New Era of Diacylglycerol Kinase, Phosphatidic Acid and Phosphatidic Acid-Binding Protein
Abstract
1. Introduction
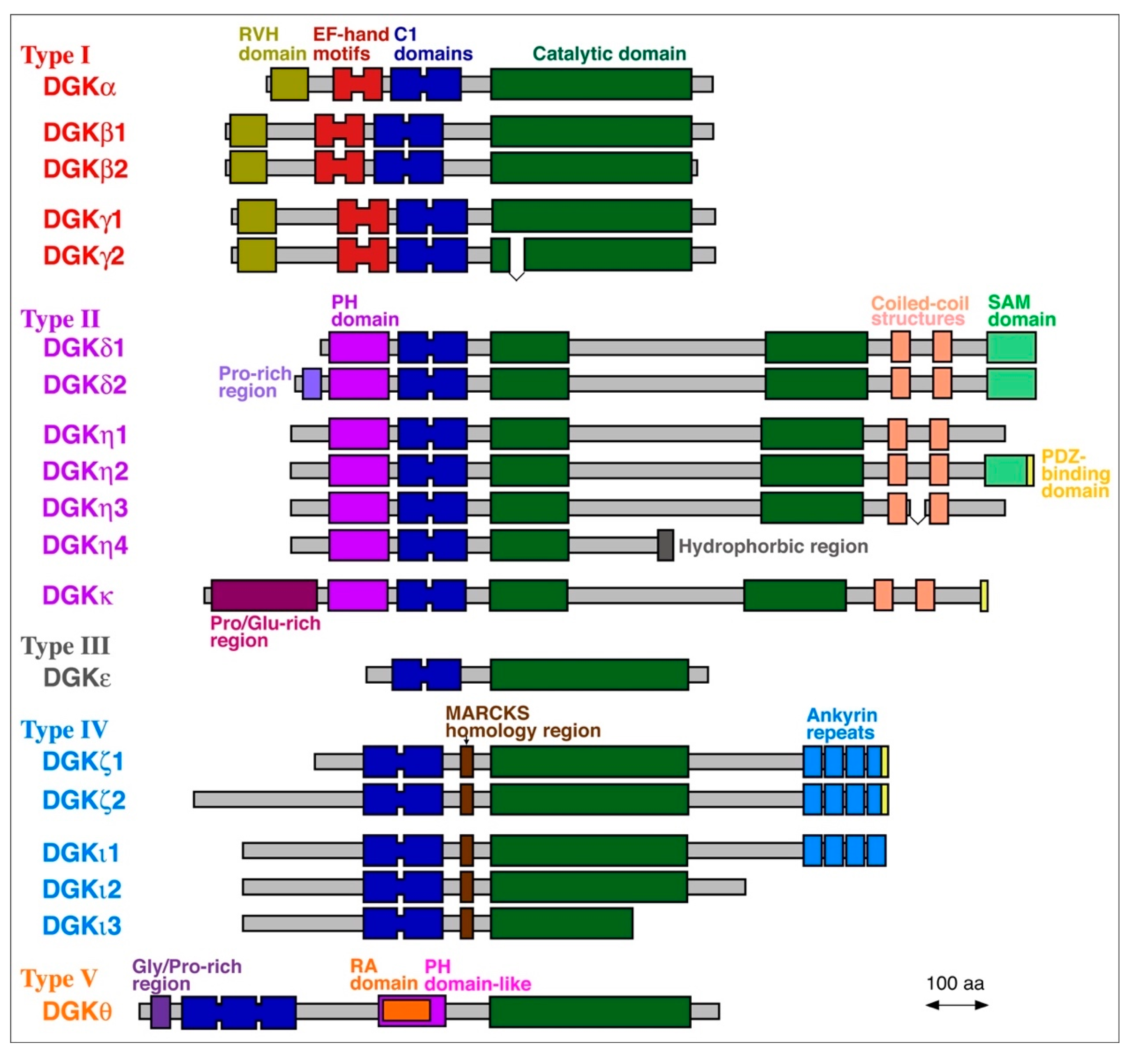
2. PA Molecular Species Produced by DGK Isozymes Except for DGKε
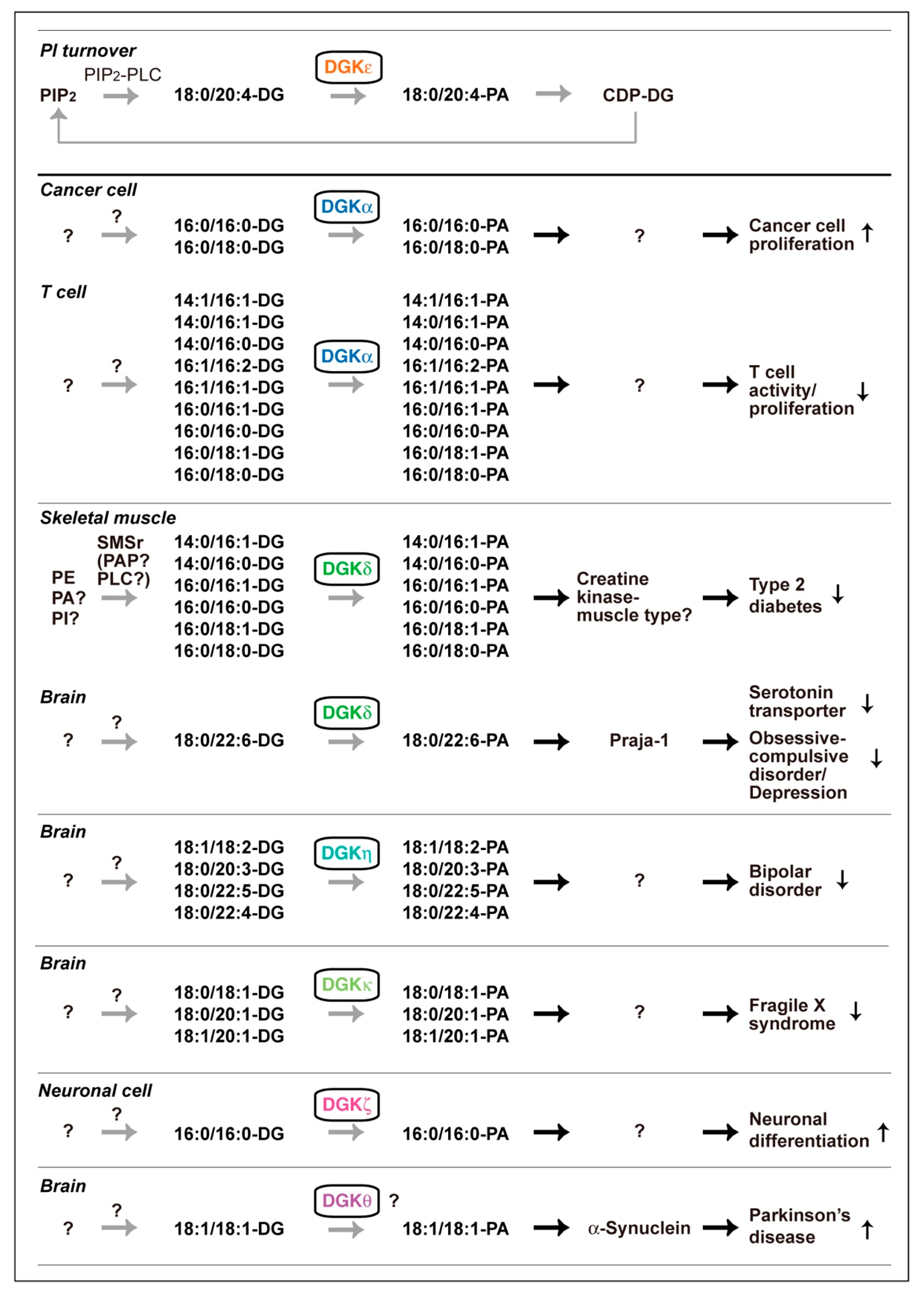
2.1. DGKα
2.2. DGKζ
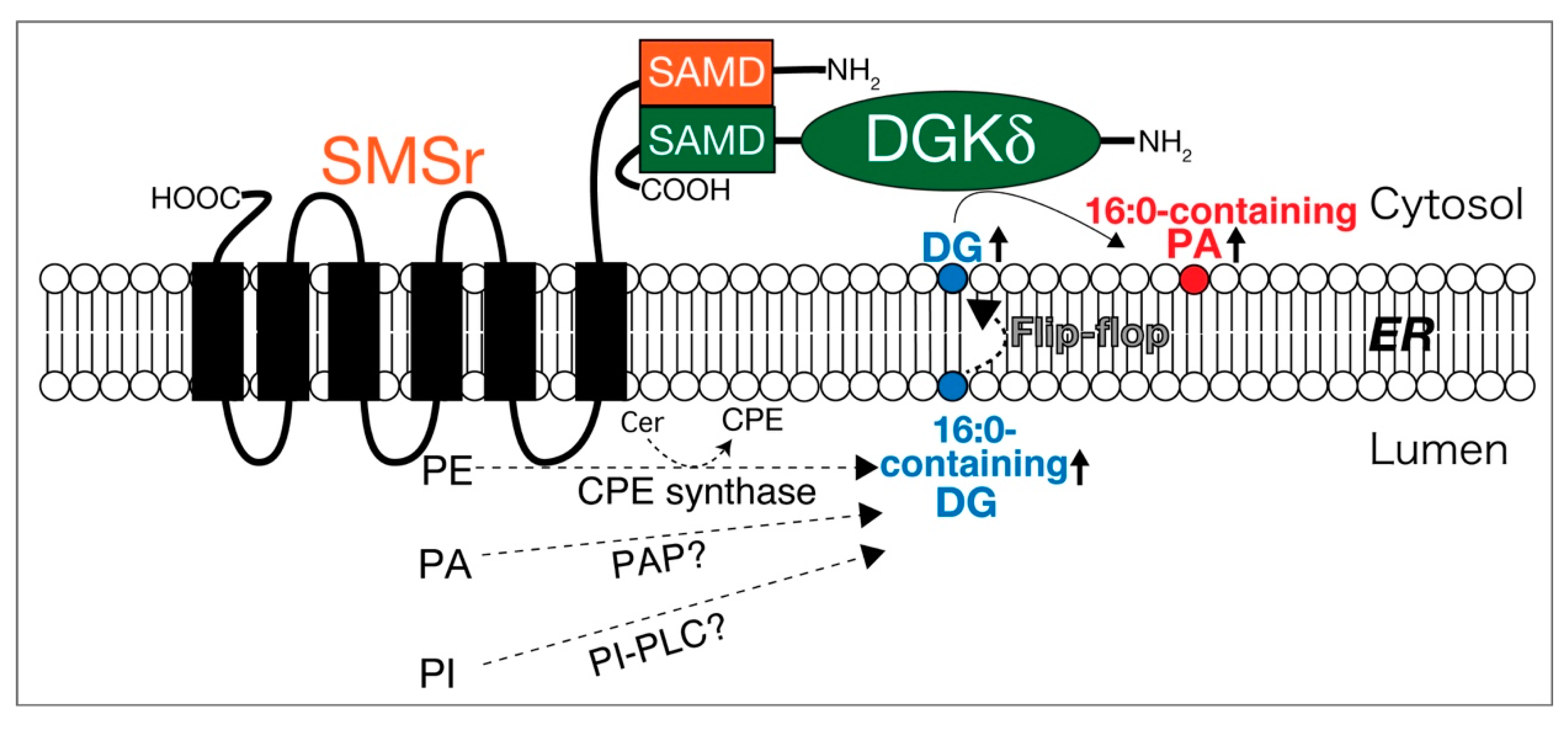
2.3. DGKδ
2.4. DGKη
2.5. DGKκ
2.6. DGKθ
3. Molecular Species Selectivity of PABP
3.1. α-Synuclein
3.2. Praja-1
3.3. Synaptojanin-1
3.4. L-Lactate Dehydrogenase (LDH) A
3.5. CKM
3.6. DGKγ
3.7. Raf-1 (C-Raf)
3.8. mTORC2
3.9. PDE4A1 and A5
3.10. Seipin
3.11. Plant PABPs
4. PA Probe
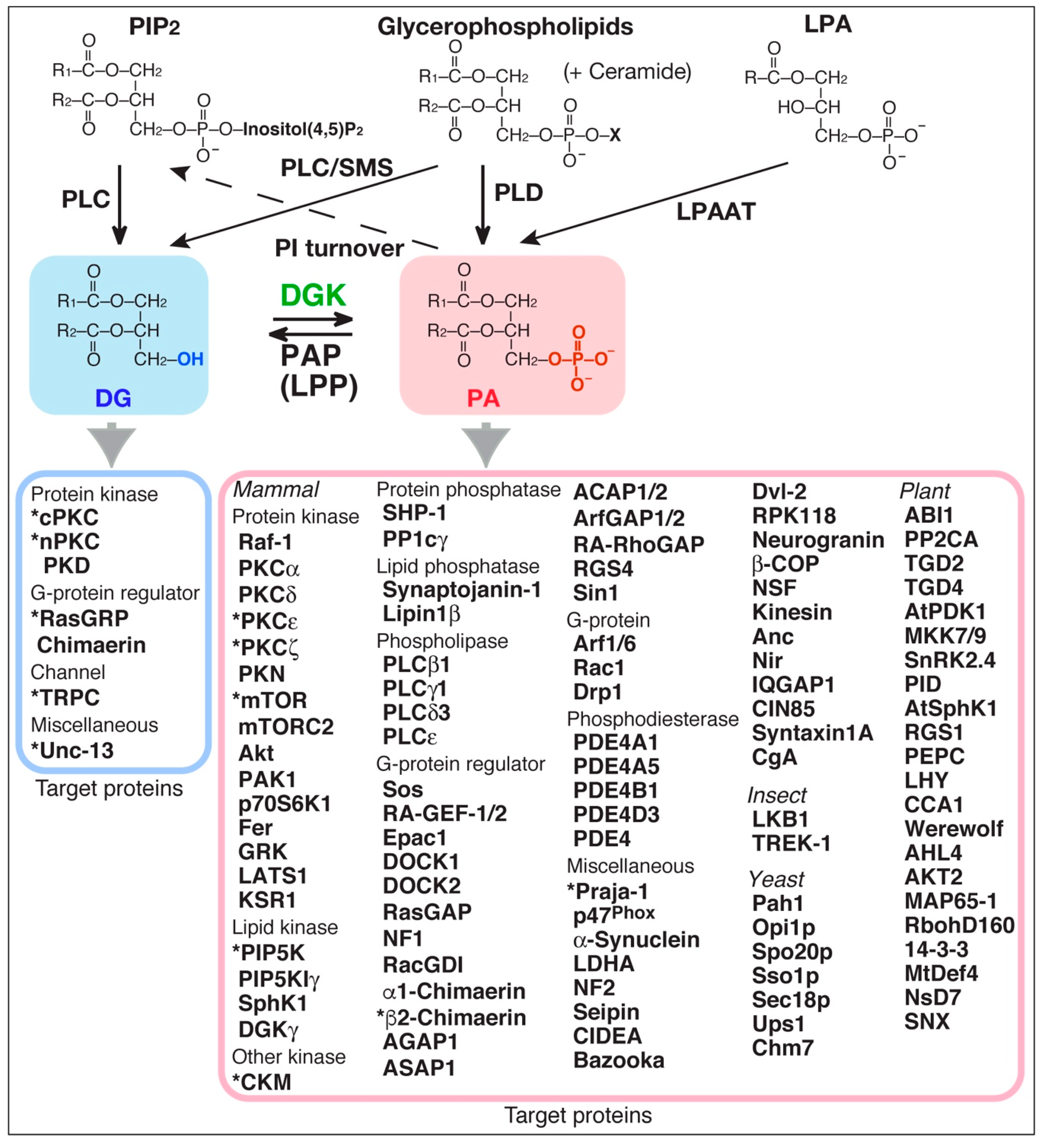
5. DG-Providing Pathway Upstream of DGK
6. Physiological Implication of Diversity of PA Molecular Species and PABPs
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| Arf | ADP-ribosylation factor |
| aPKC | Atypical protein kinase C |
| BPD | Bipolar disorder |
| CDP-DG | Cytidine diphosphate diacylglycerol |
| CL | Cardiolipin |
| CKM | Creatine kinase-muscle type |
| CPE | Ceramide phosphoethanolamine |
| cPKC | Conventional protein kinase C |
| DG | Diacylglycerol |
| DGBP | Diacylglycerol-binding protein |
| DGK | Diacylglycerol kinase |
| DHA | Docosahexaenoic acid |
| ER | Endoplasmic reticulum |
| FMR | Fragile X mental retardation |
| FXS | Fragile X syndrome |
| GRP | Guanyl nucleotide-releasing protein |
| GWAS | Genome-wide association study |
| KO | Knockout |
| LC | Liquid chromatography |
| LDH | Lactate dehydrogenase |
| LPAAT | Lysophosphatidic acid acyltransferase |
| MARCKS | Myristoylated alanine-rich C-kinase substrate |
| MS | Mass spectrometry |
| mTOR | Mammalian target of rapamycin |
| mTORC | mTOR complex |
| MUFA | Monounsaturated fatty acid |
| nPKC | Novel protein kinase C |
| OCD | Obsessive-compulsive disorder |
| PA | Phosphatidic acid |
| PABD | Phosphatidic acid-binding domain |
| PABP | Phosphatidic acid-binding protein |
| PAP | PA phosphatase |
| PC | Phosphatidylcholine |
| PDZ | Postsynaptic density 95, discs large, zonula occludens-1 |
| PE | Phosphatidylethanolamine |
| PG | Phosphatidylglycerol |
| PI | Phosphatidylinositol |
| PIP2 | Phosphatidylinositol 4,5-bisphosphate |
| PIP5K | PI-4-phosphate-5-kinase |
| PH | Pleckstrin homology |
| PKC | Protein kinase C |
| PKD | Protein kinase D |
| PLC | Phospholipase C |
| PLD | Phospholipase D |
| PS | Phosphatidylserine |
| PUFA | Polyunsaturated fatty acid |
| RA | Ras-associated |
| RVH | Recoverin homology |
| SAM | Sterile α motif |
| SFA | Saturated fatty acid |
| SphK | Sphingosine kinase |
| SMSr | Sphingomyelin synthase-related protein |
| Spo20p | Sporulation-specific protein 20 |
| T2D | Type 2 diabetes |
References
- Goto, K.; Hozumi, Y.; Kondo, H. Diacylglycerol, phosphatidic acid, and the converting enzyme, diacylglycerol kinase, in the nucleus. Biochim. Biophys. Acta 2006, 1761, 535–541. [Google Scholar] [CrossRef]
- Merida, I.; Avila-Flores, A.; Merino, E. Diacylglycerol kinases: At the hub of cell signalling. Biochem. J. 2008, 409, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Sakane, F.; Imai, S.; Kai, M.; Yasuda, S.; Kanoh, H. Diacylglycerol kinases: Why so many of them? Biochim. Biophys. Acta 2007, 1771, 793–806. [Google Scholar] [CrossRef] [PubMed]
- Sakane, F.; Mizuno, S.; Takahashi, D.; Sakai, H. Where do substrates of diacylglycerol kinases come from? Diacylglycerol kinases utilize diacylglycerol species supplied from phosphatidylinositol turnover-independent pathways. Adv. Biol. Regul. 2018, 67, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Topham, M.K.; Epand, R.M. Mammalian diacylglycerol kinases: Molecular interactions and biological functions of selected isoforms. Biochim. Biophys. Acta 2009, 1790, 416–424. [Google Scholar] [CrossRef] [PubMed]
- Hurley, J.H.; Newton, A.C.; Parker, P.J.; Blumberg, P.M.; Nishizuka, Y. Taxonomy and function of C1 protein kinase C homology domains. Protein Sci. 1997, 6, 477–480. [Google Scholar] [CrossRef] [PubMed]
- Kazanietz, M.G. Novel “nonkinase” phorbol ester receptors: The C1 domain connection. Mol. Pharmacol. 2002, 61, 759–767. [Google Scholar] [CrossRef]
- Nishizuka, Y. Intracellular signaling by hydrolysis of phospholipids and activation of protein kinase C. Science 1992, 258, 607–614. [Google Scholar] [CrossRef]
- Ron, D.; Kazanietz, M.G. New insights into the regulation of protein kinase C and novel phorbol ester receptors. FASEB J. 1999, 13, 1658–1676. [Google Scholar] [CrossRef]
- Ohno, S.; Nishizuka, Y. Protein kinase C isotypes and their specific functions: Prologue. J. Biochem. 2002, 132, 509–511. [Google Scholar] [CrossRef]
- Bruntz, R.C.; Lindsley, C.W.; Brown, H.A. Phospholipase D signaling pathways and phosphatidic acid as therapeutic targets in cancer. Pharmacol. Rev. 2014, 66, 1033–1079. [Google Scholar] [CrossRef] [PubMed]
- English, D. Phosphatidic acid: A lipid messenger involved in intracellular and extracellular signalling. Cell Signal. 1996, 8, 341–347. [Google Scholar] [CrossRef]
- Exton, J.H. Phosphatidylcholine breakdown and signal transduction. Biochim. Biophys. Acta 1994, 1212, 26–42. [Google Scholar] [CrossRef]
- Kim, S.C.; Wang, X. Phosphatidic acid: An emerging versatile class of cellular mediators. Essays Biochem. 2020. [Google Scholar] [CrossRef]
- Stace, C.L.; Ktistakis, N.T. Phosphatidic acid- and phosphatidylserine-binding proteins. Biochim. Biophys. Acta 2006, 1761, 913–926. [Google Scholar] [CrossRef]
- Zegarlinska, J.; Piascik, M.; Sikorski, A.F.; Czogalla, A. Phosphatidic acid—A simple phospholipid with multiple faces. Acta Biochim. Pol. 2018, 65, 163–171. [Google Scholar] [CrossRef]
- Zhukovsky, M.A.; Filograna, A.; Luini, A.; Corda, D.; Valente, C. Phosphatidic acid in membrane rearrangements. FEBS Lett. 2019, 593, 2428–2451. [Google Scholar] [CrossRef]
- Ghosh, S.; Moore, S.; Bell, R.M.; Dush, M. Functional analysis of a phosphatidic acid binding domain in human Raf-1 kinase: Mutations in the phosphatidate binding domain lead to tail and trunk abnormalities in developing zebrafish embryos. J. Biol. Chem. 2003, 278, 45690–45696. [Google Scholar] [CrossRef]
- Ghosh, S.; Strum, J.C.; Sciorra, V.A.; Daniel, L.; Bell, R.M. Raf-1 kinase possesses distinct binding domains for phosphatidylserine and phosphatidic acid. Phosphatidic acid regulates the translocation of Raf-1 in 12-O-tetradecanoylphorbol-13-acetate-stimulated Madin-Darby canine kidney cells. J. Biol. Chem. 1996, 271, 8472–8480. [Google Scholar] [CrossRef]
- Rizzo, M.A.; Shome, K.; Watkins, S.C.; Romero, G. The recruitment of Raf-1 to membranes is mediated by direct interaction with phosphatidic acid and is independent of association with Ras. J. Biol. Chem. 2000, 275, 23911–23918. [Google Scholar] [CrossRef]
- Corbalan-Garcia, S.; Sanchez-Carrillo, S.; Garcia-Garcia, J.; Gomez-Fernandez, J.C. Characterization of the membrane binding mode of the C2 domain of PKCε. Biochemistry 2003, 42, 11661–11668. [Google Scholar] [CrossRef] [PubMed]
- Jose Lopez-Andreo, M.; Gomez-Fernandez, J.C.; Corbalan-Garcia, S. The simultaneous production of phosphatidic acid and diacylglycerol is essential for the translocation of protein kinase Cε to the plasma membrane in RBL-2H3 cells. Mol. Biol. Cell 2003, 14, 4885–4895. [Google Scholar] [CrossRef] [PubMed]
- Limatola, C.; Schaap, D.; Moolenaar, W.H.; van Blitterswijk, W.J. Phosphatidic acid activation of protein kinase C-ζ overexpressed in COS cells: Comparison with other protein kinase C isotypes and other acidic lipids. Biochem. J. 1994, 304, 1001–1008. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Vilella-Bach, M.; Bachmann, R.; Flanigan, A.; Chen, J. Phosphatidic acid-mediated mitogenic activation of mTOR signaling. Science 2001, 294, 1942–1945. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, G.H.; Fisette, P.L.; Anderson, R.A. Type I phosphatidylinositol 4-phosphate 5-kinase isoforms are specifically stimulated by phosphatidic acid. J. Biol. Chem. 1994, 269, 11547–11554. [Google Scholar]
- Moritz, A.; DeGraan, P.N.E.; Gispen, W.H.; Wirtz, K.W.A. Phosphatidic acid is a specific activator of phosphatidylinositol-4-phosphate kinase. J. Biol. Chem. 1992, 267, 7207–7210. [Google Scholar]
- Delon, C.; Manifava, M.; Wood, E.; Thompson, D.; Krugmann, S.; Pyne, S.; Ktistakis, N.T. Sphingosine kinase 1 is an intracellular effector of phosphatidic acid. J. Biol. Chem. 2004, 279, 44763–44774. [Google Scholar] [CrossRef]
- Jones, J.A.; Hannun, Y.A. Tight binding inhibition of protein phosphatase-1 by phosphatidic acid. Specificity of inhibition by the phospholipid. J. Biol. Chem. 2002, 277, 15530–15538. [Google Scholar] [CrossRef]
- Ren, H.; Federico, L.; Huang, H.; Sunkara, M.; Drennan, T.; Frohman, M.A.; Smyth, S.S.; Morris, A.J. A phosphatidic acid binding/nuclear localization motif determines lipin1 function in lipid metabolism and adipogenesis. Mol. Biol. Cell 2010, 21, 3171–3181. [Google Scholar] [CrossRef]
- Jones, G.A.; Carpenter, G. The regulation of phospholipase C-γ 1 by phosphatidic acid. Assessment of kinetic parameters. J. Biol. Chem. 1993, 268, 20845–20850. [Google Scholar]
- Tsai, M.H.; Yu, C.L.; Stacey, D.W. A cytoplasmic protein inhibits the GTPase activity of H-Ras in a phospholipid-dependent manner. Science 1990, 250, 982–985. [Google Scholar] [CrossRef]
- Ahmed, S.; Lee, J.; Kozma, R.; Best, A.; Monfries, C.; Lim, L. A novel functional target for tumor-promoting phorbol esters and lysophosphatidic acid. The p21rac-GTPase activating protein n-chimaerin. J. Biol. Chem. 1993, 268, 10709–10712. [Google Scholar] [PubMed]
- Caloca, M.J.; Wang, H.; Kazanietz, M.G. Characterization of the Rac-GAP (Rac-GTPase-activating protein) activity of β2-chimaerin, a ‘non-protein kinase C′ phorbol ester receptor. Biochem. J. 2003, 375, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Manifava, M.; Thuring, J.W.; Lim, Z.Y.; Packman, L.; Holmes, A.B.; Ktistakis, N.T. Differential binding of traffic-related proteins to phosphatidic acid- or phosphatidylinositol (4,5)- bisphosphate-coupled affinity reagents. J. Biol. Chem. 2001, 276, 8987–8994. [Google Scholar] [CrossRef] [PubMed]
- Chae, Y.C.; Kim, J.H.; Kim, K.L.; Kim, H.W.; Lee, H.Y.; Heo, W.D.; Meyer, T.; Suh, P.G.; Ryu, S.H. Phospholipase D activity regulates integrin-mediated cell spreading and migration by inducing GTP-Rac translocation to the plasma membrane. Mol. Biol. Cell 2008, 19, 3111–3123. [Google Scholar] [CrossRef]
