Multiple-Purpose Connectivity Map Analysis Reveals the Benefits of Esculetin to Hyperuricemia and Renal Fibrosis
Abstract
1. Introduction
2. Results
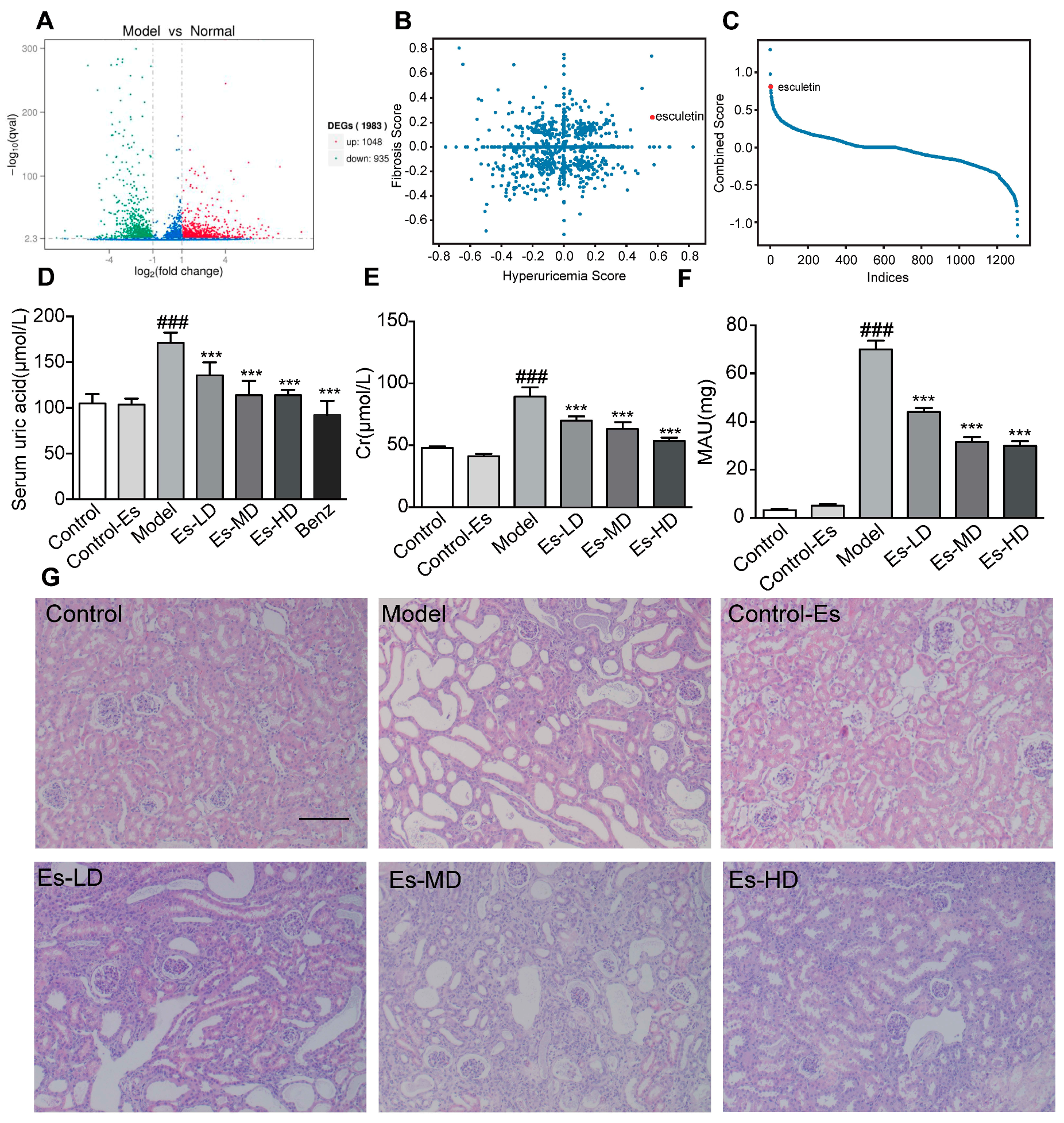
2.1. Esculetin Is the Top Scoring Candidate Compound
2.2. Esculetin Reduces SUA Levels in a Dose-Dependent Manner
2.3. Esculetin Inhibits XO Expression and Activity
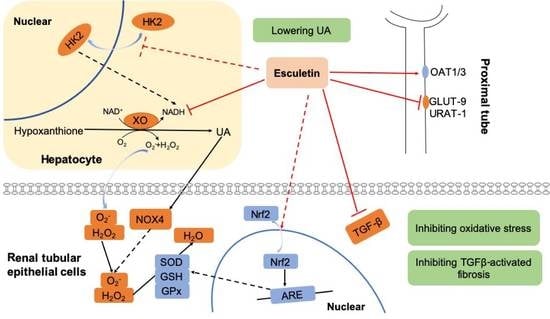
2.4. Esculetin Modulates the Expression of Renal UA Transporters
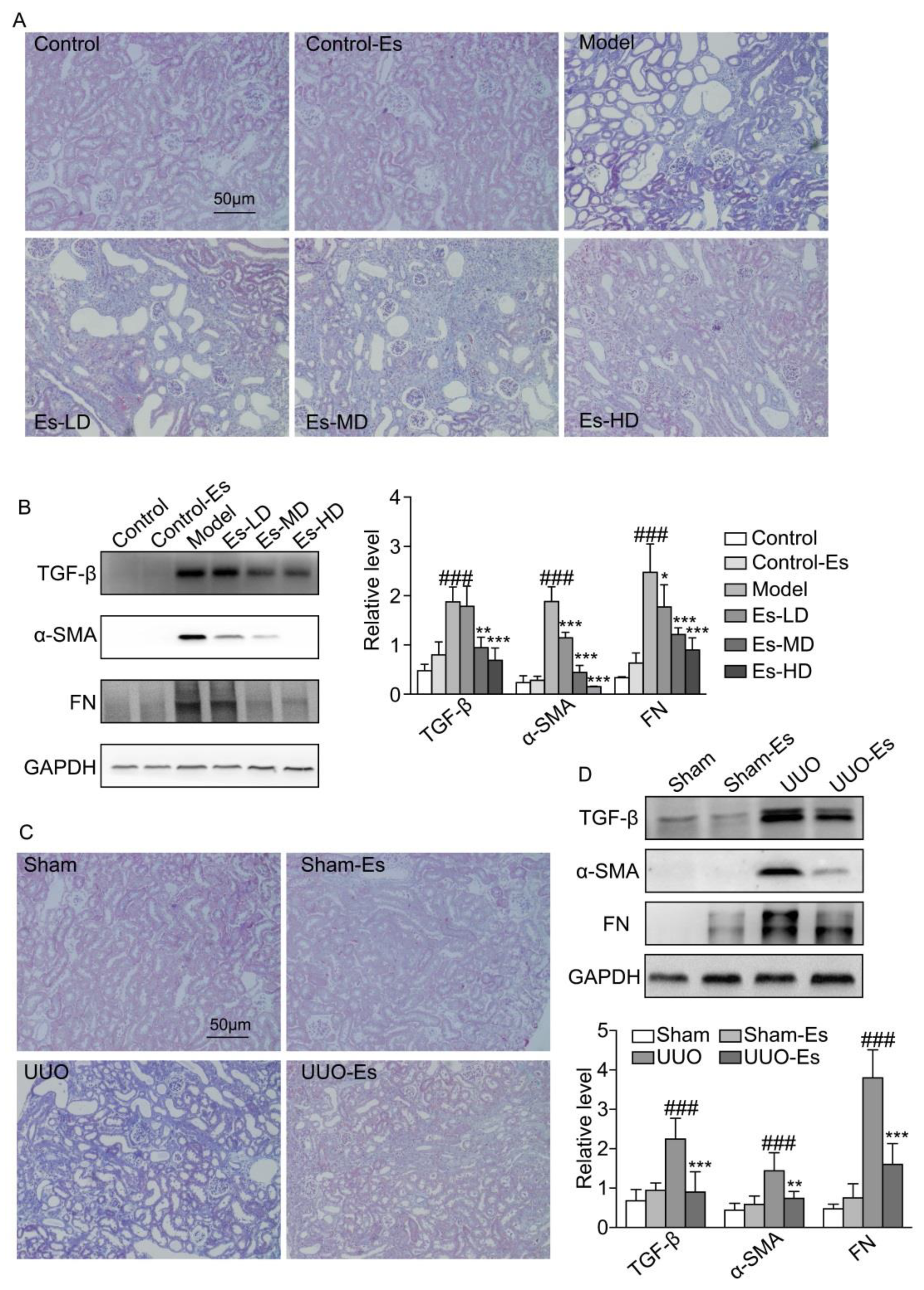
2.5. Esculetin Suppresses Renal Fibrosis in HUA and UUO Mice
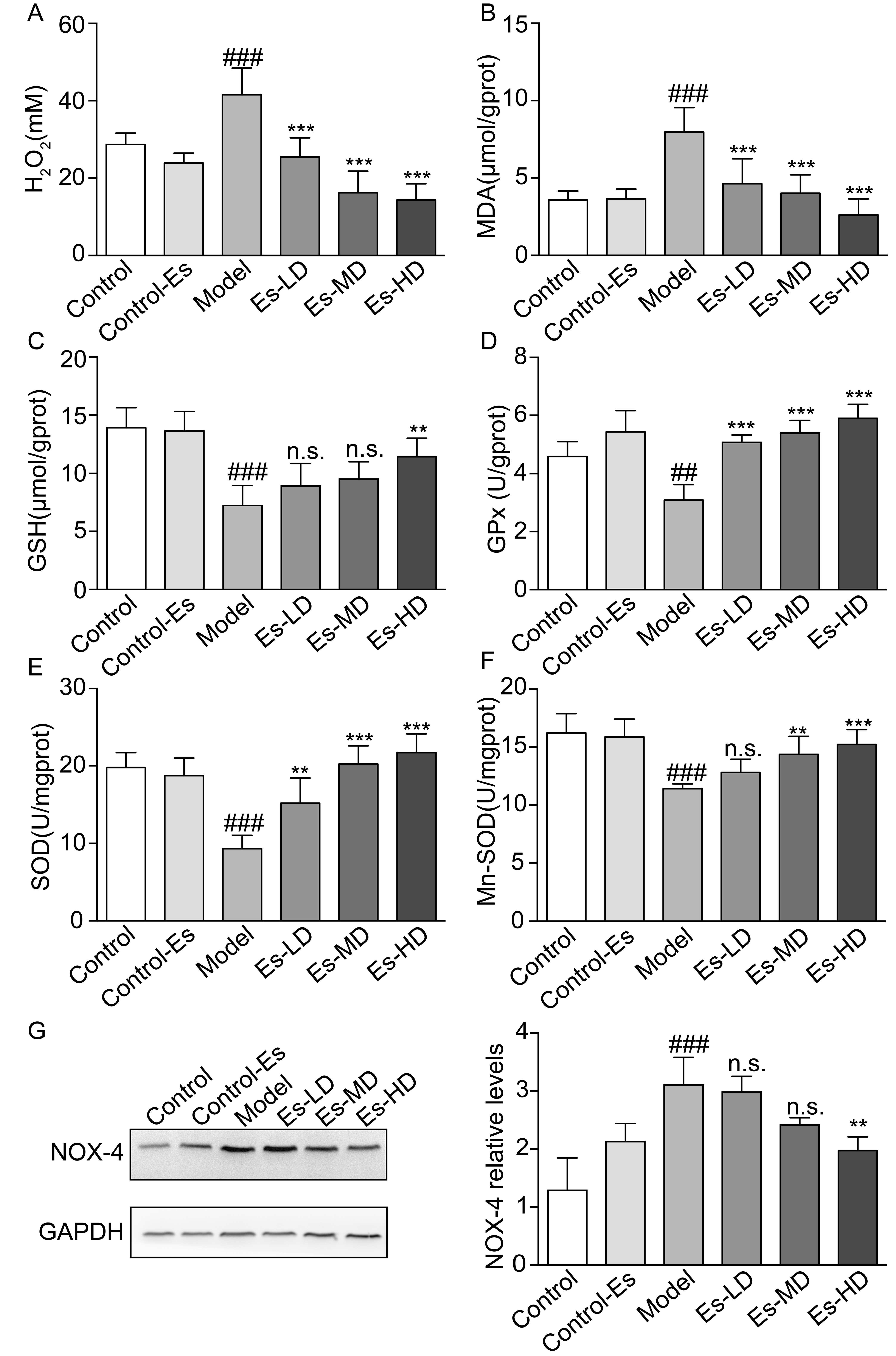
2.6. Esculetin Reduces XO-Dependent and NADPH Oxidase-Dependent Renal Oxidative Stress in HUA Mice
2.7. Esculetin Improves Oxidative Stress Dysfunction and Enhances Nuclear Nrf2 Translocation in NRK-52E Cells
2.8. Esculetin Down-Regulates HK2 in UA-Induced HEPG2 Cells
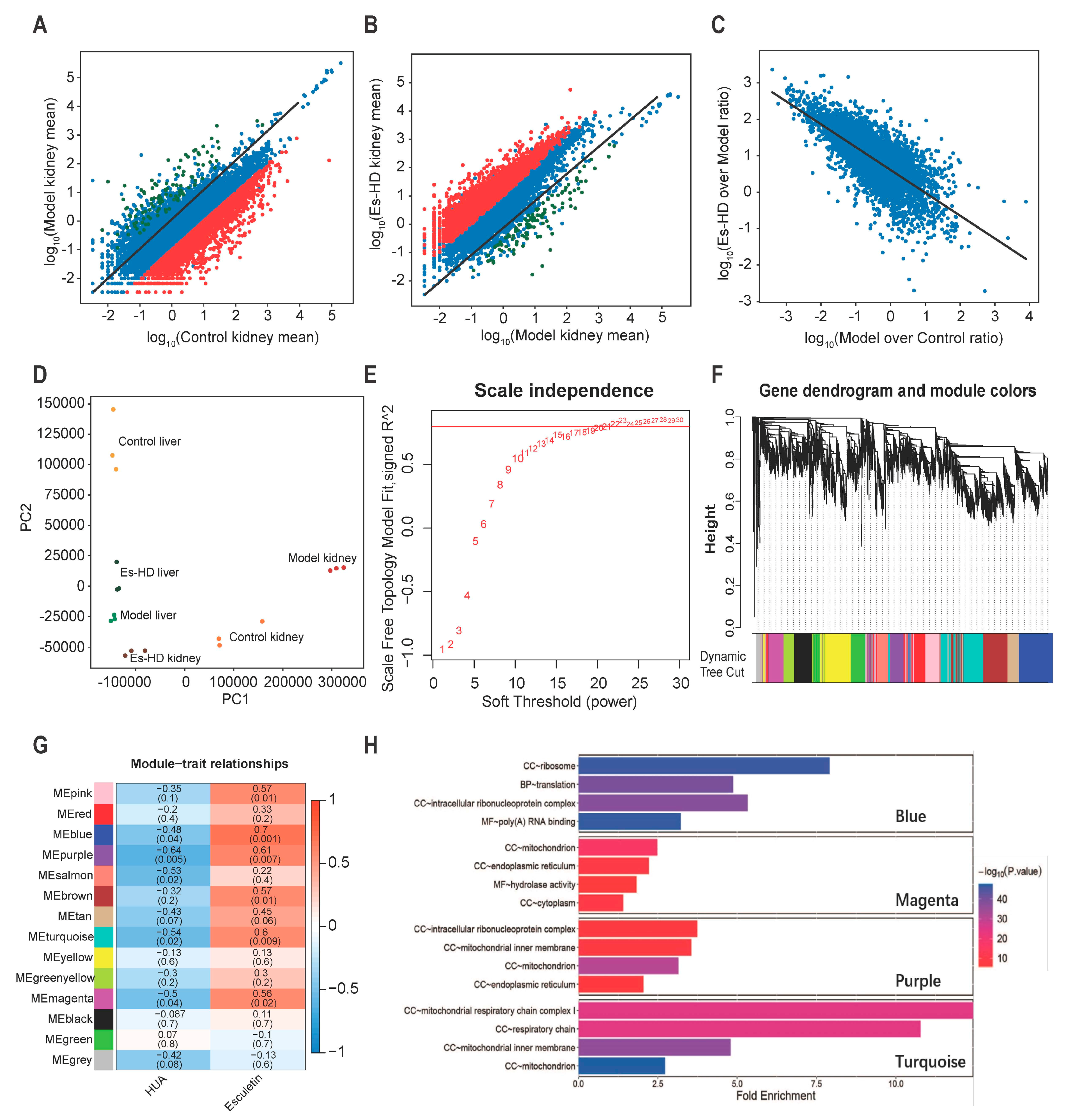
2.9. Esculetin Reverses the Transcriptome Alterations
3. Discussion
4. Materials and Methods
4.1. Connectivity Map Analysis
4.2. WGCNA
4.3. Chemical Agents
4.4. Animal Experiments and Ethics
4.5. HUA Mouse Models
4.6. UUO Model
4.7. Assessment of UA, Renal Function, Liver Function and Other Biochemical Indices
4.8. SPR Biosensor Analysis
4.9. XO Activity and IC50
4.10. Cell Culture
4.11. Western Blots Analysis
4.12. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| HUA | Hyperuricemia |
| CKD | chronic kidney disease |
| SUA | serum uric acid |
| HN | hyperuricemic nephropathy |
| UA | uric acid |
| CMAP | connectivity map |
| UUO | unilateral ureteral occlusion |
| XO | xanthine oxidase |
| HK2 | hexokinase-2 |
| GFR | glomerular filtration rate |
| ESRD | end-stage renal disease |
| RNA-Seq | RNA sequencing |
| Cr | creatinine |
| MAU | proteinuria |
| BUN | urea nitrogen |
| RU | response units |
| MDA | malondialdehyde |
| SOD | superoxide dismutase |
