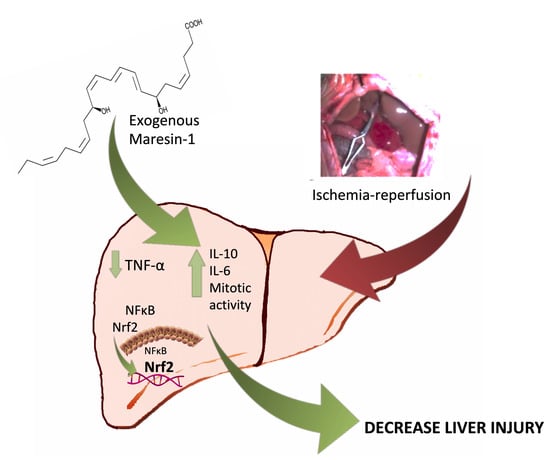
Maresin 1, a Proresolving Lipid Mediator, Ameliorates Liver Ischemia-Reperfusion Injury and Stimulates Hepatocyte Proliferation in Sprague-Dawley Rats
Abstract
1. Introduction
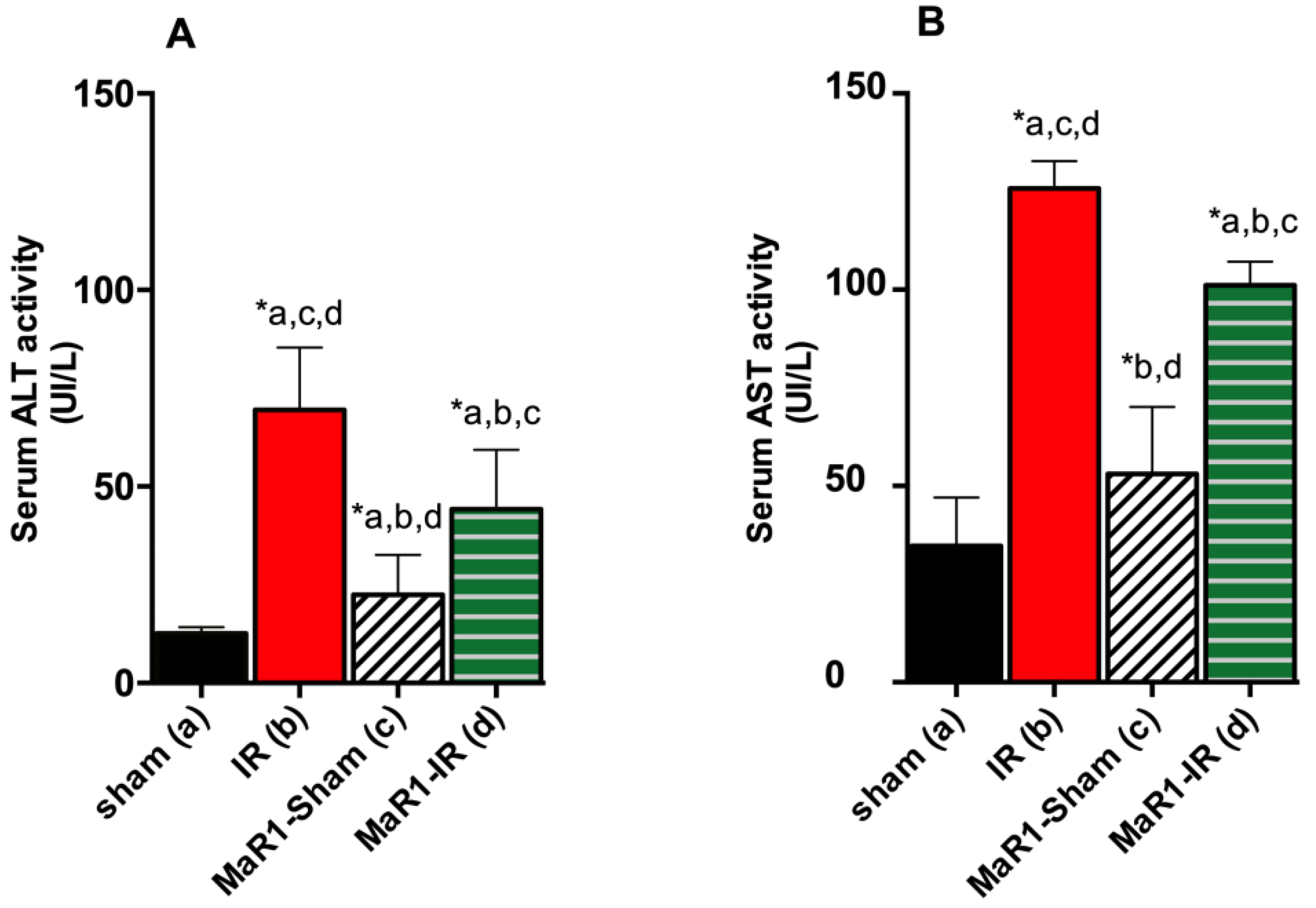
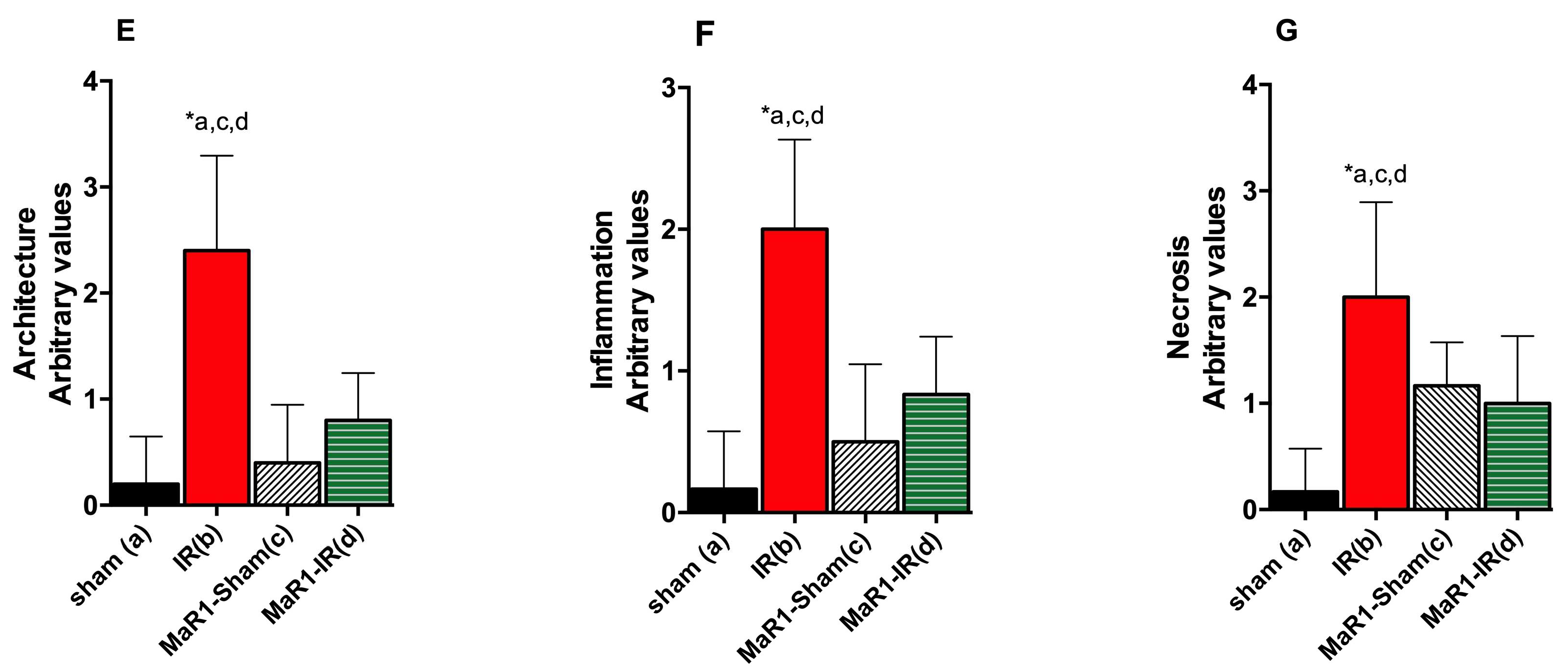
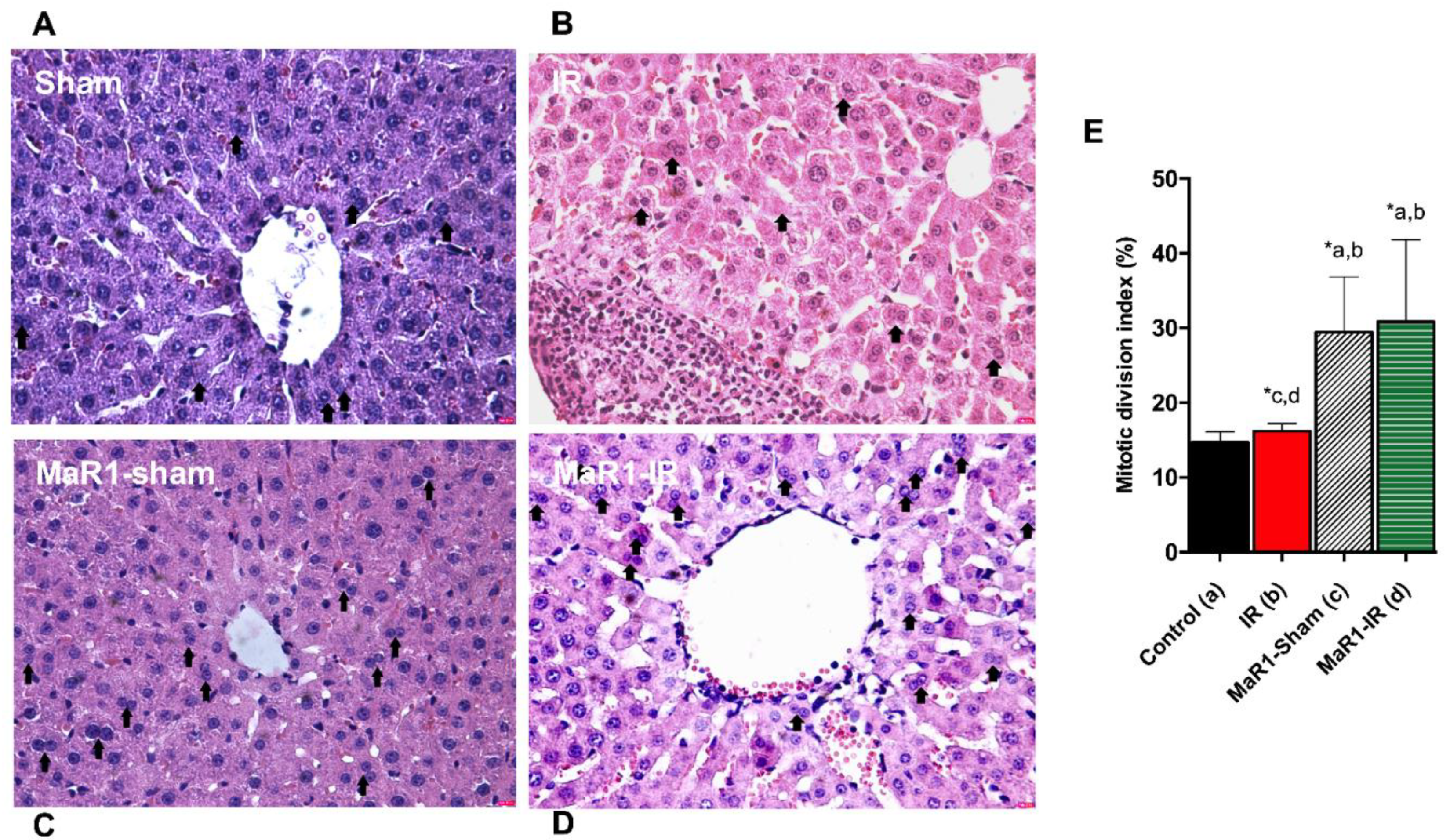
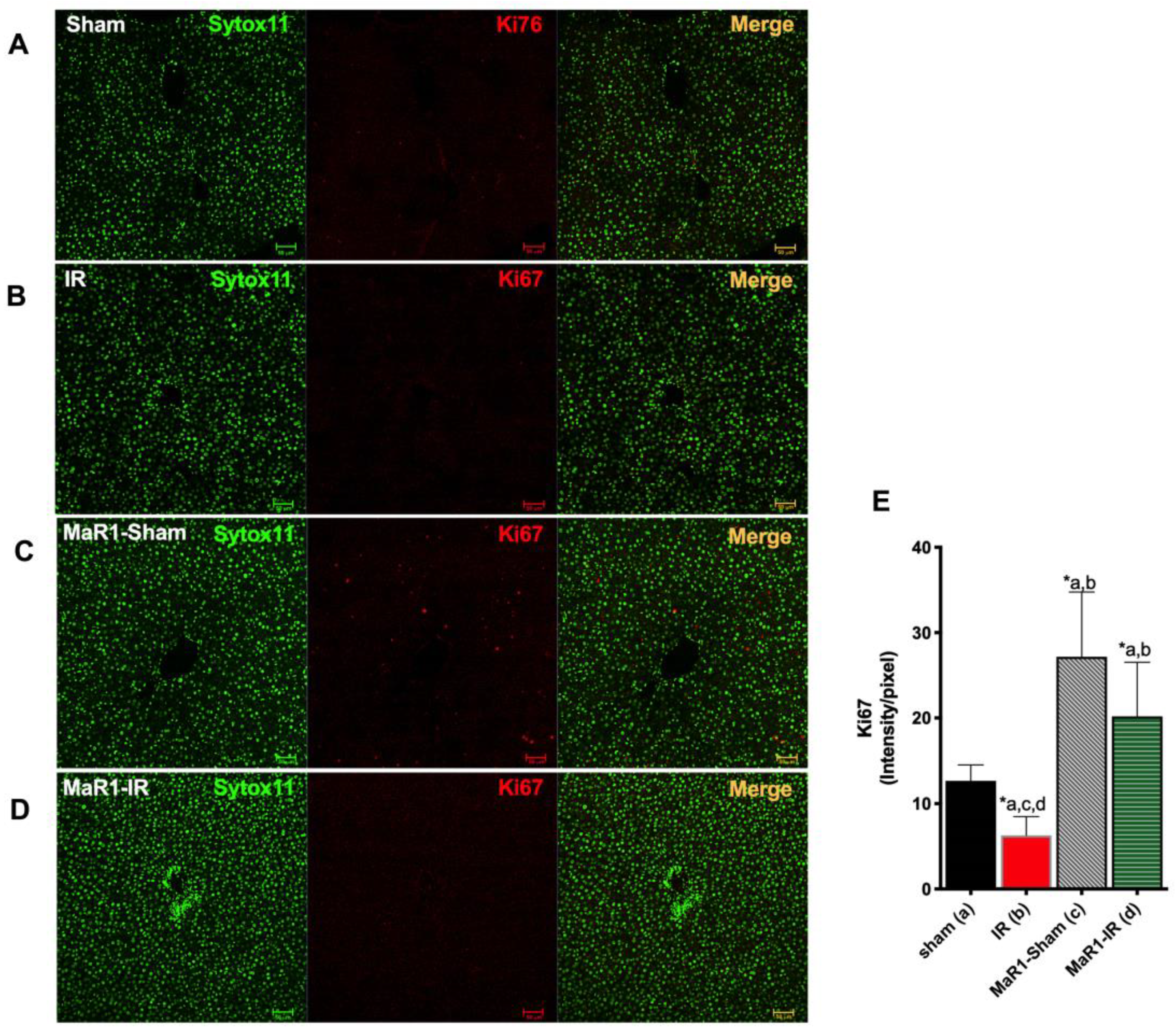
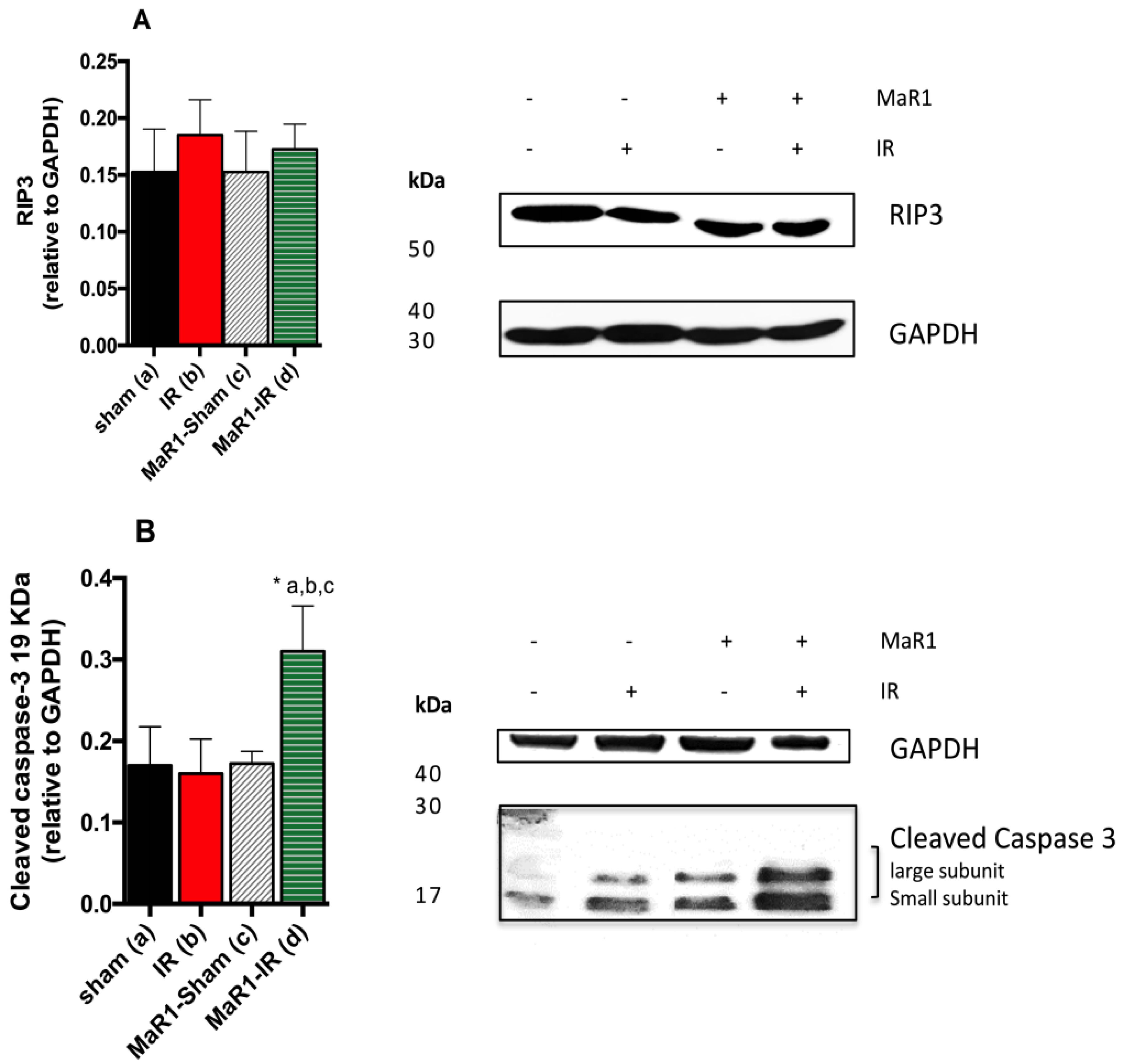
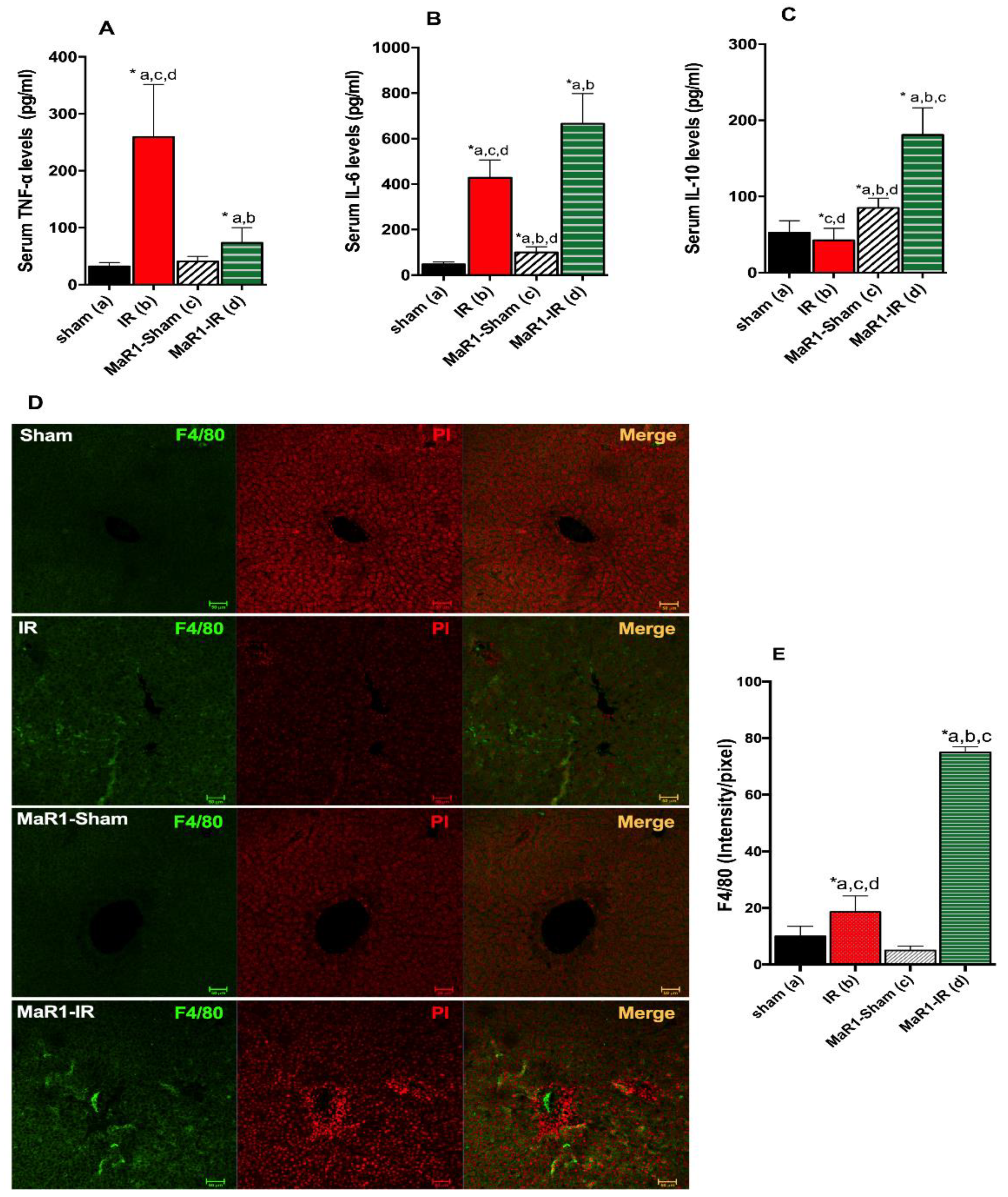
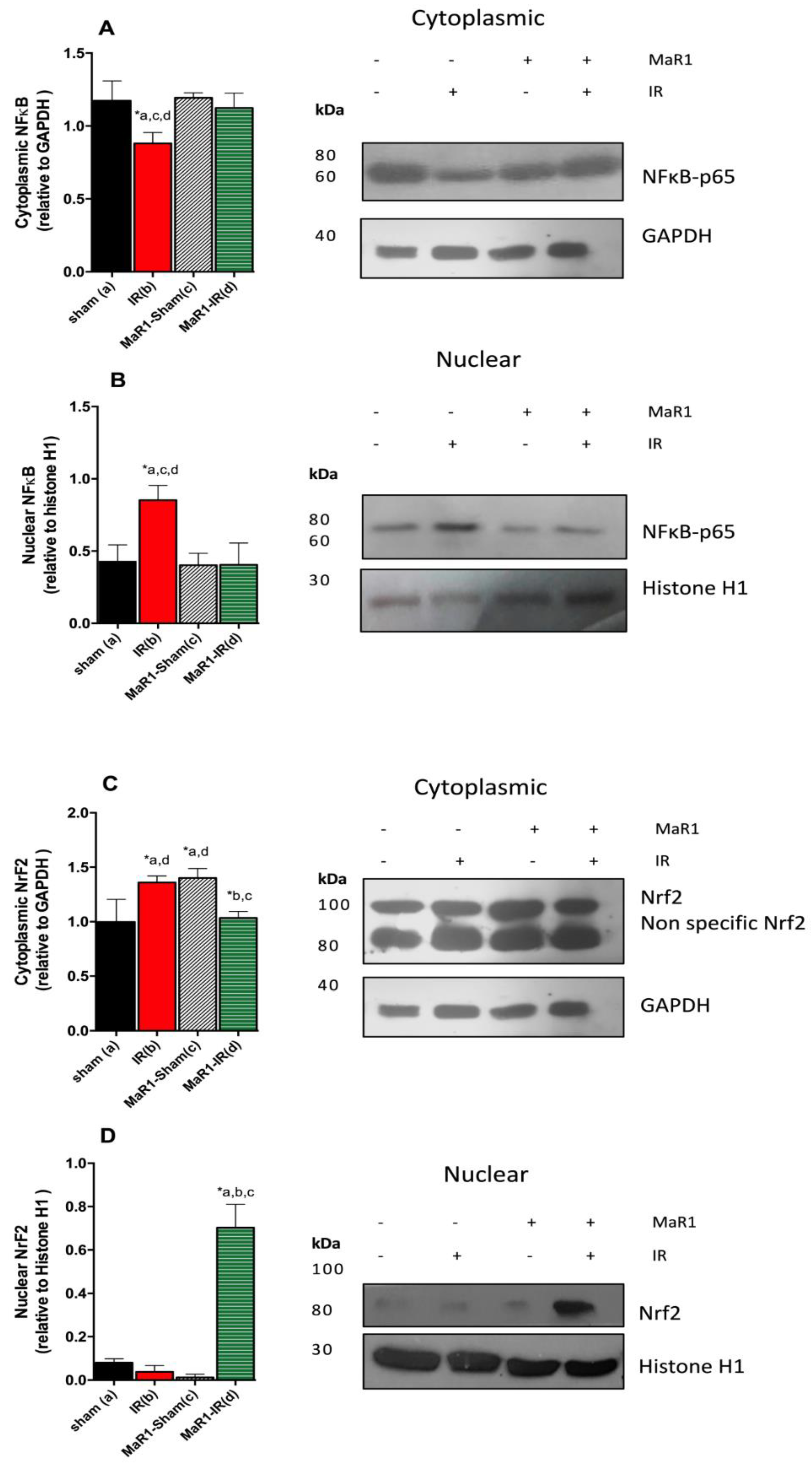
2. Results
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Model of Partial Ischemia-Reperfusion (IR) Injury
4.3. Measurement of Serum Parameters
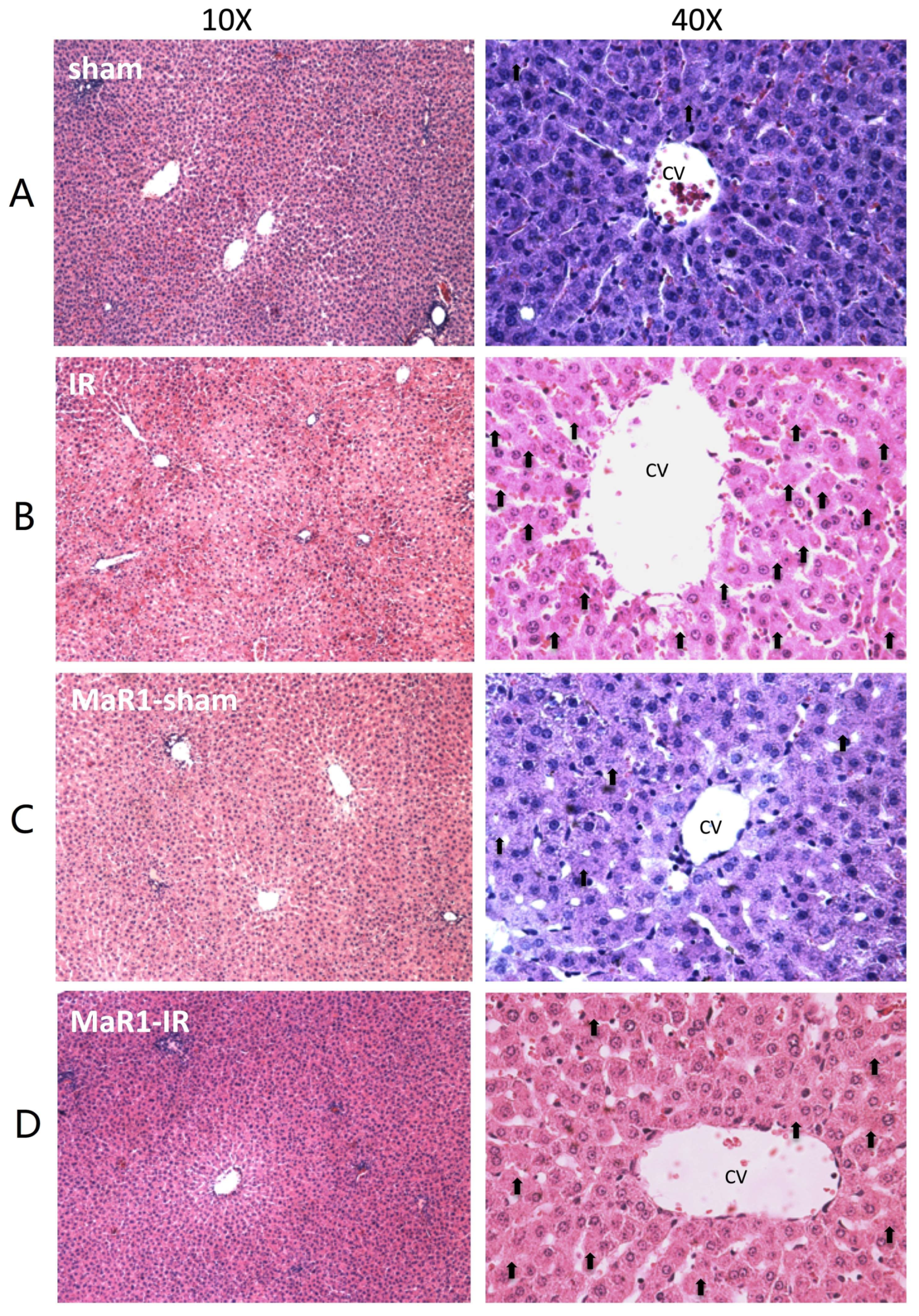
4.4. Histopathological Examination
4.5. Immunofluorescense Staining
4.6. Western Blot Analysis
4.7. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| IR | ischemia-reperfusion |
| ROS | reactive oxygen species |
| KCs | Küpffer Cells |
| PUFA | Polyunsaturated fatty acids |
| EPA | eicosapentaenoic acid |
| DHA | docosahexaenoic acid |
| MaR1 | maresin 1 |
| MAI | mitotic activity index |
| H&E | hematoxylin-eosin |
| ALT | alanine aminotransferase |
| AST | aspartate aminotransferase |
| SD | standard deviation |
| PH | partial hepatectomy |
| HCC | hepatocellular carcinoma |
| ER | endoplasmic reticulum |
References
- Lentsch, A.B. Regulatory Mechanisms of Injury and Repair after Hepatic Ischemia/Reperfusion. Scientifica 2012, 2012, 513192. [Google Scholar] [CrossRef] [PubMed]
- Nastos, C.; Kalimeris, K.; Papoutsidakis, N.; Tasoulis, M.K.; Likoudis, P.M.; Theodoraki, K.; Nastou, D.; Smyrniotis, V.; Arkadopoulos, N. Global Consequences of Liver Ischemia/Reperfusion Injury. Oxidative Med. Cell Longev. 2014, 2014, 906965. [Google Scholar] [CrossRef] [PubMed]
- Lee, L.Y.; Harberg, C.; Matkowskyj, K.A.; Cook, C.; Roenneburg, D.; Werner, S.; Johnson, J.; Foley, D.P. Overactivation of the Nuclear Factor (Erythroid-Derived 2) Like 2–Antioxidant Response Element Pathway in Hepatocytes Decreases Hepatic Ischemia/Reperfusion Injury in Mice. Liver Transpl. 2016, 22, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Kudoh, K.; Uchinami, H.; Yoshioka, M.; Seki, E.; Yamamoto, Y. Nrf2 activation protects the liver from ischemia/reperfusion injury in mice. Ann. Surg. 2014, 260, 118–127. [Google Scholar] [CrossRef] [PubMed]
