Rubrofusarin Attenuates Chronic Restraint Stress-Induced Depressive Symptoms
Abstract
1. Introduction
2. Results
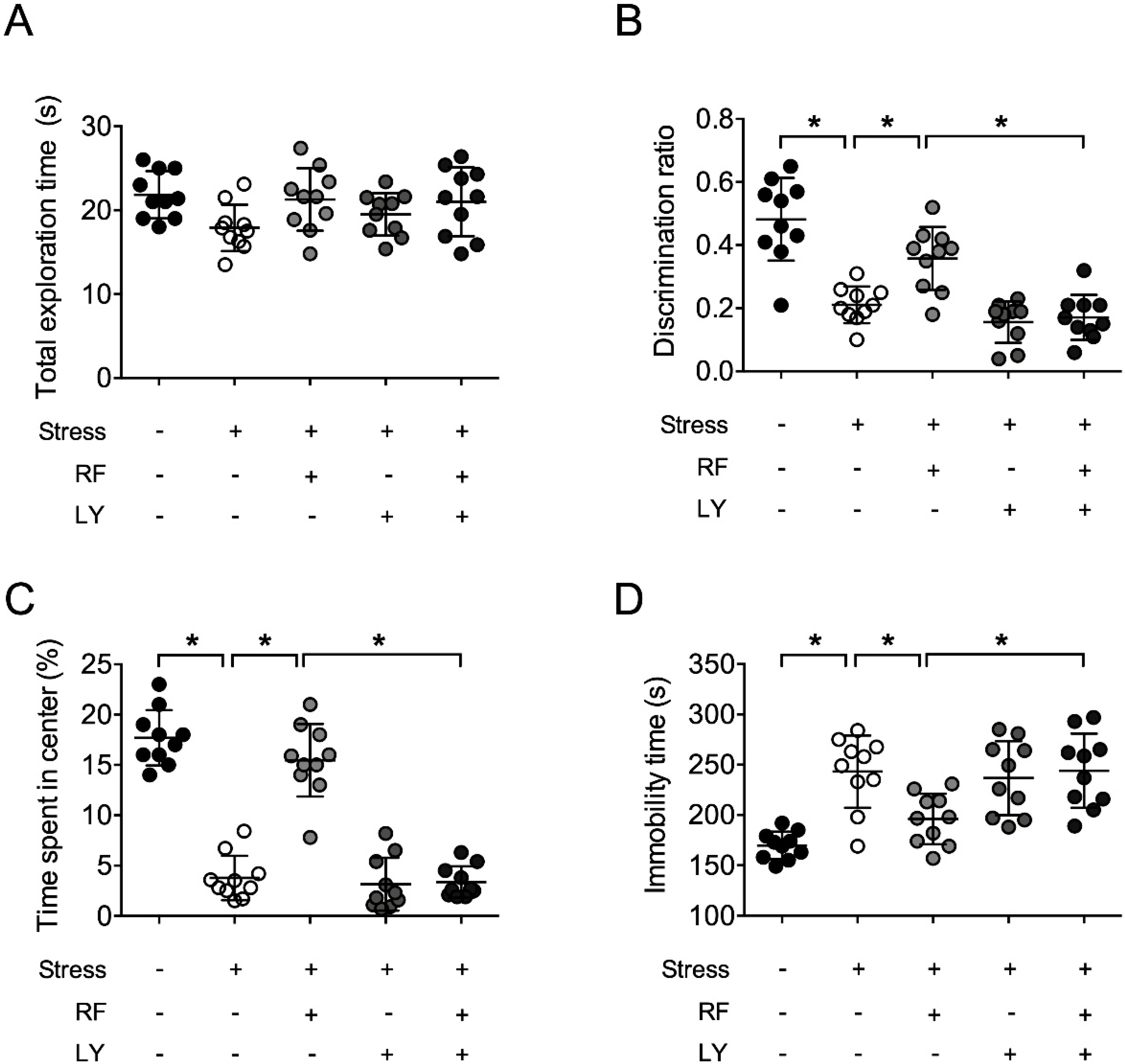
2.1. Chronic Rubrofusarin Administration Ameliorated Chronic Restraint Stress (CRS)-Induced Depressive Symptoms
2.2. Chronic Rubrofusarin Administration Rescued CRS-Induced Hippocampal Neuronal Damage
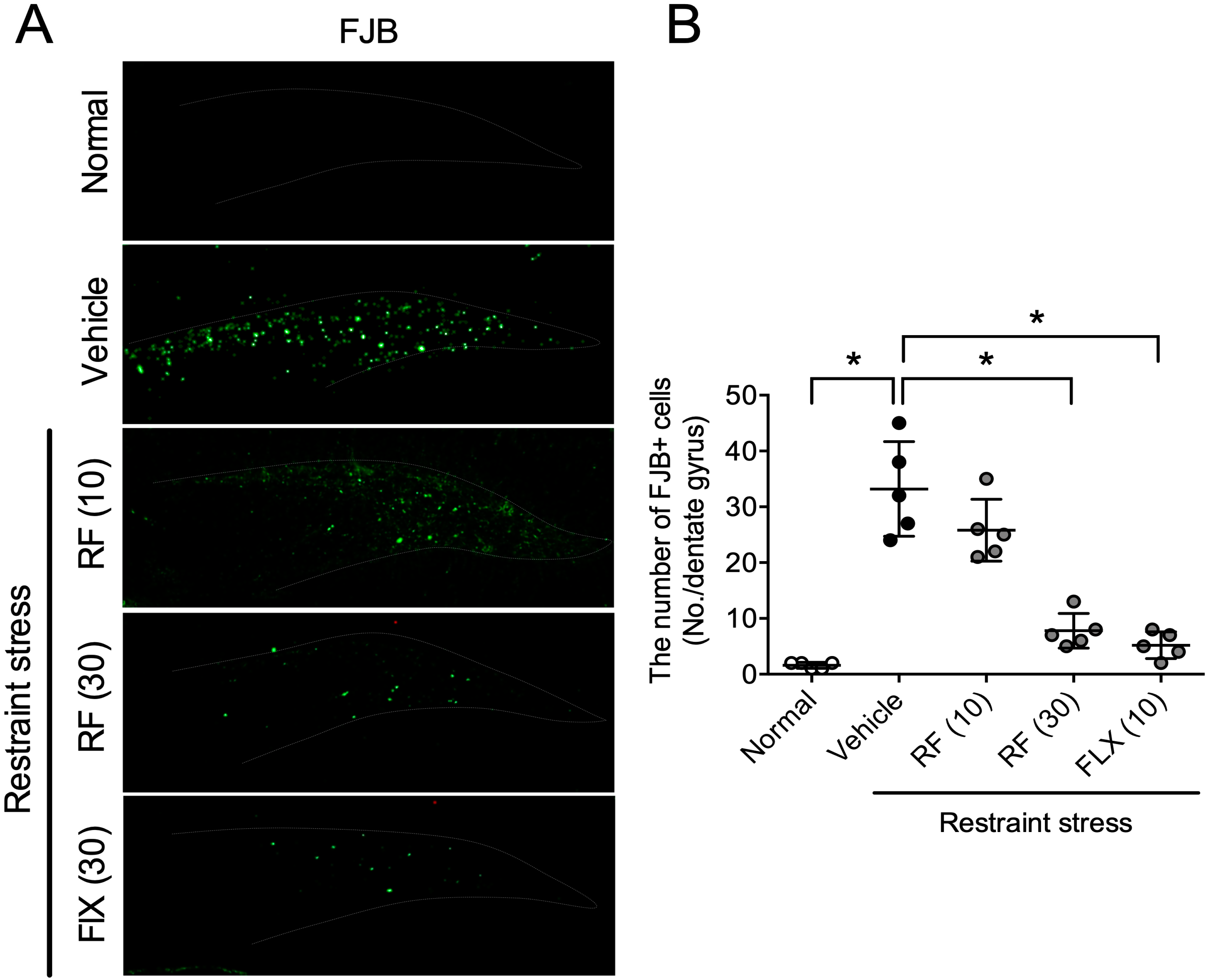
2.3. Chronic Rubrofusarin Administration Blocked CRS-Induced Reduction of Adult Neurogenesis
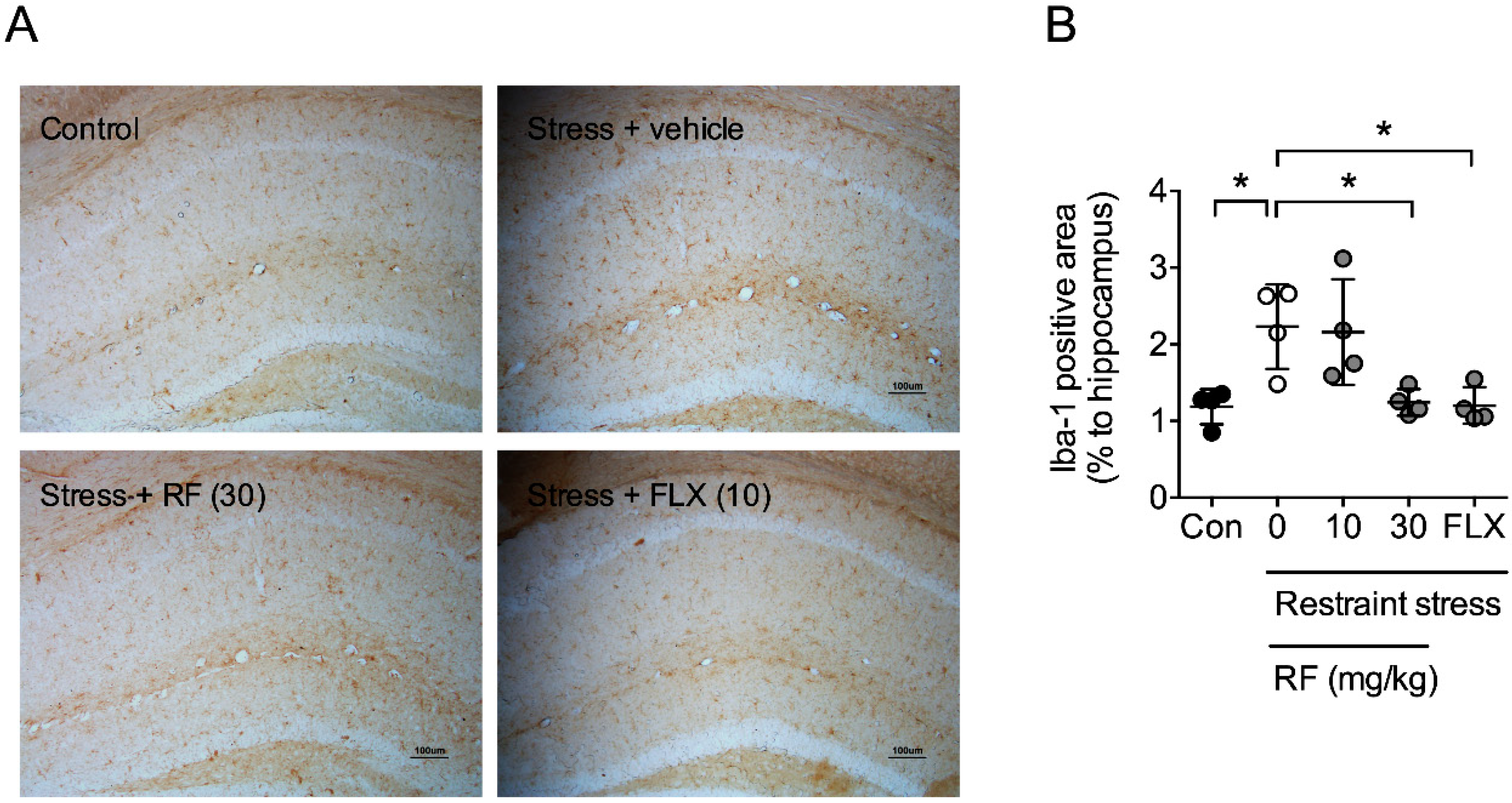
2.4. Chronic Rubrofusarin Administration Blocked CRS-Induced Neuroinflammation
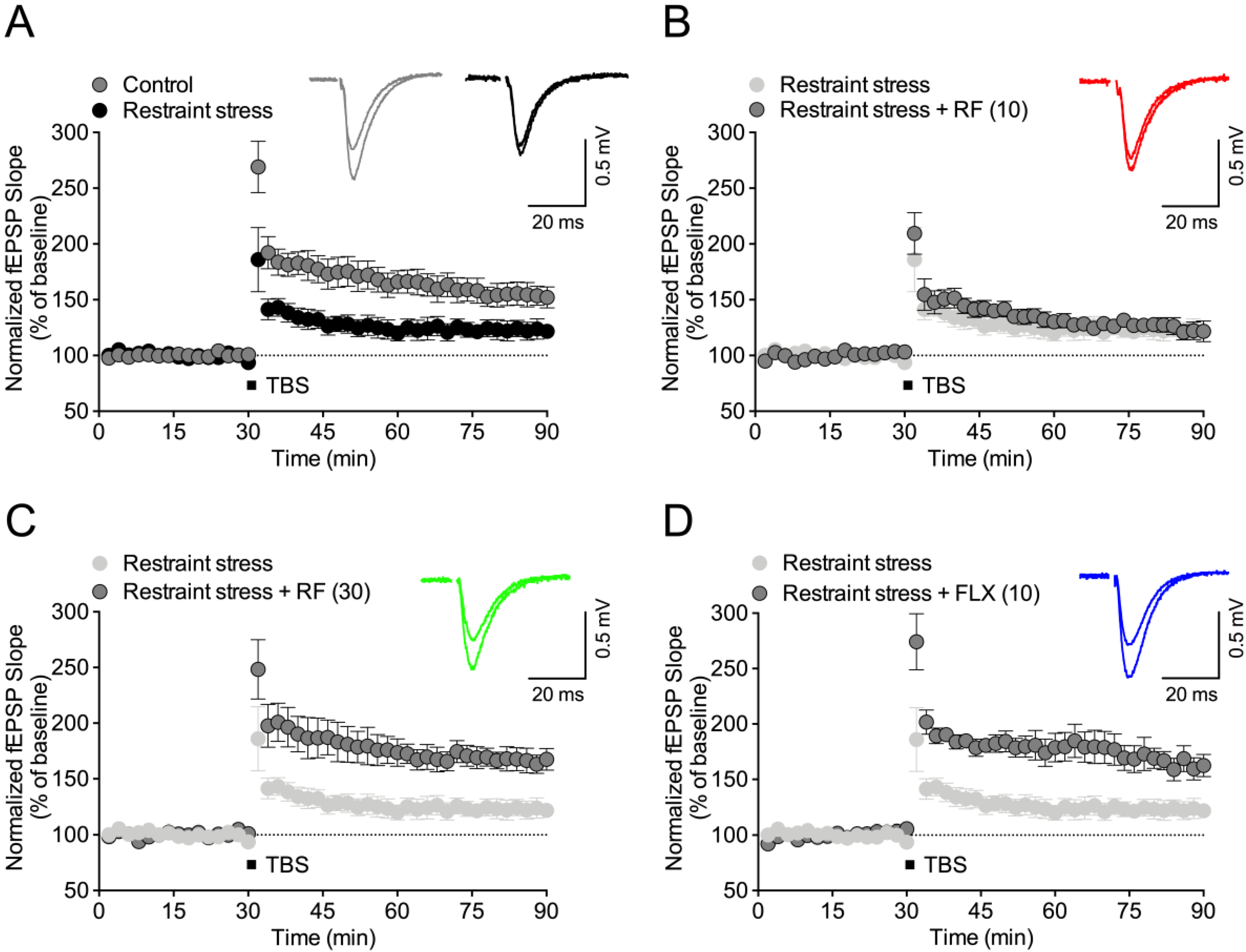
2.5. Chronic Rubrofusarin Administration Blocked CRS-Induced Synaptic Dysfunctions
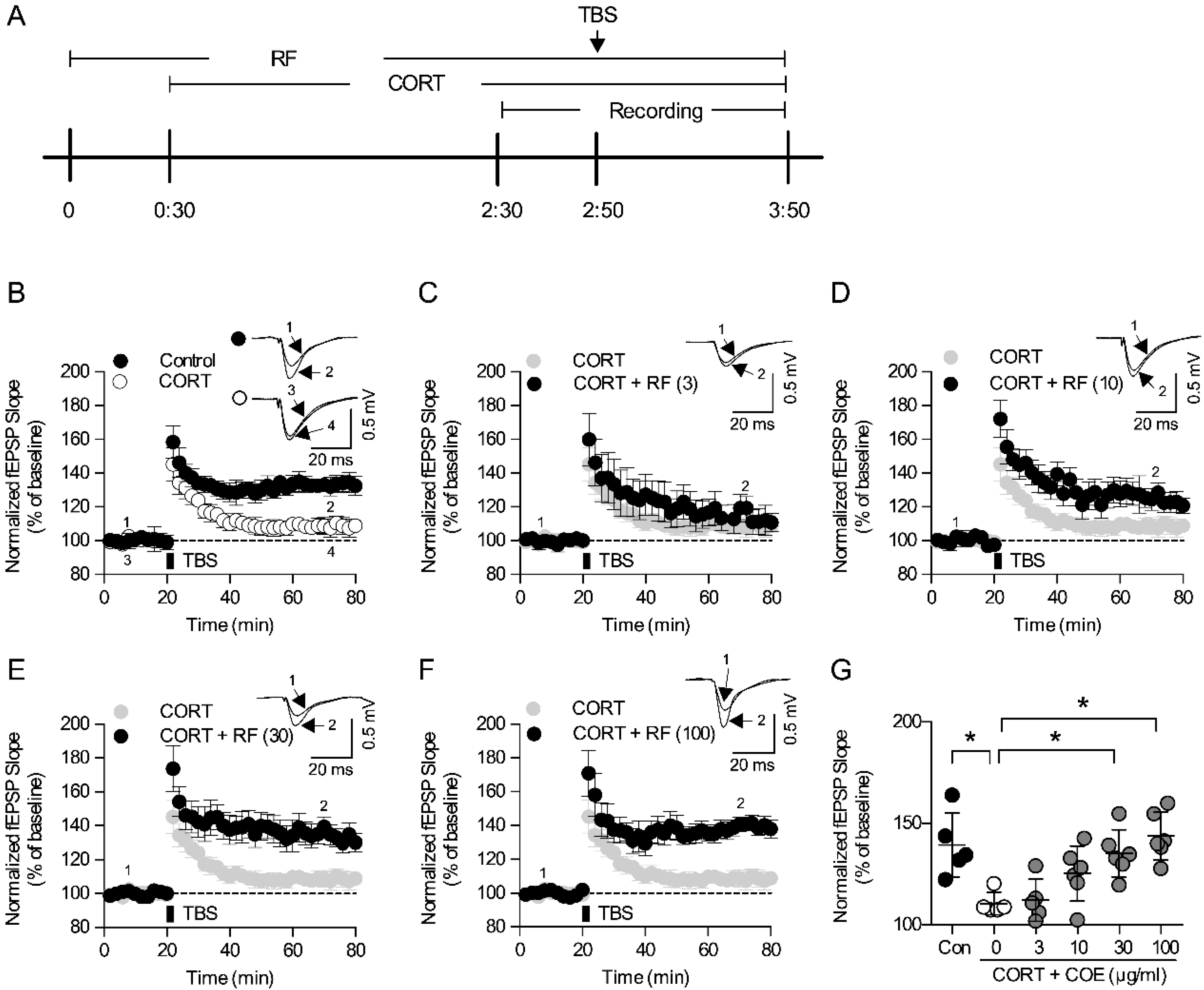
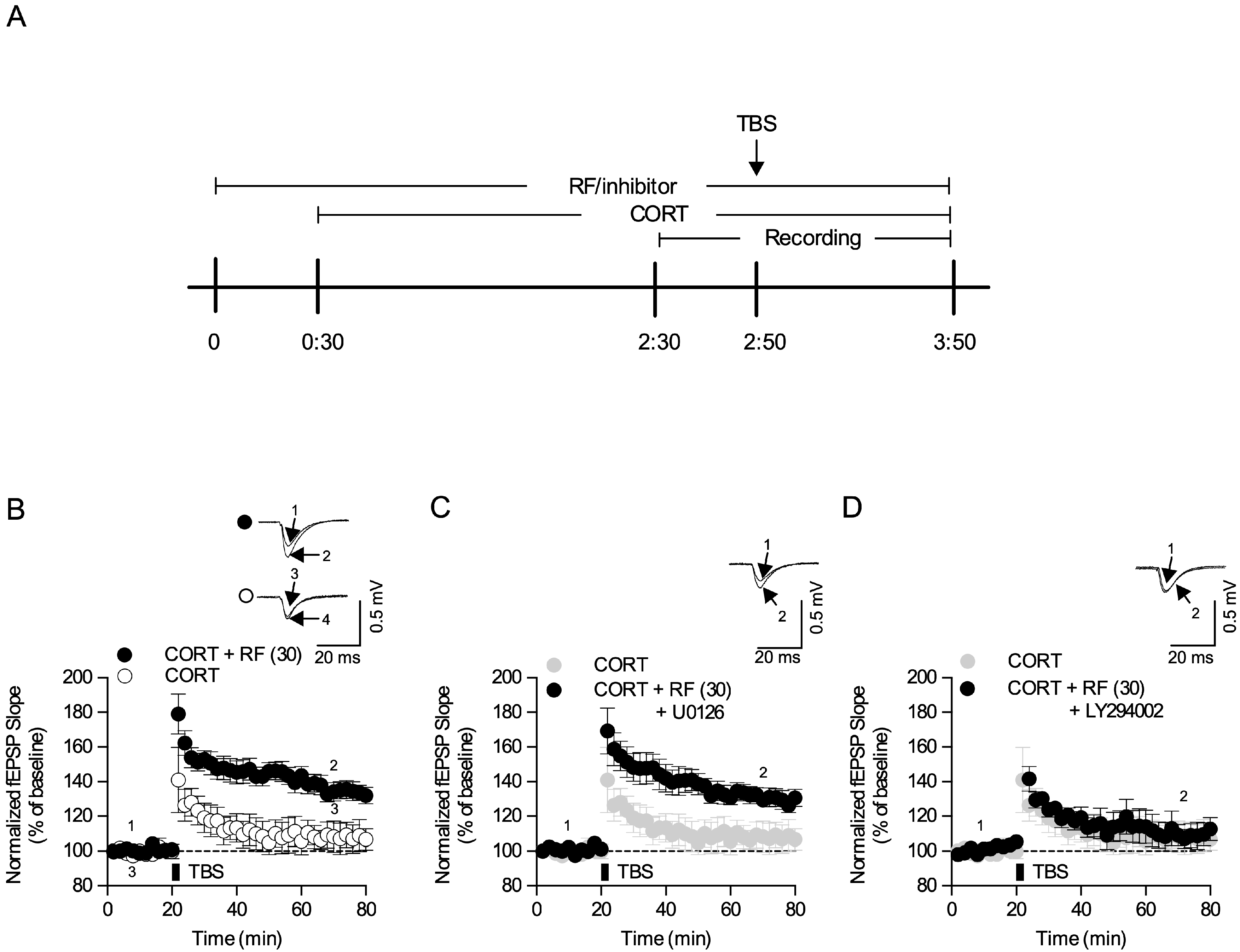
2.6. Rubrofusarin Blocked Corticosterone-Induced LTP Impairment Through Regulating the Akt Signaling
2.7. Akt Signaling Was Required for the Antidepressive Effect of Rubrofusarin
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Restraint Stress
4.3. Objective Recognition Memory Test
4.4. Forced Swimming Test
4.5. Tissue Slices Preparation
4.6. Floro-Jade B Staining
4.7. Immunohistochemistry
4.8. Hippocampal Slices Preparation and Electrophysiology
4.9. Microinfusion of Drugs
4.10. Statistics
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| CRS | Chronic restraint stress |
| REDD1 | Regulated in development and DNA damage responses 1 |
| NLRP3 | Nod-like receptor family pyrin domain containing 3 |
| GSK-3 | Glycogen synthase kinase-3 |
| PI3K | Phosphoinositide 3-kinases |
| ACSF | Artificial cerebrospinal fluid |
| fEPSP | Field excitatory postsynaptic potential |
| TBS | Theta burst stimulation |
| LTP | Long-term potentiation |
| RF | Rubrofusarin |
| FLX | Fluoxetine |
| DG | Dentate gyrus |
| CORT | Corticosterone |
| NMDAR | N-methyl-d-aspartate receptor |
| GR | Glucocorticoid receptor |
References
- Pardon, M.C.; Gould, G.G.; Garcia, A.; Phillips, L.; Cook, M.C.; Miller, S.A.; Mason, P.A.; Morilak, D.A. Stress reactivity of the brain noradrenergic system in three rat strains differing in their neuroendocrine and behavioral responses to stress: Implications for susceptibility to stress-related neuropsychiatric disorders. Neuroscience 2002, 115, 229–242. [Google Scholar] [CrossRef]
