Effects of Kifunensine on Production and N-Glycosylation Modification of Butyrylcholinesterase in a Transgenic Rice Cell Culture Bioreactor
Abstract
1. Introduction
2. Results and Discussion
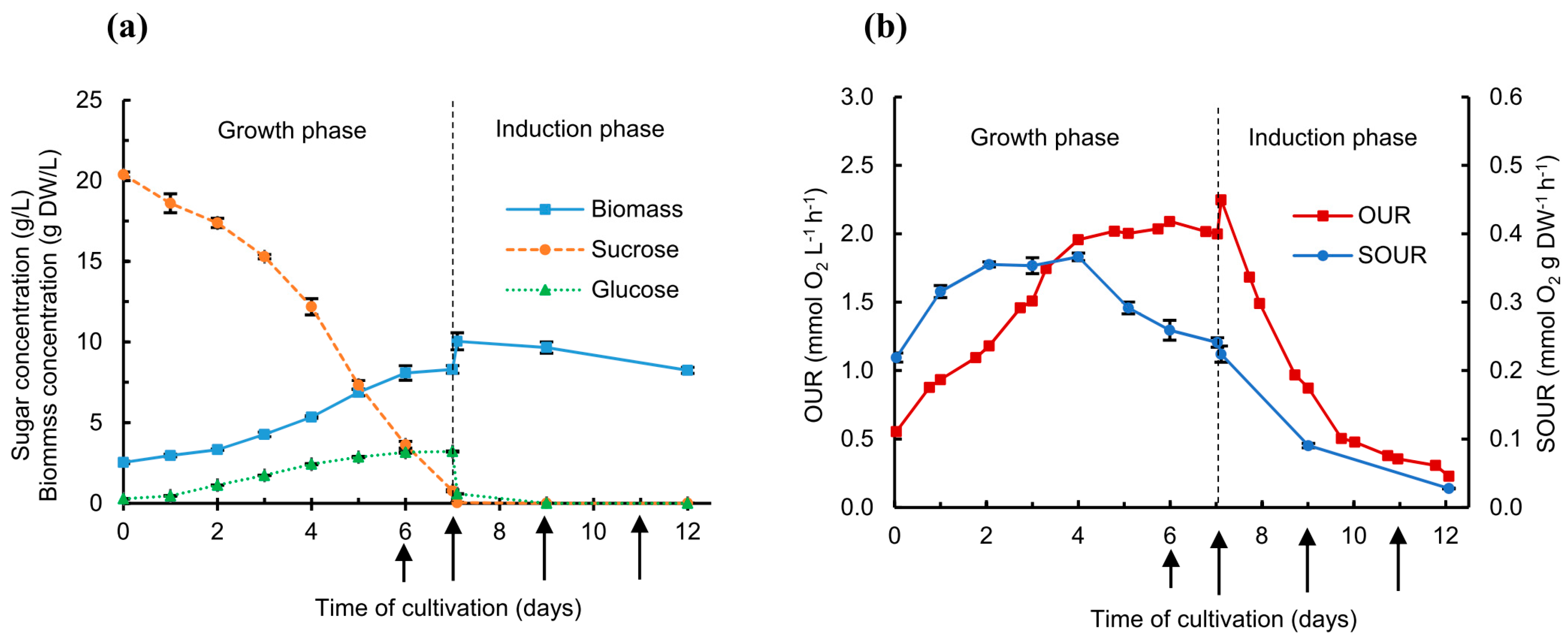
2.1. Cell Growth Kinetics, Sugar Consumption, and Oxygen Uptake Rate with Kifunensine Addition
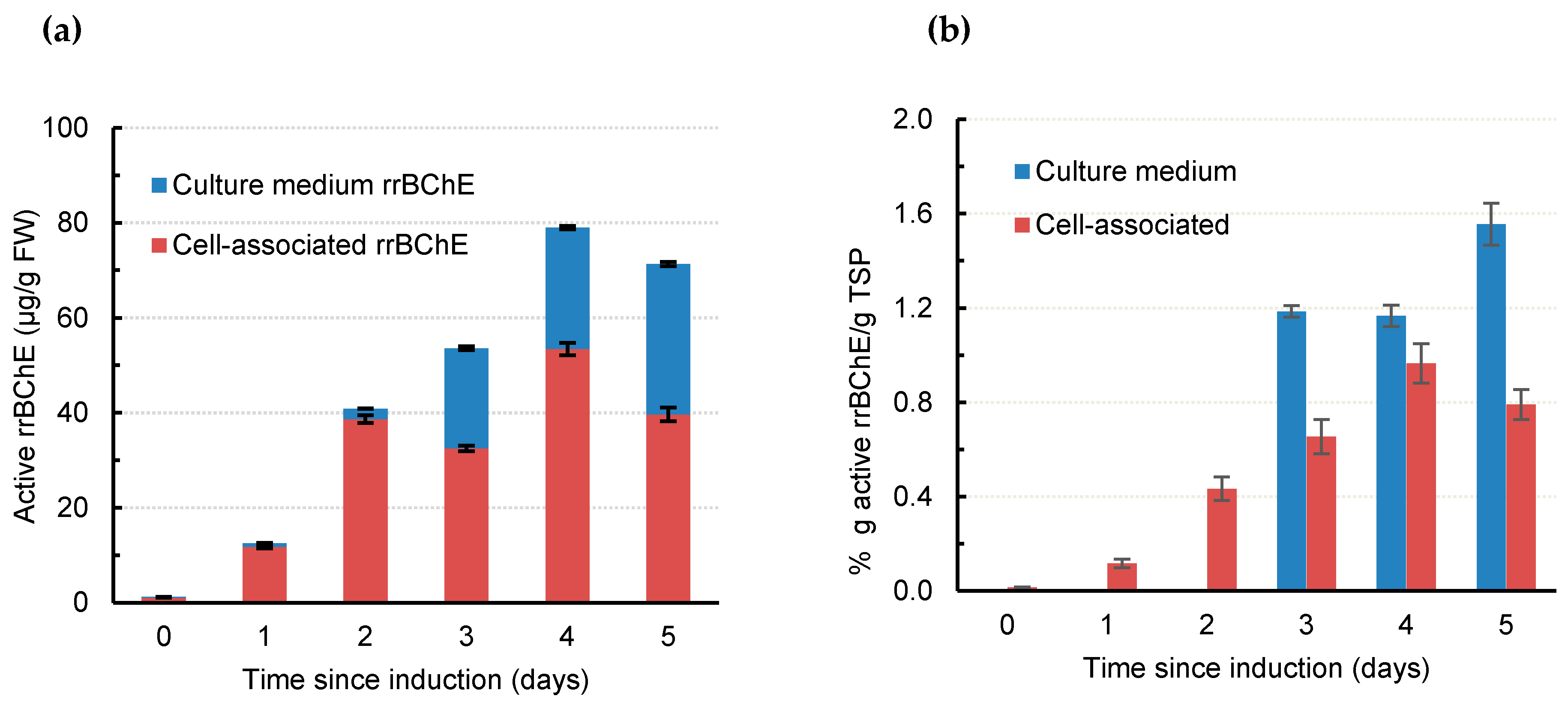
2.2. rrBChE Production and Recombinant Protein Purity in the Presence of Kifunensine
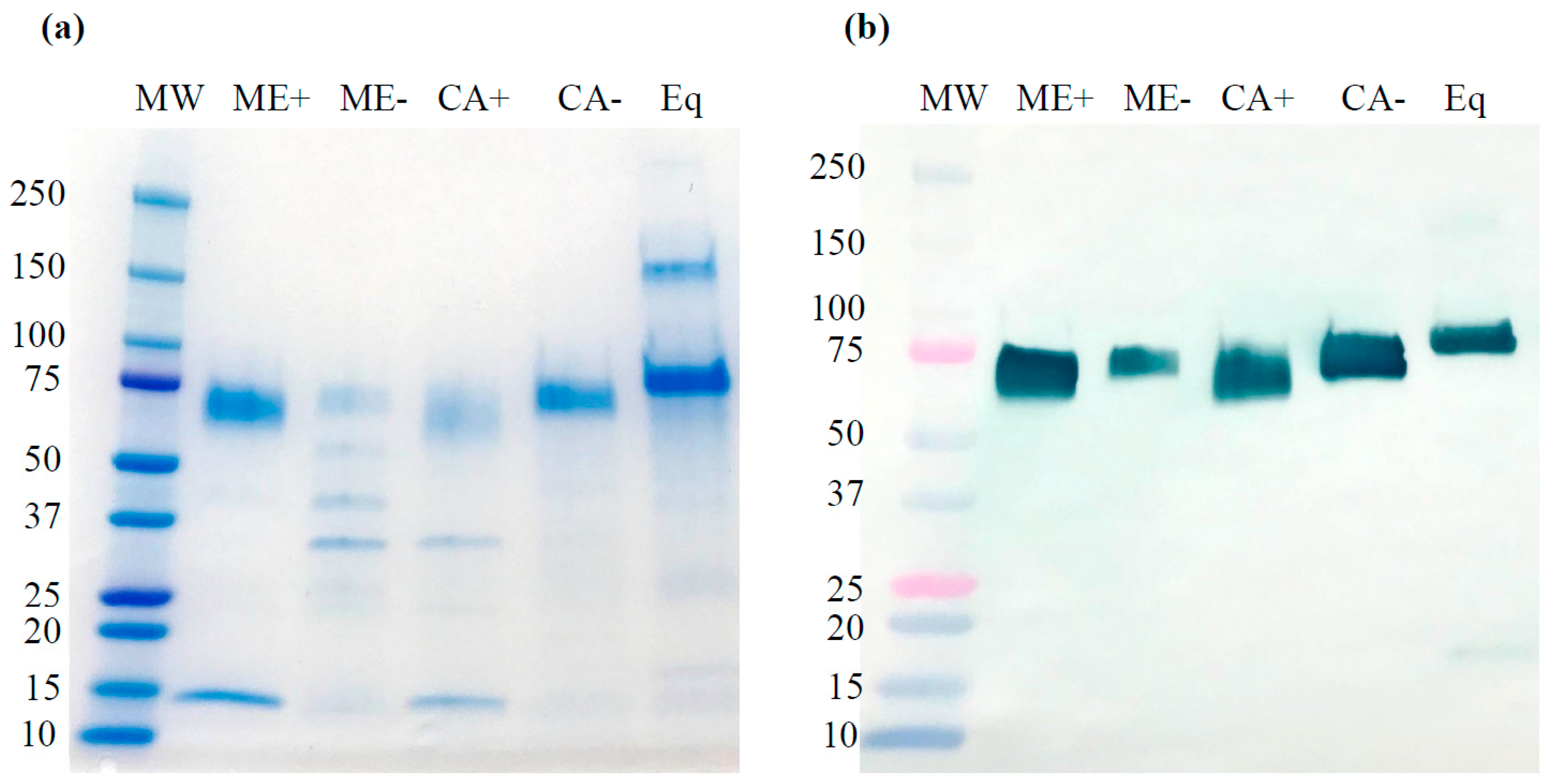
2.3. Purification, SDS-PAGE, and Western Blot Analysis of rrBChE with/without Kifunensine Treatment
2.4. Site-Specific N-glycosylation Analysis
3. Materials and Methods
3.1. Transgenic Rice Cell Suspension Inoculum
3.2. Bioreactor Operation with Kifunensine Treatment
3.3. Harvesting of Culture Medium rrBChE and Cell-Associated rrBChE
3.4. Biomass Measurements
3.5. Sugar Analysis
3.6. Active rrBChE and Total Protein Quantification
3.7. Purification of rrBChE
3.8. Gel Electrophoresis and Western Blot
3.9. Site-Specific N-glycopeptide Analysis of rrBChE
4. Conclusions and Future Prospects
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Alkanaimsh, S.; Karuppanan, K.; Guerrero, A.; Tu, A.M.; Hashimoto, B.; Hwang, M.S.; Phu, M.L.; Arzola, L.; Lebrilla, C.B.; Dandekar, A.M.; et al. Transient Expression of Tetrameric Recombinant Human Butyrylcholinesterase in Nicotiana benthamiana. Front. Plant Sci. 2016, 7, 1–13. [Google Scholar] [CrossRef]
- Alkanaimsh, S.; Corbin, J.M.; Kailemia, M.J.; Karuppanan, K.; Rodriguez, R.L.; Lebrilla, C.B.; McDonald, K.A.; Nandi, S. Purification and site-specific N-glycosylation analysis of human recombinant butyrylcholinesterase from Nicotiana benthamiana. Biochem. Eng. J. 2019, 142, 58–67. [Google Scholar] [CrossRef]
- Corbin, J.M.; Kailemia, M.J.; Cadieux, C.L.; Alkanaimsh, S.; Karuppanan, K.; Rodriguez, R.L.; Lebrilla, C.B.; Cerasoli, D.M.; McDonald, K.A.; Nandi, S. Purification, characterization, and N-glycosylation of recombinant butyrylcholinesterase from transgenic rice cell suspension cultures. Biotechnol. Bioeng. 2018, 115, 1301–1310. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Li, Q.; Kailemia, M.; Lebrilla, C.; Nandi, S.; McDonald, K. Glycoform Modification of Secreted Recombinant Glycoproteins through Kifunensine Addition during Transient Vacuum Agroinfiltration. Int. J. Mol. Sci. 2018, 19, 890. