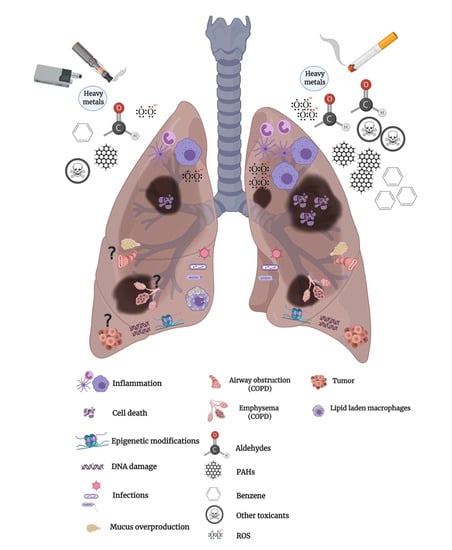
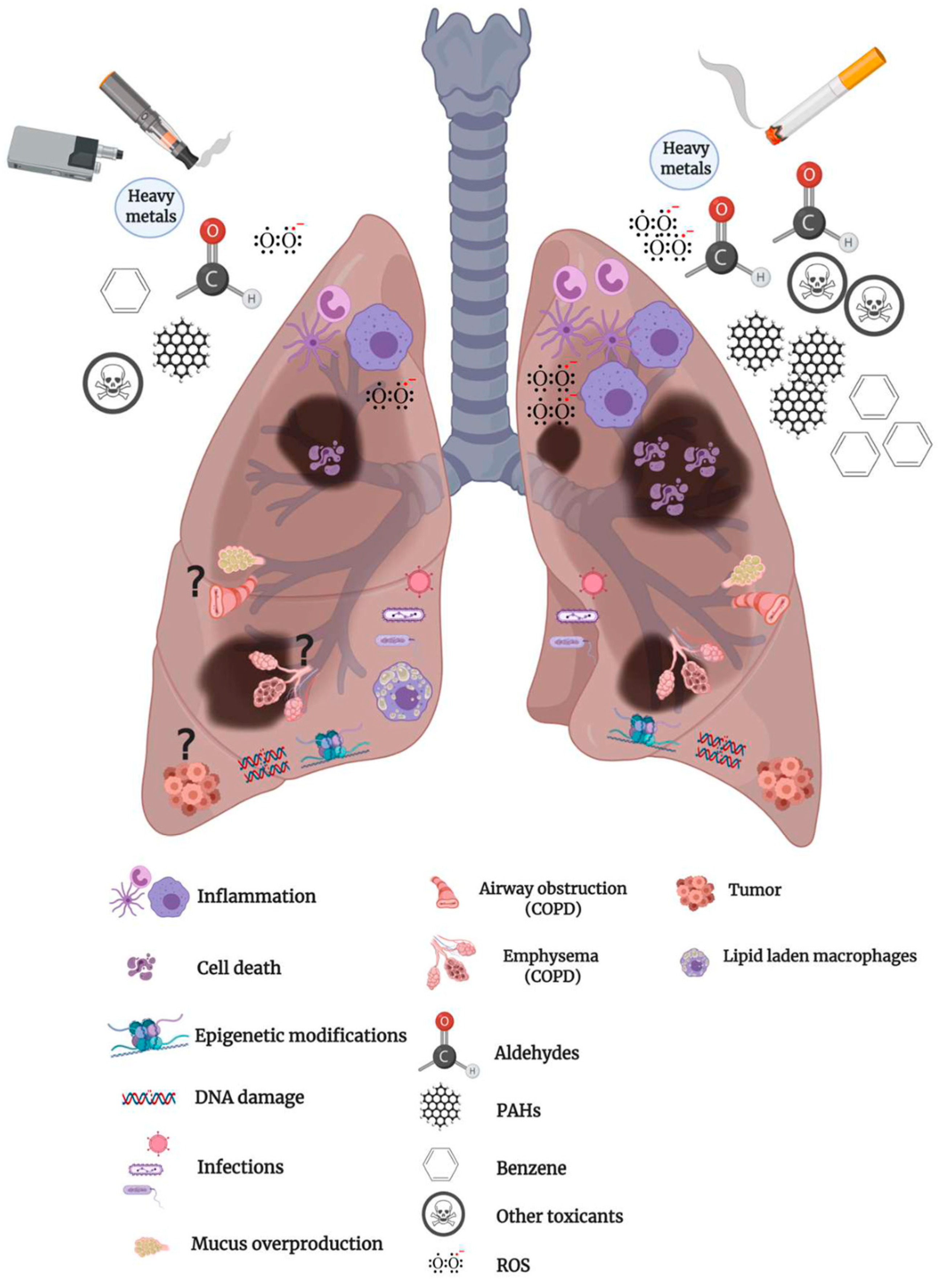
Inhalation Toxicology of Vaping Products and Implications for Pulmonary Health
Abstract
1. Introduction
2. Historical Context of Cigarette Use and Evidence for Disease Induction
3. E-Cigarette Use Patterns and Their Utility as a Smoking Cessation Aid
4. E-Cigarette Components
5. E-liquids
5.1. Solvents
5.2. Flavorings
5.3. Nicotine
5.4. Cannabinoids
6. E-Cigarette Toxicology
6.1. Ultra Fine Particles (UFP)
6.2. Aldehydes
6.3. PAHs
6.4. Metals
6.5. Other Toxicants
7. Cellular Alterations from E-cigarette Exposure
7.1. Inflammation
7.2. Oxidative Stress
7.3. DNA Damage
7.4. Host Defense
7.5. Epigenetic Modifications
8. Clinical Impact of E-Cigarette Use on the Respiratory System
8.1. Respiratory Symptoms
8.2. Airway Mechanics (Lung Function)
8.3. Acute Pulmonary Disease
8.3.1. Primary Spontaneous Pneumothorax (PSP)
8.3.2. Acute Eosinophilic Pneumonia
8.3.3. Bronchiolitis Obliterans
8.3.4. Vaping-Associated Lung Injury (EVALI)
Could EVALI Be an Exogenous Lipoid Pneumonia?
The Role of Vitamin E Acetate
9. Chronic Pulmonary Disease
9.1. COPD
9.2. Asthma
10. Limitations
11. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| EVALI | vaping product-associated lung illness |
| COPD | chronic obstructive pulmonary disease |
| PG | propylene glycol |
| VG | vegetable glycerin |
| BAL | bronchoalveolar lavage |
| THC | tetrahydrocannabinol |
| CBD | cannabidiol |
| PM | particulate matter |
| GRAS | generally recognized as safe |
| PEG | polyethylene glycol |
| MCT | medium chain triglycerides |
| nAChRs | nicotinic acetylcholine receptors |
| TSNAs | tobacco-specific nitrosamines |
| PAHs | polycyclic aromatic hydrocarbons |
| UFP | ultrafine particles |
| miRNA | micro RNA |
| AhR | aryl hydrocarbon receptor |
| XMEs | xenobiotic metabolizing enzymes |
| VOCs | volatile organic compounds |
| FDA | food and drug administration |
| FeNO | fraction of exhaled nitric oxide |
| CRP | C-reactive protein |
| NO | nitric oxide |
| NK | natural killer cells |
| PPB | parts per billion |
| ROS | reactive oxygen species |
| H2O2 | hydrogen peroxide |
| BER | base excision repair |
| NER | nucleotide excision repair |
| CD28 | cluster of differentiation 28 |
| FEV1 | fraction of expired volume in 1 s |
| FVC | forced vital capacity |
| PEF | peak expiratory flow |
| OS | impulse oscillometry system |
| PSP | primary spontaneous pneumothorax |
| AHR | airway hyperresponsiveness |
References
- Hajek, P.; Phillips-Waller, A.; Przulj, D.; Pesola, F.; Myers Smith, K.; Bisal, N.; Li, J.; Parrott, S.; Sasieni, P.; Dawkins, L.; et al. A Randomized Trial of E-Cigarettes versus Nicotine-Replacement Therapy. N. Engl. J. Med. 2019, 380, 629–637. [Google Scholar] [CrossRef] [PubMed]
- Civiletto, C.W.; Aslam, S.; Hutchison, J. Electronic Delivery (Vaping) of Cannabis and Nicotine. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2019. [Google Scholar]
- Hammond, D.; Reid, J.L.; Rynard, V.L.; Fong, G.T.; Cummings, K.M.; McNeill, A.; Hitchman, S.; Thrasher, J.F.; Goniewicz, M.L.; Bansal-Travers, M.; et al. Prevalence of vaping and smoking among adolescents in Canada, England, and the United States: repeat national cross sectional surveys. BMJ 2019, 365, l2219. [Google Scholar] [CrossRef] [PubMed]
- Carlos, W.G.; Crotty Alexander, L.E.; Gross, J.E.; Dela Cruz, C.S.; Keller, J.M.; Pasnick, S.; Jamil, S. ATS Health Alert-Vaping-associated Pulmonary Illness (VAPI). Am. J. Respir. Crit. Care Med. 2019, 200, P15–P16. [Google Scholar] [CrossRef] [PubMed]
- Perrine, C.G.; Pickens, C.M.; Boehmer, T.K.; King, B.A.; Jones, C.M.; DeSisto, C.L.; Duca, L.M.; Lekiachvili, A.; Kenemer, B.; Shamout, M.; et al. Characteristics of a Multistate Outbreak of Lung Injury Associated with E-cigarette Use, or Vaping-United States, 2019. MMWR Morb. Mortal. Wkly. Rep. 2019, 68, 860–864. [Google Scholar] [CrossRef]
- Ellington, S.; Salvatore, P.P.; Ko, J.; Danielson, M.; Kim, L.; Cyrus, A.; Wallace, M.; Board, A.; Krishnasamy, V.; King, B.A.; et al. Update: Product, Substance-Use, and Demographic Characteristics of Hospitalized Patients in a Nationwide Outbreak of E-cigarette, or Vaping, Product Use-Associated Lung Injury-United States, August 2019-January 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 44–49. [Google Scholar] [CrossRef]
- Schier, J.G.; Meiman, J.G.; Layden, J.; Mikosz, C.A.; VanFrank, B.; King, B.A.; Salvatore, P.P.; Weissman, D.N.; Thomas, J.; Melstrom, P.C.; et al. Severe Pulmonary Disease Associated with Electronic-Cigarette-Product Use-Interim Guidance. MMWR Morb. Mortal. Wkly. Rep. 2019, 68, 787–790. [Google Scholar] [CrossRef]
- Butt, Y.M.; Smith, M.L.; Tazelaar, H.D.; Vaszar, L.T.; Swanson, K.L.; Cecchini, M.J.; Boland, J.M.; Bois, M.C.; Boyum, J.H.; Froemming, A.T.; et al. Pathology of Vaping-Associated Lung Injury. N. Engl. J. Med. 2019, 381, 1780–1781. [Google Scholar] [CrossRef]
- Bernhard, D. Cigarette Smoke Toxicity: Linking Individual Chemicals to Human Diseases; John Wiley & Sons: Hoboken, NJ, USA, 2011. [Google Scholar]
- Kucharska, M.; Wesolowski, W.; Czerczak, S.; Socko, R. [Testing of the composition of e-cigarette liquids-Manufacturer-declared vs. true contents in a selected series of products]. Med. Pr. 2016, 67, 239–253. [Google Scholar] [CrossRef]
- Bacon, C.W.; Wenger, R.; Bullock, J.F. Chemical Changes in Tobacco during Flue-Curing. Ind. Eng. Chem. 1952, 44, 292–296. [Google Scholar] [CrossRef]
- Wahlberg, I.; Karlsson, K.; Austin, D.J.; Junker, N.; Roeraade, J.; Enzell, C.R.; Johnson, W.H. Effects of flue-curing and ageing on the volatile, neutral and acidic constituents of Virginia tobacco. Phytochemistry 1977, 16, 1217–1231. [Google Scholar] [CrossRef]
- Sen, S. The global cigarette: origins and evolution of British American Tobacco, 1880–1945. [Review of: Cox, H. The global cigarette: origins and evolution of British American Tobacco, 1880–1945. New York: Oxford U. Pr., 2000]. Am. Hist. Rev. 2002, 107, 164–165. [Google Scholar] [CrossRef] [PubMed]
- Sleight, V.J. A brief history of the electronic cigarette. J. Lung Pulm. Respir. Res. 2016, 3, 135–136. [Google Scholar] [CrossRef][Green Version]
- Hill, J. Cautions Against the Immoderate Use of Snuff. Founded on the Known Qualities of the Tobacco Plant; and the Effects it Must Produce When This Way Taken into the Body; Printed for Baldwin, R. and Jackson, J., Eds. London; 1761. [Google Scholar]
- Proctor, R.N. Tobacco and the global lung cancer epidemic. Nat. Rev. Cancer 2001, 1, 82–86. [Google Scholar] [CrossRef] [PubMed]
