Characterization and Dye Decolorization Potential of Two Laccases from the Marine-Derived Fungus Pestalotiopsis sp.
Abstract
1. Introduction
2. Results
2.1. Target Selection, Aspergillus Niger Transformation, Screening, and Protein Purification
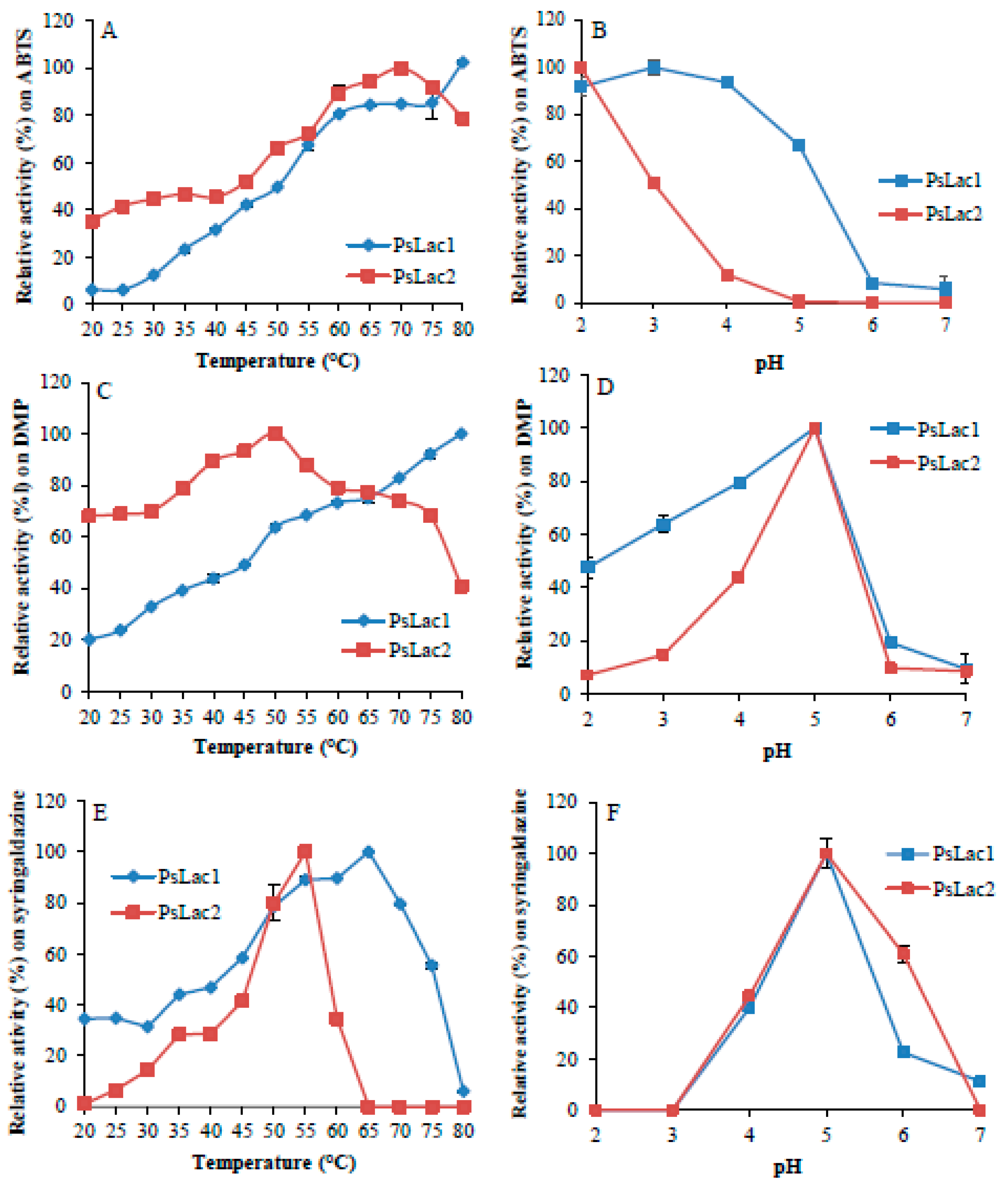
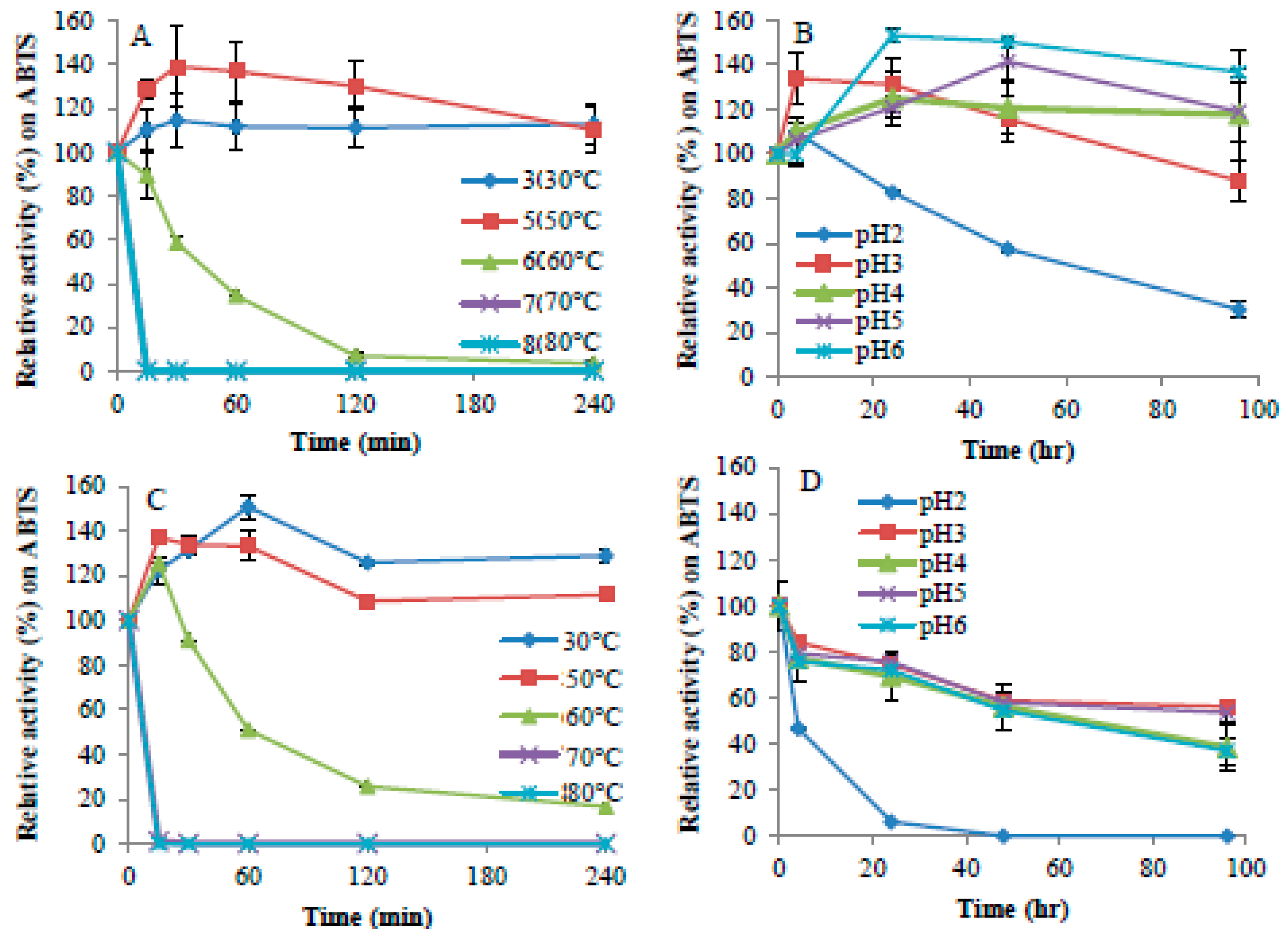
2.2. Enzyme Activity and Stability at Different pH and Temperature
2.3. Substrate Specificity and Kinetic Properties
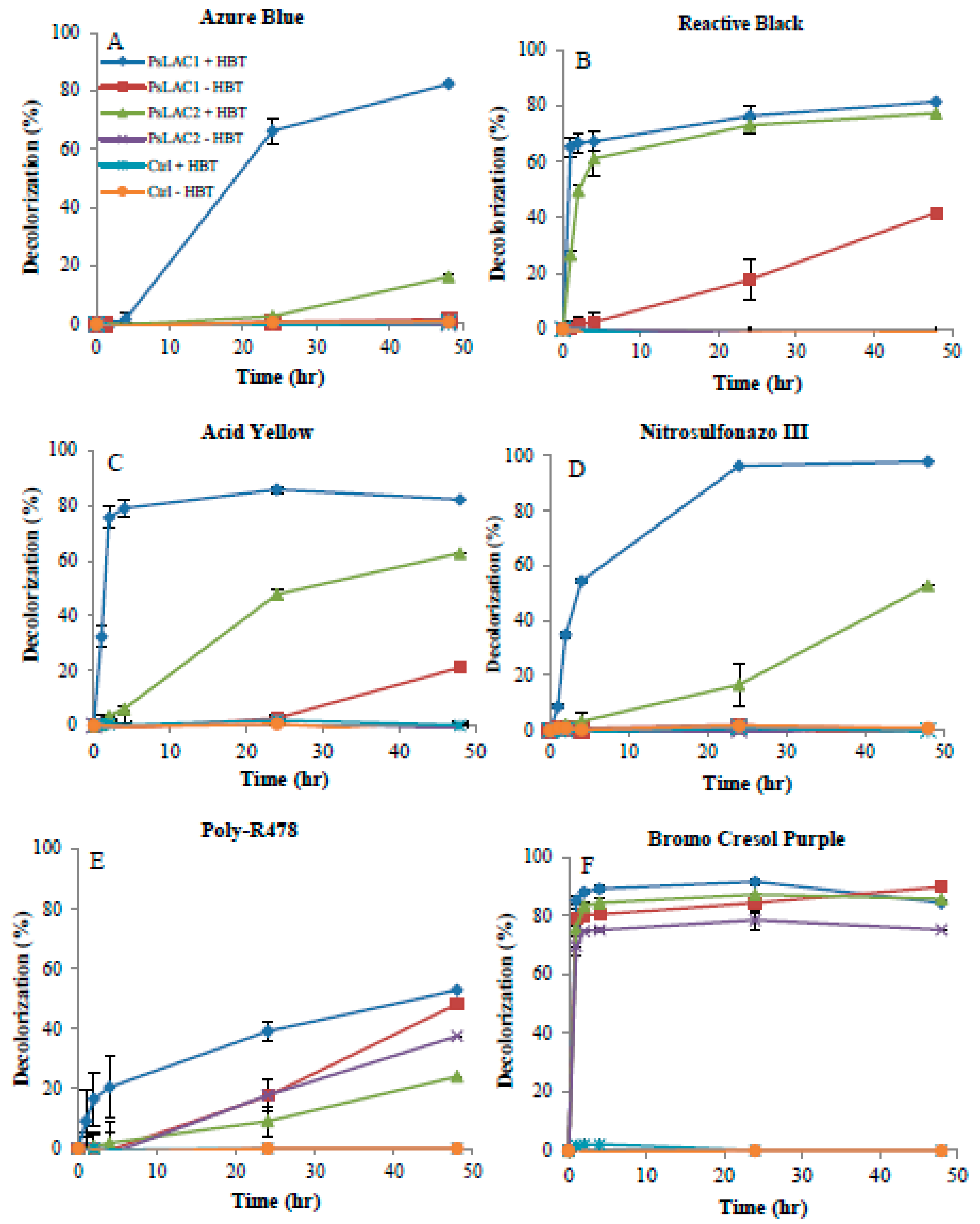
2.4. Decolorization of Commercial Dyes
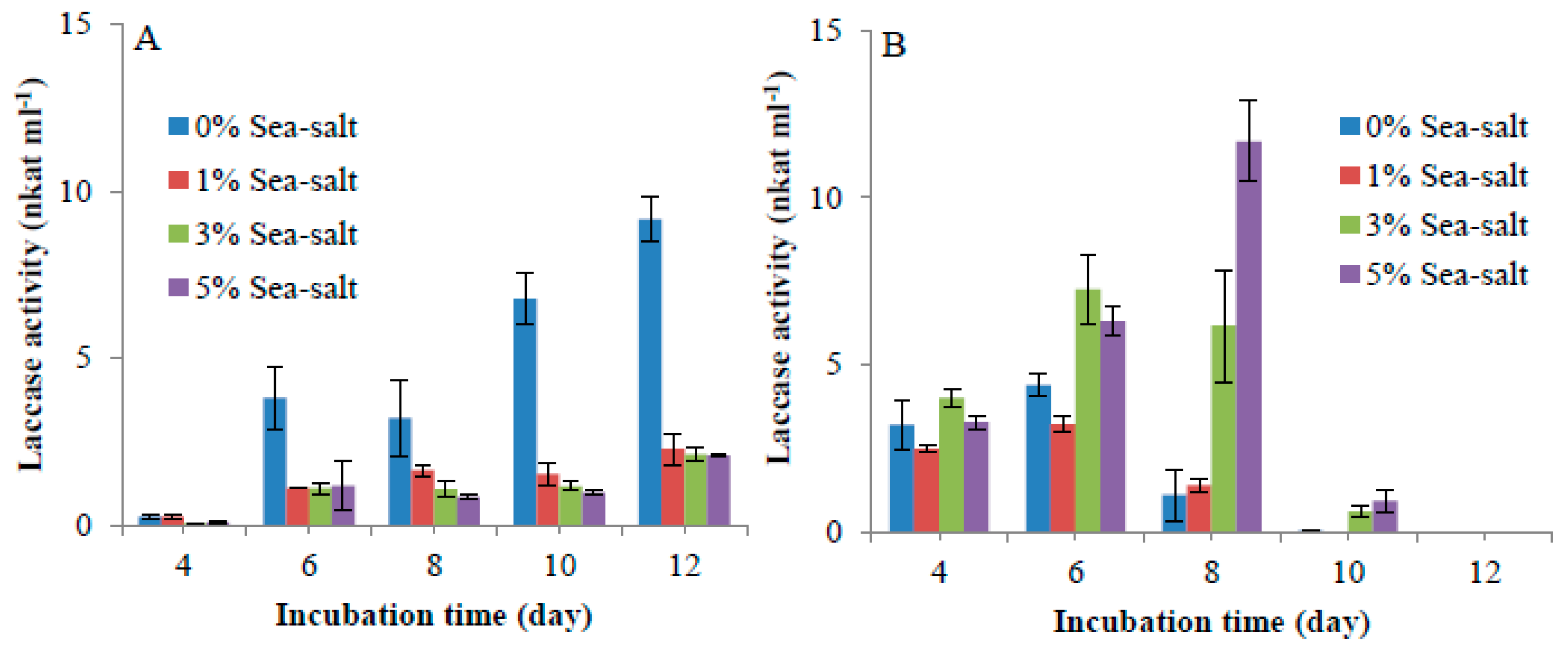
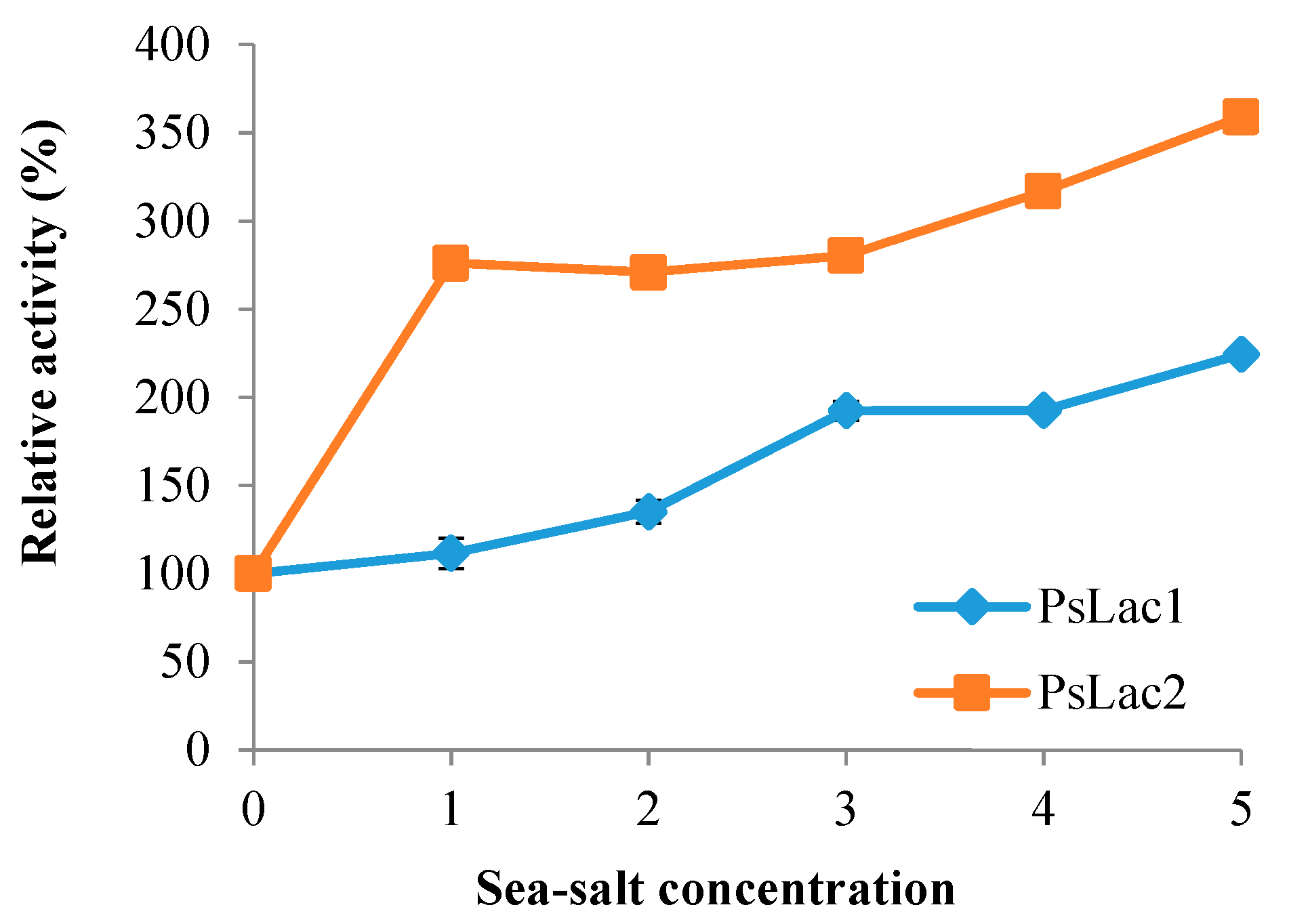
2.5. Influence of Sea Salt on Laccase Activity
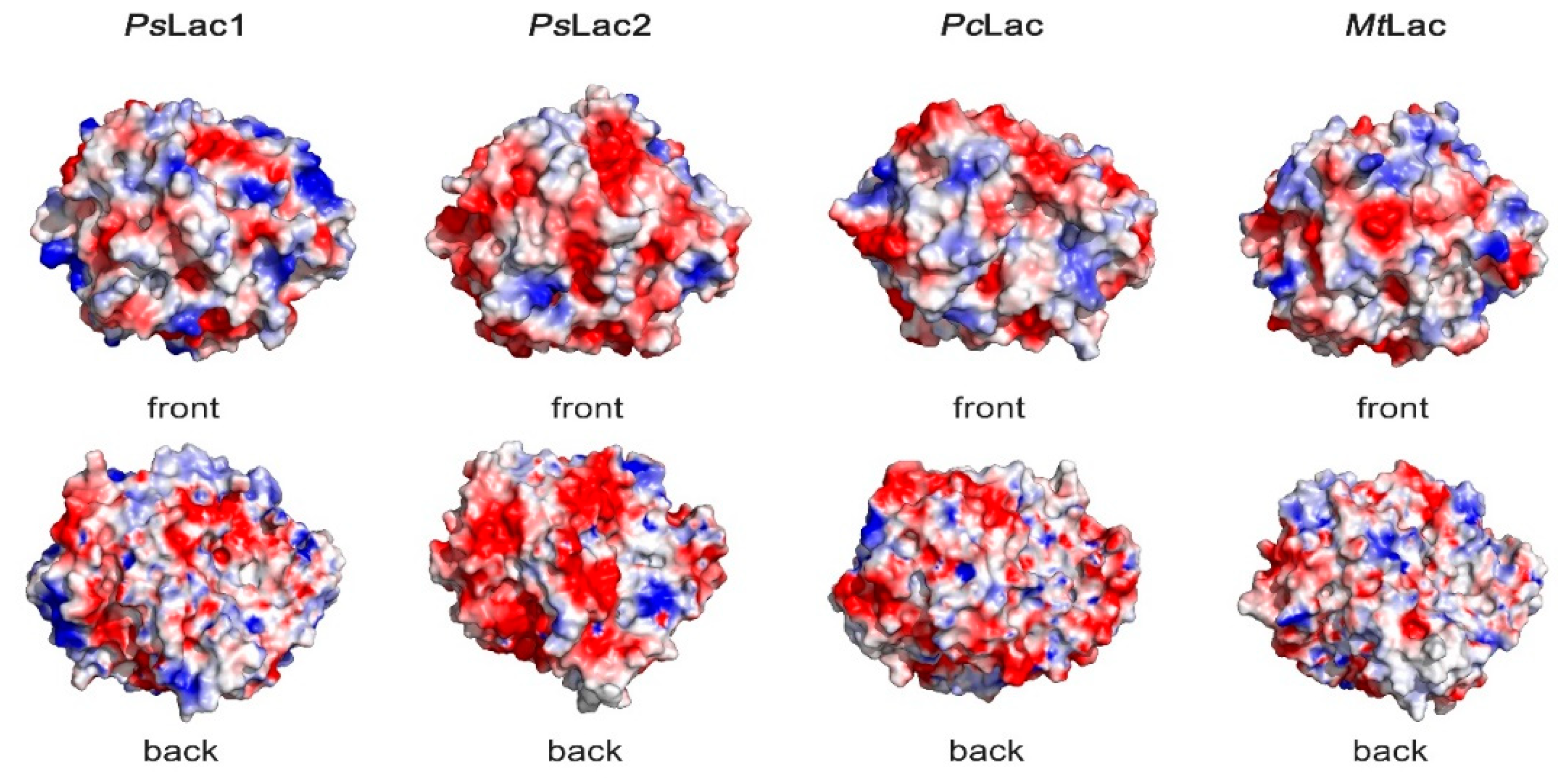
2.6. Surface Charges of Fungal Laccases
3. Discussion
4. Materials and Methods
4.1. Strains and Culture Conditions
4.2. Cloning and Expression of PsLac-Encoding Genes
4.3. Transformation, Screening of Transformants, and Laccase Activity Assay
4.4. Influence of Salt on Laccase Production
4.5. Purification of Recombinant Laccases
4.6. Bioinformatic Analysis
4.7. Influence of Temperature and pH
4.8. The Substrates Specificity and Kinetics
4.9. Decolorization Properties
4.10. Influence of Sea Salt on Laccase Activity
4.11. Sequence Information
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Baldrian, P. Fungal laccases—Occurrence and properties. FEMS Microbiol. Rev. 2006, 30, 215–242. [Google Scholar] [CrossRef]
- Rodríguez Couto, S.; Toca Herrera, J.L. Industrial and biotechnological applications of laccases: A review. Biotechnol. Adv. 2006, 24, 500–513. [Google Scholar] [CrossRef]
- Thurston, C.F. The structure and function of fungal laccases. Microbiology 1994, 140, 19–26. [Google Scholar] [CrossRef]
- Riva, S. Laccases: Blue enzymes for green chemistry. Trends Biotechnol. 2006, 24, 219–226. [Google Scholar] [CrossRef]
- Chen, S.; Ge, W.; Buswell, J.A. Molecular cloning of a new laccase from the edible straw mushroom Volvariella volvacea: Possible involvement in fruit body development. FEMS Microbiol. Lett. 2004, 230, 171–176. [Google Scholar] [CrossRef]
- De Vries, O.M.H.; Kooistra, W.H.C.F.; Wessels, J.G.H. Formation of an extracellular laccase by a Schizophyllum commune dikaryon. Microbiology 1986, 132, 2817–2826. [Google Scholar] [CrossRef][Green Version]
- Piscitelli, A.; Pezzella, C.; Giardina, P.; Faraco, V.; Sannia, G. Heterologous laccase production and its role in industrial applications. Bioeng. Bugs 2010, 1, 252–262. [Google Scholar] [CrossRef]