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Karuppanan, K.; Bernardi, A.; Li, Q.; Kommineni, V.; Dandekar, A.M.; Lebrilla, C.B.; Faller, R.; McDonald, K.A.; Nandi, S. Effects of N-Glycosylation on the Structure, Function, and Stability of a Plant-Made Fc-Fusion Anthrax Decoy Protein. Front. Plant Sci. 2019, 10, 768. [Google Scholar] [CrossRef]
- Helenius, A.; Aebi, M. Intracellular Functions of N-Linked Glycans. Science. 2001, 291, 2364–2369. [Google Scholar] [CrossRef]
- Ruiz-May, E.; Kim, S.J.; Brandizzi, F.; Rose, J. The Secreted Plant N-Glycoproteome and Associated Secretory Pathways. Front. Plant. Sci. 2012, 3, 117. [Google Scholar] [CrossRef]
- Gomord, V.; Fitchette, A.C.; Menu-Bouaouiche, L.; Saint-Jore-Dupas, C.; Plasson, C.; Michaud, D.; Faye, L. Plant-specific glycosylation patterns in the context of therapeutic protein production. Plant Biotechnol. J. 2010, 8, 564–587. [Google Scholar] [CrossRef]
- Shaaltiel, Y.; Tekoah, Y. Plant specific N-glycans do not have proven adverse effects in humans. Nat. Biotechnol. 2016, 34, 706–708. [Google Scholar] [CrossRef]
- Mercx, S.; Smargiasso, N.; Chaumont, F.; De Pauw, E.; Boutry, M.; Navarre, C. Inactivation of the β(1,2)-xylosyltransferase and the α(1,3)-fucosyltransferase genes in Nicotiana tabacum BY-2 Cells by a Multiplex CRISPR/Cas9 Strategy Results in Glycoproteins without Plant-Specific Glycans. Front. Plant Sci. 2017, 8, 403. [Google Scholar] [CrossRef]
- Hanania, U.; Ariel, T.; Tekoah, Y.; Fux, L.; Sheva, M.; Gubbay, Y.; Weiss, M.; Oz, D.; Azulay, Y.; Turbovski, A.; et al. Establishment of a tobacco BY2 cell line devoid of plant-specific xylose and fucose as a platform for the production of biotherapeutic proteins. Plant Biotechnol. J. 2017, 15, 1120–1129. [Google Scholar] [CrossRef] [PubMed]
- Jansing, J.; Sack, M.; Augustine, S.M.; Fischer, R.; Bortesi, L. CRISPR/Cas9-mediated knockout of six glycosyltransferase genes in Nicotiana benthamiana for the production of recombinant proteins lacking β-1,2-xylose and core α-1,3-fucose. Plant Biotechnol. J. 2019, 17, 350–361. [Google Scholar] [CrossRef] [PubMed]