| GSH | reduced glutathione |
| GPx | glutathione peroxidase |
| Nox-4 | NADPH oxidase 4 |
| ROS | reactive oxygen species |
| Bar | Bardoxolone methyl |
| Keap1 | Kelch-like ECH associated protein 1 |
| Nrf2 | Nuclear factor-erythroid-2 related factor |
| ARE | antioxidant response element |
| AST | aspartate aminotransferase |
| ALT | alanine transaminase |
References
- Bardin, T.; Richette, P. Impact of comorbidities on gout and hyperuricaemia: An update on prevalence and treatment options. BMC Med. 2017, 15, 123. [Google Scholar] [CrossRef] [PubMed]
- Murray, K.; Burkard, T. Hyperuricemia, gout and cardiovascular diseases. Ther. Umsch. 2016, 73, 141–146. (In German) [Google Scholar] [CrossRef] [PubMed]
- Obermayr, R.P.; Temml, C.; Gutjahr, G.; Knechtelsdorfer, M.; Oberbauer, R.; Klauser-Braun, R. Elevated uric acid increases the risk for kidney disease. J. Am. Soc. Nephrol. 2008, 19, 2407–2413. [Google Scholar] [CrossRef] [PubMed]
- Sah, O.S.; Qing, Y.X. Associations between hyperuricemia and chronic kidney disease: A review. Nephrourol. Mon. 2015, 7, e27233. [Google Scholar]
- Mount, D.B. The kidney in hyperuricemia and gout. Curr. Opin. Nephrol. Hypertens. 2013, 22, 216–223. [Google Scholar] [CrossRef] [PubMed]
- Gibson, T. Hyperuricemia, gout and the kidney. Curr. Opin. Rheumatol. 2012, 24, 127–131. [Google Scholar] [CrossRef] [PubMed]
- Karis, E.; Crittenden, D.; Pillinger, M. Hyperuricemia, gout, and related comorbidities: Cause and effect on a two-way street. South Med. J. 2014, 107, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Vargas-Santos, A.B.; Neogi, T. Management of gout and hyperuricemia in CKD. Am. J. Kidney Dis. 2017, 70, 422–439. [Google Scholar] [CrossRef]
