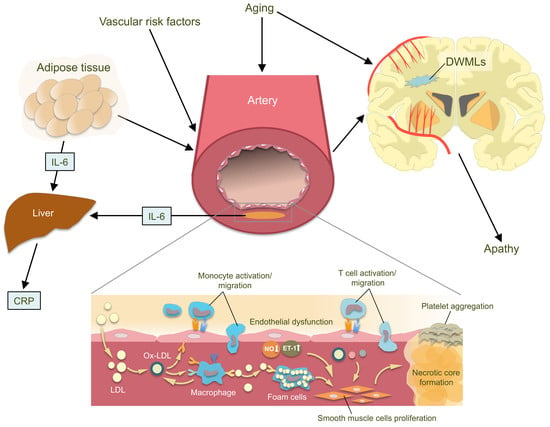
Low-Grade Inflammation Is Associated with Apathy Indirectly via Deep White Matter Lesions in Community-Dwelling Older Adults: The Sefuri Study
Abstract
1. Introduction
2. Results
2.1. Background Characteristics
2.2. Logistic Regression Analysis
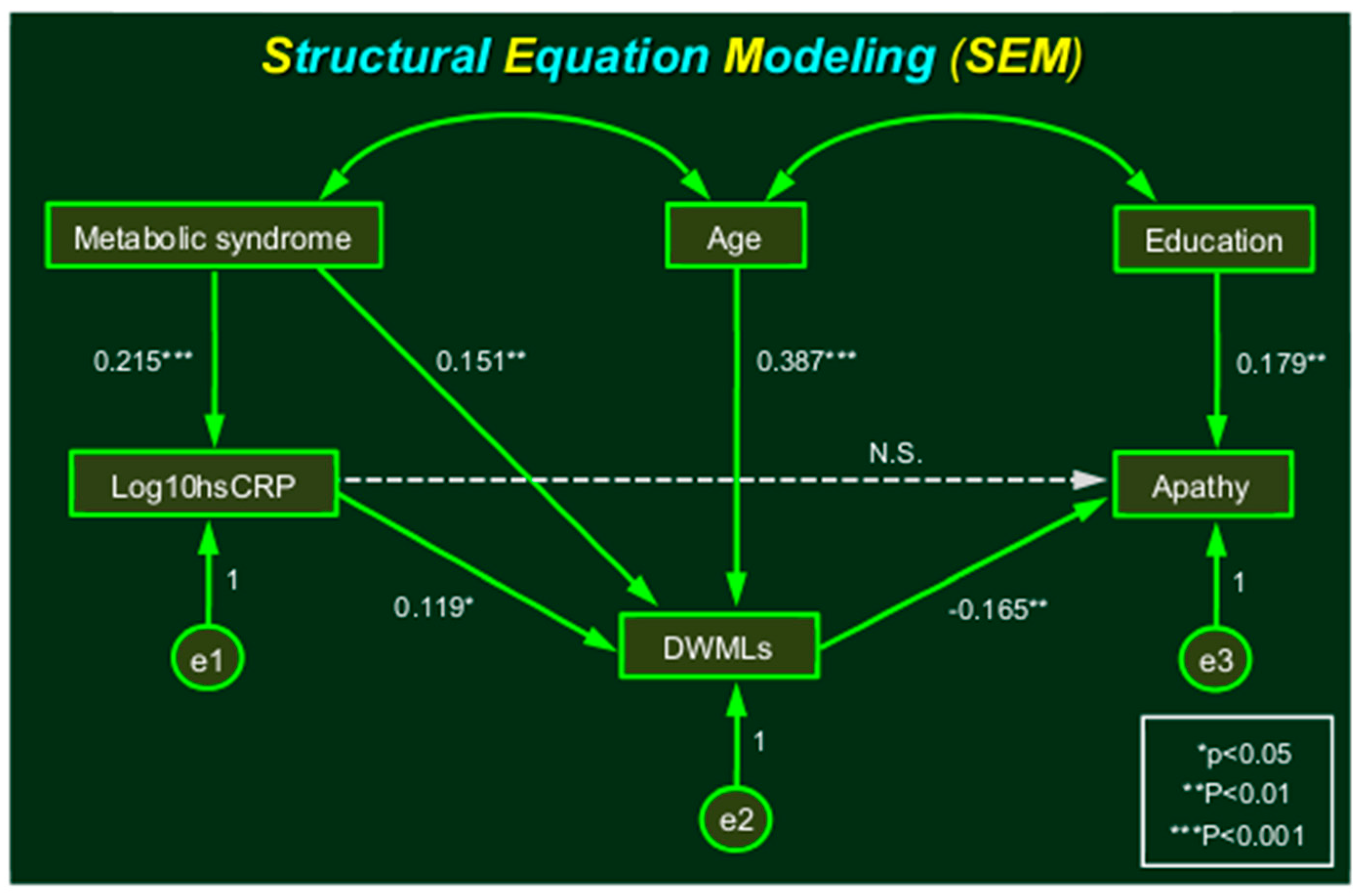
2.3. Structural Equation Modeling
3. Discussion
4. Subjects and Methods
4.1. Participants and Protocol Approval
4.2. Clinical Assessments
4.3. Apathy Scale
4.4. High-Sensitivity CRP Measurements
4.5. Assessment of MRI Findings
4.6. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AGFI | adjusted goodness of fit index |
| ANOVA | analysis of variance |
| CBF | cerebral blood flow |
| CFI | comparative fit index |
| DWMLs | deep white matter lesions |
| eGFR | estimated glomerular filtration rate |
| FLAIR | fluid attenuated inversion recovery |
| GFI | goodness of fit index |
| HDL | high-density lipoprotein |
| hsCRP | high-sensitivity C-reactive protein |
| IL-1 | interleukin-1 |
| IL-1β | interleukin-1β |
| IL-6 | interleukin-6 |
| IL-10 | interleukin-10 |
| LDL | low-density lipoprotein |
| MRI | magnetic resonance imaging |
| mST | modified Stroop test |
| Ox-LDL | oxidized low-density lipoprotein |
| RBMT | Rivermead Behavioral Memory Test |
| RMSEA | root mean square error of approximation |
| SEM | structural equation modeling |
| TNF-α | tumor necrosis factor-α |
References
- Rost, N.S.; Wolf, P.A.; Kase, C.S.; Kelly-Hayes, M.; Silbershatz, H.; Massaro, J.M.; D’Agostino, R.B.; Franzblau, C.; Wilson, P.W. Plasma concentration of C-reactive protein and risk of ischemic stroke and transient ischemic attack: The Framingham study. Stroke 2001, 32, 2575–2579. [Google Scholar] [CrossRef]
- Curb, J.D.; Abbott, R.D.; Rodriguez, B.L.; Sakkinen, P.; Popper, J.S.; Yano, K.; Tracy, R.P. C-reactive protein and the future risk of thromboembolic stroke in healthy men. Circulation 2003, 107, 2016–2020. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.J.; Thach, C.; Manolio, T.A.; Psaty, B.M.; Kuller, L.H.; Chaves, P.H.; Polak, J.F.; Sutton-Tyrrell, K.; Herrington, D.M.; Price, T.R.; et al. C-reactive protein, carotid intima-media thickness, and incidence of ischemic stroke in the elderly: The Cardiovascular Health Study. Circulation 2003, 108, 166–170. [Google Scholar] [CrossRef]