- Maxwell, K.N.; Zhou, Y.; Hancock, J.F. Rac1 Nanoscale Organization on the Plasma Membrane Is Driven by Lipid Binding Specificity Encoded in the Membrane Anchor. Mol. Cell. Biol. 2018, 38, e00186-18. [Google Scholar] [CrossRef]
- Baillie, G.S.; Huston, E.; Scotland, G.; Hodgkin, M.; Gall, I.; Peden, A.H.; MacKenzie, C.; Houslay, E.S.; Currie, R.; Pettitt, T.R.; et al. TAPAS-1, a novel microdomain within the unique N-terminal region of the PDE4A1 cAMP-specific phosphodiesterase that allows rapid, Ca2+-triggered membrane association with selectivity for interaction with phosphatidic acid. J. Biol. Chem. 2002, 277, 28298–28309. [Google Scholar] [CrossRef]
- Kassas, N.; Tanguy, E.; Thahouly, T.; Fouillen, L.; Heintz, D.; Chasserot-Golaz, S.; Bader, M.F.; Grant, N.J.; Vitale, N. Comparative Characterization of Phosphatidic Acid Sensors and Their Localization during Frustrated Phagocytosis. J. Biol. Chem. 2017, 292, 4266–4279. [Google Scholar] [CrossRef]
- Nemoz, G.; Sette, C.; Conti, M. Selective activation of rolipram-sensitive, cAMP-specific phosphodiesterase isoforms by phosphatidic acid. Mol. Pharmacol. 1997, 51, 242–249. [Google Scholar] [CrossRef]
- Lu, Q.; Murakami, C.; Hoshino, F.; Murakami, Y.; Sakane, F. Diacylglycerol kinase δ destabilizes serotonin transporter protein through the ubiquitin-proteasome system. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2020, 1865, 158608. [Google Scholar] [CrossRef]
- Karathanassis, D.; Stahelin, R.V.; Bravo, J.; Perisic, O.; Pacold, C.M.; Cho, W.; Williams, R.L. Binding of the PX domain of p47(phox) to phosphatidylinositol 3,4-bisphosphate and phosphatidic acid is masked by an intramolecular interaction. EMBO J. 2002, 21, 5057–5068. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, S.; Sasai, H.; Kume, A.; Takahashi, D.; Satoh, M.; Kado, S.; Sakane, F. Dioleoyl-phosphatidic acid selectively binds to α-synuclein and strongly induces its aggregation. FEBS Lett. 2017, 591, 784–791. [Google Scholar] [CrossRef] [PubMed]
- Yamada, H.; Mizuno, S.; Honda, S.; Takahashi, D.; Sakane, F. Characterization of alpha-synuclein N-terminal domain as a novel cellular phosphatidic acid sensor. FEBS J. 2020, 287, 2212–2234. [Google Scholar] [CrossRef]
- Baldanzi, G.; Ragnoli, B.; Malerba, M. Potential role of diacylglycerol kinases in immune-mediated diseases. Clin. Sci. (Lond.) 2020, 134, 1637–1658. [Google Scholar] [CrossRef] [PubMed]
- Goto, K.; Nakano, T.; Hozumi, Y. Diacylglycerol kinase and animal models: The pathophysiological roles in the brain and heart. Adv. Enzym. Regul. 2006, 46, 192–202. [Google Scholar] [CrossRef] [PubMed]
- Merida, I.; Andrada, E.; Gharbi, S.I.; Avila-Flores, A. Redundant and specialized roles for diacylglycerol kinases α and ζ in the control of T cell functions. Sci. Signal. 2015, 8, re6. [Google Scholar] [CrossRef]
- Sakane, F.; Imai, S.; Kai, M.; Yasuda, S.; Kanoh, H. Diacylglycerol kinases as emerging potential drug targets for a variety of diseases. Curr. Drug Targets 2008, 9, 626–640. [Google Scholar] [CrossRef]
- Shirai, Y.; Saito, N. Diacylglycerol kinase as a possible therapeutic target for neuronal diseases. J. Biomed. Sci. 2014, 21, 28. [Google Scholar] [CrossRef]
- Jones, D.R.; Sanjuan, M.A.; Stone, J.C.; Merida, I. Expression of a catalytically inactive form of diacylglycerol kinase α induces sustained signaling through RasGRP. FASEB J. 2002, 16, 595–597. [Google Scholar] [CrossRef]
- Sanjuan, M.A.; Jones, D.R.; Izquierdo, M.; Merida, I. Role of diacylglycerol kinase α in the attenuation of receptor signaling. J. Cell Biol. 2001, 153, 207–220. [Google Scholar] [CrossRef]
- Ueyama, T.; Lennartz, M.R.; Noda, Y.; Kobayashi, T.; Shirai, Y.; Rikitake, K.; Yamasaki, T.; Hayashi, S.; Sakai, N.; Seguchi, H.; et al. Superoxide production at phagosomal cup/phagosome through βI protein kinase C during FcγR-mediated phagocytosis in microglia. J. Immunol. 2004, 173, 4582–4589. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, Y.; Shirai, Y.; Matsubara, T.; Sanse, K.; Kuriyama, M.; Ohshiro, N.; Yoshino, K.; Yonezawa, K.; Ono, Y.; Saito, N. Phosphorylation and upregulation of diacylglycerol kinase γ via its interaction with protein kinase Cγ. J. Biol. Chem. 2006, 281, 31627–31637. [Google Scholar] [CrossRef] [PubMed]
- Lucas, P.; Ukhanov, K.; Leinders-Zufall, T.; Zufall, F. A diacylglycerol-gated cation channel in vomeronasal neuron dendrites is impaired in TRPC2 mutant mice: Mechanism of pheromone transduction. Neuron 2003, 40, 551–561. [Google Scholar] [CrossRef]
- Chibalin, A.V.; Leng, Y.; Vieira, E.; Krook, A.; Bjornholm, M.; Long, Y.C.; Kotova, O.; Zhong, Z.; Sakane, F.; Steiler, T.; et al. Downregulation of diacylglycerol kinase delta contributes to hyperglycemia-induced insulin resistance. Cell 2008, 132, 375–386. [Google Scholar] [CrossRef]
- Crotty, T.; Cai, J.; Sakane, F.; Taketomi, A.; Prescott, S.M.; Topham, M.K. Diacylglycerol kinase δ regulates protein kinase C and epidermal growth factor receptor signaling. Proc. Natl. Acad. Sci. USA 2006, 103, 15485–15490. [Google Scholar] [CrossRef]
- Miele, C.; Paturzo, F.; Teperino, R.; Sakane, F.; Fiory, F.; Oriente, F.; Ungaro, P.; Valentino, R.; Beguinot, F.; Formisano, P. Glucose regulates diacylglycerol intracellular levels and protein kinase C activity by modulating diacylglycerol-kinase subcellular localization. J. Biol. Chem. 2007, 282, 31835–31843. [Google Scholar] [CrossRef]
- Luo, B.; Prescott, S.M.; Topham, M.K. Association of diacylglycerol kinase ζ with protein kinase C α: Spatial regulation of diacylglycerol signaling. J. Cell Biol. 2003, 160, 929–937. [Google Scholar] [CrossRef]
- Luo, B.; Prescott, S.M.; Topham, M.K. Protein kinase C α phosphorylates and negatively regulates diacylglycerol kinase ζ. J. Biol. Chem. 2003, 278, 39542–39547. [Google Scholar] [CrossRef]
- Topham, M.K.; Prescott, S.M. Diacylglycerol kinase ζ regulates Ras activation by a novel mechanism. J. Cell Biol. 2001, 152, 1135–1144. [Google Scholar] [CrossRef]
- Regier, D.S.; Higbee, J.; Lund, K.M.; Sakane, F.; Prescott, S.M.; Topham, M.K. Diacylglycerol kinase ι regulates Ras guanyl-releasing protein 3 and inhibits Rap1 signaling. Proc. Natl. Acad. Sci. USA 2005, 102, 7595–7600. [Google Scholar] [CrossRef]
- Avila-Flores, A.; Santos, T.; Rincon, E.; Merida, I. Modulation of the mammalian target of rapamycin pathway by diacylglycerol kinase-produced phosphatidic acid. J. Biol. Chem. 2005, 280, 10091–10099. [Google Scholar] [CrossRef]
- Luo, B.; Prescott, S.M.; Topham, M.K. Diacylglycerol kinase ζ regulates phosphatidylinositol 4-phosphate 5-kinase Iα by a novel mechanism. Cell Signal. 2004, 16, 891–897. [Google Scholar] [CrossRef] [PubMed]
- Kai, M.; Yasuda, S.; Imai, S.; Toyota, M.; Kanoh, H.; Sakane, F. Diacylglycerol kinase α enhances protein kinase Cζ-dependent phosphorylation at Ser311 of p65/RelA subunit of nuclear factor-κB. FEBS Lett. 2009, 583, 3265–3268. [Google Scholar] [CrossRef] [PubMed]
- Rainero, E.; Cianflone, C.; Porporato, P.E.; Chianale, F.; Malacarne, V.; Bettio, V.; Ruffo, E.; Ferrara, M.; Benecchia, F.; Capello, D.; et al. The diacylglycerol kinase α/atypical PKC/β1 integrin pathway in SDF-1α mammary carcinoma invasiveness. PLoS ONE 2014, 9, e97144. [Google Scholar] [CrossRef]
- Yasuda, S.; Kai, M.; Imai, S.; Kanoh, H.; Sakane, F. Diacylglycerol kinase γ interacts with and activates β2-chimaerin, a Rac-specific GAP, in response to epidermal growth factor. FEBS Lett. 2007, 581, 551–557. [Google Scholar] [CrossRef]
- Lu, Q.; Murakami, C.; Murakami, Y.; Hoshino, F.; Asami, M.; Usuki, T.; Sakai, H.; Sakane, F. 1-Stearoyl-2-docosahexaenoyl-phosphatidic acid interacts with and activates Praja-1, the E3 ubiquitin ligase acting on the serotonin transporter in the brain. FEBS Lett. 2020, 594, 1787–1796. [Google Scholar] [CrossRef] [PubMed]
- Hoshino, F.; Murakami, C.; Sakai, H.; Satoh, M.; Sakane, F. Creatine kinase muscle type specifically interacts with saturated fatty acid- and/or monounsaturated fatty acid-containing phosphatidic acids. Biochem. Biophys. Res. Commun. 2019, 513, 1035–1040. [Google Scholar] [CrossRef] [PubMed]
- Hoshino, F.; Sakane, F. Polyunsaturated fatty acid-containing phosphatidic acids selectively interact with L-lactate dehydrogenase A and induce its secondary structural change and inactivation. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2020, 1865, 158768. [Google Scholar] [CrossRef]
- Tang, W.; Bunting, M.; Zimmerman, G.A.; McIntyre, T.M.; Prescott, S.M. Molecular cloning of a novel human diacylglycerol kinase highly selective for arachidonate-containing substrates. J. Biol. Chem. 1996, 271, 10237–10241. [Google Scholar] [CrossRef] [PubMed]
- Walsh, J.P.; Suen, R.; Lemaitre, R.N.; Glomset, J.A. Arachidonoyl-diacylglycerol kinase from bovine testis. J. Biol. Chem. 1994, 269, 21155–21164. [Google Scholar] [PubMed]
- De Rodriguez Turco, E.B.; Tang, W.; Topham, M.K.; Sakane, F.; Marcheselli, V.L.; Chen, C.; Taketomi, A.; Prescott, S.M.; Bazan, N.G. Diacylglycerol kinase ε regulates seizure susceptibility and long-term potentiation through arachidonoyl-inositol lipid signaling. Proc. Natl. Acad. Sci. USA 2001, 98, 4740–4745. [Google Scholar] [CrossRef] [PubMed]
- Bunting, M.; Tang, W.; Zimmerman, G.A.; McIntyre, T.M.; Prescott, S.M. Molecular cloning and characterization of a novel human diacylglycerol kinase ζ. J. Biol. Chem. 1996, 271, 10230–10236. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.; Traer, E.; McIntyre, T.M.; Zimmerman, G.A.; Prescott, S.M. The cloning and characterization of a novel human diacylglycerol kinase, DGKι. J. Biol. Chem. 1998, 273, 32746–32752. [Google Scholar] [CrossRef]
- Houssa, B.; Schaap, D.; van der Val, J.; Goto, K.; Kondo, H.; Yamakawa, A.; Shibata, M.; Takenawa, T.; van Blitterswijk, W.J. Cloning of a Novel Human Diacylglycerol Kinase (DGKθ) Containing Three Cysteine-rich Domains, a Proline-rich Region, and a Pleckstrin Homology Domain with an Overlapping Ras-associating Domain. J. Biol. Chem. 1997, 272, 10422–10428. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Takahashi, D.; Sakane, F. Expression, Purification, and Characterization of Human Diacylglycerol Kinase ζ. ACS Omega 2019, 4, 5540–5546. [Google Scholar] [CrossRef]