- Hall, B.S.; Moda, R.N.; Liston, C. Glucocorticoid Mechanisms of Functional Connectivity Changes in Stress-Related Neuropsychiatric Disorders. Neurobiol. Stress 2015, 1, 174–183. [Google Scholar] [CrossRef] [PubMed]
- Dunn, A.J.; Swiergiel, A.H.; Palamarchouk, V. Brain circuits involved in corticotropin-releasing factor-norepinephrine interactions during stress. Ann. N. Y. Acad. Sci. 2004, 1018, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Bremner, J.D. Traumatic stress: Effects on the brain. Dialogues Clin. Neurosci. 2006, 8, 445–461. [Google Scholar] [PubMed]
- Kashihara, K.; Maruyama, T.; Murota, M.; Nakahara, Y. Positive effects of acute and moderate physical exercise on cognitive function. J. Physiol. Anthropol. 2009, 28, 155–164. [Google Scholar] [CrossRef]
- Pittenger, C.; Duman, R.S. Stress, depression, and neuroplasticity: A convergence of mechanisms. Neuropsychopharmacology 2008, 33, 88–109. [Google Scholar] [CrossRef]
- Burke, H.M.; Davis, M.C.; Otte, C.; Mohr, D.C. Depression and cortisol responses to psychological stress: A meta-analysis. Psychoneuroendocrinology 2005, 30, 846–856. [Google Scholar] [CrossRef]
- Bruno, A.; Celebre, L.; Mento, C.; Rizzo, A.; Silvestri, M.C.; De Stefano, R.; Zoccali, R.A.; Muscatello, M.R.A. When Fathers Begin to Falter: A Comprehensive Review on Paternal Perinatal Depression. Int. J. Environ. Res. Public Health 2020, 17, 1139. [Google Scholar] [CrossRef]
- Thomson, K.C.; Romaniuk, H.; Greenwood, C.J.; Letcher, P.; Spry, E.; Macdonald, J.A.; McAnally, H.M.; Youssef, G.J.; McIntosh, J.; Hutchinson, D.; et al. Adolescent antecedents of maternal and paternal perinatal depression: A 36-year prospective cohort. Psychol. Med. 2020, 1–8. [Google Scholar] [CrossRef]
- Ossowska, G.; Nowa, G.; Kata, R.; Klenk-Majewska, B.; Danilczuk, Z.; Zebrowska-Lupina, I. Brain monoamine receptors in a chronic unpredictable stress model in rats. J. Neural Transm. 2001, 108, 311–319. [Google Scholar] [CrossRef]
- Hamon, M.; Blier, P. Monoamine neurocircuitry in depression and strategies for new treatments. Prog. Neuropsychopharmacol. Biol. Psychiatry 2013, 45, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Cascade, E.; Kalali, A.H.; Kennedy, S.H. Real-World Data on SSRI Antidepressant Side Effects. Psychiatry (Edgmont) 2009, 6, 16–18. [Google Scholar]
