Abstract
The root bark of Morus has long been appreciated as an antiphlogistic, diuretic and expectorant drug in Chinese herbal medicine, albeit with barely known targets and mechanisms of action. In the 1970s, the development of analytic chemistry allowed for the discovery of morusin as one of 7 different isoprene flavonoid derivatives in the root bark of Morus. However, the remarkable antioxidant capacity of morusin with the unexpected potential for health benefits over the other flavonoid derivatives has recently sparked scientific interest in the biochemical identification of target proteins and signaling pathways and further clinical relevance. In this review, we discuss recent advances in the understanding of the functional roles of morusin in multiple biological processes such as inflammation, apoptosis, metabolism and autophagy. We also highlight recent in vivo and in vitro evidence on the clinical potential of morusin treatment for multiple human pathologies including inflammatory diseases, neurological disorders, diabetes, cancer and the underlying mechanisms.
1. Introduction
The therapeutic relevance of the root bark of the mulberry tree (genus Morus) as an antiphlogistic, diuretic and expectorant drug has long been acknowledged in Chinese herbal medicine [1]. The rapid development of analytic chemistry in the 1970s led researchers to explore the constituents of this mysterious plant with remarkable clinical potential [2,3,4,5]. As a result of these efforts, a group at Toho university in Japan achieved the consecutive purification of two 2-arylbenzofuran derivatives and seven isoprene-substituted flavonoid derivatives from the benzene extracts of the root bark of a variety of plants in the genus Morus including Morus alba L. and Morus nigra L. Such discoveries, taken together with the anti-inflammatory properties of flavonoid species, significantly provoked scientific attention to the potential application of Morus-derived flavonoids to a myriad of human pathologies beyond its known benefits, leading to subsequent biochemical studies from multiple research groups in which the chemical and bioactive properties of the phytochemicals were tested.
Morusin is one of the Morus-derived flavonoids, which has been highlighted for its outstanding antioxidant capacity over the other flavonoids due to unique chemical and bioactive properties (Figure 1) [6]. Its versatile potential against human pathologies including cancer, immune dysfunction and metabolic disorders has been intensively tested in in vitro systems, although the underlying mechanisms and clinical evidence in vivo are yet to be fully explored. In this review, we describe the previously reported physiological processes induced by morusin, as well as in vitro and in vivo evidence connecting its therapeutic potential to relevant human pathologies including cancer. Finally, we highlight our recent discoveries of mechanisms regarding the strategies employed by cancer cells to resist morusin treatment and propose the cotreatment of morusin and relevant inhibitors as a potential tactic for boosting the anti-tumor capacity of the morusin.
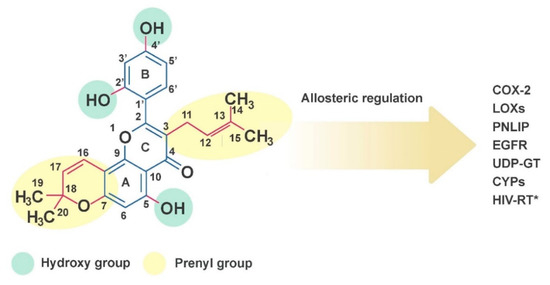
Figure 1.
The chemical structure of morusin. Molecular targets of morusin include cyclooxygenase-2 (COX-2), lipoxygenases (LOXs), pancreatic lipase (PNLIP), epidermal growth factor receptor (EGFR), UDP-glucuronosyltransferase, cytochrome P450 (CYP) and HIV reverse transcriptase (* in silico analysis only).
2. Results
2.1. Chemical Properties of Morusin
Various classes of prenylated flavonoids have been isolated from the root bark of Morus. Among these, morusin has been highlighted for its versatile effects on human physiology and pathology. Morusin is a prenylated flavone with strong antioxidant capacity, which is structurally characterized by (1) a prenyl unit at position 3, (2) hydroxy groups at 5, 2′′ and 4′ and (3) a 2,2-dimethyl pyran group across positions 7 and 8 (Figure 1) [6]. The structure-bioactivity relationship was analyzed by the comparison of many prenylated flavonoids in the number and position of prenyl moieties [7,8,9,10]. In general, attachment of hydroxy group and prenyl group on the flavone backbone affects the bioactivities of a compound depending on the position and number of functional groups. Prenylation results in a more lipophilic compound to provide high affinity with the cell membrane, while leads to decreased bioavailability and plasma absorption [10]. The prenylations at the C-3 and C-7 position in the flavone backbone of morusin greatly contribute to its high cytotoxicity against murine P-388 cells [11,12]. However, the change from a cyclic form of the prenyl group at the C-8 position of morusin to a free prenyl group in kuwanon C slightly reduced the cytotoxic effects, while inhibitory activity against β-secretase and anti-bacterial activity against E.coli as well as S. typhimurium were markedly increased [8,9]. On the other hand, cyclization of the prenyl group at C-7 of morusin reduced inhibitory activity against tyrosinase and α-glucosidase [7].
Morusin may allosterically regulate several enzymes including cyclooxygenase-2, lipoxygenases, pancreatic lipase, epidermal growth factor receptor, UDP-glucuronosyltransferase, acetylcholine esterase (AChE), matrix metalloproteinases (MMP-9 and MMP-2), cytochrome P450 and HIV reverse transcriptase (further discussed in later sections) [13,14,15,16,17]. Such abilities have been decoupled from its antioxidant and anti-inflammatory capacities, since each carbon residue appears to play differential roles in mediating biological functions. For example, a prenyl substitution on C-3 of morusin caused noncompetitive inhibition characteristics toward AChE, while non-prenylated flavonoids showed mixed inhibition kinetics [18]. In addition, hydroxylation at C-5′ of morusin determined the selective inhibition of different oxygenases. Morusin, which lacks the 5′-hydroxyl group of artonin E, was a less potent 5-lipoxygenase inhibitor. Morusin displayed broad inhibitory activities against several lipoxygenases, while artonin E, produced by hydroxylation at C-5′ of morusin, displayed inhibitory activity against 5-lipoxygenase higher by one order of magnitude [19]. Molecular docking analysis between morusin and the cytochrome isoform CYP3A4 indicated that oxygens in the pyran ring, C-5 and C-2′ are involved in hydrogen bonds with CYP3A4 and the B ring structure of morusin is involved in π-π interaction with Phe108 of CYP3A4 [16,20]. Hydrogen bond formation between the 2′-hydroxy group of morusin and Tyr393 of MMP-9 and cation-pi interactions between the flavone backbone of morusin and Tyr423 of MMP-9 were also associated with morusin binding to MMP-9 [21]. Docking analysis of morusin with 5-lipoxygenase (5-LOX) indicated that the 2′- and 4′-hydroxy groups of the B ring structure of morusin form hydrogen bonds with Val127 of both the B and F chain in 5-LOX, respectively. Moreover, the 3-prenyl group of morusin aligned into the hydrophobic groove generated by Val127, Ala128, Leu124, Leu135 and Phe131 of the 5-LOX active site [22], indicating that hydroxylation and prenylation of morusin are crucial in molecular recognition of the target protein. Molecular modeling of proteins and docking analysis was utilized to screen for ligands of GABA transporter 1. In this study, morusin was identified as the strongest potential ligand for GABA transporter 1, in which Tyr140 and Ser396 of GABA transporter 1 may be involved in hydrogen bonding with morusin [23].
We briefly mentioned the chemical properties of morusin here, since this review mainly focuses on the biological aspects of morusin treatment with various clinical relevance. More detailed physiochemical properties of flavonoids including morusin and their purification processes are well described elsewhere [3,6,7].
2.2. The Effects of Morusin Treatment on Cellular Processes
Multiple physiological processes have been proposed as effectors of morusin treatment in various biochemical studies (Figure 2). In this section, we describe the cellular processes specifically implicated in morusin-targeted human pathophysiology discussed later on.
