The Roles of CCN1/CYR61 in Pulmonary Diseases
Abstract
:1. Introduction
2. CCN1
3. CCN1 in Lung Diseases
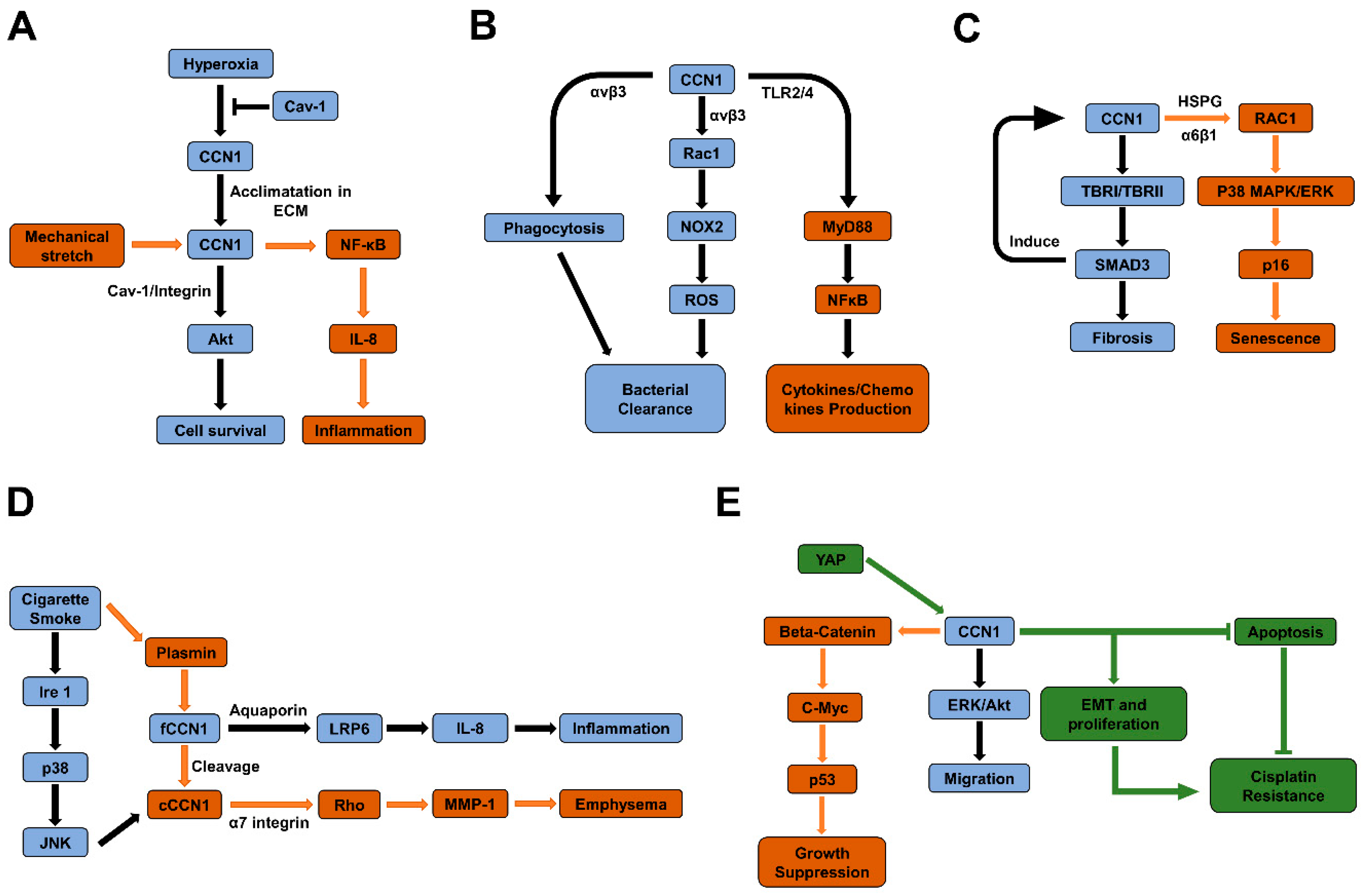
3.1. CCN1 Association with Acute Lung Injury (ALI) and Bronchopulmonary Dysplasia (BPD)
3.2. CCN1 with Chronic Obstructive Pulmonary Disease (COPD)
3.3. CCN1 with Pulmonary Fibrosis
3.4. CCN1 with Pulmonary Hypertension (PH)
3.5. CCN1 with Lung Infection
3.6. CCN1 with Lung Cancer
4. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| CCN1 | Cysteine-rich 61, connective tissue growth factor, and nephroblastoma-1 |
| ALI | Acute lung injury |
| COPD | Chronic obstructive pulmonary disease |
| PAH | Pulmonary arterial hypertension |
| IGFBP | Insulin-like growth factor binding protein |
| vWC | Willebrand factor type C repeat |
| TSP | Thrombospondin type 1 repeat |
| CT | Cysteine knot |
| TGF-β1 | Transforming growth factor β1 |
| FGF2 | Fibroblast growth factor |
| VEGF | Vascular endothelial growth factor |
| GH | Growth hormone |
| PDGF | Platelet-derived growth factor |
| BPD | Bronchopulmonary dysplasia |
| ARDS | Acute respiratory distress syndrome |
| VILI | Ventilator-induced lung injury |
| ROS | Reactive oxygen species |
| ER | Endoplasmic reticulum |
| PASMCs | Pulmonary artery smooth muscle cells |
| LPS | Lipopolysaccharide |
| NTHi | Nontypeable Haemophilus influenzae |
| NSCLC | Non-small cell lung cancer |
| EMT | Epithelial–mesenchymal transition |
| NCTD | Norcantharidin |
| YAP | Yes-associated protein 1 |
| CTGF | Connective tissue growth factor |
References
- O’Brien, T.P.; Yang, G.P.; Sanders, L.; Lau, L.F. Expression of cyr61, a growth factor-inducible immediate-early gene. Mol. Cell. Biol. 1990, 10, 3569–3577. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holbourn, K.P.; Acharya, K.R.; Perbal, B. The CCN family of proteins: Structure-function relationships. Trends Biochem. Sci. 2008, 33, 461–473. [Google Scholar] [CrossRef] [PubMed]
- Perbal, B. CCN proteins: Multifunctional signalling regulators. Lancet 2004, 363, 62–64. [Google Scholar] [CrossRef]
- Chen, C.C.; Lau, L.F. Functions and mechanisms of action of CCN matricellular proteins. Int. J. Biochem. Cell Biol. 2009, 41, 771–783. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lau, L.F. CCN1/CYR61: The very model of a modern matricellular protein. Cell. Mol. Life Sci. 2011, 68, 3149–3163. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bai, T.; Chen, C.C.; Lau, L.F. Matricellular protein CCN1 activates a proinflammatory genetic program in murine macrophages. J. Immunol. 2010, 184, 3223–3232. [Google Scholar] [CrossRef] [Green Version]
- Shimoyama, T.; Hiraoka, S.; Takemoto, M.; Koshizaka, M.; Tokuyama, H.; Tokuyama, T.; Watanabe, A.; Fujimoto, M.; Kawamura, H.; Sato, S.; et al. CCN3 inhibits neointimal hyperplasia through modulation of smooth muscle cell growth and migration. Arter. Thromb. Vasc. Biol. 2010, 30, 675–682. [Google Scholar] [CrossRef] [Green Version]
- Todorovic, V.; Chen, C.C.; Hay, N.; Lau, L.F. The matrix protein CCN1 (CYR61) induces apoptosis in fibroblasts. J. Cell Biol. 2005, 171, 559–568. [Google Scholar] [CrossRef] [Green Version]
- Jun, J.I.; Lau, L.F. The matricellular protein CCN1 induces fibroblast senescence and restricts fibrosis in cutaneous wound healing. Nat. Cell Biol. 2010, 12, 676–685. [Google Scholar] [CrossRef]
- Murphy, M.; Godson, C.; Cannon, S.; Kato, S.; Mackenzie, H.S.; Martin, F.; Brady, H.R. Suppression subtractive hybridization identifies high glucose levels as a stimulus for expression of connective tissue growth factor and other genes in human mesangial cells. J. Biol. Chem. 1999, 274, 5830–5834. [Google Scholar] [CrossRef] [Green Version]
- Twigg, S.M.; Cao, Z.; McLennan, S.V.; Burns, W.C.; Brammar, G.; Forbes, J.M.; Cooper, M.E. Renal connective tissue growth factor induction in experimental diabetes is prevented by aminoguanidine. Endocrinology 2002, 143, 4907–4915. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hughes, J.M.; Kuiper, E.J.; Klaassen, I.; Canning, P.; Stitt, A.W.; Van Bezu, J.; Schalkwijk, C.G.; Van Noorden, C.J.; Schlingemann, R.O. Advanced glycation end products cause increased CCN family and extracellular matrix gene expression in the diabetic rodent retina. Diabetologia 2007, 50, 1089–1098. [Google Scholar] [CrossRef] [Green Version]
