Insights into the Mn2+ Binding Site in the Agmatinase-Like Protein (ALP): A Critical Enzyme for the Regulation of Agmatine Levels in Mammals
Abstract
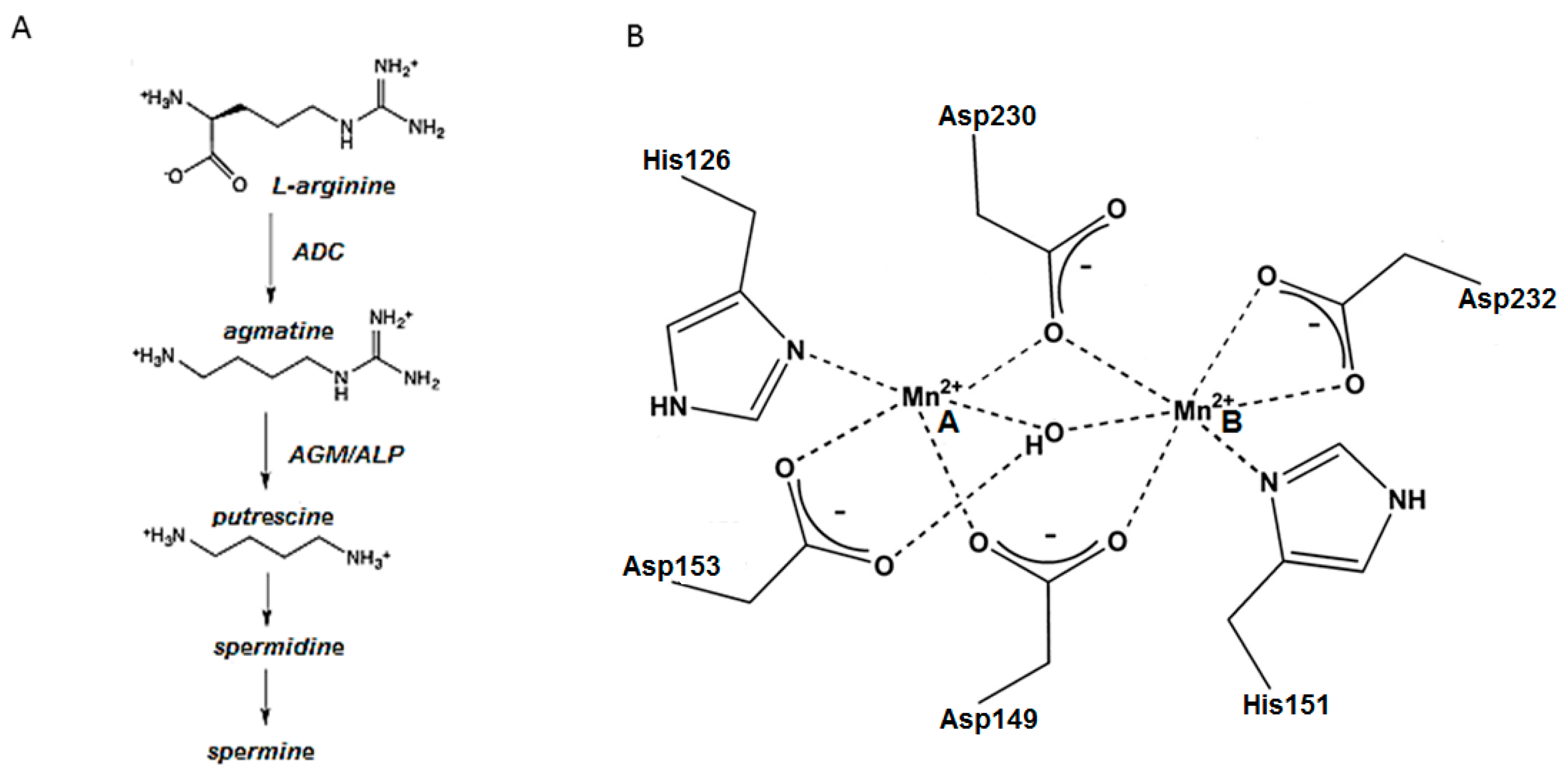
1. Introduction
2. Results and Discussion
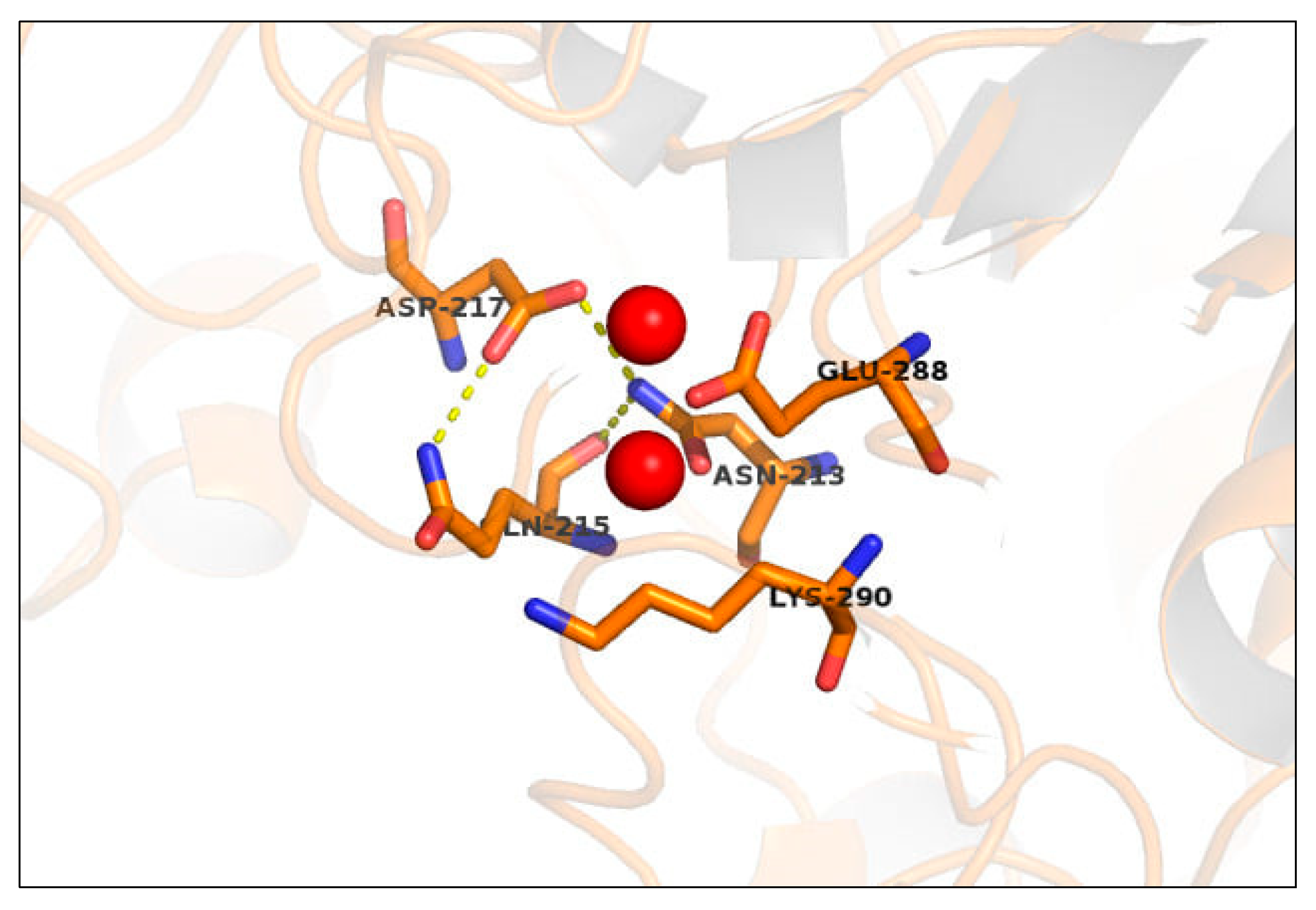
2.1. Manganese Binding Site in ALP
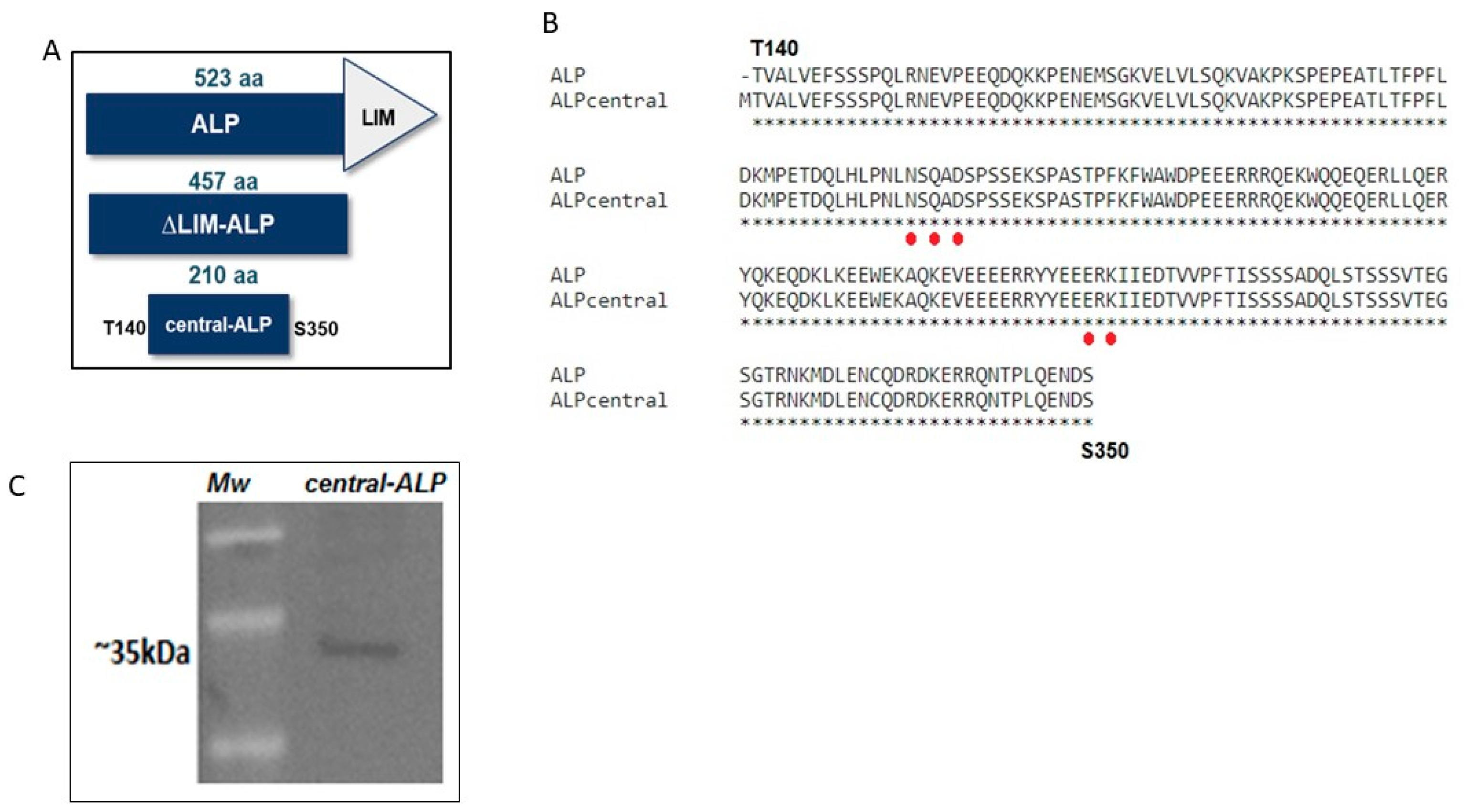
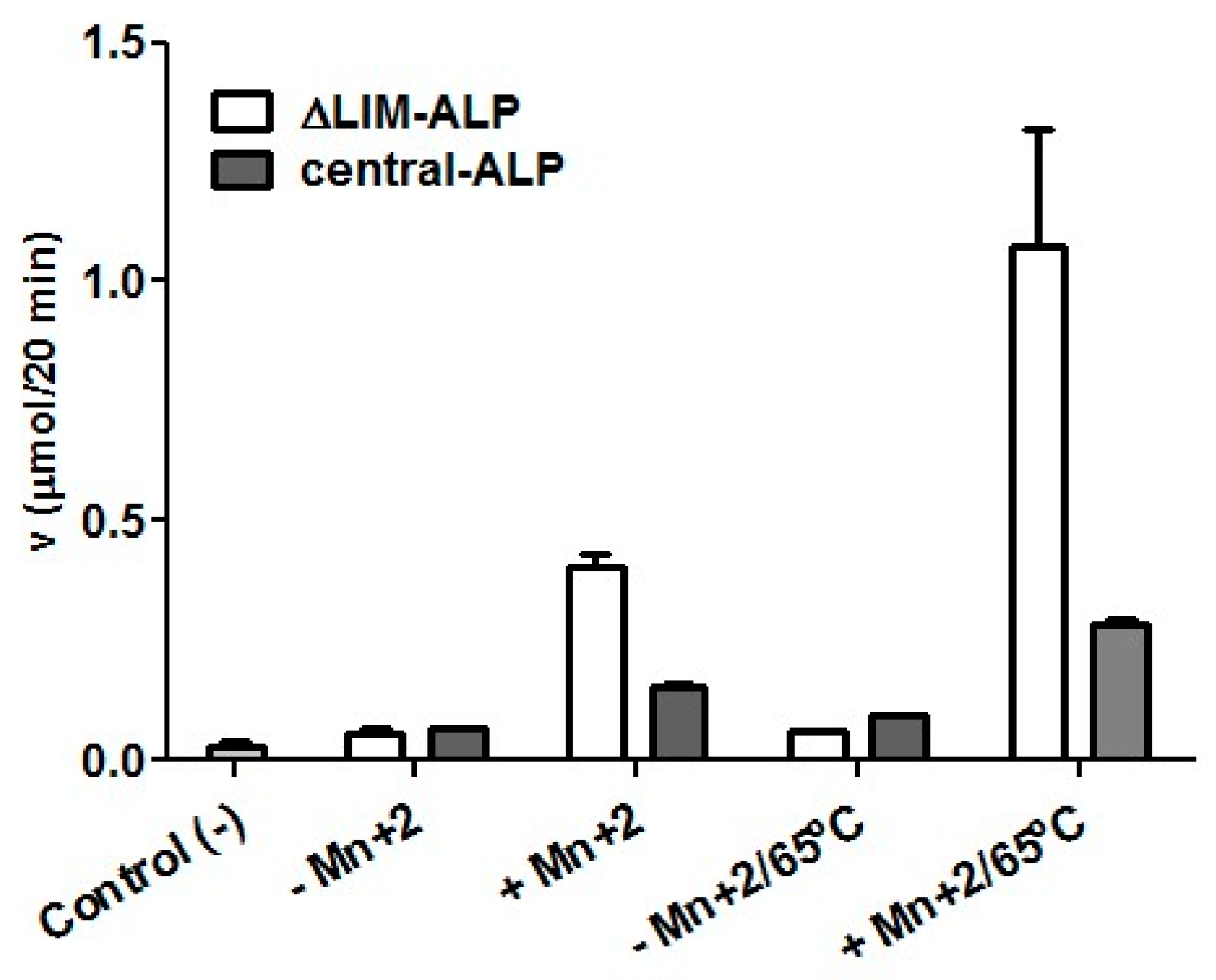
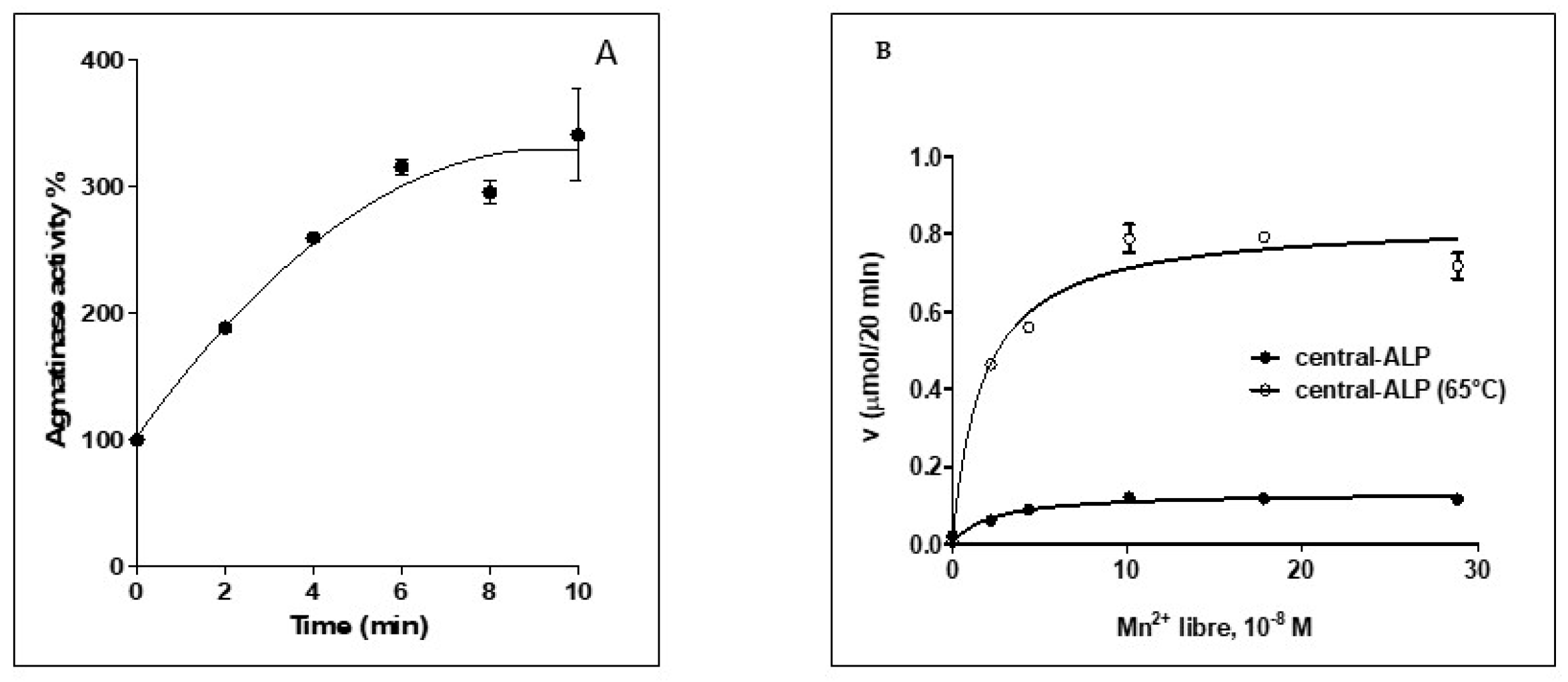
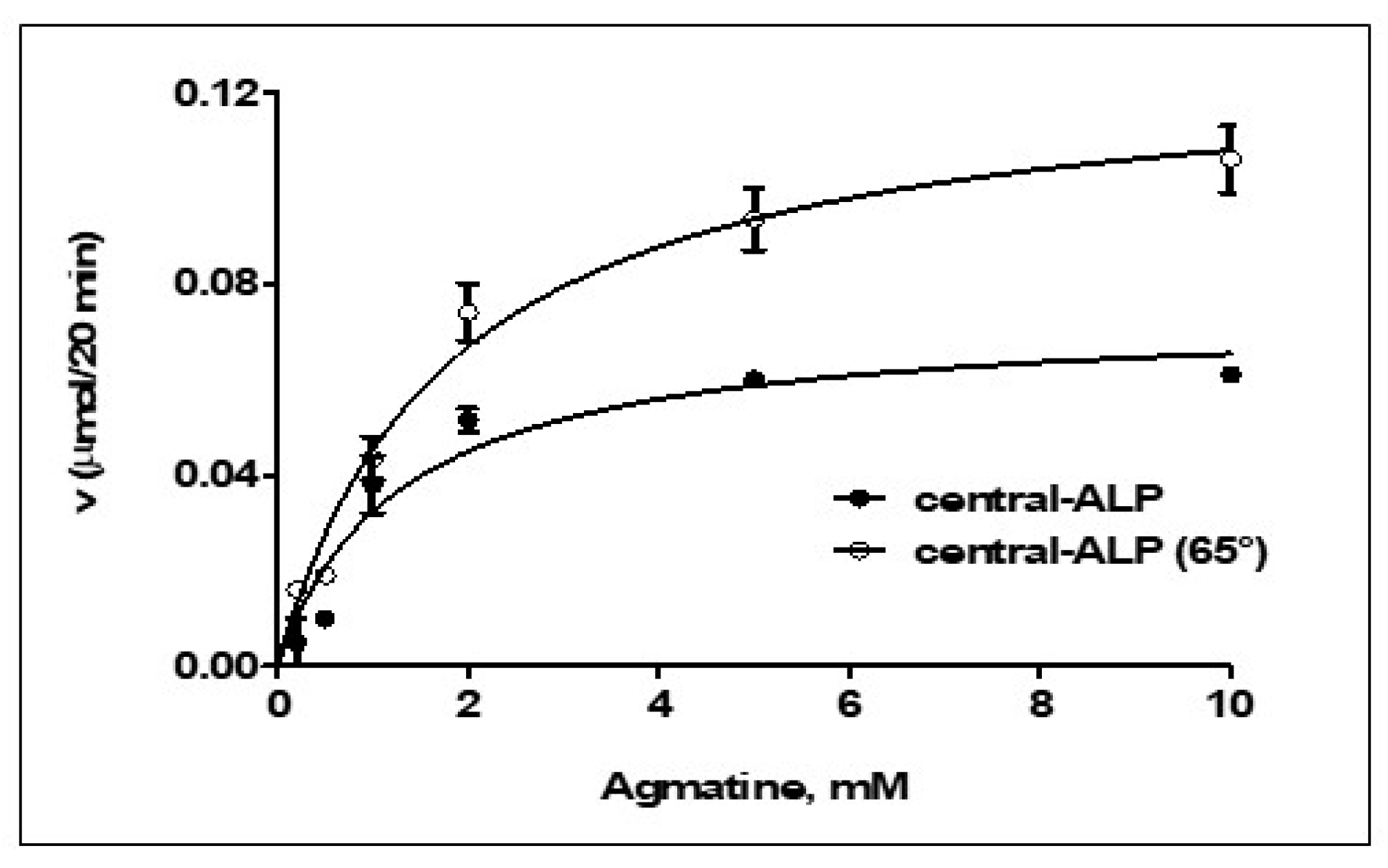
2.2. Expression and Characterization of Central-ALP
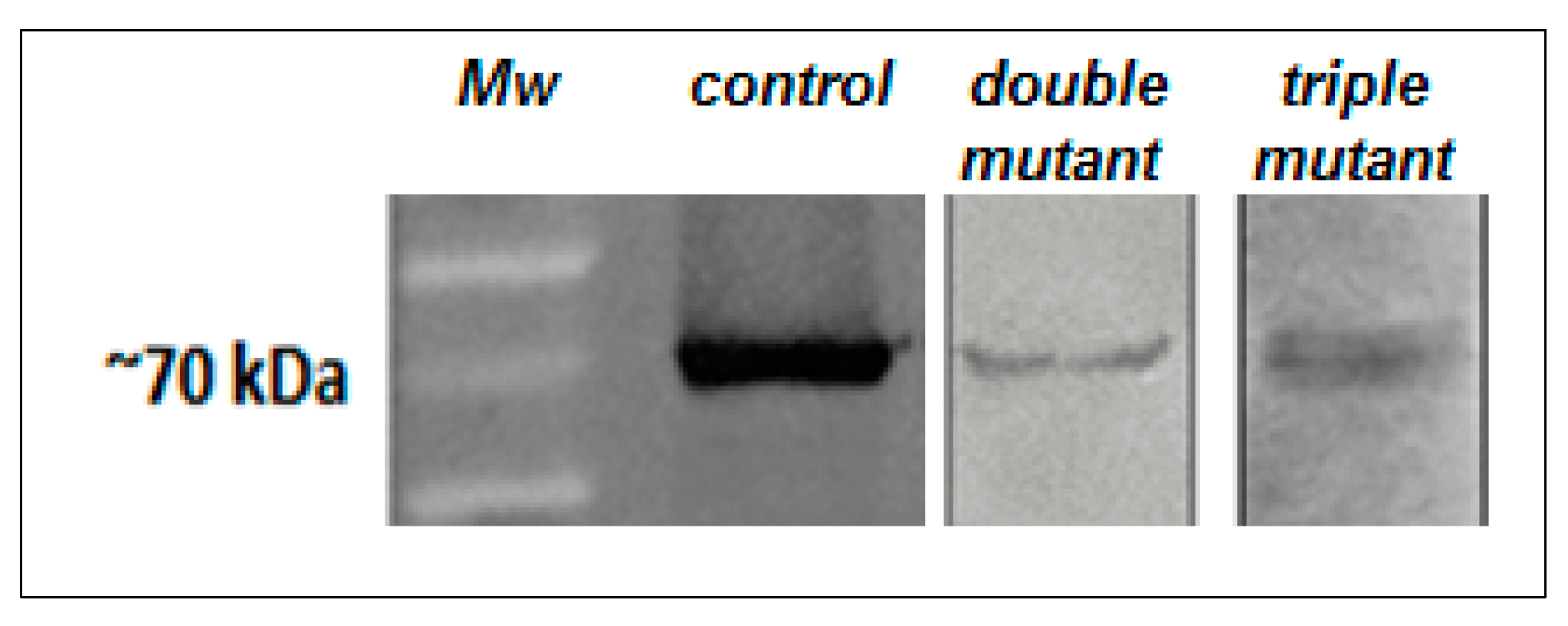
2.3. Site-Directed Mutagenesis of Putative Mn2+ Ligands
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. Molecular Modeling
4.3. Enzyme Preparations
4.4. ALP Activity Determination
4.5. Enzyme–Metal Interactions Analysis
4.6. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chan, S.L.F. Clonidine-displacing substance and its putative role in control of insulin secretion: A minireview. Gen. Pharmacol. Vasc. Syst. 1998, 31, 525–529. [Google Scholar] [CrossRef]
- Su, C.-H.; Liu, I.-M.; Chung, H.-H.; Cheng, J.-T. Activation of I2-imidazoline receptors by agmatine improved insulin sensitivity through two mechanisms in type-2 diabetic rats. Neurosci. Lett. 2009, 457, 125–128. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-P.; Chen, W.; Wu, H.-T.; Lin, K.-C.; Cheng, J.-T. Metformin can activate imidazoline I-2 Receptors to lower plasma glucose in type 1-like diabetic rats. Horm. Metab. Res. 2011, 43, 26–30. [Google Scholar] [CrossRef] [PubMed]
- Penner, B.; Smyth, D.D. Natriuresis following central and peripheral administration of agmatine in the rat. Pharmacology 1996, 53, 160–169. [Google Scholar] [CrossRef] [PubMed]
