Dynamic Changes of Heart Failure Biomarkers in Response to Parabolic Flight
Abstract
1. Introduction
2. Results
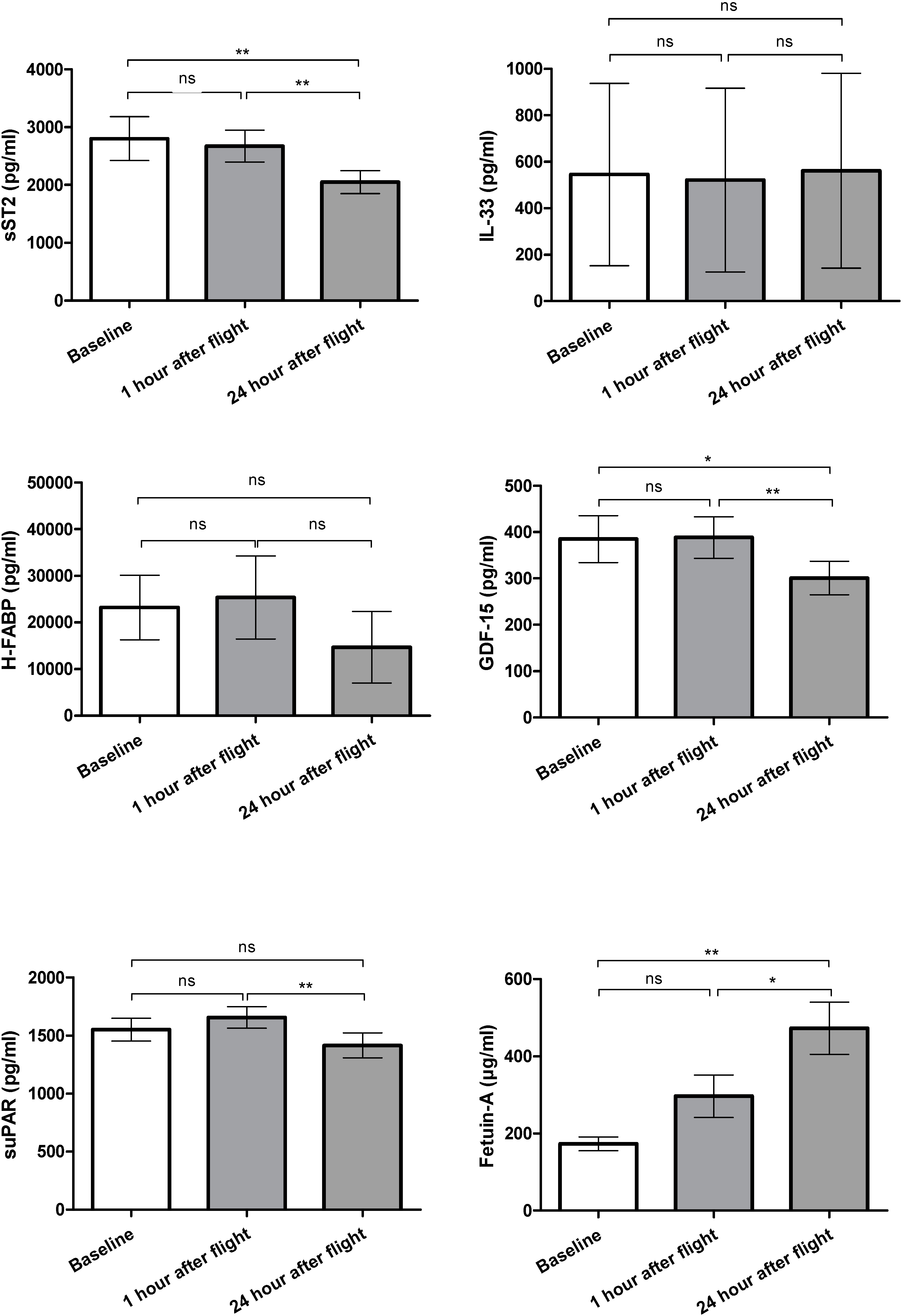
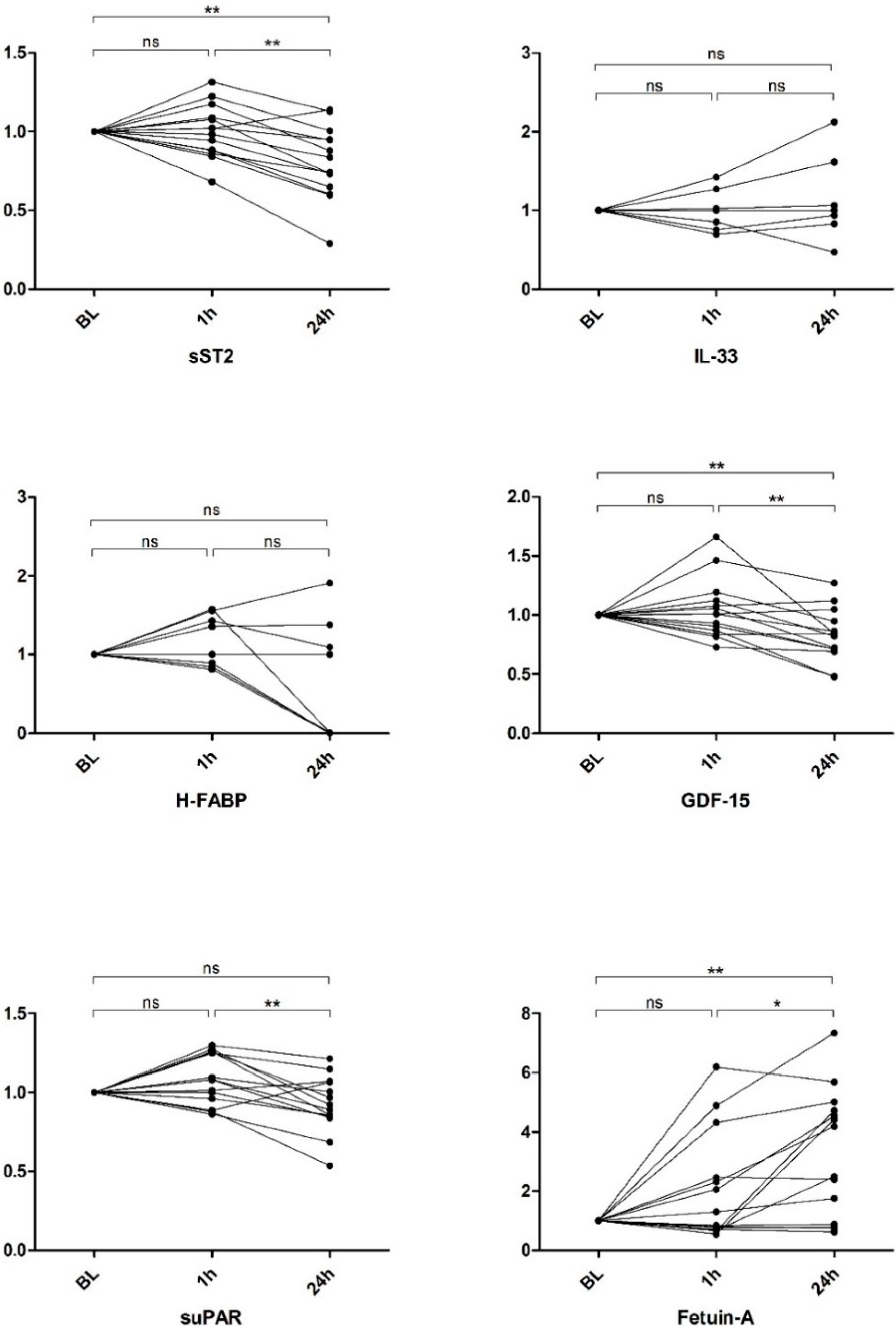
2.1. Biomarker Levels
2.1.1. sST2
2.1.2. IL-33
2.1.3. H-FABP
2.1.4. GDF-15
2.1.5. suPAR
2.1.6. Fetuin-A
2.2. Correlation Analysis
3. Discussion
4. Materials and Methods
4.1. Participants
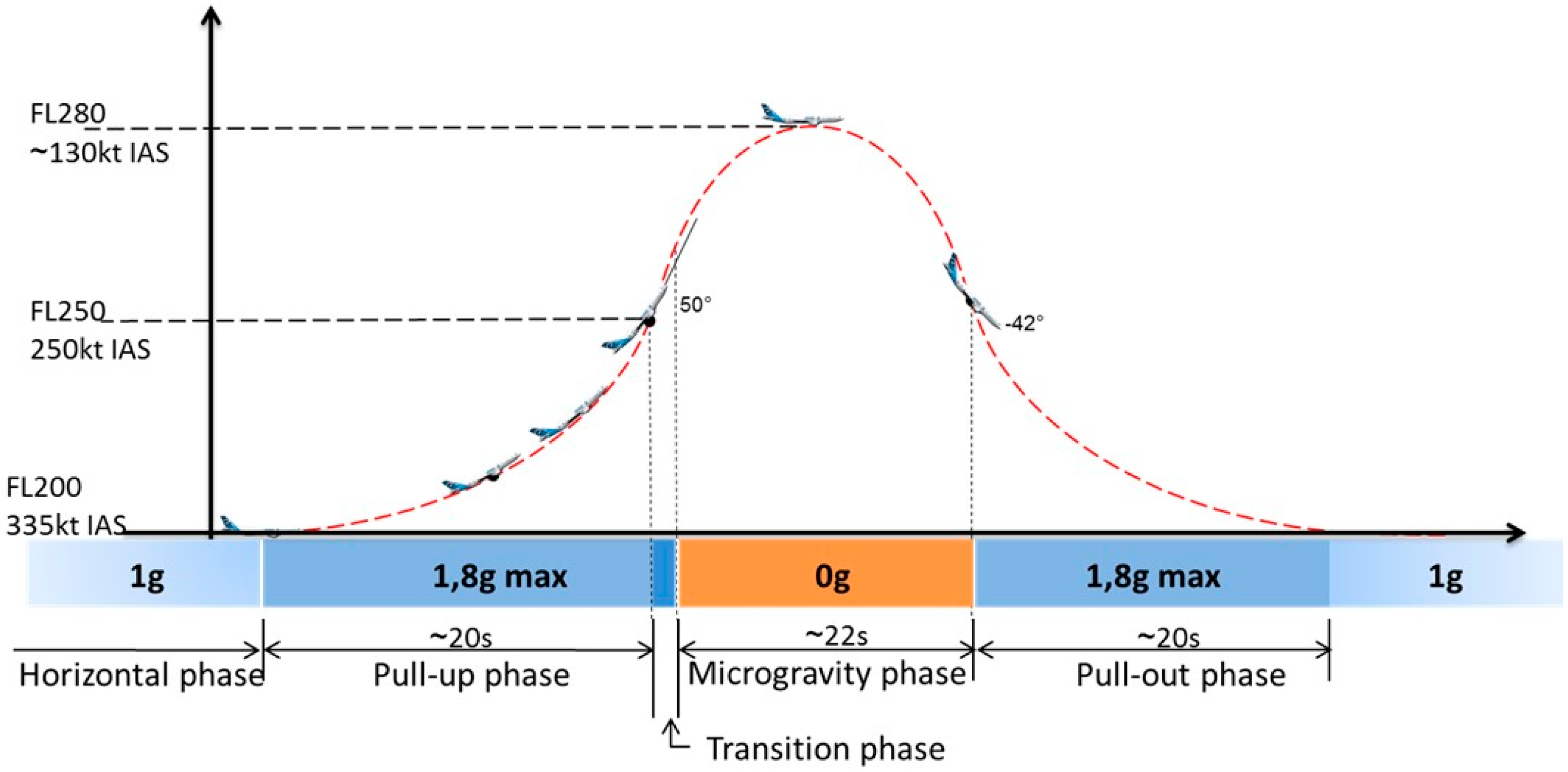
4.2. Parabolic Flight
4.3. Experimental Set-Up
4.4. Statistical Analysis
5. Conclusions
6. Limitations
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CKD | chronic kidney disease |
| DLR | Deutsches Zentrum für Luft- und Raumfahrt |
| ELISA | enzyme-linked immunosorbent assay |
| g | gravitational force |
| GDF-15 | growth differentiation factor-15 |
| H-FABP | heart-type fatty acid binding protein |
| IL-33 | interleukin-33 |
| NASA | National Aeronautics and Space Administration |
| PFC | parabolic flight campaign |
| sST2 | soluble suppression of tumorigenicity 2 |
| suPAR | soluble urokinase-type plasminogen activator receptor |
References
- Stepanek, J.; Blue, R.S.; Parazynski, S. Space Medicine in the Era of Civilian Spaceflight. N. Engl. J. Med. 2019, 380, 1053–1060. [Google Scholar] [CrossRef] [PubMed]
- Hughson, R.L.; Shoemaker, J.K.; Blaber, A.P.; Arbeille, P.; Greaves, D.K.; Pereira-Junior, P.P.; Xu, D. Cardiovascular regulation during long-duration spaceflights to the International Space Station. J. Appl. Physiol. 1985, 112, 719–727. [Google Scholar] [CrossRef] [PubMed]
- Shen, M.; Frishman, W.H. Effects of Spaceflight on Cardiovascular Physiology and Health. Cardiol. Rev. 2019, 27, 122–126. [Google Scholar] [CrossRef] [PubMed]