- Zuniga, J.; Venegas, F.; Villarreal, M.; Nunez, D.; Chandia, M.; Valenzuela, R.; Tapia, G.; Varela, P.; Videla, L.A.; Fernández, V. Protection against in vivo liver ischemia-reperfusion injury by n-3 long-chain polyunsaturated fatty acids in the rat. Free Radic. Res. 2010, 44, 854–863. [Google Scholar] [CrossRef]
- Kaur, N.; Chugh, V.; Gupta, A. Essential fatty acids as functional components of food—A review. J. Food Sci. Technol. 2014, 51, 2289–2303. [Google Scholar] [CrossRef]
- Marcon, R.; Bento, A.F.; Dutra, R.C.; Bicca, M.A.; Leite, D.F.; Calixto, J.B. Maresin 1, a proresolving lipid mediator derived from omega-3 polyunsaturated fatty acids, exerts protective actions in murine models of colitis. J. Immunol. 2013, 191, 4288–4298. [Google Scholar] [CrossRef]
- Li, R.; Wang, Y.; Zhao, E.; Wu, K.; Li, W.; Shi, L.; Wang, D.; Xie, G.; Yin, Y.; Deng, M.; et al. Maresin 1, a Proresolving Lipid Mediator, Mitigates Carbon Tetrachloride-Induced Liver Injury in Mice. Oxidative Med. Cell Longev. 2016, 2016, 9203716. [Google Scholar] [CrossRef]
- Ma, J.Q.; Ding, J.; Zhang, L.; Liu, C.M. Ursolic acid protects mouse liver against CCl4-induced oxidative stress and inflammation by the MAPK/NF-κB pathway. Environ. Toxicol. Pharmacol. 2014, 37, 975–983. [Google Scholar] [CrossRef]
- Saidi, R.F.; Kenari, S.K. Liver ischemia/reperfusion injury: An overview. J. Investig. Surg. 2014, 27, 366–379. [Google Scholar] [CrossRef]
- Yang, F.; Shang, L.; Wang, S.; Liu, Y.; Ren, H.; Zhu, W.; Shi, X. TNFα-Mediated Necroptosis Aggravates Ischemia-Reperfusion Injury in the Fatty Liver by Regulating the Inflammatory Response. Oxidative Med. Cell Longev. 2019, 12, 2301903. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Yao, J.; Zhang, F.; Xu, X.; Zhai, X.; Wang, G.; Ma, Z.; Gao, D.; Ma, X.; Fan, Q.; et al. Sirtuin 1-mediated inhibition of p66shc expression alleviates liver ischemia/reperfusion injury. Crit. Care Med. 2014, 42, e373–e381. [Google Scholar]
- Li, S.; Zheng, X.; Li, H.; Zheng, J.; Chen, X.; Liu, W.; Tai, Y.; Zhang, Y.; Wang, G.; Yang, Y. Mesenchymal Stem Cells Ameliorate Hepatic Ischemia/Reperfusion Injury via Inhibition of Neutrophil Recruitment. J. Immunol. Res. 2018, 2018. [Google Scholar] [CrossRef] [PubMed]