- Satriano, J.; Cunard, R.; Peterson, O.W.; Dousa, T.; Gabbai, F.B.; Blantz, R.C. Effects on kidney filtration rate by agmatine requires activation of ryanodine channels for nitric oxide generation. Am. J. Physiol. Ren. Physiol. 2008, 294, F795–F800. [Google Scholar] [CrossRef] [PubMed]
- Qiu, W.-W.; Zheng, R.-Y. Neuroprotective effects of receptor imidazoline 2 and its endogenous ligand agmatine. Neurosci. Bull. 2006, 22, 187–191. [Google Scholar] [PubMed]
- Mun, C.H.; Lee, W.T.; Park, K.A.; Lee, J.E. Regulation of endothelial nitric oxide synthase by agmatine after transient global cerebral ischemia in rat brain. Anat. Cell Biol. 2010, 43, 230–240. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.T.; Hong, S.; Yoon, S.H.; Kim, J.H.; Park, K.A.; Seong, G.J.; Lee, J.E. Neuroprotective effects of agmatine on oxygen-glucose deprived primary-cultured astrocytes and nuclear translocation of nuclear factor-kappa B. Brain Res. 2009, 1281, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Wu, N.; Su, R.; Chen, Y.; Lu, X.; Liu, Y.; Li, J. Imidazoline receptor antisera-selected/Nischarin regulates the effect of agmatine on the development of morphine dependence. Addict. Biol. 2012, 17, 392–408. [Google Scholar] [CrossRef] [PubMed]
- Taksande, B.G.; Kotagale, N.R.; Patel, M.R.; Shelkar, G.P.; Ugale, R.R.; Chopde, C.T. Agmatine, an endogenous imidazoline receptor ligand modulates ethanol anxiolysis and withdrawal anxiety in rats. Eur. J. Pharmacol. 2010, 637, 89–101. [Google Scholar] [CrossRef] [PubMed]
- Satriano, J.; Matsufuji, S.; Murakami, Y.; Lortie, M.J.; Schwartz, D.; Kelly, C.J.; Hayashi, S.; Blantz, R.C. Agmatine suppresses proliferation by frameshift induction of antizyme and attenuation of cellular polyamine levels. J. Biol. Chem. 1998, 273, 15313–15316. [Google Scholar] [CrossRef] [PubMed]
- Vargiu, C.; Cabella, C.; Belliardo, S.; Cravanzola, C.; Grillo, M.A.; Colombatto, S. Agmatine modulates polyamine content in hepatocytes by inducing spermidine/spermine acetyltransferase. Eur. J. Biochem. 1999, 259, 933–938. [Google Scholar] [CrossRef] [PubMed]