- Wakugawa, Y.; Kiyohara, Y.; Tanizaki, Y.; Kubo, M.; Ninomiya, T.; Hata, J.; Doi, Y.; Okubo, K.; Oishi, Y.; Shikata, K.; et al. C-reactive protein and risk of first-ever ischemic and hemorrhagic stroke in a general Japanese population: the Hisayama Study. Stroke 2006, 37, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Elkind, M.S.; Luna, J.M.; Moon, Y.P.; Liu, K.M.; Spitalnik, S.L.; Paik, M.C.; Sacco, R.L. High-sensitivity C-reactive protein predicts mortality but not stroke: The Northern Manhattan Study. Neurology 2009, 73, 1300–1307. [Google Scholar] [CrossRef]
- Shoamanesh, A.; Preis, S.R.; Beiser, A.S.; Kase, C.S.; Wolf, P.A.; Vasan, R.S.; Benjamin, E.J.; Seshadri, S.; Romero, J.R. Circulating biomarkers and incident ischemic stroke in the Framingham Offspring Study. Neurology 2016, 87, 1206–1211. [Google Scholar] [CrossRef] [PubMed]
- Luna, J.M.; Moon, Y.P.; Liu, K.M.; Spitalnik, S.; Paik, M.C.; Cheung, K.; Sacco, R.L.; Elkind, M.S. High-sensitivity C-reactive protein and interleukin-6-dominant inflammation and ischemic stroke risk: the northern Manhattan study. Stroke 2014, 45, 979–987. [Google Scholar] [CrossRef]
- Hoshi, T.; Kitagawa, K.; Yamagami, H.; Furukado, S.; Hougaku, H.; Hori, M. Relations of serum high-sensitivity C-reactive protein and interleukin-6 levels with silent brain infarction. Stroke 2005, 36, 768–772. [Google Scholar] [CrossRef]
- Van Dijk, E.J.; Prins, N.D.; Vermeer, S.E.; Vrooman, H.A.; Hofman, A.; Koudstaal, P.J.; Breteler, M.M. C-reactive protein and cerebral small-vessel disease: The Rotterdam Scan Study. Circulation 2005, 112, 900–905. [Google Scholar] [CrossRef]
- Satizabal, C.L.; Zhu, Y.C.; Mazoyer, B.; Dufouil, C.; Tzourio, C. Circulating IL-6 and CRP are associated with MRI findings in the elderly: The 3C-Dijon Study. Neurology 2012, 78, 720–727. [Google Scholar] [CrossRef]
- Walker, K.A.; Power, M.C.; Hoogeveen, R.C.; Folsom, A.R.; Ballantyne, C.M.; Knopman, D.S.; Windham, B.G.; Selvin, E.; Jack, C.R., Jr.; Gottesman, R.F. Midlife Systemic Inflammation, Late-Life White Matter Integrity, and Cerebral Small Vessel Disease: The Atherosclerosis Risk in Communities Study. Stroke 2017, 48, 3196–3202. [Google Scholar] [CrossRef]
- Schmidt, R.; Schmidt, H.; Pichler, M.; Enzinger, C.; Petrovic, K.; Niederkorn, K.; Horner, S.; Ropele, S.; Watzinger, N.; Schumacher, M.; et al. C-reactive protein, carotid atherosclerosis, and cerebral small-vessel disease: results of the Austrian Stroke Prevention Study. Stroke 2006, 37, 2910–2916. [Google Scholar] [CrossRef]
- Rosenberg, G.A.; Wallin, A.; Wardlaw, J.M.; Markus, H.S.; Montaner, J.; Wolfson, L.; Iadecola, C.; Zlokovic, B.V.; Joutel, A.; Dichgans, M.; et al. Consensus statement for diagnosis of subcortical small vessel disease. J. Cereb. Blood Flow Metab. 2016, 36, 6–25. [Google Scholar] [CrossRef]