- Lathers, C.M.; Charles, J.B.; Elton, K.F.; Holt, T.A.; Mukai, C.; Bennett, B.S.; Bungo, M.W. Acute hemodynamic responses to weightlessness in humans. J. Clin. Pharmacol. 1989, 29, 615–627. [Google Scholar] [CrossRef] [PubMed]
- Convertino, V.A. Status of cardiovascular issues related to space flight: Implications for future research directions. Respir. Physiol. Neurobiol. 2009, 169, 19. [Google Scholar] [CrossRef]
- Blaber, A.P.; Zuj, K.A.; Goswami, N. Cerebrovascular autoregulation: Lessons learned from spaceflight research. Eur. J. Appl. Physiol. 2013, 113, 1909–1917. [Google Scholar] [CrossRef]
- Cervantes, J.L.; Hong, B.Y. Dysbiosis and Immune Dysregulation in Outer Space. Int. Rev. Immunol. 2016, 35, 67–82. [Google Scholar] [CrossRef]
- Iskovitz, I.; Kassemi, M.; Thomas, J.D. Impact of weightlessness on cardiac shape and left ventricular stress/strain distributions. J. Biomech. Eng. 2013, 135, 4025464. [Google Scholar] [CrossRef]
- Pietsch, J.; Bauer, J.; Egli, M.; Infanger, M.; Wise, P.; Ulbrich, C.; Grimm, D. The effects of weightlessness on the human organism and mammalian cells. Curr. Mol. Med. 2011, 11, 350–364. [Google Scholar] [CrossRef]
- Bimpong-Buta, N.Y.; Jirak, P.; Wernly, B.; Lichtenauer, M.; Knost, T.; Abusamrah, T.; Kelm, M.; Jung, C. Blood parameter analysis after short term exposure to weightlessness in parabolic flight. Clin. Hemorheol. Microcirc. 2018, 70, 477–486. [Google Scholar] [CrossRef]
- Savic-Radojevic, A.; Pljesa-Ercegovac, M.; Matic, M.; Simic, D.; Radovanovic, S.; Simic, T. Novel Biomarkers of Heart Failure. Adv. Clin. Chem. 2017, 79, 93–152. [Google Scholar] [PubMed]