- Strasser, R.; Stadlmann, J.; Schähs, M.; Stiegler, G.; Quendler, H.; Mach, L.; Glössl, J.; Weterings, K.; Pabst, M.; Steinkellner, H. Generation of glyco-engineered Nicotiana benthamiana for the production of monoclonal antibodies with a homogeneous human-like N-glycan structure. Plant Biotechnol. J. 2008, 6, 392–402. [Google Scholar] [CrossRef] [PubMed]
- Cox, K.M.; Sterling, J.D.; Regan, J.T.; Gasdaska, J.R.; Frantz, K.K.; Peele, C.G.; Black, A.; Passmore, D.; Moldovan-Loomis, C.; Srinivasan, M.; et al. Glycan optimization of a human monoclonal antibody in the aquatic plant Lemna minor. Nat. Biotechnol. 2006, 24, 1591–1597. [Google Scholar] [CrossRef]
- Sourrouille, C.; Marquet-Blouin, E.; D’Aoust, M.-A.; Kiefer-Meyer, M.-C.; Seveno, M.; Pagny-Salehabadi, S.; Bardor, M.; Durambur, G.; Lerouge, P.; Vezina, L.; et al. Down-regulated expression of plant-specific glycoepitopes in alfalfa. Plant Biotechnol. J. 2008, 6, 702–721. [Google Scholar] [CrossRef]
- Shin, Y.-J.; Chong, Y.-J.; Yang, M.-S.; Kwon, T.-H. Production of recombinant human granulocyte macrophage-colony stimulating factor in rice cell suspension culture with a human-like N-glycan structure. Plant Biotechnol. J. 2011, 9, 1109–1119. [Google Scholar] [CrossRef]
- Sim, J.S.; Lee, Y.H.; Chung, H.Y.; Kim, Y.H.; Hahn, B.S. RNA interference-mediated knock-down of α1,3-fucose and β1,2-xylose in rice N-glycans. J. Plant Biochem. Biotechnol. 2015, 24, 242–248. [Google Scholar] [CrossRef]
- Kajiura, H.; Wasai, M.; Kasahara, S.; Takaiwa, F.; Fujiyama, K. N-glycosylation and N-glycan moieties of CTB expressed in rice seeds. Mol. Biotechnol. 2013, 54, 784–794. [Google Scholar] [CrossRef]
- Tekoah, Y.; Ko, K.; Koprowski, H.; Harvey, D.J.; Wormald, M.R.; Dwek, R.A.; Rudd, P.M. Controlled glycosylation of therapeutic antibodies in plants. Arch. Biochem. Biophys. 2004, 426, 266–278. [Google Scholar] [CrossRef]
- Sriraman, R.; Bardor, M.; Sack, M.; Vaquero, C.; Faye, L.; Fischer, R.; Finnern, R.; Lerouge, P. Recombinant anti-hCG antibodies retained in the endoplasmic reticulum of transformed plants lack core-xylose and core-α(1,3)-fucose residues. Plant Biotechnol. J. 2004, 2, 279–287. [Google Scholar] [CrossRef]
- Shaaltiel, Y.; Bartfeld, D.; Hashmueli, S.; Baum, G.; Brill-Almon, E.; Galili, G.; Dym, O.; Boldin-Adamsky, S.A.; Silman, I.; Sussman, J.L.; et al. Production of glucocerebrosidase with terminal mannose glycans for enzyme replacement therapy of Gaucher’s disease using a plant cell system. Plant Biotechnol. J. 2007, 5, 579–590. [Google Scholar] [CrossRef] [PubMed]