- Hilfiker, A.; Hilfiker-Kleiner, D.; Fuchs, M.; Kaminski, K.; Lichtenberg, A.; Rothkotter, H.J.; Schieffer, B.; Drexler, H. Expression of CYR61, an angiogenic immediate early gene, in arteriosclerosis and its regulation by angiotensin II. Circulation 2002, 106, 254–260. [Google Scholar] [CrossRef] [Green Version]
- Cicha, I.; Yilmaz, A.; Klein, M.; Raithel, D.; Brigstock, D.R.; Daniel, W.G.; Goppelt-Struebe, M.; Garlichs, C.D. Connective tissue growth factor is overexpressed in complicated atherosclerotic plaques and induces mononuclear cell chemotaxis in vitro. Arter. Thromb. Vasc. Biol. 2005, 25, 1008–1013. [Google Scholar] [CrossRef]
- Nozawa, K.; Fujishiro, M.; Kawasaki, M.; Kaneko, H.; Iwabuchi, K.; Yanagida, M.; Suzuki, F.; Miyazawa, K.; Takasaki, Y.; Ogawa, H.; et al. Connective tissue growth factor promotes articular damage by increased osteoclastogenesis in patients with rheumatoid arthritis. Arthritis Res. Ther. 2009, 11, R174. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Q.; Wu, J.; Cao, Q.; Xiao, L.; Wang, L.; He, D.; Ouyang, G.; Lin, J.; Shen, B.; Shi, Y.; et al. A critical role of Cyr61 in interleukin-17-dependent proliferation of fibroblast-like synoviocytes in rheumatoid arthritis. Arthritis Rheum. 2009, 60, 3602–3612. [Google Scholar] [CrossRef] [PubMed]
- Lai, D.; Ho, K.C.; Hao, Y.; Yang, X. Taxol resistance in breast cancer cells is mediated by the hippo pathway component TAZ and its downstream transcriptional targets Cyr61 and CTGF. Cancer Res. 2011, 71, 2728–2738. [Google Scholar] [CrossRef] [Green Version]
- Xie, D.; Nakachi, K.; Wang, H.; Elashoff, R.; Koeffler, H.P. Elevated levels of connective tissue growth factor, WISP-1, and CYR61 in primary breast cancers associated with more advanced features. Cancer Res. 2001, 61, 8917–8923. [Google Scholar] [PubMed]
- Kleer, C.G.; Zhang, Y.; Pan, Q.; van Golen, K.L.; Wu, Z.F.; Livant, D.; Merajver, S.D. WISP3 is a novel tumor suppressor gene of inflammatory breast cancer. Oncogene 2002, 21, 3172–3180. [Google Scholar] [CrossRef] [Green Version]
- Chen, P.P.; Li, W.J.; Wang, Y.; Zhao, S.; Li, D.Y.; Feng, L.Y.; Shi, X.L.; Koeffler, H.P.; Tong, X.J.; Xie, D. Expression of Cyr61, CTGF, and WISP-1 correlates with clinical features of lung cancer. PLoS ONE 2007, 2, e534. [Google Scholar] [CrossRef]
- Tong, X.; Xie, D.; O’Kelly, J.; Miller, C.W.; Muller-Tidow, C.; Koeffler, H.P. Cyr61, a member of CCN family, is a tumor suppressor in non-small cell lung cancer. J. Biol. Chem. 2001, 276, 47709–47714. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tian, C.; Zhou, Z.G.; Meng, W.J.; Sun, X.F.; Yu, Y.Y.; Li, L.; Luo, H.Z.; Yang, L.; Zhou, B.; Gu, J. Overexpression of connective tissue growth factor WISP-1 in Chinese primary rectal cancer patients. World J. Gastroenterol. 2007, 13, 3878–3882. [Google Scholar] [CrossRef] [PubMed]
- Ladwa, R.; Pringle, H.; Kumar, R.; West, K. Expression of CTGF and Cyr61 in colorectal cancer. J. Clin. Pathol. 2011, 64, 58–64. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, K.H.; Won, J.H.; Cheng, N.; Lau, L.F. The matricellular protein CCN1 in tissue injury repair. J. Cell Commun. Signal 2018, 12, 273–279. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Y.; Du, X.Y. Functional properties and intracellular signaling of CCN1/Cyr61. J. Cell Biochem. 2007, 100, 1337–1345. [Google Scholar] [CrossRef]
- Di, Y.; Zhang, Y.; Nie, Q.; Chen, X. CCN1/Cyr61-PI3K/AKT signaling promotes retinal neovascularization in oxygen-induced retinopathy. Int. J. Mol. Med. 2015, 36, 1507–1518. [Google Scholar] [CrossRef] [Green Version]
- Hsu, P.L.; Chen, J.S.; Wang, C.Y.; Wu, H.L.; Mo, F.E. Shear-Induced CCN1 Promotes Atheroprone Endothelial Phenotypes and Atherosclerosis. Circulation 2019, 139, 2877–2891. [Google Scholar] [CrossRef]
- Barranco, C. Rheumatoid arthritis: CCN1, a novel RA target? Nat. Rev. Rheumatol. 2016, 12, 561. [Google Scholar] [CrossRef]