- Sakai, H.; Kado, S.; Taketomi, A.; Sakane, F. Diacylglycerol kinase δ phosphorylates phosphatidylcholine-specific phospholipase C-dependent, palmitic acid-containing diacylglycerol species in response to high glucose levels. J. Biol. Chem. 2014, 289, 26607–26617. [Google Scholar] [CrossRef]
- Mizuno, S.; Sakai, H.; Saito, M.; Kado, S.; Sakane, F. Diacylglycerol kinase-dependent formation of phosphatidic acid molecular species during interleukin-2 activation in CTLL-2 T-lymphocytes. FEBS Open Bio 2012, 2, 267–272. [Google Scholar] [CrossRef]
- Sakane, F.; Yamada, K.; Kanoh, H.; Yokoyama, C.; Tanabe, T. Porcine diacylglycerol kinase sequence has zinc finger and E-F hand motifs. Nature 1990, 344, 345–348. [Google Scholar] [CrossRef]
- Schaap, D.; de Widt, J.; van der Wal, J.; Vandekerckhove, J.; van Damme, J.; Gussow, D.; Ploegh, H.L.; van Blitterswijk, W.J.; van der Bend, R.L. Purification, cDNA-cloning and expression of human diacylglycerol kinase. FEBS Lett. 1990, 275, 151–158. [Google Scholar] [CrossRef]
- Sakane, F.; Imai, S.; Yamada, K.; Kanoh, H. The regulatory role of EF-hand motifs of pig 80K diacylglycerol kinase as assessed using truncation and deletion mutants. Biochem. Biophys. Res. Commun. 1991, 181, 1015–1021. [Google Scholar] [CrossRef]
- Sakane, F.; Yamada, K.; Imai, S.; Kanoh, H. Porcine 80-kDa diacylglycerol kinase is a calcium-binding and calcium/phospholipid-dependent enzyme and undergoes calcium-dependent translocation. J. Biol. Chem. 1991, 266, 7096–7100. [Google Scholar] [PubMed]
- Takahashi, D.; Suzuki, K.; Sakamoto, T.; Iwamoto, T.; Murata, T.; Sakane, F. Crystal structure and calcium-induced conformational changes of diacylglycerol kinase α EF-hand domains. Protein Sci. 2019, 28, 694–706. [Google Scholar] [CrossRef]
- Takahashi, M.; Yamamoto, T.; Sakai, H.; Sakane, F. Calcium negatively regulates an intramolecular interaction between the N-terminal recoverin homology and EF-hand motif domains and the C-terminal C1 and catalytic domains of diacylglycerol kinase α. Biochem. Biophys. Res. Commun. 2012, 423, 571–576. [Google Scholar] [CrossRef]
- Yamamoto, T.; Sakai, H.; Sakane, F. EF-hand motifs of diacylglycerol kinase α interact intra-molecularly with its C1 domains. FEBS Open Bio 2014, 4, 387–392. [Google Scholar] [CrossRef]
- Merida, I.; Torres-Ayuso, P.; Avila-Flores, A.; Arranz-Nicolas, J.; Andrada, E.; Tello-Lafoz, M.; Liebana, R.; Arcos, R. Diacylglycerol kinases in cancer. Adv. Biol. Regul. 2017, 63, 22–31. [Google Scholar] [CrossRef]
- Yanagisawa, K.; Yasuda, S.; Kai, M.; Imai, S.; Yamada, K.; Yamashita, T.; Jimbow, K.; Kanoh, H.; Sakane, F. Diacylglycerol kinase α suppresses tumor necrosis factor-α-induced apoptosis of human melanoma cells through NF-κB activation. Biochim. Biophys. Acta 2007, 1771, 462–474. [Google Scholar] [CrossRef]
- Takeishi, K.; Taketomi, A.; Shirabe, K.; Toshima, T.; Motomura, T.; Ikegami, T.; Yoshizumi, T.; Sakane, F.; Maehara, Y. Diacylglycerol kinase alpha enhances hepatocellular carcinoma progression by activation of Ras-Raf-MEK-ERK pathway. J. Hepatol. 2012, 57, 77–83. [Google Scholar] [CrossRef]
- Bacchiocchi, R.; Baldanzi, G.; Carbonari, D.; Capomagi, C.; Colombo, E.; van Blitterswijk, W.J.; Graziani, A.; Fazioli, F. Activation of alpha-diacylglycerol kinase is critical for the mitogenic properties of anaplastic lymphoma kinase. Blood 2005, 106, 2175–2182. [Google Scholar] [CrossRef]
- Baldanzi, G.; Mitola, S.; Cutrupi, S.; Filigheddu, N.; van Blitterswijk, W.J.; Sinigaglia, F.; Bussolino, F.; Graziani, A. Activation of diacylglycerol kinase α is required for VEGF-induced angiogenic signaling in vitro. Oncogene 2004, 23, 4828–4838. [Google Scholar] [CrossRef] [PubMed]
- Rainero, E.; Caswell, P.T.; Muller, P.A.; Grindlay, J.; McCaffrey, M.W.; Zhang, Q.; Wakelam, M.J.; Vousden, K.H.; Graziani, A.; Norman, J.C. Diacylglycerol kinase α controls RCP-dependent integrin trafficking to promote invasive migration. J. Cell Biol. 2012, 196, 277–295. [Google Scholar] [CrossRef]
- Liu, K.; Kunii, N.; Sakuma, M.; Yamaki, A.; Mizuno, S.; Sato, M.; Sakai, H.; Kado, S.; Kumagai, K.; Kojima, H.; et al. A novel diacylglycerol kinase α-selective inhibitor, CU-3, induces cancer cell apoptosis and enhances immune response. J. Lipid Res. 2016, 57, 368–379. [Google Scholar] [CrossRef]
- Yamaki, A.; Akiyama, R.; Murakami, C.; Takao, S.; Murakami, Y.; Mizuno, S.; Takahashi, D.; Kado, S.; Taketomi, A.; Shirai, Y.; et al. Diacylglycerol kinase α-selective inhibitors induce apoptosis and reduce viability of melanoma and several other cancer cell lines. J. Cell. Biochem. 2019, 120, 10043–10056. [Google Scholar] [CrossRef]
- Olenchock, B.A.; Guo, R.; Carpenter, J.H.; Jordan, M.; Topham, M.K.; Koretzky, G.A.; Zhong, X.P. Disruption of diacylglycerol metabolism impairs the induction of T cell anergy. Nat. Immunol. 2006, 7, 1174–1181. [Google Scholar] [CrossRef] [PubMed]
- Zha, Y.; Marks, R.; Ho, A.W.; Peterson, A.C.; Janardhan, S.; Brown, I.; Praveen, K.; Stang, S.; Stone, J.C.; Gajewski, T.F. T cell anergy is reversed by active Ras and is regulated by diacylglycerol kinase-α. Nat. Immunol. 2006, 7, 1166–1173. [Google Scholar] [CrossRef]
- Foell, J.; Hewes, B.; Mittler, R.S. T cell costimulatory and inhibitory receptors as therapeutic targets for inducing anti-tumor immunity. Curr. Cancer Drug Targets 2007, 7, 55–70. [Google Scholar] [CrossRef] [PubMed]
- Prinz, P.U.; Mendler, A.N.; Masouris, I.; Durner, L.; Oberneder, R.; Noessner, E. High DGK-α and disabled MAPK pathways cause dysfunction of human tumor-infiltrating CD8+ T cells that is reversible by pharmacologic intervention. J. Immunol. 2012, 188, 5990–6000. [Google Scholar] [CrossRef]
- Noessner, E. DGK-α: A Checkpoint in Cancer-Mediated Immuno-Inhibition and Target for Immunotherapy. Front. Cell Dev. Biol. 2017, 5, 16. [Google Scholar] [CrossRef]
- Riese, M.J.; Moon, E.K.; Johnson, B.D.; Albelda, S.M. Diacylglycerol Kinases (DGKs): Novel Targets for Improving T Cell Activity in Cancer. Front. Cell Dev. Biol. 2016, 4, 108. [Google Scholar] [CrossRef]
- Murakami, Y.; Murakami, C.; Hoshino, F.; Lu, Q.; Akiyama, R.; Yamaki, A.; Takahashi, D.; Sakane, F. Palmitic acid- and/or palmitoleic acid-containing phosphatidic acids are generated by diacylglycerol kinase α in starved Jurkat T cells. Biochem. Biophys. Res. Commun. 2020, 525, 1054–1060. [Google Scholar] [CrossRef]
- Sakane, F.; Mizuno, S.; Komenoi, S. Diacylglycerol Kinases as Emerging Potential Drug Targets for a Variety of Diseases: An Update. Front. Cell Dev. Biol. 2016, 4, 82. [Google Scholar] [CrossRef] [PubMed]
- Olmez, I.; Love, S.; Xiao, A.; Manigat, L.; Randolph, P.; McKenna, B.D.; Neal, B.P.; Boroda, S.; Li, M.; Brenneman, B.; et al. Targeting the mesenchymal subtype in glioblastoma and other cancers via inhibition of diacylglycerol kinase alpha. Neuro-oncology 2018, 20, 192–202. [Google Scholar] [CrossRef] [PubMed]
- Velnati, S.; Massarotti, A.; Antona, A.; Talmon, M.; Fresu, L.G.; Galetto, A.S.; Capello, D.; Bertoni, A.; Mercalli, V.; Graziani, A.; et al. Structure activity relationship studies on Amb639752: Toward the identification of a common pharmacophoric structure for DGKα inhibitors. J. Enzym. Inhib. Med. Chem. 2020, 35, 96–108. [Google Scholar] [CrossRef] [PubMed]
- Goto, K.; Kondo, H. A 104-kDa diacylglycerol kinase containing ankyrin-like repeats localizes in the cell nucleus. Proc. Natl. Acad. Sci. USA 1996, 93, 11196–11201. [Google Scholar] [CrossRef]
- Topham, M.K.; Bunting, M.; Zimmerman, G.A.; McIntyre, T.M.; Blackshear, P.J.; Prescott, S.M. Protein kinase C regulates the nuclear localization of diacylglycerol kinase-ζ. Nature 1998, 394, 697–700. [Google Scholar] [CrossRef]
- Kim, K.; Yang, J.; Zhong, X.P.; Kim, M.H.; Kim, Y.S.; Lee, H.W.; Han, S.; Choi, J.; Han, K.; Seo, J.; et al. Synaptic removal of diacylglycerol by DGKζ and PSD-95 regulates dendritic spine maintenance. EMBO J. 2009, 28, 1170–1179. [Google Scholar] [CrossRef]
- Yakubchyk, Y.; Abramovici, H.; Maillet, J.C.; Daher, E.; Obagi, C.; Parks, R.J.; Topham, M.K.; Gee, S.H. Regulation of neurite outgrowth in N1E-115 cells through PDZ-mediated recruitment of diacylglycerol kinase ζ. Mol. Cell. Biol. 2005, 25, 7289–7302. [Google Scholar] [CrossRef] [PubMed]
- Traynor, A.E.; Schubert, D.; Allen, W.R. Alterations of lipid metabolism in response to nerve growth factor. J. Neurochem. 1982, 39, 1677–1683. [Google Scholar] [CrossRef]
- Traynor, A.E. The relationship between neurite extension and phospholipid metabolism in PC12 cells. Brain Res. 1984, 316, 205–210. [Google Scholar]
- Mizuno, S.; Kado, S.; Goto, K.; Takahashi, D.; Sakane, F. Diacylglycerol kinase ζ generates dipalmitoyl-phosphatidic acid species during neuroblastoma cell differentiation. Biochem. Biophys. Rep. 2016, 8, 352–359. [Google Scholar]
- Singh, B.K.; Kambayashi, T. The Immunomodulatory Functions of Diacylglycerol Kinase ζ. Front. Cell Dev. Biol. 2016, 4, 96. [Google Scholar] [CrossRef]
- Jung, I.Y.; Kim, Y.Y.; Yu, H.S.; Lee, M.; Kim, S.; Lee, J. CRISPR/Cas9-Mediated Knockout of DGK Improves Antitumor Activities of Human T Cells. Cancer Res. 2018, 78, 4692–4703. [Google Scholar] [CrossRef] [PubMed]
- Sakane, F.; Imai, S.; Kai, M.; Wada, I.; Kanoh, H. Molecular cloning of a novel diacylglycerol kinase isozyme with a pleckstrin homology domain and a C-terminal tail similar to those of the EPH family of protein tyrosine kinase. J. Biol. Chem. 1996, 271, 8394–8401. [Google Scholar] [CrossRef] [PubMed]
- Sakane, F.; Imai, S.; Yamada, K.; Murakami, T.; Tsushima, S.; Kanoh, H. Alternative splicing of the human diacylglycerol kinase δ gene generates two isoforms differing in their expression patterns and in regulatory functions. J. Biol. Chem. 2002, 277, 43519–43526. [Google Scholar] [CrossRef]
- Imai, S.; Kai, M.; Yamada, K.; Kanoh, H.; Sakane, F. The plasma membrane translocation of diacylglycerol kinase δ1 is negatively regulated by conventional protein kinase C-dependent phosphorylation at Ser-22 and Ser-26 within the pleckstrin homology domain. Biochem. J. 2004, 382, 957–966. [Google Scholar] [CrossRef]
- Harada, B.T.; Knight, M.J.; Imai, S.; Qiao, F.; Ramachander, R.; Sawaya, M.R.; Gingery, M.; Sakane, F.; Bowie, J.U. Regulation of enzyme localization by polymerization: Polymer formation by the SAM domain of diacylglycerol kinase δ1. Structure 2008, 16, 380–387. [Google Scholar] [CrossRef] [PubMed]