- De Berardis, D.; Campanella, D.; Gambi, F.; La Rovere, R.; Carano, A.; Conti, C.M.; Sivestrini, C.; Serroni, N.; Piersanti, D.; di Giuseppe, B.; et al. The role of C-reactive protein in mood disorders. Int. J. Immunopathol. Pharmacol. 2006, 19, 721–725. [Google Scholar] [CrossRef]
- Haapakoski, R.; Mathieu, J.; Ebmeier, K.P.; Alenius, H.; Kivimäki, M. Cumulative meta-analysis of interleukins 6 and 1β, tumour necrosis factor α and C-reactive protein in patients with major depressive disorder. Brain Behav. Immun. 2015, 49, 206–215. [Google Scholar] [CrossRef]
- Ng, A.; Tam, W.W.; Zhang, M.W.; Ho, C.S.; Husain, S.F.; McIntyre, R.S.; Ho, R.C. IL-1β, IL-6, TNF- α and CRP in Elderly Patients with Depression or Alzheimer’s disease: Systematic Review and Meta-Analysis. Sci. Rep. 2018, 8, 12050. [Google Scholar] [CrossRef]
- Eurelings, L.S.; Richard, E.; Eikelenboom, P.; van Gool, W.A.; Moll van Charante, E.P. Low-grade inflammation differentiates between symptoms of apathy and depression in community-dwelling older individuals. Int. Psychogeriatr. 2015, 27, 639–647. [Google Scholar] [CrossRef]
- Eurelings, L.S.; van Dalen, J.W.; Ter Riet, G.; Moll van Charante, E.P.; Richard, E.; van Gool, W.A.; ICARA Study Group. Apathy and depressive symptoms in older people and incident myocardial infarction, stroke, and mortality: A systematic review and meta-analysis of individual participant data. Clin. Epidemiol. 2018, 10, 363–379. [Google Scholar] [CrossRef]
- Hajjar, I.; Quach, L.; Yang, F.; Chaves, P.H.; Newman, A.B.; Mukamal, K.; Longstreth, W., Jr.; Inzitari, M.; Lipsitz, L.A. Hypertension, white matter hyperintensities, and concurrent impairments in mobility, cognition, and mood: The Cardiovascular Health Study. Circulation 2011, 123, 858–865. [Google Scholar] [CrossRef]
- Firbank, M.J.; Teodorczuk, A.; van der Flier, W.M.; Gouw, A.A.; Wallin, A.; Erkinjuntti, T.; Inzitari, D.; Wahlund, L.O.; Pantoni, L.; Poggesi, A.; et al. Relationship between progression of brain white matter changes and late-life depression: 3-year results from the LADIS study. Br. J. Psychiatry 2012, 201, 40–45. [Google Scholar] [CrossRef]
- Brookes, R.L.; Herbert, V.; Lawrence, A.J.; Morris, R.G.; Markus, H.S. Depression in small-vessel disease relates to white matter ultrastructural damage, not disability. Neurology 2014, 83, 1417–1423. [Google Scholar] [CrossRef]