- Kim, H.; Son, S.; Shin, I. Role of the CCN protein family in cancer. BMB Rep. 2018, 51, 486–492. [Google Scholar] [CrossRef] [Green Version]
- Henrot, P.; Truchetet, M.E.; Fisher, G.; Taieb, A.; Cario, M. CCN proteins as potential actionable targets in scleroderma. Exp. Dermatol. 2019, 28, 11–18. [Google Scholar] [CrossRef] [Green Version]
- Jun, J.I.; Lau, L.F. Taking aim at the extracellular matrix: CCN proteins as emerging therapeutic targets. Nat. Rev. Drug. Discov. 2011, 10, 945–963. [Google Scholar] [CrossRef] [Green Version]
- You, J.J.; Yang, C.M.; Chen, M.S.; Yang, C.H. Regulation of Cyr61/CCN1 expression by hypoxia through cooperation of c-Jun/AP-1 and HIF-1alpha in retinal vascular endothelial cells. Exp. Eye Res. 2010, 91, 825–836. [Google Scholar] [CrossRef]
- Quan, T.; Qin, Z.; Xu, Y.; He, T.; Kang, S.; Voorhees, J.J.; Fisher, G.J. Ultraviolet irradiation induces CYR61/CCN1, a mediator of collagen homeostasis, through activation of transcription factor AP-1 in human skin fibroblasts. J. Investig. Dermatol. 2010, 130, 1697–1706. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vaidya, R.; Zambrano, R.; Hummler, J.K.; Luo, S.H.; Duncan, M.R.; Young, K.; Lau, L.F.; Wu, S. Recombinant CCN1 prevents hyperoxia-induced lung injury in neonatal rats. Pediatr. Res. 2017, 82, 863–871. [Google Scholar] [CrossRef] [PubMed]
- Grote, K.; Bavendiek, U.; Grothusen, C.; Flach, I.; Hilfiker-Kleiner, D.; Drexler, H.; Schieffer, B. Stretch-inducible expression of the angiogenic factor CCN1 in vascular smooth muscle cells is mediated by Egr-1. J. Biol. Chem. 2004, 279, 55675–55681. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, J.; Zhang, W.; Tan, Z.; Zheng, C.; Tang, Y.; Ke, X.; Zhang, Y.; Liu, Y.; Li, P.; Hu, Q.; et al. Zika virus promotes CCN1 expression via the CaMKIIalpha-CREB pathway in astrocytes. Virulence 2020, 11, 113–131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gueugnon, F.; Thibault, V.C.; Kearley, J.; Petit-Courty, A.; Vallet, A.; Guillon, A.; Si-Tahar, M.; Humbles, A.A.; Courty, Y. Altered expression of the CCN genes in the lungs of mice in response to cigarette smoke exposure and viral and bacterial infections. Gene 2016, 586, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Klein, T.; Eckhard, U.; Dufour, A.; Solis, N.; Overall, C.M. Proteolytic Cleavage-Mechanisms, Function, and “Omic” Approaches for a Near-Ubiquitous Posttranslational Modification. Chem. Rev. 2018, 118, 1137–1168. [Google Scholar] [CrossRef]
- Pendurthi, U.R.; Tran, T.T.; Post, M.; Rao, L.V. Proteolysis of CCN1 by plasmin: Functional implications. Cancer Res. 2005, 65, 9705–9711. [Google Scholar] [CrossRef] [Green Version]
- Grazioli, S.; Gil, S.; An, D.; Kajikawa, O.; Farnand, A.W.; Hanson, J.F.; Birkland, T.; Chen, P.; Duffield, J.; Schnapp, L.M.; et al. CYR61 (CCN1) overexpression induces lung injury in mice. Am. J. Physiol. Lung Cell. Mol. Physiol. 2015, 308, L759–L765. [Google Scholar] [CrossRef] [Green Version]
- Li, Z.Q.; Wu, W.R.; Zhao, C.; Zhao, C.; Zhang, X.L.; Yang, Z.; Pan, J.; Si, W.K. CCN1/Cyr61 enhances the function of hepatic stellate cells in promoting the progression of hepatocellular carcinoma. Int. J. Mol. Med. 2018, 41, 1518–1528. [Google Scholar] [CrossRef] [PubMed]
- Mo, F.E.; Lau, L.F. The matricellular protein CCN1 is essential for cardiac development. Circ. Res. 2006, 99, 961–969. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sawai, K.; Mukoyama, M.; Mori, K.; Kasahara, M.; Koshikawa, M.; Yokoi, H.; Yoshioka, T.; Ogawa, Y.; Sugawara, A.; Nishiyama, H.; et al. Expression of CCN1 (CYR61) in developing, normal, and diseased human kidney. Am. J. Physiol. Physiol. 2007, 293, F1363–F1372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moon, H.G.; Kim, S.H.; Gao, J.; Quan, T.; Qin, Z.; Osorio, J.C.; Rosas, I.O.; Wu, M.; Tesfaigzi, Y.; Jin, Y. CCN1 secretion and cleavage regulate the lung epithelial cell functions after cigarette smoke. Am. J. Physiol. Lung Cell. Mol. Physiol. 2014, 307, L326–L337. [Google Scholar] [CrossRef] [Green Version]