- Ferguson, J.M. SSRI Antidepressant Medications: Adverse Effects and Tolerability. Prim Care Companion J. Clin. Psychiatry 2001, 3, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Cartwright, C.; Gibson, K.; Read, J.; Cowan, O.; Dehar, T. Long-term antidepressant use: Patient perspectives of benefits and adverse effects. Patient Prefer. Adherence 2016, 10, 1401–1407. [Google Scholar] [CrossRef]
- Delgado, P.L. Depression: The case for a monoamine deficiency. J. Clin. Psychiatry 2000, 61 (Suppl. 6), 7–11. [Google Scholar] [PubMed]
- Bottiglieri, T.; Laundy, M.; Crellin, R.; Toone, B.K.; Carney, M.W.; Reynolds, E.H. Homocysteine, folate, methylation, and monoamine metabolism in depression. J. Neurol. Neurosurg. Psychiatry 2000, 69, 228–232. [Google Scholar] [CrossRef]
- Wang, S.M.; Han, C.; Bahk, W.M.; Lee, S.J.; Patkar, A.A.; Masand, P.S.; Pae, C.U. Addressing the Side Effects of Contemporary Antidepressant Drugs: A Comprehensive Review. Chonnam Med. J. 2018, 54, 101–112. [Google Scholar] [CrossRef]
- Arroll, B.; Chin, W.Y.; Martis, W.; Goodyear-Smith, F.; Mount, V.; Kingsford, D.; Humm, S.; Blashki, G.; MacGillivray, S. Antidepressants for treatment of depression in primary care: A systematic review and meta-analysis. J. Prim Health Care 2016, 8, 325–334. [Google Scholar] [CrossRef]
- Han, Y.H.; Kee, J.Y.; Park, S.H.; Mun, J.G.; Jeon, H.D.; Park, J.; Zou, Q.P.; Liu, X.Q.; Hong, S.H. Rubrofusarin-6-beta-gentiobioside inhibits lipid accumulation and weight gain by regulating AMPK/mTOR signaling. Phytomedicine 2019, 62, 152952. [Google Scholar] [CrossRef]
- Xiao, J.; Zhang, Q.; Gao, Y.Q.; Shi, X.W.; Gao, J.M. Antifungal and antibacterial metabolites from an endophytic Aspergillus sp. associated with Melia azedarach. Nat. Prod. Res. 2014, 28, 1388–1392. [Google Scholar] [CrossRef]
- Lee, H.J.; Jung, J.H.; Kang, S.S.; Choi, J.S. A rubrofusarin gentiobioside isomer from roastedCassia tora. Arch. Pharm Res. 1997, 20, 513–515. [Google Scholar] [CrossRef] [PubMed]
- Cho, E.; Cai, M.; Kwon, H.; Jeon, J.; Moon, M.; Jun, M.; Lee, Y.C.; Yi, J.H.; Ryu, J.H.; Kim, D.H. Rubrofusarin inhibits Abeta aggregation and ameliorates memory loss in an Abeta-induced Alzheimer’s disease-like mouse model. Food Chem. Toxicol. 2019, 132, 110698. [Google Scholar] [CrossRef] [PubMed]
- Paudel, P.; Seong, S.H.; Jung, H.A.; Choi, J.S. Rubrofusarin as a Dual Protein Tyrosine Phosphate 1B and Human Monoamine Oxidase-A Inhibitor: An in Vitro and in Silico Study. ACS Omega 2019, 4, 11621–11630. [Google Scholar] [CrossRef] [PubMed]