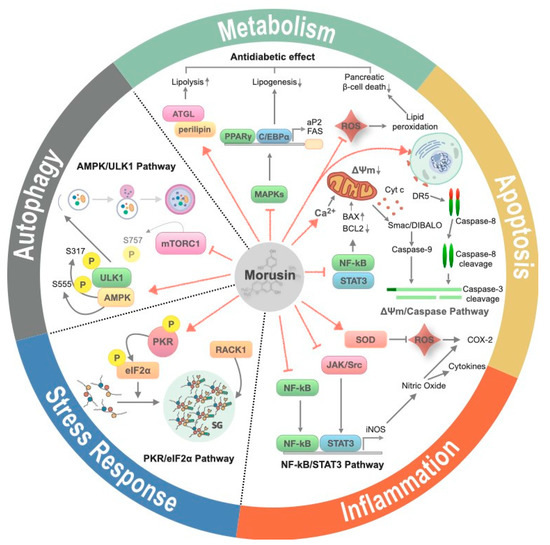
Figure 2.
The effects of morusin on cellular processes. Morusin elicits various cellular processes including suppression of inflammation, induction of apoptosis, autophagy and stress granule formation and is involved in the homeostasis of glucose and lipid metabolism. Red solid arrow and solid bar indicates activation and suppression of target molecule, respectively. Upwards and downwards arrow indicates an increase and decrease of target protein or pathway, respectively.
2.2.1. Inflammation
Many studies have confirmed the anti-inflammatory capacity of morusin in different in vivo and in vitro contexts—(1) Morusin treatment inhibits secretion of cytokines such as CCL5 and CCL17 in TNFα- and IFN-γ-stimulated keratinocytes and also inhibits the release of histamine and LTC4 in A23187-stimulated MC/9 mast cells [24]. (2) Morusin treatment inhibits PMA-induced MUC5AC production in a human pulmonary mucoepidermoid cell line NCI-H292, showing prominent anti-inflammatory effects in vitro [25]. (3) Morusin treatment ameliorates IL-1β-induced chondrocyte inflammation and abrogates osteoarthritis in destabilization of the medial meniscus model in vivo [26]. (4) Morusin treatment inhibits NO production from LPS-induced RAW264.7 cells [27]. (5) Morusin treatment protects against 2,4,6-trinitrobenzensulfonic acid (TNBS)-induced colitis in rats [28]. (6) Morusin treatment alleviates inflammatory signaling thereby controlling the outgrowth of mycoplasma pneumonia [29].
This explosion of observations on the anti-inflammatory effects of morusin have raised questions of the molecular and biochemical targets of morusin and the underlying mechanisms. In this context, the antioxidant capacity of morusin has been linked to the suppression of iNOS induction, thereby reducing nitric oxide formation [27,30,31]. This mechanism may also be associated with the neuroprotective function of morusin against NO-induced cell death in SH-SY5Y cells [31]. Although the mechanism underlying iNOS regulation by morusin has not been directly addressed yet, it may be recapitulated by the NF-κB pathway, a key intracellular pathway that governs pro-inflammatory signaling in multiple ways including reactive oxygen species (ROS) production, cytokine production and immune cell activation [32,33]. Furthermore, morusin suppressed STAT1-mediated cytokine secretion in TNF-α- and IFN-γ-stimulated keratinocytes [24]. Indeed, many studies have already emphasized that the versatile effects of morusin may be associated with downregulation of the NF-κB pathway and its crosstalk with STAT1, STAT3 and Wnt/β-catenin signaling [24,25,26,29,34,35,36,37,38,39,40]. Given that activation of the NF-κB and STAT signaling pathways is one of the downstream events of EGF receptor signaling and since morusin directly binds to the catalytic domain of the EGF receptor for inactivation of EGFR [14], it is plausible that morusin-mediated blocking of EGFR signaling may contribute to the down-regulation of NF-κB-mediated iNOS induction and NO synthesis, as well as STAT1-mediated cytokine secretion, to result in anti-inflammatory effects. In addition, several in vitro studies argued that the anti-inflammatory effects of morusin are partly attributed to its role as an allosteric inhibitor of cyclooxygenase-2 (COX-2) and lipoxygenases (LOXs), which are key rate enzymes implicated in arachidonic acid metabolism [41]. Given that arachidonic acid is a key molecule spiking intracellular pro-inflammatory signaling followed by immune cell activation [42], such regulations may be clinically relevant to diseases associated with aberrant inflammation.
2.2.2. Apoptosis
Although cytotoxicity upon morusin treatment has been observed across multiple cancer cell lines, biochemical characterization of the underlying mechanisms and clinical potential in vivo are still active areas of research. In particular, multiple recent studies have performed a thorough assessment of the biochemical targets of morusin, allowing for the identification of apoptosis as a critical tumor-killing effect of morusin.
Apoptosis is a type of tightly regulated programmed cell death, characterized by cell shrinkage, nuclear fragmentation, chromatin condensation and chromosomal DNA fragmentation [43]. Apoptosis can be subclassified into extrinsic and intrinsic pathways according to the origin of stimuli (intracellular vs extracellular signals). This biological process has been appreciated as an attractive druggable target in numerous pathological contexts especially including cancer [44]. Morusin effects on apoptosis in cancer cells has been mostly linked to its capacity to suppress the NF-κB pathway, since suppressors of the intrinsic apoptotic pathway, such as cIAP and Bcl-xL, are representative downstream targets of p50/p65, the core transcription factor of the NF-κB pathway [43]. The potential involvement of the NF-κB pathway was first suggested by a study in which morusin treatment led to inhibition of the NF-κB pathway and activation of intrinsic apoptosis in human colorectal cancer cells [35]. Such observations were also substantiated by studies across multiple cancer cell lines in which morusin treatment resulted in concomitant NF-κB pathway inhibition and activation of apoptosis [40,45,46].
In addition, several studies have suggested the possibility of STAT3 as a component of the mechanisms underlying the pro-apoptotic effects of morusin [14,36,38,47,48], which may be relevant since (1) ROS formation is both a consequence and a driving force of STAT3 activation [49] and (2) the STAT3 and NF-κB pathways synergistically promote transcriptional activation of suppressors of the intrinsic apoptotic pathway [50]. Consistently, STAT3 target genes, such as anti-apoptotic genes encoding Bcl-xL, Bcl-2, XIAP, survivin and cell cycle regulators (c-Myc and cyclin D), are down-regulated upon morusin treatment, while pro-apoptotic Bax expression was induced [36,38,40,47,51]. In addition, the administration of morusin reduces mitochondrial membrane potential resulting in the release of cytochrome c and Smac/DIABLO and thus, apoptosis is facilitated by the activation of caspase-9 and caspase-3 [35,48,52].
Turning off signaling pathways implicated in cell survival and proliferation has been functionally associated with the activation of apoptosis [53]. In this regard, morusin also exerts its pro-apoptotic effects by inhibiting relevant pathways including the PI3K-AKT and MAPK signaling pathways [35,54,55]. However, it is still unclear if such mechanisms can be dissociated from morusin effects on the NF-κB pathway, which has cross-talk with the PI3K-AKT, MAPK pathways in many physiological and pathological contexts [54,55].
Finally, some evidence has suggested that morusin treatment also results in the activation of extrinsic apoptotic pathways, in which morusin strongly increased expression of the death receptor DR5 at the transcriptional level and conferred sensitization of glioblastoma to TRAIL signaling [35,38]. However, thorough assessment of the underlying mechanisms may be further required to interpret morusin effects on the extrinsic apoptotic pathways, considering the intimate crosstalk between the intrinsic and extrinsic apoptotic pathways [56].
2.2.3. Metabolism
Metabolism plays key roles in energy homeostasis and signal transduction, which are fundamental aspects of organisms. One of the unique aspects of metabolism is the remarkable metabolic flexibility in response to various stimuli [57]; metabolic pathways are dramatically rewired as an adaptive response to specific physiological contexts such as starvation and energy overload. Such adaptations have also been proposed as either the consequence or cause of numerous human pathologies including metabolic disorders, cardiovascular diseases and cancer.