- Roth, S.; Spiess, A.C. Laccases for biorefinery applications: A critical review on challenges and perspectives. Bioprocess Biosyst. Eng. 2015, 38, 2285–2313. [Google Scholar] [CrossRef]
- Sette, L.; Costa Bonugli Santos, R. Ligninolytic enzymes from marine-derived fungi: Production and applications. In Marine Enzymes for Biocatalysis: Sources, Biocatalytic Characteristics and Bioprocesses of Marine Enzymes; Woodhead Publishing: Sawston, Cambridge, UK, 2013; pp. 403–427. ISBN 978-1-907568-80-0. [Google Scholar]
- Bonugli-Santos, R.C.; Dos Santos Vasconcelos, M.R.; Passarini, M.R.Z.; Vieira, G.A.L.; Lopes, V.C.P.; Mainardi, P.H.; Dos Santos, J.A.; de Azevedo Duarte, L.; Otero, I.V.R.; da Silva Yoshida, A.M.; et al. Marine-derived fungi: Diversity of enzymes and biotechnological applications. Front. Microbiol. 2015, 6, 269. [Google Scholar] [CrossRef]
- Raghukumar, C. Marine fungal biotechnology: An ecological perspective. Fungal Divers. 2008, 31, 19–35. [Google Scholar]
- Raghukumar, C. Marine fungi and their enzymes for decolorization of colored effluents. In Marine microbiology: Facts and opportunities; Ramaiah, N., Ed.; National Institute of Oceanography: Goa, India, 2004; pp. 145–158. [Google Scholar]
- Damare, S.; Raghukumar, C.; Muraleedharan, U.D.; Raghukumar, S. Deep-sea fungi as a source of alkaline and cold-tolerant proteases. Enz. Microb. Technol. 2006, 39, 172–181. [Google Scholar] [CrossRef]
- Gianfreda, L.; Xu, F.; Bollag, J.-M. Laccases: A useful group of oxidoreductive enzymes. Biorem. J. 1999, 3, 1–26. [Google Scholar] [CrossRef]
- Rédou, V.; Kumar, A.; Hainaut, M.; Henrissat, B.; Record, E.; Barbier, G.; Burgaud, G. Draft genome sequence of the deep-sea basidiomycetous yeast Cryptococcus sp. Strain Mo29 reveals its biotechnological potential. Genome Announc. 2016, 4, e00461-16. [Google Scholar] [CrossRef]
- Rédou, V.; Kumar, A.; Hainaut, M.; Henrissat, B.; Record, E.; Barbier, G.; Burgaud, G. Draft genome sequence of the deep-sea ascomycetous filamentous fungus Cadophora malorum Mo12 from the Mid-Atlantic Ridge reveals its biotechnological potential. Genome Announc. 2016, 4, e00467-16. [Google Scholar] [CrossRef]