- Imai, S.; Sakane, F.; Kanoh, H. Phorbol ester-regulated oligomerization of diacylglycerol kinase δ linked to its phosphorylation and translocation. J. Biol. Chem. 2002, 277, 35323–35332. [Google Scholar] [CrossRef]
- Knight, M.J.; Joubert, M.K.; Plotkowski, M.L.; Kropat, J.; Gingery, M.; Sakane, F.; Merchant, S.S.; Bowie, J.U. Zinc Binding Drives Sheet Formation by the SAM Domain of Diacylglycerol Kinase δ. Biochemistry 2010, 49, 9667–9676. [Google Scholar] [CrossRef]
- Wada, Y.; Sakiyama, S.; Sakai, H.; Sakane, F. Myristic Acid Enhances Diacylglycerol Kinase δ-Dependent Glucose Uptake in Myotubes. Lipids 2016, 51, 897–903. [Google Scholar] [CrossRef]
- Sakiyama, S.; Usuki, T.; Sakai, H.; Sakane, F. Regulation of diacylglycerol kinase δ2 expression in C2C12 skeletal muscle cells by free fatty acids. Lipids 2014, 49, 633–640. [Google Scholar] [CrossRef]
- Takato, T.; Iwata, K.; Murakami, C.; Wada, Y.; Sakane, F. Chronic administration of myristic acid improves hyperglycaemia in the Nagoya-Shibata-Yasuda mouse model of congenital type 2 diabetes. Diabetologia 2017, 60, 2076–2083. [Google Scholar] [CrossRef]
- Usuki, T.; Sakai, H.; Shionoya, T.; Sato, N.; Sakane, F. Expression and localization of type II diacylglycerol kinase isozymes δ and η in the developing mouse brain. J. Histochem. Cytochem. 2015, 63, 57–68. [Google Scholar] [CrossRef] [PubMed]
- Usuki, T.; Takato, T.; Lu, Q.; Sakai, H.; Bando, K.; Kiyonari, H.; Sakane, F. Behavioral and pharmacological phenotypes of brain-specific diacylglycerol kinase δ-knockout mice. Brain Res. 2016, 1648, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.; Komenoi, S.; Usuki, T.; Takahashi, D.; Sakane, F. Abnormalities of the serotonergic system in diacylglycerol kinase δ-deficient mouse brain. Biochem. Biophys. Res. Commun. 2018, 497, 1031–1037. [Google Scholar] [CrossRef]
- Mouri, A.; Sasaki, A.; Watanabe, K.; Sogawa, C.; Kitayama, S.; Mamiya, T.; Miyamoto, Y.; Yamada, K.; Noda, Y.; Nabeshima, T. MAGE-D1 regulates expression of depression-like behavior through serotonin transporter ubiquitylation. J. Neurosci. 2012, 32, 4562–4580. [Google Scholar] [CrossRef]
- Nakashima, S. Protein kinase C α (PKCα): Regulation and biological function. J. Biochem. 2002, 132, 669–675. [Google Scholar] [CrossRef] [PubMed]
- Ochoa, W.F.; Corbalan-Garcia, S.; Eritja, R.; Rodriguez-Alfaro, J.A.; Gomez-Fernandez, J.C.; Fita, I.; Verdaguer, N. Additional binding sites for anionic phospholipids and calcium ions in the crystal structures of complexes of the C2 domain of protein kinase Cα. J. Mol. Biol. 2002, 320, 277–291. [Google Scholar] [CrossRef]
- Yokozeki, T.; Homma, K.; Kuroda, S.; Kikkawa, U.; Ohno, S.; Takahashi, M.; Imahori, K.; Kanaho, Y. Phosphatidic acid-dependent phosphorylation of a 29-kDa protein by protein kinase Calpha in bovine brain cytosol. J. Neurochem. 1998, 71, 410–417. [Google Scholar] [CrossRef]
- Aris, J.P.; Basta, P.V.; Holmes, W.D.; Ballas, L.M.; Moomaw, C.; Rankl, N.B.; Blobel, G.; Loomis, C.R.; Burns, D.J. Molecular and biochemical characterization of a recombinant human PKC-δ family member. Biochim. Biophys. Acta 1993, 1174, 171–181. [Google Scholar] [CrossRef]
- Nakanishi, H.; Exton, J.H. Purification and characterization of the ζ isoform of protein kinase C from bovine kidney. J. Biol. Chem. 1992, 267, 16347–16354. [Google Scholar]
- Khan, W.A.; Blobe, G.C.; Richards, A.L.; Hannun, Y.A. Identification, partial purification, and characterization of a novel phospholipid-dependent and fatty acid-activated protein kinase from human platelets. J. Biol. Chem. 1994, 269, 9729–9735. [Google Scholar]
- Zhang, C.; Wendel, A.A.; Keogh, M.R.; Harris, T.E.; Chen, J.; Coleman, R.A. Glycerolipid signals alter mTOR complex 2 (mTORC2) to diminish insulin signaling. Proc. Natl. Acad. Sci. USA 2012, 109, 1667–1672. [Google Scholar] [CrossRef]
- Bruntz, R.C.; Taylor, H.E.; Lindsley, C.W.; Brown, H.A. Phospholipase D2 mediates survival signaling through direct regulation of Akt in glioblastoma cells. J. Biol. Chem. 2014, 289, 600–616. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, K.; Coppola, D.; Challa, S.; Fang, B.; Chen, Y.A.; Zhu, W.; Lopez, A.S.; Koomen, J.; Engelman, R.W.; Rivera, C.; et al. Ack1 mediated AKT/PKB tyrosine 176 phosphorylation regulates its activation. PLoS ONE 2010, 5, e9646. [Google Scholar] [CrossRef] [PubMed]
- Bokoch, G.M.; Reilly, A.M.; Daniels, R.H.; King, C.C.; Olivera, A.; Spiegel, S.; Knaus, U.G. A GTPase-independent mechanism of p21-activated kinase activation. Regulation by sphingosine and other biologically active lipids. J. Biol. Chem. 1998, 273, 8137–8144. [Google Scholar] [CrossRef] [PubMed]
- Lehman, N.; Ledford, B.; Di Fulvio, M.; Frondorf, K.; McPhail, L.C.; Gomez-Cambronero, J. Phospholipase D2-derived phosphatidic acid binds to and activates ribosomal p70 S6 kinase independently of mTOR. FASEB J. 2007, 21, 1075–1087. [Google Scholar] [CrossRef]
- Itoh, T.; Hasegawa, J.; Tsujita, K.; Kanaho, Y.; Takenawa, T. The Tyrosine Kinase Fer Is a Downstream Target of the PLD-PA Pathway that Regulates Cell Migration. Sci. Signal. 2009, 2, ra52. [Google Scholar] [CrossRef] [PubMed]
- Debburman, S.K.; Ptasienski, J.; Boetticher, E.; Lomasney, J.W.; Benovic, J.L.; Hosey, M.M. Lipid-Mediated Regulation of G-Protein-Coupled Receptor Kinase-2 and Kinase-3. J. Biol. Chem. 1995, 270, 5742–5747. [Google Scholar] [CrossRef]
- Han, H.; Qi, R.; Zhou, J.J.; Ta, A.P.; Yang, B.; Nakaoka, H.J.; Seo, G.; Guan, K.L.; Luo, R.; Wang, W. Regulation of the Hippo Pathway by Phosphatidic Acid-Mediated Lipid-Protein Interaction. Mol. Cell 2018, 72, 328–340.e8. [Google Scholar] [CrossRef]
- Kraft, C.A.; Garrido, J.L.; Fluharty, E.; Leiva-Vega, L.; Romero, G. Role of phosphatidic acid in the coupling of the ERK cascade. J. Biol. Chem. 2008, 283, 36636–36645. [Google Scholar] [CrossRef]
- Roach, A.N.; Wang, Z.; Wu, P.; Zhang, F.; Chan, R.B.; Yonekubo, Y.; Di Paolo, G.; Gorfe, A.A.; Du, G. Phosphatidic acid regulation of PIPKI is critical for actin cytoskeletal reorganization. J. Lipid Res. 2012, 53, 2598–2609. [Google Scholar] [CrossRef]
- Takeshita, E.; Kume, A.; Maeda, Y.; Sakai, H.; Sakane, F. Diacylglycerol kinase γ is a novel anionic phospholipid binding protein with a selective binding preference. Biochem. Biophys. Res. Commun. 2014, 444, 617–621. [Google Scholar] [CrossRef] [PubMed]
- Frank, C.; Keilhack, H.; Opitz, F.; Zschornig, O.; Bohmer, F.D. Binding of phosphatidic acid to the protein-tyrosine phosphatase SHP-1 as a basis for activity modulation. Biochemistry 1999, 38, 11993–12002. [Google Scholar] [CrossRef] [PubMed]
- Litosch, I. Phosphatidic acid modulates G protein regulation of phospholipase C-beta1 activity in membranes. Cell Signal. 2002, 14, 259–263. [Google Scholar] [CrossRef]
- Litosch, I. Regulation of phospholipase C-β activity by phosphatidic acid: Isoform dependence, role of protein kinase C, and G protein subunits. Biochemistry 2003, 42, 1618–1623. [Google Scholar] [CrossRef] [PubMed]
- Litosch, I.; Pujari, R.; Lee, S.J. Phosphatidic acid regulates signal output by G protein coupled receptors through direct interaction with phospholipase C-β1. Cell Signal. 2009, 21, 1379–1384. [Google Scholar] [CrossRef]
- Pawelczyk, T.; Matecki, A. Phospholipase C-δ3 binds with high specificity to phosphatidylinositol 4,5-bisphosphate and phosphatidic acid in bilayer membranes. Eur. J. Biochem. 1999, 262, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Murthy, S.N.; Chung, P.H.; Lin, L.; Lomasney, J.W. Activation of phospholipase Cepsilon by free fatty acids and cross talk with phospholipase D and phospholipase A2. Biochemistry 2006, 45, 10987–10997. [Google Scholar] [CrossRef]
- Zhao, C.; Du, G.; Skowronek, K.; Frohman, M.A.; Bar-Sagi, D. Phospholipase D2-generated phosphatidic acid couples EGFR stimulation to Ras activation by Sos. Nat. Cell Biol. 2007, 9, 706–712. [Google Scholar] [CrossRef]
- Consonni, S.V.; Brouwer, P.M.; van Slobbe, E.S.; Bos, J.L. The PDZ domain of the guanine nucleotide exchange factor PDZGEF directs binding to phosphatidic acid during brush border formation. PLoS ONE 2014, 9, e98253. [Google Scholar] [CrossRef]
- Momoi, Y.; Nishikimi, A.; Du, G.; Kataoka, T.; Katagiri, K. Phosphatidic acid regulates subcellular distribution of RA-GEFs critical for chemokine-dependent migration. Biochem. Biophys. Res. Commun. 2020, 524, 325–331. [Google Scholar] [CrossRef]
- Consonni, S.V.; Gloerich, M.; Spanjaard, E.; Bos, J.L. cAMP regulates DEP domain-mediated binding of the guanine nucleotide exchange factor Epac1 to phosphatidic acid at the plasma membrane. Proc. Natl. Acad. Sci. USA 2012, 109, 3814–3819. [Google Scholar] [CrossRef] [PubMed]
- Sanematsu, F.; Nishikimi, A.; Watanabe, M.; Hongu, T.; Tanaka, Y.; Kanaho, Y.; Cote, J.F.; Fukui, Y. Phosphatidic acid-dependent recruitment and function of the Rac activator DOCK1 during dorsal ruffle formation. J. Biol. Chem. 2013, 288, 8092–8100. [Google Scholar] [CrossRef] [PubMed]
- Nishikimi, A.; Fukuhara, H.; Su, W.; Hongu, T.; Takasuga, S.; Mihara, H.; Cao, Q.; Sanematsu, F.; Kanai, M.; Hasegawa, H.; et al. Sequential regulation of DOCK2 dynamics by two phospholipids during neutrophil chemotaxis. Science 2009, 324, 384–387. [Google Scholar] [CrossRef]
- Bollag, G.; McCormick, F. Differential regulation of rasGAP and neurofibromatosis gene product activities. Nature 1991, 351, 576–579. [Google Scholar] [CrossRef]
- Chuang, T.H.; Bohl, B.P.; Bokoch, G.M. Biologically active lipids are regulators of RacGDI complexation. J. Biol. Chem. 1993, 268, 26206–26211. [Google Scholar] [PubMed]
- Caloca, M.J.; Wang, H.; Delemos, A.; Wang, S.; Kazanietz, M.G. Phorbol esters and related analogs regulate the subcellular localization of β2-chimaerin, a non-protein kinase C phorbol ester receptor. J. Biol. Chem. 2001, 276, 18303–18312. [Google Scholar] [CrossRef] [PubMed]
- Nie, Z.; Stanley, K.T.; Stauffer, S.; Jacques, K.M.; Hirsch, D.S.; Takei, J.; Randazzo, P.A. AGAP1, an endosome-associated, phosphoinositide-dependent ADP-ribosylation factor GTPase-activating protein that affects actin cytoskeleton. J. Biol. Chem. 2002, 277, 48965–48975. [Google Scholar] [CrossRef]
- Brown, M.T.; Andrade, J.; Radhakrishna, H.; Donaldson, J.G.; Cooper, J.A.; Randazzo, P.A. ASAP1, a phospholipid-dependent arf GTPase-activating protein that associates with and is phosphorylated by Src. Mol. Cell Biol. 1998, 18, 7038–7051. [Google Scholar] [CrossRef]