- Ciccone, M.M.; Cortese, F.; Gesualdo, M.; Riccardi, R.; Di Nunzio, D.; Moncelli, M.; Iacoviello, M.; Scicchitano, P. A novel cardiac bio-marker: ST2: A review. Molecules 2013, 18, 15314–15328. [Google Scholar] [CrossRef]
- Ye, X.D.; He, Y.; Wang, S.; Wong, G.T.; Irwin, M.G.; Xia, Z. Heart-type fatty acid binding protein (H-FABP) as a biomarker for acute myocardial injury and long-term post-ischemic prognosis. Acta Pharmacol. Sin. 2018, 39, 1155–1163. [Google Scholar] [CrossRef] [PubMed]
- Wollert, K.C.; Kempf, T.; Wallentin, L. Growth Differentiation Factor 15 as a Biomarker in Cardiovascular Disease. Clin. Chem. 2017, 63, 140–151. [Google Scholar] [CrossRef] [PubMed]
- Eugen-Olsen, J.; Giamarellos-Bourboulis, E.J. suPAR: The unspecific marker for disease presence, severity and prognosis. Int. J. Antimicrob. Agents 2015, 46, 31. [Google Scholar] [CrossRef] [PubMed]
- Jirak, P.; Stechemesser, L.; More, E.; Franzen, M.; Topf, A.; Mirna, M.; Paar, V.; Pistulli, R.; Kretzschmar, D.; Wernl, B.; et al. Clinical implications of fetuin-A. Adv. Clin. Chem. 2019, 89, 79–130. [Google Scholar] [PubMed]
- Dieplinger, B.; Mueller, T. Soluble ST2 in heart failure. Clin. Chim. Acta 2015, 443, 57–70. [Google Scholar] [CrossRef]
- Caiani, E.G.; Weinert, L.; Lang, R.M.; Vaida, P. The role of echocardiography in the assessment of cardiac function in weightlessness-Our experience during parabolic flights. Respir. Physiol. Neurobiol. 2009, 169, 16. [Google Scholar] [CrossRef]
- Norsk, P.; Asmar, A.; Damgaard, M.; Christensen, N.J. Fluid shifts, vasodilatation and ambulatory blood pressure reduction during long duration spaceflight. J. Physiol. 2015, 593, 573–584. [Google Scholar] [CrossRef]
- Kakkar, R.; Lee, R.T. The IL-33/ST2 pathway: Therapeutic target and novel biomarker. Nat. Rev. Drug Discov. 2008, 7, 827–840. [Google Scholar] [CrossRef]