- Van Sloten, T.T.; Sigurdsson, S.; van Buchem, M.A.; Phillips, C.L.; Jonsson, P.V.; Ding, J.; Schram, M.T.; Harris, T.B.; Gudnason, V.; Launer, L.J. Cerebral Small Vessel Disease and Association With Higher Incidence of Depressive Symptoms in a General Elderly Population: The AGES-Reykjavik Study. Am. J. Psychiatry 2015, 172, 570–578. [Google Scholar] [CrossRef] [PubMed]
- Yao, H.; Takashima, Y.; Araki, Y.; Uchino, A.; Yuzuriha, T.; Hashimoto, M. Leisure-Time Physical Inactivity Associated with Vascular Depression or Apathy in Community-Dwelling Elderly Subjects: The Sefuri Study. J. Stroke Cerebrovasc. Dis. 2015, 24, 2625–2631. [Google Scholar] [CrossRef]
- Lavretsky, H.; Zheng, L.; Weiner, M.W.; Mungas, D.; Reed, B.; Kramer, J.H.; Jagust, W.; Chui, H.; Mack, W.J. The MRI brain correlates of depressed mood, anhedonia, apathy, and anergia in older adults with and without cognitive impairment or dementia. Int. J. Geriatr. Psychiatry 2008, 23, 1040–1050. [Google Scholar] [CrossRef] [PubMed]
- Grool, A.M.; Gerritsen, L.; Zuithoff, N.P.; Mali, W.P.; van der Graaf, Y.; Geerlings, M.I. Lacunar infarcts in deep white matter are associated with higher and more fluctuating depressive symptoms during three years follow-up. Biol. Psychiatry 2013, 73, 169–176. [Google Scholar] [CrossRef]
- Taylor, W.D.; Aizenstein, H.J.; Alexopoulos, G.S. The vascular depression hypothesis: Mechanisms linking vascular disease with depression. Mol. Psychiatry 2013, 18, 963–974. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, G.A.; Bjerke, M.; Wallin, A. Multimodal markers of inflammation in the subcortical ischemic vascular disease type of vascular cognitive impairment. Stroke 2014, 45, 1531–1538. [Google Scholar] [CrossRef] [PubMed]
- De Reuck, J. The human periventricular arterial blood supply and the anatomy of cerebral infarctions. Eur. Neurol. 1971, 5, 321–334. [Google Scholar] [CrossRef]
- Furuta, A.; Ishii, N.; Nishihara, Y.; Horie, A. Medullary arteries in aging and dementia. Stroke 1991, 22, 442–446. [Google Scholar] [CrossRef]
- Fisher, C.M. Binswanger’s encephalopathy: A review. J. Neurol. 1989, 236, 65–79. [Google Scholar] [CrossRef]
- Yao, H.; Sadoshima, S.; Kuwabara, Y.; Ichiya, Y.; Fujishima, M. Cerebral blood flow and oxygen metabolism in patients with vascular dementia of the Binswanger type. Stroke 1990, 21, 1694–1699. [Google Scholar] [CrossRef] [PubMed]
- Yao, H.; Sadoshima, S.; Ibayashi, S.; Kuwabara, Y.; Ichiya, Y.; Fujishima, M. Leukoaraiosis and dementia in hypertensive patients. Stroke 1992, 23, 1673–1677. [Google Scholar] [CrossRef] [PubMed]