- Bove, M.; Cicero, A.F.; Veronesi, M.; Borghi, C. An evidence-based review on urate-lowering treatments: Implications for optimal treatment of chronic hyperuricemia. Vasc. Health Risk Manag. 2017, 13, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Grassi, D.; Pontremoli, R.; Bocale, R.; Ferri, C.; Desideri, G. Therapeutic approaches to chronic hyperuricemia and gout. High Blood Press. Cardiovasc. Prev. 2014, 21, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Stamp, L.K.; Chapman, P.T.; Palmer, S.C. Allopurinol and kidney function: An update. Joint Bone Spine 2016, 83, 19–24. [Google Scholar] [CrossRef]
- Pascual, E.; Sivera, F.; Andrés, M. Managing gout in the patient with renal impairment. Drugs Aging 2018, 35, 263–273. [Google Scholar] [CrossRef]
- Jones, G.; Panova, E.; Day, R. Guideline development for the management of gout: Role of combination therapy with a focus on lesinurad. Drug Des. Dev. Ther. 2017, 11, 3077–3081. [Google Scholar] [CrossRef] [PubMed]
- Schlesinger, N. The safety of treatment options available for gout. Expert Opin. Drug Saf. 2017, 16, 429–436. [Google Scholar] [CrossRef] [PubMed]
- Fang, P.; He, B.; Yu, M.; Shi, M.; Zhu, Y.; Zhang, Z.; Bo, P. Treatment with celastrol protects against obesity through suppression of galanin-induced fat intake and activation of PGC-1alpha/GLUT4 axis-mediated glucose consumption. Biochim. Biophys. Acta. Mol. Basis Dis. 2019, 1865, 1341–1350. [Google Scholar] [CrossRef] [PubMed]
- Verhaeghe, C.; Remouchamps, C.; Hennuy, B.; Vanderplasschen, A.; Chariot, A.; Tabruyn, S.P.; Oury, C.; Bours, V. Role of IKK and ERK pathways in intrinsic inflammation of cystic fibrosis airways. Biochem. Pharmacol. 2007, 73, 1982–1994. [Google Scholar] [CrossRef] [PubMed]
- Sheikh, T.; Gupta, P.; Gowda, P.; Patrick, S.; Sen, E. Hexokinase 2 and nuclear factor erythroid 2-related factor 2 transcriptionally coactivate xanthine oxidoreductase expression in stressed glioma cells. J. Biol. Chem. 2018, 293, 4767–4777. [Google Scholar] [CrossRef]