- Schmitz, J.; Owyang, A.; Oldham, E.; Song, Y.; Murphy, E.; McClanahan, T.K.; Zurawski, G.; Moshrefi, M.; Qin, J.; Li, X.; et al. IL-33, an interleukin-1-like cytokine that signals via the IL-1 receptor-related protein ST2 and induces T helper type 2-associated cytokines. Immunity 2005, 23, 479–490. [Google Scholar] [CrossRef] [PubMed]
- Cayrol, C.; Girard, J.P. Interleukin-33 (IL-33): A nuclear cytokine from the IL-1 family. Immunol. Rev. 2018, 281, 154–168. [Google Scholar] [CrossRef] [PubMed]
- Griesenauer, B.; Paczesny, S. The ST2/IL-33 Axis in Immune Cells during Inflammatory Diseases. Front. Immunol. 2017, 8. [Google Scholar] [CrossRef]
- Otaki, Y.; Watanabe, T.; Kubota, I. Heart-type fatty acid-binding protein in cardiovascular disease: A systemic review. Clin. Chim. Acta 2017, 474, 44–53. [Google Scholar] [CrossRef]
- Min, K.W.; Liggett, J.L.; Silva, G.; Wu, W.W.; Wang, R.; Shen, R.F.; Elin, T.E.; Baek, S.J. NAG-1/GDF15 accumulates in the nucleus and modulates transcriptional regulation of the Smad pathway. Oncogene 2016, 35, 377–388. [Google Scholar] [CrossRef] [PubMed]
- Thuno, M.; Macho, B.; Eugen-Olsen, J. suPAR: The molecular crystal ball. Dis. Markers 2009, 27, 157–172. [Google Scholar] [CrossRef] [PubMed]
- Kawauchi, T.; Ishimaru, K.; Nakamura, Y.; Nakano, N.; Hara, M.; Ogawa, H.; Okumura, K.; Shibata, S.; Nakao, A. Clock-dependent temporal regulation of IL-33/ST2-mediated mast cell response. Allergol. Int. 2017, 66, 472–478. [Google Scholar] [CrossRef]
- Pelsers, M.M.; Chapelle, J.P.; Knapen, M.; Vermeer, C.; Muijtjens, A.M.; Hermens, W.T. Influence of age and sex and day-to-day and within-day biological variation on plasma concentrations of fatty acid-binding protein and myoglobin in healthy subjects. Clin. Chem. 1999, 45, 441–443. [Google Scholar]