- Piletz, J.E.; Aricioglu, F.; Cheng, J.-T.; Fairbanks, C.A.; Gilad, V.H.; Haenisch, B.; Halaris, A.; Hong, S.; Lee, J.E.; Li, J.; et al. Agmatine: Clinical applications after 100 years in translation. Drug Discov. Today 2013, 18, 880–893. [Google Scholar] [CrossRef] [PubMed]
- Wu, N.; Su, R.-B.; Li, J. Agmatine and imidazoline receptors: Their role in opioid analgesia, tolerance and dependence. Cell. Mol. Neurobiol. 2008, 28, 629–641. [Google Scholar] [CrossRef] [PubMed]
- Dixit, M.P.; Thakre, P.P.; Pannase, A.S.; Aglawe, M.M.; Taksande, B.G.; Kotagale, N.R. Imidazoline binding sites mediates anticompulsive-like effect of agmatine in marble-burying behavior in mice. Eur. J. Pharmacol. 2014, 732, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Barua, S.; Kim, J.Y.; Kim, J.Y.; Kim, J.H.; Lee, J.E. Therapeutic Effect of Agmatine on Neurological Disease: Focus on Ion Channels and Receptors. Neurochem. Res. 2019, 44, 735–750. [Google Scholar] [CrossRef] [PubMed]
- Moretti, M.; Matheus, F.C.; de Oliveira, P.A.; Neis, V.B.; Ben, J.; Walz, R.; Rodrigues, A.L.S.; Prediger, R.D. Role of agmatine in neurodegenerative diseases and epilepsy. Front. Biosci. 2014, 6, 341–359. [Google Scholar] [CrossRef]
- Uribe, E.; Reyes, M.-B.; Martínez, I.; Mella, K.; Salas, M.; Tarifeño-Saldivia, E.; López, V.; García-Robles, M.; Martínez-Oyanedel, J.; Figueroa, M.; et al. Functional analysis of the Mn2+ requirement in the catalysis of ureohydrolases arginase and agmatinase—A historical perspective. J. Inorg. Biochem. 2020, 202, 110812. [Google Scholar] [CrossRef]
- Scolnick, L.R.; Kanyo, Z.F.; Cavalli, R.C.; Ash, D.E.; Christianson, D.W. Altering the binuclear manganese cluster of arginase diminishes thermostability and catalytic function. Biochemistry 1997, 36, 10558–10565. [Google Scholar] [CrossRef] [PubMed]