- Zhang, B.; Horvath, S. A general framework for weighted gene co-expression network analysis. Stat. Appl. Genet. Mol. Biol. 2005, 4. [Google Scholar] [CrossRef]
- Langfelder, P.; Horvath, S. WGCNA: An R package for weighted correlation network analysis. BMC Bioinform. 2008, 9, 559. [Google Scholar] [CrossRef]
- Ling, X.; Bochu, W. A review of phytotherapy of gout: Perspective of new pharmacological treatments. Pharmazie 2014, 69, 243–256. [Google Scholar]
- Chen, C.; Lü, J.M.; Yao, Q. Hyperuricemia-related diseases and xanthine oxidoreductase (XOR) inhibitors: An overview. Med. Sci. Monit. 2016, 22, 2501–2512. [Google Scholar] [CrossRef] [PubMed]
- So, A.; Thorens, B. Uric acid transport and disease. J. Clin. Invest. 2010, 120, 1791–1799. [Google Scholar] [CrossRef] [PubMed]
- Bach, M.H.; Simkin, P.A. Uricosuric drugs: The once and future therapy for hyperuricemia? Curr. Opin. Rheumatol. 2014, 26, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhai, T.; Ma, R.; Luo, C.; Wang, H.; Liu, L. Effects of uric acid-lowering therapy on the progression of chronic kidney disease: A systematic review and meta-analysis. Ren. Fail. 2018, 40, 289–297. [Google Scholar] [CrossRef]
- Sampson, A.L.; Singer, R.F.; Walters, G.D. Uric acid lowering therapies for preventing or delaying the progression of chronic kidney disease. Cochrane Database Syst. Rev. 2017, 10, CD009460. [Google Scholar] [CrossRef]
- Mandal, A.K.; Mount, D.B. The molecular physiology of uric acid homeostasis. Annu. Rev. Physiol. 2015, 77, 323–345. [Google Scholar] [CrossRef]
- Feig, D.I.; Kang, D.H.; Johnson, R.J. Uric acid and cardiovascular risk. N. Engl. J. Med. 2008, 359, 1811–1821. [Google Scholar] [CrossRef]
- Jalal, D.I. Hyperuricemia, the kidneys, and the spectrum of associated diseases: A narrative review. Curr. Med. Res. Opin. 2016, 32, 1863–1869. [Google Scholar] [CrossRef]
- Desai, J.; Steiger, S.; Anders, H.J. Molecular pathophysiology of gout. Trends Mol. Med. 2017, 23, 756–768. [Google Scholar] [CrossRef]