- Tsai, V.W.; Macia, L.; Feinle-Bisset, C.; Manandhar, R.; Astrup, A.; Raben, A.; Lorenze, J.K.; Schmidt, P.T.; Wiklund, F.; Pedersen, N.L.; et al. Serum Levels of Human MIC-1/GDF15 Vary in a Diurnal Pattern, Do Not Display a Profile Suggestive of a Satiety Factor and Are Related to BMI. PLoS ONE 2015, 10, e0133362. [Google Scholar] [CrossRef]
- Andersen, O.; Eugen-Olsen, J.; Kofoed, K.; Iversen, J.; Haugaard, S.B. Soluble urokinase plasminogen activator receptor is a marker of dysmetabolism in HIV-infected patients receiving highly active antiretroviral therapy. J. Med. Virol. 2008, 80, 209–216. [Google Scholar] [CrossRef]
- Chappuis, S.; Ripperger, J.A.; Schnell, A.; Rando, G.; Jud, C.; Wahli, W.; Albrecht, U. Role of the circadian clock gene Per2 in adaptation to cold temperature. Mol. Metab. 2013, 2, 184–193. [Google Scholar] [CrossRef]
- Crnko, S.; Printezi, M.I.; Jansen, T.P.J.; Leiteris, L.; van der Meer, M.G.; Schutte, H.; van Faassen, M.; du Bre, B.C.; de Jonge, N.; Asselsberg, F.W.; et al. Prognostic biomarker soluble ST2 exhibits diurnal variation in chronic heart failure patients. ESC Heart Fail. 2020, 31, 12673. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.J.; Thakkar, B.; Chamberland, J.P.; Mantzoros, C.S. Circulating fetuin-A levels are not affected by short and long-term energy deprivation and/or by leptin administration. Metabolism 2014, 63, 754–759. [Google Scholar] [CrossRef]