- Shi, L.; Dong, N.; Ji, D.; Huang, X.; Ying, Z.; Wang, X.; Chen, C. Lipopolysaccharide-induced CCN1 production enhances interleukin-6 secretion in bronchial epithelial cells. Cell Biol. Toxicol. 2018, 34, 39–49. [Google Scholar] [CrossRef]
- Quan, T.; Qin, Z.; Robichaud, P.; Voorhees, J.J.; Fisher, G.J. CCN1 contributes to skin connective tissue aging by inducing age-associated secretory phenotype in human skin dermal fibroblasts. J. Cell Commun. Signal. 2011, 5, 201–207. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Y.; Li, H.; Liang, X.; Du, H.; Suo, Y.; Chen, H.; Liu, W.; Duan, R.; Huang, X.; Li, Q. The CCN1 (CYR61) protein promotes skin growth by enhancing epithelial-mesenchymal transition during skin expansion. J. Cell. Mol. Med. 2020, 24, 1460–1473. [Google Scholar] [CrossRef]
- Malik, A.R.; Liszewska, E.; Jaworski, J. Matricellular proteins of the Cyr61/CTGF/NOV (CCN) family and the nervous system. Front. Cell. Neurosci. 2015, 9, 237. [Google Scholar] [CrossRef] [Green Version]
- Kim, K.H.; Chen, C.C.; Monzon, R.I.; Lau, L.F. Matricellular protein CCN1 promotes regression of liver fibrosis through induction of cellular senescence in hepatic myofibroblasts. Mol. Cell. Biol. 2013, 33, 2078–2090. [Google Scholar] [CrossRef] [Green Version]
- Muramatsu, Y.; Tsujie, M.; Kohda, Y.; Pham, B.; Perantoni, A.O.; Zhao, H.; Jo, S.K.; Yuen, P.S.; Craig, L.; Hu, X.; et al. Early detection of cysteine rich protein 61 (CYR61, CCN1) in urine following renal ischemic reperfusion injury. Kidney Int. 2002, 62, 1601–1610. [Google Scholar] [CrossRef] [Green Version]
- Su, J.L.; Chiou, J.; Tang, C.H.; Zhao, M.; Tsai, C.H.; Chen, P.S.; Chang, Y.W.; Chien, M.H.; Peng, C.Y.; Hsiao, M.; et al. CYR61 regulates BMP-2-dependent osteoblast differentiation through the {alpha}v{beta}3 integrin/integrin-linked kinase/ERK pathway. J. Biol. Chem. 2010, 285, 31325–31336. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, S.F.; Grigoryev, D.N.; Taylor, A.D.; Nonas, S.; Sammani, S.; Ye, S.Q.; Garcia, J.G. Bioinformatic identification of novel early stress response genes in rodent models of lung injury. Am. J. Physiol. Lung Cell. Mol. Physiol. 2005, 289, L468–L477. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Kelly, J.; Chung, A.; Lemp, N.; Chumakova, K.; Yin, D.; Wang, H.J.; Said, J.; Gui, D.; Miller, C.W.; Karlan, B.Y.; et al. Functional domains of CCN1 (Cyr61) regulate breast cancer progression. Int. J. Oncol. 2008, 33, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Holloway, S.E.; Beck, A.W.; Girard, L.; Jaber, M.R.; Barnett, C.C., Jr.; Brekken, R.A.; Fleming, J.B. Increased expression of Cyr61 (CCN1) identified in peritoneal metastases from human pancreatic cancer. J. Am. Coll. Surg. 2005, 200, 371–377. [Google Scholar] [CrossRef]
- Emre, Y.; Imhof, B.A. Matricellular protein CCN1/CYR61: A new player in inflammation and leukocyte trafficking. Semin. Immunopathol. 2014, 36, 253–259. [Google Scholar] [CrossRef]
- Chai, J.; Norng, M.; Modak, C.; Reavis, K.M.; Mouazzen, W.; Pham, J. CCN1 induces a reversible epithelial-mesenchymal transition in gastric epithelial cells. Lab. Investig. 2010, 90, 1140–1151. [Google Scholar] [CrossRef] [Green Version]
- You, J.J.; Yang, C.H.; Chen, M.S.; Yang, C.M. Cysteine-rich 61, a member of the CCN family, as a factor involved in the pathogenesis of proliferative diabetic retinopathy. Investig. Ophthalmol. Vis. Sci. 2009, 50, 3447–3455. [Google Scholar] [CrossRef]
- Chintalapudi, M.R.; Markiewicz, M.; Kose, N.; Dammai, V.; Champion, K.J.; Hoda, R.S.; Trojanowska, M.; Hsu, T. Cyr61/CCN1 and CTGF/CCN2 mediate the proangiogenic activity of VHL-mutant renal carcinoma cells. Carcinogenesis 2008, 29, 696–703. [Google Scholar] [CrossRef] [Green Version]
- Kim, K.H.; Chen, C.C.; Alpini, G.; Lau, L.F. CCN1 induces hepatic ductular reaction through integrin alphavbeta(5)-mediated activation of NF-kappaB. J. Clin. Investig. 2015, 125, 1886–1900. [Google Scholar] [CrossRef] [Green Version]