- Webster, A.C.; Nagler, E.V.; Morton, R.L.; Masson, P. Chronic kidney disease. Lancet 2017, 389, 1238–1252. [Google Scholar] [CrossRef]
- Yang, Z.; Wang, X.; Jiang, L.; Tan, R.; Xiong, M.; He, W.; Fang, L.; Wen, P.; Yang, J. Uric acid increases fibronectin synthesis through upregulation of lysyl oxidase expression in rat renal tubular epithelial cells. Am. J. Physiol. Renal Physiol. 2010, 299, F336–F346. [Google Scholar] [CrossRef] [PubMed]
- Bergamini, C.; Cicoira, M.; Rossi, A.; Vassanelli, C. Oxidative stress and hyperuricaemia: Pathophysiology, clinical relevance, and therapeutic implications in chronic heart failure. Eur. J. Heart Fail. 2009, 11, 444–452. [Google Scholar] [CrossRef] [PubMed]
- Isaka, Y.; Takabatake, Y.; Takahashi, A.; Saitoh, T.; Yoshimori, T. Hyperuricemia-induced inflammasome and kidney diseases. Nephrol. Dial. Transplant. 2016, 31, 890–896. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Sheng, Y.; Liu, C.; Li, K.; Huang, X.; Huang, J.; Xu, K. Nox4 has a crucial role in uric acidinduced oxidative stress and apoptosis in renal tubular cells. Mol. Med. Rep. 2016, 13, 4343–4348. [Google Scholar] [CrossRef]
- Casalena, G.; Daehn, I.; Bottinger, E. Transforming growth factor-beta, bioenergetics, and mitochondria in renal disease. Semin. Nephrol. 2012, 32, 295–303. [Google Scholar] [CrossRef]
- Chen, L.; Yang, T.; Lu, D.W.; Zhao, H.; Feng, Y.L.; Chen, H.; Chen, D.Q.; Vaziri, N.D.; Zhao, Y.Y. Central role of dysregulation of TGF-beta/Smad in CKD progression and potential targets of its treatment. Biomed. Pharmacother. 2018, 101, 670–681. [Google Scholar] [CrossRef]
- Chevalier, R.L.; Forbes, M.S.; Thornhill, B.A. Ureteral obstruction as a model of renal interstitial fibrosis and obstructive nephropathy. Kidney Int. 2009, 75, 1145–1152. [Google Scholar] [CrossRef]