- Wynder, E.L.; Graham, E.A.; Croninger, A.B. Experimental production of carcinoma with cigarette tar. Cancer Res. 1953, 13, 855–864. [Google Scholar] [PubMed]
- Alberg, A.J.; Shopland, D.R.; Cummings, K.M. The 2014 Surgeon General’s report: commemorating the 50th Anniversary of the 1964 Report of the Advisory Committee to the US Surgeon General and updating the evidence on the health consequences of cigarette smoking. Am. J. Epidemiol. 2014, 179, 403–412. [Google Scholar] [CrossRef]
- WHO. Cancer. Available online: https://www.who.int/cancer/PRGlobocanFinal.pdf (accessed on 20 January 2020).
- WHO. Available online: https://www.who.int/news-room/fact-sheets/detail/chronic-obstructive-pulmonary-disease-(copd) (accessed on 20 January 2020).
- Gilbert, H.A. Smokeless Non-Tobacco Cigarette. U.S. Patent No. 3,200,819, 17 April 1963. [Google Scholar]
- Hon, L. Electronic Cigarette. U.S. Patent No. 8,689,805, 11 February 2009. [Google Scholar]
- Bullen, C. Electronic cigarettes for smoking cessation. Curr. Cardiol. Rep. 2014, 16, 538. [Google Scholar] [CrossRef]
- Polosa, R.; Caponnetto, P.; Morjaria, J.B.; Papale, G.; Campagna, D.; Russo, C. Effect of an electronic nicotine delivery device (e-Cigarette) on smoking reduction and cessation: a prospective 6-month pilot study. BMC Public Health 2011, 11, 786. [Google Scholar] [CrossRef]
- Rouabhia, M. Impact of Electronic Cigarettes on Oral Health: a Review. J. Can. Dent. Assoc. 2020, 86, 1488–2159. [Google Scholar]
- Group, I. North America E-Cigarette Market: Industry Trends, Share, Size, Growth, Opportunity and Forecast 2019–2024 ID: 4894046 Report December 2019 Region: North America 88 pages IMARC Group. Available online: https://www.mordorintelligence.com/industry-reports/global-e-cigarettes-market-industry (accessed on 20 January 2020).
- Soneji, S.S.; Knutzen, K.E.; Villanti, A.C. Use of Flavored E-Cigarettes Among Adolescents, Young Adults, and Older Adults: Findings From the Population Assessment for Tobacco and Health Study. Public Health Rep. 2019, 134, 282–292. [Google Scholar] [CrossRef]
- El-Khoury Lesueur, F.; Bolze, C.; Melchior, M. Factors associated with successful vs. unsuccessful smoking cessation: Data from a nationally representative study. Addict. Behav. 2018, 80, 110–115. [Google Scholar] [CrossRef]
- O’Connell, G.; Pritchard, J.D.; Prue, C.; Thompson, J.; Verron, T.; Graff, D.; Walele, T. A randomised, open-label, cross-over clinical study to evaluate the pharmacokinetic profiles of cigarettes and e-cigarettes with nicotine salt formulations in US adult smokers. Intern. Emerg. Med. 2019, 14, 853–861. [Google Scholar] [CrossRef] [PubMed]
- Caponnetto, P.; Campagna, D.; Cibella, F.; Morjaria, J.B.; Caruso, M.; Russo, C.; Polosa, R. EffiCiency and Safety of an eLectronic cigAreTte (ECLAT) as tobacco cigarettes substitute: a prospective 12-month randomized control design study. PLoS ONE 2013, 8, e66317. [Google Scholar] [CrossRef] [PubMed]
- Herrington, J.S.; Myers, C. Electronic cigarette solutions and resultant aerosol profiles. J. Chromatogr. A 2015, 1418, 192–199. [Google Scholar] [CrossRef] [PubMed]
- McNeill, A.; Brose, L.S.; Calder, R.; Bauld, L.; Robson, D. Evidence Review of E-Cigarettes and Heated Tobacco Products. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/684963/Evidence_review_of_e-cigarettes_and_heated_tobacco_products_2018.pdf (accessed on 25 January 2020).
- Substance Abuse and Mental Health Services Administration (US); Office of the Surgeon General (US). Smoking Cessation: A Report of the Surgeon General; U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health: Atlanta, GA, USA, 2020. [Google Scholar]
- Zhu, S.H.; Sun, J.Y.; Bonnevie, E.; Cummins, S.E.; Gamst, A.; Yin, L.; Lee, M. Four hundred and sixty brands of e-cigarettes and counting: implications for product regulation. Tob. Control. 2014, 23 (Suppl. 3), iii3–iii9. [Google Scholar] [CrossRef]
- Williams, M.; Talbot, P. Design Features in Multiple Generations of Electronic Cigarette Atomizers. Int. J. Environ. Res. Public Health 2019, 16, 2904. [Google Scholar] [CrossRef]
- Peace, M.R.; Mulder, H.A.; Baird, T.R.; Butler, K.E.; Friedrich, A.K.; Stone, J.W.; Turner, J.B.M.; Poklis, A.; Poklis, J.L. Evaluation of Nicotine and the Components of e-Liquids Generated from e-Cigarette Aerosols. J. Anal. Toxicol. 2018, 42, 537–543. [Google Scholar] [CrossRef]
- Prochaska, J.J. The public health consequences of e-cigarettes: a review by the National Academies of Sciences. A call for more research, a need for regulatory action. Addiction 2019, 114, 587–589. [Google Scholar] [CrossRef]
- Rowell, T.R.; Tarran, R. Will chronic e-cigarette use cause lung disease? Am. J. Physiol. Lung Cell. Mol. Physiol. 2015, 309, L1398–L1409. [Google Scholar] [CrossRef]
- Lozier, M.J.; Wallace, B.; Anderson, K.; Ellington, S.; Jones, C.M.; Rose, D.; Baldwin, G.; King, B.A.; Briss, P.; Mikosz, C.A. Update: Demographic, Product, and Substance-Use Characteristics of Hospitalized Patients in a Nationwide Outbreak of E-cigarette, or Vaping, Product Use-Associated Lung Injuries-United States, December 2019. MMWR Morb. Mortal. Wkly. Rep. 2019, 68, 1142–1148. [Google Scholar] [CrossRef]
- Uryupin, A.B.; Peregudov, A.S.; Kochetkov, K.A.; Bulatnikova, L.N.; Kiselev, S.S.; Nekrasov, Y.S. Qualitative and quantitative compositions of fluids for electronic cigarettes. Pharm. Chem. J. 2013, 46. [Google Scholar] [CrossRef]
- Phillips, B.; Titz, B.; Kogel, U.; Sharma, D.; Leroy, P.; Xiang, Y.; Vuillaume, G.; Lebrun, S.; Sciuscio, D.; Ho, J.; et al. Toxicity of the main electronic cigarette components, propylene glycol, glycerin, and nicotine, in Sprague-Dawley rats in a 90-day OECD inhalation study complemented by molecular endpoints. Food Chem. Toxicol. 2017, 109 Pt 1, 315–332. [Google Scholar] [CrossRef]
- Hahn, J.; Monakhova, Y.B.; Hengen, J.; Kohl-Himmelseher, M.; Schussler, J.; Hahn, H.; Kuballa, T.; Lachenmeier, D.W. Electronic cigarettes: overview of chemical composition and exposure estimation. Tob. Induc. Dis. 2014, 12, 23. [Google Scholar] [CrossRef] [PubMed]
- National Academies of Sciences, Engineering, and Medicine. Public Health Consequences of E-Cigarettes; Eaton, D.L., Kwan, L.Y., Stratton, K., Eds.; National Academies Press: Washington, DC, USA, 2018. [Google Scholar]
- FDA Lung Illnesses Associated with Use of Vaping Products. Available online: https://www.fda.gov/news-events/public-health-focus/lung-illnesses-associated-use-vaping-products#Analysis (accessed on 23 December 2019).