- Laiglesia, L.M.; Lorente-Cebrián, S.; López-Yoldi, M.; Lanas, R.; Sáinz, N.; Martínez, J.A.; Moreno-Aliaga, M.J. Maresin 1 inhibits TNF-alpha-induced lipolysis and autophagy in 3T3-L1 adipocytes. J. Cell Physiol. 2018, 233, 2238–2246. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, E.S.; Saleh, S.M.; El Hoseeny, M.; El Shaarawy, A. Effect of omega3 on hepatic regeneration in adult living donors undergoing hepatic resections for liver transplantation: A randomized controlled trial. J. Crit. Care 2016, 31, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Jackson, L.N.; Larson, S.D.; Silva, S.R.; Rychahou, P.G.; Chen, L.A.; Qiu, S.; Rajaraman, S.; Evers, B.M. PI3K/Akt activation is critical for early hepatic regeneration after partial hepatectomy. Am. J. Physiol. Gastrointest. Liver Physiol. 2008, 294, G1401–G1410. [Google Scholar] [CrossRef]
- Dalli, J.; Vlasakov, I.; Riley, I.R.; Rodriguez, A.R.; Spur, B.W.; Petasis, N.A.; Chiang, N.; Serhan, C.N. Maresin conjugates in tissue regeneration biosynthesis enzymes in human macrophages. Proc. Natl. Acad. Sci. USA 2016, 113, 12232–12237. [Google Scholar] [CrossRef]
- Hong, J.M.; Kim, S.J.; Lee, S.M. Role of necroptosis in autophagy signaling during hepatic ischemia and reperfusion. Toxicol. Appl. Pharmacol. 2016, 308, 1–10. [Google Scholar] [CrossRef]
- Saeed, W.K.; Jun, D.W.; Jang, K.; Chae, Y.J.; Lee, J.S.; Kang, H.T. Does necroptosis have a crucial role in hepatic ischemia-reperfusion injury? PLoS ONE 2017, 12, e0184752. [Google Scholar] [CrossRef]
- Canbek, O. Apoptosis induced by boric anhydrite (B2O3) after partial hepatectomy in rat liver. Bratisl. Med. 2016, 117, 231–234. [Google Scholar]
- Suzuki, T.; Tsukamoto, I. Manganese-induced apoptosis in hepatocytes after partial hepatectomy. Eur. J. Pharmacol. 2005, 525, 48–53. [Google Scholar] [CrossRef]
- Martínez-Fernández, L.; González-Muniesa, P.; Laiglesia, L.M.; Sáinz, N.; Prieto-Hontoria, P.L.; Escoté, X.; Odriozola, L.; Corrales, F.J.; Arbones-Mainar, J.M.; Martínez, J.A.; et al. Maresin 1 improves insulin sensitivity and attenuates adipose tissue inflammation in ob/ob and diet-induced obese mice. FASEB J. 2017, 31, 2135–2145. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.W.; Lee, S.M. Resolvin D1 protects the liver from ischemia/reperfusion injury by enhancing M2 macrophagepolarization and efferocytosis. Biochim. Biophys. Acta 2016, 1861, 1025–1035. [Google Scholar] [CrossRef] [PubMed]