- Nezu, T.; Yokota, C.; Uehara, T.; Yamauchi, M.; Fukushima, K.; Toyoda, K.; Matsumoto, M.; Iida, H.; Minematsu, K. Preserved acetazolamide reactivity in lacunar patients with severe white-matter lesions: 15O-labeled gas and H2O positron emission tomography studies. J. Cereb. Blood Flow Metab. 2012, 32, 844–850. [Google Scholar] [CrossRef]
- Bernbaum, M.; Menon, B.K.; Fick, G.; Smith, E.E.; Goyal, M.; Frayne, R.; Coutts, S.B. Reduced blood flow in normal white matter predicts development of leukoaraiosis. J. Cereb. Blood Flow Metab. 2015, 35, 1610–1615. [Google Scholar] [CrossRef]
- Yakushiji, Y.; Werring, D.J. Cerebrovascular disease: Lobar cerebral microbleeds signal early cognitive impairment. Nat. Rev. Neurol. 2016, 12, 680–682. [Google Scholar] [CrossRef]
- Ross, R. Atherosclerosis—An inflammatory disease. N. Engl. J. Med. 1999, 340, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Hansson, G.K. Inflammation, atherosclerosis, and coronary artery disease. N. Engl. J. Med. 2005, 352, 1685–1695. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.Y.; Li, C.J.; Hou, M.F.; Chu, P.Y. New Insights into the Role of Inflammation in the Pathogenesis of Atherosclerosis. Int. J. Mol. Sci. 2017, 18, 2034. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; Wilson, P.W.; Grundy, S.M. Should C-reactive protein be added to metabolic syndrome and to assessment of global cardiovascular risk? Circulation 2004, 109, 2818–2825. [Google Scholar] [CrossRef]
- Devaraj, S.; Singh, U.; Jialal, I. Human C-reactive protein and the metabolic syndrome. Curr. Opin. Lipidol. 2009, 20, 182–189. [Google Scholar] [CrossRef]
- Srikanthan, K.; Feyh, A.; Visweshwar, H.; Shapiro, J.I.; Sodhi, K. Systematic Review of Metabolic Syndrome Biomarkers: A Panel for Early Detection, Management, and Risk Stratification in the West Virginian Population. Int. J. Med. Sci. 2016, 13, 25–38. [Google Scholar] [CrossRef] [PubMed]
- Rogero, M.M.; Calder, P.C. Obesity, Inflammation, Toll-Like Receptor 4 and Fatty Acids. Nutrients 2018, 10, 432. [Google Scholar] [CrossRef] [PubMed]
- Bokura, H.; Yamaguchi, S.; Iijima, K.; Nagai, A.; Oguro, H. Metabolic syndrome is associated with silent ischemic brain lesions. Stroke 2008, 39, 1607–1609. [Google Scholar] [CrossRef] [PubMed]