- Fujiyama, K.; Misaki, R.; Sakai, Y.; Omasa, T.; Seki, T. Change in glycosylation pattern with extension of endoplasmic reticulum retention signal sequence of mouse antibody produced by suspension-cultured tobacco BY2 cells. J. Biosci. Bioeng. 2009, 107, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Kommineni, V.; Markert, M.; Ren, Z.; Palle, S.; Carrillo, B.; Deng, J.; Tejeda, A.; Nandi, S.; McDonald, K.A.; Marcel, S.; et al. In Vivo Glycan Engineering via the Mannosidase I Inhibitor (Kifunensine) Improves Efficacy of Rituximab Manufactured in Nicotiana benthamiana Plants. Int. J. Mol. Sci. 2019, 20, 194. [Google Scholar] [CrossRef] [PubMed]
- Roychowdhury, S.; Oh, Y.J.; Kajiura, H.; Hamorsky, K.T.; Fujiyama, K.; Matoba, N. Hydroponic Treatment of Nicotiana benthamiana with Kifunensine Modifies the N-glycans of Recombinant Glycoprotein Antigens to Predominantly Man9 High-Mannose Type upon Transient Overexpression. Front. Plant Sci. 2018, 9, 62. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.-Y.; Park, H.; Hong, J.K.; Kim, S.-D.; Kwon, J.-Y.; You, S.; Do, J.; Lee, D.-Y.; Kim, H.H.; Kim, D.-I. N-glycan Remodeling Using Mannosidase Inhibitors to Increase High-mannose Glycans on Acid α-Glucosidase in Transgenic Rice Cell Cultures. Sci. Rep. 2018, 8, 16130. [Google Scholar] [CrossRef] [PubMed]
- Hamorsky, K.T.; Kouokam, J.C.; Jurkiewicz, J.M.; Nelson, B.; Moore, L.J.; Husk, A.S.; Kajiura, H.; Fujiyama, K.; Matoba, N. N-Glycosylation of cholera toxin B subunit in Nicotiana benthamiana: Impacts on host stress response, production yield and vaccine potential. Sci. Rep. 2015, 5, 8003. [Google Scholar] [CrossRef]
- Bennett, L.; Yang, Q.; Berquist, B.; Giddens, J.; Ren, Z.; Kommineni, V.; Murray, R.; White, E.; Holtz, B.; Wang, L.-X.; et al. Implementation of Glycan Remodeling to Plant-Made Therapeutic Antibodies. Int. J. Mol. Sci. 2018, 19, 421. [Google Scholar] [CrossRef]
- Elbein, A.D.; Tropea, J.E.; Mitchell, M.; Kaushal, G.P. Kifunensine, a potent inhibitor of the glycoprotein processing mannosidase I. J. Biol. Chem. 1990, 265, 15599–15605. [Google Scholar]