- Micheau, O.; Tschopp, J. Induction of TNF receptor I-mediated apoptosis via two sequential signaling complexes. Cell 2003, 114, 181–190. [Google Scholar] [CrossRef]
- Faletti, L.; Peintner, L.; Neumann, S.; Sandler, S.; Grabinger, T.; Mac Nelly, S.; Merfort, I. TNFα sensitizes hepatocytes to FasL-induced apoptosis by NFκB-mediated Fas upregulation. Cell Death Dis. 2018, 9, 909. [Google Scholar] [CrossRef] [PubMed]
- Kilicoglu, B.; Kilicoglu, S.S.; Tuncal, S.; Onalan, A.K.; Celepli, S.; Tasova, V.; Namuslu, M.; Erdemli, E. Ultrastructural view of a promising anti TNF-α agent on hepatic ischaemia reperfusion injury. Bratisl. Lek Listy. 2015, 116, 601–607. [Google Scholar] [CrossRef]
- Zúñiga, J.; Cancino, M.; Medina, F.; Varela, P.; Vargas, R.; Tapia, G.; Videla, L.A.; Fernández, V. N-3 PUFA supplementation triggers PPAR-α activation and PPAR-α/NF-κB interaction: Anti-inflammatory implications in liver ischemia-reperfusion injury. PLoS ONE 2011, 6, e28502. [Google Scholar] [CrossRef]
- Calder, P.C. Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochim. Biophys. Acta 2015, 1851, 469–484. [Google Scholar] [CrossRef]
- Tao, Y.; Wang, M.; Chen, E.; Tang, H. Liver Regeneration: Analysis of the Main Relevant Signaling Molecules. Mediat. Inflamm. 2017, 2017, 4256352. [Google Scholar] [CrossRef]
- Kang, L.I.; Mars, W.M.; Michalopoulos, G. Signals and Cells Involved in Regulating Liver Regeneration. Cells 2012, 1, 1261–1292. [Google Scholar] [CrossRef]
- Li, R.; Wang, Y.; Ma, Z.; Ma, M.; Wang, D.; Xie, G.; Yin, Y.; Zhang, P.; Tao, K. Maresin 1 Mitigates Inflammatory Response and Protects Mice from Sepsis. Mediat. Inflamm. 2016, 2016, 3798465. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Y.; Wu, Y.; Zhao, H.; Sun, H.; Gao, S. Maresin I mitigate renal ischemia/reperfusion injury in mice via inhibition of the TLR4/MAPK/NF-κB pathways and activation of the NrF2 pathway. Drug Des. Dev. Ther. 2019, 12, 739–745. [Google Scholar] [CrossRef] [PubMed]
