NR2F6 as a Prognostic Biomarker in HNSCC
Abstract
:1. Introduction
2. Results
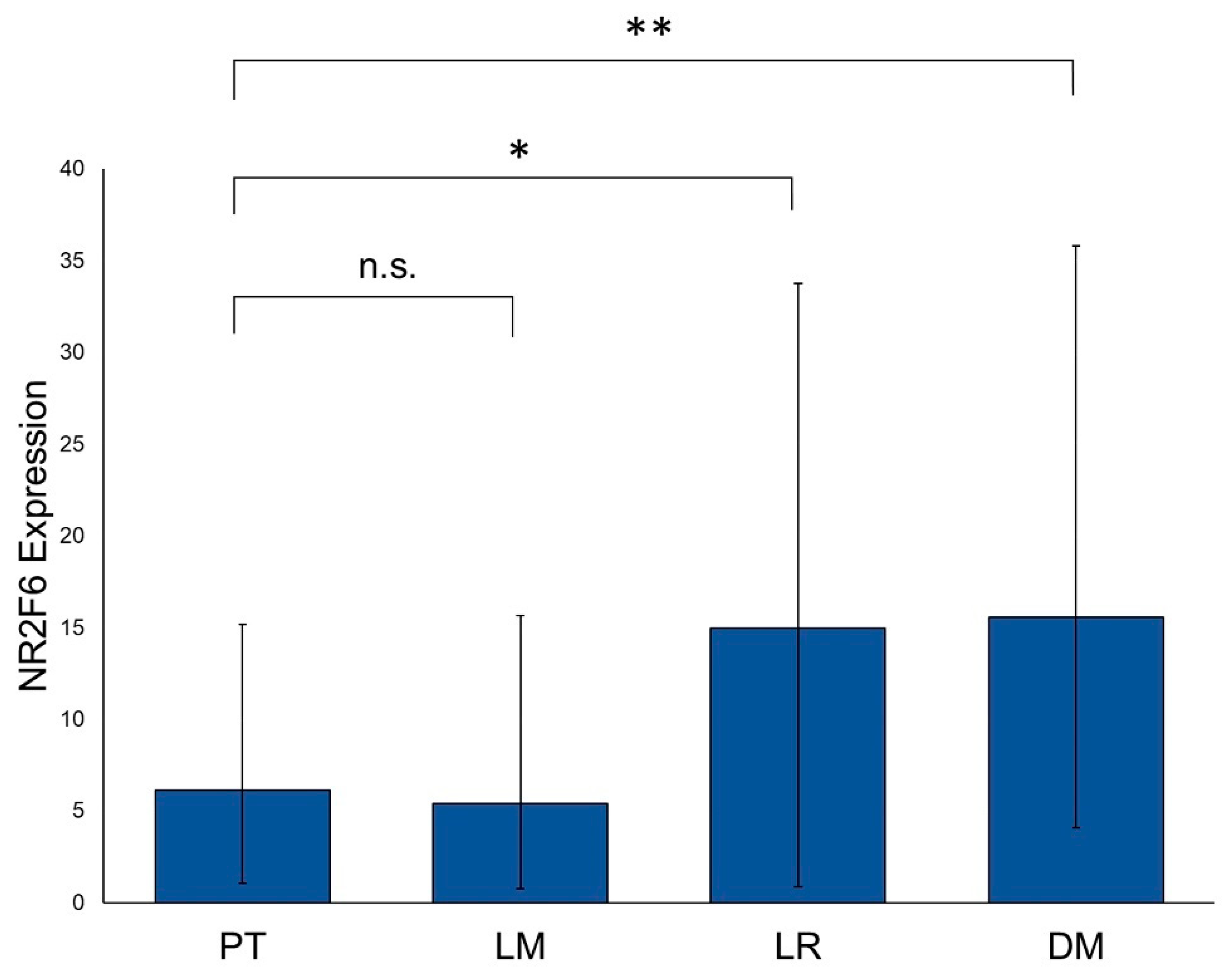
2.1. NR2F6 Expression in Primary Tumors, Lymph Node Metastases, Distant Metastases, and Local Recurrences
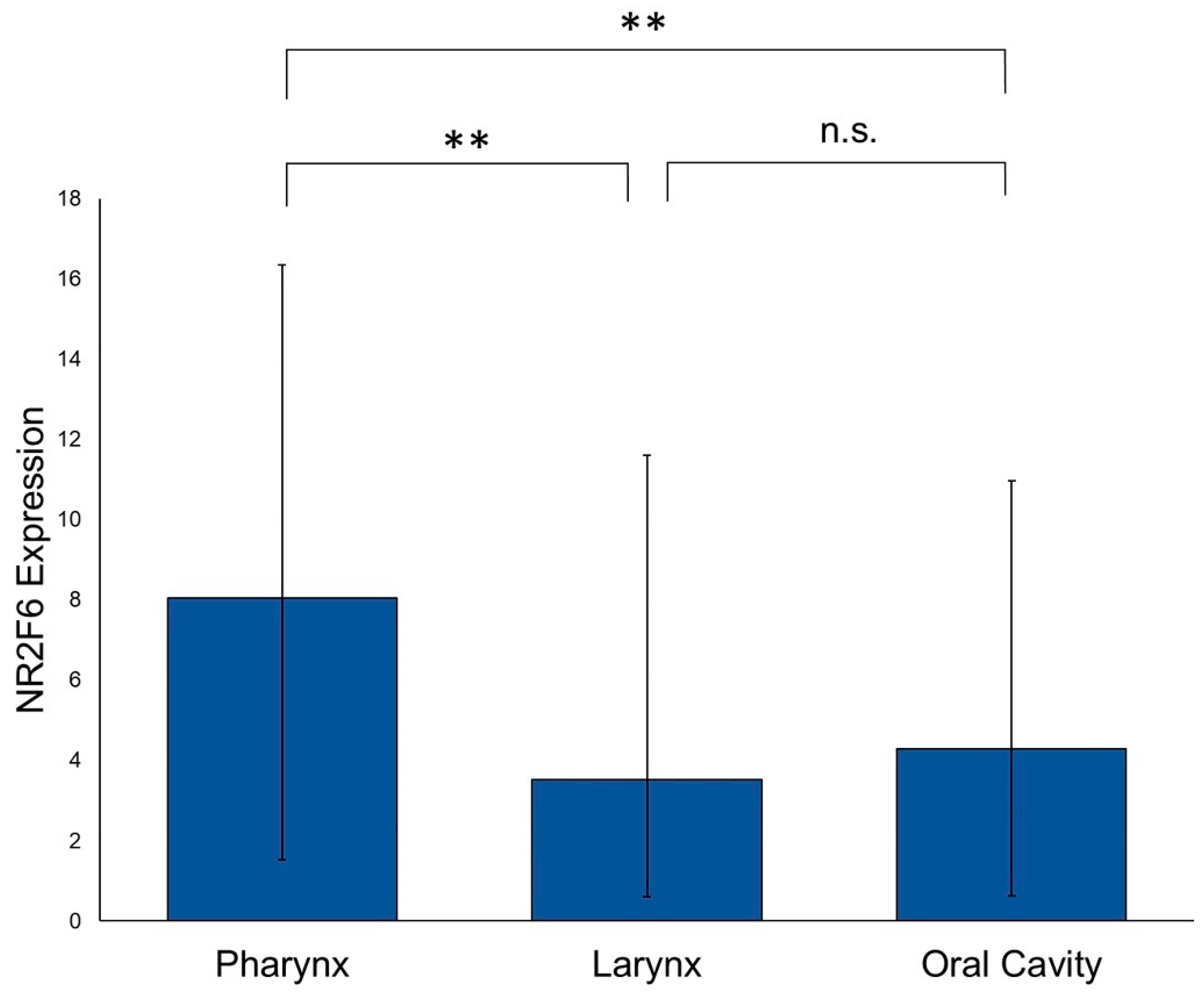
2.2. NR2F6 Expression in Different HNSCC Tumor Sites
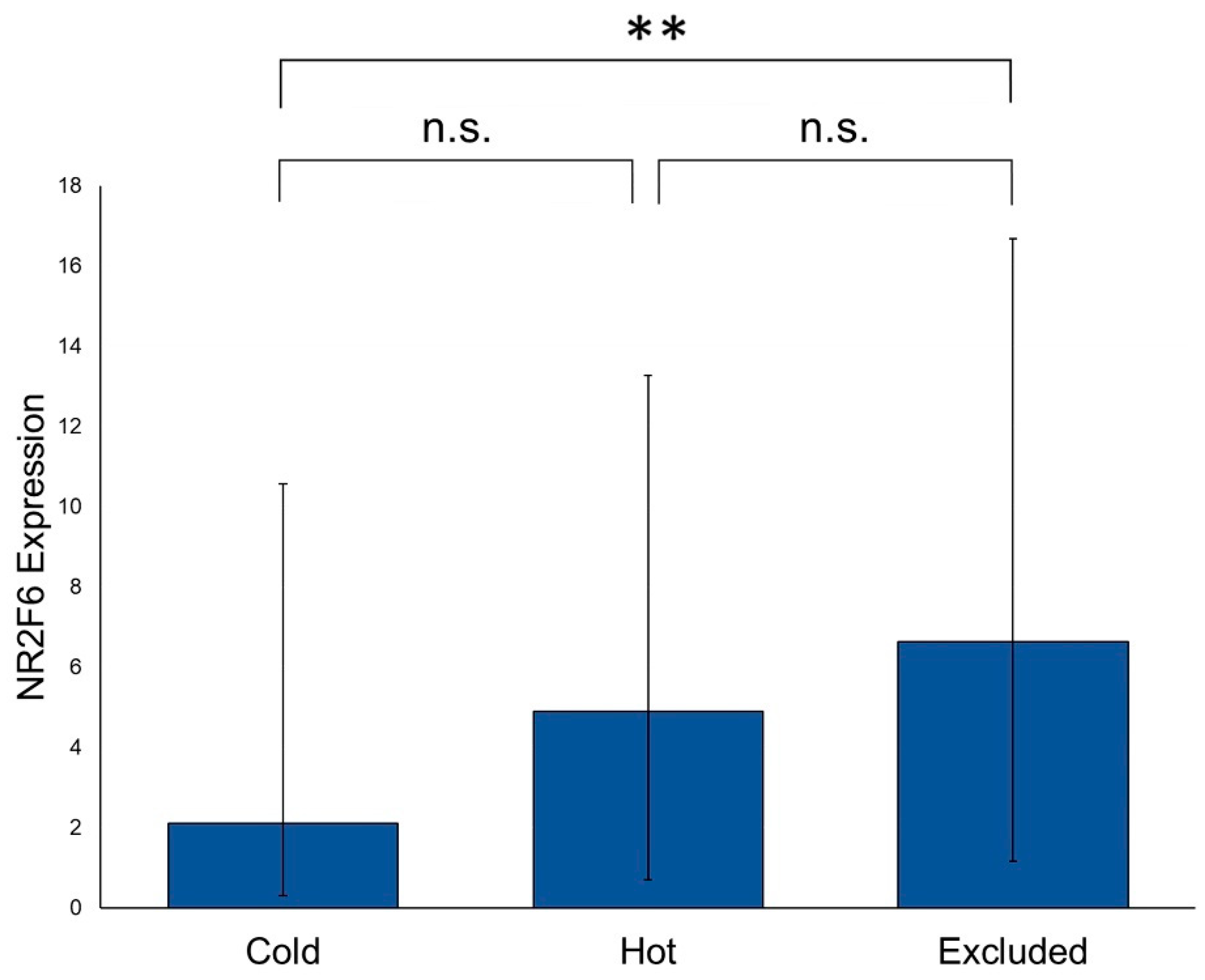
2.3. NR2F6 Expression Is Higher in Primary HNSCCs with Immune Cell Infiltration than in Primary HNSCCs without Immune Cell Infiltration
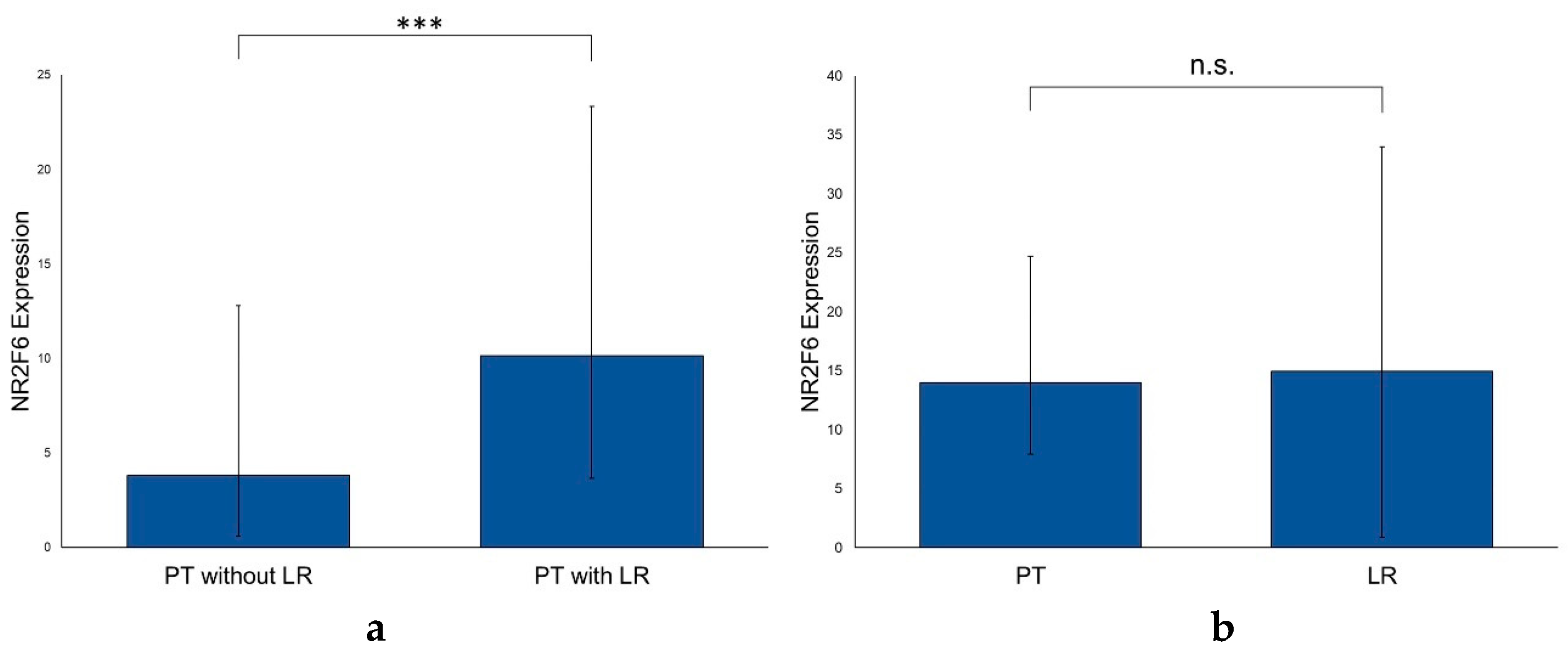
2.4. NR2F6 Expression Is Higher in Primary HNSCCs with Subsequent Local Recurrences
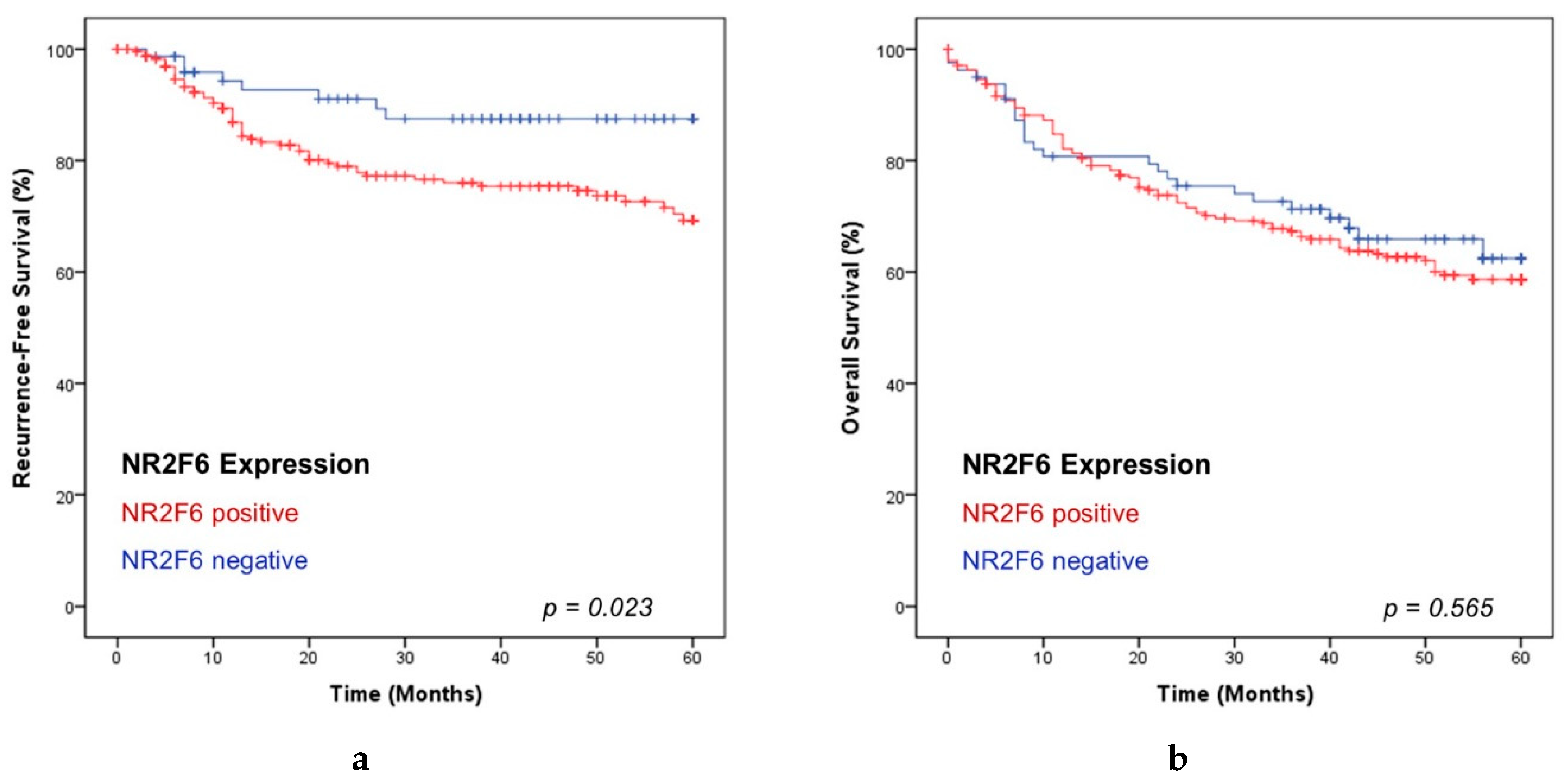
2.5. NR2F6-Expressing HNSCCs Are Associated with a Short Recurrence-Free Survival
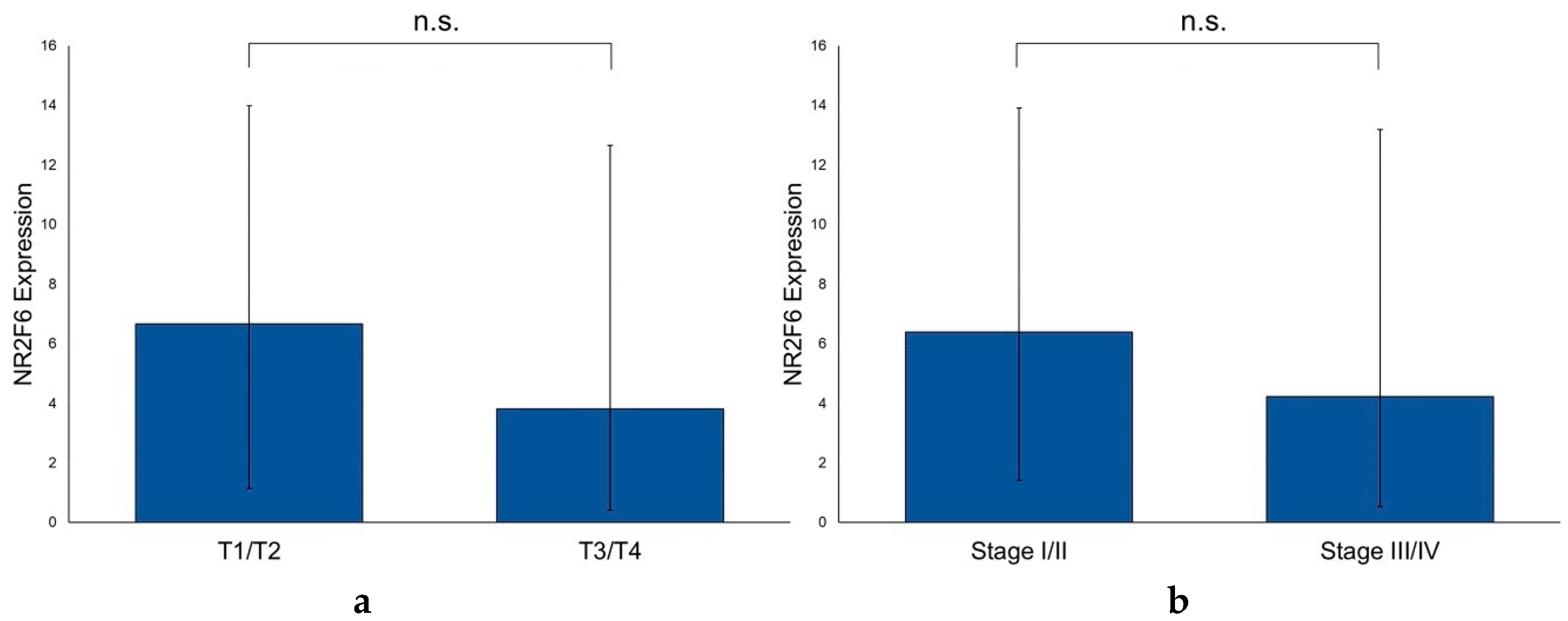
2.6. The NR2F6 Expression Is Independent of T and UICC Stages and the p16 Status of PTs
3. Discussion
4. Materials and Methods
4.1. Patient Data and Tumor Material
4.2. Immunohistochemistry and Evaluation of Stained Slides
4.3. Pathological Evaluation of Immune Cell Infiltration
4.4. Statistical Analysis and Visualization
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CTLA-4 | Cytotoxic T lymphocyte-associated protein 4 |
| DM | Distant metastasis |
| FFPE | Formalin-fixed paraffin-embedded |
| HE | Hematoxylin-eosin |
| HNSCC | Head and neck squamous cell carcinoma |
| HPV | Human papillomavirus |
| HR | Hazard Ratio |
| IFN-γ | Interferon-γ |
| IL-21 | Interleukin-21 |
| LM | Lymph node metastasis |
| LR | Local recurrence |
| NR2F6 | Nuclear receptor family 2 subgroup 6 |
| PD-1 | Programmed cell death-1 |
| PD-L1 | Programmed cell death ligand-1 |
| PT | Primary tumor |
| ROI | Region of Interest |
| TMA | Tissue microarray |