- Ash, D.E. Structure and function of arginases. J. Nutr. 2004, 134, 2760S–2764S. [Google Scholar] [CrossRef] [PubMed]
- Uribe, E.; Salas, M.; Enríquez, S.; Orellana, M.S.; Carvajal, N. Cloning and functional expression of a rodent brain cDNA encoding a novel protein with agmatinase activity, but not belonging to the arginase family. Arch. Biochem. Biophys. 2007, 461, 146–150. [Google Scholar] [CrossRef] [PubMed]
- Castro, V.; Fuentealba, P.; Henríquez, A.; Vallejos, A.; Benítez, J.; Lobos, M.; Díaz, B.; Carvajal, N.; Uribe, E. Evidence for an inhibitory LIM domain in a rat brain agmatinase-like protein. Arch. Biochem. Biophys. 2011, 512, 107–110. [Google Scholar] [CrossRef] [PubMed]
- Mella, C.; Martínez, F.; de Los Angeles García, M.; Nualart, F.; Castro, V.; Bustos, P.; Carvajal, N.; Uribe, E. Expression and localization of an agmatinase-like protein in the rat brain. Histochem. Cell Biol. 2010, 134, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Romero, N.; Benítez, J.; Garcia, D.; González, A.; Bennun, L.; García-Robles, M.A.; López, V.; Wilson, L.A.; Schenk, G.; Carvajal, N.; et al. Mammalian agmatinases constitute unusual members in the family of Mn2+-dependent ureahydrolases. J. Inorg. Biochem. 2017, 166, 122–125. [Google Scholar] [CrossRef] [PubMed]
- Iyer, R.K.; Kim, H.K.; Tsoa, R.W.; Grody, W.W.; Cederbaum, S.D. Cloning and characterization of human agmatinase. Mol. Genet. Metab. 2002, 75, 209–218. [Google Scholar] [CrossRef] [PubMed]