- Nandi, S.; Khush, G.S. Strategies to Increase Heterologous Protein Expression in Rice Grains. In Recent Advancements in Gene Expression and Enabling Technologies in Crop Plants; Azhakanandam, K., Silverstone, A., Daniell, H., Davey, M.R., Eds.; Springer: New York, NY, USA, 2015; pp. 241–262. ISBN 978-1-4939-2202-4. [Google Scholar]
- Ranjhan, S.; Karrer, E.E.; Rodriguez, R.L. Localizing a-Amylase Gene Expression in Germinated Rice Grains. Plant Cell Physiol. 1992, 33, 73–79. [Google Scholar] [CrossRef]
- Rodriguez, R.L.; Huang, N.; Sutliff, T.; Ranjhan, S.; Karrer, E.; Litts, J. Organization, structure, and expression of the rice α- amylase multigene family. In Rice Genetics II: Rice Genetics Collection; Banta, S.J., Argosino, G.S., Eds.; World Scientific Publishing Company: Singapore, Singapore, 2008; Volume 2, pp. 417–429. ISBN 978-981-281-866-9. [Google Scholar]
- Huang, N.; Koizumi, N.; Reinl, S.; Rodriguez, R.L. Structural organization and differential expression of rice α-amylase genes. Nucleic Acids Res. 1990, 18, 7007–7014. [Google Scholar] [CrossRef]
- Mitsunaga, S.; Rodriguez, R.L.; Yamaguchi, J. Sequence-specific interactions of a nuclear protein factor with the promoter region of a rice gene for α-amylase, RAmy3D. Nucleic Acids Res. 1994, 22, 1948–1953. [Google Scholar] [CrossRef] [PubMed]
- Karrer, E.; Rodriguez, R. Metabolic regulation of rice alpha-amylase and sucrose synthase genes in planta. Plant J. 1992, 2, 517–523. [Google Scholar] [CrossRef] [PubMed]
- Huang, N.; Chandler, J.; Thomas, B.R.; Koizumi, N.; Rodriguez, R.L. Metabolic regulation of α-amylase gene expression in transgenic cell cultures of rice (Oryza sativa L.). Plant Mol. Biol. 1993, 23, 737–747. [Google Scholar] [CrossRef] [PubMed]
- Terashima, M.; Murai, Y.; Kawamura, M.; Nakanishi, S.; Stoltz, T.; Chen, L.; Drohan, W.; Rodriguez, R.L.; Katoh, S. Production of functional human α1-antitrypsin by plant cell culture. Appl. Microbiol. Biotechnol. 1999, 52, 516–523. [Google Scholar] [CrossRef]
- Huang, J.; Sutliff, T.D.; Wu, L.; Nandi, S.; Benge, K.; Terashima, M.; Ralston, A.H.; Drohan, W.; Huang, N.; Rodriguez, R.L. Expression and Purification of Functional Human α-1-Antitrypsin from Cultured Plant Cells. Biotechnol. Prog. 2001, 17, 126–133. [Google Scholar] [CrossRef]