- Kadakol, A.; Sharma, N.; Kulkarni, Y.A.; Gaikwad, A.B. Esculetin: A phytochemical endeavor fortifying effect against non-communicable diseases. Biomed. Pharmacother. 2016, 84, 1442–1448. [Google Scholar] [CrossRef]
- Figueroa-Guiñez, R.; Matos, M.J.; Vazquez-Rodriguez, S.; Santana, L.; Uriarte, E.; Borges, F.; Olea-Azar, C.; Diego Maya, J. Interest of antioxidant agents in parasitic diseases. The case study of coumarins. Curr. Top. Med. Chem. 2015, 15, 850–856. [Google Scholar] [CrossRef]
- Filipsky, T.; Riha, M.; Macakova, K.; Anzenbacherová, E.; Karlickova, J.; Mladenka, P. Antioxidant effects of coumarins include direct radical scavenging, metal chelation and inhibition of ROS-producing enzymes. Curr. Top. Med. Chem. 2015, 15, 415–431. [Google Scholar] [CrossRef]
- He, Y.; Li, C.; Ma, Q.; Chen, S. Esculetin inhibits oxidative stress and apoptosis in H9c2 cardiomyocytes following hypoxia/reoxygenation injury. Biochem. Biophys. Res. Commun. 2018, 501, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Kang, K.A.; Zhang, R.; Piao, M.J.; Ko, D.O.; Wang, Z.H.; Chae, S.W.; Kang, S.S.; Lee, K.H.; Kang, H.K.; et al. Protective effect of esculetin against oxidative stress-induced cell damage via scavenging reactive oxygen species. Acta Pharmacol. Sin. 2008, 29, 1319–1326. [Google Scholar] [CrossRef] [PubMed]
- Ozal, S.A.; Turkekul, K.; Gurlu, V.; Guclu, H.; Erdogan, S. Esculetin protects human retinal pigment epithelial cells from lipopolysaccharide-induced inflammation and cell death. Curr. Eye Res. 2018, 43, 1169–1176. [Google Scholar] [CrossRef] [PubMed]
- Arora, R.; Sawney, S.; Saini, V.; Steffi, C.; Tiwari, M.; Saluja, D. Esculetin induces antiproliferative and apoptotic response in pancreatic cancer cells by directly binding to KEAP1. Mol. Cancer 2016, 15, 64. [Google Scholar] [CrossRef] [PubMed]