- Kumar, A.; Henrissat, B.; Arvas, M.; Syed, M.F.; Thieme, N.; Benz, J.P.; Sørensen, J.L.; Record, E.; Pöggeler, S.; Kempken, F. De novo assembly and genome analyses of the marine-derived Scopulariopsis brevicaulis strain LF580 unravels life-style traits and anticancerous scopularide biosynthetic gene cluster. PLoS ONE 2015, 10, e0140398. [Google Scholar] [CrossRef]
- Kumar, A.; Sørensen, J.L.; Hansen, F.T.; Arvas, M.; Syed, M.F.; Hassan, L.; Benz, J.P.; Record, E.; Henrissat, B.; Pöggeler, S.; et al. Genome sequencing and analyses of two marine fungi from the North Sea unraveled a plethora of novel biosynthetic gene clusters. Sci. Rep. 2018, 8, 10187. [Google Scholar] [CrossRef]
- Kamei, I.; Daikoku, C.; Tsutsumi, Y.; Kondo, R. Saline-dependent regulation of manganese peroxidase genes in the hypersaline-tolerant white rot fungus Phlebia sp. strain MG-60. Appl. Environ. Microbiol. 2008, 74, 2709–2716. [Google Scholar] [CrossRef]
- Spalding, M.; Blasco, F.; Filed, C. World Mangrove Atlas; International Society for Mangrove Ecosystems: Okinawa, Japan, 1997; pp. 1–198. [Google Scholar]
- Hyde, K.D.; Lee, S.Y. Ecology of mangrove fungi and their role in nutrient cycling: What gaps occur in our knowledge? Hydrobiologia 1995, 295, 107–118. [Google Scholar] [CrossRef]
- Arfi, Y.; Chevret, D.; Henrissat, B.; Berrin, J.-G.; Levasseur, A.; Record, E. Characterization of salt-adapted secreted lignocellulolytic enzymes from the mangrove fungus Pestalotiopsis sp. Nat. Commun. 2013, 4, 1810. [Google Scholar] [CrossRef]
- Patel, I.; Kracher, D.; Ma, S.; Garajova, S.; Haon, M.; Faulds, C.B.; Berrin, J.-G.; Ludwig, R.; Record, E. Salt-responsive lytic polysaccharide monooxygenases from the mangrove fungus Pestalotiopsis sp. NCi6. Biotechnol. Biofuels 2016, 9, 108. [Google Scholar] [CrossRef]
- Maharachchikumbura, S.S.N.; Hyde, K.D.; Groenewald, J.Z.; Xu, J.; Crous, P.W. Pestalotiopsis revisited. Stud. Mycol. 2014, 79, 121–186. [Google Scholar] [CrossRef]
- Xu, J.; Yang, X.; Lin, Q. Chemistry and biology of Pestalotiopsis-derived natural products. Chem. Biol. 2014, 2, 721–727. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, X.; Liu, L.; Xiang, M.; Wang, W.; Sun, X.; Che, Y.; Guo, L.; Liu, G.; Guo, L.; et al. Genomic and transcriptomic analysis of the endophytic fungus Pestalotiopsis fici reveals its lifestyle and high potential for synthesis of natural products. BMC Genom. 2015, 16, 28. [Google Scholar] [CrossRef]
