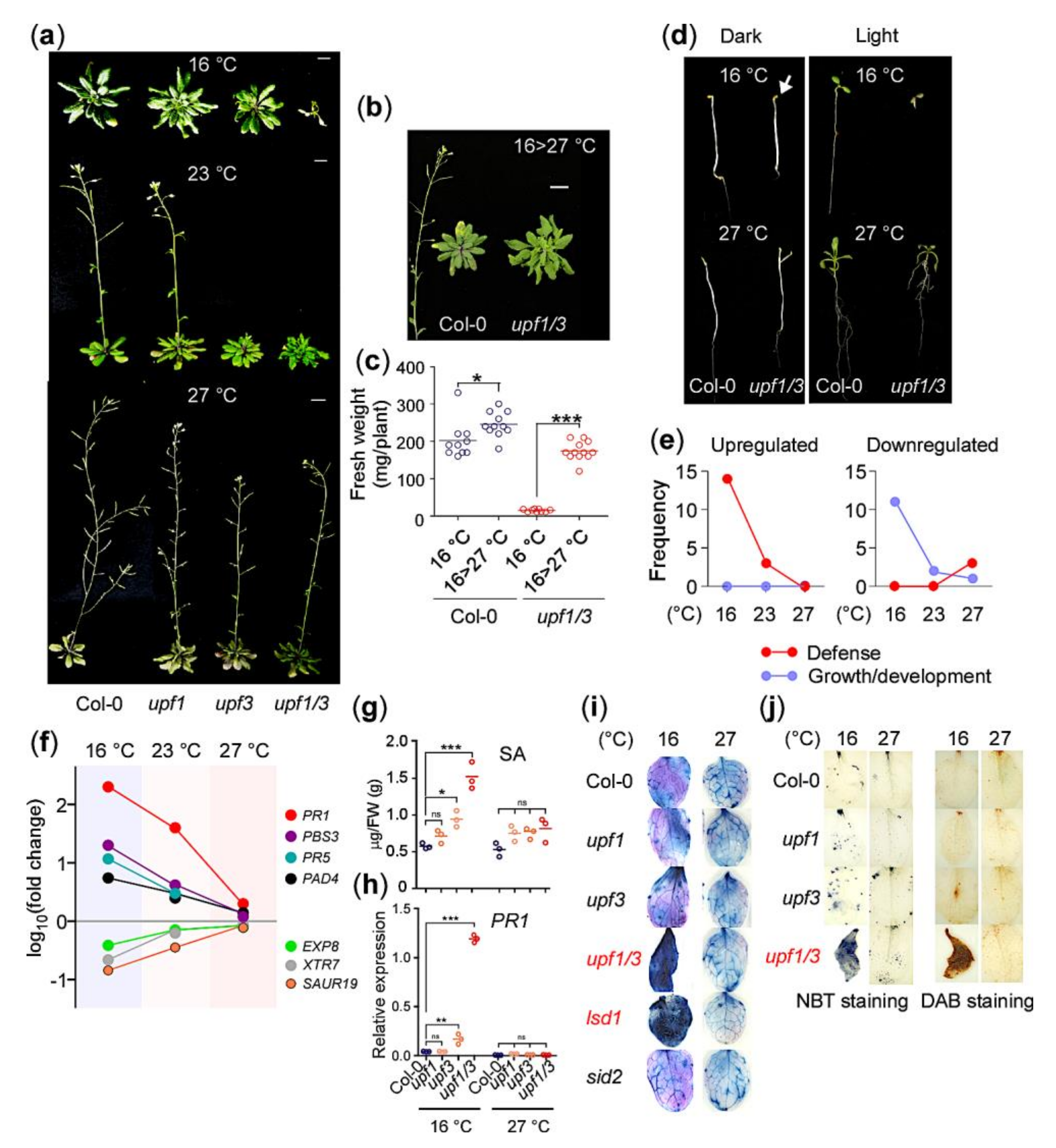
2.1. The NMD-Deficient upf1 upf3 Mutants Exhibit Autoimmunity at 16 °C
2.2. Identification of AT1G72910, AT1G72940, and ADR1-LIKE 2 as NLR Genes Regulated by NMD at 16 °C
2.3. AT1G72910, AT1G72940, and ADR1-LIKE 2 Are Required for Pathogen Response at Lower Temperatures
2.4. Knockdown of AT1G72910, AT1G72940, and ADR1-LIKE 2 Can Rescue the Growth Arrest Phenotype of upf1/3 Mutants
2.5. Overexpression of AT1G72910, AT1G72940, and ADR1-LIKE 2 Results in Autoimmunity in WT Plants
3. Discussion
4. Materials and Methods
4.1. Plant Materials and Growth Conditions
4.2. Inhibition of Translation by Cycloheximide (CHX)
4.3. Analyses of Cell Death and Pathogen Assays
4.4. RNA Sequencing
4.5. Public Transcriptome Data and Bioinformatics Analyses
4.6. qPCR Expression Analyses
4.7. Quantification of Phytohormones and Chlorophyll Contents
4.8. Determination of mRNA Degradation Kinetics
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| NLR | Nucleotide-binding, Leucine-rich Repeat |
| SA | Salicylic acid |
| CHX | Cycloheximide |
| DEGs | Differentially expressed genes |
| HR | Hypersensitive response |
References
- Hua, J. Modulation of plant immunity by light, circadian rhythm, and temperature. Curr. Opin. Plant Biol. 2013, 16, 406–413. [Google Scholar] [CrossRef] [PubMed]
- Huot, B.; Castroverde, C.D.M.; Velásquez, A.C.; Hubbard, E.; Pulman, J.A.; Yao, J.; Childs, K.L.; Tsuda, K.; Montgomery, B.L.; He, S.Y. Dual impact of elevated temperature on plant defence and bacterial virulence in Arabidopsis. Nat. Commun. 2017, 8, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Gangappa, S.N.; Berriri, S.; Kumar, S.V. PIF4 Coordinates Thermosensory Growth and Immunity in Arabidopsis. Curr. Biol. 2017, 27, 243–249. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schweingruber, C.; Rufener, S.C.; Zünd, D.; Yamashita, A.; Mühlemann, O. Nonsense-mediated mRNA decay — Mechanisms of substrate mRNA recognition and degradation in mammalian cells. Biochim. Biophys. Acta (BBA) Bioenergy 2013, 1829, 612–623. [Google Scholar] [CrossRef] [PubMed]
- Isken, O.; Maquat, L.E. The multiple lives of NMD factors: Balancing roles in gene and genome regulation. Nat. Rev. Genet. 2008, 9, 699–712. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Drechsel, G.; Kahles, A.; Kesarwani, A.K.; Stauffer, E.; Behr, J.; Drewe, P.; Rätsch, G.; Wachter, A. Nonsense-Mediated Decay of Alternative Precursor mRNA Splicing Variants Is a Major Determinant of the Arabidopsis Steady State Transcriptome. Plant Cell 2013, 25, 3726–3742. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kurihara, Y.; Matsui, A.; Hanada, K.; Kawashima, M.; Ishida, J.; Morosawa, T.; Tanaka, M.; Kaminuma, E.; Mochizuki, Y.; Matsushima, A.; et al. Genome-wide suppression of aberrant mRNA-like noncoding RNAs by NMD in Arabidopsis. Proc. Natl. Acad. Sci. USA 2009, 106, 2453–2458. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jeong, H.-J.; Kim, Y.J.; Kim, S.H.; Kim, Y.-H.; Lee, I.-J.; Shin, J.S. Nonsense-Mediated mRNA Decay Factors, UPF1 and UPF3, Contribute to Plant Defense. Plant Cell Physiol. 2011, 52, 2147–2156. [Google Scholar] [CrossRef] [Green Version]
- Riehs-Kearnan, N.; Gloggnitzer, J.; Dekrout, B.; Jonak, C.; Riha, K. Aberrant growth and lethality of Arabidopsis deficient in nonsense-mediated RNA decay factors is caused by autoimmune-like response. Nucleic Acids Res. 2012, 40, 5615–5624. [Google Scholar] [CrossRef] [Green Version]
- Bari, R.; Jones, J.D.G. Role of plant hormones in plant defence responses. Plant Mol. Biol. 2008, 69, 473–488. [Google Scholar] [CrossRef]
- Pieterse, C.M.; Van Der Does, D.; Zamioudis, C.; Leon-Reyes, A.; Van Wees, S.C. Hormonal Modulation of Plant Immunity. Annu. Rev. Cell Dev. Biol. 2012, 28, 489–521. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wildermuth, M.C.; Dewdney, J.; Wu, G.; Ausubel, F.M. Isochorismate synthase is required to synthesize salicylic acid for plant defence. Nat. Cell Biol. 2001, 414, 562–565. [Google Scholar] [CrossRef]
- Chen, Z.; Zheng, Z.; Huang, J.; Lai, Z.; Fan, B. Biosynthesis of salicylic acid in plants. Plant Signal. Behav. 2009, 4, 493–496. [Google Scholar] [CrossRef]
- Lee, H.-J.; Park, Y.-J.; Seo, P.J.; Kim, J.-H.; Sim, H.-J.; Kim, S.-G.; Park, C.-M. Systemic Immunity Requires SnRK2.8-Mediated Nuclear Import of NPR1 in Arabidopsis. Plant Cell 2015, 27, 3425–3438. [Google Scholar] [CrossRef] [Green Version]
- Tada, Y.; Spoel, S.H.; Pajerowska-Mukhtar, K.; Mou, Z.; Song, J.; Wang, C.; Zuo, J.; Dong, X. Plant Immunity Requires Conformational Charges of NPR1 via S-Nitrosylation and Thioredoxins. Science 2008, 321, 952–956. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jones, J.D.G.; Dangl, J.L. The plant immune system. Nat. Cell Biol. 2006, 444, 323–329. [Google Scholar] [CrossRef] [Green Version]
- Fu, Z.Q.; Dong, X. Systemic Acquired Resistance: Turning Local Infection into Global Defense. Annu. Rev. Plant Biol. 2013, 64, 839–863. [Google Scholar] [CrossRef] [Green Version]
- Dodds, P.N.; Rathjen, J.P. Plant immunity: Towards an integrated view of plant–pathogen interactions. Nat. Rev. Genet. 2010, 11, 539–548. [Google Scholar] [CrossRef]