- Trexler, M.M.; McDonald, K.A.; Jackman, A.P. A Cyclical Semicontinuous Process for Production of Human α1-Antitrypsin Using Metabolically Induced Plant Cell Suspension Cultures. Biotechnol. Prog. 2005, 21, 321–328. [Google Scholar] [CrossRef]
- Corbin, J.M.; Hashimoto, B.I.; Karuppanan, K.; Kyser, Z.R.; Wu, L.; Roberts, B.A.; Noe, A.R.; Rodriguez, R.L.; McDonald, K.A.; Nandi, S. Semicontinuous Bioreactor Production of Recombinant Butyrylcholinesterase in Transgenic Rice Cell Suspension Cultures. Front. Plant Sci. 2016, 7, 412. [Google Scholar] [CrossRef]
- McDonald, K.A.; Lo, M.H.; Trombly, D.M.; Xie, Q.; Jackman, A.P. Production of human α-1-antitrypsin from transgenic rice cell culture in a membrane bioreactor. Biotechnol. Prog. 2005, 21, 728–734. [Google Scholar] [CrossRef]
- Lockridge, O. Review of human butyrylcholinesterase structure, function, genetic variants, history of use in the clinic, and potential therapeutic uses. Pharmacol. Ther. 2015, 148, 34–46. [Google Scholar] [CrossRef]
- Trexler, M.M.; McDonald, K.A.; Jackman, A.P. Bioreactor Production of Human α1-Antitrypsin Using Metabolically Regulated Plant Cell Cultures. Biotechnol. Prog. 2002, 18, 501–508. [Google Scholar] [CrossRef]
- Macharoen, K.; McDonald, K.A.; Nandi, S. Simplified bioreactor processes for recombinant butyrylcholinesterase production in transgenic rice cell suspension cultures. Biochem. Eng. J. 2020, 163, 107751. [Google Scholar] [CrossRef]
- Park, C.I.; Lee, S.J.; Kang, S.H.; Jung, H.S.; Kim, D.I.; Lim, S.M. Fed-batch cultivation of transgenic rice cells for the production of hCTLA4Ig using concentrated amino acids. Process. Biochem. 2010, 45, 67–74. [Google Scholar] [CrossRef]
- Liebminger, E.; Hüttner, S.; Vavra, U.; Fischl, R.; Schoberer, J.; Grass, J.; Blaukopf, C.; Seifert, G.J.; Altmann, F.; Mach, L.; et al. Class I α-Mannosidases Are Required for N-Glycan Processing and Root Development in Arabidopsis thaliana. Plant Cell 2009, 21, 3850–3867. [Google Scholar] [CrossRef] [PubMed]
- Moussalli, M.; Pipe, S.W.; Hauri, H.-P.; Nichols, W.C.; Ginsburg, D.; Kaufman, R.J. Mannose-dependent Endoplasmic Reticulum (ER)-Golgi Intermediate Compartment-53-mediated ER to Golgi Trafficking of Coagulation Factors V and VIII. J. Biol. Chem. 1999, 274, 32539–32542. [Google Scholar] [CrossRef]
- Marcus, N.Y.; Perlmutter, D.H. Glucosidase and Mannosidase Inhibitors Mediate Increased Secretion of Mutant α1 Antitrypsin Z. J. Biol. Chem. 2000, 275, 1987–1992. [Google Scholar] [CrossRef]