- Schut, F.; de Vries, E.J.; Gottschal, J.C.; Robertson, B.R.; Harder, W.; Prins, R.A.; Button, D.K. Isolation of typical marine bacteria by dilution culture: Growth, maintenance, and characteristics of isolates under Laboratory CONDITIONS. Appl. Environ. Microbiol. 1993, 59, 2150–2160. [Google Scholar]
- Chen, H.-Y.; Xue, D.-S.; Feng, X.-Y.; Yao, S.-J. Screening and production of ligninolytic enzyme by a marine-derived fungal Pestalotiopsis sp. J63. Appl. Bochem. Biotechnol. 2011, 165, 1754–1769. [Google Scholar] [CrossRef]
- Camarero, S.; Ibarra, D.; Martínez, M.J.; Martínez, A.T. Lignin-derived compounds as efficient laccase mediators for decolorization of different types of recalcitrant dyes. Appl. Environ. Microbiol. 2005, 71, 1775–1784. [Google Scholar] [CrossRef]
- Levasseur, A.; Lomascolo, A.; Chabrol, O.; Ruiz-Dueñas, F.J.; Boukhris-Uzan, E.; Piumi, F.; Kües, U.; Ram, A.F.J.; Murat, C.; Haon, M.; et al. The genome of the white-rot fungus Pycnoporus cinnabarinus: A basidiomycete model with a versatile arsenal for lignocellulosic biomass breakdown. BMC Genom. 2014, 15, 486. [Google Scholar] [CrossRef]
- Van den Brink, J.; Samson, R.A.; Hagen, F.; Boekhout, T.; de Vries, R.P. Phylogeny of the industrial relevant, thermophilic genera Myceliophthora and Corynascus. Fungal Div. 2012, 52, 197–207. [Google Scholar] [CrossRef]
- Kern, M.; McGeehan, J.E.; Streeter, S.D.; Martin, R.N.A.; Besser, K.; Elias, L.; Eborall, W.; Malyon, G.P.; Payne, C.M.; Himmel, M.E.; et al. Structural characterization of a unique marine animal family 7 cellobiohydrolase suggests a mechanism of cellulase salt tolerance. Proc. Nat. Acad. Sci. USA 2013, 110, 10189–10194. [Google Scholar] [CrossRef]
- Eggert, C.; Temp, U.; Eriksson, K.E. The ligninolytic system of the white rot fungus Pycnoporus cinnabarinus: Purification and characterization of the laccase. Appl. Environ. Microbiol. 1996, 62, 1151–1158. [Google Scholar]
- Theerachat, M.; Guieysse, D.; Morel, S.; Remaud-Siméon, M.; Chulalaksananukul, W. Laccases from marine organisms and their applications in the biodegradation of toxic and environmental pollutants: A Review. Appl. Biochem. Biotechnol. 2018, 187, 583–611. [Google Scholar] [CrossRef]
- Brander, S.; Mikkelsen, J.D.; Kepp, K.P. Characterization of an alkali- and halide-resistant laccase expressed in E. coli: CotA from Bacillus clausii. PLoS ONE 2014, 9, e99402. [Google Scholar] [CrossRef] [PubMed]