Morusin treatment was shown to exert beneficial effects on systemic and cellular metabolism. Those effects have also been mostly associated with the antioxidant capacity of morusin. Multiple studies have shown that morusin treatment reduces reactive oxygen species in metabolically relevant organs, in which redox homeostasis is tightly associated with general metabolic fitness including increased mitochondrial performance and the amelioration of dysregulated fuel metabolism [58]. For example, treatment of Morus alba root bark extract including morusin reduces lipid peroxidation, thereby leading to hypoglycemic effects in vivo in a streptozotocin-induced mouse model of type I diabetes. Such beneficial effects may be associated with decreasing oxidative stress and preservation of pancreatic β-cell integrity by reducing cell death [59,60].
It has recently been shown that morusin also serves as an allosteric inhibitor of metabolic enzymes including pancreatic lipase, UDP-glucuronosyltransferase (UGT) and cytochrome p450 (CYP450) [16,61]. Such findings may be clinically relevant, since these enzymes have been implicated in the dysregulation of lipid homeostasis and endoplasmic reticulum stress, which play key roles in the pathogenesis of various metabolic disorders including diabetes [62,63]. Of note, CYP450 and other ER-resident lipid oxygenases including CYP1A2, CYP2C9, CYP2D6, CYP2E1, CYP3A4 and CYP2C19 have been proposed as metabolizing enzymes of morusin per se [17,20], although it is unclear whether the mechanisms contribute to the drug resistance or whether the intermediates mediate the beneficial effects of morusin.
Morusin has also been highlighted as a potent de novo lipogenesis inhibitor as well as a lipolysis stimulator [64]. The study was performed in 3T3-L1 and primary adipocytes in which morusin treatment leads to reduced lipid build-up and increased lipid breakdown. Such observations may be associated with changes in multiple processes including down-regulation of the MAPK pathway downstream of insulin receptor signaling, adipogenic transcription factors (PPARγand C/EBPα) and lipogenic factors (aP2 and FAS), while expression of lipolytic factors (HSL, ATGL and perilipin) are enhanced by morusin administration in differentiated 3T3-L1 adipocytes. However, the trans-differentiation of glioblastoma multiforme (GBM) cancer stem cells into adipocyte-like cells was induced by morusin treatment, in which the expression of adipogenic proteins including PPARγ, Adipsin D and aP2 were enhanced in a dose-dependent manner [39]. A similar result was observed with breast cancer cells in which differentiation of breast cancer cells into adipocyte-like cells was induced by morusin treatment and concomitantly expression of adipogenic protein was increased [65]. The detailed clinical potential of morusin treatment for metabolic disorders including diabetes has been discussed in a later section.
2.2.4. Autophagy
Autophagy is a physiological process through which unnecessary and dysfunctional cellular components are removed or recycled [66]. Various extracellular and intracellular signals induce autophagy and thus autophagy plays a key role in the intricate regulation of cellular homeostasis including cell fate determination, fuel metabolism and mitochondrial fitness. In this context, autophagy has been implicated in numerus human pathologies particularly associated with energy stress and mitochondrial dysfunction, such as cancer and neurodegeneration, respectively. This notion, together with previous reports in which drug resistance of cancer cells are tightly associated with the capacity of cells to induce autophagy [67], brought us to the question as to whether cancer cells employ autophagy as a protective mechanism against morusin-induced cell death. Biochemical evidence in our study showed that morusin treatment leads to mTOR1 inhibition and the subsequent activation of AMPK, resulting in ULK1-mediated autophagy activation. Such autophagy activation is associated with the reduced apoptosis induced by morusin (Figure 3), corroborating the idea that activation of autophagy confers drug resistance to cancer cells [68]. This mechanism may be noteworthy in the context of the therapeutic relevance of morusin for neurological disorders, given the anti-cell death effects of morusin on neuronal cells [31] and the intimate connection between autophagy and neurodegenerative diseases [69].
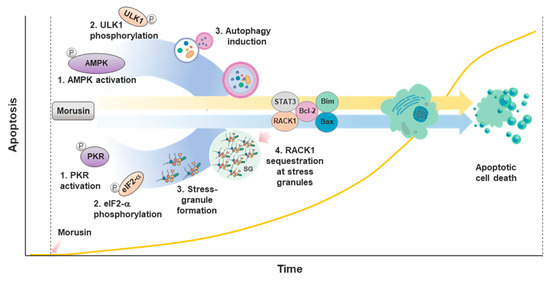
Figure 3.
Morusin-induced autophagy and stress granule (SG) formation inhibits induction of apoptosis at early times of the stress response. Morusin induces apoptosis, autophagy and SG formation. However, induction of autophagy and SG formation occurs ahead of apoptosis induction at the early times of morusin treatment, resulting in a delay of the induction of apoptosis.
2.2.5. Stress Response
Cells have evolved multiple physiological strategies to cope with diverse environmental and intracellular stresses. Among these is stress granule (SG) formation, during which macromolecular aggregates of mRNA and proteins are assembled, leading to the attenuation of translation and activation of various physiological pathways for DNA damage repair and cell survival [70]. Since study of the SGs has mainly focused on biochemical or mechanistic perspectives, the physiological and pathological relevance of the SGs is still enigmatic. Recent in vitro and in vivo clinical evidence has proposed the potential implication of SGs in cancers and neurodegenerative diseases, warranting a more detailed examination of the underlying mechanisms [71,72]. In particular, the SGs are induced by various types of cytotoxic drugs, thereby contributing to drug resistance [73]. Our recent study showed that SG formation is induced by morusin treatment [74]. PKR was identified as a target of morusin in an un-biased systemic phospho-antibody array. PKR activation and subsequent eIF2α phosphorylation are key biochemical events for morusin-induced SG formation, which leads to the retention of RACK1 within the SGs. Since RACK1 is a pro-apoptotic molecule that activates intrinsic apoptotic pathways and together with our data in which RACK1 manipulation decouples SG formation and morusin-induced cell death, we suggested that morusin-mediated cell death may at least in part be attributed to RACK1 activation (Figure 3).
Although our study only highlighted the potential connection between SG formation and the intrinsic apoptotic pathway, it may also be of interest to study the biochemical mechanism(s) and the pathological relevance of interplay with other pathways including the extrinsic apoptotic pathways also activated by morusin, as well as inflammatory signaling, given the involvement of PKR in this mechanism.
2.3. Clinical Potential of Morusin Treatment in Pathophysiology
Exploding number of causality studies linking specific cellular processes to human pathologies, together with morusin implication in the processes, warranted assessment of the clinical potential of morusin in various disease settings in vivo and in vitro. Here, we discuss recent advances in the understanding of the potential application of morusin in treating common human diseases.
2.3.1. Inflammatory Diseases
The anti-inflammatory capacity of morusin has been tested in various models of inflammatory diseases, which was initiated with several in vitro studies on morusin-mediated inhibition of iNOS, cyclooxygenases and lipoxygenases [27,41]—(1) Lee et al. demonstrated that morusin reduces musin secretion in NCI-H292 cancer cells with mucoepidermoid characteristics following chronic inflammation [25]. This in vitro observation was concordant with in vivo results in which morusin treatment alleviates the hypersecretion of airway mucin in a rat model of bronchitis induced by sulfur dioxide treatment. (2) Morusin displays potential anti-allergic and anti-inflammatory effects on atopic dermatitis, a common chronic inflammatory skin disease, which was well-demonstrated by Jin et al., in their in vitro study with MC/9 mast cells and HaCat keratinocytes [24]. Mechanistically, they proposed the dual roles of morusin in modulating inflammatory signals in both keratinocytes and immune cells via STAT1/NF-κB and lipoxygenase, respectively. (3) Morusin treatment inhibits the NF-κB signaling pathway and thereby dampens IL-1β-induced chondrocyte inflammation and osteoarthritis using mouse chondrocytes and destabilization of the medial meniscus (DMM) model, a mouse model of osteoarthritis [26]. (4) Morusin treatment has been tested in a chemical-induced rat model of colitis. Histological observations following morusin gavage in the rats showed a significant reduction of tissue damage score and pro-inflammatory markers (TGF-β1 and IL-1β) and an increase in the level of antioxidant enzymes (superoxide dismutase and catalase), indicating that morusin may have therapeutic potential in treating inflammatory bowel disease [28].