- Maekawa, T.; A Kufer, T.; Schulze-Lefert, P. NLR functions in plant and animal immune systems: so far and yet so close. Nat. Immunol. 2011, 12, 817–826. [Google Scholar] [CrossRef]
- Bernoux, M.; Burdett, H.; Williams, S.J.; Zhang, X.; Chen, C.; Newell, K.; Lawrence, G.J.; Kobe, B.; Ellis, J.G.; Anderson, P.A.; et al. Comparative Analysis of the Flax Immune Receptors L6 and L7 Suggests an Equilibrium-Based Switch Activation Model. Plant Cell 2016, 28, 146–159. [Google Scholar] [CrossRef] [Green Version]
- Staiger, D.; Korneli, C.; Lummer, M.; Navarro, L. Emerging role for RNA-based regulation in plant immunity. N. Phytol. 2012, 197, 394–404. [Google Scholar] [CrossRef]
- Alcázar, R.; Parker, J.E. The impact of temperature on balancing immune responsiveness and growth in Arabidopsis. Trends Plant Sci. 2011, 16, 666–675. [Google Scholar] [CrossRef] [PubMed]
- Gloggnitzer, J.; Akimcheva, S.; Srinivasan, A.; Kusenda, B.; Riehs, N.; Stampfl, H.; Bautor, J.; Dekrout, B.; Jonak, C.; Jiménez-Gómez, J.M.; et al. Nonsense-Mediated mRNA Decay Modulates Immune Receptor Levels to Regulate Plant Antibacterial Defense. Cell Host Microbe 2014, 16, 376–390. [Google Scholar] [CrossRef] [Green Version]
- Jung, H.W.; Panigrahi, G.K.; Jung, G.Y.; Lee, Y.J.; Shin, K.H.; Sahoo, A.; Choi, E.S.; Lee, E.; Kim, K.M.; Yang, S.H.; et al. Pathogen-Associated Molecular Pattern-Triggered Immunity Involves Proteolytic Degradation of Core Nonsense-Mediated mRNA Decay Factors During the Early Defense Response. Plant Cell 2020, 32, 1081–1101. [Google Scholar] [CrossRef] [Green Version]
- Arciga-Reyes, L.; Wootton, L.; Kieffer, M.; Davies, B. UPF1 is required for nonsense-mediated mRNA decay (NMD) and RNAi in Arabidopsis. Plant J. 2006, 47, 480–489. [Google Scholar] [CrossRef]
- Nasim, Z.; Fahim, M.; Ahn, J.H. Possible Role of MADS AFFECTING FLOWERING 3 and B-BOX DOMAIN PROTEIN 19 in Flowering Time Regulation of Arabidopsis Mutants with Defects in Nonsense-Mediated mRNA Decay. Front. Plant Sci. 2017, 8. [Google Scholar] [CrossRef] [Green Version]
- Rayson, S.; Arciga-Reyes, L.; Wootton, L.; Zabala, M.D.T.; Truman, W.; Graham, N.; Grant, M.; Davies, B. A Role for Nonsense-Mediated mRNA Decay in Plants: Pathogen Responses Are Induced in Arabidopsis thaliana NMD Mutants. PLoS ONE 2012, 7, e31917. [Google Scholar] [CrossRef]
- Raxwal, V.K.; Simpson, C.G.; Gloggnitzer, J.; Entinze, J.C.; Guo, W.; Zhang, R.; Brown, J.W.; Riha, K. Nonsense-mediated RNA Decay Factor UPF1 is Critical for Post-transcriptional and Post-translational Gene Regulation in Arabidopsis. Plant Cell 2020. [Google Scholar] [CrossRef]