- Xian, W.; Li, T.; Li, L.; Hu, L.; Gao, J. Maresin 1 attenuates the inflammatory response and mitochondrial damage in mice with cerebral ischemia/reperfusion in SIRT1-dependent manner. Brain Res. 2019, 1711, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.; Wu, Y.; Zhao, F.; Wang, J. Maresin 1 Ameliorates Lung Ischemia/Reperfusion Injury by Suppressing Oxidative Stress via Activation of the Nrf-2-Mediated HO-1 Signaling Pathway. Oxidative Med. Cell Longev. 2017, 2017, 9634803. [Google Scholar] [CrossRef]
- Akira, S.; Isshiki, H.; Nakajima, T.; Kinoshita, S.; Nishio, Y.; Natsuka, S.; Kishimoto, T. Regulation of expression of the interleukin 6 gene: Structure and function of the transcription factor NF-IL6. Ciba Found Symp. 1992, 167, 47–62. [Google Scholar]
- Kamimukai, N.; Togo, S.; Hasegawa, S.; Tuboca, T.; Kurosawa, H.; Li, X.K.; Suzuki, S.; Shimada, H. Expression of Bcl-2 family reduce apoptotic hepatocyte after excessive hepatectomy. Eur. Sur. Res. 2001, 33, 8–15. [Google Scholar] [CrossRef]
- Schmidt-Arras, D.; Rose-John, A. IL-6 pathway in the liver: From physiopatology to therapy. J. Hepatol. 2016, 64, 1403–1415. [Google Scholar] [CrossRef]
- Trautwein, C.; Caelles, C.; van der Geer, P.; Hunter, T.; Karin, M.; Chojkier, M. Transactivation by NF-IL6/LAP is enhanced by phosphorylation of its activation domain. Nature 1993, 364, 544–547. [Google Scholar] [CrossRef]
- Li, M.; Zhou, X.; Mei, J.; Geng, X.; Zhou, Y.; Zhang, W.; Xu, C. Study on the activity of the signaling pathways regulating hepatocytes from G0 phase into G1 phase during rat liver regeneration. Cell Mol. Biol. Lett. 2014, 19, 181–200. [Google Scholar] [CrossRef]
- Cressman, D.E.; Greenbaum, L.E.; DeAngelis, R.A.; Ciliberto, G.; Furth, E.E.; Poli, V.; Taub, R. Liver failure and defective hepatocyte regeneration in interleukin-6-deficient mice. Science 1996, 274, 1379–1383. [Google Scholar] [CrossRef]
- Wan, S.; LeClerc, J.L.; Schmartz, D.; Barvais, L.; Huynh, C.H.; Deviere, J.; DeSmet, J.M.; Vincent, J.L. Hepatic release of interleukin-10 during cardiopulmonary bypass in steroid-pretreated patients. Am. Heart J. 1997, 133, 335–339. [Google Scholar] [CrossRef]