- Kosmider, L.; Sobczak, A.; Fik, M.; Knysak, J.; Zaciera, M.; Kurek, J.; Goniewicz, M.L. Carbonyl compounds in electronic cigarette vapors: effects of nicotine solvent and battery output voltage. Nicotine Tob. Res. 2014, 16, 1319–1326. [Google Scholar] [CrossRef] [PubMed]
- Muthumalage, T.; Lamb, T.; Friedman, M.R.; Rahman, I. E-cigarette flavored pods induce inflammation, epithelial barrier dysfunction, and DNA damage in lung epithelial cells and monocytes. Sci. Rep. 2019, 9, 19035. [Google Scholar] [CrossRef] [PubMed]
- Czoli, C.D.; Goniewicz, M.L.; Palumbo, M.; Leigh, N.; White, C.M.; Hammond, D. Identification of flavouring chemicals and potential toxicants in e-cigarette products in Ontario, Canada. Can. J. Public Health 2019, 110, 542–550. [Google Scholar] [CrossRef] [PubMed]
- Behar, R.Z.; Luo, W.; Lin, S.C.; Wang, Y.; Valle, J.; Pankow, J.F.; Talbot, P. Distribution, quantification and toxicity of cinnamaldehyde in electronic cigarette refill fluids and aerosols. Tob. Control. 2016, 25 (Suppl. 2), ii94–ii102. [Google Scholar] [CrossRef]
- Farley, F.; Robert, E. Thayer (1935–2014). Am. Psychol. 2015, 70, 281. [Google Scholar] [CrossRef]
- Kosmider, L.; Sobczak, A.; Prokopowicz, A.; Kurek, J.; Zaciera, M.; Knysak, J.; Smith, D.; Goniewicz, M.L. Cherry-flavoured electronic cigarettes expose users to the inhalation irritant, benzaldehyde. Thorax 2016, 71, 376–377. [Google Scholar] [CrossRef]
- Wu, Q.; Jiang, D.; Minor, M.; Chu, H.W. Electronic cigarette liquid increases inflammation and virus infection in primary human airway epithelial cells. PLoS ONE 2014, 9, e108342. [Google Scholar] [CrossRef]
- Muthumalage, T.; Prinz, M.; Ansah, K.O.; Gerloff, J.; Sundar, I.K.; Rahman, I. Inflammatory and Oxidative Responses Induced by Exposure to Commonly Used e-Cigarette Flavoring Chemicals and Flavored e-Liquids without Nicotine. Front. Physiol. 2017, 8, 1130. [Google Scholar] [CrossRef]
- Behar, R.Z.; Hua, M.; Talbot, P. Puffing topography and nicotine intake of electronic cigarette users. PLoS ONE 2015, 10, e0117222. [Google Scholar] [CrossRef] [PubMed]
- Benowitz, N.L.; Hukkanen, J.; Jacob, P. Nicotine chemistry, metabolism, kinetics and biomarkers. Handb. Exp. Pharm. 2009, 192, 29–60. [Google Scholar] [CrossRef]
- Benowitz, N.L.; Fraiman, J.B. Cardiovascular effects of electronic cigarettes. Nat. Rev. Cardiol. 2017, 14, 447–456. [Google Scholar] [CrossRef] [PubMed]
- Bhatnagar, A. E-Cigarettes and Cardiovascular Disease Risk: Evaluation of Evidence, Policy Implications, and Recommendations. Curr. Cardiovasc. Risk Rep. 2016, 10, 24. [Google Scholar] [CrossRef]
- Lam, D.C.; Luo, S.Y.; Fu, K.H.; Lui, M.M.; Chan, K.H.; Wistuba, I.I.; Gao, B.; Tsao, S.W.; Ip, M.S.; Minna, J.D. Nicotinic acetylcholine receptor expression in human airway correlates with lung function. Am. J. Physiol. Lung Cell. Mol. Physiol. 2016, 310, L232–L239. [Google Scholar] [CrossRef]
- Benowitz, N.L.; Gourlay, S.G. Cardiovascular toxicity of nicotine: implications for nicotine replacement therapy. J. Am. Coll. Cardiol. 1997, 29, 1422–1431. [Google Scholar] [CrossRef]
- Kozlowski, L.T.; Mehta, N.Y.; Sweeney, C.T.; Schwartz, S.S.; Vogler, G.P.; Jarvis, M.J.; West, R.J. Filter ventilation and nicotine content of tobacco in cigarettes from Canada, the United Kingdom, and the United States. Tob Control. 1998, 7, 369–375. [Google Scholar] [CrossRef]
- Chun, L.F.; Moazed, F.; Calfee, C.S.; Matthay, M.A.; Gotts, J.E. Pulmonary toxicity of e-cigarettes. Am. J. Physiol. Lung Cell. Mol. Physiol. 2017, 313, L193–l206. [Google Scholar] [CrossRef]
- Goniewicz, M.L.; Boykan, R.; Messina, C.R.; Eliscu, A.; Tolentino, J. High exposure to nicotine among adolescents who use Juul and other vape pod systems (‘pods’). Tob. Control. 2019, 28, 676–677. [Google Scholar] [CrossRef]
- Pankow, J.F.; Kim, K.; McWhirter, K.J.; Luo, W.; Escobedo, J.O.; Strongin, R.M.; Duell, A.K.; Peyton, D.H. Benzene formation in electronic cigarettes. PLoS ONE 2017, 12, e0173055. [Google Scholar] [CrossRef]
- Rao, P.; Liu, J.; Springer, M.L. JUUL and Combusted Cigarettes Comparably Impair Endothelial Function. Tob. Regul. Sci. 2020, 6, 30–37. [Google Scholar] [CrossRef] [PubMed]
- King, B.A.; Gammon, D.G.; Marynak, K.L.; Rogers, T. Electronic Cigarette Sales in the United States, 2013–2017. JAMA 2018, 320, 1379–1380. [Google Scholar] [CrossRef] [PubMed]
- Jackler, R.K.; Ramamurthi, D. Nicotine arms race: JUUL and the high-nicotine product market. Tob. Control. 2019, 28, 623–628. [Google Scholar] [CrossRef] [PubMed]
- Lafaye, G.; Karila, L.; Blecha, L.; Benyamina, A. Cannabis, cannabinoids, and health. Dialogues Clin. Neurosci. 2017, 19, 309–316. [Google Scholar] [PubMed]
- Hoffman, D.; Brunnemann, K.D.; Gori, G.B.; Wynder, E.L. On the Carcinogenicity of marijuana smoke. In Recent Advances in Phytochemistry; Runeckles, V.C., Ed.; Plenum Press: New York, NY, USA, 1975; pp. 63–81. [Google Scholar]
- Schwartz, R. Legalize marijuana without the smoke. CMAJ 2017, 189, E137–E138. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Blohm, E.; Sell, P.; Neavyn, M. Cannabinoid toxicity in pediatrics. Curr. Opin. Pediatrics 2019, 31, 256–261. [Google Scholar] [CrossRef]
- Stepanov, I.; Fujioka, N. Bringing attention to e-cigarette pH as an important element for research and regulation. Tob. Control. 2015, 24, 413–414. [Google Scholar] [CrossRef]
- Traboulsi, H.; Guerrina, N.; Iu, M.; Maysinger, D.; Ariya, P.; Baglole, C.J. Inhaled Pollutants: The Molecular Scene behind Respiratory and Systemic Diseases Associated with Ultrafine Particulate Matter. Int. J. Mol. Sci. 2017, 18, 243. [Google Scholar] [CrossRef]
- Ghio, A.J.; Hilborn, E.D.; Stonehuerner, J.G.; Dailey, L.A.; Carter, J.D.; Richards, J.H.; Crissman, K.M.; Foronjy, R.F.; Uyeminami, D.L.; Pinkerton, K.E. Particulate matter in cigarette smoke alters iron homeostasis to produce a biological effect. Am. J. Respir. Crit. Care Med. 2008, 178, 1130–1138. [Google Scholar] [CrossRef]
- Zhao, J.; Zhang, Y.; Sisler, J.D.; Shaffer, J.; Leonard, S.S.; Morris, A.M.; Qian, Y.; Bello, D.; Demokritou, P. Assessment of reactive oxygen species generated by electronic cigarettes using acellular and cellular approaches. J. Hazard. Mater. 2018, 344, 549–557. [Google Scholar] [CrossRef]
- Sen, C. Assessing Ultrafine Particles Exposure in Electronic Cigarette Vape Shops; University of California: California, CA, USA, 2016. [Google Scholar]
- Volesky, K.D.; Maki, A.; Scherf, C.; Watson, L.; Van Ryswyk, K.; Fraser, B.; Weichenthal, S.A.; Cassol, E.; Villeneuve, P.J. The influence of three e-cigarette models on indoor fine and ultrafine particulate matter concentrations under real-world conditions. Environ. Pollut. 2018, 243 Pt B, 882–889. [Google Scholar] [CrossRef]
- Lampos, S.; Kostenidou, E.; Farsalinos, K.; Zagoriti, Z.; Ntoukas, A.; Dalamarinis, K.; Savranakis, P.; Lagoumintzis, G.; Poulas, K. Real-Time Assessment of E-Cigarettes and Conventional Cigarettes Emissions: Aerosol Size Distributions, Mass and Number Concentrations. Toxics 2019, 7, 45. [Google Scholar] [CrossRef] [PubMed]
- Mikheev, V.B.; Brinkman, M.C.; Granville, C.A.; Gordon, S.M.; Clark, P.I. Real-Time Measurement of Electronic Cigarette Aerosol Size Distribution and Metals Content Analysis. Nicotine Tob. Res. 2016, 18, 1895–1902. [Google Scholar] [CrossRef]
- Floyd, E.L.; Queimado, L.; Wang, J.; Regens, J.L.; Johnson, D.L. Electronic cigarette power affects count concentration and particle size distribution of vaping aerosol. PLoS ONE 2018, 13, e0210147. [Google Scholar] [CrossRef]
- Pisinger, C. A Systematic Review of Health Effects of Lectronic Cigarettes. Available online: https://www.who.int/tobacco/industry/product_regulation/BackgroundPapersENDS3_4November-.pdf (accessed on 28 April 2020).