- DeNicola, G.M.; Karreth, F.A.; Humpton, T.J.; Gopinathan, A.; Wei, C.; Frese, K.; Mangal, D.; Kenneth, H.Y.; Yeo, C.J.; Calhoun, E.S.; et al. Oncogene-induced Nrf2 transcription promotes ROS detoxification and tumorigenesis. Nature 2011, 475, 106–109. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, M.; Kensler, T.W.; Motohashi, H. The KEAP1-NRF2 system: A thiol-based sensor-effector apparatus for maintaining redox homeostasis. Physiol. Rev. 2018, 98, 1169–1203. [Google Scholar] [CrossRef] [PubMed]
- Iwanaga, T.; Kobayashi, D.; Hirayama, M.; Maeda, T.; Tamai, I. Involvement of uric acid transporter in increased renal clearance of the xanthine oxidase inhibitor oxypurinol induced by a uricosuric agent, benzbromarone. Drug Metab. Dispos. 2005, 33, 1791–1795. [Google Scholar] [CrossRef] [PubMed]
- Stamp, L.K.; Haslett, J.; Frampton, C.; White, D.; Gardner, D.; Stebbings, S.; Taylor, G.; Grainger, R.; Kumar, R.; Kumar, S.; et al. The safety and efficacy of benzbromarone in gout in Aotearoa New Zealand. Intern. Med. J. 2016, 46, 1075–1080. [Google Scholar] [CrossRef]
- Sun, P.; Zhu, J.J.; Wang, T.; Huang, Q.; Zhou, Y.R.; Yu, B.W.; Jiang, H.L.; Wang, H.Y. Benzbromarone aggravates hepatic steatosis in obese individuals. Biochim. Biophys. Acta Mol. Basis Dis. 2018, 1864, 2067–2077. [Google Scholar] [CrossRef]
- Felser, A.; Lindinger, P.W.; Schnell, D.; Kratschmar, D.V.; Odermatt, A.; Mies, S.; Jenö, P.; Krähenbühl, S. Hepatocellular toxicity of benzbromarone: Effects on mitochondrial function and structure. Toxicology 2014, 324, 136–146. [Google Scholar] [CrossRef]
- Wang, J.; Lu, M.L.; Dai, H.L.; Zhang, S.P.; Wang, H.X.; Wei, N. Esculetin, a coumarin derivative, exerts in vitro and in vivo antiproliferative activity against hepatocellular carcinoma by initiating a mitochondrial-dependent apoptosis pathway. Braz. J. Med. Biol. Res. 2015, 48, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Choi, R.Y.; Ham, J.R.; Lee, M.K. Esculetin prevents non-alcoholic fatty liver in diabetic mice fed high-fat diet. Chem. Biol. Interact. 