- Schoenfeld, T.J.; McCausland, H.C.; Morris, H.D.; Padmanaban, V.; Cameron, H.A. Stress and Loss of Adult Neurogenesis Differentially Reduce Hippocampal Volume. Biol. Psychiatry 2017, 82, 914–923. [Google Scholar] [CrossRef]
- Barha, C.K.; Brummelte, S.; Lieblich, S.E.; Galea, L.A. Chronic restraint stress in adolescence differentially influences hypothalamic-pituitary-adrenal axis function and adult hippocampal neurogenesis in male and female rats. Hippocampus 2011, 21, 1216–1227. [Google Scholar] [CrossRef]
- Pan, Y.; Chen, X.Y.; Zhang, Q.Y.; Kong, L.D. Microglial NLRP3 inflammasome activation mediates IL-1beta-related inflammation in prefrontal cortex of depressive rats. Brain Behav. Immun. 2014, 41, 90–100. [Google Scholar] [CrossRef]
- Kreisel, T.; Frank, M.G.; Licht, T.; Reshef, R.; Ben-Menachem-Zidon, O.; Baratta, M.V.; Maier, S.F.; Yirmiya, R. Dynamic microglial alterations underlie stress-induced depressive-like behavior and suppressed neurogenesis. Mol. Psychiatry 2014, 19, 699–709. [Google Scholar] [CrossRef]
- Duman, R.S.; Aghajanian, G.K. Synaptic dysfunction in depression: Potential therapeutic targets. Science 2012, 338, 68–72. [Google Scholar] [CrossRef]
- Slavich, G.M.; Irwin, M.R. From stress to inflammation and major depressive disorder: A social signal transduction theory of depression. Psychol. Bull. 2014, 140, 774–815. [Google Scholar] [CrossRef]
- Breslau, N.; Davis, G.C. Chronic stress and major depression. Arch. Gen. Psychiatry 1986, 43, 309–314. [Google Scholar] [CrossRef]
- Tekieh, E.; Riahi, E.; Kazemi, M.; Sahraei, H.; Tavakoli, H.; Aliyary, H.; Hajinasrollah, M.; Salehi, M.; Meftahi, G.; Saberi, M. Role of basal stress hormones and amygdala dimensions in stress coping strategies of male rhesus monkeys in response to a hazard-reward conflict. Iran. J. Basic Med. Sci. 2017, 20, 951–957. [Google Scholar] [PubMed]
- Hoglund, E.; Silva, P.I.M.; Vindas, M.A.; Overli, O. Contrasting Coping Styles Meet the Wall: A Dopamine Driven Dichotomy in Behavior and Cognition. Front. Neurosci. 2017, 11, 383. [Google Scholar] [CrossRef] [PubMed]
- Alfonso, J.; Frasch, A.C.; Flugge, G. Chronic stress, depression and antidepressants: Effects on gene transcription in the hippocampus. Rev. Neurosci. 2005, 16, 43–56. [Google Scholar] [CrossRef] [PubMed]
- Tafet, G.E.; Smolovich, J. Psychoneuroendocrinological studies on chronic stress and depression. Ann. N. Y. Acad. Sci. 2004, 1032, 276–278. [Google Scholar] [CrossRef] [PubMed]