- Jackson, T.R.; Brown, F.D.; Nie, Z.; Miura, K.; Foroni, L.; Sun, J.; Hsu, V.W.; Donaldson, J.G.; Randazzo, P.A. ACAPs are arf6 GTPase-activating proteins that function in the cell periphery. J. Cell Biol. 2000, 151, 627–638. [Google Scholar] [CrossRef]
- Randazzo, P.A. Resolution of two ADP-ribosylation factor 1 GTPase-activating proteins from rat liver. Biochem. J. 1997, 324, 413–419. [Google Scholar] [CrossRef]
- Kurooka, T.; Yamamoto, Y.; Takai, Y.; Sakisaka, T. Dual regulation of RA-RhoGAP activity by phosphatidic acid and Rap1 during neurite outgrowth. J. Biol. Chem. 2011, 286, 6832–6843. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, Y.S.; Tu, Y.; Barker, S.A.; Yang, F. Regulators of G-protein signaling (RGS) 4, insertion into model membranes and inhibition of activity by phosphatidic acid. J. Biol. Chem. 2003, 278, 11115–11122. [Google Scholar] [CrossRef]
- Tu, Y.; Wilkie, T.M. Allosteric regulation of GAP activity by phospholipids in regulators of G-protein signaling. Methods Enzymol. 2004, 389, 89–105. [Google Scholar]
- Schroder, W.A.; Buck, M.; Cloonan, N.; Hancock, J.F.; Suhrbier, A.; Sculley, T.; Bushell, G. Human Sin1 contains Ras-binding and pleckstrin homology domains and suppresses Ras signalling. Cell Signal. 2007, 19, 1279–1289. [Google Scholar] [CrossRef]
- Adachi, Y.; Iijima, M.; Sesaki, H. An unstructured loop that is critical for interactions of the stalk domain of Drp1 with saturated phosphatidic acid. Small GTPases 2018, 9, 472–479. [Google Scholar] [CrossRef]
- El Bawab, S.; Macovschi, O.; Sette, C.; Conti, M.; Lagarde, M.; Nemoz, G.; Prigent, A.F. Selective stimulation of a cAMP-specific phosphodiesterase (PDE4A5) isoform by phosphatidic acid molecular species endogenously formed in rat thymocytes. Eur. J. Biochem. 1997, 247, 1151–1157. [Google Scholar] [CrossRef]
- Grange, M.; Sette, C.; Cuomo, M.; Conti, M.; Lagarde, M.; Prigent, A.F.; Nemoz, G. The cAMP-specific phosphodiesterase PDE4D3 is regulated by phosphatidic acid binding. Consequences for cAMP signaling pathway and characterization of a phosphatidic acid binding site. J. Biol. Chem. 2000, 275, 33379–33387. [Google Scholar] [CrossRef]
- Savany, A.; Abriat, C.; Nemoz, G.; Lagarde, M.; Prigent, A.F. Activation of a cyclic nucleotide phosphodiesterase 4 (PDE4) from rat thymocytes by phosphatidic acid. Cell Signal. 1996, 8, 511–516. [Google Scholar] [CrossRef]
- Yan, R.; Qian, H.; Lukmantara, I.; Gao, M.; Du, X.; Yan, N.; Yang, H. Human SEIPIN Binds Anionic Phospholipids. Dev. Cell 2018, 47, 248–256.e4. [Google Scholar] [CrossRef] [PubMed]
- Barneda, D.; Planas-Iglesias, J.; Gaspar, M.L.; Mohammadyani, D.; Prasannan, S.; Dormann, D.; Han, G.S.; Jesch, S.A.; Carman, G.M.; Kagan, V.; et al. The brown adipocyte protein CIDEA promotes lipid droplet fusion via a phosphatidic acid-binding amphipathic helix. Elife 2015, 4, e07485. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.G.; Harris, T.J. Interactions between the PDZ domains of Bazooka (Par-3) and phosphatidic acid: In vitro characterization and role in epithelial development. Mol. Biol. Cell 2012, 23, 3743–3753. [Google Scholar] [CrossRef] [PubMed]
- Capelluto, D.G.; Zhao, X.; Lucas, A.; Lemkul, J.A.; Xiao, S.; Fu, X.; Sun, F.; Bevan, D.R.; Finkielstein, C.V. Biophysical and molecular-dynamics studies of phosphatidic acid binding by the Dvl-2 DEP domain. Biophys. J. 2014, 106, 1101–1111. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, H.; Zhang, Z.; Gao, J.; Sugiyama, G.; Takeuchi, T.; Hirata, M. Second basic pockets contribute to the localization of PX domains by binding to phosphatidic acid. Adv. Biol. Regul. 2012, 52, 183–194. [Google Scholar] [CrossRef] [PubMed]
- Baudier, J.; Deloulme, J.C.; Van Dorsselaer, A.; Black, D.; Matthes, H.W. Purification and characterization of a brain-specific protein kinase C substrate, neurogranin (p17). Identification of a consensus amino acid sequence between neurogranin and neuromodulin (GAP43) that corresponds to the protein kinase C phosphorylation site and the calmodulin-binding domain. J. Biol. Chem. 1991, 266, 229–237. [Google Scholar]
- Epand, R.M.; Epand, R.F.; Berno, B.; Pelosi, L.; Brandolin, G. Association of phosphatidic acid with the bovine mitochondrial ADP/ATP carrier. Biochemistry 2009, 48, 12358–12364. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kedan, A.; Marom, M.; Gavert, N.; Keinan, O.; Selitrennik, M.; Laufman, O.; Lev, S. The phosphatidylinositol-transfer protein Nir2 binds phosphatidic acid and positively regulates phosphoinositide signalling. EMBO Rep. 2013, 14, 891–899. [Google Scholar] [CrossRef]
- Wang, Z.; Cai, M.; Tay, L.W.R.; Zhang, F.; Wu, P.; Huynh, A.; Cao, X.; Di Paolo, G.; Peng, J.; Milewicz, D.M.; et al. Phosphatidic acid generated by PLD2 promotes the plasma membrane recruitment of IQGAP1 and neointima formation. FASEB J. 2019, 33, 6713–6725. [Google Scholar] [CrossRef]
- Zheng, X.; Zhang, J.; Liao, K. The basic amino acids in the coiled-coil domain of CIN85 regulate its interaction with c-Cbl and phosphatidic acid during epidermal growth factor receptor (EGFR) endocytosis. BMC Biochem. 2014, 15, 13. [Google Scholar] [CrossRef][Green Version]
- Lam, A.D.; Tryoen-Toth, P.; Tsai, B.; Vitale, N.; Stuenkel, E.L. SNARE-catalyzed fusion events are regulated by Syntaxin1A-lipid interactions. Mol. Biol. Cell 2008, 19, 485–497. [Google Scholar] [CrossRef]
- Carmon, O.; Laguerre, F.; Riachy, L.; Delestre-Delacour, C.; Wang, Q.; Tanguy, E.; Jeandel, L.; Cartier, D.; Thahouly, T.; Haeberle, A.M.; et al. Chromogranin A preferential interaction with Golgi phosphatidic acid induces membrane deformation and contributes to secretory granule biogenesis. FASEB J. 2020, 34, 6769–6790. [Google Scholar] [CrossRef]
- Dogliotti, G.; Kullmann, L.; Dhumale, P.; Thiele, C.; Panichkina, O.; Mendl, G.; Houben, R.; Haferkamp, S.; Puschel, A.W.; Krahn, M.P. Membrane-binding and activation of LKB1 by phosphatidic acid is essential for development and tumour suppression. Nat. Commun. 2017, 8, 15747. [Google Scholar] [CrossRef]
- Chung, H.W.; Petersen, E.N.; Cabanos, C.; Murphy, K.R.; Pavel, M.A.; Hansen, A.S.; Ja, W.W.; Hansen, S.B. A Molecular Target for an Alcohol Chain-Length Cutoff. J. Mol. Biol. 2019, 431, 196–209. [Google Scholar] [CrossRef] [PubMed]
- Kwiatek, J.M.; Carman, G.M. Yeast phosphatidic acid phosphatase Pah1 hops and scoots along the membrane phospholipid bilayer. J. Lipid Res. 2020, 61, 1232–1243. [Google Scholar] [CrossRef] [PubMed]
- Loewen, C.J.; Gaspar, M.L.; Jesch, S.A.; Delon, C.; Ktistakis, N.T.; Henry, S.A.; Levine, T.P. Phospholipid metabolism regulated by a transcription factor sensing phosphatidic acid. Science 2004, 304, 1644–1647. [Google Scholar] [CrossRef]
- Horchani, H.; de Saint-Jean, M.; Barelli, H.; Antonny, B. Interaction of the Spo20 membrane-sensor motif with phosphatidic acid and other anionic lipids, and influence of the membrane environment. PLoS ONE 2014, 9, e113484. [Google Scholar] [CrossRef]
- Nakanishi, H.; de los Santos, P.; Neiman, A.M. Positive and negative regulation of a SNARE protein by control of intracellular localization. Mol. Biol. Cell 2004, 15, 1802–1815. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Wilson, K.A.; Rice-Stitt, T.; Neiman, A.M.; McNew, J.A. In vitro fusion catalyzed by the sporulation-specific t-SNARE light-chain Spo20p is stimulated by phosphatidic acid. Traffic 2007, 8, 1630–1643. [Google Scholar] [CrossRef]
- Starr, M.L.; Hurst, L.R.; Fratti, R.A. Phosphatidic Acid Sequesters Sec18p from cis-SNARE Complexes to Inhibit Priming. Traffic 2016, 17, 1091–1109. [Google Scholar] [CrossRef]
- Starr, M.L.; Sparks, R.P.; Arango, A.S.; Hurst, L.R.; Zhao, Z.; Lihan, M.; Jenkins, J.L.; Tajkhorshid, E.; Fratti, R.A. Phosphatidic acid induces conformational changes in Sec18 protomers that prevent SNARE priming. J. Biol. Chem. 2019, 294, 3100–3116. [Google Scholar] [CrossRef]
- Watanabe, Y.; Tamura, Y.; Kawano, S.; Endo, T. Structural and mechanistic insights into phospholipid transfer by Ups1-Mdm35 in mitochondria. Nat. Commun. 2015, 6, 7922. [Google Scholar] [CrossRef]
- Yu, F.; He, F.; Yao, H.; Wang, C.; Wang, J.; Li, J.; Qi, X.; Xue, H.; Ding, J.; Zhang, P. Structural basis of intramitochondrial phosphatidic acid transport mediated by Ups1-Mdm35 complex. EMBO Rep. 2015, 16, 813–823. [Google Scholar] [CrossRef] [PubMed]
- Thaller, D.; Tong, D.; Marklew, C.; Borah, S.; Ciani, B.; Lusk, C.P. Direct PA-binding by Chm7 is required for nuclear envelope surveillance at herniations. bioRxiv 2020. [Google Scholar] [CrossRef]
- Zhang, W.; Qin, C.; Zhao, J.; Wang, X. Phospholipase Dα1-derived phosphatidic acid interacts with ABI1 phosphatase 2C and regulates abscisic acid signaling. Proc. Natl. Acad. Sci. USA 2004, 101, 9508–9513. [Google Scholar] [CrossRef] [PubMed]
- Qu, Y.N.; Song, P.; Hu, Y.W.; Jin, X.; Jia, Q.R.; Zhang, X.D.; Chen, L.; Zhang, Q. Regulation of stomatal movement by cortical microtubule organization in response to darkness and ABA signaling in Arabidopsis. Plant Growth Regul. 2018, 84, 467–479. [Google Scholar] [CrossRef]
- Lu, B.; Benning, C. A 25-amino acid sequence of the Arabidopsis TGD2 protein is sufficient for specific binding of phosphatidic acid. J. Biol. Chem. 2009, 284, 17420–17427. [Google Scholar] [CrossRef]
- Roston, R.; Gao, J.; Xu, C.; Benning, C. Arabidopsis chloroplast lipid transport protein TGD2 disrupts membranes and is part of a large complex. Plant J. 2011, 66, 759–769. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Anderson, N.S.; Benning, C. The phosphatidic acid binding site of the Arabidopsis trigalactosyldiacylglycerol 4 (TGD4) protein required for lipid import into chloroplasts. J. Biol. Chem. 2013, 288, 4763–4771. [Google Scholar] [CrossRef]
- Anthony, R.G.; Henriques, R.; Helfer, A.; Meszaros, T.; Rios, G.; Testerink, C.; Munnik, T.; Deak, M.; Koncz, C.; Bogre, L. A protein kinase target of a PDK1 signalling pathway is involved in root hair growth in Arabidopsis. EMBO J. 2004, 23, 572–581. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Zhuang, B.; Wu, Q.; Zhang, H.; Nie, J.; Jing, W.; Yang, L.; Zhang, W. Phosphatidic acid promotes the activation and plasma membrane localization of MKK7 and MKK9 in response to salt stress. Plant Sci. 2019, 287, 110190. [Google Scholar] [CrossRef]