References
- Parker, S.L.; Tong, T.; Bolden, S.; Wingo, P.A. Cancer statistics, 1996. CA Cancer J. Clin. 1996, 46, 5–27. [Google Scholar] [CrossRef]
- Vokes, E.E.; Weichselbaum, R.R.; Lippman, S.M.; Hong, W.K. Head and neck cancer. N. Engl. J. Med. 1993, 328, 184–194. [Google Scholar] [CrossRef] [PubMed]
- Larson, D.L.; Lindberg, R.D.; Lane, E.; Goepfert, H. Major complications of radiotherapy in cancer of the oral cavity and oropharynx. A 10 year retrospective study. Am. J. Surg. 1983, 146, 531–536. [Google Scholar] [CrossRef]
- Ervin, T.J.; Clark, J.R.; Weichselbaum, R.R.; Fallon, B.G.; Miller, D.; Fabian, R.L.; Posner, M.R.; Norris, C.M.; Tuttle, S.A.; Schoenfeld, D.A. An analysis of induction and adjuvant chemotherapy in the multidisciplinary treatment of squamous-cell carcinoma of the head and neck. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 1987, 5, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Chin, D.; Boyle, G.M.; Porceddu, S.; Theile, D.R.; Parsons, P.G.; Coman, W.B. Head and neck cancer: Past, present and future. Expert Rev. Anticancer Ther. 2006, 6, 1111–1118. [Google Scholar] [CrossRef]
- Bernier, J. A multidisciplinary approach to squamous cell carcinomas of the head and neck: An update. Curr. Opin. Oncol. 2008, 20, 249–255. [Google Scholar] [CrossRef]
- Stell, P.M.; Rawson, N.S. Adjuvant chemotherapy in head and neck cancer. Br. J. Cancer 1990, 61, 779–787. [Google Scholar] [CrossRef] [Green Version]
- Murdoch, D. Standard, and novel cytotoxic and molecular-targeted, therapies for HNSCC: An evidence-based review. Curr. Opin. Oncol. 2007, 19, 216–221. [Google Scholar] [CrossRef]