- Mammarella, N.D.; Cheng, Z.; Fu, Z.Q.; Daudi, A.; Bolwell, G.P.; Dong, X.; Ausubel, F.M. Apoplastic peroxidases are required for salicylic acid-mediated defense against Pseudomonas syringae. Phytochemistry 2015, 112, 110–121. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Neuenschwander, U.; Vernooij, B.; Friedrich, L.; Uknes, S.; Kessmann, H.; Ryals, J. Is hydrogen peroxide a second messenger of salicylic acid in systemic acquired resistance? Plant J. 1995, 8, 227–233. [Google Scholar] [CrossRef]
- Feys, B.J.; Moisan, L.J.; Newman, M.; Parker, J.E. Direct interaction between the Arabidopsis disease resistance signaling proteins, EDS1 and PAD4. EMBO J. 2001, 20, 5400–5411. [Google Scholar] [CrossRef]
- Rustérucci, C.; Aviv, D.H.; Holt, B.F.; Dangl, J.L.; Parker, J.E. The disease resistance signaling components EDS1 and PAD4 are essential regulators of the cell death pathway controlled by LSD1 in Arabidopsis. Plant Cell 2001, 13, 2211–2224. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johnson, M.A.; Pérez-Amador, M.A.; Lidder, P.; Green, P.J. Mutants of Arabidopsis defective in a sequence-specific mRNA degradation pathway. Proc. Natl. Acad. Sci. USA 2000, 97, 13991–13996. [Google Scholar] [CrossRef] [Green Version]
- Xia, S.; Cheng, Y.T.; Huang, S.; Win, J.; Soards, A.; Jinn, T.-L.; Jones, J.D.; Kamoun, S.; Chen, S.; Zhang, Y.; et al. Regulation of Transcription of Nucleotide-Binding Leucine-Rich Repeat-Encoding Genes SNC1 and RPP4 via H3K4 Trimethylation. Plant Physiol. 2013, 162, 1694–1705. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Loon, L.C. The Intelligent Behavior of Plants. Trends Plant Sci. 2016, 21, 286–294. [Google Scholar] [CrossRef]
- Cui, H.; Tsuda, K.; Parker, J.E. Effector-Triggered Immunity: From Pathogen Perception to Robust Defense. Annu. Rev. Plant Biol. 2015, 66, 487–511. [Google Scholar] [CrossRef]
- Sano, S.; Aoyama, M.; Nakai, K.; Shimotani, K.; Yamasaki, K.; Sato, M.H.; Tojo, D.; Suwastika, I.N.; Nomura, H.; Eshiina, T. Light-dependent expression of flg22-induced defense genes in Arabidopsis. Front. Plant Sci. 2014, 5, 531. [Google Scholar] [CrossRef] [Green Version]
- Zhou, N.; Tootle, T.L.; Tsui, F.; Klessig, D.F.; Glazebrook, J. PAD4 Functions Upstream from Salicylic Acid to Control Defense Responses in Arabidopsis. Plant Cell 1998, 10, 1021–1030. [Google Scholar] [CrossRef] [Green Version]
- Roberts, M.R.; Paul, N.D. Seduced by the dark side: Integrating molecular and ecological perspectives on the influence of light on plant defence against pests and pathogens. N. Phytol. 2006, 170, 677–699. [Google Scholar] [CrossRef] [Green Version]