- Ago, Y.; Arikawa, S.; Yata, M.; Yano, K.; Abe, M.; Takuma, K.; Matsuda, T. Antidepressant-like effects of the glucocorticoid receptor antagonist RU-43044 are associated with changes in prefrontal dopamine in mouse models of depression. Neuropharmacology 2008, 55, 1355–1363. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, E.T.; Caldwell, J.L.; Streicher, J.; Ghisays, V.; Balmer, N.J.; Estrada, C.M.; Solomon, M.B. Differential effects of imipramine and CORT118335 (Glucocorticoid receptor modulator/mineralocorticoid receptor antagonist) on brain-endocrine stress responses and depression-like behavior in female rats. Behav. Brain Res. 2018, 336, 99–110. [Google Scholar] [CrossRef]
- Natarajan, R.; Forrester, L.; Chiaia, N.L.; Yamamoto, B.K. Chronic-Stress-Induced Behavioral Changes Associated with Subregion-Selective Serotonin Cell Death in the Dorsal Raphe. J. Neurosci. 2017, 37, 6214–6223. [Google Scholar] [CrossRef]
- Vyas, S.; Rodrigues, A.J.; Silva, J.M.; Tronche, F.; Almeida, O.F.; Sousa, N.; Sotiropoulos, I. Chronic Stress and Glucocorticoids: From Neuronal Plasticity to Neurodegeneration. Neural Plast. 2016, 2016. [Google Scholar] [CrossRef]
- Nair, A.; Bonneau, R.H. Stress-induced elevation of glucocorticoids increases microglia proliferation through NMDA receptor activation. J. Neuroimmunol. 2006, 171, 72–85. [Google Scholar] [CrossRef]
- Takahashi, T.; Kimoto, T.; Tanabe, N.; Hattori, T.A.; Yasumatsu, N.; Kawato, S. Corticosterone acutely prolonged N-methyl-d-aspartate receptor-mediated Ca2+ elevation in cultured rat hippocampal neurons. J. Neurochem. 2002, 83, 1441–1451. [Google Scholar] [CrossRef]
- Rami, A. Ischemic neuronal death in the rat hippocampus: The calpain-calpastatin-caspase hypothesis. Neurobiol. Dis. 2003, 13, 75–88. [Google Scholar] [CrossRef]
- Yamashima, T. Ca2+-dependent proteases in ischemic neuronal death: A conserved ‘calpain-cathepsin cascade’ from nematodes to primates. Cell Calcium 2004, 36, 285–293. [Google Scholar] [CrossRef] [PubMed]
- Deng, W.; Aimone, J.B.; Gage, F.H. New neurons and new memories: How does adult hippocampal neurogenesis affect learning and memory? Nat. Rev. Neurosci. 2010, 11, 339–350. [Google Scholar] [CrossRef] [PubMed]