- Sher, D.J.; Yan, J.; Day, A.; Sumer, B.D.; Pham, N.-L.; Khan, S.; Zhu, H. Comparative effectiveness of primary radiotherapy versus surgery in elderly patients with locally advanced oropharyngeal squamous cell carcinoma. Oral Oncol. 2019, 88, 18–26. [Google Scholar] [CrossRef]
- Tsai, S.Y.; Tsai, M.J. Chick ovalbumin upstream promoter-transcription factors (COUP-TFs): Coming of age. Endocr. Rev. 1997, 18, 229–240. [Google Scholar] [CrossRef] [Green Version]
- Takamoto, N.; You, L.-R.; Moses, K.; Chiang, C.; Zimmer, W.E.; Schwartz, R.J.; DeMayo, F.J.; Tsai, M.-J.; Tsai, S.Y. COUP-TFII is essential for radial and anteroposterior patterning of the stomach. Dev. Camb. Engl. 2005, 132, 2179–2189. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Dufau, M.L. Nuclear orphan receptors regulate transcription of the gene for the human luteinizing hormone receptor. J. Biol. Chem. 2000, 275, 2763–2770. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, X.G.; Park, K.S.; Kaneshige, M.; Bhat, M.K.; Zhu, Q.; Mariash, C.N.; McPhie, P.; Cheng, S.Y. The orphan nuclear receptor Ear-2 is a negative coregulator for thyroid hormone nuclear receptor function. Mol. Cell. Biol. 2000, 20, 2604–2618. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chu, K.; Zingg, H.H. The nuclear orphan receptors COUP-TFII and Ear-2 act as silencers of the human oxytocin gene promoter. J. Mol. Endocrinol. 1997, 19, 163–172. [Google Scholar] [CrossRef]
- Chu, K.; Boutin, J.M.; Breton, C.; Zingg, H.H. Nuclear orphan receptors COUP-TFII and Ear-2: Presence in oxytocin-producing uterine cells and functional interaction with the oxytocin gene promoter. Mol. Cell. Endocrinol. 1998, 137, 145–154. [Google Scholar] [CrossRef]