- García, D.; Ordenes, P.; Benítez, J.; González, A.; García-Robles, M.A.; López, V.; Carvajal, N.; Uribe, E. Cloning of two LIMCH1 isoforms: Characterization of their distribution in rat brain and their agmatinase activity. Histochem. Cell Biol. 2016, 145, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Feuerstein, R.; Wang, X.; Song, D.; Cooke, N.E.; Liebhaber, S.A. The LIM/double zinc-finger motif functions as a protein dimerization domain. Proc. Natl. Acad. Sci. USA 1994, 91, 10655–10659. [Google Scholar] [CrossRef] [PubMed]
- Kadrmas, J.L.; Beckerle, M.C. The LIM domain: From the cytoskeleton to the nucleus. Nat. Rev. Mol. Cell Biol. 2004, 5, 920–931. [Google Scholar] [CrossRef] [PubMed]
- Quiñones, M.; Cofre, J.; Benítez, J.; García, D.; Romero, N.; González, A.; Carvajal, N.; García, M.; López, V.; Schenk, G.; et al. Insight on the interaction of an agmatinase-like protein with Mn2+ activator ions. J. Inorg. Biochem. 2015, 145, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Carvajal, N.; López, V.; Salas, M.; Uribe, E.; Herrera, P.; Cerpa, J. Manganese is essential for catalytic activity ofescherichia coliagmatinase. Biochem. Biophys. Res. Commun. 1999, 258, 808–811. [Google Scholar] [CrossRef] [PubMed]
- Carvajal, N.; Orellana, M.S.; Salas, M.; Enríquez, P.; Alarcón, R.; Uribe, E.; López, V. Kinetic studies and site-directed mutagenesis of Escherichia coli agmatinase. A role for Glu274 in binding and correct positioning of the substrate guanidinium group. Arch. Biochem. Biophys. 2004, 430, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Cama, E.; Colleluori, D.M.; Emig, F.A.; Shin, H.; Kim, S.W.; Kim, N.N.; Traish, A.M.; Ash, D.E.; Christianson, D.W. Human arginase II: Crystal structure and physiological role in male and female sexual arousal. Biochemistry 2003, 42, 8445–8451. [Google Scholar] [CrossRef] [PubMed]
- Alarcón, R.; Orellana, M.S.; Neira, B.; Uribe, E.; García, J.R.; Carvajal, N. Mutational analysis of substrate recognition by human arginase type I—Agmatinase activity of the N130D variant. Febs J. 2006, 273, 5625–5631. [Google Scholar] [CrossRef] [PubMed]