- Onder, S.; David, E.; Tacal, O.; Schopfer, L.M.; Lockridge, O. Hupresin Retains Binding Capacity for Butyrylcholinesterase and Acetylcholinesterase after Sanitation with Sodium Hydroxide. Front. Pharmacol. 2017, 8, 713. [Google Scholar] [CrossRef]
- Kolarich, D.; Weber, A.; Pabst, M.; Stadlmann, J.; Teschner, W.; Ehrlich, H.; Schwarz, H.-P.; Altmann, F. Glycoproteomic characterization of butyrylcholinesterase from human plasma. Proteomics 2008, 8, 254–263. [Google Scholar] [CrossRef]
- Bernardi, A.; Kirschner, K.N.; Faller, R. Structural analysis of human glycoprotein butyrylcholinesterase using atomistic molecular dynamics: The importance of glycosylation site ASN241. PLoS ONE 2017, 12, e0187994. [Google Scholar] [CrossRef]
- McDonald, K.A.; Jackman, A.P.; Hurst, S. Characterization of plant suspension cultures using the focused beam reflectance technique. Biotechnol. Lett. 2001, 23, 317–324. [Google Scholar] [CrossRef]
- Huang, Y.-J.; Huang, Y.; Baldassarre, H.; Wang, B.; Lazaris, A.; Leduc, M.; Bilodeau, A.S.; Bellemare, A.; Cote, M.; Herskovits, P.; et al. Recombinant human butyrylcholinesterase from milk of transgenic animals to protect against organophosphate poisoning. Proc. Natl. Acad. Sci. USA 2007, 104, 13603–13608. [Google Scholar] [CrossRef]
- Ellman, G.L.; Courtney, K.D.; Andres, V.; Featherstone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar] [CrossRef]
- Corbin, J.M.; McNulty, M.J.; Macharoen, K.; McDonald, K.A.; Nandi, S. Techno-economic analysis of semicontinuous bioreactor production of biopharmaceuticals in transgenic rice cell suspension cultures. Biotechnol. Bioeng. 2020, 117, 3053–3065. [Google Scholar] [CrossRef]
 ), N-acetylglucosamine; green circle (
), N-acetylglucosamine; green circle ( ), mannose; yellow circle (
), mannose; yellow circle ( ), galactose; red triangle (
), galactose; red triangle ( ), fucose; orange star (
), fucose; orange star ( ), xylose. (c) The relative abundance (%) of overall (combining all sites) N-glycoform distributions of rrBChE with and without kifunensine treatment. CA and ME stand for cell-associated rrBChE and culture medium rrBChE, respectively, while + and - refer to with and without kifunensine treatment, respectively.