- Jimenez-Juarez, N.; Roman-Miranda, R.; Baeza, A.; Sánchez-Amat, A.; Vazquez-Duhalt, R.; Valderrama, B. Alkali and halide-resistant catalysis by the multipotent oxidase from Marinomonas mediterranea. J. Biotechnol. 2005, 117, 73–82. [Google Scholar] [CrossRef]
- Ruijssenaars, H.J.; Hartmans, S. A cloned Bacillus halodurans multicopper oxidase exhibiting alkaline laccase activity. Appl. Microbiol. Biotechnol. 2004, 65, 177–182. [Google Scholar] [CrossRef]
- Atalla, M.M.; Zeinab, H.K.; Eman, R.H.; Amani, A.Y.; Abeer, A.A.E.A. Characterization and kinetic properties of the purified Trematosphaeria mangrovei laccase enzyme. Saudi J. Biol. Sci. 2013, 20, 373–381. [Google Scholar] [CrossRef]
- D’Souza-Ticlo, D.; Garg, S.; Raghukumar, C. Effects and interactions of medium components on laccase from a marine-derived fungus using response surface methodology. Mar. Drugs 2009, 7, 672–688. [Google Scholar] [CrossRef] [PubMed]
- Mainardi, P.H.; Feitosa, V.A.; Brenelli de Paiva, L.B.; Bonugli-Santos, R.C.; Squina, F.M.; Pessoa, A.; Sette, L.D. Laccase production in bioreactor scale under saline condition by the marine-derived basidiomycete Peniophora sp. CBMAI 1063. Fungal Biol. 2018, 122, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Farnet, A.M.; Criquet, S.; Tagger, S.; Gil, G.; Le Petit, J. Purification, partial characterization, and reactivity with aromatic compounds of two laccases from Marasmius quercophilus strain 17. Can. J. Microbiol. 2000, 46, 189–194. [Google Scholar] [CrossRef]
- Record, E.; Punt, P.J.; Chamkha, M.; Labat, M.; van Den Hondel, C.A.M.J.J.; Asther, M. Expression of the Pycnoporus cinnabarinus laccase gene in Aspergillus niger and characterization of the recombinant enzyme. Eur. J. Biochem. 2002, 269, 602–609. [Google Scholar] [CrossRef]
- Kiiskinen, L.-L.; Viikari, L.; Kruus, K. Purification and characterisation of a novel laccase from the ascomycete Melanocarpus albomyces. Appl. Microbiol. Biotechnol. 2002, 59, 198–204. [Google Scholar]
- Fields, P.A. Review: Protein function at thermal extremes: Balancing stability and flexibility. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2001, 129, 417–431. [Google Scholar] [CrossRef]
- Gromiha, M.M.; Pathak, M.C.; Saraboji, K.; Ortlund, E.A.; Gaucher, E.A. Hydrophobic environment is a key factor for the stability of thermophilic proteins. Proteins 2013, 81, 715–721. [Google Scholar] [CrossRef]