- Dutta, A.; Mazumder, M.; Alam, M.; Gourinath, S.; Sau, A.K. Metal-induced change in catalytic loop positioning in Helicobacter pylori arginase alters catalytic function. Biochem. J. 2019, 476, 3595–3614. [Google Scholar] [CrossRef] [PubMed]
- Mitić, N.; Miraula, M.; Selleck, C.; Hadler, K.S.; Uribe, E.; Pedroso, M.M.; Schenk, G. Chapter three—Catalytic mechanisms of metallohydrolases containing two metal ions. In Advances in Protein Chemistry and Structural Biology; Metal-Containing Enzymes; Christov, C.Z., Ed.; Academic Press: New York, NY, USA, 2014; Volume 97, pp. 49–81. [Google Scholar]
- Cofre, J.; Montes, P.; Vallejos, A.; Benítez, J.; García, D.; Martínez-Oyanedel, J.; Carvajal, N.; Uribe, E. Further insight into the inhibitory action of a LIM/double zinc-finger motif of an agmatinase-like protein. J. Inorg. Biochem. 2014, 132, 92–95. [Google Scholar] [CrossRef] [PubMed]
- Harding, M.M. The architecture of metal coordination groups in proteins. Acta Crystallogr. D Biol Crystallogr. 2004, 60, 849–859. [Google Scholar] [CrossRef] [PubMed]
- Khrustaleva, T.A. Secondary structure preferences of Mn2+ binding sites in bacterial proteins. Adv. Bioinform. 2014, 2014, 501841. [Google Scholar] [CrossRef] [PubMed]
- Wei, T.; Quareshy, M.; Zhang, Y.; Scanlan, D.; Chen, Y. Manganese Is essential for PlcP metallophosphoesterase activity involved in lipid remodeling in abundant marine heterotrophic bacteria. Appl. Environ. Microbiol. 2018, 84, 1109–1118. [Google Scholar] [CrossRef] [PubMed]
- Phelan, E.K.; Miraula, M.; Selleck, C.; Ollis, D.L.; Schenk, G.; Mitić, N. Metallo-β-lactamases: A major threat to human health. Am. J. Mol. Biol. 2014, 4, 89–104. [Google Scholar] [CrossRef]