- Pink, B.; Mueller, M.J.; Berger, S. Light conditions influence specific defence responses in incompatible plant?pathogen interactions: Uncoupling systemic resistance from salicylic acid and PR-1 accumulation. Planta 2004, 219, 673–683. [Google Scholar] [CrossRef]
- Wittkopp, N.; Huntzinger, E.; Weiler, C.; Saulière, J.; Schmidt, S.; Sonawane, M.; Izaurralde, E. Nonsense-Mediated mRNA Decay Effectors Are Essential for Zebrafish Embryonic Development and Survival. Mol. Cell. Biol. 2009, 29, 3517–3528. [Google Scholar] [CrossRef] [Green Version]
- Bruno, I.G.; Karam, R.; Huang, L.; Bhardwaj, A.; Lou, C.H.; Shum, E.Y.; Song, H.-W.; Corbett, M.A.; Gifford, W.D.; Gecz, J.; et al. Identification of a MicroRNA that Activates Gene Expression by Repressing Nonsense-Mediated RNA Decay. Mol. Cell 2011, 42, 500–510. [Google Scholar] [CrossRef] [Green Version]
- Filichkin, S.A.; Cumbie, J.S.; Dharmawardhana, P.; Jaiswal, P.; Chang, J.H.; Palusa, S.G.; Reddy, A.; Megraw, M.; Mockler, T.C. Environmental Stresses Modulate Abundance and Timing of Alternatively Spliced Circadian Transcripts in Arabidopsis. Mol. Plant 2015, 8, 207–227. [Google Scholar] [CrossRef] [Green Version]
- Aarts, N.; Metz, M.; Holub, E.; Staskawicz, B.J.; Daniels, M.J.; Parker, J.E. Different requirements for EDS1 and NDR1 by disease resistance genes define at least two R gene-mediated signaling pathways in Arabidopsis. Proc. Natl. Acad. Sci. USA 1998, 95, 10306–10311. [Google Scholar] [CrossRef] [Green Version]
- Gassmann, W. Alternative Splicing in Plant Defense. In The Future of HIV-1 Therapeutics; Springer Science and Business Media LLC: New York, NY, USA, 2008; Volume 326, pp. 219–233. [Google Scholar]
- Kim, S.H.; Gao, F.; Bhattacharjee, S.; Adiasor, J.A.; Nam, J.C.; Gassmann, W. The Arabidopsis Resistance-Like Gene SNC1 Is Activated by Mutations in SRFR1 and Contributes to Resistance to the Bacterial Effector AvrRps4. PLoS Pathog. 2010, 6, e1001172. [Google Scholar] [CrossRef]
- Karasov, T.L.; Chae, E.; Herman, J.J.; Bergelson, J. Mechanisms to Mitigate the Trade-Off between Growth and Defense. Plant Cell 2017, 29, 666–680. [Google Scholar] [CrossRef] [Green Version]
- Heidrich, K.; Tsuda, K.; Blanvillain-Baufumé, S.; Wirthmueller, L.; Bautor, J.; Parker, J.E. Arabidopsis TNL-WRKY domain receptor RRS1 contributes to temperature-conditioned RPS4 auto-immunity. Front. Plant Sci. 2013, 4, 403. [Google Scholar] [CrossRef] [Green Version]
- Ehori, K.; Watanabe, Y. UPF3 suppresses aberrant spliced mRNA in Arabidopsis. Plant J. 2005, 43, 530–540. [Google Scholar] [CrossRef]