- Xu, F. Effects of redox potential and hydroxide inhibition on the pH activity profile of fungal laccases. J. Biol. Chem. 1997, 272, 924–928. [Google Scholar] [CrossRef]
- Li, K.; Xu, F.; Eriksson, K.E. Comparison of fungal laccases and redox mediators in oxidation of a nonphenolic lignin model compound. Appl. Environ. Microbiol. 1999, 65, 2654–2660. [Google Scholar]
- Abadulla, E.; Tzanov, T.; Costa, S.; Robra, K.H.; Cavaco-Paulo, A.; Gübitz, G.M. Decolorization and detoxification of textile dyes with a laccase from Trametes hirsuta. Appl. Environ. Microbiol. 2000, 66, 3357–3362. [Google Scholar] [CrossRef]
- Zille, A.; Tzanov, T.; Gübitz, G.M.; Cavaco-Paulo, A. Immobilized laccase for decolourization of reactive black 5 dyeing effluent. Biotechnol. Lett. 2003, 25, 1473–1477. [Google Scholar] [CrossRef]
- Muheim, A.; Fiechter, A.; Harvey, P.J.; Schoemaker, H.E. On the mechanism of oxidation of non-phenolic lignin model compounds by the laccase-ABTS couple. Holzforschung 1992, 46, 121–126. [Google Scholar] [CrossRef]
- Bourbonnais, R.; Paice, M.G. Oxidation of non-phenolic substrates. An expanded role for laccase in lignin biodegradation. FEBS Lett. 1990, 267, 99–102. [Google Scholar] [CrossRef]
- Kuddus, M.; Joseph, B.; Wasudev Ramteke, P. Production of laccase from newly isolated Pseudomonas putida and its application in bioremediation of synthetic dyes and industrial effluents. Biocat. Agric. Biotechnol. 2013, 2, 333–338. [Google Scholar] [CrossRef]
- Sathiya, M.; Periyar, S.; Sasikalaveni, A.; Murugesan, K.; Kalaichelvan, P.T. Decolorization of textile dyes and their effluents using white rot fungi. Afr. J. Biotechnol. 2007, 6, 424–429. [Google Scholar]
- Saparrat, M.C.N.; Hammer, E. Decolorization of synthetic dyes by the deuteromycete Pestalotiopsis guepinii CLPS no. 786 strain. J. Basic Microbiol. 2006, 46, 28–33. [Google Scholar] [CrossRef]
- Yanto, D.H.Y.; Tachibana, S.; Itoh, K. Biodecolorization of textile dyes by immobilized Enzymes in a vertical bioreactor system. Proced. Environ. Sci. 2014, 20, 235–244. [Google Scholar] [CrossRef]
- Kim, H.; Lee, S.; Ryu, S.; Choi, H.T. Decolorization of remazol brilliant blue R by a purified laccase of Polyporus brumalis. Appl. Biochem. Biotechnol. 2012, 166, 159–164. [Google Scholar] [CrossRef]
- Paul, S.; Bag, S.K.; Das, S.; Harvill, E.T.; Dutta, C. Molecular signature of hypersaline adaptation: Insights from genome and proteome composition of halophilic prokaryotes. Gen. Biol. 2008, 9, R70. [Google Scholar] [CrossRef]