- Zhang, L.-J.; Wang, X.-Z. Interleukin-10 and chronic liver disease. World J. Gastroenterol. 2006, 12, 1681–1685. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.H.; Chen, M.H.; Guo, Q.L.; Chen, Z.X.; Chen, Q.D.; Wang, X.Z. Interleukin-10 induces senescence of activated hepatic stellate cells via STAT3-p53 pathway to attenuate liver fibrosis. Cell Signal. 2019, 12, 109445. [Google Scholar] [CrossRef] [PubMed]
- Céspedes, N.; Tamayo, A.; Rodriguez, M.J.; Zúñiga-Hernández, J. EPA plus DHA improves survival related to a decrease of injury after extended liver ischemia in Sprague-Dawley rats. Ann. Hepatol. 2019, S1665-2681, 32254–32259. [Google Scholar] [CrossRef]
- Yang, T.; Xu, G.; Newton, P.T.; Chagin, A.S.; Mkrtchian, S.; Carlström, M.; Zhang, X.M.; Harris, R.A.; Cooter, M.; Berger, M.; et al. Maresin 1 attenuates neuroinflammation in a mouse model of perioperative neurocognitive disorders. Br. J. Anaesth. 2019, 122, 350–360. [Google Scholar] [CrossRef]
- Munir, F.; Jamshed, M.B.; Shahid, N.; Muhammad, S.A.; Bhandari, A.; Zhang, Q. Protective effects of maresin 1 against inflammation in experimentally induced acute pancreatitis and related lung injury. Am. J. Physiol. Gastrointest. Liver Physiol. 2019, 317, G333–G341. [Google Scholar] [CrossRef]
- Wan, J.; Benkdane, M.; Teixeira-Clerc, F.; Bonnafous, S.; Louvet, A.; Lafdil, F.; Pecker, F.; Tran, A.; Gual, P.; Mallat, A.; et al. M2 Kupffer cells promote M1 Kupffer cell apoptosis: A protective mechanism against alcoholic and nonalcoholic fatty liver disease. Hepatology 2014, 59, 130–142. [Google Scholar] [CrossRef]
- Ohuchi, K.; Ono, Y.; Joho, M.; Tsuruma, K.; Ogami, S.; Yamane, S.; Funato, M.; Kaneko, H.; Nakamura, S.; Hara, H.; et al. A Docosahexaenoic Acid-Derived Pro-resolving Agent, Maresin 1, Protects Motor Neuron Cells Death. Neurochem. Res. 2018, 43, 1413–1423. [Google Scholar] [CrossRef]
- Schreck, R.; Rieber, P.; Baeuerle, P.A. Reactive oxygen intermediates as apparently widely used messengers in the activation of the NF-kappa B transcription factor and HIV-1. EMBO J. 1991, 10, 2247–2258. [Google Scholar] [CrossRef]
- Tam, A.B.; Mercado, E.L.; Hoffmann, A.; Niwa, M. ER stress activates NF-kappaB by integrating functions of basal IKK activity, IRE1 and PERK. PLoS ONE 2012, 7, e45078. [Google Scholar] [CrossRef]