- Manigrasso, M.; Buonanno, G.; Fuoco, F.C.; Stabile, L.; Avino, P. Aerosol deposition doses in the human respiratory tree of electronic cigarette smokers. Environ. Pollut. 2015, 196, 257–267. [Google Scholar] [CrossRef]
- Vriens, A.; Nawrot, T.S.; Saenen, N.D.; Provost, E.B.; Kicinski, M.; Lefebvre, W.; Vanpoucke, C.; Van Deun, J.; De Wever, O.; Vrijens, K.; et al. Recent exposure to ultrafine particles in school children alters miR-222 expression in the extracellular fraction of saliva. Environ. Health 2016, 15, 80. [Google Scholar] [CrossRef] [PubMed]
- Ogunwale, M.A.; Li, M.; Ramakrishnam Raju, M.V.; Chen, Y.; Nantz, M.H.; Conklin, D.J.; Fu, X.A. Aldehyde Detection in Electronic Cigarette Aerosols. ACS Omega 2017, 2, 1207–1214. [Google Scholar] [CrossRef] [PubMed]
- Farsalinos, K.E.; Voudris, V.; Poulas, K. E-cigarettes generate high levels of aldehydes only in ‘dry puff’ conditions. Addiction 2015, 110, 1352–1356. [Google Scholar] [CrossRef]
- Troutt, W.D.; DiDonato, M.D. Carbonyl Compounds Produced by Vaporizing Cannabis Oil Thinning Agents. J. Altern. Complement. Med. 2017, 23, 879–884. [Google Scholar] [CrossRef] [PubMed]
- Guerrina, N.; Traboulsi, H.; Eidelman, D.H.; Baglole, C.J. The Aryl Hydrocarbon Receptor and the Maintenance of Lung Health. Int. J. Mol. Sci. 2018, 19, 3882. [Google Scholar] [CrossRef]
- Jia, C.; Batterman, S. A critical review of naphthalene sources and exposures relevant to indoor and outdoor air. Int. J. Environ. Res. Public Health 2010, 7, 2903–2939. [Google Scholar] [CrossRef] [PubMed]
- Carratt, S.A.; Kovalchuk, N.; Ding, X.; Van Winkle, L.S. Metabolism and Lung Toxicity of Inhaled Naphthalene: Effects of Postnatal Age and Sex. Toxicol. Sci. 2019, 170, 536–548. [Google Scholar] [CrossRef]
- Pelkonen, O.; Nebert, D.W. Metabolism of polycyclic aromatic hydrocarbons: etiologic role in carcinogenesis. Pharmacol. Rev. 1982, 34, 189–222. [Google Scholar] [PubMed]
- Alexandrov, K.; Rojas, M.; Rolando, C. DNA Damage by Benzo(a)pyrene in Human Cells Is Increased by Cigarette Smoke and Decreased by a Filter Containing Rosemary Extract, Which Lowers Free Radicals. Cancer Res. 2006, 66, 11938–11945. [Google Scholar] [CrossRef]
- Wagner, K.A.; Flora, J.W.; Melvin, M.S.; Avery, K.C.; Ballentine, R.M.; Brown, A.P.; McKinney, W.J. An evaluation of electronic cigarette formulations and aerosols for harmful and potentially harmful constituents (HPHCs) typically derived from combustion. Regul. Toxicol. Pharmacol. 2018, 95, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Margham, J.; McAdam, K.; Forster, M.; Liu, C.; Wright, C.; Mariner, D.; Proctor, C. Chemical Composition of Aerosol from an E-Cigarette: A Quantitative Comparison with Cigarette Smoke. Chem. Res. Toxicol. 2016, 29, 1662–1678. [Google Scholar] [CrossRef] [PubMed]
- Pasquale Avino, A.R. Maurizio Manigrasso4, The inorganic fraction in e-liquids used in vapor products including e-cigarettes. J. Radioanal. Nucl. Chem. 2019, 322, 6. [Google Scholar]
- Papaefstathiou, E.; Stylianou, M.; Agapiou, A. Main and side stream effects of electronic cigarettes. J. Environ. Manag. 2019, 238, 10–17. [Google Scholar] [CrossRef]
- Gray, N.; Halstead, M.; Gonzalez-Jimenez, N.; Valentin-Blasini, L.; Watson, C.; Pappas, R.S. Analysis of Toxic Metals in Liquid from Electronic Cigarettes. Int. J. Environ. Res. Public Health 2019, 16, 4450. [Google Scholar] [CrossRef]
- Agency for Toxic Substances & Disease Registry Public Health Statement for Chromium. Available online: https://www.atsdr.cdc.gov/phs/phs.asp?id=60&tid=17 (accessed on 20 December 2019).
- Prokopowicz, A.; Sobczak, A.; Szula-Chraplewska, M.; Ochota, P.; Kosmider, L. Exposure to Cadmium and Lead in Cigarette Smokers Who Switched to Electronic Cigarettes. Nicotine Tob. Res. 2019, 21, 1198–1205. [Google Scholar] [CrossRef]
- GASP of Colorado (Group to Alleviate Smoking Pollution). Harmful Chemicals in Electronic Cigarettes. Available online: http://www.gaspforair.org/gasp/gedc/pdf/E-CigSmoke.pdf (accessed on 7 January 2020).