- Lanyi, J.K. Salt-dependent properties of proteins from extremely halophilic bacteria. Bacteriol. Rev. 1974, 38, 272–290. [Google Scholar]
- Gordon, C.L.; Khalaj, V.; Ram, A.F.; Archer, D.B.; Brookman, J.L.; Trinci, A.P.; Jeenes, D.J.; Doonan, J.H.; Wells, B.; Punt, P.J.; et al. Glucoamylase: Green fluorescent protein fusions to monitor protein secretion in Aspergillus niger. Microbiology 2000, 146, 415–426. [Google Scholar] [CrossRef]
- Punt, P.J.; van den Hondel, C.A. Transformation of filamentous fungi based on hygromycin B and phleomycin resistance markers. Meth. Enzymol. 1992, 216, 447–457. [Google Scholar]
- Van Hartingsveldt, W.; Mattern, I.E.; van Zeijl, C.M.; Pouwels, P.H.; van den Hondel, C.A. Development of a homologous transformation system for Aspergillus niger based on the pyrG gene. Mol. Gen. Genet. 1987, 206, 71–75. [Google Scholar] [CrossRef]
- Edgar, R.C. MUSCLE: A multiple sequence alignment method with reduced time and space complexity. BMC Bioinform. 2004, 5, 113. [Google Scholar] [CrossRef]
- Chojnacki, S.; Cowley, A.; Lee, J.; Foix, A.; Lopez, R. Programmatic access to bioinformatics tools from EMBL-EBI update: 2017. Nucl. Acids Res. 2017, 45, W550–W553. [Google Scholar] [CrossRef]
- Armenteros, J.J.A.; Tsirigos, K.D.; Sønderby, C.K.; Petersen, T.N.; Winther, O.; Brunak, S.; von Heijne, G.; Nielsen, H. SignalP 5.0 improves signal peptide predictions using deep neural networks. Nat. Biotechnol. 2019, 37, 420–423. [Google Scholar] [CrossRef] [PubMed]
- Kelley, L.A.; Mezulis, S.; Yates, C.M.; Wass, M.N.; Sternberg, M.J.E. The Phyre2 web portal for protein modeling, prediction and analysis. Nat. Protoc. 2015, 10, 845–858. [Google Scholar] [CrossRef] [PubMed]
- Surwase, S.V.; Deshpande, K.K.; Phugare, S.S.; Jadhav, J.P. Biotransformation studies of textile dye Remazol Orange 3R. 3 Biotech 2013, 3, 267–275. [Google Scholar] [CrossRef][Green Version]

| Purification Steps | Volume (mL) | Total Activity (nkat) | Protein (mg) | Specific Activity (nkat mg−1) | Activity Yield (%) | Purification Factor (Fold) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PsLAC1 | PsLAC2 | PsLAC1 | PsLAC2 | PsLAC1 | PsLAC2 | PsLAC1 | PsLAC2 | PsLAC1 | PsLAC2 | PsLAC1 | PsLAC2 | |
| Culture medium | 1800 | 700 | 20081 | 20055 | 45 | 160 | 446 | 125 | 100 | 100 | 1.0 | 1.0 |