- Dearborn, J.L.; Schneider, A.L.; Sharrett, A.R.; Mosley, T.H.; Bezerra, D.C.; Knopman, D.S.; Selvin, E.; Jack, C.R.; Coker, L.H.; Alonso, A.; et al. Obesity, Insulin Resistance, and Incident Small Vessel Disease on Magnetic Resonance Imaging: Atherosclerosis Risk in Communities Study. Stroke 2015, 46, 3131–3136. [Google Scholar] [CrossRef] [PubMed]
- Vermeer, S.E.; Den Heijer, T.; Koudstaal, P.J.; Oudkerk, M.; Hofman, A.; Breteler, M.M.; Rotterdam Scan Study. Incidence and risk factors of silent brain infarcts in the population-based Rotterdam Scan Study. Stroke 2003, 34, 392–396. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M. From C-Reactive Protein to Interleukin-6 to Interleukin-1: Moving Upstream To Identify Novel Targets for Atheroprotection. Circ. Res. 2016, 118, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Aday, A.W.; Ridker, P.M. Antiinflammatory Therapy in Clinical Care: The CANTOS Trial and Beyond. Front. Cardiovasc. Med. 2018, 5, 62. [Google Scholar] [CrossRef]
- Yao, H.; Fukuda, K.; Araki, Y.; Takashima, Y.; Uchino, A.; Yuzuriha, T.; Hashimoto, M. Parity As a Protective Biomarker Against Silent Brain Infarction in Community-Dwelling Older Adults: The Sefuri Study. J. Stroke Cerebrovasc. Dis. 2018. [Google Scholar] [CrossRef] [PubMed]
- Doi, Y.; Ninomiya, T.; Hata, J.; Yonemoto, K.; Arima, H.; Kubo, M.; Tanizaki, Y.; Iwase, M.; Iida, M.; Kiyohara, Y. Proposed criteria for metabolic syndrome in Japanese based on prospective evidence: The Hisayama study. Stroke 2009, 40, 1187–1194. [Google Scholar] [CrossRef]
- Yao, H.; Araki, Y.; Takashima, Y.; Uchino, A.; Yuzuriha, T.; Hashimoto, M. Chronic Kidney Disease and Subclinical Brain Infarction Increase the Risk of Vascular Cognitive Impairment: The Sefuri Study. J. Stroke Cerebrovasc. Dis. 2017, 26, 420–424. [Google Scholar] [CrossRef] [PubMed]
- Starkstein, S.E.; Mayberg, H.S.; Preziosi, T.J.; Andrezejewski, P.; Leiguarda, R.; Robinson, R.G. Reliability, validity, and clinical correlates of apathy in Parkinson’s disease. J. Neuropsychiatry. Clin. Neurosci. 1992, 4, 134–139. [Google Scholar] [PubMed]
- Yao, H.; Takashima, Y.; Mori, T.; Uchino, A.; Hashimoto, M.; Yuzuriha, T.; Miwa, Y.; Sasaguri, T. Hypertension and white matter lesions are independently associated with apathetic behavior in healthy elderly subjects: The Sefuri brain MRI study. Hypertens. Res. 2009, 32, 586–590. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, M.; Araki, Y.; Takashima, Y.; Nogami, K.; Uchino, A.; Yuzuriha, T.; Yao, H. Physical inactivity associated with vascular depression or apathy leads to hippocampal atrophy and memory dysfunction in community-dwelling elderly subjects: The Sefuri study. Brain Behav. 2016, 7, e00620. [Google Scholar] [CrossRef]