- Edwards, S.H.; Rossiter, L.M.; Taylor, K.M.; Holman, M.R.; Zhang, L.; Ding, Y.S.; Watson, C.H. Tobacco-Specific Nitrosamines in the Tobacco and Mainstream Smoke of U.S. Commercial Cigarettes. Chem. Res. Toxicol. 2017, 30, 540–551. [Google Scholar] [CrossRef] [PubMed]
- Hecht, S.S.; Hoffmann, D. Tobacco-specific nitrosamines, an important group of carcinogens in tobacco and tobacco smoke. Carcinogenesis 1988, 9, 875–884. [Google Scholar] [CrossRef] [PubMed]
- Konstantinou, E.; Fotopoulou, F.; Drosos, A.; Dimakopoulou, N.; Zagoriti, Z.; Niarchos, A.; Makrynioti, D.; Kouretas, D.; Farsalinos, K.; Lagoumintzis, G.; et al. Tobacco-specific nitrosamines: A literature review. Food Chem. Toxicol. 2018, 118, 198–203. [Google Scholar] [CrossRef]
- Kim, H.J.; Shin, H.S. Determination of tobacco-specific nitrosamines in replacement liquids of electronic cigarettes by liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2013, 1291, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Gaurav, R. Vaping Away Epithelial Integrity. Am. J. Respir. Cell Mol. Biol. 2019, 61, 127–129. [Google Scholar] [CrossRef] [PubMed]
- Greenwood, M.F.; Holland, P. The mammalian respiratory tract surface. A scanning electron microscopic study. Lab. Investig. J. Tech. Methods Pathol. 1972, 27, 296–304. [Google Scholar]
- Borgas, D.; Chambers, E.; Newton, J.; Ko, J.; Rivera, S.; Rounds, S.; Lu, Q. Cigarette Smoke Disrupted Lung Endothelial Barrier Integrity and Increased Susceptibility to Acute Lung Injury via Histone Deacetylase 6. Am. J. Respir. Cell Mol. Biol. 2016, 54, 683–696. [Google Scholar] [CrossRef]
- Bengalli, R.; Ferri, E.; Labra, M.; Mantecca, P. Lung Toxicity of Condensed Aerosol from E-CIG Liquids: Influence of the Flavor and the In Vitro Model Used. Int. J. Environ. Res. Public Health 2017, 14, 1254. [Google Scholar] [CrossRef]
- Gerloff, J.; Sundar, I.K.; Freter, R.; Sekera, E.R.; Friedman, A.E.; Robinson, R.; Pagano, T.; Rahman, I. Inflammatory Response and Barrier Dysfunction by Different e-Cigarette Flavoring Chemicals Identified by Gas Chromatography-Mass Spectrometry in e-Liquids and e-Vapors on Human Lung Epithelial Cells and Fibroblasts. Appl. In Vitro Toxicol. 2017, 3, 28–40. [Google Scholar] [CrossRef]
- Hwang, J.H.; Lyes, M.; Sladewski, K.; Enany, S.; McEachern, E.; Mathew, D.P.; Das, S.; Moshensky, A.; Bapat, S.; Pride, D.T.; et al. Electronic cigarette inhalation alters innate immunity and airway cytokines while increasing the virulence of colonizing bacteria. J. Mol. Med. 2016, 94, 667–679. [Google Scholar] [CrossRef]
- Schweitzer, K.S.; Chen, S.X.; Law, S.; Van Demark, M.; Poirier, C.; Justice, M.J.; Hubbard, W.C.; Kim, E.S.; Lai, X.; Wang, M.; et al. Endothelial disruptive proinflammatory effects of nicotine and e-cigarette vapor exposures. Am. J. Physiol. Lung Cell. Mol. Physiol. 2015, 309, L175–L187. [Google Scholar] [CrossRef] [PubMed]
- King, P.T. Inflammation in chronic obstructive pulmonary disease and its role in cardiovascular disease and lung cancer. Clin. Transl. Med. 2015, 4, 68. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, N.H. Smoking, immunity, and DNA damage. Transl. Lung Cancer Res. 2019, 8 (Suppl. 1), S3–S6. [Google Scholar] [CrossRef]
- Jaspers, I. Cigarette smoke effects on innate immune mechanisms in the nasal mucosa. Potential effects on the microbiome. Ann. Am. Thorac. Soc. 2014, 11 (Suppl. 1), S38–S42. [Google Scholar] [CrossRef]
- Lerner, C.A.; Sundar, I.K.; Yao, H.; Gerloff, J.; Ossip, D.J.; McIntosh, S.; Robinson, R.; Rahman, I. Vapors produced by electronic cigarettes and e-juices with flavorings induce toxicity, oxidative stress, and inflammatory response in lung epithelial cells and in mouse lung. PLoS ONE 2015, 10, e0116732. [Google Scholar] [CrossRef]
- Glynos, C.; Bibli, S.I.; Katsaounou, P.; Pavlidou, A.; Magkou, C.; Karavana, V.; Topouzis, S.; Kalomenidis, I.; Zakynthinos, S.; Papapetropoulos, A. Comparison of the effects of e-cigarette vapor with cigarette smoke on lung function and inflammation in mice. Am. J. Physiol. Lung Cell. Mol. Physiol. 2018, 315, L662–L672. [Google Scholar] [CrossRef] [PubMed]
- Reidel, B.; Radicioni, G.; Clapp, P.W.; Ford, A.A.; Abdelwahab, S.; Rebuli, M.E.; Haridass, P.; Alexis, N.E.; Jaspers, I.; Kesimer, M. E-Cigarette Use Causes a Unique Innate Immune Response in the Lung, Involving Increased Neutrophilic Activation and Altered Mucin Secretion. Am. J. Respir. Crit. Care Med. 2018, 197, 492–501. [Google Scholar] [CrossRef] [PubMed]
- Harvanko, A.M.; McCubbin, A.K.; Ashford, K.B.; Kelly, T.H. Electronic cigarette liquid and device parameters and aerosol characteristics: A survey of regular users. Addict. Behav. 2018, 84, 201–206. [Google Scholar] [CrossRef]
- Escobar, Y.H.; Nipp, G.; Cui, T.; Petters, S.S.; Surratt, J.D.; Jaspers, I. In Vitro Toxicity and Chemical Characterization of Aerosol Derived from Electronic Cigarette Humectants Using a Newly Developed Exposure System. Chem. Res. Toxicol. 2020. [Google Scholar] [CrossRef]
- Cirillo, S.; Vivarelli, F.; Turrini, E.; Fimognari, C.; Burattini, S.; Falcieri, E.; Rocchi, M.B.L.; Cardenia, V.; Rodriguez-Estrada, M.T.; Paolini, M.; et al. The customizable e-cigarette resistance influences toxicological outcomes: lung degeneration, inflammation and oxidative stress-induced in a rat model. Toxicol. Sci. 2019, 172, 132–145. [Google Scholar] [CrossRef]
- Vardavas, C.I.; Anagnostopoulos, N.; Kougias, M.; Evangelopoulou, V.; Connolly, G.N.; Behrakis, P.K. Short-term pulmonary effects of using an electronic cigarette: impact on respiratory flow resistance, impedance, and exhaled nitric oxide. Chest 2012, 141, 1400–1406. [Google Scholar] [CrossRef] [PubMed]
- Boulay, M.E.; Henry, C.; Bosse, Y.; Boulet, L.P.; Morissette, M.C. Acute effects of nicotine-free and flavour-free electronic cigarette use on lung functions in healthy and asthmatic individuals. Respir. Res. 2017, 18, 33. [Google Scholar] [CrossRef] [PubMed]
- Mehta, D.R.; Ashkar, A.A.; Mossman, K.L. The nitric oxide pathway provides innate antiviral protection in conjunction with the type I interferon pathway in fibroblasts. PLoS ONE 2012, 7, e31688. [Google Scholar] [CrossRef] [PubMed]
- Coleman, J.W. Nitric oxide in immunity and inflammation. Int. Immunopharmacol. 2001, 1, 1397–1406. [Google Scholar] [CrossRef]
- Verini, M.; Consilvio, N.P.; Di Pillo, S.; Cingolani, A.; Spagnuolo, C.; Rapino, D.; Scaparrotta, A.; Chiarelli, F. FeNO as a Marker of Airways Inflammation: The Possible Implications in Childhood Asthma Management. J. Allergy 2010, 2010, 691425. [Google Scholar] [CrossRef] [PubMed]
- Lakhan, S.E.; Kirchgessner, A. Anti-inflammatory effects of nicotine in obesity and ulcerative colitis. J. Transl. Med. 2011, 9, 129. [Google Scholar] [CrossRef]
- Sui, H.X.; Ke, S.Z.; Xu, D.D.; Lu, N.N.; Wang, Y.N.; Zhang, Y.H.; Gao, F.G. Nicotine induces TIPE2 upregulation and Stat3 phosphorylation contributes to cholinergic anti-inflammatory effect. Int. J. Oncol. 2017, 51, 987–995. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. How Tobacco Smoke Causes Disease: The Biology and Behavioral Basis for Smoking-Attributable Disease. 2010. Available online: https://www.ncbi.nlm.nih.gov/pubmed/21452462 (accessed on 22 December 2019).