- Boyes, D.C.; Zayed, A.M.; Ascenzi, R.; McCaskill, A.J.; Hoffman, N.E.; Davis, K.R.; Görlach, J. Growth Stage–Based Phenotypic Analysis of Arabidopsis. Plant Cell 2001, 13, 1499–1510. [Google Scholar] [CrossRef] [Green Version]
- Van Wees, S.C. Phenotypic Analysis of Arabidopsis Mutants: Trypan Blue Stain for Fungi, Oomycetes, and Dead Plant Cells. Cold Spring Harb. Protoc. 2008, 2008, 4982. [Google Scholar] [CrossRef] [Green Version]
- Thordal-Christensen, H.; Zhang, Z.; Wei, Y.; Collinge, D.B. Subcellular localization of H2O2 in plants. H2O2 accumulation in papillae and hypersensitive response during the barley-powdery mildew interaction. Plant J. 1997, 11, 1187–1194. [Google Scholar] [CrossRef]
- Liu, X.; Sun, Y.; Kørner, C.J.; Du, X.; Vollmer, M.E.; Pajerowska-Mukhtar, K.M. Bacterial Leaf Infiltration Assay for Fine Characterization of Plant Defense Responses using the Arabidopsis thaliana-Pseudomonas syringae Pathosystem. J. Vis. Exp. 2015. [Google Scholar] [CrossRef] [Green Version]
- Rallapalli, G.; Kemen, E.M.; Robert-Seilaniantz, A.; Segonzac, C.; Etherington, G.J.; Sohn, K.H.; MacLean, D.; Jones, J.D.G. EXPRSS: An Illumina based high-throughput expression-profiling method to reveal transcriptional dynamics. BMC Genom. 2014, 15, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Bazin, J.; Mariappan, K.; Jiang, Y.; Blein, T.; Voelz, R.; Crespi, M.; Hirt, H. Role of MPK4 in pathogen-associated molecular pattern-triggered alternative splicing in Arabidopsis. PLoS Pathog. 2020, 16, e1008401. [Google Scholar] [CrossRef]
- Latrasse, D.; Jégu, T.; Li, H.; Zelicourt, A.D.J.D.; Raynaud, C.; Legras, S.; Gust, A.; Samajova, O.; Veluchamy, A.; Rayapuram, N.; et al. MAPK-triggered chromatin reprogramming by histone deacetylase in plant innate immunity. Genome Biol. 2017, 18, 1–19. [Google Scholar] [CrossRef]
- Caarls, L.; Van Der Does, D.; Hickman, R.; Jansen, W.; Van Verk, M.C.; Silvia, P.; Lorenzo, O.; Solano, R.; Pieterse, C.M.J.; Van Wees, S.C. Assessing the Role of ETHYLENE RESPONSE FACTOR Transcriptional Repressors in Salicylic Acid-Mediated Suppression of Jasmonic Acid-Responsive Genes. Plant Cell Physiol. 2016, 58, 266–278. [Google Scholar] [CrossRef]
- Trapnell, C.; Roberts, A.; A Goff, L.; Pertea, G.; Kim, D.; Kelley, D.R.; Pimentel, H.; Salzberg, S.L.; Rinn, J.L.; Pachter, L. Differential gene and transcript expression analysis of RNA-seq experiments with TopHat and Cufflinks. Nat. Protoc. 2012, 7, 562–578. [Google Scholar] [CrossRef] [Green Version]
- Goff, L.A.; Trapnell, C.; Kelley, D. CummeRbund: Visualization and exploration of Cufflinks high-throughput sequencing data. R Package Vers. 2012, 2. [Google Scholar]