- Yu, J.; Feng, Z.; Tan, L.; Pu, L.; Kong, L. Interleukin-11 protects mouse liver from warm ischemia/reperfusion (WI/Rp) injury. Clin. Res. Hepatol. Gastroenterol. 2016, 40, 562–570. [Google Scholar] [CrossRef] [PubMed]
- Fernández, V.; Tapia, G.; Varela, P.; Gaete, L.; Vera, G.; Mora, C.; Vial, M.T.; Videla, L.A. Causal role of oxidative stress in liver preconditioning by thyroid hormone in rats. Free Radic. Biol. Med. 2008, 44, 1724–1731. [Google Scholar] [CrossRef] [PubMed]
- Videla, L.A. Oxidative stress signaling underlying liver disease and hepatoprotective mechanisms. World J. Hepatol. 2009, 1, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Firat, O.; Makay, O.; Yeniay, L.; Gokce, G.; Yenisey, C.; Coker, A. Omega-3 fatty acids inhibit oxidative stress in a rat model of liver regeneration. Ann. Surg. Treat. Res. 2017, 93, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Firat, T.; Söyler, G.; Töre, F.; Şit, M.; Kiyan, A.; Özgen, U.; Kükner, A. 4-(3,4-dihydroxybenzoyloxymethyl)phenyl-O-β-D-glucopyranoside effect in liver regeneration. Histol. Histopathol. 2018, 5, 18056. [Google Scholar]
- Köhler, U.A.; Kurinna, S.; Schwitter, D.; Marti, A.; Schäfer, M.; Hellerbrand, C.; Speicher, T.; Werner, S. Activated Nrf2 impairs liver regeneration in mice by activation of genes involved in cell-cycle control and apoptosis. Hepatology 2014, 60, 670–678. [Google Scholar] [CrossRef]
- Dayoub, R.; Vogel, A.; Schuett, J.; Lupke, M.; Spieker, S.M.; Kettern, N.; Hildt, E.; Melter, M.; Weiss, T.S. Nrf2 activates augmenter of liver regeneration (ALR) via antioxidant response element and links oxidative stress to liver regeneration. Mol. Med. 2013, 19, 237–244. [Google Scholar] [CrossRef]
- Fernández, V.; Castillo, I.; Tapia, G.; Romanque, P.; Uribe-Echevarrıía, S.; Uribe, M.; Cartier-Ugarte, D.; Santander, G.; Vial, M.T.; Videla, L.A. Thyroid hormone preconditioning: Protection against ischemia-reperfusion liver injury in the rat. Hepatology 2007, 45, 170–177. [Google Scholar] [CrossRef]
- Korourian, S.; Hakkak, R.; Ronis, M.J.; Shelnutt, S.R.; Waldron, J.; Ingelman-Sunberg, M.; Badger, T.M. Diet and risk of ethanol-induced hepatoxicity: Carbohydrate-fat relationships in rats. Toxicol. Sci. 1999, 47, 110–117. [Google Scholar] [CrossRef]