- Cameron, H.A.; Glover, L.R. Adult neurogenesis: Beyond learning and memory. Annu. Rev. Psychol. 2015, 66, 53–81. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.S.; Sahay, A.; Hen, R. Increasing Adult Hippocampal Neurogenesis is Sufficient to Reduce Anxiety and Depression-Like Behaviors. Neuropsychopharmacology 2015, 40, 2368–2378. [Google Scholar] [CrossRef]
- Sahay, A.; Hen, R. Adult hippocampal neurogenesis in depression. Nat. Neurosci. 2007, 10, 1110–1115. [Google Scholar] [CrossRef]
- Ming, G.L.; Song, H. DISC1 partners with GSK3beta in neurogenesis. Cell 2009, 136, 990–992. [Google Scholar] [CrossRef]
- Kitagishi, Y.; Kobayashi, M.; Kikuta, K.; Matsuda, S. Roles of PI3K/AKT/GSK3/mTOR Pathway in Cell Signaling of Mental Illnesses. Depress. Res. Treat. 2012, 2012. [Google Scholar] [CrossRef]
- Manchia, M.; Rybakowski, J.K.; Sani, G.; Kessing, L.V.; Murru, A.; Alda, M.; Tondo, L. Lithium and bipolar depression. Bipolar Disord. 2019, 21, 458–459. [Google Scholar] [CrossRef]
- Cussotto, S.; Cryan, J.F.; O’Leary, O.F. The hippocampus and dorsal raphe nucleus are key brain areas associated with the antidepressant effects of lithium augmentation of desipramine. Neurosci. Lett. 2017, 648, 14–20. [Google Scholar] [CrossRef]
- Planchez, B.; Surget, A.; Belzung, C. Adult hippocampal neurogenesis and antidepressants effects. Curr. Opin. Pharmacol. 2019, 50, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Wohleb, E.S.; Terwilliger, R.; Duman, C.H.; Duman, R.S. Stress-Induced Neuronal Colony Stimulating Factor 1 Provokes Microglia-Mediated Neuronal Remodeling and Depressive-like Behavior. Biol. Psychiatry 2018, 83, 38–49. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.L.; Han, Q.Q.; Gong, W.Q.; Pan, D.H.; Wang, L.Z.; Hu, W.; Yang, M.; Li, B.; Yu, J.; Liu, Q. Microglial activation mediates chronic mild stress-induced depressive- and anxiety-like behavior in adult rats. J. Neuroinflamm. 2018, 15, 21. [Google Scholar] [CrossRef] [PubMed]
- Alcocer-Gomez, E.; Ulecia-Moron, C.; Marin-Aguilar, F.; Rybkina, T.; Casas-Barquero, N.; Ruiz-Cabello, J.; Ryffel, B.; Apetoh, L.; Ghiringhelli, F.; Bullon, P.; et al. Stress-Induced Depressive Behaviors Require a Functional NLRP3 Inflammasome. Mol. Neurobiol. 2016, 53, 4874–4882. [Google Scholar] [CrossRef]
- Feng, X.; Zhao, Y.; Yang, T.; Song, M.; Wang, C.; Yao, Y.; Fan, H. Glucocorticoid-Driven NLRP3 Inflammasome Activation in Hippocampal Microglia Mediates Chronic Stress-Induced Depressive-Like Behaviors. Front. Mol. Neurosci. 2019, 12, 210. [Google Scholar] [CrossRef]