- Ray, P.D.; Huang, B.W.; Tsuji, Y. Reactive oxygen species (ROS) homeostasis and redox regulation in cellular signaling. Cell. Signal. 2012, 24, 981–990. [Google Scholar] [CrossRef]
- Zhao, J.; Hopke, P.K. Concentration of Reactive Oxygen Species (ROS) in Mainstream and Sidestream Cigarette Smoke. Aerosol Sci. Technol. 2012, 46, 191–197. [Google Scholar] [CrossRef]
- Lerner, C.A.; Sundar, I.K.; Watson, R.M.; Elder, A.; Jones, R.; Done, D.; Kurtzman, R.; Ossip, D.J.; Robinson, R.; McIntosh, S.; et al. Environmental health hazards of e-cigarettes and their components: Oxidants and copper in e-cigarette aerosols. Environ. Pollut. 2015, 198, 100–107. [Google Scholar] [CrossRef]
- Anderson, C.; Majeste, A.; Hanus, J.; Wang, S. E-Cigarette Aerosol Exposure Induces Reactive Oxygen Species, DNA Damage, and Cell Death in Vascular Endothelial Cells. Toxicol. Sci. 2016, 154, 332–340. [Google Scholar] [CrossRef] [PubMed]
- Mathiassen, S.G.; De Zio, D.; Cecconi, F. Autophagy and the Cell Cycle: A Complex Landscape. Front. Oncol. 2017, 7, 51. [Google Scholar] [CrossRef] [PubMed]
- Shivalingappa, P.C.; Hole, R.; Westphal, C.V.; Vij, N. Airway Exposure to E-Cigarette Vapors Impairs Autophagy and Induces Aggresome Formation. Antioxid. Redox Signal. 2016, 24, 186–204. [Google Scholar] [CrossRef] [PubMed]
- Bodas, M.; Van Westphal, C.; Carpenter-Thompson, R.; Mohanty, D.K.; Vij, N. Nicotine exposure induces bronchial epithelial cell apoptosis and senescence via ROS mediated autophagy-impairment. Free Radic. Biol. Med. 2016, 97, 441–453. [Google Scholar] [CrossRef]
- Schweitzer, K.S.; Hatoum, H.; Brown, M.B.; Gupta, M.; Justice, M.J.; Beteck, B.; Van Demark, M.; Gu, Y.; Presson, R.G., Jr.; Hubbard, W.C.; et al. Mechanisms of lung endothelial barrier disruption induced by cigarette smoke: role of oxidative stress and ceramides. Am. J. Physiol. Lung Cell. Mol. Physiol. 2011, 301, L836–L846. [Google Scholar] [CrossRef]
- Peluso, M.; Munnia, A.; Piro, S.; Armillis, A.; Ceppi, M.; Matullo, G.; Puntoni, R. Smoking, DNA adducts and number of risk DNA repair alleles in lung cancer cases, in subjects with benign lung diseases and in controls. J. Nucleic Acids 2010, 2010, 386798. [Google Scholar] [CrossRef]
- Queimado, L.; Wagener, T.; Ganapathy, V. Electronic cigarette aerosols induce DNA damage and reduce DNA repair: Consistency across species. Proc. Natl. Acad. Sci. USA 2018, 115, E5437–E5438. [Google Scholar] [CrossRef]
- Lee, H.W.; Park, S.H.; Weng, M.W.; Wang, H.T.; Huang, W.C.; Lepor, H.; Wu, X.R.; Chen, L.C.; Tang, M.S. E-cigarette smoke damages DNA and reduces repair activity in mouse lung, heart, and bladder as well as in human lung and bladder cells. Proc. Natl. Acad. Sci. USA 2018, 115, E1560–E1569. [Google Scholar] [CrossRef]
- Ganapathy, V.; Manyanga, J.; Brame, L.; McGuire, D.; Sadhasivam, B.; Floyd, E.; Rubenstein, D.A.; Ramachandran, I.; Wagener, T.; Queimado, L. Electronic cigarette aerosols suppress cellular antioxidant defenses and induce significant oxidative DNA damage. PLoS ONE 2017, 12, e0177780. [Google Scholar] [CrossRef]
- Espinoza-Derout, J.; Shao, X.M.; Bankole, E.; Hasan, K.M.; Mtume, N.; Liu, Y.; Sinha-Hikim, A.P.; Friedman, T.C. Hepatic DNA Damage Induced by Electronic Cigarette Exposure Is Associated With the Modulation of NAD+/PARP1/SIRT1 Axis. Front. Endocrinol. 2019, 10, 320. [Google Scholar] [CrossRef]
- Flach, S.; Maniam, P.; Manickavasagam, J. E-cigarettes and head and neck cancers: A systematic review of the current literature. Clin. Otolaryngol. 2019, 44, 749–756. [Google Scholar] [CrossRef] [PubMed]
- Thorne, D.; Larard, S.; Baxter, A.; Meredith, C.; Gaa, M. The comparative in vitro assessment of e-cigarette and cigarette smoke aerosols using the gammaH2AX assay and applied dose measurements. Toxicol. Lett. 2017, 265, 170–178. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, N.; Walker, G.C. Mechanisms of DNA damage, repair, and mutagenesis. Environ. Mol. Mutagenes. 2017, 58, 235–263. [Google Scholar] [CrossRef]
- Corriden, R.; Moshensky, A.; Bojanowski, C.M.; Meier, A.; Chien, J.; Nelson, R.K.; Crotty Alexander, L.E. E-cigarette use increases susceptibility to bacterial infection by impairment of human neutrophil chemotaxis, phagocytosis, and NET formation. Am. J. Physiol. Cell Physiol. 2020, 318, C205–C214. [Google Scholar] [CrossRef] [PubMed]
- Noah, T.L.; Zhou, H.; Jaspers, I. Alteration of the nasal responses to influenza virus by tobacco smoke. Curr. Opin. Allergy Clin. Immunol. 2012, 12, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Luo, G.; Chen, J.; Jiang, R.; Zhu, J.; Hu, N.; Huang, W.; Cheng, G.; Jia, M.; Su, B.; et al. Cigarette smoke attenuates phagocytic ability of macrophages through down-regulating Milk fat globule-EGF factor 8 (MFG-E8) expressions. Sci. Rep. 2017, 7, 42642. [Google Scholar] [CrossRef]
- Monick, M.M.; Powers, L.S.; Walters, K.; Lovan, N.; Zhang, M.; Gerke, A.; Hansdottir, S.; Hunninghake, G.W. Identification of an autophagy defect in smokers’ alveolar macrophages. J. Immunol. 2010, 185, 5425–5435. [Google Scholar] [CrossRef]
- Qiu, F.; Liang, C.L.; Liu, H.; Zeng, Y.Q.; Hou, S.; Huang, S.; Lai, X.; Dai, Z. Impacts of cigarette smoking on immune responsiveness: Up and down or upside down? Oncotarget 2017, 8, 268–284. [Google Scholar] [CrossRef]
- Mian, M.F.; Pek, E.A.; Mossman, K.L.; Stampfli, M.R.; Ashkar, A.A. Exposure to cigarette smoke suppresses IL-15 generation and its regulatory NK cell functions in poly I:C-augmented human PBMCs. Mol. Immunol. 2009, 46, 3108–3116. [Google Scholar] [CrossRef]
- Javed, F.; Kellesarian, S.V.; Sundar, I.K.; Romanos, G.E.; Rahman, I. Recent updates on electronic cigarette aerosol and inhaled nicotine effects on periodontal and pulmonary tissues. Oral Dis. 2017, 23, 1052–1057. [Google Scholar] [CrossRef]
- Ween, M.P.; Whittall, J.J.; Hamon, R.; Reynolds, P.N.; Hodge, S.J. Phagocytosis and Inflammation: Exploring the effects of the components of E-cigarette vapor on macrophages. Physiol. Rep. 2017, 5, e13370. [Google Scholar] [CrossRef] [PubMed]
- Madison, M.C.; Landers, C.T.; Gu, B.H.; Chang, C.Y.; Tung, H.Y.; You, R.; Hong, M.J.; Baghaei, N.; Song, L.Z.; Porter, P.; et al. Electronic cigarettes disrupt lung lipid homeostasis and innate immunity independent of nicotine. J. Clin. Investig. 2019, 129, 4290–4304. [Google Scholar] [CrossRef] [PubMed]
- Martin, E.M.; Clapp, P.W.; Rebuli, M.E.; Pawlak, E.A.; Glista-Baker, E.; Benowitz, N.L.; Fry, R.C.; Jaspers, I. E-cigarette use results in suppression of immune and inflammatory-response genes in nasal epithelial cells similar to cigarette smoke. Am. J. Physiol. Lung Cell. Mol. Physiol. 2016, 311, L135–L144. [Google Scholar] [CrossRef]
- Sokol, C.L.; Luster, A.D. The chemokine system in innate immunity. Cold Spring Harb. Perspect. Biol. 2015, 7, a016303. [Google Scholar] [CrossRef] [PubMed]
- Edmead, C.E.; Lamb, J.R.; Hoyne, G.F. The T cell surface protein, CD28. Int. J. Biochem. Cell Biol. 1997, 29, 1053–1057. [Google Scholar] [CrossRef]
- Chen, H.; Li, G.; Chan, Y.L.; Chapman, D.G.; Sukjamnong, S.; Nguyen, T.; Annissa, T.; McGrath, K.C.; Sharma, P.; Oliver, B.G. Maternal E-Cigarette Exposure in Mice Alters DNA Methylation and Lung Cytokine Expression in Offspring. Am. J. Respir. Cell Mol. Biol. 2018, 58, 366–377. [Google Scholar] [CrossRef] [PubMed]
- Zago, M.; Rico de Souza, A.; Hecht, E.; Rousseau, S.; Hamid, Q.; Eidelman, D.H.; Baglole, C.J. The NF-kappaB family member RelB regulates microRNA miR-146a to suppress cigarette smoke-induced COX-2 protein expression in lung fibroblasts. Toxicol. Lett. 2014, 226, 107–116. [Google Scholar] [CrossRef]
- Rogers, S.; de Souza, A.R.; Zago, M.; Iu, M.; Guerrina, N.; Gomez, A.; Matthews, J.; Baglole, C.J. Aryl hydrocarbon receptor (AhR)-dependent regulation of pulmonary miRNA by chronic cigarette smoke exposure. Sci. Rep. 2017, 7, 40539. [Google Scholar] [CrossRef]
- Izzotti, A.; Calin, G.A.; Arrigo, P.; Steele, V.E.; Croce, C.M.; De Flora, S. Downregulation of microRNA expression in the lungs of rats exposed to cigarette smoke. FASEB J. 2009, 23, 806–812. [Google Scholar] [CrossRef]
- Pottelberge, G.R.; Mestdagh, P.; Bracke, K.R.; Thas, O.; Durme, Y.M.; Joos, G.F.; Vandesompele, J.; Brusselle, G.G. MicroRNA expression in induced sputum of smokers and patients with chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 2011, 183, 898–906. [Google Scholar] [CrossRef]
- Petrek, H.; Yu, A.M. MicroRNAs in non-small cell lung cancer: Gene regulation, impact on cancer cellular processes, and therapeutic potential. Pharm. Res. Perspect 2019, 7, e00528. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Zhu, Z.; Guo, X.; Kong, X. The roles of microRNAs in the pathogenesis of chronic obstructive pulmonary disease. Int. Immunopharmacol. 2019, 67, 335–347. [Google Scholar] [CrossRef] [PubMed]
- Solleti, S.K.; Bhattacharya, S.; Ahmad, A.; Wang, Q.; Mereness, J.; Rangasamy, T.; Mariani, T.J. MicroRNA expression profiling defines the impact of electronic cigarettes on human airway epithelial cells. Sci. Rep. 2017, 7, 1081. [Google Scholar] [CrossRef] [PubMed]
- Xue, M.; Zhuo, Y.; Shan, B. MicroRNAs, Long Noncoding RNAs, and Their Functions in Human Disease. Methods Mol. Biol. 2017, 1617, 1–25. [Google Scholar]
- Tommasi, S.; Caliri, A.W.; Caceres, A.; Moreno, D.E.; Li, M.; Chen, Y.; Siegmund, K.D.; Besaratinia, A. Deregulation of Biologically Significant Genes and Associated Molecular Pathways in the Oral Epithelium of Electronic Cigarette Users. Int. J. Mol. Sci. 2019, 20, 738. [Google Scholar] [CrossRef]
- Association, C.L. Vaping—What You Need to Know. Available online: https://www.lung.ca/lung-health/vaping-what-you-need-know (accessed on 12 February 2020).