Numerous microbes and viruses take advantage of host cellular processes and machineries to maximize their infection capacities. One such strategy is to provoke cellular and systemic inflammatory responses. Morusin has been administered to a few models of infectious diseases, such as an in vivo mouse model of mycoplasma pneumonia infection, in which morusin treatment efficiently suppressed mycoplasma pneumonia via the inhibition of Wnt/β-catenin and NF-κB signaling [29]. Morusin also appears to exert anti-microbial and anti-viral activities on microorganisms per se; Pang et al. and We et al., demonstrated that morusin has the remarkable capacity to target Staphylococcus aureus and clinical methicillin-resistant Staphylococcus aureus (MRSA) in vitro and in vivo [75,76]. The mechanism of action may be associated with the chemical properties of morusin as an isopentenyl leading to an increase in membrane permeability, inhibition of the phospholipid-repair system and dissipation of the proton motive force of the bacteria [77]. Mechanisms underlying the anti-viral effects of morusin are less clear. One possible explanation may be that morusin exerts its effects by allosteric inhibition of essential viral components. Of note, an in silico analysis proposed that morusin could directly inhibit HIV-1 reverse transcriptase activity [78] and SARS-CoV-2 protease [79].
2.3.2. Neurological Disorders
In Chinese medicine, Morus alba has been used as a neuroprotective herb. The neuroprotective functions of morusin and the implications for disease have recently been appreciated at the cellular and molecular levels. For example, morusin treatment protects neuroblastoma SH-SY5Y cells from nitric oxide-induced cell death [31]. This in vitro observation was also recapitulated by a follow-up study in which the in vivo relevance of the antioxidant capacities of morusin in neuroprotection was tested [80]. In the study, authors used a rat model of memory disorder induced by aluminum trichloride (AlCl3), showing that morusin ameliorates the impaired memory and learning capacity by decreasing the AlCl3-induced rise in brain acetylcholinesterase (AChE) activity and brain oxidative stress levels. Although in vivo studies of the effects of morusin on neurological disorders are very limited, morusin may exert beneficial functions against neurodegenerative diseases including Alzheimer’s disease, given that (1) morusin shows inhibitory activity against AChE, butyrylcholinesterase (BChE) and β-site amyloid precursor protein cleaving enzyme 1 (BACE1), which play important roles in the prevention and treatment of Alzheimer’s disease [18,81] and (2) morusin treatment activates autophagy in normal cells and the dysregulation of autophagy significantly contributes to the pathogenesis of neurodegenerative disorders [68,69]. Accordingly, an integrative approach of pharmacokinetics and a structural bioinformatics approach of 210 plant compounds revealed that morusin displays high potential as an anti-Alzheimer drug [82], corroborating the idea of the clinical potential of morusin in targeting neurodegenerative diseases.
2.3.3. Diabetes
Diabetes mellitus is a pathological condition in which the body displays impaired capacity to maintain coordinated systemic fuel metabolism upon various nutrient perturbations, particularly perturbations of glucose, due to (1) pancreatic β-cell dysfunction followed by reduced serum levels of insulin (Type I diabetes), and/or (2) reduced insulin sensitivity of systemic cells and relatively reduced insulin secretion from pancreatic β-cells (Type II diabetes) [83]. Although numerous factors have been implicated in such confounding metabolic disorders, the causalities and underlying mechanisms are not fully understood. Among these, the reduced lipid storage capacity of adipocytes and aberrant lipid metabolism with ROS accumulation in metabolically relevant organs including adipocytes, liver, muscle and pancreatic β-cells have been tightly associated with the pathogenesis of the disease [84,85]. In this context, multiple in vivo and in vitro evidence linked the versatile effects of morusin on the metabolic pathways to its clinical potential in treating diabetes. For example, two independent studies showed that morusin treatment ameliorates hyperglycemia and dysregulated lipid homeostasis in a mice model of type I diabetes induced by streptozotocin treatment that specifically kills pancreatic β-cells [59,60]. Such mechanisms may involve morusin-mediated alleviation of the peroxidation of lipid species spilled over from adipocytes, due to insulin secretion levels insufficient to maintain the fat storage capacity of adipocytes.
Although no studies have yet tested the effects of morusin on the pathogenesis of type II diabetes using relevant in vivo models (e.g., ob/ob, db/db mouse or HFD-fed mice), morusin may also exert anti-diabetes effects in this context, considering that morusin potentially regulates systemic ROS levels, lipogenesis, lipolysis, C/EBPβ and PPARγ signaling, arachidonic acid metabolism and ER-resident proteins implicated in ER stress [16,59,64], all of which have been frequently implicated in type II diabetes as well as type I diabetes [42,63,86,87].
2.3.4. Cancer
The implication of morusin in multiple biological pathways that cancers take advantage of, described above, led to many studies testing the antitumoral capacities of morusin in vitro, with recent initiation of research on its clinical potential in vivo. Indeed, very early studies on the chemical properties of morusin already appreciated its antitumoral potential [6], connecting the strong antioxidant capacity of morusin to the aberrant ROS production broadly relevant to the pathogenesis of cancers [88].
The effects of morusin on specific subtypes of cancers and the underlying biochemical mechanisms have recently been studied using various in vitro and in vivo model systems (Table 1). For example, Wang et al., demonstrated the clinical potential of morusin in targeting the stemness capacity of human cervical carcinoma in vitro [40]. Such an approach may be relevant, as NF-κB is a well-established upstream transcription factor of genes for stemness and metastatic capacities of various cancers including cervical cancers [33]. In this study, morusin treatment was shown to decrease the expression of Oct4, SOX2, ALDH1, as well as epithelial-to-mesenchymal markers, thereby decreasing the stemness signature of the cancer cells. Morusin-mediated inhibition of the NF-κB pathway also leads to reduced expression of Bcl-2 and upregulation of pro-apoptotic proteins including Bax and caspase-3, which results in the apoptotic death of the cancer cells. Morusin may also display antitumor effects independent of its capacity to activate apoptosis at non-cytotoxic concentrations (lower than 10 μM), since morusin promotes VDAC-mediated Ca2+ influx into mitochondria leading to mitochondrial Ca2+ overload and mitochondrial dysfunction and subsequent paraptosis-like cell death observed in an in vitro and in vivo model of epithelial ovarian cancer with apoptotic resistance [37].

Table 1.
The Effects of Morusin on Subtypes of Cancer.
Morusin treatment exerts beneficial effects against liver cancers as well. The intravenous injection of morusin in a xenograft mouse model of hepatocarcinoma significantly reduced tumor growth without any side effects. The histological and biochemical evidence provided in this study indicated that the mechanism may be associated with the activation of p53, survivin, cyclin B1 and caspase-3 and a decrease in the expression of NF-κB [47]. Moreover, morusin treatment also suppresses the metastatic capacity of the human hepatoma SK-Hep1 cell line, which may be biochemically associated with inhibition of STAT3 and NF-κB followed by the reduction of the expression of metastatic markers, such as vimentin, α2- and β1-integrins [34]. Such in vitro observations were also confirmed in an experiment with immunocompromised mice xenografted with SK-Hep1 cells, in which morusin treatment significantly decreased lung colonization of the cancer cells. Following the report that morusin effectively inhibits the growth of tumors in mice transplanted with H22 liver tumor cells [46], similar inhibitory effects of morusin on tumor xenografts were observed for liver, ovary, breast, kidney and gastric cancer cells [37,47,54,65,90]. In addition to inhibitory effects on tumor growth, tube formation of human umbilical vein endothelial cells (HUVEC) and expression of angiogenesis-related genes were also inhibited by morusin treatment in vitro [47,52], suggesting the anti-metastatic potential of morusin.
Morusin also targets the STAT3 pathway and its downstream targets, including survivin, cyclin B1, to exert antitumor activity in prostate and pancreatic cancers [36,48]. In the case of pancreatic cancer, pancreatic lipase may be another relevant target of morusin, considering (1) the strong association between pancreatic cancer and the levels of pancreatic lipase [48] and (2) the potential role of morusin in the allosteric regulation of pancreatic lipase [61].