- Ladias, J.A.; Hadzopoulou-Cladaras, M.; Kardassis, D.; Cardot, P.; Cheng, J.; Zannis, V.; Cladaras, C. Transcriptional regulation of human apolipoprotein genes ApoB, ApoCIII, and ApoAII by members of the steroid hormone receptor superfamily HNF-4, ARP-1, EAR-2, and EAR-3. J. Biol. Chem. 1992, 267, 15849–15860. [Google Scholar]
- Liu, X.; Huang, X.; Sigmund, C.D. Identification of a nuclear orphan receptor (Ear2) as a negative regulator of renin gene transcription. Circ. Res. 2003, 92, 1033–1040. [Google Scholar] [CrossRef] [Green Version]
- Hermann-Kleiter, N.; Baier, G. Orphan nuclear receptor NR2F6 acts as an essential gatekeeper of Th17 CD4+ T cell effector functions. Cell Commun. Signal. CCS 2014, 12, 38. [Google Scholar] [CrossRef] [Green Version]
- Olson, W.J.; Jakic, B.; Labi, V.; Schoeler, K.; Kind, M.; Klepsch, V.; Baier, G.; Hermann-Kleiter, N. Orphan Nuclear Receptor NR2F6 Suppresses T Follicular Helper Cell Accumulation through Regulation of IL-21. Cell Rep. 2019, 28, 2878–2891.e5. [Google Scholar] [CrossRef]
- Ichim, C.V.; Dervović, D.D.; Zúñiga-Pflücker, J.C.; Wells, R.A. The orphan nuclear receptor Ear-2 (Nr2f6) is a novel negative regulator of T cell development. Exp. Hematol. 2014, 42, 46–58. [Google Scholar] [CrossRef]
- Klepsch, V.; Hermann-Kleiter, N.; Do-Dinh, P.; Jakic, B.; Offermann, A.; Efremova, M.; Sopper, S.; Rieder, D.; Krogsdam, A.; Gamerith, G.; et al. Nuclear receptor NR2F6 inhibition potentiates responses to PD-L1/PD-1 cancer immune checkpoint blockade. Nat. Commun. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Hermann-Kleiter, N.; Klepsch, V.; Wallner, S.; Siegmund, K.; Klepsch, S.; Tuzlak, S.; Villunger, A.; Kaminski, S.; Pfeifhofer-Obermair, C.; Gruber, T.; et al. The Nuclear Orphan Receptor NR2F6 Is a Central Checkpoint for Cancer Immune Surveillance. Cell Rep. 2015, 12, 2072–2085. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klepsch, V.; Pommermayr, M.; Humer, D.; Brigo, N.; Hermann-Kleiter, N.; Baier, G. Targeting the orphan nuclear receptor NR2F6 in T cells primes tumors for immune checkpoint therapy. Cell Commun. Signal. CCS 2020, 18, 8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, H.; Zhang, W.; Niu, C.; Lin, C.; Wu, X.; Jian, Y.; Li, Y.; Ye, L.; Dai, Y.; Ouyang, Y.; et al. Nuclear orphan receptor NR2F6 confers cisplatin resistance in epithelial ovarian cancer cells by activating the Notch3 signaling pathway. Int. J. Cancer 2019, 145, 1921–1934. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Long, H.; Zheng, Q.; Bo, X.; Xiao, X.; Li, B. Circular RNA circRHOT1 promotes hepatocellular carcinoma progression by initiation of NR2F6 expression. Mol. Cancer 2019, 18, 119. [Google Scholar] [CrossRef] [PubMed]
- Li, X.-B.; Jiao, S.; Sun, H.; Xue, J.; Zhao, W.-T.; Fan, L.; Wu, G.-H.; Fang, J. The orphan nuclear receptor EAR2 is overexpressed in colorectal cancer and it regulates survivability of colon cancer cells. Cancer Lett. 2011, 309, 137–144. [Google Scholar] [CrossRef]
- Niu, C.; Sun, X.; Zhang, W.; Li, H.; Xu, L.; Li, J.; Xu, B.; Zhang, Y. NR2F6 Expression Correlates with Pelvic Lymph Node Metastasis and Poor Prognosis in Early-Stage Cervical Cancer. Int. J. Mol. Sci. 2016, 17, 1694. [Google Scholar] [CrossRef] [Green Version]
- Ribbat-Idel, J.; Perner, S.; Kuppler, P.; Klapper, L.; Krupar, R.; Watermann, C.; Paulsen, F.-O.; Offermann, A.; Bruchhage, K.-L.; Wollenberg, B.; et al. Immunologic “Cold” Squamous Cell Carcinomas of the Head and Neck are Associated with an Unfavorable Prognosis. Cancers Manuscript in preparation for submission. 2020. [Google Scholar]