- Julkowska, M.M.; McLoughlin, F.; Galvan-Ampudia, C.S.; Rankenberg, J.M.; Kawa, D.; Klimecka, M.; Haring, M.A.; Munnik, T.; Kooijman, E.E.; Testerink, C. Identification and functional characterization of the Arabidopsis Snf1-related protein kinase SnRK2.4 phosphatidic acid-binding domain. Plant Cell Environ. 2015, 38, 614–624. [Google Scholar] [CrossRef]
- Wang, P.; Shen, L.; Guo, J.; Jing, W.; Qu, Y.; Li, W.; Bi, R.; Xuan, W.; Zhang, Q.; Zhang, W. Phosphatidic Acid Directly Regulates PINOID-Dependent Phosphorylation and Activation of the PIN-FORMED2 Auxin Efflux Transporter in Response to Salt Stress. Plant Cell 2019, 31, 250–271. [Google Scholar] [CrossRef] [PubMed]
- Pandit, S.; Dalal, V.; Mishra, G. Identification of novel phosphatidic acid binding domain on sphingosine kinase 1 of Arabidopsis thaliana. Plant Physiol. Biochem. 2018, 128, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Roy Choudhury, S.; Pandey, S. Phosphatidic acid binding inhibits RGS1 activity to affect specific signaling pathways in Arabidopsis. Plant J. 2017, 90, 466–477. [Google Scholar] [CrossRef] [PubMed]
- Testerink, C.; Dekker, H.L.; Lim, Z.Y.; Johns, M.K.; Holmes, A.B.; Koster, C.G.; Ktistakis, N.T.; Munnik, T. Isolation and identification of phosphatidic acid targets from plants. Plant J. 2004, 39, 527–536. [Google Scholar] [CrossRef]
- Kim, S.C.; Nusinow, D.A.; Sorkin, M.L.; Pruneda-Paz, J.; Wang, X. Interaction and Regulation Between Lipid Mediator Phosphatidic Acid and Circadian Clock Regulators. Plant Cell 2019, 31, 399–416. [Google Scholar] [CrossRef]
- Yao, H.; Wang, G.; Guo, L.; Wang, X. Phosphatidic acid interacts with a MYB transcription factor and regulates its nuclear localization and function in Arabidopsis. Plant Cell 2013, 25, 5030–5042. [Google Scholar] [CrossRef]
- Cai, G.; Kim, S.C.; Li, J.; Zhou, Y.; Wang, X. Transcriptional Regulation of Lipid Catabolism during Seedling Establishment. Mol. Plant 2020, 13, 984–1000. [Google Scholar] [CrossRef]
- Shen, L.; Tian, Q.; Yang, L.; Zhang, H.; Shi, Y.; Shen, Y.; Zhou, Z.; Wu, Q.; Zhang, Q.; Zhang, W. Phosphatidic acid directly binds with rice potassium channel OsAKT2 to inhibit its activity. Plant J. 2020, 102, 649–665. [Google Scholar] [CrossRef]
- Zhang, Q.; Lin, F.; Mao, T.; Nie, J.; Yan, M.; Yuan, M.; Zhang, W. Phosphatidic acid regulates microtubule organization by interacting with MAP65-1 in response to salt stress in Arabidopsis. Plant Cell 2012, 24, 4555–4576. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhu, H.; Zhang, Q.; Li, M.; Yan, M.; Wang, R.; Wang, L.; Welti, R.; Zhang, W.; Wang, X. Phospholipase Dα1 and phosphatidic acid regulate NADPH oxidase activity and production of reactive oxygen species in ABA-mediated stomatal closure in Arabidopsis. Plant Cell 2009, 21, 2357–2377. [Google Scholar] [CrossRef]
- Sagaram, U.S.; El-Mounadi, K.; Buchko, G.W.; Berg, H.R.; Kaur, J.; Pandurangi, R.S.; Smith, T.J.; Shah, D.M. Structural and functional studies of a phosphatidic acid-binding antifungal plant defensin MtDef4: Identification of an RGFRRR motif governing fungal cell entry. PLoS ONE 2013, 8, e82485. [Google Scholar] [CrossRef]
- Kvansakul, M.; Lay, F.T.; Adda, C.G.; Veneer, P.K.; Baxter, A.A.; Phan, T.K.; Poon, I.K.; Hulett, M.D. Binding of phosphatidic acid by NsD7 mediates the formation of helical defensin-lipid oligomeric assemblies and membrane permeabilization. Proc. Natl. Acad. Sci. USA 2016, 113, 11202–11207. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.L.; Yao, H.Y.; Jia, L.H.; Tan, J.F.; Xu, Z.H.; Zheng, W.M.; Xue, H.W. Phospholipase D-derived phosphatidic acid promotes root hair development under phosphorus deficiency by suppressing vacuolar degradation of PIN-FORMED2. New Phytol. 2020, 226, 142–155. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, M.; Hossain, S.; Al Mamun, A.; Matsuzaki, K.; Arai, H. Docosahexaenoic acid: One molecule diverse functions. Crit. Rev. Biotechnol. 2017, 37, 579–597. [Google Scholar] [CrossRef]
- Meneses, A.; Liy-Salmeron, G. Serotonin and emotion, learning and memory. Rev. Neurosci. 2012, 23, 543–553. [Google Scholar] [CrossRef]
- Joel, D. Current animal models of obsessive compulsive disorder: A critical review. Prog. Neuro Psychopharmacol. Biol. Psychiatry 2006, 30, 374–388. [Google Scholar] [CrossRef]
- Yadin, E.; Friedman, E.; Bridger, W.H. Spontaneous alternation behavior: An animal model for obsessive-compulsive disorder? Pharmacol. Biochem. Behav. 1991, 40, 311–315. [Google Scholar] [CrossRef]
- Klauck, T.M.; Xu, X.; Mousseau, B.; Jaken, S. Cloning and characterization of a glucocorticoid-induced diacylglycerol kinase. J. Biol. Chem. 1996, 271, 19781–19788. [Google Scholar] [CrossRef]
- Murakami, T.; Sakane, F.; Imai, S.; Houkin, K.; Kanoh, H. Identification and characterization of two splice variants of human diacylglycerol kinase η. J. Biol. Chem. 2003, 278, 34364–34372. [Google Scholar] [CrossRef]
- Kume, A.; Kawase, K.; Komenoi, S.; Usuki, T.; Takeshita, E.; Sakai, H.; Sakane, F. The Pleckstrin Homology Domain of Diacylglycerol Kinase eta Strongly and Selectively Binds to Phosphatidylinositol 4,5-Bisphosphate. J. Biol. Chem. 2016, 291, 8150–8161. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, S.; Kai, M.; Imai, S.; Takeishi, K.; Taketomi, A.; Toyota, M.; Kanoh, H.; Sakane, F. Diacylglycerol kinase η augments C-Raf activity and B-Raf/C-Raf heterodimerization. J. Biol. Chem. 2009, 284, 29559–29570. [Google Scholar] [CrossRef]
- Baum, A.E.; Akula, N.; Cabanero, M.; Cardona, I.; Corona, W.; Klemens, B.; Schulze, T.G.; Cichon, S.; Rietschel, M.; Nothen, M.M.; et al. A genome-wide association study implicates diacylglycerol kinase eta (DGKH) and several other genes in the etiology of bipolar disorder. Mol. Psychiatry 2008, 13, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Ollila, H.M.; Soronen, P.; Silander, K.; Palo, O.M.; Kieseppa, T.; Kaunisto, M.A.; Lonnqvist, J.; Peltonen, L.; Partonen, T.; Paunio, T. Findings from bipolar disorder genome-wide association studies replicate in a Finnish bipolar family-cohort. Mol. Psychiatry 2009, 14, 351–353. [Google Scholar] [CrossRef] [PubMed]
- Squassina, A.; Manchia, M.; Congiu, D.; Severino, G.; Chillotti, C.; Ardau, R.; Piccardi, M.; Zompo, M.D. The diacylglycerol kinase eta gene and bipolar disorder: A replication study in a Sardinian sample. Mol. Psychiatry 2009, 14, 350–351. [Google Scholar] [CrossRef] [PubMed]
- Weber, H.; Kittel-Schneider, S.; Gessner, A.; Domschke, K.; Neuner, M.; Jacob, C.P.; Buttenschon, H.N.; Boreatti-Hummer, A.; Volkert, J.; Herterich, S.; et al. Cross-disorder analysis of bipolar risk genes: Further evidence of DGKH as a risk gene for bipolar disorder, but also unipolar depression and adult ADHD. Neuropsychopharmacol. Off. Publ. Am. Coll. Neuropsychopharmacol. 2011, 36, 2076–2085. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.; Wang, T.; Li, T.; Li, Y.; Chen, P.; Zhao, Q.; Liu, J.; Li, J.; Feng, G.; He, L.; et al. Common SNPs and haplotypes in DGKH are associated with bipolar disorder and schizophrenia in the Chinese Han population. Mol. Psychiatry 2011, 16, 473–475. [Google Scholar] [CrossRef]
- Isozaki, T.; Komenoi, S.; Lu, Q.; Usuki, T.; Tomokata, S.; Matsutomo, D.; Sakai, H.; Bando, K.; Kiyonari, H.; Sakane, F. Deficiency of diacylglycerol kinase η induces lithium-sensitive mania-like behavior. J. Neurochem. 2016, 138, 448–456. [Google Scholar] [CrossRef]
- Komenoi, S.; Suzuki, Y.; Asami, M.; Murakami, C.; Hoshino, F.; Chiba, S.; Takahashi, D.; Kado, S.; Sakane, F. Microarray analysis of gene expression in the diacylglycerol kinase η knockout mouse brain. Biochem. Biophys. Rep. 2019, 19, 100660. [Google Scholar] [CrossRef]
- Imai, S.; Kai, M.; Yasuda, S.; Kanoh, H.; Sakane, F. Identification and characterization of a novel human type II diacylglycerol kinase, DGKκ. J. Biol. Chem. 2005, 280, 39870–39881. [Google Scholar] [CrossRef]
- Tabet, R.; Moutin, E.; Becker, J.A.; Heintz, D.; Fouillen, L.; Flatter, E.; Krezel, W.; Alunni, V.; Koebel, P.; Dembele, D.; et al. Fragile X Mental Retardation Protein (FMRP) controls diacylglycerol kinase activity in neurons. Proc. Natl. Acad. Sci. USA 2016, 113, E3619–E3628. [Google Scholar] [CrossRef]
- Pankratz, N.; Wilk, J.B.; Latourelle, J.C.; DeStefano, A.L.; Halter, C.; Pugh, E.W.; Doheny, K.F.; Gusella, J.F.; Nichols, W.C.; Foroud, T.; et al. Genomewide association study for susceptibility genes contributing to familial Parkinson disease. Hum. Genet. 2009, 124, 593–605. [Google Scholar] [CrossRef] [PubMed]
- Simon-Sanchez, J.; van Hilten, J.J.; van de Warrenburg, B.; Post, B.; Berendse, H.W.; Arepalli, S.; Hernandez, D.G.; de Bie, R.M.; Velseboer, D.; Scheffer, H.; et al. Genome-wide association study confirms extant PD risk loci among the Dutch. Eur. J. Hum. Genet. EJHG 2011, 19, 655–661. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Hwarng, G.; Cooper, D.E.; Grevengoed, T.J.; Eaton, J.M.; Natarajan, V.; Harris, T.E.; Coleman, R.A. Inhibited insulin signaling in mouse hepatocytes is associated with increased phosphatidic acid but not diacylglycerol. J. Biol. Chem. 2015, 290, 3519–3528. [Google Scholar] [CrossRef] [PubMed]
- Ruiperez, V.; Darios, F.; Davletov, B. Alpha-synuclein, lipids and Parkinson’s disease. Prog. Lipid Res. 2010, 49, 420–428. [Google Scholar] [CrossRef] [PubMed]
- Spillantini, M.G.; Schmidt, M.L.; Lee, V.M.; Trojanowski, J.Q.; Jakes, R.; Goedert, M. α-synuclein in Lewy bodies. Nature 1997, 388, 839–840. [Google Scholar] [CrossRef]
- Kubo, S.; Nemani, V.M.; Chalkley, R.J.; Anthony, M.D.; Hattori, N.; Mizuno, Y.; Edwards, R.H.; Fortin, D.L. A combinatorial code for the interaction of α-synuclein with membranes. J. Biol. Chem. 2005, 280, 31664–31672. [Google Scholar] [CrossRef]
- Bussell, R., Jr.; Eliezer, D. Effects of Parkinson’s disease-linked mutations on the structure of lipid-associated α-synuclein. Biochemistry 2004, 43, 4810–4818. [Google Scholar] [CrossRef]
- Davidson, W.S.; Jonas, A.; Clayton, D.F.; George, J.M. Stabilization of α-synuclein secondary structure upon binding to synthetic membranes. J. Biol. Chem. 1998, 273, 9443–9449. [Google Scholar] [CrossRef]
- Jiang, Z.; Hess, S.K.; Heinrich, F.; Lee, J.C. Molecular details of α-synuclein membrane association revealed by neutrons and photons. J. Phys. Chem. B 2015, 119, 4812–4823. [Google Scholar] [CrossRef]