- Sponder, M.; Lichtenauer, M.; Wernly, B.; Paar, V.; Hoppe, U.; Emich, M.; Fritzer-Szekeres, M.; Litschauer, B.; Strametz-Juranek, J. Serum heart-type fatty acid-binding protein decreases and soluble isoform of suppression of tumorigenicity 2 increases significantly by long-term physical activity. J. Investig. Med. 2019, 67, 833–840. [Google Scholar] [CrossRef] [PubMed]
- Masyuk, M.; Wernly, B.; Lichtenauer, M.; Franz, M.; Kabisch, B.; Muessig, J.M.; Zimmermann, G.; Lauten, A.; Schulze, P.C.; Hoppe, U.C.; et al. Prognostic relevance of serum lactate kinetics in critically ill patients. Intensive Care Med. 2019, 45, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Bimpong-Buta, N.Y.; Jirak, P.; Wernly, B.; Lichtenauer, M.; Masyuk, M.; Muessig, J.M.; Braun, K.; Kaya, S.; Kelm, M.; Jung, C. Analysis of human microcirculation in weightlessness: Study protocol and pre-study experiments. Clin. Hemorheol. Microcirc. 2018, 14. [Google Scholar] [CrossRef]
- Shelhamer, M. Parabolic flight as a spaceflight analog. J. Appl. Physiol. 1985, 120, 1442–1448. [Google Scholar] [CrossRef]
| n | Sex | Age (y) | Height (m) | Weight (kg) | BMI (kg/m2) | BSA (m2) | BP Systolic (mmHg) | BP Mean (mmHg) | BP Diastolic (mmHg) | Heart Rate (bpm) |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | M | 40 | 1.76 | 93 | 30 | 2.13 | 128 | 91 | 69 | 77 |
| 2 | F | 30 | 1.62 | 52 | 20 | 1.52 | 96 | 87 | 67 | 78 |
| 3 | M | 22 | 1.88 | 86 | 24 | 2.11 | 131 | 94 | 78 | 103 |
| 4 | M | 28 | 1.83 | 83 | 25 | 2.05 | 103 | 84 | 70 | 95 |
| 5 | M | 23 | 1.93 | 92 | 25 | 2.22 | 111 | 93 | 82 | 51 |
| 6 | M | 29 | 1.91 | 82 | 22 | 2.08 | 107 | 97 | 79 | 58 |
| 7 | F | 23 | 1.64 | 54 | 20 | 1.56 | 109 | 102 | 95 | 95 |
| 8 | F | 25 | 1.72 | 75 | 25 | 1.89 | 109 | 92 | 77 | 69 |
| 9 | F | 30 | 1.70 | 62 | 21 | 1.71 | 112 | 90 | 80 | 80 |
| 10 | M | 31 | 1.77 | 78 | 25 | 1.95 | 106 | 87 | 76 | 55 |