- Long, Q.Z.; Zhou, M.; Liu, X.G.; Du, Y.F.; Fan, J.H.; Li, X.; He, D.L. Interaction of CCN1 with alphavbeta3 integrin induces P-glycoprotein and confers vinblastine resistance in renal cell carcinoma cells. Anticancer Drugs 2013, 24, 810–817. [Google Scholar] [CrossRef]
- Chen, C.C.; Young, J.L.; Monzon, R.I.; Chen, N.; Todorovic, V.; Lau, L.F. Cytotoxicity of TNFalpha is regulated by integrin-mediated matrix signaling. EMBO J. 2007, 26, 1257–1267. [Google Scholar] [CrossRef]
- Mo, F.E.; Muntean, A.G.; Chen, C.C.; Stolz, D.B.; Watkins, S.C.; Lau, L.F. CYR61 (CCN1) is essential for placental development and vascular integrity. Mol. Cell. Biol. 2002, 22, 8709–8720. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, M.H.; Kim, A.K.; Manandhar, S.; Oh, S.Y.; Jang, G.H.; Kang, L.; Lee, D.W.; Hyeon, D.Y.; Lee, S.H.; Lee, H.E.; et al. CCN1 interlinks integrin and hippo pathway to autoregulate tip cell activity. Elife 2019, 8. [Google Scholar] [CrossRef] [PubMed]
- Lau, L.F. Cell surface receptors for CCN proteins. J. Cell Commun. Signal. 2016, 10, 121–127. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jun, J.I.; Lau, L.F. CCN1 is an opsonin for bacterial clearance and a direct activator of Toll-like receptor signaling. Nat. Commun. 2020, 11, 1242. [Google Scholar] [CrossRef] [PubMed]
- Walsh, C.T.; Stupack, D.; Brown, J.H. G protein-coupled receptors go extracellular: RhoA integrates the integrins. Mol. Interv. 2008, 8, 165–173. [Google Scholar] [CrossRef]
- Grzeszkiewicz, T.M.; Kirschling, D.J.; Chen, N.; Lau, L.F. CYR61 stimulates human skin fibroblast migration through Integrin alpha vbeta 5 and enhances mitogenesis through integrin alpha vbeta 3, independent of its carboxyl-terminal domain. J. Biol. Chem. 2001, 276, 21943–21950. [Google Scholar] [CrossRef] [Green Version]
- Rother, M.; Krohn, S.; Kania, G.; Vanhoutte, D.; Eisenreich, A.; Wang, X.; Westermann, D.; Savvatis, K.; Dannemann, N.; Skurk, C.; et al. Matricellular signaling molecule CCN1 attenuates experimental autoimmune myocarditis by acting as a novel immune cell migration modulator. Circulation 2010, 122, 2688–2698. [Google Scholar] [CrossRef] [Green Version]
- Leu, S.J.; Lam, S.C.; Lau, L.F. Pro-angiogenic activities of CYR61 (CCN1) mediated through integrins alphavbeta3 and alpha6beta1 in human umbilical vein endothelial cells. J. Biol. Chem. 2002, 277, 46248–46255. [Google Scholar] [CrossRef] [Green Version]
- Jin, Y.; Kim, H.P.; Ifedigbo, E.; Lau, L.F.; Choi, A.M. Cyr61 protects against hyperoxia-induced cell death via Akt pathway in pulmonary epithelial cells. Am. J. Respir. Cell Mol. Biol. 2005, 33, 297–302. [Google Scholar] [CrossRef]
- Moon, H.G.; Qin, Z.; Quan, T.; Xie, L.; Dela Cruz, C.S.; Jin, Y. Matrix protein CCN1 induced by bacterial DNA and CpG ODN limits lung inflammation and contributes to innate immune homeostasis. Mucosal Immunol. 2015, 8, 243–253. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.G.; Zheng, Y.; An, C.H.; Kim, Y.K.; Jin, Y. CCN1 secretion induced by cigarette smoking extracts augments IL-8 release from bronchial epithelial cells. PLoS ONE 2013, 8, e68199. [Google Scholar] [CrossRef] [PubMed]
- Brigstock, D.R. The connective tissue growth factor/cysteine-rich 61/nephroblastoma overexpressed (CCN) family. Endocr. Rev. 1999, 20, 189–206. [Google Scholar] [PubMed] [Green Version]
- Confalonieri, M.; Salton, F.; Fabiano, F. Acute respiratory distress syndrome. Eur. Respir. Rev. 2017, 26, 160116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thompson, B.T.; Chambers, R.C.; Liu, K.D. Acute Respiratory Distress Syndrome. N. Engl. J. Med. 2017, 377, 562–572. [Google Scholar] [CrossRef]
- Phua, J.; Badia, J.R.; Adhikari, N.K.; Friedrich, J.O.; Fowler, R.A.; Singh, J.M.; Scales, D.C.; Stather, D.R.; Li, A.; Jones, A.; et al. Has mortality from acute respiratory distress syndrome decreased over time?: A systematic review. Am. J. Respir. Crit. Care Med. 2009, 179, 220–227. [Google Scholar] [CrossRef] [Green Version]
- Perkowski, S.; Sun, J.; Singhal, S.; Santiago, J.; Leikauf, G.D.; Albelda, S.M. Gene expression profiling of the early pulmonary response to hyperoxia in mice. Am. J. Respir. Cell Mol. Biol. 2003, 28, 682–696. [Google Scholar] [CrossRef]