- Cohen, E.E.W.; Bell, R.B.; Bifulco, C.B.; Burtness, B.; Gillison, M.L.; Harrington, K.J.; Le, Q.-T.; Lee, N.Y.; Leidner, R.; Lewis, R.L.; et al. The Society for Immunotherapy of Cancer consensus statement on immunotherapy for the treatment of squamous cell carcinoma of the head and neck (HNSCC). J. Immunother. Cancer 2019, 7, 184. [Google Scholar] [CrossRef] [Green Version]
- Chaudhary, S.; Ganguly, K.; Muniyan, S.; Pothuraju, R.; Sayed, Z.; Jones, D.T.; Batra, S.K.; Macha, M.A. Immunometabolic Alterations by HPV Infection: New Dimensions to Head and Neck Cancer Disparity. JNCI J. Natl. Cancer Inst. 2019, 111, 233–244. [Google Scholar] [CrossRef] [Green Version]
- Petersen, J.F.; Timmermans, A.J.; van Dijk, B.A.C.; Overbeek, L.I.H.; Smit, L.A.; Hilgers, F.J.M.; Stuiver, M.M.; van den Brekel, M.W.M. Trends in treatment, incidence and survival of hypopharynx cancer: A 20-year population-based study in the Netherlands. Eur. Arch. Otorhinolaryngol. 2018, 275, 181–189. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saloura, V.; Izumchenko, E.; Zuo, Z.; Bao, R.; Korzinkin, M.; Ozerov, I.; Zhavoronkov, A.; Sidransky, D.; Bedi, A.; Hoque, M.O.; et al. Immune profiles in primary squamous cell carcinoma of the head and neck. Oral Oncol. 2019, 96, 77–88. [Google Scholar] [CrossRef] [PubMed]
- Krupar, R.; Hautmann, M.G.; Pathak, R.R.; Varier, I.; McLaren, C.; Gaag, D.; Hellerbrand, C.; Evert, M.; Laban, S.; Idel, C.; et al. Immunometabolic Determinants of Chemoradiotherapy Response and Survival in Head and Neck Squamous Cell Carcinoma. Am. J. Pathol. 2018, 188, 72–83. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krupar, R.; Watermann, C.; Idel, C.; Ribbat-Idel, J.; Offermann, A.; Pasternack, H.; Kirfel, J.; Sikora, A.G.; Perner, S. In silico analysis reveals EP300 as a panCancer inhibitor of anti-tumor immune response via metabolic modulation. Sci. Rep. 2020, 10, 9389. [Google Scholar] [CrossRef]
- Piccirillo, J.F. Importance of Comorbidity in Head and Neck Cancer. Laryngoscope 2000, 110, 593–602. [Google Scholar] [CrossRef]
- Massa, S.T.; Osazuwa-Peters, N.; Christopher, K.M.; Arnold, L.D.; Schootman, M.; Walker, R.J.; Varvares, M.A. Competing causes of death in the head and neck cancer population. Oral Oncol. 2017, 65, 8–15. [Google Scholar] [CrossRef]
- Lydiatt, W.M.; Patel, S.G.; O’Sullivan, B.; Brandwein, M.S.; Ridge, J.A.; Migliacci, J.C.; Loomis, A.M.; Shah, J.P. Head and Neck cancers-major changes in the American Joint Committee on cancer eighth edition cancer staging manual. CA. Cancer J. Clin. 2017, 67, 122–137. [Google Scholar] [CrossRef]
- Idel, C.; Ribbat-Idel, J.; Kuppler, P.; Krupar, R.; Offermann, A.; Vogel, W.; Rades, D.; Kirfel, J.; Wollenberg, B.; Perner, S. EVI1 as a Marker for Lymph Node Metastasis in HNSCC. Int. J. Mol. Sci. 2020, 21, 854. [Google Scholar] [CrossRef] [Green Version]
- Scheble, V.J.; Braun, M.; Beroukhim, R.; Mermel, C.H.; Ruiz, C.; Wilbertz, T.; Stiedl, A.-C.; Petersen, K.; Reischl, M.; Kuefer, R.; et al. ERG rearrangement is specific to prostate cancer and does not occur in any other common tumor. Mod. Pathol. 2010, 23, 1061–1067. [Google Scholar] [CrossRef] [Green Version]
- Braun, M.; Goltz, D.; Shaikhibrahim, Z.; Vogel, W.; Böhm, D.; Scheble, V.; Sotlar, K.; Fend, F.; Tan, S.-H.; Dobi, A.; et al. ERG protein expression and genomic rearrangement status in primary and metastatic prostate cancer--a comparative study of two monoclonal antibodies. Prostate Cancer Prostatic Dis. 2012, 15, 165–169. [Google Scholar] [CrossRef] [Green Version]