- Society, C.C. What You Need to Know about E-Cigarettes. Available online: https://www.cancer.ca/en/prevention-and-screening/reduce-cancer-risk/make-healthy-choices/live-smoke-free/what-you-need-to-know-about-e-cigarettes/?region=qc (accessed on 12 February 2020).
- Moritz, E.D.; Zapata, L.B.; Lekiachvili, A.; Glidden, E.; Annor, F.B.; Werner, A.K.; Ussery, E.N.; Hughes, M.M.; Kimball, A.; DeSisto, C.L.; et al. Update: Characteristics of Patients in a National Outbreak of E-cigarette, or Vaping, Product Use-Associated Lung Injuries - United States, October 2019. MMWR Morb. Mortal. Wkly. Rep. 2019, 68, 985–989. [Google Scholar]
- Czogala, J.; Goniewicz, M.L.; Fidelus, B.; Zielinska-Danch, W.; Travers, M.J.; Sobczak, A. Secondhand exposure to vapors from electronic cigarettes. Nicotine Tob. Res. 2014, 16, 655–662. [Google Scholar] [CrossRef]
- Palamidas, A.; Tsikrika, S.; Katsaounou, P.; Vakali, S.; Gennimata, S.-A.; Kaltsakas, G.; Gratziou, C.; Koulouris, N. Acute effects of short term use of e-cigarettes on airways physiology and respiratory symptoms in smokers with and without airways obstructive diseases and in healthy non smokers. Tob. Prev. Cessat. 2017, 3, 5. [Google Scholar] [CrossRef]
- McConnell, R.; Barrington-Trimis, J.L.; Wang, K.; Urman, R.; Hong, H.; Unger, J.; Samet, J.; Leventhal, A.; Berhane, K. Electronic Cigarette Use and Respiratory Symptoms in Adolescents. Am. J. Respir. Crit. Care Med. 2017, 195, 1043–1049. [Google Scholar] [CrossRef]
- Cho, J.H.; Paik, S.Y. Association between Electronic Cigarette Use and Asthma among High School Students in South Korea. PLoS ONE 2016, 11, e0151022. [Google Scholar] [CrossRef]
- Varughese, S.; Teschke, K.; Brauer, M.; Chow, Y.; van Netten, C.; Kennedy, S.M. Effects of theatrical smokes and fogs on respiratory health in the entertainment industry. Am. J. Ind. Med. 2005, 47, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Wieslander, G.; Norback, D.; Lindgren, T. Experimental exposure to propylene glycol mist in aviation emergency training: Acute ocular and respiratory effects. Occup. Environ. Med. 2001, 58, 649–655. [Google Scholar] [CrossRef] [PubMed]
- Bahl, V.; Lin, S.; Xu, N.; Davis, B.; Wang, Y.H.; Talbot, P. Comparison of electronic cigarette refill fluid cytotoxicity using embryonic and adult models. Reprod. Toxicol. 2012, 34, 529–537. [Google Scholar] [CrossRef]
- Flouris, A.D.; Chorti, M.S.; Poulianiti, K.P.; Jamurtas, A.Z.; Kostikas, K.; Tzatzarakis, M.N.; Wallace Hayes, A.; Tsatsakis, A.M.; Koutedakis, Y. Acute impact of active and passive electronic cigarette smoking on serum cotinine and lung function. Inhal. Toxicol. 2013, 25, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Vink, G.R.; Arets, H.G.; van der Laag, J.; van der Ent, C.K. Impulse oscillometry: a measure for airway obstruction. Pediatr. Pulmonol. 2003, 35, 214–219. [Google Scholar] [CrossRef]
- Walele, T.; Bush, J.; Koch, A.; Savioz, R.; Martin, C.; O’Connell, G. Evaluation of the safety profile of an electronic vapour product used for two years by smokers in a real-life setting. Regul. Toxicol. Pharmacol. 2018, 92, 226–238. [Google Scholar] [CrossRef]
- Gupta, D.; Hansell, A.; Nichols, T.; Duong, T.; Ayres, J.G.; Strachan, D. Epidemiology of pneumothorax in England. Thorax 2000, 55, 666–671. [Google Scholar] [CrossRef]
- Bonilla, A.; Blair, A.J.; Alamro, S.M.; Ward, R.A.; Feldman, M.B.; Dutko, R.A.; Karagounis, T.K.; Johnson, A.L.; Folch, E.E.; Vyas, J.M. Recurrent spontaneous pneumothoraces and vaping in an 18-year-old man: a case report and review of the literature. J. Med. Case Rep. 2019, 13, 283. [Google Scholar] [CrossRef]
- Allen, J.N.; Pacht, E.R.; Gadek, J.E.; Davis, W.B. Acute eosinophilic pneumonia as a reversible cause of noninfectious respiratory failure. N. Engl. J. Med. 1989, 321, 569–574. [Google Scholar] [CrossRef]
- Philit, F.; Etienne-Mastroianni, B.; Parrot, A.; Guerin, C.; Robert, D.; Cordier, J.F. Idiopathic acute eosinophilic pneumonia: a study of 22 patients. Am. J. Respir. Crit. Care Med. 2002, 166, 1235–1239. [Google Scholar] [CrossRef]
- Kanwal, R.; Kullman, G.; Piacitelli, C.; Boylstein, R.; Sahakian, N.; Martin, S.; Fedan, K.; Kreiss, K. Evaluation of flavorings-related lung disease risk at six microwave popcorn plants. J. Occup. Environ. Med. 2006, 48, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Kreiss, K.; Gomaa, A.; Kullman, G.; Fedan, K.; Simoes, E.J.; Enright, P.L. Clinical bronchiolitis obliterans in workers at a microwave-popcorn plant. N. Engl. J. Med. 2002, 347, 330–338. [Google Scholar] [CrossRef]
- Allen, J.G.; Flanigan, S.S.; LeBlanc, M.; Vallarino, J.; MacNaughton, P.; Stewart, J.H.; Christiani, D.C. Flavoring Chemicals in E-Cigarettes: Diacetyl, 2,3-Pentanedione, and Acetoin in a Sample of 51 Products, Including Fruit-, Candy-, and Cocktail-Flavored E-Cigarettes. Environ. Health Perspect. 2016, 124, 733–739. [Google Scholar] [CrossRef] [PubMed]
- Landman, S.T.; Dhaliwal, I.; Mackenzie, C.A.; Martinu, T.; Steele, A.; Bosma, K.J. Life-threatening bronchiolitis related to electronic cigarette use in a Canadian youth. Can. Med. Assoc. J. 2019, 191, E1321. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Latest Outbreak Information. Available online: https://www.cdc.gov/tobacco/basic_information/e-cigarettes/severe-lung-disease.html (accessed on 17 December 2019).
- The Official Website of the Government of Canada. Vaping-Associated Lung Illness. Available online: https://www.canada.ca/en/public-health/services/diseases/vaping-pulmonary-illness.html (accessed on 17 December 2019).