- Rappley, I.; Myers, D.S.; Milne, S.B.; Ivanova, P.T.; Lavoie, M.J.; Brown, H.A.; Selkoe, D.J. Lipidomic profiling in mouse brain reveals differences between ages and genders, with smaller changes associated with α-synuclein genotype. J. Neurochem. 2009, 111, 15–25. [Google Scholar] [CrossRef]
- Reeve, A.; Simcox, E.; Turnbull, D. Ageing and Parkinson’s disease: Why is advancing age the biggest risk factor? Ageing Res. Rev. 2014, 14, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Taylor, K.S.; Cook, J.A.; Counsell, C.E. Heterogeneity in male to female risk for Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 2007, 78, 905–906. [Google Scholar] [CrossRef]
- Wooten, G.F.; Currie, L.J.; Bovbjerg, V.E.; Lee, J.K.; Patrie, J. Are men at greater risk for Parkinson’s disease than women? J. Neurol. Neurosurg. Psychiatry 2004, 75, 637–639. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, A.; Masuda, Y.; Iwai, K.; Ikeda, K.; Watanabe, K. A RING finger protein Praja1 regulates Dlx5-dependent transcription through its ubiquitin ligase activity for the Dlx/Msx-interacting MAGE/Necdin family protein, Dlxin-1. J. Biol. Chem. 2002, 277, 22541–22546. [Google Scholar] [CrossRef] [PubMed]
- McPherson, P.S.; Garcia, E.P.; Slepnev, V.I.; David, C.; Zhang, X.; Grabs, D.; Sossin, W.S.; Bauerfeind, R.; Nemoto, Y.; De Camilli, P. A presynaptic inositol-5-phosphatase. Nature 1996, 379, 353–357. [Google Scholar] [CrossRef] [PubMed]
- Montesinos, M.L.; Castellano-Munoz, M.; Garcia-Junco-Clemente, P.; Fernandez-Chacon, R. Recycling and EH domain proteins at the synapse. Brain Res. Brain Res. Rev. 2005, 49, 416–428. [Google Scholar] [CrossRef]
- Augoff, K.; Hryniewicz-Jankowska, A.; Tabola, R. Lactate dehydrogenase 5: An old friend and a new hope in the war on cancer. Cancer Lett. 2015, 358, 1–7. [Google Scholar] [CrossRef]
- Terlecki, G.; Czapinska, E.; Rogozik, K.; Lisowski, M.; Gutowicz, J. Investigation of the interaction of pig muscle lactate dehydrogenase with acidic phospholipids at low pH. Biochim. Biophys. Acta 2006, 1758, 133–144. [Google Scholar] [CrossRef]
- Crane, C.A.; Austgen, K.; Haberthur, K.; Hofmann, C.; Moyes, K.W.; Avanesyan, L.; Fong, L.; Campbell, M.J.; Cooper, S.; Oakes, S.A.; et al. Immune evasion mediated by tumor-derived lactate dehydrogenase induction of NKG2D ligands on myeloid cells in glioblastoma patients. Proc. Natl. Acad. Sci. USA 2014, 111, 12823–12828. [Google Scholar] [CrossRef]
- Fantin, V.R.; St-Pierre, J.; Leder, P. Attenuation of LDH-A expression uncovers a link between glycolysis, mitochondrial physiology, and tumor maintenance. Cancer Cell 2006, 9, 425–434. [Google Scholar] [CrossRef]
- Koukourakis, M.; Tsolou, A.; Pouliliou, S.; Lamprou, I.; Papadopoulou, M.; Ilemosoglou, M.; Kostoglou, G.; Ananiadou, D.; Sivridis, E.; Giatromanolaki, A. Blocking LDHA glycolytic pathway sensitizes glioblastoma cells to radiation and temozolomide. Biochem. Biophys. Res. Commun. 2017, 491, 932–938. [Google Scholar] [CrossRef] [PubMed]
- Vander Heiden, M.G.; Cantley, L.C.; Thompson, C.B. Understanding the Warburg effect: The metabolic requirements of cell proliferation. Science 2009, 324, 1029–1033. [Google Scholar] [CrossRef] [PubMed]
- Gallo, M.; Sapio, L.; Spina, A.; Naviglio, D.; Calogero, A.; Naviglio, S. Lactic dehydrogenase and cancer: An overview. Front. Biosci. (Landmark Ed.) 2015, 20, 1234–1249. [Google Scholar]
- Valvona, C.J.; Fillmore, H.L.; Nunn, P.B.; Pilkington, G.J. The Regulation and Function of Lactate Dehydrogenase A: Therapeutic Potential in Brain Tumor. Brain Pathol. 2016, 26, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Chesnelong, C.; Chaumeil, M.M.; Blough, M.D.; Al-Najjar, M.; Stechishin, O.D.; Chan, J.A.; Pieper, R.O.; Ronen, S.M.; Weiss, S.; Luchman, H.A.; et al. Lactate dehydrogenase A silencing in IDH mutant gliomas. Neuro-oncology 2014, 16, 686–695. [Google Scholar] [CrossRef]
- Mack, N.; Mazzio, E.A.; Bauer, D.; Flores-Rozas, H.; Soliman, K.F. Stable shRNA Silencing of Lactate Dehydrogenase A (LDHA) in Human MDA-MB-231 Breast Cancer Cells Fails to Alter Lactic Acid Production, Glycolytic Activity, ATP or Survival. Anticancer Res. 2017, 37, 1205–1212. [Google Scholar]
- Zdralevic, M.; Brand, A.; Di Ianni, L.; Dettmer, K.; Reinders, J.; Singer, K.; Peter, K.; Schnell, A.; Bruss, C.; Decking, S.M.; et al. Double genetic disruption of lactate dehydrogenases A and B is required to ablate the “Warburg effect” restricting tumor growth to oxidative metabolism. J. Biol. Chem. 2018, 293, 15947–15961. [Google Scholar] [CrossRef]
- Wildburger, N.C.; Wood, P.L.; Gumin, J.; Lichti, C.F.; Emmett, M.R.; Lang, F.F.; Nilsson, C.L. ESI-MS/MS and MALDI-IMS Localization Reveal Alterations in Phosphatidic Acid, Diacylglycerol, and DHA in Glioma Stem Cell Xenografts. J. Proteome Res. 2015, 14, 2511–2519. [Google Scholar] [CrossRef]
- Aragno, M.; Mastrocola, R.; Catalano, M.G.; Brignardello, E.; Danni, O.; Boccuzzi, G. Oxidative stress impairs skeletal muscle repair in diabetic rats. Diabetes 2004, 53, 1082–1088. [Google Scholar] [CrossRef]
- Jevric-Causevic, A.; Malenica, M.; Dujic, T. Creatine kinase activity in patients with diabetes mellitus type I and type II. Bosn. J. Basic Med. Sci. 2006, 6, 5–9. [Google Scholar] [CrossRef]
- Odum, E.P.; Young, E.E. Elevated cardiac troponin I, creatine kinase and myoglobin and their relationship with cardiovascular risk factors in patients with type 2 diabetes. Diabetes Metab. Syndr. 2018, 12, 141–145. [Google Scholar] [CrossRef]
- Popovich, B.K.; Boheler, K.R.; Dillmann, W.H. Diabetes decreases creatine kinase enzyme activity and mRNA level in the rat heart. Am. J. Physiol. 1989, 257, E573–E577. [Google Scholar] [CrossRef]
- Tsushima, S.; Kai, M.; Yamada, K.; Imai, S.; Houkin, K.; Kanoh, H.; Sakane, F. Diacylglycerol kinase γ serves as an upstream suppressor of Rac1 and lamellipodium formation. J. Biol. Chem. 2004, 279, 28603–28613. [Google Scholar] [CrossRef] [PubMed]
- Tsumagari, R.; Kakizawa, S.; Kikunaga, S.; Fujihara, Y.; Ueda, S.; Yamanoue, M.; Saito, N.; Ikawa, M.; Shirai, Y. DGKγ Knock-Out Mice Show Impairments in Cerebellar Motor Coordination, LTD, and the Dendritic Development of Purkinje Cells through the Activation of PKCγ. eNeuro 2020, 7. [Google Scholar] [CrossRef]
- Ferraz-Nogueira, J.P.; Diez-Guerra, F.J.; Llopis, J. Visualization of phosphatidic acid fluctuations in the plasma membrane of living cells. PLoS ONE 2014, 9, e102526. [Google Scholar] [CrossRef]
- Potocky, M.; Pleskot, R.; Pejchar, P.; Vitale, N.; Kost, B.; Zarsky, V. Live-cell imaging of phosphatidic acid dynamics in pollen tubes visualized by Spo20p-derived biosensor. New Phytol. 2014, 203, 483–494. [Google Scholar] [CrossRef]
- Schwarz, K.; Natarajan, S.; Kassas, N.; Vitale, N.; Schmitz, F. The synaptic ribbon is a site of phosphatidic acid generation in ribbon synapses. J. Neurosci. 2011, 31, 15996–16011. [Google Scholar] [CrossRef]
- Yamada, H.; Hoshino, F.; Lu, Q.; Sakane, F. Cellular phosphatidic acid sensor, α-synuclein N-terminal domain, detects endogenous phosphatidic acid in macrophagic phagosomes and neuronal growth cones. Biochem. Biophys. Rep. 2020, 22, 100769. [Google Scholar] [CrossRef]
- Vacaru, A.M.; Tafesse, F.G.; Ternes, P.; Kondylis, V.; Hermansson, M.; Brouwers, J.F.; Somerharju, P.; Rabouille, C.; Holthuis, J.C. Sphingomyelin synthase-related protein SMSr controls ceramide homeostasis in the ER. J. Cell Biol. 2009, 185, 1013–1027. [Google Scholar] [CrossRef] [PubMed]
- Cabukusta, B.; Kol, M.; Kneller, L.; Hilderink, A.; Bickert, A.; Mina, J.G.; Korneev, S.; Holthuis, J.C. ER residency of the ceramide phosphoethanolamine synthase SMSr relies on homotypic oligomerization mediated by its SAM domain. Sci. Rep. 2017, 7, 41290. [Google Scholar] [CrossRef]
- Cabukusta, B.; Köhlen, J.A.; Richter, C.P.; You, C.; Holthuis, J.C. Monitoring Changes in the Oligomeric State of a Candidate Endoplasmic Reticulum (ER) Ceramide Sensor by Single-molecule Photobleaching. J. Biol. Chem. 2016, 291, 24735–24746. [Google Scholar] [CrossRef] [PubMed]
- Murakami, C.; Hoshino, F.; Sakai, H.; Hayashi, Y.; Yamashita, A.; Sakane, F. Diacylglycerol kinase δ and sphingomyelin synthase-related protein functionally interact via their sterile α motif domains. J. Biol. Chem. 2020, 295, 2932–2947. [Google Scholar] [CrossRef] [PubMed]
- Ogushi, F.; Ishitsuka, R.; Kobayashi, T.; Sugita, Y. Rapid flip-flop motions of diacylglycerol and ceramide in phospholipid bilayers. Chem. Phys. Lett. 2012, 522, 96–102. [Google Scholar] [CrossRef]
- Kooijman, E.E.; Burger, K.N. Biophysics and function of phosphatidic acid: A molecular perspective. Biochim. Biophys. Acta 2009, 1791, 881–888. [Google Scholar] [CrossRef] [PubMed]
- Kamiya, Y.; Mizuno, S.; Komenoi, S.; Sakai, H.; Sakane, F. Activation of conventional and novel protein kinase C isozymes by different diacylglycerol molecular species. Biochem. Biophys. Rep. 2016, 7, 361–366. [Google Scholar] [CrossRef]
- Peng, X.; Frohman, M.A. Mammalian phospholipase D physiological and pathological roles. Acta Physiol. (Oxf.) 2012, 204, 219–226. [Google Scholar] [CrossRef]
- Bradley, R.M.; Duncan, R.E. The lysophosphatidic acid acyltransferases (acylglycerophosphate acyltransferases) family: One reaction, five enzymes, many roles. Curr. Opin. Lipidol. 2018, 29, 110–115. [Google Scholar] [CrossRef]
- Eto, M.; Shindou, H.; Shimizu, T. A novel lysophosphatidic acid acyltransferase enzyme (LPAAT4) with a possible role for incorporating docosahexaenoic acid into brain glycerophospholipids. Biochem. Biophys. Res. Commun. 2014, 443, 718–724. [Google Scholar] [CrossRef]
- Zhukovsky, M.A.; Filograna, A.; Luini, A.; Corda, D.; Valente, C. The Structure and Function of Acylglycerophosphate Acyltransferase 4/Lysophosphatidic Acid Acyltransferase Delta (AGPAT4/LPAATδ). Front. Cell Dev. Biol. 2019, 7, 147. [Google Scholar] [CrossRef]
- De Kroon, A.I. Metabolism of phosphatidylcholine and its implications for lipid acyl chain composition in Saccharomyces cerevisiae. Biochim. Biophys. Acta 2007, 1771, 343–352. [Google Scholar] [CrossRef] [PubMed]
- Wagner, S.; Paltauf, F. Generation of glycerophospholipid molecular species in the yeast Saccharomyces cerevisiae. Fatty acid pattern of phospholipid classes and selective acyl turnover at sn-1 and sn-2 positions. Yeast 1994, 10, 1429–1437. [Google Scholar] [CrossRef]