- Sasaki, M.; Hirai, T.; Taoka, T.; Higano, S.; Wakabayashi, C.; Matsusue, E.; Ida, M. Discriminating between silent cerebral infarction and deep white matter hyperintensity using combinations of three types of magnetic resonance images: A multicenter observer performance study. Neuroradiology 2008, 50, 753–758. [Google Scholar] [CrossRef]
- Yakushiji, Y.; Charidimou, A.; Hara, M.; Noguchi, T.; Nishihara, M.; Eriguchi, M.; Nanri, Y.; Nishiyama, M.; Werring, D.J.; Hara, H. Topography and associations of perivascular spaces in healthy adults: The Kashima scan study. Neurology 2014, 83, 2116–2123. [Google Scholar] [CrossRef]
- Fazekas, F.; Kleinert, R.; Offenbacher, H.; Schmidt, R.; Kleinert, G.; Payer, F.; Radner, H.; Lechner, H. Pathologic correlates of incidental MRI white matter signal hyperintensities. Neurology 1993, 43, 1683–1689. [Google Scholar] [CrossRef] [PubMed]
- Haenlein, M.; Kaplan, A.M. A beginner’s guide to partial least squares analysis. Underst. Stat. 2004, 3, 283–297. [Google Scholar] [CrossRef]
| C-Reactive Protein Tertiles | p for Trend | ||||||
|---|---|---|---|---|---|---|---|
| Low (n = 87) | Medium (n = 86) | High (n = 86) | |||||
| High-Sensitivity C-Reactive Protein | 0.020–0.366 mg/L | 0.379–0.982 mg/L | 1.001–12.617 mg/L | ||||
| Age, mean (S.D.), years | 68.8 | (7.3) | 67.7 | (6.3) | 68.8 | (7.3) | NS |
| Male, n (%) | 38 | (43.7) | 46 | (53.5) | 38 | (44.2) | NS |
| Body mass index, mean (S.D.), kg/m2 | 22.5 | (3.1) | 23.8 | (3.7) | 24.2 | (3.5) | 0.004 |
| Metabolic syndrome, n (%) | 5 | (5.7) | 15 | (17.4) | 14 | (16.3) | 0.043 |
| Waist circumference, mean (S.D.), cm | 81.7 | (7.5) | 85.6 | (9.9) | 87.1 | (9.6) | <0.001 |
| Hypertension, n (%) | 34 | (39.1) | 32 | (37.2) | 38 | (44.2) | NS |
| Systolic BP, mean (S.D.), mmHg | 139.8 | (19.7) | 139.5 | (18.0) | 145.2 | (18.8) | 0.089 |
| Diastolic BP, mean (S.D.), mmHg | 80.9 | (10.9) | 82.1 | (10.4) | 83.0 | (10.9) | NS |
| Diabetes mellitus, n (%) | 8 | (9.2) | 14 | (16.3) | 17 | (19.8) | 0.140 |
| Hyperlipidemia, n (%) | 24 | (27.6) | 32 | (37.2) | 30 | (34.9) | NS |
| Ischemic heart disease, n (%) | 4 | (4.6) | 9 | (10.5) | 1 | (1.2) | 0.024 |
| Chronic kidney disease, n (%) | 15 | (17.2) | 12 | (14.0) | 20 | (23.3) | NS |
| Alcohol, n (%) | 32 | (36.8) | 41 | (47.7) | 30 | (34.9) | 0.180 |
| Smoking, n (%) | 6 | (7.0) | 13 | (15.1) | 6 | (7.0) | 0.110 |
| Hemoglobin A1c, mean (S.D.), % | 5.56 | (0.51) | 5.78 | (0.96) | 5.74 | (0.63) | 0.103 |