- Ghinai, I.; Navon, L.; Gunn, J.K.L.; Duca, L.M.; Brister, S.; Love, S.; Brink, R.; Fajardo, G.; Johnson, J.; Saathoff-Huber, L.; et al. Characteristics of Persons Who Report Using Only Nicotine-Containing Products Among Interviewed Patients with E-cigarette, or Vaping, Product Use-Associated Lung Injury-Illinois, August-December 2019. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 84–89. [Google Scholar] [CrossRef]
- Miller, G.J.; Beadnell, H.M.; Ashcroft, M.T. Diffuse pulmonary fibrosis and blackfat-tobacco smoking in Guyana. Lancet 1968, 2, 259–260. [Google Scholar] [CrossRef]
- Miller, G.J.; Ashcroft, M.T.; Beadnell, H.M.; Wagner, J.C.; Pepys, J. The lipoid pneumonia of blackfat tobacco smokers in Guyana. Q. J. Med. 1971, 40, 457–470. [Google Scholar]
- Imminent, O. Blackfat tobacco smoker’s lung. Br. Med. J. 1972, 12, 393. [Google Scholar]
- Layden, J.E.; Ghinai, I.; Pray, I.; Kimball, A.; Layer, M.; Tenforde, M.; Navon, L.; Hoots, B.; Salvatore, P.P.; Elderbrook, M.; et al. Pulmonary Illness Related to E-Cigarette Use in Illinois and Wisconsin — Preliminary Report. N. Engl. J. Med. 2019, 382, 903–916. [Google Scholar] [CrossRef]
- Maddock, S.D.; Cirulis, M.M.; Callahan, S.J.; Keenan, L.M.; Pirozzi, C.S.; Raman, S.M.; Aberegg, S.K. Pulmonary Lipid-Laden Macrophages and Vaping. N. Engl. J. Med. 2019, 381, 1488–1489. [Google Scholar] [CrossRef]
- Corwin, R.W.; Irwin, R.S. The lipid-laden alveolar macrophage as a marker of aspiration in parenchymal lung disease. Am. Rev. Respir. Dis. 1985, 132, 576–581. [Google Scholar] [PubMed]
- Taylor, J.; Wiens, T.; Peterson, J.; Saravia, S.; Lunda, M.; Hanson, K.; Wogen, M.; D’Heilly, P.; Margetta, J.; Bye, M.; et al. Characteristics of E-cigarette, or Vaping, Products Used by Patients with Associated Lung Injury and Products Seized by Law Enforcement-Minnesota, 2018 and 2019. MMWR Morb. Mortal. Wkly. Rep. 2019, 68, 1096–1100. [Google Scholar] [CrossRef] [PubMed]
- Blount, B.C.; Karwowski, M.P.; Shields, P.G.; Morel-Espinosa, M.; Valentin-Blasini, L.; Gardner, M.; Braselton, M.; Brosius, C.R.; Caron, K.T.; Chambers, D.; et al. Vitamin E Acetate in Bronchoalveolar-Lavage Fluid Associated with EVALI. N. Engl. J. Med. 2020, 382, 697–705. [Google Scholar] [CrossRef] [PubMed]
- Bhat, T.A.; Kalathil, S.G.; Bogner, P.N.; Blount, B.C.; Goniewicz, M.L.; Thanavala, Y.M. An Animal Model of Inhaled Vitamin E Acetate and EVALI-like Lung Injury. N. Engl. J. Med. 2020, 382, 1175–1177. [Google Scholar] [CrossRef] [PubMed]
- Osei, A.D.; Mirbolouk, M.; Orimoloye, O.A.; Dzaye, O.; Uddin, S.M.I.; Dardari, Z.A.; DeFilippis, A.P.; Bhatnagar, A.; Blaha, M.J. The association between e-cigarette use and asthma among never combustible cigarette smokers: behavioral risk factor surveillance system (BRFSS) 2016 & 2017. BMC Pulm. Med. 2019, 19, 180. [Google Scholar]
- Osei, A.D.; Mirbolouk, M.; Orimoloye, O.A.; Dzaye, O.; Uddin, S.M.I.; Benjamin, E.J.; Hall, M.E.; DeFilippis, A.P.; Bhatnagar, A.; Biswal, S.S.; et al. Association Between E-Cigarette Use and Chronic Obstructive Pulmonary Disease by Smoking Status: Behavioral Risk Factor Surveillance System 2016 and 2017. Am. J. Prev. Med. 2019, 58, 336–342. [Google Scholar] [CrossRef]
- Lozano, R.; Naghavi, M.; Foreman, K.; Lim, S.; Shibuya, K.; Aboyans, V.; Abraham, J.; Adair, T.; Aggarwal, R.; Ahn, S.Y.; et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2095–2128. [Google Scholar] [CrossRef]
- Yoshida, T.; Tuder, R.M. Pathobiology of cigarette smoke-induced chronic obstructive pulmonary disease. Physiol Rev. 2007, 87, 1047–1082. [Google Scholar] [CrossRef]
- Garcia-Arcos, I.; Geraghty, P.; Baumlin, N.; Campos, M.; Dabo, A.J.; Jundi, B.; Cummins, N.; Eden, E.; Grosche, A.; Salathe, M.; et al. Chronic electronic cigarette exposure in mice induces features of COPD in a nicotine-dependent manner. Thorax 2016, 71, 1119–1129. [Google Scholar] [CrossRef]
- Reinikovaite, V.; Rodriguez, I.E.; Karoor, V.; Rau, A.; Trinh, B.B.; Deleyiannis, F.W.; Taraseviciene-Stewart, L. The effects of electronic cigarette vapour on the lung: direct comparison to tobacco smoke. Eur. Respir. J. 2018, 51, 1701661. [Google Scholar] [CrossRef]
- Olfert, I.M.; DeVallance, E.; Hoskinson, H.; Branyan, K.W.; Clayton, S.; Pitzer, C.R.; Sullivan, D.P.; Breit, M.J.; Wu, Z.; Klinkhachorn, P.; et al. Chronic exposure to electronic cigarettes results in impaired cardiovascular function in mice. J. Appl. Physiol. 2018, 124, 573–582. [Google Scholar] [CrossRef] [PubMed]
- Polosa, R.; Morjaria, J.B.; Prosperini, U.; Russo, C.; Pennisi, A.; Puleo, R.; Caruso, M.; Caponnetto, P. Health effects in COPD smokers who switch to electronic cigarettes: a retrospective-prospective 3-year follow-up. Int. J. Chron. Obstruct. Pulmon. Dis. 2018, 13, 2533–2542. [Google Scholar] [CrossRef] [PubMed]
- Coogan, P.F.; Castro-Webb, N.; Yu, J.; O’Connor, G.T.; Palmer, J.R.; Rosenberg, L. Active and passive smoking and the incidence of asthma in the Black Women’s Health Study. Am. J. Respir. Crit. Care Med. 2015, 191, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Chapman, D.G.; Casey, D.T.; Ather, J.L.; Aliyeva, M.; Daphtary, N.; Lahue, K.G.; van der Velden, J.L.; Janssen-Heininger, Y.M.W.; Irvin, C.G. The Effect of Flavored E-cigarettes on Murine Allergic Airways Disease. Sci. Rep. 2019, 9, 13671. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.B.; Kim, S.H. Inhallation of e-Cigarette Cartridge Solution Aggravates Allergen-induced Airway Inflammation and Hyper-responsiveness in Mice. Toxicol. Res. 2014, 30, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.O.; Nonnemaker, J.M.; Bradfield, B.; Hensel, E.C.; Robinson, R.J. Examining Daily Electronic Cigarette Puff Topography Among Established and Nonestablished Cigarette Smokers in their Natural Environment. Nicotine Tob. Res. 2018, 20, 1283–1288. [Google Scholar] [CrossRef] [PubMed]
- St Helen, G.; Ross, K.C.; Dempsey, D.A.; Havel, C.M.; Jacob, P., 3rd; Benowitz, N.L. Nicotine Delivery and Vaping Behavior During ad Libitum E-cigarette Access. Tob. Regul. Sci. 2016, 2, 363–376. [Google Scholar] [CrossRef]
- Kyriakos, C.N.; Filippidis, F.T.; Hitchman, S.; Girvalaki, C.; Tzavara, C.; Demjen, T.; Fernandez, E.; Mons, U.; Trofor, A.; Tountas, Y.; et al. Characteristics and correlates of electronic cigarette product attributes and undesirable events during e-cigarette use in six countries of the EUREST-PLUS ITC Europe Surveys. Tob. Induc. Dis. 2018, 16, A1. [Google Scholar] [CrossRef]
- Sussan, T.E.; Gajghate, S.; Thimmulappa, R.K.; Ma, J.; Kim, J.H.; Sudini, K.; Consolini, N.; Cormier, S.A.; Lomnicki, S.; Hasan, F.; et al. Exposure to electronic cigarettes impairs pulmonary anti-bacterial and anti-viral defenses in a mouse model. PLoS ONE 2015, 10, e0116861. [Google Scholar] [CrossRef]
- Song, M.A.; Reisinger, S.A.; Freudenheim, J.L.; Brasky, T.M.; Mathe, E.A.; McElroy, J.P.; Nickerson, Q.A.; Weng, D.Y.; Wewers, M.D.; Shields, P.G. Effects of Electronic Cigarette Constituents on the Human Lung: A Pilot Clinical Trial. Cancer Prev. Res. 2020, 13, 145–152. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Traboulsi, H.; Cherian, M.; Abou Rjeili, M.; Preteroti, M.; Bourbeau, J.; Smith, B.M.; Eidelman, D.H.; Baglole, C.J. Inhalation Toxicology of Vaping Products and Implications for Pulmonary Health. Int. J. Mol. Sci. 2020, 21, 3495. https://doi.org/10.3390/ijms21103495
Traboulsi H, Cherian M, Abou Rjeili M, Preteroti M, Bourbeau J, Smith BM, Eidelman DH, Baglole CJ. Inhalation Toxicology of Vaping Products and Implications for Pulmonary Health. International Journal of Molecular Sciences. 2020; 21(10):3495. https://doi.org/10.3390/ijms21103495
Chicago/Turabian StyleTraboulsi, Hussein, Mathew Cherian, Mira Abou Rjeili, Matthew Preteroti, Jean Bourbeau, Benjamin M. Smith, David H. Eidelman, and Carolyn J. Baglole. 2020. "Inhalation Toxicology of Vaping Products and Implications for Pulmonary Health" International Journal of Molecular Sciences 21, no. 10: 3495. https://doi.org/10.3390/ijms21103495
APA StyleTraboulsi, H., Cherian, M., Abou Rjeili, M., Preteroti, M., Bourbeau, J., Smith, B. M., Eidelman, D. H., & Baglole, C. J. (2020). Inhalation Toxicology of Vaping Products and Implications for Pulmonary Health. International Journal of Molecular Sciences, 21(10), 3495. https://doi.org/10.3390/ijms21103495