| Ultrafiltration | 200 | 200 | 29404 | 41218 | 58 | 63 | 507 | 654 | 146 | 206 | 1.1 | 5.2 |
| DEAE | 200 | 30 | 88174 | 54015 | 62 | 33 | 1422 | 1636 | 439 | 269 | 3.2 | 13 |
| Gel filtration | 2 | 5 | 84244 | 55545 | 20 | 12 | 4212 | 4629 | 420 | 276 | 9.4 | 37 |
| Substrate | Structure | PsLac1 | PsLac2 | ||||
|---|---|---|---|---|---|---|---|
| Km (mM) | Kcat (s−1) | Kcat/Km (s−1 mM−1) | Km (mM) | Kcat (s−1) | Kcat/Km (s−1 mM−1) | ||
| ABTS | 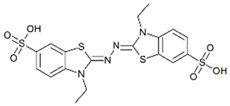 | 0.024 | 0.696 | 29.245 | 0.100 | 0.431 | 4.331 |
| DMP | 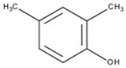 | 0.100 | 2.441 | 24.532 | 0.020 | 0.178 | 9.102 |
| Syringaldazine | 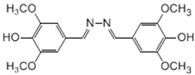 | 0.004 | 0.365 | 82.936 | 0.101 | 0.573 | 5.672 |
| O-dianisidine | 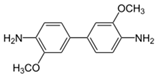 | 0.647 | 0.057 | 0.089 | 0.144 | 0.748 | 5.187 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wikee, S.; Hatton, J.; Turbé-Doan, A.; Mathieu, Y.; Daou, M.; Lomascolo, A.; Kumar, A.; Lumyong, S.; Sciara, G.; Faulds, C.B.; et al. Characterization and Dye Decolorization Potential of Two Laccases from the Marine-Derived Fungus Pestalotiopsis sp. Int. J. Mol. Sci. 2019, 20, 1864. https://doi.org/10.3390/ijms20081864
Wikee S, Hatton J, Turbé-Doan A, Mathieu Y, Daou M, Lomascolo A, Kumar A, Lumyong S, Sciara G, Faulds CB, et al. Characterization and Dye Decolorization Potential of Two Laccases from the Marine-Derived Fungus Pestalotiopsis sp. International Journal of Molecular Sciences. 2019; 20(8):1864. https://doi.org/10.3390/ijms20081864
Chicago/Turabian StyleWikee, Saowanee, Juliette Hatton, Annick Turbé-Doan, Yann Mathieu, Marianne Daou, Anne Lomascolo, Abhishek Kumar, Saisamorn Lumyong, Giuliano Sciara, Craig B. Faulds, and et al. 2019. "Characterization and Dye Decolorization Potential of Two Laccases from the Marine-Derived Fungus Pestalotiopsis sp." International Journal of Molecular Sciences 20, no. 8: 1864. https://doi.org/10.3390/ijms20081864
APA StyleWikee, S., Hatton, J., Turbé-Doan, A., Mathieu, Y., Daou, M., Lomascolo, A., Kumar, A., Lumyong, S., Sciara, G., Faulds, C. B., & Record, E. (2019). Characterization and Dye Decolorization Potential of Two Laccases from the Marine-Derived Fungus Pestalotiopsis sp. International Journal of Molecular Sciences, 20(8), 1864. https://doi.org/10.3390/ijms20081864