The antitumor capacity of morusin has also been tested in glioblastoma. For example, the stemness capacity of glioblastoma multiforme (GBM) cancer stem cells (GSCs) were targeted by morusin treatment in vivo and in vitro [39]. This observation may be mechanistically attributable to the expression of genes downstream of NF-κB, which was already proposed in the previously described study with the cervical carcinoma model [40]. Of note, the reduced stemness of the cancers upon morusin treatment results in adipocyte-like trans-differentiation, followed by activation of intrinsic apoptosis [39]. The underlying mechanisms may also involve the activation of extrinsic apoptosis, as morusin treatment promotes expression of an extrinsic receptor, Death receptor 5, thereby sensitizing the glioblastoma to TRAIL, an antitumor chemical that mimics ligands for the extrinsic receptor [38].
Morusin may also employ other various cellular processes independent of NF-κB for targeting cancers—(1) An in vitro study of gastric cancer showed that morusin governs the expression of c-Myc and multiple downstream genes including CDKs and cyclins implicated in cell proliferation, thereby exerting antitumor capacity [90]. (2) The PI3K/AKT pathway has been proposed as a target of morusin in treating osteosarcoma in vitro [55]. (3) Morusin may directly target the MAPK pathway in renal carcinoma in vitro, although this should be further validated considering the intimate crosstalk between the NF-κB and MAPK pathway [54]. (4) C/EBPβ- and PPARγ-mediated lipoapoptosis may be therapeutically relevant targets of morusin in breast cancers, proposed in a study using multiple breast cancer cell lines and a xenograft model of breast cancers [64]. This may be of great importance considering that the capacity of mitochondrial fatty acid oxidation and fatty acid availability associated with host metabolism have recently emerged as key factors contributing to the pathogenesis of breast cancers [91,92]. (5) Studies with a panel of lung cancer cell lines showed that morusin treatment not only modulates conventional targets including NF-κB, STAT3 and downstream genes such as VEGF but may also directly bind and dephosphorylate EGFR on an active site, which would contribute to the further reduction of STAT3/NF-κB activity [14].
A cancer cell continuously undergoes dynamic remodeling of numerous cellular processes, which confer various benefits such as drug resistance. In this context, the two recent studies from our lab have highlighted the biochemical mechanisms by which cancer cells gain resistance against the cytotoxicity of morusin [68,74]. As described in the previous section, cancer cells promote autophagy and SG formation in response to morusin treatment, thereby desensitizing the apoptotic signals exerted by morusin treatment. Indeed, autophagy and SGs have already been proposed as the ‘shield pathways’ of cancers against various drug treatments. This notion, together with multiple biochemical studies in which morusin synergized with inhibitors of either autophagy or SG formation to boost its antitumor capacity, proposed the cotreatment of morusin and the inhibitors as a therapeutically relevant strategy for targeting cancers with drug resistance. However, whether such resistant mechanisms are still relevant in the context of various subtypes of cancers in vivo should be addressed.
3. Conclusions
The well-acknowledged functions of the root bark of Morus in Chinese herbal medicine have been expanded to the potential application of individual phytochemicals extracted from such arcane plants to treat multiple chronic diseases common in modern society. Morusin has been appreciated as a phytochemical with the unique physiochemical properties of isoprene flavonoids, allowing for versatile salutary effects including antioxidant and tumor killing activities. Morusin reduces ROS formation and inflammation at non-cytotoxic concentrations (lower than 10 μM), while it induces ROS formation and cytotoxicity against cancer cells at high concentrations (higher than 20 μM). A growing body of evidence, described here, highlighted the biological properties of morusin in multiple in vitro and in vivo pathophysiological settings and proposed clinical potential. The as yet unraveled clinical potential of morusin is waiting for thorough scientific assessment of the effects of morusin on human pathology with an accompanying understanding of the underlying biochemical mechanisms and effective platforms, which may allow for bona fide application of morusin in patients with various pathologies in the near future.
Author Contributions
Conceptualization, D.W.C., S.W.C. and C.Y.C.; writing—original draft preparation, D.W.C., S.W.C., S.-G.L. and C.Y.C.; writing—review and editing, S.-G.L. and C.Y.C.; visualization, S.W.C. and D.W.C.; supervision, C.Y.C.; project administration, C.Y.C.; funding acquisition, C.Y.C. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the National Research Foundation of Korea grant (2020-R1F1A1068120 and SRC-2017R1A5A1014560 to C.Y.C.) funded by the Korea Ministry of Science and ICT (Information and Communications Technology).
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- Yang, S.; Wang, B.L.; Li, Y. Advances in the pharmacological study of Morus alba L. Yao Xue Xue Bao 2014, 49, 824–831. [Google Scholar] [PubMed]
- Nomura, T.; Fukai, T.; Matsumoto, J.; Ohmori, T. Constituents of the cultivated mulberry tree. Planta Med. 1982, 46, 28–32. [Google Scholar] [CrossRef] [PubMed]
- Nomura, T.; Fukai, T.; Shimada, T.; Chen, I.S. Components of Root Bark of Morus australis. Planta Med. 1983, 49, 90–94. [Google Scholar] [CrossRef] [PubMed]
- Nomura, T.; Fukai, T.; Hano, Y. Constituents of the Chinese crude drug “sang-bai-pi” (Morus root bark). Planta Med. 1983, 47, 30–34. [Google Scholar] [CrossRef] [PubMed]
- Hano, Y.; Hirakura, K.; Nomura, T.; Terada, S.; Fukushima, K. Components of root bark of morus lhou1 1. Structures of two new natural diels-alder adducts, kuwanons N and o. Planta Med. 1984, 50, 127–130. [Google Scholar] [CrossRef]
- Nomura, T.; Fukai, T.; Hano, Y.; Yoshizawa, S.; Suganuma, M.; Fujiki, H. Chemistry and anti-tumor promoting activity of Morus flavonoids. Prog. Clin. Biol. Res. 1988, 280, 267–281. [Google Scholar]
- Yan, J.; Ruan, J.; Huang, P.; Sun, F.; Zheng, D.; Zhang, Y.; Wang, T. The structure-activity relationship review of the main bioactive constituents of Morus genus plants. J. Nat. Med. 2020, 74, 331–340. [Google Scholar] [CrossRef]