- Alcocer-Gomez, E.; de Miguel, M.; Casas-Barquero, N.; Nunez-Vasco, J.; Sanchez-Alcazar, J.A.; Fernandez-Rodriguez, A.; Cordero, M.D. NLRP3 inflammasome is activated in mononuclear blood cells from patients with major depressive disorder. Brain Behav. Immun. 2014, 36, 111–117. [Google Scholar] [CrossRef]
- Ashraf, A.; Mahmoud, P.A.; Reda, H.; Mansour, S.; Helal, M.H.; Michel, H.E.; Nasr, M. Silymarin and silymarin nanoparticles guard against chronic unpredictable mild stress induced depressive-like behavior in mice: Involvement of neurogenesis and NLRP3 inflammasome. J. Psychopharmacol. 2019, 33, 615–631. [Google Scholar] [CrossRef]
- Yue, N.; Huang, H.; Zhu, X.; Han, Q.; Wang, Y.; Li, B.; Liu, Q.; Wu, G.; Zhang, Y.; Yu, J. Activation of P2X7 receptor and NLRP3 inflammasome assembly in hippocampal glial cells mediates chronic stress-induced depressive-like behaviors. J. Neuroinflamm. 2017, 14, 102. [Google Scholar] [CrossRef]
- Yi, J.H.; Brown, C.; Whitehead, G.; Piers, T.; Lee, Y.S.; Perez, C.M.; Regan, P.; Whitcomb, D.J.; Cho, K. Glucocorticoids activate a synapse weakening pathway culminating in tau phosphorylation in the hippocampus. Pharmacol. Res. 2017, 121, 42–51. [Google Scholar] [CrossRef]
- Wiegert, O.; Pu, Z.; Shor, S.; Joels, M.; Krugers, H. Glucocorticoid receptor activation selectively hampers N-methyl-d-aspartate receptor dependent hippocampal synaptic plasticity in vitro. Neuroscience 2005, 135, 403–411. [Google Scholar] [CrossRef]
- Li, Z.; Sheng, M. Caspases in synaptic plasticity. Mol. Brain 2012, 5, 15. [Google Scholar] [CrossRef]
- Yang, C.H.; Huang, C.C.; Hsu, K.S. Behavioral stress modifies hippocampal synaptic plasticity through corticosterone-induced sustained extracellular signal-regulated kinase/mitogen-activated protein kinase activation. J. Neurosci. 2004, 24, 11029–11034. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.J.; Foy, M.R.; Thompson, R.F. Behavioral stress modifies hippocampal plasticity through N-methyl-d-aspartate receptor activation. Proc. Natl. Acad. Sci. USA 1996, 93, 4750–4753. [Google Scholar] [CrossRef] [PubMed]
- Furay, A.R.; Bruestle, A.E.; Herman, J.P. The role of the forebrain glucocorticoid receptor in acute and chronic stress. Endocrinology 2008, 149, 5482–5490. [Google Scholar] [CrossRef] [PubMed]
- Zunszain, P.A.; Anacker, C.; Cattaneo, A.; Carvalho, L.A.; Pariante, C.M. Glucocorticoids, cytokines and brain abnormalities in depression. Prog. Neuropsychopharmacol. Biol. Psychiatry 2011, 35, 722–729. [Google Scholar] [CrossRef] [PubMed]