- Devaiah, S.P.; Roth, M.R.; Baughman, E.; Li, M.; Tamura, P.; Jeannotte, R.; Welti, R.; Wang, X. Quantitative profiling of polar glycerolipid species from organs of wild-type Arabidopsis and a phospholipase Dα1 knockout mutant. Phytochemistry 2006, 67, 1907–1924. [Google Scholar] [CrossRef] [PubMed]
- Narasimhan, R.; Wang, G.; Li, M.; Roth, M.; Welti, R.; Wang, X. Differential changes in galactolipid and phospholipid species in soybean leaves and roots under nitrogen deficiency and after nodulation. Phytochemistry 2013, 96, 81–91. [Google Scholar] [CrossRef]
| Organism | Protein | PA Species | Affinity (Kd)/ PA Species | PA-Binding Region | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 16:0/16:0 | 16:0/18:1 | 18:1/18:1 | 18:0/18:1 | 18:0/18:0 | 18:0/20:4 | 18:0/22:6 | Other | Mixture | ||||
| Mammal | Raf-1 (C-Raf) | 50 | – | 75 | – | – | – | – | 100 (16:1/18:1) | – | 78 nM/ 18:1/18:1-PA | FRNEVAVLRKTRHVNILLFMGYMTKDNLAIVTQWCEG |
| PKC | – | – | – | – | – | – | – | – | 100 | – | KLKLIPDPKNESKQKTKTIRSTLN | |
| PKC | – | – | – | – | – | – | – | – | 100 | – | – | |
| PKC | – | 100 | – | – | – | – | – | – | – | 20–313 µM/ 16:0/18:1-PA | SLKPTAWSLRHAVGPRPQTF……VFHDAPIGYDDFVA (part of C2 domain) | |
| PKC | – | – | – | – | – | – | – | – | 100 | – | – | |
| PKN | – | – | – | – | – | – | – | – | 100 | 15.9 µM/ PA mixture | – | |
| mTOR | – | – | – | – | – | – | – | – | 100 | – | RNVKGMFEVLEPLHAMMERGPQTLKETSFNQAYGRDLMEAQEWCRKYMKSGNVKDLTQAWDLYYHVFR | |
| mTORC2 | 100 | 0 | 0 | – | – | – | – | – | – | – | – | |
| Akt | – | – | – | – | – | – | – | 100 (16:0/20:4) | – | – | PH domain | |
| PAK1 | – | – | – | – | – | 100 | – | – | 100 | – | – | |
| p70S6K1 | – | 100 | – | – | – | – | – | – | – | – | – | |
| Fer | – | – | – | – | – | – | – | – | 100 | – | SMERKERLSK FESIRHSIAG | |
| GRK | – | – | – | – | – | – | – | – | 100 | – | – | |
| LATS1 | 100 | – | – | – | – | – | – | – | – | – | MOB binding domain, aa 601–751 | |
| KSR1 | 75 | - | 75 | - | - | - | - | 100 (16:1/18:1) | - | 1.2 µM/ 18:1/18:1-PA | FKKEVMNYRQTRHENVVLFMGACMNPPHLA | |
| PIP5K | – | – | – | – | – | – | – | – | 100 | – | – | |
| PIP5KI | – | 100 | – | – | – | – | – | – | – | – | KPERDVLMQDFYVVESIFFPSEGSNLTPAHHFQ | |
| SphK1 | 100 | – | – | – | – | – | – | – | – | – | – | |
| DGK | – | – | 100 | – | 30 | 100 | – | 40 (14:0/14:0) | 100 | 6–13 pmol/ 18:1/18:1-PA (Overlay) | RVH domain and EF-hand motifs, aa 1–259 | |
| CKM | 86 | 57 | 93 | – | 100 | 14 | 14 | – | – | 2.0 µM/ 16:0/16:0-PA | – | |
| SHP-1 | 100 | – | – | – | – | – | – | – | – | 0.04 μM/ 16:0/16:0-PA | SSKHKEDVYENLHTKNKREEKVKKQRSADKEKSKGSLKRK | |
| PP1c | – | – | 100 | – | – | – | – | – | – | 1.37 µM/ 18:1/18:1-PA | GAMMSVDETLMCSFQILKPAEKKKPNATRPVTPPRGMITKQAKK | |
| Synaptojanin-1 | 20 | 30 | 40 | – | 30 | 100 | 100 | – | – | – | – | |
| Lipin1 | – | – | – | – | – | – | – | – | 100 | – | VVKKRRKRRRKSQLDSLKR | |
| PLC 1 | 89 | – | – | – | 88 | – | – | 100 (14:0/14:0) | 100 | 15 mol%/ PA mixture | C-terminus, aa 944-955 | |
| PLC 1 | – | – | – | – | – | – | – | – | 100 | – | – | |
| PLC 3 | – | – | – | – | – | – | – | – | 100 | – | – | |
| PLC | – | – | – | – | – | – | – | – | 100 | 160 µM/ PA mixture | – | |
| Sos | – | – | – | – | – | – | – | – | 100 | 0.2–0.5 µM/ PA mixture | HF domain, aa 97–99 (RKR); PH domain, aa KSNHGQPRLPGA | |
| RA-GEF-1/2 (PDZ-GEF) | – | – | – | – | – | – | – | – | 100 | – | CDC25 homology domain, aa 919–967; PDZ domain, aa K428 and R429 | |
| Epac1 | – | – | – | – | – | – | – | – | 100 | – | RDRKYHLRLYRQCCSGR | |
| DOCK1 | – | – | – | – | – | – | – | – | 100 | – | C-terminal domain, aa 1610–1698 | |
| DOCK2 | – | – | – | – | – | – | – | – | 100 | – | EYGVREMPDFEDRRVGRPRSMRSKKRT | |
| RasGAP | – | – | – | – | – | 75 | – | 100 (18:2/18:2) | – | – | – | |
| NF1 | – | – | – | – | – | 100 | – | – | – | 12 µM/ 18:0/20:4-PA | – | |
| RacGDI | – | – | 100 | – | – | 100 | – | – | – | – | – | |
| 1-Chimaerin | – | – | – | – | – | – | – | – | 100 | – | – | |
| 2-Chimaerin | – | – | – | – | – | – | – | – | 100 | – | – | |
| AGAP1 | – | – | – | – | – | – | – | – | 100 | – | GTP binding protein-like domain, adjacent to PH domain | |
| ASAP1 | – | – | – | – | – | – | – | – | 100 | – | – | |
| ACAP1/2 | – | – | – | – | – | – | – | – | 100 | – | – | |
| ArfGAP1/2 | – | – | – | – | – | – | – | – | 100 | – | – | |
| RA-RhoGAP | – | – | – | – | – | – | – | – | 100 | – | RAELKRGLQRQERHLFLFND | |
| RGS4 | – | – | – | – | – | – | – | – | 100 | 8 mol%/ PA mixture | N-terminal 57 aa | |
| Sin1 | – | – | – | – | – | – | – | – | 100 | – | PH domain | |
| Arf1/6 | – | – | – | – | – | – | – | – | 100 | – | – | |
| Rac1 | – | – | – | – | – | – | – | 100 (14:0/14:0) | – | – | AVLCPPPVKKRKRKCLLL | |
| Drp1 | 85 | – | 100 | – | – | – | – | – | – | – | Stalk domain TAKYIETSEL | |
| PDE4A1 | – | 100 | 77 | 85 | – | – | 38 | 69 (16:0/18:2) | – | 6.8 µM/ 18:0/18:1-PA | PWLVGWWDQFKR (requires calcium) | |
| PDE4A5 | 100 | 23 | – | 18 | 53 | 0 | 27 | – | – | 1.44 µM/ 16:0/16:0-PA | – | |
| PDE4B1 | – | – | – | – | – | – | – | – | 100 | – | – | |
| PDE4D3 | – | – | – | – | – | – | – | – | 100 | – | DPMTSPGSGLILQANFVHSQRRESFLYRS | |
| PDE4 | – | 74 | 100 | 78 | – | 94 | – | – | 89 | – | – | |
| Praja-1 | – | <5 | 25 | – | 25 | 13 | 100 | – | – | – | – | |
| p47phox | – | 100 | – | – | – | – | – | – | – | 1.2 nM/ 16:0/18:1-PA | VYRRFTEIYEFHKTLKEMFPIEAGAINPENRIIPHLPAPKWFD (part of PX domain) | |
| -Synuclein | 19 | 32 | 100 | – | <5 | 32 | – | – | – | 6.6 µM/ 18:1/18:1-PA | N-terminal 60 aa | |
| LDHA | 44 | 22 | 22 | 22 | 100 | 100 | 100 | – | – | 3.7 µM/ 18:0/22:6-PA | – | |
| NF2 | 100 | – | – | – | – | – | – | – | – | – | – | |
| Seipin | <50 | 100 | – | – | <50 | – | – | – | 100 | – | C2-like domain, aa 60–200 | |
| CIDEA | – | – | – | – | – | – | – | 100 (12:0/12:0) | – | – | C-terminal domain, RCTSFKAVLRNLLRFMS | |
| Bazooka (Par-3) | 100 | – | – | – | – | – | – | – | – | – | PDZ2 domain, aa 458–546; PDZ3 domain, aa 635–682 | |
| Dvl-2 | 100 | 100 | – | – | – | – | – | – | – | – | GLLKAGLIRHTVNKITFSEQ | |
| RPK118 | – | – | – | – | – | – | – | – | 100 | – | KRYSDFKKLHKELW | |
| Neurogranin | 100 | – | – | – | – | – | – | – | 100 | – | IQ motif, aa 29-47 | |
| -COP | – | – | – | – | – | – | – | – | 100 | – | – | |
| NSF | – | – | – | – | – | – | – | – | 100 | – | – | |
| Kinesin | – | – | – | – | – | – | – | – | 100 | – | – | |
| Anc | – | – | <5 | – | – | – | – | – | 100 | – | – | |
| Nir | – | – | – | – | – | – | – | – | 100 | – | IVAGYGSPKDVAVYAALGLSPSQTYIVGR | |
| IQGAP1 | – | – | 100 | – | – | – | – | – | – | – | KKEKIQTGKK | |
| CIN85 | – | – | – | – | – | – | – | – | 100 | – | LDEEKKIRLRLQMEVNDIKK | |
| Syntaxin1A | 100 | - | - | - | - | - | - | - | - | EC50 2.0 pmol/ 16:0/16:0-PA (Overlay) | DTKKAVKYQSKARRKKIMI | |
| CgA | – | – | – | 100 | – | – | 100 | – | – | – | – | |
| Insect | LKB1 | – | – | – | – | – | – | – | – | 100 | – | VKKKGSALKRRAKKLTSCISVRKLSHCRTS |
| TREK-1 | – | – | – | – | – | – | – | – | 100 | 15.7 µM/ PA mixture | – | |
| Yeast | Pah1 | ~100 | ~100 | ~100 | 100 | – | – | – | ~100 (16:0/18:2) ~100 (18:0/18:2) | – | Km 0.65 mol%/ 18:1/18:1-PA | – |
| Opi1p | – | 100 | 83 | 83 | – | – | 75 | 88 (16:0/18:2) | 100 | 4.5 µM/ 18:0/18:1-PA | KRQKLSRAIAKGKDNLKEYKLNMSIESKKR | |
| Spo20p | – | 87 | 80 | 100 | – | – | 72 | 67 (16:0/18:2) | – | 2.2 µM/ 18:0/18:1-PA | RLHVKLKSLRNKIHKQLH | |
| Sso1p | – | 100 | – | – | – | – | – | – | – | – | KAVKSARKARRNKIRCWLIV | |
| Sec18p | – | 100 | – | – | – | – | – | – | – | 1.4 µM/ 8:0/8:0-PA | ATP binding site (D1–D2 domain) | |
| Ups1 | – | 100 | – | – | – | – | – | – | – | – | R25, K61, K155, I78, V106 | |
| Chm7 | – | 100 | – | – | – | – | – | – | – | – | RKGFAKAARSAKESTNMYKSRK | |
| Plant | ABI1 | 0 | – | 100 | – | 10 | – | – | – | – | – | ESRKVLISRINSPNLMKESAAADIVVVDISAG |
| PP2CA | 10 | – | 100 | – | 10 | – | – | 100 (18:2/18:2) | 100 | – | – | |
| TGD2 | – | – | – | – | – | – | – | – | 100 | 39.6% (w/w) PA mixture | aa 201–225 | |
| TGD4 | – | – | 100 | – | – | – | – | – | – | – | aa 1–80, 110–145 | |
| AtPDK1 | – | – | – | – | – | – | – | – | 100 | – | PH domain aa 391–491 | |
| MKK7/9 | <10 | 70 | 70 | 50 | 30 | – | – | – | 100 | – | – | |
| SnRK2.4 | – | 100 | – | – | – | – | – | – | – | – | aa 261–302 | |
| PID | 0 | 30 | 30 | <5 | 0 | – | – | 100 (18:2/18:2) | 30 | – | LALKKKMHR | |
| AtSphK1 | – | – | – | – | – | – | – | – | 100 | 0.3 µM/ PA mixture | VSGDGI | |
| RGS1 | – | – | – | – | – | – | – | – | 100 | 0.3 mM/ PA mixture | PLLSQISLKK | |
| PEPC | – | – | – | – | – | – | – | – | 100 | – | – | |
| LHY | 80 | 30 | – | – | – | – | – | – | 100 | 0.18 µM/ 16:0/16:0-PA | – | |
| CCA1 | 100 | 30 | – | – | – | – | – | – | 30 | 0.12 µM/ 16:0/16:0-PA | – | |
| Werewolf | – | 100 | 100 | – | – | – | – | 100 (18:2/18:2) | 100 | 3.99 µM/ 18:1/18:1-PA | RIAKKTGLKRCGKSCRLRWMNYL | |
| AHL4 | 0 | 100 | 100 | – | 0 | – | – | – | – | – | – | |
| AKT2 | <10 | <10 | <10 | <10 | <10 | – | – | 100 (16:0/18:2) | – | – | aa 317–855 | |
| MAP65-1 | <5 | 50 | 50 | 30 | <5 | – | – | 100 (18:2/18:2) | 100 | – | ARILVSKIPAM | |
| RbohD160 | <5 | 100 | 100 | – | <5 | – | – | 100 (16:0/18:2) | 100 | – | SRELRRVSFRRPSPAVRRFDR | |
| 14-3-3 protein | – | – | – | – | – | – | – | – | 100 | EC50 48.5 µM/ PA mixture | LSVAYKNVIGARRASWRIIS | |
| MtDef4 | 100 | 100 | 100 | – | – | – | – | 0 (8:0/8:0) | – | – | RGFRRR | |
| NsD7 | – | – | – | – | – | – | – | – | 100 | – | DGHCSKILRR | |
| SNX | – | – | 100 | – | – | – | – | – | – | – | – | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sakane, F.; Hoshino, F.; Murakami, C. New Era of Diacylglycerol Kinase, Phosphatidic Acid and Phosphatidic Acid-Binding Protein. Int. J. Mol. Sci. 2020, 21, 6794. https://doi.org/10.3390/ijms21186794
Sakane F, Hoshino F, Murakami C. New Era of Diacylglycerol Kinase, Phosphatidic Acid and Phosphatidic Acid-Binding Protein. International Journal of Molecular Sciences. 2020; 21(18):6794. https://doi.org/10.3390/ijms21186794
Chicago/Turabian StyleSakane, Fumio, Fumi Hoshino, and Chiaki Murakami. 2020. "New Era of Diacylglycerol Kinase, Phosphatidic Acid and Phosphatidic Acid-Binding Protein" International Journal of Molecular Sciences 21, no. 18: 6794. https://doi.org/10.3390/ijms21186794
APA StyleSakane, F., Hoshino, F., & Murakami, C. (2020). New Era of Diacylglycerol Kinase, Phosphatidic Acid and Phosphatidic Acid-Binding Protein. International Journal of Molecular Sciences, 21(18), 6794. https://doi.org/10.3390/ijms21186794