- Shimada, I.; Matsui, K.; Brinkmann, B.; Hohoff, C.; Hiraga, K.; Tabuchi, Y.; Takasaki, I.; Kato, I.; Kawaguchi, H.; Takasawa, K.; et al. Novel transcript profiling of diffuse alveolar damage induced by hyperoxia exposure in mice: Normalization by glyceraldehyde 3-phosphate dehydrogenase. Int. J. Leg. Med. 2008, 122, 373–383. [Google Scholar] [CrossRef] [Green Version]
- Dolinay, T.; Kaminski, N.; Felgendreher, M.; Kim, H.P.; Reynolds, P.; Watkins, S.C.; Karp, D.; Uhlig, S.; Choi, A.M. Gene expression profiling of target genes in ventilator-induced lung injury. Physiol. Genom. 2006, 26, 68–75. [Google Scholar] [CrossRef]
- Zhang, Y.; Guf, P.; Yao, S.L.; Yang, D.; Lv, Y.; Ding, D.F. Stretch-induced Expression of CYR61 Increases the Secretion of IL-8 in A549 Cells via the NF-kappabeta/lkappabeta Pathway. Curr. Med. Sci. 2018, 38, 672–678. [Google Scholar] [CrossRef]
- Kurundkar, A.R.; Kurundkar, D.; Rangarajan, S.; Locy, M.L.; Zhou, Y.; Liu, R.M.; Zmijewski, J.; Thannickal, V.J. The matricellular protein CCN1 enhances TGF-beta1/SMAD3-dependent profibrotic signaling in fibroblasts and contributes to fibrogenic responses to lung injury. FASEB J. 2016, 30, 2135–2150. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jin, Y.; Kim, H.P.; Cao, J.; Zhang, M.; Ifedigbo, E.; Choi, A.M. Caveolin-1 regulates the secretion and cytoprotection of Cyr61 in hyperoxic cell death. FASEB J. 2009, 23, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Zemans, R.L.; McClendon, J.; Aschner, Y.; Briones, N.; Young, S.K.; Lau, L.F.; Kahn, M.; Downey, G.P. Role of beta-catenin-regulated CCN matricellular proteins in epithelial repair after inflammatory lung injury. Am. J. Physiol. Lung Cell. Mol. Physiol. 2013, 304, L415–L427. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wallace, M.J.; Probyn, M.E.; Zahra, V.A.; Crossley, K.; Cole, T.J.; Davis, P.G.; Morley, C.J.; Hooper, S.B. Early biomarkers and potential mediators of ventilation-induced lung injury in very preterm lambs. Respir. Res. 2009, 10, 19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adeloye, D.; Chua, S.; Lee, C.; Basquill, C.; Papana, A.; Theodoratou, E.; Nair, H.; Gasevic, D.; Sridhar, D.; Campbell, H.; et al. Global and regional estimates of COPD prevalence: Systematic review and meta-analysis. J. Glob. Health 2015, 5, 020415. [Google Scholar] [CrossRef]
- Celli, B.R.; Wedzicha, J.A. Update on Clinical Aspects of Chronic Obstructive Pulmonary Disease. N. Engl. J. Med. 2019, 381, 1257–1266. [Google Scholar] [CrossRef] [Green Version]
- Ning, W.; Li, C.J.; Kaminski, N.; Feghali-Bostwick, C.A.; Alber, S.M.; Di, Y.P.; Otterbein, S.L.; Song, R.; Hayashi, S.; Zhou, Z.; et al. Comprehensive gene expression profiles reveal pathways related to the pathogenesis of chronic obstructive pulmonary disease. Proc. Natl. Acad. Sci. USA 2004, 101, 14895–14900. [Google Scholar] [CrossRef] [Green Version]
- Du, Y.; Ding, Y.; Chen, X.; Mei, Z.; Ding, H.; Wu, Y.; Jie, Z. MicroRNA-181c inhibits cigarette smoke-induced chronic obstructive pulmonary disease by regulating CCN1 expression. Respir. Res. 2017, 18, 155. [Google Scholar] [CrossRef] [Green Version]
- Richeldi, L.; Collard, H.R.; Jones, M.G. Idiopathic pulmonary fibrosis. Lancet 2017, 389, 1941–1952. [Google Scholar] [CrossRef]
- Kulkarni, T.; Kurundkar, A.R.; Kim, Y.I.; de Andrade, J.; Luckhardt, T.; Thannickal, V.J. The senescence-associated matricellular protein CCN1 in plasma of human subjects with idiopathic pulmonary fibrosis. Respir. Med. 2020, 161, 105821. [Google Scholar] [CrossRef]
- Simonneau, G.; Montani, D.; Celermajer, D.S.; Denton, C.P.; Gatzoulis, M.A.; Krowka, M.; Williams, P.G.; Souza, R. Haemodynamic definitions and updated clinical classification of pulmonary hypertension. Eur. Respir. J. 2019, 53, 1801913. [Google Scholar] [CrossRef] [PubMed]
- Hoeper, M.M.; Bogaard, H.J.; Condliffe, R.; Frantz, R.; Khanna, D.; Kurzyna, M.; Langleben, D.; Manes, A.; Satoh, T.; Torres, F.; et al. Definitions and diagnosis of pulmonary hypertension. J. Am. Coll. Cardiol. 2013, 62, D42–D50. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, S.J.; Zhang, M.; Hu, K.; Lin, L.; Zhang, D.; Jin, Y. CCN1 suppresses pulmonary vascular smooth muscle contraction in response to hypoxia. Pulm. Circ. 2015, 5, 716–722. [Google Scholar] [CrossRef] [Green Version]