- Freitas, A.E.; Egea, J.; Buendia, I.; Navarro, E.; Rada, P.; Cuadrado, A.; Rodrigues, A.L.; Lopez, M.G. Agmatine induces Nrf2 and protects against corticosterone effects in hippocampal neuronal cell line. Mol. Neurobiol. 2015, 51, 1504–1519. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.Y.; Wang, X.; Wu, Y.; Doble, B.W.; Patel, S.; Woodgett, J.R.; Snider, W.D. GSK-3 is a master regulator of neural progenitor homeostasis. Nat. Neurosci. 2009, 12, 1390–1397. [Google Scholar] [CrossRef]
- Bradley, C.A.; Peineau, S.; Taghibiglou, C.; Nicolas, C.S.; Whitcomb, D.J.; Bortolotto, Z.A.; Kaang, B.K.; Cho, K.; Wang, Y.T.; Collingridge, G.L. A pivotal role of GSK-3 in synaptic plasticity. Front. Mol. Neurosci. 2012, 5, 13. [Google Scholar] [CrossRef]
- Green, H.F.; Nolan, Y.M. GSK-3 mediates the release of IL-1beta, TNF-alpha and IL-10 from cortical glia. Neurochem. Int. 2012, 61, 666–671. [Google Scholar] [CrossRef]
- Paxinos, G.; Franklin, K.B.J. The Mouse Brain in Stereotaxic Coordinate; Gulf Professional Publishing: Houston, TX, USA, 2004. [Google Scholar]



© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yi, J.H.; Jeon, J.; Kwon, H.; Cho, E.; Yun, J.; Lee, Y.C.; Ryu, J.H.; Park, S.J.; Cho, J.H.; Kim, D.H. Rubrofusarin Attenuates Chronic Restraint Stress-Induced Depressive Symptoms. Int. J. Mol. Sci. 2020, 21, 3454. https://doi.org/10.3390/ijms21103454
Yi JH, Jeon J, Kwon H, Cho E, Yun J, Lee YC, Ryu JH, Park SJ, Cho JH, Kim DH. Rubrofusarin Attenuates Chronic Restraint Stress-Induced Depressive Symptoms. International Journal of Molecular Sciences. 2020; 21(10):3454. https://doi.org/10.3390/ijms21103454
Chicago/Turabian StyleYi, Jee Hyun, Jieun Jeon, Huiyoung Kwon, Eunbi Cho, Jeanho Yun, Young Choon Lee, Jong Hoon Ryu, Se Jin Park, Jong Hyun Cho, and Dong Hyun Kim. 2020. "Rubrofusarin Attenuates Chronic Restraint Stress-Induced Depressive Symptoms" International Journal of Molecular Sciences 21, no. 10: 3454. https://doi.org/10.3390/ijms21103454
APA StyleYi, J. H., Jeon, J., Kwon, H., Cho, E., Yun, J., Lee, Y. C., Ryu, J. H., Park, S. J., Cho, J. H., & Kim, D. H. (2020). Rubrofusarin Attenuates Chronic Restraint Stress-Induced Depressive Symptoms. International Journal of Molecular Sciences, 21(10), 3454. https://doi.org/10.3390/ijms21103454