- Cho, J.K.; Ryu, Y.B.; Curtis-Long, M.J.; Kim, J.Y.; Kim, D.; Lee, S.; Lee, W.S.; Park, K.H. Inhibition and structural reliability of prenylated flavones from the stem bark of Morus lhou on β-secretase (BACE-1). Bioorg. Med. Chem. Lett. 2011, 21, 2945–2948. [Google Scholar] [CrossRef]
- Syah, Y.M.; Juliawaty, L.D.; Achmad, S.A.; Hakim, E.H.; Takayama, H.; Said, I.M.; Latip, J. Phenolic constituents from the wood of Morus australis with cytotoxic activity. Z. Naturforsch. C J. Biosci. 2008, 63, 35–39. [Google Scholar] [CrossRef]
- Chen, X.; Mukwaya, E.; Wong, M.S.; Zhang, Y. A systematic review on biological activities of prenylated flavonoids. Pharm. Biol. 2014, 52, 655–660. [Google Scholar] [CrossRef]
- Hakim, E.H.; Achmad, S.A.; Juliawaty, L.D.; Makmur, L.; Syah, Y.M.; Aimi, N.; Kitajima, M.; Takayama, H.; Ghisalberti, E.L. Prenylated flavonoids and related compounds of the Indonesian Artocarpus (Moraceae). J. Nat. Med. 2006, 60, 161–184. [Google Scholar] [CrossRef] [PubMed]
- Dat, N.T.; Binh, P.T.; le Quynh, T.P.; Van Minh, C.; Huong, H.T.; Lee, J.J. Cytotoxic prenylated flavonoids from Morus alba. Fitoterapia 2010, 81, 1224–1227. [Google Scholar] [CrossRef]
- Bahramann, E. The pathology of testicular neoplasms. Z. Urol. Nephrol. 1976, 69, 91–97. [Google Scholar] [PubMed]
- Park, H.J.; Min, T.R.; Chi, G.Y.; Choi, Y.H.; Park, S.H. Induction of apoptosis by morusin in human non-small cell lung cancer cells by suppression of EGFR/STAT3 activation. Biochem. Biophys. Res. Commun. 2018, 505, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Parant, M.R.; Vial, H.J. Rapid and serial determination of protein kinase C activity and of the associated [3H]PDBu binding using a 96-well microtiter plate and a cell harvester. Anal. Biochem. 1990, 184, 283–290. [Google Scholar] [CrossRef]
- Shi, X.; Yang, S.; Zhang, G.; Song, Y.; Su, D.; Liu, Y.; Guo, F.; Shan, L.; Cai, J. The different metabolism of morusin in various species and its potent inhibition against UDP-glucuronosyltransferase (UGT) and cytochrome p450 (CYP450) enzymes. Xenobiotica 2016, 46, 467–476. [Google Scholar] [CrossRef] [PubMed]
- Hou, C.; Liu, W.; Liang, Z.; Han, W.; Li, J.; Ye, L.; Liu, M.; Cai, Z.; Zhao, J.; Chen, Y.; et al. UGT-mediated metabolism plays a dominant role in the pharmacokinetic behavior and the disposition of morusin in vivo and in vitro. J. Pharm. Biomed. Anal. 2018, 154, 339–353. [Google Scholar] [CrossRef]
- Kim, J.Y.; Lee, W.S.; Kim, Y.S.; Curtis-Long, M.J.; Lee, B.W.; Ryu, Y.B.; Park, K.H. Isolation of cholinesterase-inhibiting flavonoids from Morus lhou. J. Agric. Food Chem. 2011, 59, 4589–4596. [Google Scholar] [CrossRef]
- Reddy, G.R.; Ueda, N.; Hada, T.; Sackeyfio, A.C.; Yamamoto, S.; Hano, Y.; Aida, M.; Nomura, T. A prenylflavone, artonin E, as arachidonate 5-lipoxygenase inhibitor. Biochem. Pharmacol. 1991, 41, 115–118. [Google Scholar] [CrossRef]
- Shi, X.; Mackie, B.; Zhang, G.; Yang, S.; Song, Y.; Su, D.; Liu, Y.; Shan, L. Identification of the Metabolic Enzyme Involved Morusin Metabolism and Characterization of Its Metabolites by Ultraperformance Liquid Chromatography Quadrupole Time-of-Flight Mass Spectrometry (UPLC/Q-TOF-MS/MS). Evid. Based Complement. Altern. Med. eCAM 2016, 2016, 9240103. [Google Scholar] [CrossRef]
- Agarwal, S.; Mohamed, M.S.; Raveendran, S.; Rochani, A.K.; Maekawa, T.; Kumar, D.S. Formulation, characterization and evaluation of morusin loaded niosomes for potentiation of anticancer therapy. RSC Adv. 2018, 8, 32621–32636. [Google Scholar] [CrossRef]
- Tseng, T.-H.; Chuang, S.-K.; Hu, C.-C.; Chang, C.-F.; Huang, Y.-C.; Lin, C.-W.; Lee, Y.-J. The synthesis of morusin as a potent antitumor agent. Tetrahedron 2010, 66, 1335–1340. [Google Scholar] [CrossRef]
- Nitya Bankupalli, S.K.G.; Shaik, M. Comparative modeling of sodium- and chloride-dependent GABA transporter 1 and docking studies with natural compounds. J. Appl. Biol. Biotechnol. 2020, 8, 12–21. [Google Scholar]
- Jin, S.E.; Ha, H.; Shin, H.K.; Seo, C.S. Anti-Allergic and Anti-Inflammatory Effects of Kuwanon G and Morusin on MC/9 Mast Cells and HaCaT Keratinocytes. Molecules 2019, 24, 265. [Google Scholar] [CrossRef]
- Lee, H.J.; Ryu, J.; Park, S.H.; Woo, E.R.; Kim, A.R.; Lee, S.K.; Kim, Y.S.; Kim, J.O.; Hong, J.H.; Lee, C.J. Effects of Morus alba L. and Natural Products Including Morusin on In Vivo Secretion and In Vitro Production of Airway MUC5AC Mucin. Tuberc. Respir. Dis. 2014, 77, 65–72. [Google Scholar] [CrossRef]
- Jia, Y.; He, W.; Zhang, H.; He, L.; Wang, Y.; Zhang, T.; Peng, J.; Sun, P.; Qian, Y. Morusin Ameliorates IL-1β-Induced Chondrocyte Inflammation and Osteoarthritis via NF-κB Signal Pathway. Drug Des. Dev. Ther. 2020, 14, 1227–1240. [Google Scholar] [CrossRef]
- Cheon, B.S.; Kim, Y.H.; Son, K.S.; Chang, H.W.; Kang, S.S.; Kim, H.P. Effects of prenylated flavonoids and biflavonoids on lipopolysaccharide-induced nitric oxide production from the mouse macrophage cell line RAW 264.7. Planta Med. 2000, 66, 596–600. [Google Scholar] [CrossRef]
- Vochyanova, Z.; Pokorna, M.; Rotrekl, D.; Smekal, V.; Fictum, P.; Suchy, P.; Gajdziok, J.; Smejkal, K.; Hosek, J. Prenylated flavonoid morusin protects against TNBS-induced colitis in rats. PLoS ONE 2017, 12, e0182464. [Google Scholar] [CrossRef]
- Chen, C.; Wang, J.; Chen, J.; Zhou, L.; Wang, H.; Chen, J.; Xu, Z.; Zhu, S.; Liu, W.; Yu, R.; et al. Morusin alleviates mycoplasma pneumonia via the inhibition of Wnt/beta-catenin and NF-kappaB signaling. Biosci. Rep. 2019, 39. [Google Scholar] [CrossRef]
- Yang, Z.G.; Matsuzaki, K.; Takamatsu, S.; Kitanaka, S. Inhibitory effects of constituents from Morus alba var. multicaulis on differentiation of 3T3-L1 cells and nitric oxide production in RAW264.7 cells. Molecules 2011, 16, 6010–6022. [Google Scholar] [CrossRef]
- Lee, H.J.; da Lyu, H.; Koo, U.; Nam, K.W.; Hong, S.S.; Kim, K.O.; Kim, K.H.; Lee, D.; Mar, W. Protection of prenylated flavonoids from Mori Cortex Radicis (Moraceae) against nitric oxide-induced cell death in neuroblastoma SH-SY5Y cells. Arch. Pharm. Res. 2012, 35, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Arias-Salvatierra, D.; Silbergeld, E.K.; Acosta-Saavedra, L.C.; Calderon-Aranda, E.S. Role of nitric oxide produced by iNOS through NF-κB pathway in migration of cerebellar granule neurons induced by Lipopolysaccharide. Cell Signal. 2011, 23, 425–435. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Lenardo, M.J.; Baltimore, D. 30 Years of NF-kappaB: A Blossoming of Relevance to Human Pathobiology. Cell 2017, 168, 37–57. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.L.; Lai, D.Y.; Lee, Y.J.; Chen, N.F.; Tseng, T.H. Antitumor progression potential of morusin suppressing STAT3 and NFkappaB in human hepatoma SK-Hep1 cells. Toxicol. Lett. 2015, 232, 490–498. [Google Scholar] [CrossRef]