- Goodman, Z. Grading and staging systems for inflammation and fibrosis in chronic liver disease. J. Hepatol. 2007, 47, 598–607. [Google Scholar] [CrossRef]
- Ishak, K.; Baptista, A.; Bianchi, L.; Callea, F.; De Groote, J.; Gudat, F.; Denk, H.; Desmet, V.; Korb, G.; MacSween, R.N.; et al. Histological grading and staging of chronic hepatitis. J. Hepatol. 1995, 22, 696–699. [Google Scholar] [CrossRef]
- Al-Janabi, S.; van Slooten, H.J.; Visser, M.; van der Ploeg, T.; van Diest, P.J.; Jiwa, M. Evaluation of mitotic activity index in breast cancer using whole slide digital images. PLoS ONE 2013, 8, e82576. [Google Scholar] [CrossRef] [PubMed]
- Deryckere, F.; Gannon, F. A one-hour minipreparation technique for extraction of DNA-binding proteins from animal tissues. Biotechniques 1994, 16, 405. [Google Scholar] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Soto, G.; Rodríguez, M.J.; Fuentealba, R.; Treuer, A.V.; Castillo, I.; González, D.R.; Zúñiga-Hernández, J. Maresin 1, a Proresolving Lipid Mediator, Ameliorates Liver Ischemia-Reperfusion Injury and Stimulates Hepatocyte Proliferation in Sprague-Dawley Rats. Int. J. Mol. Sci. 2020, 21, 540. https://doi.org/10.3390/ijms21020540
Soto G, Rodríguez MJ, Fuentealba R, Treuer AV, Castillo I, González DR, Zúñiga-Hernández J. Maresin 1, a Proresolving Lipid Mediator, Ameliorates Liver Ischemia-Reperfusion Injury and Stimulates Hepatocyte Proliferation in Sprague-Dawley Rats. International Journal of Molecular Sciences. 2020; 21(2):540. https://doi.org/10.3390/ijms21020540
Chicago/Turabian StyleSoto, Gonzalo, María José Rodríguez, Roberto Fuentealba, Adriana V. Treuer, Iván Castillo, Daniel R. González, and Jessica Zúñiga-Hernández. 2020. "Maresin 1, a Proresolving Lipid Mediator, Ameliorates Liver Ischemia-Reperfusion Injury and Stimulates Hepatocyte Proliferation in Sprague-Dawley Rats" International Journal of Molecular Sciences 21, no. 2: 540. https://doi.org/10.3390/ijms21020540
APA StyleSoto, G., Rodríguez, M. J., Fuentealba, R., Treuer, A. V., Castillo, I., González, D. R., & Zúñiga-Hernández, J. (2020). Maresin 1, a Proresolving Lipid Mediator, Ameliorates Liver Ischemia-Reperfusion Injury and Stimulates Hepatocyte Proliferation in Sprague-Dawley Rats. International Journal of Molecular Sciences, 21(2), 540. https://doi.org/10.3390/ijms21020540