- Harris, T.; Wu, G.; Massiah, M.; Mildvan, A. Mutational, kinetic, and NMR studies of the roles of conserved glutamate residues and of lysine-39 in the mechanism of the MutT pyrophosphohydrolase. Biochemistry 2000, 39, 1655–1674. [Google Scholar] [CrossRef] [PubMed]
- Dudev, T.; Lin, Y.; Dudev, M.; Lim, C. First−second shell interactions in metal binding sites in proteins: A PDB survey and DFT/CDM calculations. J. Am. Chem. Soc. 2003, 125, 3168–3180. [Google Scholar] [CrossRef] [PubMed]
- Orellana, M.S.; López, V.; Uribe, E.; Fuentes, M.; Salas, M.; Carvajal, N. Insights into the interaction of human liver arginase with tightly and weakly bound manganese ions by chemical modification and site-directed mutagenesis studies. Arch. Biochem Biophys 2002, 403, 155–159. [Google Scholar] [CrossRef]
- Salas, M.; López, V.; Uribe, E.; Carvajal, N. Studies on the interaction of Escherichia coli agmatinase with manganese ions: Structural and kinetic studies of the H126N and H151N variants. J. Inorg. Biochem. 2004, 98, 1032–1036. [Google Scholar] [CrossRef] [PubMed]
- Benítez, J.; García, D.; Romero, N.; González, A.; Martínez-Oyanedel, J.; Figueroa, M.; Salas, M.; López, V.; García-Robles, M.; Dodd, P.R.; et al. Metabolic strategies for the degradation of the neuromodulator agmatine in mammals. Metabolism 2018, 81, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Canutescu, A.A.; Shelenkov, A.A.; Dunbrack, R.L. A graph-theory algorithm for rapid protein side-chain prediction. Protein Sci. 2003, 12, 2001–2014. [Google Scholar] [CrossRef] [PubMed]
| Km (mM) | kcat (s−¹) | Kact Mn2+ (M) | |
|---|---|---|---|
| ALP-wt | 3.0 ± 0.20 | 0.9 ± 0.2 | 1.88 × 10−8 |
| ∆LIM-ALP | 1.2 ± 0.04 | 10 ± 1 | 3.6 × 10−8 |
| ∆LIM-ALP/D217A | No activity | No activity | No activity |
| ∆LIM-ALP/E287A/K289A | No activity | No activity | No activity |
| ∆LIM-ALP/N213A/Q215A/D217A | No activity | No activity | No activity |