- Lee, J.C.; Won, S.J.; Chao, C.L.; Wu, F.L.; Liu, H.S.; Ling, P.; Lin, C.N.; Su, C.L. Morusin induces apoptosis and suppresses NF-kappaB activity in human colorectal cancer HT-29 cells. Biochem. Biophys. Res. Commun. 2008, 372, 236–242. [Google Scholar] [CrossRef]
- Lim, S.L.; Park, S.Y.; Kang, S.; Park, D.; Kim, S.H.; Um, J.Y.; Jang, H.J.; Lee, J.H.; Jeong, C.H.; Jang, J.H.; et al. Morusin induces cell death through inactivating STAT3 signaling in prostate cancer cells. Am. J. Cancer Res. 2015, 5, 289–299. [Google Scholar]
- Xue, J.; Li, R.; Zhao, X.; Ma, C.; Lv, X.; Liu, L.; Liu, P. Morusin induces paraptosis-like cell death through mitochondrial calcium overload and dysfunction in epithelial ovarian cancer. Chem. Biol. Interact. 2018, 283, 59–74. [Google Scholar] [CrossRef]
- Park, D.; Ha, I.J.; Park, S.Y.; Choi, M.; Lim, S.L.; Kim, S.H.; Lee, J.H.; Ahn, K.S.; Yun, M.; Lee, S.G. Morusin Induces TRAIL Sensitization by Regulating EGFR and DR5 in Human Glioblastoma Cells. J. Nat. Prod. 2016, 79, 317–323. [Google Scholar] [CrossRef]
- Guo, H.; Liu, C.; Yang, L.; Dong, L.; Wang, L.; Wang, Q.; Li, H.; Zhang, J.; Lin, P.; Wang, X. Morusin inhibits glioblastoma stem cell growth in vitro and in vivo through stemness attenuation, adipocyte transdifferentiation and apoptosis induction. Mol. Carcinog. 2016, 55, 77–89. [Google Scholar] [CrossRef]
- Wang, L.; Guo, H.; Yang, L.; Dong, L.; Lin, C.; Zhang, J.; Lin, P.; Wang, X. Morusin inhibits human cervical cancer stem cell growth and migration through attenuation of NF-κB activity and apoptosis induction. Mol. Cell. Biochem. 2013, 379, 7–18. [Google Scholar] [CrossRef]
- Chi, Y.S.; Jong, H.G.; Son, K.H.; Chang, H.W.; Kang, S.S.; Kim, H.P. Effects of naturally occurring prenylated flavonoids on enzymes metabolizing arachidonic acid: Cyclooxygenases and lipoxygenases. Biochem. Pharmacol. 2001, 62, 1185–1191. [Google Scholar] [CrossRef]
- Hanna, V.S.; Hafez, E.A.A. Synopsis of arachidonic acid metabolism: A review. J. Adv. Res. 2018, 11, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Elmore, S. Apoptosis: A review of programmed cell death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef] [PubMed]
- Pistritto, G.; Trisciuoglio, D.; Ceci, C.; Garufi, A.; D’Orazi, G. Apoptosis as anticancer mechanism: Function and dysfunction of its modulators and targeted therapeutic strategies. Aging 2016, 8, 603–619. [Google Scholar] [CrossRef] [PubMed]
- Cheng, P.S.; Hu, C.C.; Wang, C.J.; Lee, Y.J.; Chung, W.C.; Tseng, T.H. Involvement of the antioxidative property of morusin in blocking phorbol ester-induced malignant transformation of JB6 P(+) mouse epidermal cells. Chem. Biol. Interact. 2017, 264, 34–42. [Google Scholar] [CrossRef]
- Wan, L.Z.; Ma, B.; Zhang, Y.Q. Preparation of morusin from Ramulus mori and its effects on mice with transplanted H22 hepatocarcinoma. Biofactors 2014, 40, 636–645. [Google Scholar] [CrossRef]
- Gao, L.; Wang, L.; Sun, Z.; Li, H.; Wang, Q.; Yi, C.; Wang, X. Morusin shows potent antitumor activity for human hepatocellular carcinoma in vitro and in vivo through apoptosis induction and angiogenesis inhibition. Drug Des. Dev. Ther. 2017, 11, 1789–1802. [Google Scholar] [CrossRef]
- Kim, C.; Kim, J.H.; Oh, E.Y.; Nam, D.; Lee, S.G.; Lee, J.; Kim, S.H.; Shim, B.S.; Ahn, K.S. Blockage of STAT3 Signaling Pathway by Morusin Induces Apoptosis and Inhibits Invasion in Human Pancreatic Tumor Cells. Pancreas 2016, 45, 409–419. [Google Scholar] [CrossRef]
- Simon, A.R.; Rai, U.; Fanburg, B.L.; Cochran, B.H. Activation of the JAK-STAT pathway by reactive oxygen species. Am. J. Physiol. 1998, 275, C1640–C1652. [Google Scholar] [CrossRef]
- Grivennikov, S.I.; Karin, M. Dangerous liaisons: STAT3 and NF-kappaB collaboration and crosstalk in cancer. Cytokine Growth Factor Rev. 2010, 21, 11–19. [Google Scholar] [CrossRef]
- Kang, S.; Kim, E.O.; Kim, S.H.; Lee, J.H.; Ahn, K.S.; Yun, M.; Lee, S.G. Morusin induces apoptosis by regulating expression of Bax and Survivin in human breast cancer cells. Oncol. Lett. 2017, 13, 4558–4562. [Google Scholar] [CrossRef] [PubMed]
- Yin, X.L.; Lv, Y.; Wang, S.; Zhang, Y.Q. Morusin suppresses A549 cell migration and induces cell apoptosis by downregulating the expression of COX--2 and VEGF genes. Oncol. Rep. 2018, 40, 504–510. [Google Scholar] [CrossRef] [PubMed]
- Portt, L.; Norman, G.; Clapp, C.; Greenwood, M.; Greenwood, M.T. Anti-apoptosis and cell survival: A review. Biochim. Biophys. Acta 2011, 1813, 238–259. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Luo, J.; Luo, X.; Jia, W.; Fang, Z.; Yi, S.; Li, L. Morusin exerts anti-cancer activity in renal cell carcinoma by disturbing MAPK signaling pathways. Ann. Transl. Med. 2020, 8, 327. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Weng, Q.; Chen, J.; Han, J. Morusin inhibited human osteosarcoma via PI3K-AKT signaling pathway. Curr. Pharm. Biotechnol. 2020. [Google Scholar] [CrossRef]
- Roy, S.; Nicholson, D.W. Cross-talk in cell death signaling. J. Exp. Med. 2000, 192, F21–F25. [Google Scholar] [CrossRef]
- Goodpaster, B.H.; Sparks, L.M. Metabolic Flexibility in Health and Disease. Cell Metab. 2017, 25, 1027–1036. [Google Scholar] [CrossRef]
- Apel, K.; Hirt, H. Reactive oxygen species: Metabolism, oxidative stress and signal transduction. Annu. Rev. Plant Biol. 2004, 55, 373–399. [Google Scholar] [CrossRef]
- Singab, A.N.; El-Beshbishy, H.A.; Yonekawa, M.; Nomura, T.; Fukai, T. Hypoglycemic effect of Egyptian Morus alba root bark extract: Effect on diabetes and lipid peroxidation of streptozotocin-induced diabetic rats. J. Ethnopharmacol. 2005, 100, 333–338. [Google Scholar] [CrossRef]
- Abd El-Mawla, A.M.; Mohamed, K.M.; Mostafa, A.M. Induction of Biologically Active Flavonoids in Cell Cultures of Morus nigra and Testing their Hypoglycemic Efficacy. Sci. Pharm. 2011, 79, 951–961. [Google Scholar] [CrossRef]
- Hou, X.D.; Ge, G.B.; Weng, Z.M.; Dai, Z.R.; Leng, Y.H.; Ding, L.L.; Jin, L.L.; Yu, Y.; Cao, Y.F.; Hou, J. Natural constituents from Cortex Mori Radicis as new pancreatic lipase inhibitors. Bioorg. Chem. 2018, 80, 577–584. [Google Scholar] [CrossRef] [PubMed]
- Foufelle, F.; Fromenty, B. Role of endoplasmic reticulum stress in drug-induced toxicity. Pharmacol. Res. Perspect. 2016, 4, e00211. [Google Scholar] [CrossRef] [PubMed]