), xylose. (c) The relative abundance (%) of overall (combining all sites) N-glycoform distributions of rrBChE with and without kifunensine treatment. CA and ME stand for cell-associated rrBChE and culture medium rrBChE, respectively, while + and - refer to with and without kifunensine treatment, respectively.
 ), N-acetylglucosamine; green circle (
), N-acetylglucosamine; green circle ( ), mannose; yellow circle (
), mannose; yellow circle ( ), galactose; red triangle (
), galactose; red triangle ( ), fucose; orange star (
), fucose; orange star ( ), xylose. (c) The relative abundance (%) of overall (combining all sites) N-glycoform distributions of rrBChE with and without kifunensine treatment. CA and ME stand for cell-associated rrBChE and culture medium rrBChE, respectively, while + and - refer to with and without kifunensine treatment, respectively.
), xylose. (c) The relative abundance (%) of overall (combining all sites) N-glycoform distributions of rrBChE with and without kifunensine treatment. CA and ME stand for cell-associated rrBChE and culture medium rrBChE, respectively, while + and - refer to with and without kifunensine treatment, respectively.

| tgrowth (Days) | x0 (g DW/L) | xf (g DW/L) | µmax (Day−1) | τD (Days) | Max OUR (mmol O2 L−1 h−1) | Max SOUR (mmol O2 g DW−1 h−1) |
|---|---|---|---|---|---|---|
| 7 | 2.53 ± 0.08 | 8.29 ± 0.24 | 0.21 ± 0.02 | 3.3 ± 0.4 | 2.09 ± 0.01 | 0.37 ± 0.01 |
| tinduction (Days) | x’0 (g DW/L) | x’f (g DW/L) | g FW/ g DW Ratio† | Max total rrBChE Production (µg/g FW; mg/L) | Max Volumetric Productivity † (µg L−1 Day−1) | Max Specific Productivity † (μg g DW−1 Day−1) |
|---|---|---|---|---|---|---|
| 5 | 10.04 ± 0.53 | 8.24 ± 0.19 | 11.5 ± 0.3 | 79.0 ± 2.2; 7.5 ± 0.4 | 680 ± 34 | 82.5 ± 4.6 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Macharoen, K.; Li, Q.; Márquez-Escobar, V.A.; Corbin, J.M.; Lebrilla, C.B.; Nandi, S.; McDonald, K.A. Effects of Kifunensine on Production and N-Glycosylation Modification of Butyrylcholinesterase in a Transgenic Rice Cell Culture Bioreactor. Int. J. Mol. Sci. 2020, 21, 6896. https://doi.org/10.3390/ijms21186896
Macharoen K, Li Q, Márquez-Escobar VA, Corbin JM, Lebrilla CB, Nandi S, McDonald KA. Effects of Kifunensine on Production and N-Glycosylation Modification of Butyrylcholinesterase in a Transgenic Rice Cell Culture Bioreactor. International Journal of Molecular Sciences. 2020; 21(18):6896. https://doi.org/10.3390/ijms21186896
Chicago/Turabian StyleMacharoen, Kantharakorn, Qiongyu Li, Veronica A. Márquez-Escobar, Jasmine M. Corbin, Carlito B. Lebrilla, Somen Nandi, and Karen A. McDonald. 2020. "Effects of Kifunensine on Production and N-Glycosylation Modification of Butyrylcholinesterase in a Transgenic Rice Cell Culture Bioreactor" International Journal of Molecular Sciences 21, no. 18: 6896. https://doi.org/10.3390/ijms21186896
APA StyleMacharoen, K., Li, Q., Márquez-Escobar, V. A., Corbin, J. M., Lebrilla, C. B., Nandi, S., & McDonald, K. A. (2020). Effects of Kifunensine on Production and N-Glycosylation Modification of Butyrylcholinesterase in a Transgenic Rice Cell Culture Bioreactor. International Journal of Molecular Sciences, 21(18), 6896. https://doi.org/10.3390/ijms21186896