- Maere, S.; Heymans, K.; Kuiper, M. BiNGO: A Cytoscape plugin to assess overrepresentation of Gene Ontology categories in Biological Networks. Bioinformatics 2005, 21, 3448–3449. [Google Scholar] [CrossRef] [Green Version]
- Proost, S.; Van Bel, M.; Vaneechoutte, D.; Van De Peer, Y.; Inzé, D.; Mueller-Roeber, B.; Vandepoele, K. PLAZA 3.0: An access point for plant comparative genomics. Nucleic Acids Res. 2014, 43, D974–D981. [Google Scholar] [CrossRef] [Green Version]
- Hong, S.M.; Bahn, S.C.; Lyu, A.; Jung, H.S.; Ahn, J.H. Identification and Testing of Superior Reference Genes for a Starting Pool of Transcript Normalization in Arabidopsis. Plant Cell Physiol. 2010, 51, 1694–1706. [Google Scholar] [CrossRef] [Green Version]
- Nehela, Y.; Hijaz, F.; Elzaawely, A.A.; El-Zahaby, H.M.; Killiny, N. Phytohormone profiling of the sweet orange (Citrus sinensis (L.) Osbeck) leaves and roots using GC–MS-based method. J. Plant Physiol. 2016, 199, 12–17. [Google Scholar] [CrossRef]
- Hijaz, F.; Killiny, N. Collection and Chemical Composition of Phloem Sap from Citrus sinensis L. Osbeck (Sweet Orange). PLoS ONE 2014, 9, e101830. [Google Scholar] [CrossRef] [Green Version]
- Frye, C.A.; Tang, D.; Innes, R.W. Negative regulation of defense responses in plants by a conserved MAPKK kinase. Proc. Natl. Acad. Sci. USA 2001, 98, 373–378. [Google Scholar] [CrossRef]
- Ratnadiwakara, M.; Änkö, M.-L. mRNA Stability Assay Using Transcription Inhibition by Actinomycin D in Mouse Pluripotent Stem Cells. Bio-Protocol 2018, 8, 3072. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nasim, Z.; Fahim, M.; Gawarecka, K.; Susila, H.; Jin, S.; Youn, G.; Ahn, J.H. Role of AT1G72910, AT1G72940, and ADR1-LIKE 2 in Plant Immunity under Nonsense-Mediated mRNA Decay-Compromised Conditions at Low Temperatures. Int. J. Mol. Sci. 2020, 21, 7986. https://doi.org/10.3390/ijms21217986
Nasim Z, Fahim M, Gawarecka K, Susila H, Jin S, Youn G, Ahn JH. Role of AT1G72910, AT1G72940, and ADR1-LIKE 2 in Plant Immunity under Nonsense-Mediated mRNA Decay-Compromised Conditions at Low Temperatures. International Journal of Molecular Sciences. 2020; 21(21):7986. https://doi.org/10.3390/ijms21217986
Chicago/Turabian StyleNasim, Zeeshan, Muhammad Fahim, Katarzyna Gawarecka, Hendry Susila, Suhyun Jin, Geummin Youn, and Ji Hoon Ahn. 2020. "Role of AT1G72910, AT1G72940, and ADR1-LIKE 2 in Plant Immunity under Nonsense-Mediated mRNA Decay-Compromised Conditions at Low Temperatures" International Journal of Molecular Sciences 21, no. 21: 7986. https://doi.org/10.3390/ijms21217986