- Elfaki, I.; Mir, R.; Almutairi, F.M.; Duhier, F.M.A. Cytochrome P450: Polymorphisms and Roles in Cancer, Diabetes and Atherosclerosis. Asian Pac. J. Cancer Prev. 2018, 19, 2057–2070. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.R.; Kim, J.E.; Choi, J.Y.; Park, J.J.; Kim, H.R.; Song, B.R.; Park, J.W.; Kang, M.J.; Choi, Y.W.; Kim, K.M.; et al. Morusin Functions as a Lipogenesis Inhibitor as Well as a Lipolysis Stimulator in Differentiated 3T3-L1 and Primary Adipocytes. Molecules 2018, 23, 2004. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Wang, Q.; Dong, L.; Liu, C.; Sun, Z.; Gao, L.; Wang, X. Morusin suppresses breast cancer cell growth in vitro and in vivo through C/EBPβ and PPARγ mediated lipoapoptosis. J. Exp. Clin. Cancer Res. 2015, 34, 137. [Google Scholar] [CrossRef] [PubMed]
- Dikic, I.; Elazar, Z. Mechanism and medical implications of mammalian autophagy. Nat. Rev. Mol. Cell Biol. 2018, 19, 349–364. [Google Scholar] [CrossRef]
- Li, X.; Zhou, Y.; Li, Y.; Yang, L.; Ma, Y.; Peng, X.; Yang, S.; Liu, J.; Li, H. Autophagy: A novel mechanism of chemoresistance in cancers. Biomed. Pharmacother. 2019, 119, 109415. [Google Scholar] [CrossRef]
- Cho, S.W.; Na, W.; Choi, M.; Kang, S.J.; Lee, S.G.; Choi, C.Y. Autophagy inhibits cell death induced by the anti-cancer drug morusin. Am. J. Cancer Res. 2017, 7, 518–530. [Google Scholar]
- Fujikake, N.; Shin, M.; Shimizu, S. Association Between Autophagy and Neurodegenerative Diseases. Front. Neurosci. 2018, 12, 255. [Google Scholar] [CrossRef]
- Buchan, J.R.; Parker, R. Eukaryotic stress granules: The ins and outs of translation. Mol. Cell 2009, 36, 932–941. [Google Scholar] [CrossRef]
- Wolozin, B.; Ivanov, P. Stress granules and neurodegeneration. Nat. Rev. Neurosci. 2019, 20, 649–666. [Google Scholar] [CrossRef] [PubMed]
- Protter, D.S.W.; Parker, R. Principles and Properties of Stress Granules. Trends Cell Biol. 2016, 26, 668–679. [Google Scholar] [CrossRef] [PubMed]
- Yague, E.; Raguz, S. Escape from stress granule sequestration: Another way to drug resistance? Biochem. Soc. Trans. 2010, 38, 1537–1542. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.J.; Choi, D.W.; Cho, S.W.; Han, J.; Yang, S.; Choi, C.Y. Stress Granule Formation Attenuates RACK1-Mediated Apoptotic Cell Death Induced by Morusin. Int. J. Mol. Sci. 2020, 21, 5360. [Google Scholar] [CrossRef]
- Zuo, G.Y.; Yang, C.X.; Han, J.; Li, Y.Q.; Wang, G.C. Synergism of prenylflavonoids from Morus alba root bark against clinical MRSA isolates. Phytomedicine 2018, 39, 93–99. [Google Scholar] [CrossRef]
- Pang, D.; Liao, S.; Wang, W.; Mu, L.; Li, E.; Shen, W.; Liu, F.; Zou, Y. Destruction of the cell membrane and inhibition of cell phosphatidic acid biosynthesis in Staphylococcus aureus: An explanation for the antibacterial mechanism of morusin. Food Funct. 2019, 10, 6438–6446. [Google Scholar] [CrossRef]
- Wu, S.C.; Han, F.; Song, M.R.; Chen, S.; Li, Q.; Zhang, Q.; Zhu, K.; Shen, J.Z. Natural Flavones from Morus alba against Methicillin-Resistant Staphylococcus aureus via Targeting the Proton Motive Force and Membrane Permeability. J. Agric. Food Chem. 2019, 67, 10222–10234. [Google Scholar] [CrossRef]
- Syahdi, R.R.; Mun’im, A.; Suhartanto, H.; Yanuar, A. Virtual screening of Indonesian herbal database as HIV-1 reverse transcriptase inhibitor. Bioinformation 2012, 8, 1206–1210. [Google Scholar] [CrossRef]
- Singh, S.; Florez, H. Coronavirus disease 2019 drug discovery through molecular docking. F1000Research 2020, 9, 502. [Google Scholar] [CrossRef]
- Gupta, G.; Chellappan, D.K.; Agarwal, M.; Ashwathanarayana, M.; Nammi, S.; Pabreja, K.; Dua, K. Pharmacological Evaluation of the Recuperative Effect of Morusin Against Aluminium Trichloride (AlCl3)-Induced Memory Impairment in Rats. Cent. Nerv. Syst. Agents Med. Chem. 2017, 17, 196–200. [Google Scholar] [CrossRef]
- Kuk, E.B.; Jo, A.R.; Oh, S.I.; Sohn, H.S.; Seong, S.H.; Roy, A.; Choi, J.S.; Jung, H.A. Anti-Alzheimer’s disease activity of compounds from the root bark of Morus alba L. Arch. Pharm. Res. 2017, 40, 338–349. [Google Scholar] [CrossRef] [PubMed]
- Borah, K.; Sharma, S.; Silla, Y. Structural bioinformatics-based identification of putative plant based lead compounds for Alzheimer Disease Therapy. Comput. Biol. Chem. 2019, 78, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Kharroubi, A.T.; Darwish, H.M. Diabetes mellitus: The epidemic of the century. World J. Diabetes 2015, 6, 850–867. [Google Scholar] [CrossRef] [PubMed]
- Guilherme, A.; Virbasius, J.V.; Puri, V.; Czech, M.P. Adipocyte dysfunctions linking obesity to insulin resistance and type 2 diabetes. Nat. Rev. Mol. Cell Biol. 2008, 9, 367–377. [Google Scholar] [CrossRef] [PubMed]
- Athyros, V.G.; Doumas, M.; Imprialos, K.P.; Stavropoulos, K.; Georgianou, E.; Katsimardou, A.; Karagiannis, A. Diabetes and lipid metabolism. Hormones 2018, 17, 61–67. [Google Scholar] [CrossRef]
- Tsalamandris, S.; Antonopoulos, A.S.; Oikonomou, E.; Papamikroulis, G.A.; Vogiatzi, G.; Papaioannou, S.; Deftereos, S.; Tousoulis, D. The Role of Inflammation in Diabetes: Current Concepts and Future Perspectives. Eur. Cardiol. 2019, 14, 50–59. [Google Scholar] [CrossRef]
- Kim, H.I.; Ahn, Y.H. Role of peroxisome proliferator-activated receptor-gamma in the glucose-sensing apparatus of liver and beta-cells. Diabetes 2004, 53 (Suppl. 1), S60–S65. [Google Scholar] [CrossRef]
- Kumari, S.; Badana, A.K.; Murali, M.G.; Shailender, G.; Malla, R. Reactive Oxygen Species: A Key Constituent in Cancer Survival. Biomark. Insights 2018, 13, 1177271918755391. [Google Scholar] [CrossRef]
- Agarwal, S.; Mohamed, M.S.; Mizuki, T.; Maekawa, T.; Sakthi Kumar, D. Chlorotoxin modified morusin-PLGA nanoparticles for targeted glioblastoma therapy. J. Mater. Chem. B 2019, 7, 5896–5919. [Google Scholar] [CrossRef]
- Wang, F.; Zhang, D.; Mao, J.; Ke, X.X.; Zhang, R.; Yin, C.; Gao, N.; Cui, H. Morusin inhibits cell proliferation and tumor growth by down-regulating c-Myc in human gastric cancer. Oncotarget 2017, 8, 57187–57200. [Google Scholar] [CrossRef]
- Hao, J.; Zhang, Y.; Yan, X.; Yan, F.; Sun, Y.; Zeng, J.; Waigel, S.; Yin, Y.; Fraig, M.M.; Egilmez, N.K.; et al. Circulating Adipose Fatty Acid Binding Protein Is a New Link Underlying Obesity-Associated Breast/Mammary Tumor Development. Cell Metab. 2018, 28, 689–705.e685. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Fahrmann, J.F.; Lee, H.; Li, Y.J.; Tripathi, S.C.; Yue, C.; Zhang, C.; Lifshitz, V.; Song, J.; Yuan, Y.; et al. JAK/STAT3-Regulated Fatty Acid beta-Oxidation Is Critical for Breast Cancer Stem Cell Self-Renewal and Chemoresistance. Cell Metab. 2018, 27, 1357. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).