- Queisser, A.; Hagedorn, S.; Wang, H.; Schaefer, T.; Konantz, M.; Alavi, S.; Deng, M.; Vogel, W.; von Mässenhausen, A.; Kristiansen, G.; et al. Ecotropic viral integration site 1, a novel oncogene in prostate cancer. Oncogene 2017, 36, 1573–1584. [Google Scholar] [CrossRef] [PubMed]
- Braun, M.; Kirsten, R.; Rupp, N.J.; Moch, H.; Fend, F.; Wernert, N.; Kristiansen, G.; Perner, S. Quantification of protein expression in cells and cellular subcompartments on immunohistochemical sections using a computer supported image analysis system. Histol. Histopathol. 2013, 28, 605–610. [Google Scholar] [CrossRef] [PubMed]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Klapper, L.; Ribbat-Idel, J.; Kuppler, P.; Paulsen, F.-O.; Bruchhage, K.-L.; Rades, D.; Offermann, A.; Kirfel, J.; Wollenberg, B.; Idel, C.; et al. NR2F6 as a Prognostic Biomarker in HNSCC. Int. J. Mol. Sci. 2020, 21, 5527. https://doi.org/10.3390/ijms21155527
Klapper L, Ribbat-Idel J, Kuppler P, Paulsen F-O, Bruchhage K-L, Rades D, Offermann A, Kirfel J, Wollenberg B, Idel C, et al. NR2F6 as a Prognostic Biomarker in HNSCC. International Journal of Molecular Sciences. 2020; 21(15):5527. https://doi.org/10.3390/ijms21155527
Chicago/Turabian StyleKlapper, Luise, Julika Ribbat-Idel, Patrick Kuppler, Finn-Ole Paulsen, Karl-Ludwig Bruchhage, Dirk Rades, Anne Offermann, Jutta Kirfel, Barbara Wollenberg, Christian Idel, and et al. 2020. "NR2F6 as a Prognostic Biomarker in HNSCC" International Journal of Molecular Sciences 21, no. 15: 5527. https://doi.org/10.3390/ijms21155527