| Central-ALP | 1.2 ± 0.37 | 0.4 ± 0.5 | 1.7 × 10−8 |
| Central-ALP (65 °C) | 1.2 ± 0.37 | 0.8 ± 0.4 | 2.2 × 10−8 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reyes, M.-B.; Martínez-Oyanedel, J.; Navarrete, C.; Mardones, E.; Martínez, I.; Salas, M.; López, V.; García-Robles, M.; Tarifeño-Saldivia, E.; Figueroa, M.; et al. Insights into the Mn2+ Binding Site in the Agmatinase-Like Protein (ALP): A Critical Enzyme for the Regulation of Agmatine Levels in Mammals. Int. J. Mol. Sci. 2020, 21, 4132. https://doi.org/10.3390/ijms21114132
Reyes M-B, Martínez-Oyanedel J, Navarrete C, Mardones E, Martínez I, Salas M, López V, García-Robles M, Tarifeño-Saldivia E, Figueroa M, et al. Insights into the Mn2+ Binding Site in the Agmatinase-Like Protein (ALP): A Critical Enzyme for the Regulation of Agmatine Levels in Mammals. International Journal of Molecular Sciences. 2020; 21(11):4132. https://doi.org/10.3390/ijms21114132
Chicago/Turabian StyleReyes, María-Belen, José Martínez-Oyanedel, Camila Navarrete, Erika Mardones, Ignacio Martínez, Mónica Salas, Vasthi López, María García-Robles, Estefania Tarifeño-Saldivia, Maximiliano Figueroa, and et al. 2020. "Insights into the Mn2+ Binding Site in the Agmatinase-Like Protein (ALP): A Critical Enzyme for the Regulation of Agmatine Levels in Mammals" International Journal of Molecular Sciences 21, no. 11: 4132. https://doi.org/10.3390/ijms21114132
APA StyleReyes, M.-B., Martínez-Oyanedel, J., Navarrete, C., Mardones, E., Martínez, I., Salas, M., López, V., García-Robles, M., Tarifeño-Saldivia, E., Figueroa, M., García, D., & Uribe, E. (2020). Insights into the Mn2+ Binding Site in the Agmatinase-Like Protein (ALP): A Critical Enzyme for the Regulation of Agmatine Levels in Mammals. International Journal of Molecular Sciences, 21(11), 4132. https://doi.org/10.3390/ijms21114132