- Gao, L.; Fan, Y.; Hao, Y.; Yuan, P.; Liu, D.; Jing, Z.; Zhang, Z. Cysteine-rich 61 (Cyr61) upregulated in pulmonary arterial hypertension promotes the proliferation of pulmonary artery smooth muscle cells. Int. J. Med. Sci. 2017, 14, 820–828. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fan, Y.; Zhao, J.; Qian, J.; Hao, Y.; Wang, Q.; Gao, L.; Li, M.; Zeng, X.; Zhang, Z. Cysteine-rich protein 61 as a novel biomarker in systemic lupus erythematosus-associated pulmonary arterial hypertension. Clin. Exp. Rheumatol. 2019, 37, 623–632. [Google Scholar] [PubMed]
- Cookson, W.; Cox, M.J.; Moffatt, M.F. New opportunities for managing acute and chronic lung infections. Nat. Rev. Microbiol. 2018, 16, 111–120. [Google Scholar] [CrossRef]
- Hviid, C.V.; Erdem, J.S.; Kunke, D.; Ahmed, S.M.; Kjeldsen, S.F.; Wang, Y.Y.; Attramadal, H.; Aasen, A.O. The matri-cellular proteins ‘cysteine-rich, angiogenic-inducer, 61’ and ‘connective tissue growth factor’ are regulated in experimentally-induced sepsis with multiple organ dysfunction. Innate Immun. 2012, 18, 717–726. [Google Scholar] [CrossRef] [Green Version]
- Hakansson, H.F.; Smailagic, A.; Brunmark, C.; Miller-Larsson, A.; Lal, H. Altered lung function relates to inflammation in an acute LPS mouse model. Pulm. Pharmacol. Ther. 2012, 25, 399–406. [Google Scholar] [CrossRef]
- Cohen, T.S.; Jones-Nelson, O.; Hotz, M.; Cheng, L.; Miller, L.S.; Suzich, J.; Stover, C.K.; Sellman, B.R.S. aureus blocks efferocytosis of neutrophils by macrophages through the activity of its virulence factor alpha toxin. Sci. Rep. 2016, 6, 35466. [Google Scholar] [CrossRef]
- Jemal, A.; Bray, F.; Center, M.M.; Ferlay, J.; Ward, E.; Forman, D. Global cancer statistics. CA Cancer J. Clin. 2011, 61, 69–90. [Google Scholar] [CrossRef] [Green Version]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef]
- Mori, A.; Desmond, J.C.; Komatsu, N.; O’Kelly, J.; Miller, C.W.; Legaspi, R.; Marchevsky, A.M.; McKenna, R.J., Jr.; Koeffler, H.P. CYR61: A new measure of lung cancer outcome. Cancer Investig. 2007, 25, 738–741. [Google Scholar] [CrossRef]
- Molina, J.R.; Yang, P.; Cassivi, S.D.; Schild, S.E.; Adjei, A.A. Non-small cell lung cancer: Epidemiology, risk factors, treatment, and survivorship. Mayo Clin. Proc. 2008, 83, 584–594. [Google Scholar] [CrossRef]
- Tong, X.; O’Kelly, J.; Xie, D.; Mori, A.; Lemp, N.; McKenna, R.; Miller, C.W.; Koeffler, H.P. Cyr61 suppresses the growth of non-small-cell lung cancer cells via the beta-catenin-c-myc-p53 pathway. Oncogene 2004, 23, 4847–4855. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Yuan, N.; Lin, L.; Yin, L.; Qu, Y. Targeting cysteine-rich angiogenic inducer-61 by antibody immunotherapy suppresses growth and migration of non-small cell lung cancer. Exp. Ther. Med. 2018, 16, 730–738. [Google Scholar] [CrossRef]
- Jandova, J.; Beyer, T.E.; Meuillet, E.J.; Watts, G.S. The matrix protein CCN1/CYR61 is required for alpha(V)beta5-mediated cancer cell migration. Cell Biochem. Funct. 2012, 30, 687–695. [Google Scholar] [CrossRef]
- Sabile, A.A.; Arlt, M.J.; Muff, R.; Bode, B.; Langsam, B.; Bertz, J.; Jentzsch, T.; Puskas, G.J.; Born, W.; Fuchs, B. Cyr61 expression in osteosarcoma indicates poor prognosis and promotes intratibial growth and lung metastasis in mice. J. Bone Miner. Res. 2012, 27, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Fromigue, O.; Hamidouche, Z.; Vaudin, P.; Lecanda, F.; Patino, A.; Barbry, P.; Mari, B.; Marie, P.J. CYR61 downregulation reduces osteosarcoma cell invasion, migration, and metastasis. J. Bone Miner. Res. 2011, 26, 1533–1542. [Google Scholar] [CrossRef] [PubMed]
- Habel, N.; Vilalta, M.; Bawa, O.; Opolon, P.; Blanco, J.; Fromigue, O. Cyr61 silencing reduces vascularization and dissemination of osteosarcoma tumors. Oncogene 2015, 34, 3207–3213. [Google Scholar] [CrossRef]