| 11 | F | 24 | 1.73 | 63 | 21 | 1.74 | 124 | 94 | 81 | 82 |
| 12 | M | 37 | 1.92 | 91 | 25 | 2.20 | 128 | 83 | 54 | 107 |
| 13 | M | 31 | 1.82 | 86 | 26 | 2.10 | 126 | 99 | 86 | 87 |
| 14 | F | 31 | 1.79 | 76 | 24 | 1.90 | 108 | 85 | 73 | 78 |
| Mean | m = 8 | 28.9 | 1.79 | 76.6 | 23.8 | 1.9 | 114.1 | 91.3 | 76.2 | 79.6 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jirak, P.; Wernly, B.; Lichtenauer, M.; Paar, V.; Franz, M.; Knost, T.; Abusamrah, T.; Kelm, M.; Muessig, J.M.; Bimpong-Buta, N.-Y.; et al. Dynamic Changes of Heart Failure Biomarkers in Response to Parabolic Flight. Int. J. Mol. Sci. 2020, 21, 3467. https://doi.org/10.3390/ijms21103467
Jirak P, Wernly B, Lichtenauer M, Paar V, Franz M, Knost T, Abusamrah T, Kelm M, Muessig JM, Bimpong-Buta N-Y, et al. Dynamic Changes of Heart Failure Biomarkers in Response to Parabolic Flight. International Journal of Molecular Sciences. 2020; 21(10):3467. https://doi.org/10.3390/ijms21103467
Chicago/Turabian StyleJirak, Peter, Bernhard Wernly, Michael Lichtenauer, Vera Paar, Marcus Franz, Thorben Knost, Thaer Abusamrah, Malte Kelm, Johanna M. Muessig, Nana-Yaw Bimpong-Buta, and et al. 2020. "Dynamic Changes of Heart Failure Biomarkers in Response to Parabolic Flight" International Journal of Molecular Sciences 21, no. 10: 3467. https://doi.org/10.3390/ijms21103467
APA StyleJirak, P., Wernly, B., Lichtenauer, M., Paar, V., Franz, M., Knost, T., Abusamrah, T., Kelm, M., Muessig, J. M., Bimpong-Buta, N.-Y., & Jung, C. (2020). Dynamic Changes of Heart Failure Biomarkers in Response to Parabolic Flight. International Journal of Molecular Sciences, 21(10), 3467. https://doi.org/10.3390/ijms21103467