| LDL cholesterol, mean (S.D.), mg/dL | 117.5 | (29.0) | 125.1 | (31.4) | 126.7 | (38.2) | 0.150 |
| HDL cholesterol, mean (S.D.), mg/dL | 73.5 | (15.7) | 65.2 | (18.5) | 63.8 | (15.7) | <0.001 |
| Triglyceridel, mean (S.D.), mg/dL | 95.6 | (59.7) | 135.8 | (107.8) | 125.9 | (87.9) | 0.007 |
| Uric acid, mean (S.D.), mg/dL | 4.69 | (1.34) | 5.05 | (1.21) | 5.38 | (1.29) | 0.002 |
| eGFR, mean (S.D.), mL/min/1.73 m2 | 74.0 | (14.6) | 76.1 | (15.4) | 71.9 | (14.7) | 0.183 |
| MRI Findings | DWMLs | Confluent DWMLs | Confluent DWMLs | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Model 1 | Model 2 | ||||||||
| OR | 95% CI | p | OR | 95% CI | p | OR | 95% CI | p | |
| Age, /10 years | 2.456 | 1.647–3.663 | <0.001 | 4.405 | 2.419–8.021 | <0.001 | 5.065 | 2.683–9.559 | <0.001 |
| log10 hsCRP | 3.024 | 1.305–7.008 | 0.010 | 2.878 | 1.218–6.800 | 0.016 | |||
| Hypertension | 2.620 | 1.508–4.553 | 0.001 | ||||||
| Metabolic syndrome * | 3.211 | 1.141–9.003 | 0.027 | 1.996 | 1.228–3.243 | 0.005 | |||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yao, H.; Mizoguchi, Y.; Monji, A.; Yakushiji, Y.; Takashima, Y.; Uchino, A.; Yuzuriha, T.; Hashimoto, M. Low-Grade Inflammation Is Associated with Apathy Indirectly via Deep White Matter Lesions in Community-Dwelling Older Adults: The Sefuri Study. Int. J. Mol. Sci. 2019, 20, 1905. https://doi.org/10.3390/ijms20081905
Yao H, Mizoguchi Y, Monji A, Yakushiji Y, Takashima Y, Uchino A, Yuzuriha T, Hashimoto M. Low-Grade Inflammation Is Associated with Apathy Indirectly via Deep White Matter Lesions in Community-Dwelling Older Adults: The Sefuri Study. International Journal of Molecular Sciences. 2019; 20(8):1905. https://doi.org/10.3390/ijms20081905
Chicago/Turabian StyleYao, Hiroshi, Yoshito Mizoguchi, Akira Monji, Yusuke Yakushiji, Yuki Takashima, Akira Uchino, Takefumi Yuzuriha, and Manabu Hashimoto. 2019. "Low-Grade Inflammation Is Associated with Apathy Indirectly via Deep White Matter Lesions in Community-Dwelling Older Adults: The Sefuri Study" International Journal of Molecular Sciences 20, no. 8: 1905. https://doi.org/10.3390/ijms20081905
APA StyleYao, H., Mizoguchi, Y., Monji, A., Yakushiji, Y., Takashima, Y., Uchino, A., Yuzuriha, T., & Hashimoto, M. (2019). Low-Grade Inflammation Is Associated with Apathy Indirectly via Deep White Matter Lesions in Community-Dwelling Older Adults: The Sefuri Study. International Journal of Molecular Sciences, 20(8), 1905. https://doi.org/10.3390/ijms20081905