- Huang, Y.T.; Lan, Q.; Lorusso, G.; Duffey, N.; Ruegg, C. The matricellular protein CYR61 promotes breast cancer lung metastasis by facilitating tumor cell extravasation and suppressing anoikis. Oncotarget 2017, 8, 9200–9215. [Google Scholar] [CrossRef] [Green Version]
- Jin, D.; Wu, Y.; Shao, C.; Gao, Y.; Wang, D.; Guo, J. Norcantharidin reverses cisplatin resistance and inhibits the epithelial mesenchymal transition of human nonsmall lung cancer cells by regulating the YAP pathway. Oncol. Rep. 2018, 40, 609–620. [Google Scholar] [PubMed]
- Zhang, D.; Zhang, Y.; Cai, Z.; Tu, Y.; Hu, Z. Dexamethasone and lenvatinib inhibit migration and invasion of non-small cell lung cancer by regulating EKR/AKT and VEGF signal pathways. Exp. Ther. Med. 2020, 19, 762–770. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hirschfeld, M.; zur Hausen, A.; Bettendorf, H.; Jager, M.; Stickeler, E. Alternative splicing of Cyr61 is regulated by hypoxia and significantly changed in breast cancer. Cancer Res. 2009, 69, 2082–2090. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yan, L.; Lee, S.; Lazzaro, D.R.; Aranda, J.; Grant, M.B.; Chaqour, B. Single and Compound Knock-outs of MicroRNA (miRNA)-155 and Its Angiogenic Gene Target CCN1 in Mice Alter Vascular and Neovascular Growth in the Retina via Resident Microglia. J. Biol. Chem. 2015, 290, 23264–23281. [Google Scholar] [CrossRef] [Green Version]
- Niwa, Y.; Suzuki, T.; Dohmae, N.; Simizu, S. O-Fucosylation of CCN1 is required for its secretion. FEBS Lett. 2015, 589, 3287–3293. [Google Scholar] [CrossRef] [Green Version]

| Tissues/Organs | Cell Types | References |
|---|---|---|
| Lung | Epithelial cell | [44,45] |
| Skin | Fibroblasts | [46,47] |
| Nervous system | Neurons | [48] |
| Liver | Hepatic stellate cells, fibroblasts | [41,49] |
| Kidney | HUVEC, podocytes | [43,50] |
| Bone | Osteoblast cell | [51] |
| Heart | Endocardial cells, mesenchymal cells | [42] |
| Muscle | Muscle progenitor cells | [52] |
| Intestine | Intestinal epithelial cell | [24] |
| Lymph node | Cancer cells | [53] |
| Pancreas | Epithelial | [54] |
| Spleen | Spleen dendritic cells | [50,55] |
| Stomach | Epithelial | [56] |
| Eye | Chorioretinal vessel endothelial cells | [57] |
| Cell Types Involved | Biological Functions | Membrane Receptors | References |
|---|---|---|---|
| Fibroblast, smooth muscle cells | Cell adhesion | Aα6β1 integrin and HSPGs | [4,57] |
| Fibroblast | Apoptosis | Aα6β1 integrin | [61] |
| Endothelial cell | Cell survival | αVβ3 integrin | [57,62] |
| Astrocytoma cells | Proliferation | α5, α6 and β1 integrins | [66] |
| Osteoblast | Differentiation | αVβ3 integrin | [51] |
| Fibroblast | Migration | αVβ5 integrin | [4,67] |
| Endothelial cell | Angiogenesis | αVβ3 integrin | [31,55] |
| Macrophages | Inflammation and bacterial clearance | αMβ2 integrin, αVβ3 integrins and TLR2/4 | [6,65,68] |
| Endothelial cell | Survival | αVβ3 integrin | [4,69] |
| Fibroblasts | Senescence | α6β1 integrin and HSPGs | [4,8] |
| Epithelial cell | Cell death | α7 integrin | [44,70] |
| Epithelial cell | Innate immune homeostasis | αVβ6 integrin and LRP6 | [71,72] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhu, Y.; Almuntashiri, S.; Han, Y.; Wang, X.; Somanath, P.R.; Zhang, D. The Roles of CCN1/CYR61 in Pulmonary Diseases. Int. J. Mol. Sci. 2020, 21, 7810. https://doi.org/10.3390/ijms21217810
Zhu Y, Almuntashiri S, Han Y, Wang X, Somanath PR, Zhang D. The Roles of CCN1/CYR61 in Pulmonary Diseases. International Journal of Molecular Sciences. 2020; 21(21):7810. https://doi.org/10.3390/ijms21217810
Chicago/Turabian StyleZhu, Yin, Sultan Almuntashiri, Yohan Han, Xiaoyun Wang, Payaningal R. Somanath, and Duo Zhang. 2020. "The Roles of CCN1/CYR61 in Pulmonary Diseases" International Journal of Molecular Sciences 21, no. 21: 7810. https://doi.org/10.3390/ijms21217810
APA StyleZhu, Y., Almuntashiri, S., Han, Y., Wang, X., Somanath, P. R., & Zhang, D. (2020). The Roles of CCN1/CYR61 in Pulmonary Diseases. International Journal of Molecular Sciences, 21(21), 7810. https://doi.org/10.3390/ijms21217810