2016, 260, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.; Raj, P.; Goru, S.K.; Kadakol, A.; Malek, V.; Sharma, N.; Gaikwad, A.B. Esculetin ameliorates hepatic fibrosis in high fat diet induced non-alcoholic fatty liver disease by regulation of FoxO1 mediated pathway. Pharmacol. Rep. 2017, 69, 666–672. [Google Scholar] [CrossRef] [PubMed]
- Prabakaran, D.; Ashokkumar, N. Protective effect of esculetin on hyperglycemia-mediated oxidative damage in the hepatic and renal tissues of experimental diabetic rats. Biochimie 2013, 95, 366–373. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Kong, W.; Wang, L.; Zhang, T.; Huang, B.; Meng, J.; Yang, B.; Xie, Z.; Zhou, H. Multiple-Purpose Connectivity Map Analysis Reveals the Benefits of Esculetin to Hyperuricemia and Renal Fibrosis. Int. J. Mol. Sci. 2020, 21, 7695. https://doi.org/10.3390/ijms21207695
Wang Y, Kong W, Wang L, Zhang T, Huang B, Meng J, Yang B, Xie Z, Zhou H. Multiple-Purpose Connectivity Map Analysis Reveals the Benefits of Esculetin to Hyperuricemia and Renal Fibrosis. International Journal of Molecular Sciences. 2020; 21(20):7695. https://doi.org/10.3390/ijms21207695
Chicago/Turabian StyleWang, Yiming, Weikaixin Kong, Liang Wang, Tianyu Zhang, Boyue Huang, Jia Meng, Baoxue Yang, Zhengwei Xie, and Hong Zhou. 2020. "Multiple-Purpose Connectivity Map Analysis Reveals the Benefits of Esculetin to Hyperuricemia and Renal Fibrosis" International Journal of Molecular Sciences 21, no. 20: 7695. https://doi.org/10.3390/ijms21207695
APA StyleWang, Y., Kong, W., Wang, L., Zhang, T., Huang, B., Meng, J., Yang, B., Xie, Z., & Zhou, H. (2020). Multiple-Purpose Connectivity Map Analysis Reveals the Benefits of Esculetin to Hyperuricemia and Renal Fibrosis. International Journal of Molecular Sciences, 21(20), 7695. https://doi.org/10.3390/ijms21207695